Frailty Syndrome as a Transition from Compensation to Decompensation: Application to the Biomechanical Regulation of Gait
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Description of Participant Groups
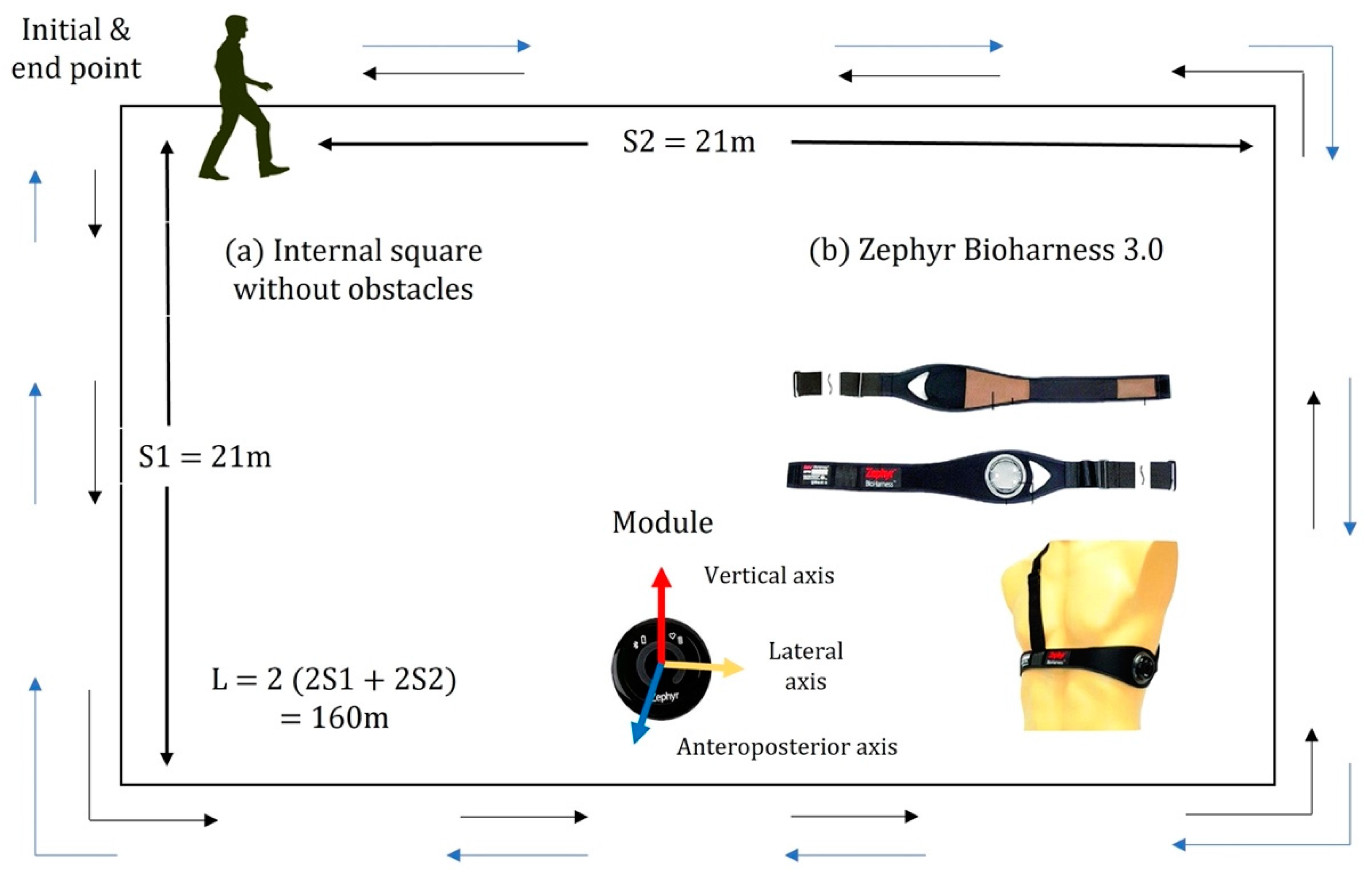
2.3. Materials
2.4. Design and Procedure
2.5. Calculation of Gait Parameters
2.6. Statistical Analysis
3. Results
4. Discussion
Strengths, Limitations, and Implications of This Study
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Abellan van Kan, G.; Rolland, Y.; Andrieu, S.; Bauer, J.; Beauchet, O.; Bonnefoy, M.; Cesari, M.; Donini, L.M.; Gillette Guyonnet, S.; Inzitari, M.; et al. Gait speed at usual pace as a predictor of adverse outcomes in community-dwelling older people an International Academy on Nutrition and Aging (IANA) Task Force. J. Nutr. Health Aging 2009, 13, 881–889. [Google Scholar] [CrossRef] [PubMed]
- Bergman, H.; Ferrucci, L.; Guralnik, J.; Hogan, D.B.; Hummel, S.; Karunananthan, S.; Wolfson, C. Frailty: An emerging research and clinical paradigm--issues and controversies. J. Gerontol. A. Biol. Sci. Med. Sci. 2007, 62, 731–737. [Google Scholar] [CrossRef] [PubMed]
- Varadhan, R.; Seplaki, C.S.; Xue, Q.L.; Bandeen-Roche, K.; Fried, L.P. Stimulus-response paradigm for characterizing the loss of resilience in homeostatic regulation associated with frailty. Mech. Ageing Dev. 2008, 129, 666–670. [Google Scholar] [CrossRef] [PubMed]
- Cesari, M.; Gambassi, G.; van Kan, G.A.; Vellas, B. The frailty phenotype and the frailty index: Different instruments for different purposes. Age Ageing 2014, 43, 10–12. [Google Scholar] [CrossRef]
- Lopez, D.; Flicker, L.; Dobson, A. Validation of the frail scale in a cohort of older Australian women. J. Am. Geriatr. Soc. 2012, 60, 171–173. [Google Scholar] [CrossRef]
- Rockwood, K.; Song, X.; MacKnight, C.; Bergman, H.; Hogan, D.B.; McDowell, I.; Mitnitski, A. A global clinical measure of fitness and frailty in elderly people. CMAJ Can. Med. Assoc. J. J. Assoc. Med. Can. 2005, 173, 489–495. [Google Scholar] [CrossRef]
- Rolfson, D.B.; Majumdar, S.R.; Tsuyuki, R.T.; Tahir, A.; Rockwood, K. Validity and reliability of the Edmonton Frail Scale. Age Ageing 2006, 35, 526–529. [Google Scholar] [CrossRef]
- Saedi, A.A.; Feehan, J.; Phu, S.; Duque, G. Current and emerging biomarkers of frailty in the elderly. Clin. Interv. Aging 2019, 14, 389–398. [Google Scholar] [CrossRef]
- Olde Rikkert, M.G.M.; Melis, R.J.F. Rerouting Geriatric Medicine by Complementing Static Frailty Measures With Dynamic Resilience Indicators of Recovery Potential. Front. Physiol. 2019, 10, 723. Available online: https://www.frontiersin.org/articles/10.3389/fphys.2019.00723 (accessed on 14 October 2022). [CrossRef]
- Fried, L.P.; Tangen, C.M.; Walston, J.; Newman, A.B.; Hirsch, C.; Gottdiener, J.; Seeman, T.; Tracy, R.; Kop, W.J.; Burke, G.; et al. Frailty in older adults: Evidence for a phenotype. J. Gerontol. A. Biol. Sci. Med. Sci. 2001, 56, M146–M156. [Google Scholar] [CrossRef]
- Roche, N.; Chorin, F.; Gerus, P.; Deshayes, M.; Guerin, O.; Zory, R. Effects of age, gender, frailty and falls on spatiotemporal gait parameters: A retrospective cohort study. Eur. J. Phys. Rehabil. Med. 2021, 57, 923–930. [Google Scholar] [CrossRef] [PubMed]
- Fritz, S.; Lusardi, M. White paper: “walking speed: The sixth vital sign”. J. Geriatr. Phys. Ther. 2001 2009, 32, 46–49. [Google Scholar] [CrossRef]
- Song, S.; Geyer, H. Predictive neuromechanical simulations indicate why walking performance declines with ageing. J. Physiol. 2018, 596, 1199–1210. [Google Scholar] [CrossRef] [PubMed]
- Xie, Y.J.; Liu, E.Y.; Anson, E.R.; Agrawal, Y. Age-related imbalance is associated with slower walking speed: Analysis from the National Health and Nutrition Examination Survey. J. Geriatr. Phys. Ther. 2001 2017, 40, 183–189. [Google Scholar] [CrossRef] [PubMed]
- Ogawa, N.; Nanayama Tanaka, C.; Ishido, M.; Nakamura, T.; Nishiwaki, M. Poor Walking Speed Is Associated With Higher Segment-Specific Arterial Stiffness in Older Adult Japanese Community Dwellers: A Cross-Sectional Study. Front. Physiol. 2020, 11, 587215. Available online: https://www.frontiersin.org/articles/10.3389/fphys.2020.587215 (accessed on 10 March 2023). [CrossRef]
- Muro-de-la-Herran, A.; Garcia-Zapirain, B.; Mendez-Zorrilla, A. Gait Analysis Methods: An Overview of Wearable and Non-Wearable Systems, Highlighting Clinical Applications. Sensors 2014, 14, 3362–3394. [Google Scholar] [CrossRef]
- Salchow-Hömmen, C.; Skrobot, M.; Jochner, M.C.E.; Schauer, T.; Kühn, A.A.; Wenger, N. Review—Emerging Portable Technologies for Gait Analysis in Neurological Disorders. Front. Hum. Neurosci. 2022, 16, 11. Available online: https://www.frontiersin.org/articles/10.3389/fnhum.2022.768575 (accessed on 10 March 2023). [CrossRef]
- Jarchi, D.; Pope, J.; Lee, T.K.M.; Tamjidi, L.; Mirzaei, A.; Sanei, S. A Review on Accelerometry-Based Gait Analysis and Emerging Clinical Applications. IEEE Rev. Biomed. Eng. 2018, 11, 177–194. [Google Scholar] [CrossRef]
- van Emmerik, R.E.A.; Ducharme, S.W.; Amado, A.C.; Hamill, J. Comparing dynamical systems concepts and techniques for biomechanical analysis. J. Sport Health Sci. 2016, 5, 3–13. [Google Scholar] [CrossRef]
- MacKay-Lyons, M. Central pattern generation of locomotion: A review of the evidence. Phys. Ther. 2002, 82, 69–83. [Google Scholar] [CrossRef]
- Stergiou, N. Biomechanics and Gait Analysis; Elsevier: Amsterdam, The Netherlands, 2020; ISBN 978-0-12-813440-5. [Google Scholar]
- Dingwell, J.B.; Cusumano, J.P. Nonlinear time series analysis of normal and pathological human walking. Chaos Interdiscip. J. Nonlinear Sci. 2000, 10, 848–863. [Google Scholar] [CrossRef] [PubMed]
- Gan, Z.; Yesilevskiy, Y.; Zaytsev, P.; Remy, C.D. All common bipedal gaits emerge from a single passive model. J. R. Soc. Interface 2018, 15, 20180455. [Google Scholar] [CrossRef]
- Montero-Odasso, M.; Muir, S.W.; Hall, M.; Doherty, T.J.; Kloseck, M.; Beauchet, O.; Speechley, M. Gait variability is associated with frailty in community-dwelling older adults. J. Gerontol. A. Biol. Sci. Med. Sci. 2011, 66, 568–576. [Google Scholar] [CrossRef] [PubMed]
- Shimada, H.; Kim, H.; Yoshida, H.; Suzukawa, M.; Makizako, H.; Yoshida, Y.; Saito, K.; Suzuki, T. Relationship between Age-Associated Changes of Gait and Falls and Life-Space in Elderly People. J. Phys. Ther. Sci. 2010, 22, 419–424. [Google Scholar] [CrossRef]
- McGibbon, C.A. Toward a better understanding of gait changes with age and disablement: Neuromuscular adaptation. Exerc. Sport Sci. Rev. 2003, 31, 102–108. [Google Scholar] [CrossRef]
- Thaler-Kall, K.; Peters, A.; Thorand, B.; Grill, E.; Autenrieth, C.S.; Horsch, A.; Meisinger, C. Description of spatio-temporal gait parameters in elderly people and their association with history of falls: Results of the population-based cross-sectional KORA-Age study. BMC Geriatr. 2015, 15, 32. [Google Scholar] [CrossRef]
- Shkuratova, N.; Morris, M.E.; Huxham, F. Effects of age on balance control during walking. Arch. Phys. Med. Rehabil. 2004, 85, 582–588. [Google Scholar] [CrossRef]
- Rota, V.; Perucca, L.; Simone, A.; Tesio, L. Walk ratio (step length/cadence) as a summary index of neuromotor control of gait: Application to multiple sclerosis. Int. J. Rehabil. Res. Int. Z. Rehabil. Rev. Int. Rech. Readapt. 2011, 34, 265–269. [Google Scholar] [CrossRef]
- Zijlstra, A.; de Bruin, E.D.; Bruins, N.; Zijlstra, W. The step length-frequency relationship in physically active community-dwelling older women. Eur. J. Appl. Physiol. 2008, 104, 427–434. [Google Scholar] [CrossRef]
- Martínez-Ramírez, A.; Martinikorena, I.; Gómez, M.; Lecumberri, P.; Millor, N.; Rodríguez-Mañas, L.; García García, F.J.; Izquierdo, M. Frailty assessment based on trunk kinematic parameters during walking. J. Neuroeng. Rehabil. 2015, 12, 48. [Google Scholar] [CrossRef]
- Menz, H.B.; Lord, S.R.; Fitzpatrick, R.C. Age-related differences in walking stability. Age Ageing 2003, 32, 137–142. [Google Scholar] [CrossRef] [PubMed]
- Marigold, D.S.; Patla, A.E. Age-related changes in gait for multi-surface terrain. Gait Posture 2008, 27, 689–696. [Google Scholar] [CrossRef] [PubMed]
- Samson, M.M.; Crowe, A.; de Vreede, P.L.; Dessens, J.A.; Duursma, S.A.; Verhaar, H.J. Differences in gait parameters at a preferred walking speed in healthy subjects due to age, height and body weight. Aging 2001, 13, 16–21. [Google Scholar] [CrossRef] [PubMed]
- Khoo, Michael Physiological Control Systems: Analysis, Simulation, and Estimation|IEEE eBooks|IEEE Xplore. Available online: https://ieeexplore.ieee.org/book/5263864 (accessed on 10 March 2023).
- Zile, M.R.; Bennett, T.D.; St John Sutton, M.; Cho, Y.K.; Adamson, P.B.; Aaron, M.F.; Aranda, J.M.; Abraham, W.T.; Smart, F.W.; Stevenson, L.W.; et al. Transition from chronic compensated to acute decompensated heart failure: Pathophysiological insights obtained from continuous monitoring of intracardiac pressures. Circulation 2008, 118, 1433–1441. [Google Scholar] [CrossRef]
- Ferrucci, L.; Gonzalez-Freire, M.; Fabbri, E.; Simonsick, E.; Tanaka, T.; Moore, Z.; Salimi, S.; Sierra, F.; de Cabo, R. Measuring biological aging in humans: A quest. Aging Cell 2020, 19, e13080. [Google Scholar] [CrossRef]
- Álvarez-Millán, L.; Lerma, C.; Castillo-Castillo, D.; Quispe-Siccha, R.M.; Pérez-Pacheco, A.; Rivera-Sánchez, J.; Fossion, R. Chronotropic Response and Heart Rate Variability before and after a 160 m Walking Test in Young, Middle-Aged, Frail, and Non-Frail Older Adults. Int. J. Environ. Res. Public. Health 2022, 19, 8413. [Google Scholar] [CrossRef]
- Khoo, M.C. Physiological Control Systems: Analysis, Simulation, and Estimation, 2nd ed.; Wiley: Hoboken, NJ, USA, 2018; Available online: https://www.wiley.com/en-us/Physiological+Control+Systems%3A+Analysis%2C+Simulation%2C+and+Estimation%2C+2nd+Edition-p-9781119058809 (accessed on 13 October 2022).
- Salem, B.E.; Brecht, M.-L.; Ekstrand, M.L.; Faucette, M.; Nyamathi, A.M. Correlates of physical, psychological, and social frailty among formerly incarcerated, homeless women. Health Care Women Int. 2019, 40, 788–812. [Google Scholar] [CrossRef]
- Piggott, D.A.; Muzaale, A.D.; Mehta, S.H.; Brown, T.T.; Patel, K.V.; Leng, S.X.; Kirk, G.D. Frailty, HIV Infection, and Mortality in an Aging Cohort of Injection Drug Users. PLoS ONE 2013, 8, e54910. [Google Scholar] [CrossRef]
- Ness, K.K.; Armstrong, G.T.; Kundu, M.; Wilson, C.L.; Tchkonia, T.; Kirkland, J.L. Frailty in childhood cancer survivors. Cancer 2015, 121, 1540–1547. [Google Scholar] [CrossRef]
- Dhar, P.; Sharma, V.K.; Hota, K.B.; Das, S.K.; Hota, S.K.; Srivastava, R.B.; Singh, S.B. Autonomic Cardiovascular Responses in Acclimatized Lowlanders on Prolonged Stay at High Altitude: A Longitudinal Follow Up Study. PLoS ONE 2014, 9, e84274. [Google Scholar] [CrossRef]
- Ruiz-Ruiz, L.; Jimenez, A.R.; Garcia-Villamil, G.; Seco, F. Detecting Fall Risk and Frailty in Elders with Inertial Motion Sensors: A Survey of Significant Gait Parameters. Sensors 2021, 21, 6918. [Google Scholar] [CrossRef]
- Álvarez-Millán, L.A.; Castillo-Castillo, D.; Pérez-Pacheco, A.; Quispe-Siccha, R.; Angelova, M.; Rivera-Sánchez, J.; Fossion, R. Time-series based evaluation of physiological alterations in age-associated frailty during walking at self-selected velocity. AIP Conf. Proc. 2019, 2090, 050002. [Google Scholar] [CrossRef]
- Ibikunle, A.F.; Adedoyin, R.A.; Awotidebe, T.O.; Fasakin, O.M.; Okonji, A.M.; Odetunde, M.O.; Olawoye, A.A. Validation of Three-Minute Walk Test for the Assessment of Functional Capacity among Patients with Hypertension. J. Clin. Exp. Cardiolog. 2020, 11, 662. [Google Scholar]
- Studenski, S.; Perera, S.; Patel, K.; Rosano, C.; Faulkner, K.; Inzitari, M.; Brach, J.; Chandler, J.; Cawthon, P.; Connor, E.B.; et al. Gait Speed and Survival in Older Adults. JAMA 2011, 305, 50–58. [Google Scholar] [CrossRef]
- Schwenk, M.; Howe, C.; Saleh, A.; Mohler, J.; Grewal, G.; Armstrong, D.; Najafi, B. Frailty and Technology: A Systematic Review of Gait Analysis in Those with Frailty. Gerontology 2014, 60, 79–89. [Google Scholar] [CrossRef] [PubMed]
- Dotov, D.G.; Bardy, B.G.; Dalla Bella, S. The role of environmental constraints in walking: Effects of steering and sharp turns on gait dynamics. Sci. Rep. 2016, 6, 28374. [Google Scholar] [CrossRef]
- Zhong, R.; Rau, P.-L.P.; Yan, X. Gait Assessment of Younger and Older Adults with Portable Motion-Sensing Methods: A User Study. Mob. Inf. Syst. 2019, 2019, e1093514. [Google Scholar] [CrossRef]
- Hof, A.L. Scaling gait data to body size. Gait Posture 1996, 4, 222–223. [Google Scholar] [CrossRef]
- Zhang, Q.; Guo, H.; Gu, H.; Zhao, X. Gender-associated factors for frailty and their impact on hospitalization and mortality among community-dwelling older adults: A cross-sectional population-based study. PeerJ 2018, 6, e4326. [Google Scholar] [CrossRef]
- Aldhahi, M.I. Effect of Gait Alteration on Fatigability during Walking in Adult Women with High Body Fat Composition. Med. Kaunas Lith. 2022, 59, 85. [Google Scholar] [CrossRef]
- Kressig, R.W.; Gregor, R.J.; Oliver, A.; Waddell, D.; Smith, W.; O’Grady, M.; Curns, A.T.; Kutner, M.; Wolf, S.L. Temporal and spatial features of gait in older adults transitioning to frailty. Gait Posture 2004, 20, 30–35. [Google Scholar] [CrossRef]
- Duque-Ramírez, J.R.; Vélez-Uribe, J.D.; Gómez-Suárez, N.E.; Manrique-Hernández, R.D. Gait parameters in a sample of healthy Colombian adults aged between 18 and 25 years: A cross-sectional study. Rev. Fac. Med. 2020, 68, 84–91. [Google Scholar] [CrossRef]
- Tudor-Locke, C.; Aguiar, E.J.; Han, H.; Ducharme, S.W.; Schuna, J.M.; Barreira, T.V.; Moore, C.C.; Busa, M.A.; Lim, J.; Sirard, J.R.; et al. Walking cadence (steps/min) and intensity in 21–40 year olds: CADENCE-adults. Int. J. Behav. Nutr. Phys. Act. 2019, 16, 8. [Google Scholar] [CrossRef] [PubMed]
- Apsega, A.; Petrauskas, L.; Alekna, V.; Daunoraviciene, K.; Sevcenko, V.; Mastaviciute, A.; Vitkus, D.; Tamulaitiene, M.; Griskevicius, J. Wearable Sensors Technology as a Tool for Discriminating Frailty Levels During Instrumented Gait Analysis. Appl. Sci. 2020, 10, 8451. [Google Scholar] [CrossRef]
- Zancan, A.; Sozzi, S.; Schieppati, M. Basic Spatiotemporal Gait Variables of Young and Older Healthy Volunteers Walking Along a Novel Figure-of-8 Path. Front. Neurol. 2021, 12, 698160. Available online: https://www.frontiersin.org/articles/10.3389/fneur.2021.698160 (accessed on 10 March 2023). [CrossRef] [PubMed]
- Bronstein, A.; Brandt, T.; Brandt, T. (Eds.) Clinical Disorders of Balance, Posture and Gait, 2nd ed.; Oxford University Press: London, UK; New York, NY, USA, 2004; ISBN 978-0-340-80657-9. [Google Scholar]
- Fielding, R.A.; Vellas, B.; Evans, W.J.; Bhasin, S.; Morley, J.E.; Newman, A.B.; Abellan van Kan, G.; Andrieu, S.; Bauer, J.; Breuille, D.; et al. Sarcopenia: An undiagnosed condition in older adults. Current consensus definition: Prevalence, etiology, and consequences. International working group on sarcopenia. J. Am. Med. Dir. Assoc. 2011, 12, 249–256. [Google Scholar] [CrossRef] [PubMed]
- Winter, D.A. Anatomy, Biomechanics and Control) of Balance during Standing and Walking; Waterloo Biomechanics: Waterloo, ON, Canada, 1995; ISBN 978-0-9699420-0-9. [Google Scholar]
- Baumgart, C.; Hoppe, M.W.; Freiwald, J. Long-Term Adaptations to Unexpected Surface Perturbations: Postural Control During Stance and Gait in Train Conductors. J. Mot. Behav. 2016, 48, 341–347. [Google Scholar] [CrossRef]
- Nowak, A.; Hubbard, R.E. Falls and frailty: Lessons from complex systems. J. R. Soc. Med. 2009, 102, 98–102. [Google Scholar] [CrossRef]
- McCrory, J.L.; Chambers, A.J.; Daftary, A.; Redfern, M.S. The pregnant “waddle”: An evaluation of torso kinematics in pregnancy. J. Biomech. 2014, 47, 2964–2968. [Google Scholar] [CrossRef] [PubMed]
- Browning, R.C. Locomotion Mechanics in Obese Adults and Children. Curr. Obes. Rep. 2012, 1, 152–159. [Google Scholar] [CrossRef][Green Version]
- Stoffregen, T.A. Functional Control of Stance in Older Adults. Kinesiol. Rev. 2016, 5, 23–29. [Google Scholar] [CrossRef]
- Schrack, J.A.; Simonsick, E.M.; Ferrucci, L. The Energetic Pathway to Mobility Loss: An Emerging New Framework for Longitudinal Studies on Aging. J. Am. Geriatr. Soc. 2010, 58, S329–S336. [Google Scholar] [CrossRef] [PubMed]
- Quinlan, N.; Marcantonio, E.R.; Inouye, S.K.; Gill, T.M.; Kamholz, B.; Rudolph, J.L. Vulnerability: The crossroads of frailty and delirium. J. Am. Geriatr. Soc. 2011, 59 (Suppl. S2), S262–S268. [Google Scholar] [CrossRef] [PubMed]
- Taffett, G.E. Physiology of Aging. In Geriatric Medicine: An Evidence-Based Approach; Cassel, C.K., Leipzig, R.M., Cohen, H.J., Larson, E.B., Meier, D.E., Capello, C.F., Eds.; Springer: New York, NY, USA, 2003; pp. 27–35. ISBN 978-0-387-22621-7. [Google Scholar]
- Modell, H.; Cliff, W.; Michael, J.; McFarland, J.; Wenderoth, M.P.; Wright, A. A physiologist’s view of homeostasis. Adv. Physiol. Educ. 2015, 39, 259–266. [Google Scholar] [CrossRef]
- Michael, J.; Cliff, W.; McFarland, J.; Modell, H.; Wright, A. The Core Concepts of Physiology; Springer: New York, NY, ISA, 2017; ISBN 978-1-4939-6907-4. [Google Scholar]
- Carpenter, R.H.S. Homeostasis: A plea for a unified approach. Adv. Physiol. Educ. 2004, 28, 180–187. [Google Scholar] [CrossRef] [PubMed]
- Billman, G.E. Homeostasis: The Underappreciated and Far Too Often Ignored Central Organizing Principle of Physiology. Front. Physiol. 2020, 11, 200. [Google Scholar] [CrossRef]
- Fossion, R.; Fossion, J.P.J.; Rivera, A.L.; Lecona, O.A.; Toledo-Roy, J.C.; García-Pelagio, K.P.; García-Iglesias, L.; Estañol, B. Homeostasis from a Time-Series Perspective: An Intuitive Interpretation of the Variability of Physiological Variables. In Quantitative Models for Microscopic to Macroscopic Biological Macromolecules and Tissues; Olivares-Quiroz, L., Resendis-Antonio, O., Eds.; Springer International Publishing: Cham, Switzerland, 2018; pp. 87–109. ISBN 978-3-319-73975-5. [Google Scholar]
- Fossion, R.; Rivera, A.L.; Estañol, B. A physicist’s view of homeostasis: How time series of continuous monitoring reflect the function of physiological variables in regulatory mechanisms. Physiol. Meas. 2018, 39, 084007. [Google Scholar] [CrossRef] [PubMed]
- Fossion, R.; Zapata-Fonseca, L. The Scientific Method as a Point of Departure in Aging Research. In Aging Research—Methodological Issues; García-Peña, C., Gutiérrez-Robledo, L.M., Pérez-Zepeda, M.U., Eds.; Springer International Publishing: Cham, Switzerland, 2018; pp. 11–24. ISBN 978-3-319-95387-8. [Google Scholar]
- Fossion, R.; Rivera, A.L.; Alvarez-Milláan, L.; García-Iglesias, L.; Lecona, O.; Robles-Cabrera, A.; Esta˜nol, B. A Time-Series Approach to Assess Physiological and Biomechanical Regulatory Mechanisms. In 2019-20 MATRIX Annals; de Gier, J., Praeger, C.E., Tao, T., Eds.; MATRIX Book Series; Springer International Publishing: Cham, Switzerland, 2021; pp. 265–277. ISBN 978-3-030-62497-2. [Google Scholar]
- Rector, J.L.; Gijzel, S.M.W.; van de Leemput, I.A.; van Meulen, F.B.; Olde Rikkert, M.G.M.; Melis, R.J.F. Dynamical indicators of resilience from physiological time series in geriatric inpatients: Lessons learned. Exp. Gerontol. 2021, 149, 111341. [Google Scholar] [CrossRef] [PubMed]
- Nakazato, Y.; Sugiyama, T.; Ohno, R.; Shimoyama, H.; Leung, D.L.; Cohen, A.A.; Kurane, R.; Hirose, S.; Watanabe, A.; Shimoyama, H. Estimation of homeostatic dysregulation and frailty using biomarker variability: A principal component analysis of hemodialysis patients. Sci. Rep. 2020, 10, 10314. [Google Scholar] [CrossRef]
- Rivera, A.L.; Estanol, B.; Senties-Madrid, H.; Fossion, R.; Toledo-Roy, J.C.; Mendoza-Temis, J.; Morales, I.O.; Landa, E.; Robles-Cabrera, A.; Moreno, R.; et al. Heart Rate and Systolic Blood Pressure Variability in the Time Domain in Patients with Recent and Long-Standing Diabetes Mellitus. PLoS ONE 2016, 11, e0148378. [Google Scholar] [CrossRef]
| Variable | Group C1 (n = 27) | Group C2 (n = 16) | Group nF (n = 15) | Group F (n = 31) | p-Value between All Groups |
|---|---|---|---|---|---|
| Age (years) (% female) | 22.3 ± 0.4 (59%) | 48.5 ± 2.2 (62%) | 72.7 ± 2.5 ¶,# (33%) | 78.5 ± 1 ¶,# (71%) | 0.000 |
| Weight (kg) | 66.8 ± 2.4 | 70.7 ± 4.4 | 69.3 ± 3.1 | 61.6 ± 2.2 | 0.154 |
| Height (m) | 1.7 ± 0.02 | 1.6 ± 0.02 | 1.6 ± 0.02 | 1.5 ± 0.02 ¶ | 0.000 |
| BMI (kg/m2) | 23.9 ± 0.6 | 27.4 ± 1.5 | 26.8 ± 1.03 | 26.3 ± 0.7 | 0.060 |
| Variable | Group C1 (n = 27) | Group C2 (n = 16) | Group nF (n = 15) | Group F (n = 31) | p-Value between All Groups |
|---|---|---|---|---|---|
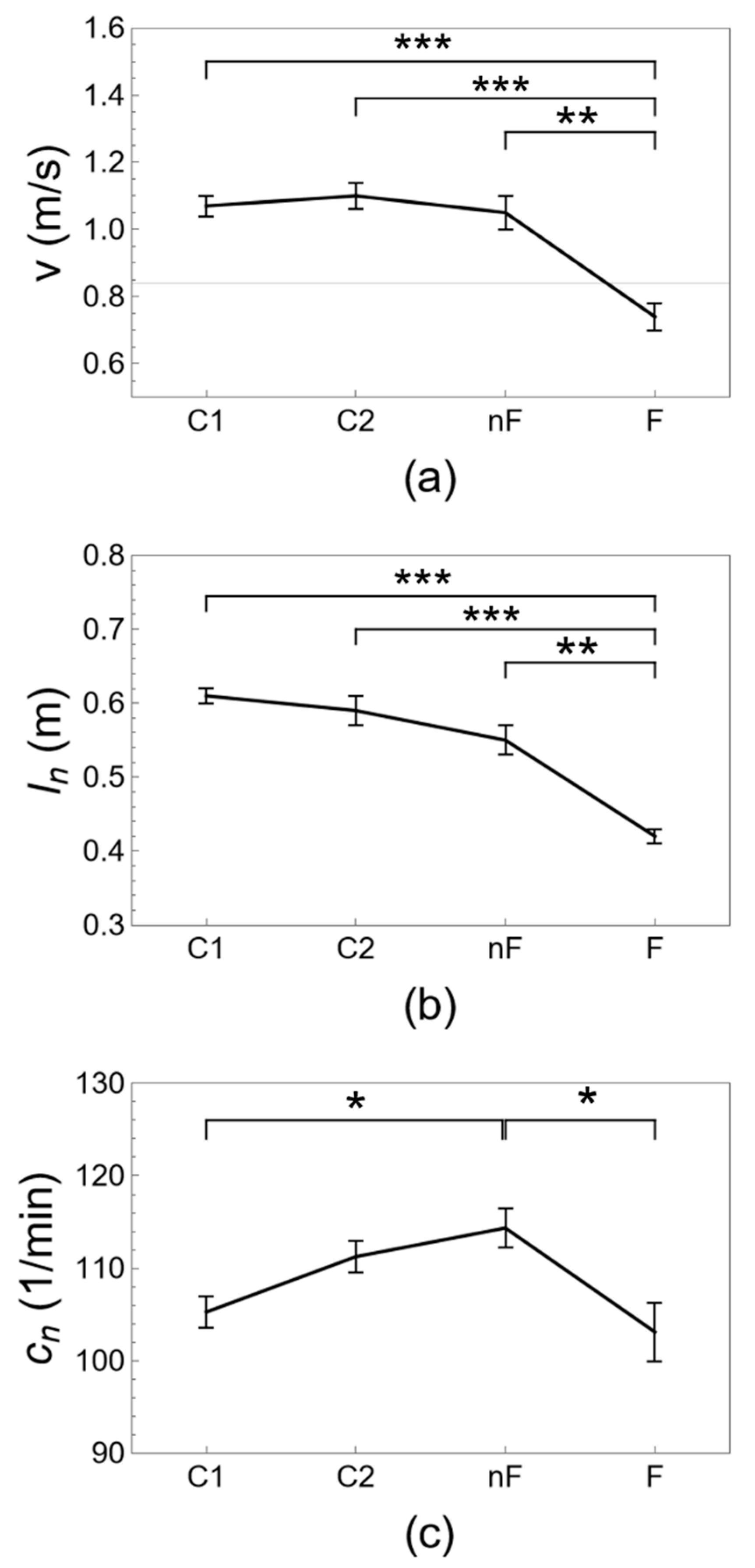
| v (m/s) | 1.08 ± 0.03 | 1.09 ± 0.05 | 1.05 ± 0.06 | 0.74 ± 0.05 ¶,#,§ | 0.000 |
| l (m) | 0.61 ± 0.01 | 0.59 ± 0.04 | 0.55 ± 0.02 | 0.42 ± 0.03 ¶,#,§ | 0.000 |
| ln (m) | 0.61 ± 0.01 | 0.6 ± 0.02 | 0.55 ± 0.02 | 0.42 ± 0.02 ¶,#,§ | 0.000 |
| c (1/min) | 105.26 ± 1.72 | 111.25 ± 1.78 | 114.35 ± 3.21 ¶ | 103.1 ± 3.1 § | 0.005 |
| cn (1/min) | 105.64 ± 1.7 | 110.62 ± 1.7 | 114.05 ± 2.1 ¶ | 103.53 ± 3.2 § | 0.005 |
| Variable | Group C1 (n = 27) | Group C2 (n = 16) | Group nF (n = 15) | Group F (n = 31) | p-Value between All Groups |
|---|---|---|---|---|---|
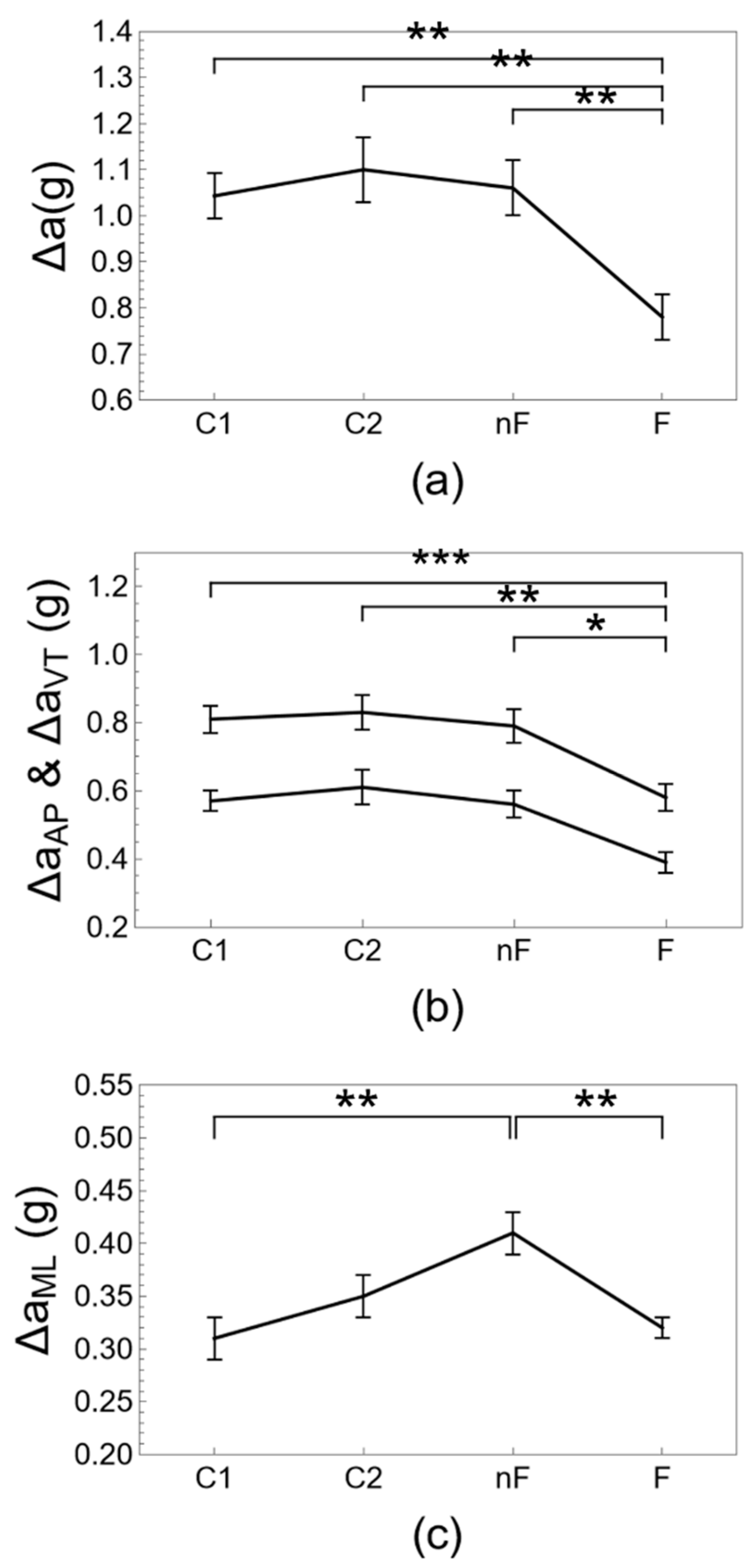
| Δa (g) | 1.04 ± 0.05 | 1.10 ± 0.07 | 1.06 ± 0.06 | 0.80 ± 0.05 ¶,#,§ | 0.000 |
| ΔaAP (g) | 0.57 ± 0.03 | 0.61 ± 0.05 | 0.56 ± 0.04 | 0.39 ± 0.03 ¶,#,§ | 0.000 |
| ΔaVT (g) | 0.81 ± 0.04 | 0.83 ± 0.05 | 0.79 ± 0.05 | 0.58 ± 0.04 ¶,#,§ | 0.000 |
| ΔaML (g) | 0.31 ± 0.02 | 0.35 ± 0.02 | 0.41 ± 0.02 ¶ | 0.32 ± 0.01 § | 0.002 |
| RMS(a) (g) | 0.12 ± 0.007 | 0.13 ± 0.008 | 0.12 ± 0.008 | 0.09 ± 0.006 ¶,#,§ | 0.000 |
| RMS() (g) | 0.13 ± 0.007 | 0.14 ± 0.01 | 0.14 ± 0.008 | 0.1 ± 0.006 ¶,#,§ | 0.001 |
| RMS() (g) | 0.2 ± 0.01 | 0.22 ± 0.02 | 0.22 ± 0.02 | 0.14 ± 0.01 ¶,#,§ | 0.000 |
| RMS () (g) | 0.08 ± 0.003 | 0.09 ± 0.004 | 0.1 ± 0.003 ¶ | 0.09 ± 0.003 | 0.002 |
| Variable. | Group C1 (n = 27) | Group C2 (n = 16) | Group nF (n = 15) | Group F (n = 31) | p-Value between All Groups |
|---|---|---|---|---|---|
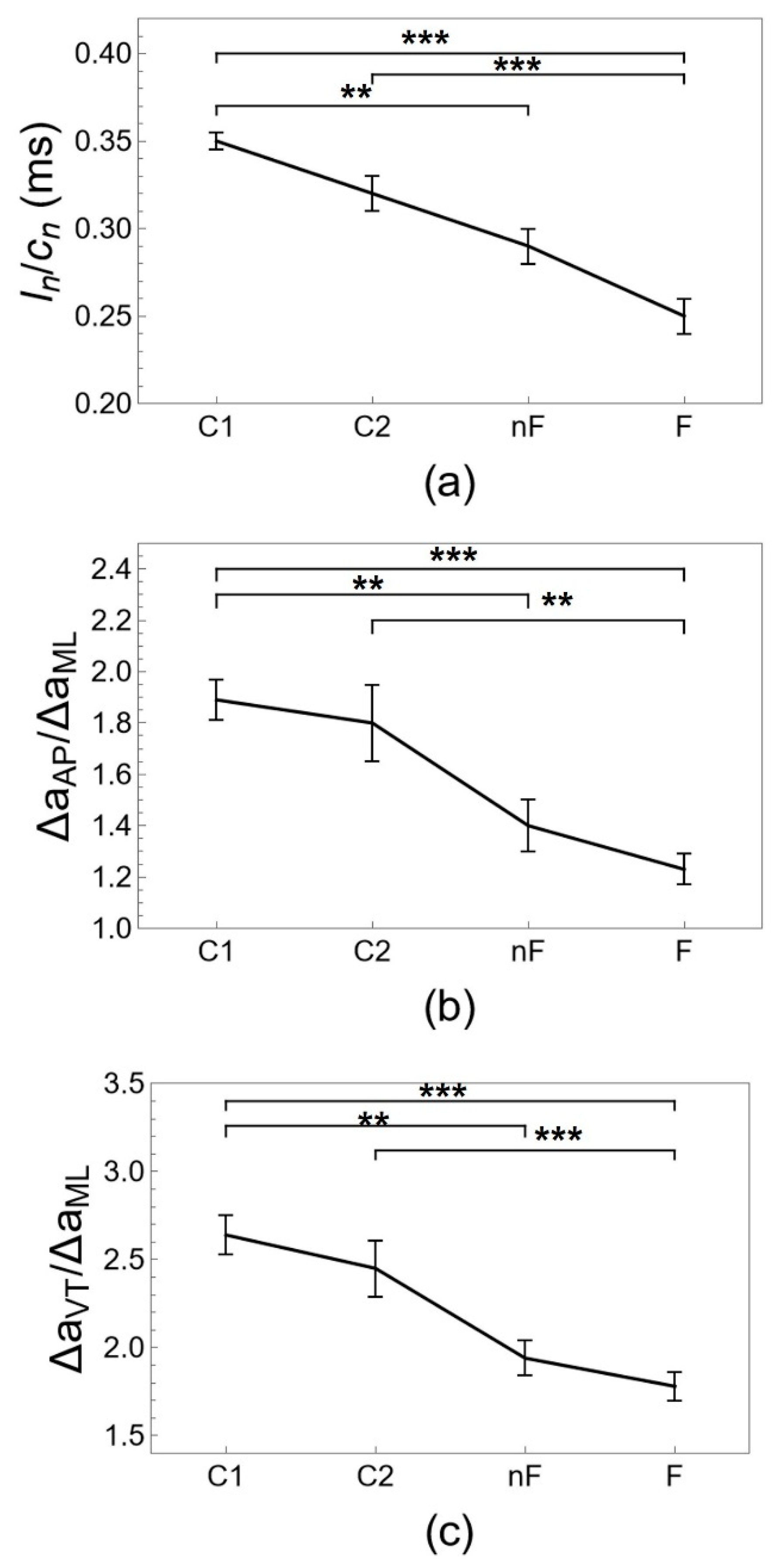
| l/c (m.s) | 0.35 ± 0.006 | 0.32 ± 0.012 | 0.29 ± 0.01 ¶ | 0.25 ± 0.01 ¶,# | 0.000 |
| ln/cn (m.s) | 0.34 ± 0.006 | 0.32 ± 0.01 | 0.29 ± 0.01 ¶ | 0.25 ± 0.01 ¶,# | 0.000 |
| ΔaAP/ΔaML | 1.9 ± 0.08 | 1.8 ± 0.15 | 1.4 ± 0.1 ¶ | 1.2 ± 0.06 ¶,# | 0.000 |
| ΔaVT/ΔaML | 2.6 ± 0.11 | 2.4 ± 0.16 | 1.9 ± 0.1 ¶ | 1.8 ± 0.08 ¶,# | 0.000 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Álvarez-Millán, L.; Castillo-Castillo, D.; Quispe-Siccha, R.; Pérez-Pacheco, A.; Angelova, M.; Rivera-Sánchez, J.; Fossion, R. Frailty Syndrome as a Transition from Compensation to Decompensation: Application to the Biomechanical Regulation of Gait. Int. J. Environ. Res. Public Health 2023, 20, 5995. https://doi.org/10.3390/ijerph20115995
Álvarez-Millán L, Castillo-Castillo D, Quispe-Siccha R, Pérez-Pacheco A, Angelova M, Rivera-Sánchez J, Fossion R. Frailty Syndrome as a Transition from Compensation to Decompensation: Application to the Biomechanical Regulation of Gait. International Journal of Environmental Research and Public Health. 2023; 20(11):5995. https://doi.org/10.3390/ijerph20115995
Chicago/Turabian StyleÁlvarez-Millán, Lesli, Daniel Castillo-Castillo, Rosa Quispe-Siccha, Argelia Pérez-Pacheco, Maia Angelova, Jesús Rivera-Sánchez, and Ruben Fossion. 2023. "Frailty Syndrome as a Transition from Compensation to Decompensation: Application to the Biomechanical Regulation of Gait" International Journal of Environmental Research and Public Health 20, no. 11: 5995. https://doi.org/10.3390/ijerph20115995
APA StyleÁlvarez-Millán, L., Castillo-Castillo, D., Quispe-Siccha, R., Pérez-Pacheco, A., Angelova, M., Rivera-Sánchez, J., & Fossion, R. (2023). Frailty Syndrome as a Transition from Compensation to Decompensation: Application to the Biomechanical Regulation of Gait. International Journal of Environmental Research and Public Health, 20(11), 5995. https://doi.org/10.3390/ijerph20115995









