Accessibility of Psychological Treatments for Bulimia Nervosa: A Review of Efficacy and Engagement in Online Self-Help Treatments
Abstract
1. Introduction
1.1. Bulimia Nervosa
1.1.1. Clinical Description of Bulimia Nervosa
1.1.2. Epidemiology
1.1.3. Burden and Cost
1.1.4. Course of Illness
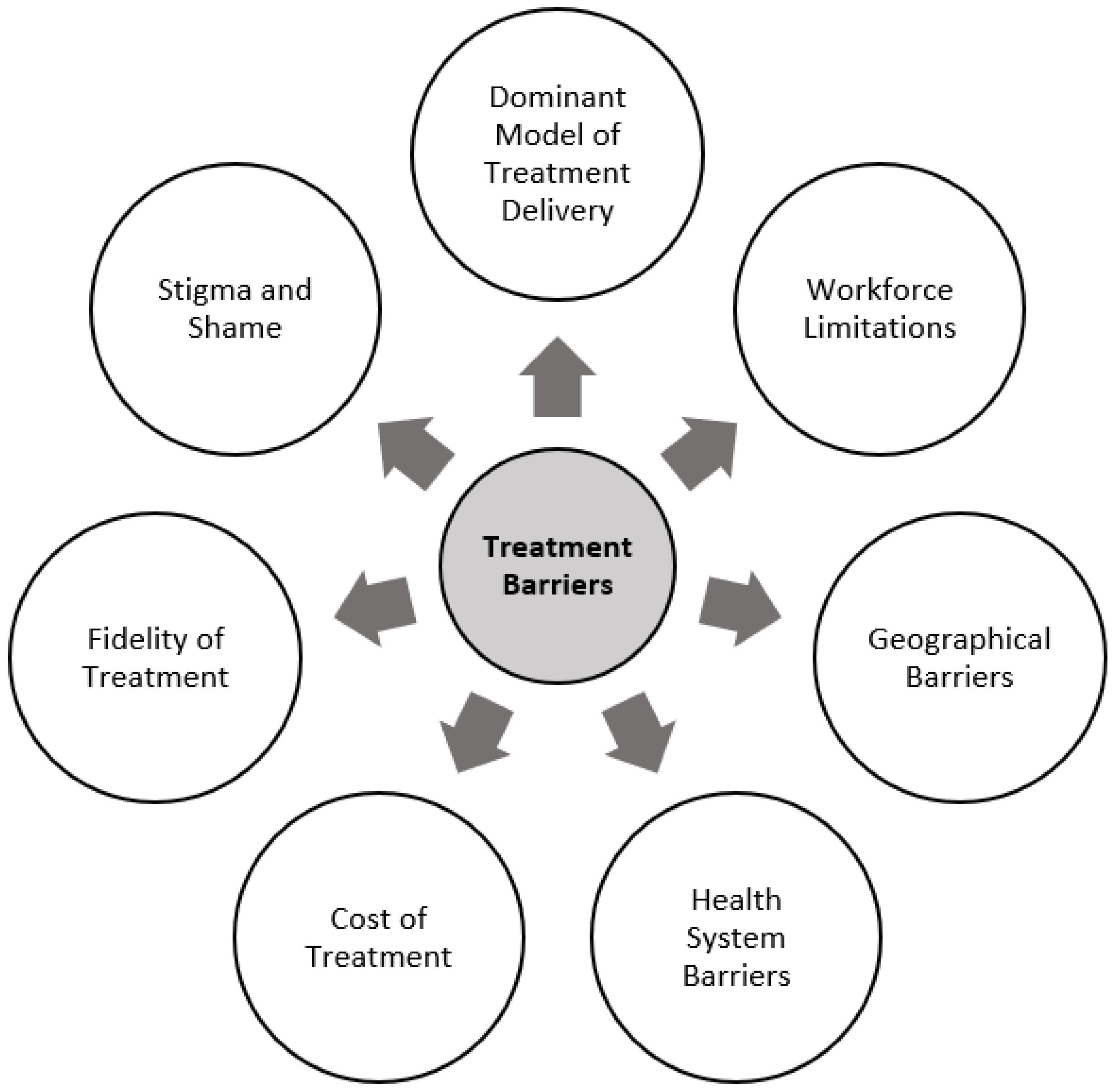
2. Treatment Barriers
2.1. Dominant Model of Treatment Delivery
2.2. Workforce Limitations
2.3. Geographical Barriers
2.4. Health System Barriers
2.5. Cost of Treatment
2.6. Fidelity of Treatment
2.7. Stigma and Shame
3. Treatment of Bulimia Nervosa
- Interpersonal Psychotherapy
- Dialectical Behaviour Therapy
- Psychoanalytic Therapy
- Pharmacological Treatments
3.1. Cognitive Behavioural Therapy
3.1.1. Cognitive Behavioural Model of Bulimia Nervosa
3.1.2. Cognitive Behavioural Treatment for Bulimia Nervosa
3.1.3. Enhanced Cognitive Behavioural Therapy
3.1.4. Clinician-Supported and Pure Self-Help Cognitive Behavioural Therapy
4. Digital Mental Health Interventions
4.1. Digital Interventions for Eating Disorders
4.1.1. eTherapy Programs for Eating Disorders
4.1.2. eTherapy Programs for Bulimia Nervosa
4.1.3. Smartphone Applications
5. Engagement and Adherence
5.1. Human Support
5.2. Digital Design
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
References
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders: DSM-5-TR, 5th ed.; American Psychiatric Association: Arlington, VA, USA, 2022. [Google Scholar]
- Russell, G. Bulimia Nervosa: An Ominous Variant of Anorexia Nervosa. Psychol. Med. 1979, 9, 429–448. [Google Scholar] [CrossRef] [PubMed]
- Castillo, M.; Weiselberg, E. Bulimia Nervosa/Purging Disorder. Curr. Probl. Pediatr. Adolesc. Health Care 2017, 47, 85–94. [Google Scholar] [CrossRef] [PubMed]
- Gordon, R.A. The History of Bulimia Nervosa. In The Wiley Handbook of Eating Disorders; John Wiley & Sons, Ltd: Hoboken, NJ, USA, 2015; pp. 25–38. ISBN 978-1-118-57408-9. [Google Scholar]
- Russell, G.F.M. The History of Bulimia Nervosa. In Handbook of Treatment for Eating Disorders, 2nd ed.; The Guilford Press: New York, NY, USA, 1997; pp. 11–24. ISBN 978-1-57230-186-3. [Google Scholar]
- Habermas, T. The Psychiatric History of Anorexia Nervosa and Bulimia Nervosa: Weight Concerns and Bulimic Symptoms in Early Case Reports. Int. J. Eat. Disord. 1989, 8, 259–273. [Google Scholar] [CrossRef]
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders: DSM-3, 3rd ed.; American Psychiatric Association: Arlington, VA, USA, 1980. [Google Scholar]
- Huelin, R.; Iheanacho, I.; Payne, K.; Sandman, K. What’s in a Name? Systematic and Non-Systematic Literature Reviews, and Why the Distinction Matters. Evidence 2015, 34–37. Available online: https://www.evidera.com/wp-content/uploads/2015/06/Whats-in-a-Name-Systematic-and-Non-Systematic-Literature-Reviews-and-Why-the-Distinction-Matters.pdf (accessed on 6 November 2022).
- Fitzsimmons-Craft, E.E.; Ciao, A.C.; Accurso, E.C.; Pisetsky, E.M.; Peterson, C.B.; Byrne, C.E.; Le Grange, D. Subjective and Objective Binge Eating in Relation to Eating Disorder Symptomatology, Depressive Symptoms, and Self-Esteem among Treatment-Seeking Adolescents with Bulimia Nervosa. Eur. Eat. Disord. Rev. 2014, 22, 230–236. [Google Scholar] [CrossRef] [PubMed]
- Fairburn, C.G.; Harrison, P.J. Eating Disorders. Lancet 2003, 361, 407–416. [Google Scholar] [CrossRef] [PubMed]
- Keski-Rahkonen, A.; Hoek, H.W.; Linna, M.S.; Raevuori, A.; Sihvola, E.; Bulik, C.M.; Rissanen, A.; Kaprio, J. Incidence and Outcomes of Bulimia Nervosa: A Nationwide Population-Based Study. Psychol. Med. 2009, 39, 823–831. [Google Scholar] [CrossRef] [PubMed]
- Forrest, L.N.; Jones, P.J.; Ortiz, S.N.; Smith, A.R. Core Psychopathology in Anorexia Nervosa and Bulimia Nervosa: A Network Analysis. Int. J. Eat. Disord. 2018, 51, 668–679. [Google Scholar] [CrossRef] [PubMed]
- Tabri, N.; Murray, H.B.; Thomas, J.J.; Franko, D.L.; Herzog, D.B.; Eddy, K.T. Overvaluation of Body Shape/Weight and Engagement in Non-Compensatory Weight-Control Behaviors in Eating Disorders: Is There a Reciprocal Relationship? Psychol. Med. 2015, 45, 2951–2958. [Google Scholar] [CrossRef]
- Bulik, C.M.; Marcus, M.D.; Zerwas, S.; Levine, M.D.; La Via, M. The Changing “Weightscape” of Bulimia Nervosa. Am. J. Psychiatry 2012, 169, 1031–1036. [Google Scholar] [CrossRef]
- Le Grange, D.; Loeb, K.L. Early Identification and Treatment of Eating Disorders: Prodrome to Syndrome. Early Interv. Psychiatry 2007, 1, 27–39. [Google Scholar] [CrossRef]
- Bagaric, M.; Touyz, S.; Heriseanu, A.; Conti, J.; Hay, P. Are Bulimia Nervosa and Binge Eating Disorder Increasing? Results of a Population-Based Study of Lifetime Prevalence and Lifetime Prevalence by Age in South Australia. Eur. Eat. Disord. Rev. 2020, 28, 260–268. [Google Scholar] [CrossRef] [PubMed]
- Hail, L.; Le Grange, D. Bulimia Nervosa in Adolescents: Prevalence and Treatment Challenges. Adolesc. Health Med. Ther. 2018, 9, 11–16. [Google Scholar] [CrossRef] [PubMed]
- Nagl, M.; Jacobi, C.; Paul, M.; Beesdo-Baum, K.; Höfler, M.; Lieb, R.; Wittchen, H.-U. Prevalence, Incidence, and Natural Course of Anorexia and Bulimia Nervosa among Adolescents and Young Adults. Eur. Child Adolesc. Psychiatry 2016, 25, 903–918. [Google Scholar] [CrossRef] [PubMed]
- Romano, K.A.; Lipson, S.K.; Beccia, A.L.; Quatromoni, P.A.; Gordon, A.R.; Murgueitio, J. Changes in the Prevalence and Sociodemographic Correlates of Eating Disorder Symptoms from 2013 to 2020 among a Large National Sample of U.S. Young Adults: A Repeated Cross-Sectional Study. Int. J. Eat. Disord. 2022, 55, 776–789. [Google Scholar] [CrossRef]
- Van Eeden, A.E.; van Hoeken, D.; Hoek, H.W. Incidence, Prevalence and Mortality of Anorexia Nervosa and Bulimia Nervosa. Curr. Opin. Psychiatry 2021, 34, 515–524. [Google Scholar] [CrossRef]
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders: DSM-5, 5th ed.; American Psychiatric Association: Arlington, VA, USA, 2013; ISBN 0-89042-554-X. [Google Scholar]
- Jones, J.M.; Bennett, S.; Olmsted, M.P.; Lawson, M.L.; Rodin, G. Disordered Eating Attitudes and Behaviours in Teenaged Girls: A School-Based Study. CMAJ 2001, 165, 547–552. [Google Scholar]
- Swanson, S.A.; Crow, S.J.; Le Grange, D.; Swendsen, J.; Merikangas, K.R. Prevalence and Correlates of Eating Disorders in Adolescents: Results From the National Comorbidity Survey Replication Adolescent Supplement. Arch. Gen. Psychiatry 2011, 68, 714–723. [Google Scholar] [CrossRef]
- Hudson, J.I.; Hiripi, E.; Pope, H.G.; Kessler, R.C. The Prevalence and Correlates of Eating Disorders in the National Comorbidity Survey Replication. Biol. Psychiatry 2007, 61, 348–358. [Google Scholar] [CrossRef]
- Micali, N.; Hagberg, K.W.; Petersen, I.; Treasure, J.L. The Incidence of Eating Disorders in the UK in 2000-2009: Findings from the General Practice Research Database. BMJ Open 2013, 3, e002646. [Google Scholar] [CrossRef]
- Kessler, R.C.; Berglund, P.A.; Chiu, W.T.; Deitz, A.C.; Hudson, J.I.; Shahly, V.; Aguilar-Gaxiola, S.; Alonso, J.; Angermeyer, M.C.; Benjet, C.; et al. The Prevalence and Correlates of Binge Eating Disorder in the World Health Organization World Mental Health Surveys. Biol. Psychiatry 2013, 73, 904–914. [Google Scholar] [CrossRef]
- Hilbert, A.; Pike, K.M.; Goldschmidt, A.B.; Wilfley, D.E.; Fairburn, C.G.; Dohm, F.-A.; Walsh, B.T.; Striegel Weissman, R. Risk Factors across the Eating Disorders. Psychiatry Res. 2014, 220, 500–506. [Google Scholar] [CrossRef] [PubMed]
- Favaro, A.; Caregaro, L.; Tenconi, E.; Bosello, R.; Santonastaso, P. Time Trends in Age at Onset of Anorexia Nervosa and Bulimia Nervosa. J. Clin. Psychiatry 2009, 70, 1715–1721. [Google Scholar] [CrossRef] [PubMed]
- Santomauro, D.F.; Melen, S.; Mitchison, D.; Vos, T.; Whiteford, H.; Ferrari, A.J. The Hidden Burden of Eating Disorders: An Extension of Estimates from the Global Burden of Disease Study 2019. Lancet Psychiatry 2021, 8, 320–328. [Google Scholar] [CrossRef] [PubMed]
- Sachs, K.; Mehler, P.S. Medical Complications of Bulimia Nervosa and Their Treatments. Eat. Weight Disord. 2016, 21, 13–18. [Google Scholar] [CrossRef] [PubMed]
- Udo, T.; Grilo, C.M. Psychiatric and Medical Correlates of DSM-5 Eating Disorders in a Nationally Representative Sample of Adults in the United States. Int. J. Eat. Disord. 2019, 52, 42–50. [Google Scholar] [CrossRef] [PubMed]
- Arcelus, J.; Mitchell, A.J.; Wales, J.; Nielsen, S. Mortality Rates in Patients With Anorexia Nervosa and Other Eating Disorders: A Meta-Analysis of 36 Studies. Arch. Gen. Psychiatry 2011, 68, 724–731. [Google Scholar] [CrossRef] [PubMed]
- Grilo, C.M.; White, M.A.; Masheb, R.M. DSM-IV Psychiatric Disorder Comorbidity and Its Correlates in Binge Eating Disorder. Int. J. Eat. Disord. 2009, 42, 228–234. [Google Scholar] [CrossRef] [PubMed]
- Chen, E.Y.; Le Grange, D. Subtyping Adolescents with Bulimia Nervosa. Behav. Res. Ther. 2007, 45, 2813–2820. [Google Scholar] [CrossRef]
- Carroll, J.M.; Touyz, S.W.; Beumont, P.J.V. Specific Comorbidity between Bulimia Nervosa and Personality Disorders. Int. J. Eat. Disord. 1996, 19, 159–170. [Google Scholar] [CrossRef]
- Nagata, T.; Kawarada, Y.; Kiriike, N.; Iketani, T. Multi-Impulsivity of Japanese Patients with Eating Disorders: Primary and Secondary Impulsivity. Psychiatry Res. 2000, 94, 239–250. [Google Scholar] [CrossRef]
- Crow, S.J.; Smiley, N. Costs and Cost-Effectiveness in Eating Disorders. In The Oxford Handbook of Eating Disorders; Oxford University Press: Oxford, UK; New York, NY, USA, 2010; p. 408. [Google Scholar]
- Striegel-Moore, R.H.; Dohm, F.-A.; Kraemer, H.C.; Schreiber, G.B.; Crawford, P.B.; Daniels, S.R. Health Services Use in Women with a History of Bulimia Nervosa or Binge Eating Disorder. Int. J. Eat. Disord. 2005, 37, 11–18. [Google Scholar] [CrossRef] [PubMed]
- Crow, S.J.; Frisch, M.J.; Peterson, C.B.; Croll, J.; Raatz, S.K.; Nyman, J.A. Monetary Costs Associated with Bulimia. Int. J. Eat. Disord. 2009, 42, 81–83. [Google Scholar] [CrossRef] [PubMed]
- van Hoeken, D.; Hoek, H.W. Review of the Burden of Eating Disorders: Mortality, Disability, Costs, Quality of Life, and Family Burden. Curr. Opin. Psychiatry 2020, 33, 521–527. [Google Scholar] [CrossRef] [PubMed]
- Ágh, T.; Kovács, G.; Supina, D.; Pawaskar, M.; Herman, B.K.; Vokó, Z.; Sheehan, D.V. A Systematic Review of the Health-Related Quality of Life and Economic Burdens of Anorexia Nervosa, Bulimia Nervosa, and Binge Eating Disorder. Eat. Weight Disord. EWD 2016, 21, 353–364. [Google Scholar] [CrossRef]
- Erskine, H.E.; Whiteford, H.A.; Pike, K.M. The Global Burden of Eating Disorders. Curr. Opin. Psychiatry 2016, 29, 346–353. [Google Scholar] [CrossRef]
- Streatfeild, J.; Hickson, J.; Austin, S.B.; Hutcheson, R.; Kandel, J.S.; Lampert, J.G.; Myers, E.M.; Richmond, T.K.; Samnaliev, M.; Velasquez, K.; et al. Social and Economic Cost of Eating Disorders in the United States: Evidence to Inform Policy Action. Int. J. Eat. Disord. 2021, 54, 851–868. [Google Scholar] [CrossRef]
- Deloitte Access Economics. Paying the Price: The Economic and Social Impact of Eating Disorders in Australia; The Butterfly Foundation: Crows Nest, NSW, Australia, 2012. [Google Scholar]
- Tannous, W.K.; Hay, P.; Girosi, F.; Heriseanu, A.I.; Ahmed, M.U.; Touyz, S. The Economic Cost of Bulimia Nervosa and Binge Eating Disorder: A Population-Based Study. Psychol. Med. 2021, 1–15. [Google Scholar] [CrossRef]
- Mond, J.M.; Hay, P.J. Functional Impairment Associated with Bulimic Behaviors in a Community Sample of Men and Women. Int. J. Eat. Disord. 2007, 40, 391–398. [Google Scholar] [CrossRef]
- Quadflieg, N.; Fichter, M.M. Long-Term Outcome of Inpatients with Bulimia Nervosa—Results from the Christina Barz Study. Int. J. Eat. Disord. 2019, 52, 834–845. [Google Scholar] [CrossRef]
- Steinhausen, H.-C.; Weber, S. The Outcome of Bulimia Nervosa: Findings from One-Quarter Century of Research. Am. J. Psychiatry 2009, 166, 1331–1341. [Google Scholar] [CrossRef]
- Wonderlich, S.; Mitchell, J.E.; Crosby, R.D.; Myers, T.C.; Kadlec, K.; Lahaise, K.; Swan-Kremeier, L.; Dokken, J.; Lange, M.; Dinkel, J.; et al. Minimizing and Treating Chronicity in the Eating Disorders: A Clinical Overview. Int. J. Eat. Disord. 2012, 45, 467–475. [Google Scholar] [CrossRef] [PubMed]
- Eddy, K.T.; Tabri, N.; Thomas, J.J.; Murray, H.B.; Keshaviah, A.; Hastings, E.; Edkins, K.; Krishna, M.; Herzog, D.B.; Keel, P.K.; et al. Recovery From Anorexia Nervosa and Bulimia Nervosa at 22-Year Follow-Up. J. Clin. Psychiatry 2017, 78, 184–189. [Google Scholar] [CrossRef] [PubMed]
- Hart, L.M.; Granillo, M.T.; Jorm, A.F.; Paxton, S.J. Unmet Need for Treatment in the Eating Disorders: A Systematic Review of Eating Disorder Specific Treatment Seeking among Community Cases. Clin. Psychol. Rev. 2011, 31, 727–735. [Google Scholar] [CrossRef] [PubMed]
- Eisenberg, D.; Nicklett, E.J.; Roeder, K.; Kirz, N.E. Eating Disorder Symptoms among College Students: Prevalence, Persistence, Correlates, and Treatment-Seeking. J. Am. Coll. Health 2011, 59, 700–707. [Google Scholar] [CrossRef] [PubMed]
- Meyer, D.F. Help-Seeking for Eating Disorders in Female Adolescents. J. Coll. Stud. Psychother. 2001, 15, 23–36. [Google Scholar] [CrossRef]
- Hamilton, A.; Mitchison, D.; Basten, C.; Byrne, S.; Goldstein, M.; Hay, P.; Heruc, G.; Thornton, C.; Touyz, S. Understanding Treatment Delay: Perceived Barriers Preventing Treatment-Seeking for Eating Disorders. Aust. N. Z. J. Psychiatry 2021, 56, 248–259. [Google Scholar] [CrossRef]
- National Eating Disorders Collaboration. Practical Responses to Eating Disorders A Guide to Implementing Responses to Eating Disorders in General Health Services; National Eating Disorders Collaboration: New York, NY, USA, 2014. [Google Scholar]
- Kazdin, A.E.; Fitzsimmons-Craft, E.E.; Wilfley, D.E. Addressing Critical Gaps in the Treatment of Eating Disorders. Int. J. Eat. Disord. 2017, 50, 170–189. [Google Scholar] [CrossRef]
- Shidhaye, R.; Lund, C.; Chisholm, D. Closing the Treatment Gap for Mental, Neurological and Substance Use Disorders by Strengthening Existing Health Care Platforms: Strategies for Delivery and Integration of Evidence-Based Interventions. Int. J. Ment. Health Syst. 2015, 9, 40. [Google Scholar] [CrossRef]
- Bauer, S.; Moessner, M. Harnessing the Power of Technology for the Treatment and Prevention of Eating Disorders. Int. J. Eat. Disord. 2013, 46, 508–515. [Google Scholar] [CrossRef]
- Cooper, Z.; Bailey-Straebler, S. Disseminating Evidence-Based Psychological Treatments for Eating Disorders. Curr. Psychiatry Rep. 2015, 17, 551. [Google Scholar] [CrossRef]
- Kazdin, A.E.; Blase, S.L. Rebooting Psychotherapy Research and Practice to Reduce the Burden of Mental Illness. Perspect. Psychol. Sci. 2011, 6, 21–37. [Google Scholar] [CrossRef] [PubMed]
- Striegel Weissman, R.; Rosselli, F. Reducing the Burden of Suffering from Eating Disorders: Unmet Treatment Needs, Cost of Illness, and the Quest for Cost-Effectiveness. Behav. Res. Ther. 2017, 88, 49–64. [Google Scholar] [CrossRef] [PubMed]
- Regan, P.; Cachelin, F.M.; Minnick, A.M. Initial treatment seeking from professional health care providers for eating disorders: A review and synthesis of potential barriers to and facilitators of “first contact”. Int. J. Eat. Disord. 2017, 50, 190–209. [Google Scholar] [CrossRef] [PubMed]
- Ali, K.; Farrer, L.; Fassnacht, D.B.; Gulliver, A.; Bauer, S.; Griffiths, K.M. Perceived Barriers and Facilitators towards Help-Seeking for Eating Disorders: A Systematic Review. Int. J. Eat. Disord. 2017, 50, 9–21. [Google Scholar] [CrossRef]
- Australian Psychological Society. Underfunded and Overworked: Psychologists on the Brink; Australian Psychological Society: Melbourne, Australia, 2022. [Google Scholar]
- The Royal Australian and New Zealand College of Psychiatrists. Psychiatry Workforce Shortage Should Be Top Priority; The Royal Australian and New Zealand College of Psychiatrists: Melbourne, Australia, 2021. [Google Scholar]
- National Eating Disorders Collaboration. Eating Disorders Prevention, Treatment and Management: An Evidence Review.; Commissioned by the Commonwealth Department of Health and Aging: Canberra, Australia, 2010. [Google Scholar]
- National Rural Health Alliance. National Rural Health Alliance Inc. Yearbook Annual Report 2014–2015; National Rural Health Alliance: Deakin, Australia, 2015. [Google Scholar]
- Productivity Commission. Productivity Commision Inquiry into Mental Health—Final Report; Mental Health Australia: Canberra, Australia, 2020. [Google Scholar]
- Stuhldreher, N.; Konnopka, A.; Wild, B.; Herzog, W.; Zipfel, S.; Löwe, B.; König, H.-H. Cost-of-Illness Studies and Cost-Effectiveness Analyses in Eating Disorders: A Systematic Review. Int. J. Eat. Disord. 2012, 45, 476–491. [Google Scholar] [CrossRef]
- The National Eating Disorders Collaboration. Eating Disorders Factsheet; Australian Government Department of Health: Canberra, Australia, 2019. [Google Scholar]
- Hay, P. A New Era in Service Provision for People with Eating Disorders in Australia: 2019 Medicare Benefit Schedule Items and the Role of Psychiatrists. Australas. Psychiatry 2020, 28, 125–127. [Google Scholar] [CrossRef]
- Duckett, S.; Stobart, A.; Lin, L. Not so Universal: How to Reduce Out-of-Pocket Healthcare Payments.; Grattan Institute: Carlton, Australia, 2022. [Google Scholar]
- Australian Bureau of Statistics. Patient Experiences in Australia: Summary of Findings; Australian Bureau of Statistics: Canberra, Australia, 2020. [Google Scholar]
- Callander, E.J.; Corscadden, L.; Levesque, J.-F. Out-of-Pocket Healthcare Expenditure and Chronic Disease—Do Australians Forgo Care Because of the Cost? Aust. J. Prim. Health 2017, 23, 15–22. [Google Scholar] [CrossRef]
- Waller, G. Treatment Protocols for Eating Disorders: Clinicians’ Attitudes, Concerns, Adherence and Difficulties Delivering Evidence-Based Psychological Interventions. Curr. Psychiatry Rep. 2016, 18, 36. [Google Scholar] [CrossRef]
- Waller, G.; Turner, H. Therapist Drift Redux: Why Well-Meaning Clinicians Fail to Deliver Evidence-Based Therapy, and How to Get Back on Track. Behav. Res. Ther. 2016, 77, 129–137. [Google Scholar] [CrossRef]
- Tobin, D.L.; Banker, J.D.; Weisberg, L.; Bowers, W. I Know What You Did Last Summer (and It Was Not CBT): A Factor Analytic Model of International Psychotherapeutic Practice in the Eating Disorders. Int. J. Eat. Disord. 2007, 40, 754–757. [Google Scholar] [CrossRef]
- Becker, A.E.; Hadley Arrindell, A.; Perloe, A.; Fay, K.; Striegel-Moore, R.H. A Qualitative Study of Perceived Social Barriers to Care for Eating Disorders: Perspectives from Ethnically Diverse Health Care Consumers. Int. J. Eat. Disord. 2010, 43, 633–647. [Google Scholar] [CrossRef] [PubMed]
- Puhl, R.; Suh, Y. Stigma and Eating and Weight Disorders. Curr. Psychiatry Rep. 2015, 17, 552. [Google Scholar] [CrossRef] [PubMed]
- Hay, P.; Chinn, D.; Forbes, D.; Madden, S.; Newton, R.; Sugenor, L.; Touyz, S.; Ward, W. Royal Australian and New Zealand College of Psychiatrists Clinical Practice Guidelines for the Treatment of Eating Disorders. Aust. N. Z. J. Psychiatry 2014, 48, 977–1008. [Google Scholar] [CrossRef] [PubMed]
- Hay, P.P.; Bacaltchuk, J.; Stefano, S.; Kashyap, P. Psychological Treatments for Bulimia Nervosa and Binging. Cochrane Database Syst. Rev. 2009, 2009, CD000562. [Google Scholar] [CrossRef] [PubMed]
- Linardon, J.; Fairburn, C.G.; Fitzsimmons-Craft, E.E.; Wilfley, D.E.; Brennan, L. The Empirical Status of the Third-Wave Behaviour Therapies for the Treatment of Eating Disorders: A Systematic Review. Clin. Psychol. Rev. 2017, 58, 125–140. [Google Scholar] [CrossRef]
- Jacobs, M.J.; Welch, R.R.; Wilfley, D.E. Interpersonal Psychotherapy for Anorexia Nervosa, Bulimia Nervosa, and Binge Eating Disorder. In Clinical Handbook of Eating Disorders; CRC Press: Boca Raton, FL, USA, 2004; ISBN 978-0-429-22858-2. [Google Scholar]
- Linardon, J.; Wade, T.D.; de la Piedad Garcia, X.; Brennan, L. The Efficacy of Cognitive-Behavioral Therapy for Eating Disorders: A Systematic Review and Meta-Analysis. J. Consult. Clin. Psychol. 2017, 85, 1080–1094. [Google Scholar] [CrossRef]
- Agras, W.S.; Walsh, B.T.; Fairburn, C.G.; Wilson, G.T.; Kraemer, H.C. A Multicenter Comparison of Cognitive-Behavioral Therapy and Interpersonal Psychotherapy for Bulimia Nervosa. Arch. Gen. Psychiatry 2000, 57, 459–466. [Google Scholar] [CrossRef]
- Fairburn, C.G.; Jones, R.; Peveler, R.C.; Hope, R.A.; O’Connor, M. Psychotherapy and Bulimia Nervosa: Longer-Term Effects of Interpersonal Psychotherapy, Behavior Therapy, and Cognitive Behavior Therapy. Arch. Gen. Psychiatry 1993, 50, 419–428. [Google Scholar] [CrossRef]
- Fairburn, C.G.; Bailey-Straebler, S.; Basden, S.; Doll, H.A.; Jones, R.; Murphy, R.; O’Connor, M.E.; Cooper, Z. A Transdiagnostic Comparison of Enhanced Cognitive Behaviour Therapy (CBT-E) and Interpersonal Psychotherapy in the Treatment of Eating Disorders. Behav. Res. Ther. 2015, 70, 64–71. [Google Scholar] [CrossRef]
- Safer, D.L.; Telch, C.F.; Agras, W.S. Dialectical Behavior Therapy for Bulimia Nervosa. Am. J. Psychiatry 2001, 158, 632–634. [Google Scholar] [CrossRef]
- Wisniewski, L.; Kelly, E. The Application of Dialectical Behavior Therapy to the Treatment of Eating Disorders. Cogn. Behav. Pract. 2003, 10, 131–138. [Google Scholar] [CrossRef]
- Safer, D.L.; Telch, C.F.; Chen, E.Y. Dialectical Behavior Therapy for Binge Eating and Bulimia; Guilford Press: New York, NY, USA, 2009; ISBN 978-1-60623-265-1. [Google Scholar]
- Bankoff, S.M.; Karpel, M.G.; Forbes, H.E.; Pantalone, D.W. A Systematic Review of Dialectical Behavior Therapy for the Treatment of Eating Disorders. Eat. Disord. 2012, 20, 196–215. [Google Scholar] [CrossRef] [PubMed]
- Bachar, E.; Latzer, Y.; Kreitler, S.; Berry, E.M. Empirical Comparison of Two Psychological Therapies: Self Psychology and Cognitive Orientation in the Treatment of Anorexia and Bulimia. J. Psychother. Pract. Res. 1999, 8, 115–128. [Google Scholar] [PubMed]
- Garner, D.M.; Rockert, W.; Davis, R.; Garner, M.V.; Olmsted, M.P.; Eagle, M. Comparison of Cognitive-Behavioral and Supportive-Expressive Therapy for Bulimia Nervosa. Am. J. Psychiatry 1993, 150, 37–46. [Google Scholar] [CrossRef]
- Poulsen, S.; Lunn, S.; Daniel, S.I.F.; Folke, S.; Mathiesen, B.B.; Katznelson, H.; Fairburn, C.G. A Randomized Controlled Trial of Psychoanalytic Psychotherapy or Cognitive-Behavioral Therapy for Bulimia Nervosa. Am. J. Psychiatry 2014, 171, 109–116. [Google Scholar] [CrossRef]
- Hay, P.J.; Claudino, A.M. Bulimia Nervosa: Online Interventions. BMJ Clin. Evid. 2015, 2015, 1009. [Google Scholar]
- McElroy, S.L.; Guerdjikova, A.I.; Mori, N.; Romo-Nava, F. Progress in Developing Pharmacologic Agents to Treat Bulimia Nervosa. CNS Drugs 2019, 33, 31–46. [Google Scholar] [CrossRef]
- Mitchell, J.E.; Agras, S.; Wonderlich, S. Treatment of Bulimia Nervosa: Where Are We and Where Are We Going? Int. J. Eat. Disord. 2007, 40, 95–101. [Google Scholar] [CrossRef]
- Keshen, A.; Bartel, S.; Frank, G.K.W.; Svedlund, N.E.; Nunes, A.; Dixon, L.; Ali, S.I.; Kaplan, A.S.; Hay, P.; Touyz, S.; et al. The Potential Role of Stimulants in Treating Eating Disorders. Int. J. Eat. Disord. 2022, 55, 318–331. [Google Scholar] [CrossRef]
- Keshen, A.R.; Dixon, L.; Ali, S.I.; Helson, T.; Nunes, A.; Milliken, H.; Gamberg, S.; Sadek, J.; Kaplan, A.; McElroy, S.L. A Feasibility Study Evaluating Lisdexamfetamine Dimesylate for the Treatment of Adults with Bulimia Nervosa. Int. J. Eat. Disord. 2021, 54, 872–878. [Google Scholar] [CrossRef]
- Hay, P. A Systematic Review of Evidence for Psychological Treatments in Eating Disorders: 2005–2012. Int. J. Eat. Disord. 2013, 46, 462–469. [Google Scholar] [CrossRef] [PubMed]
- Hay, P. Current Approach to Eating Disorders: A Clinical Update. Intern. Med. J. 2020, 50, 24–29. [Google Scholar] [CrossRef] [PubMed]
- NICE Guidelines. Recommendations Eating Disorders: Recognition and Treatment. Available online: https://www.nice.org.uk/guidance/ng69/chapter/Recommendations#treating-bulimia-nervosa (accessed on 24 March 2022).
- Shapiro, J.R.; Berkman, N.D.; Brownley, K.A.; Sedway, J.A.; Lohr, K.N.; Bulik, C.M. Bulimia Nervosa Treatment: A Systematic Review of Randomized Controlled Trials. Int. J. Eat. Disord. 2007, 40, 321–336. [Google Scholar] [CrossRef] [PubMed]
- Fairburn, C.G.; Kirk, J.; O’Connor, M.; Cooper, P.J. A Comparison of Two Psychological Treatments for Bulimia Nervosa. Behav. Res. Ther. 1986, 24, 629–643. [Google Scholar] [CrossRef]
- Fairburn, C.G.; Cooper, Z.; Shafran, R. Cognitive Behaviour Therapy for Eating Disorders: A “Transdiagnostic” Theory and Treatment. Behav. Res. Ther. 2003, 41, 509–528. [Google Scholar] [CrossRef]
- Fairburn, C.G.; Cooper, P.J. Eating Disorders. In Cognitive Behaviour Therapy for Psychiatric Problems. A Practical Guide; Oxford University Press: Oxford, UK, 1989; pp. 277–314. [Google Scholar]
- Fairburn, C.G.; Marcus, M.D.; Wilson, G.T. Cognitive-Behavioral Therapy for Binge Eating and Bulimia Nervosa: A Comprehensive Treatment Manual. In Binge Eating: Nature, Assessment, and Treatment; Guilford Press: New York, NY, USA, 1993; pp. 361–404. ISBN 978-0-89862-995-8. [Google Scholar]
- Waller, G.; Gray, E.; Hinrichsen, H.; Mountford, V.; Lawson, R.; Patient, E. Cognitive-Behavioral Therapy for Bulimia Nervosa and Atypical Bulimic Nervosa: Effectiveness in Clinical Settings. Int. J. Eat. Disord. 2014, 47, 13–17. [Google Scholar] [CrossRef]
- Wilson, G.T.; Fairburn, C.C.; Agras, W.S.; Walsh, B.T.; Kraemer, H. Cognitive-Behavioral Therapy for Bulimia Nervosa: Time Course and Mechanisms of Change. J. Consult. Clin. Psychol. 2002, 70, 267–274. [Google Scholar] [CrossRef]
- Cooper, P.J.; Steere, J. A Comparison of Two Psychological Treatments for Bulimia Nervosa: Implications for Models of Maintenance. Behav. Res. Ther. 1995, 33, 875–885. [Google Scholar] [CrossRef]
- Wilson, G.T.; Grilo, C.M.; Vitousek, K.M. Psychological Treatment of Eating Disorders. Am. Psychol. 2007, 62, 199–216. [Google Scholar] [CrossRef]
- Wilson, G.T.; Loeb, K.L.; Walsh, B.T.; Labouvie, E.; Petkova, E.; Liu, X.; Waternaux, C. Psychological versus Pharmacological Treatments of Bulimia Nervosa: Predictors and Processes of Change. J. Consult. Clin. Psychol. 1999, 67, 451–459. [Google Scholar] [CrossRef]
- Chen, E.; Touyz, S.W.; Beumont, P.J.V.; Fairburn, C.G.; Griffiths, R.; Butow, P.; Russell, J.; Schotte, D.E.; Gertler, R.; Basten, C. Comparison of Group and Individual Cognitive-Behavioral Therapy for Patients with Bulimia Nervosa. Int. J. Eat. Disord. 2003, 33, 241–254. [Google Scholar] [CrossRef] [PubMed]
- Castellini, G.; Mannucci, E.; Lo Sauro, C.; Benni, L.; Lazzeretti, L.; Ravaldi, C.; Rotella, C.M.; Faravelli, C.; Ricca, V. Different Moderators of Cognitive-Behavioral Therapy on Subjective and Objective Binge Eating in Bulimia Nervosa and Binge Eating Disorder: A Three-Year Follow-up Study. Psychother. Psychosom. 2012, 81, 11–20. [Google Scholar] [CrossRef] [PubMed]
- Linardon, J.; de la Piedad Garcia, X.; Brennan, L. Predictors, Moderators, and Mediators of Treatment Outcome Following Manualised Cognitive-Behavioural Therapy for Eating Disorders: A Systematic Review. Eur. Eat. Disord. Rev. 2017, 25, 3–12. [Google Scholar] [CrossRef] [PubMed]
- Fairburn, C.G. Eating Disorders: The Transdiagnostic View and the Cognitive Behavioral Theory. In Cognitive Behavior Therapy and Eating Disorders; Guilford Press: New York, NY, USA, 2008; pp. 7–22. ISBN 978-1-59385-709-7. [Google Scholar]
- Fairburn, C.G.; Cooper, Z.; Shafran, R.; Wilson, G.T. Eating Disorders: A Transdiagnostic Protocol. In Clinical Handbook of Psychological Disorders: A Step-by-Step Treatment Manual, 4th ed.; The Guilford Press: New York, NY, USA, 2008; pp. 578–614. ISBN 978-1-59385-572-7. [Google Scholar]
- De Jong, M.; Schoorl, M.; Hoek, H.W. Enhanced Cognitive Behavioural Therapy for Patients with Eating Disorders: A Systematic Review. Curr. Opin. Psychiatry 2018, 31, 436–444. [Google Scholar] [CrossRef]
- Groff, S.E. Is Enhanced Cognitive Behavioral Therapy an Effective Intervention in Eating Disorders? A Review. J. Evid. Inf. Soc. Work 2015, 12, 272–288. [Google Scholar] [CrossRef]
- Svaldi, J.; Schmitz, F.; Baur, J.; Hartmann, A.S.; Legenbauer, T.; Thaler, C.; von Wietersheim, J.; de Zwaan, M.; Tuschen-Caffier, B. Efficacy of Psychotherapies and Pharmacotherapies for Bulimia Nervosa. Psychol. Med. 2019, 49, 898–910. [Google Scholar] [CrossRef]
- Atwood, M.E.; Friedman, A. A Systematic Review of Enhanced Cognitive Behavioral Therapy (CBT-E) for Eating Disorders. Int. J. Eat. Disord. 2020, 53, 311–330. [Google Scholar] [CrossRef]
- Slade, E.; Keeney, E.; Mavranezouli, I.; Dias, S.; Fou, L.; Stockton, S.; Saxon, L.; Waller, G.; Turner, H.; Serpell, L.; et al. Treatments for Bulimia Nervosa: A Network Meta-Analysis. Psychol. Med. 2018, 48, 2629–2636. [Google Scholar] [CrossRef]
- Perkins, S.J.; Murphy, R.; Schmidt, U.; Williams, C. Self-Help and Guided Self-Help for Eating Disorders. Cochrane Database Syst. Rev. 2006, CD004191. [Google Scholar] [CrossRef]
- Wilson, G.T.; Zandberg, L.J. Cognitive-Behavioral Guided Self-Help for Eating Disorders: Effectiveness and Scalability. Clin. Psychol. Rev. 2012, 32, 343–357. [Google Scholar] [CrossRef]
- Yim, S.H.; Schmidt, U. Self-Help Treatment of Eating Disorders. Psychiatr. Clin. North Am. 2019, 42, 231–241. [Google Scholar] [CrossRef] [PubMed]
- Fairburn, C.G. Overcoming Binge Eating; Guilford Press: New York, NY, USA, 1995. [Google Scholar]
- Cooper, P. Overcoming Bulimia Nervosa and Binge Eating: A Guide to Recovery; Robinson: Hachette, UK, 1995; ISBN 978-1-4721-0571-4. [Google Scholar]
- Schmidt, U.; Treasure, J.; Alexander, J. Getting Better Bite by Bite: A Survival Kit for Sufferers of Bulimia Nervosa and Binge Eating Disorders, 2nd ed.; Routledge: London, UK, 2015; ISBN 978-1-315-75401-7. [Google Scholar]
- Grilo, C.M.; Mitchell, J.E. The Treatment of Eating Disorders: A Clinical Handbook; Guilford Press: New York, NY, USA, 2009; ISBN 978-1-60623-447-1. [Google Scholar]
- Cuijpers, P.; Donker, T.; van Straten, A.; Li, J.; Andersson, G. Is Guided Self-Help as Effective as Face-to-Face Psychotherapy for Depression and Anxiety Disorders? A Systematic Review and Meta-Analysis of Comparative Outcome Studies. Psychol. Med. 2010, 40, 1943–1957. [Google Scholar] [CrossRef] [PubMed]
- Cooper, P.J.; Coker, S.; Fleming, C. Self-Help for Bulimia Nervosa: A Preliminary Report. Int. J. Eat. Disord. 1994, 16, 401–404. [Google Scholar] [CrossRef] [PubMed]
- Cooper, P.J.; Coker, S.; Fleming, C. An Evaluation of the Efficacy of Supervised Cognitive Behavioral Self-Help for Bulimia Nervosa. J. Psychosom. Res. 1996, 40, 281–287. [Google Scholar] [CrossRef] [PubMed]
- Treasure, J.; Schmidt, U.; Troop, N.; Tiller, J.; Todd, G.; Turnbull, S. Sequential Treatment for Bulimia Nervosa Incorporating A Self-Care Manual. Br. J. Psychiatry 1996, 168, 94–98. [Google Scholar] [CrossRef]
- Traviss-Turner, G.D.; West, R.M.; Hill, A.J. Guided Self-Help for Eating Disorders: A Systematic Review and Metaregression. Eur. Eat. Disord. Rev. 2017, 25, 148–164. [Google Scholar] [CrossRef]
- Palmer, R.L.; Birchall, H.; Mcgrain, L.; Sullivan, V. Self-Help for Bulimic Disorders: A Randomised Controlled Trial Comparing Minimal Guidance with Face-to-Face or Telephone Guidance. Br. J. Psychiatry 2002, 181, 230–235. [Google Scholar] [CrossRef]
- Striegel-Moore, R.H.; Wilson, G.T.; DeBar, L.; Perrin, N.; Lynch, F.; Rosselli, F.; Kraemer, H.C. Cognitive Behavioral Guided Self-Help for the Treatment of Recurrent Binge Eating. J. Consult. Clin. Psychol. 2010, 78, 312–321. [Google Scholar] [CrossRef]
- Banasiak, S.J.; Paxton, S.J.; Hay, P. Guided Self-Help for Bulimia Nervosa in Primary Care: A Randomized Controlled Trial. Psychol. Med. 2005, 35, 1283–1294. [Google Scholar] [CrossRef]
- Schmidt, U.; Lee, S.; Beecham, J.; Perkins, S.; Treasure, J.; Yi, I.; Winn, S.; Robinson, P.; Murphy, R.; Keville, S.; et al. A Randomized Controlled Trial of Family Therapy and Cognitive Behavior Therapy Guided Self-Care for Adolescents With Bulimia Nervosa and Related Disorders. Am. J. Psychiatry 2007, 164, 591–598. [Google Scholar] [CrossRef]
- Mitchell, J.E.; Agras, S.; Crow, S.; Halmi, K.; Fairburn, C.G.; Bryson, S.; Kraemer, H. Stepped Care and Cognitive–Behavioural Therapy for Bulimia Nervosa: Randomised Trial. Br. J. Psychiatry 2011, 198, 391–397. [Google Scholar] [CrossRef]
- Thiels, C.; Schmidt, U.; Troop, N.; Treasure, J.; Garthe, R. Binge Frequency Predicts Outcome in Guided Self-Care Treatment of Bulimia Nervosa. Eur. Eat. Disord. Rev. 2000, 8, 272–278. [Google Scholar] [CrossRef]
- Bennett-Levy, J.; Richards, D.A.; Farrand, P. Low Intensity CBT Interventions: A Revolution in Mental Health Care. In Low Intensity CBT Interventions; Oxford University Press: Oxford, UK, 2010; pp. 3–18. [Google Scholar]
- Harvey, A.G.; Gumport, N.B. Evidence-Based Psychological Treatments for Mental Disorders: Modifiable Barriers to Access and Possible Solutions. Behav. Res. Ther. 2015, 68, 1–12. [Google Scholar] [CrossRef]
- Bower, P.; Gilbody, S. Stepped Care in Psychological Therapies: Access, Effectiveness and Efficiency: Narrative Literature Review. Br. J. Psychiatry 2005, 186, 11–17. [Google Scholar] [CrossRef]
- Lavori, P.W.; Dawson, R. Adaptive Treatment Strategies in Chronic Disease. Annu. Rev. Med. 2008, 59, 443–453. [Google Scholar] [CrossRef]
- Wilson, G.T.; Vitousek, K.M.; Loeb, K.L. Stepped Care Treatment for Eating Disorders. J. Consult. Clin. Psychol. 2000, 68, 564–572. [Google Scholar] [CrossRef]
- Van Straten, A.; Hill, J.; Richards, D.A.; Cuijpers, P. Stepped Care Treatment Delivery for Depression: A Systematic Review and Meta-Analysis. Psychol. Med. 2015, 45, 231–246. [Google Scholar] [CrossRef]
- Crow, S.J.; Agras, W.S.; Halmi, K.A.; Fairburn, C.G.; Mitchell, J.E.; Nyman, J.A. A Cost Effectiveness Analysis of Stepped Care Treatment for Bulimia Nervosa. Int. J. Eat. Disord. 2013, 46, 302–307. [Google Scholar] [CrossRef]
- Haderlein, T.P. Efficacy of Technology-Based Eating Disorder Treatment: A Meta-Analysis. Curr. Psychol. 2022, 41, 174–184. [Google Scholar] [CrossRef]
- Aardoom, J.J.; Dingemans, A.E.; Spinhoven, P.; Van Furth, E.F. Treating Eating Disorders over the Internet: A Systematic Review and Future Research Directions. Int. J. Eat. Disord. 2013, 46, 539–552. [Google Scholar] [CrossRef]
- Andersson, G.; Titov, N.; Dear, B.F.; Rozental, A.; Carlbring, P. Internet-Delivered Psychological Treatments: From Innovation to Implementation. World Psychiatry 2019, 18, 20–28. [Google Scholar] [CrossRef]
- Sysko, R. Self-Help Treatments for Eating Disorders. In Eating Disorders and Obesity: A Comprehensive Handbook; Guildford Publications: New York, NY, USA, 2017. [Google Scholar]
- Linardon, J.; Fuller-Tyszkiewicz, M. Attrition and Adherence in Smartphone-Delivered Interventions for Mental Health Problems: A Systematic and Meta-Analytic Review. J. Consult. Clin. Psychol. 2020, 88, 1–13. [Google Scholar] [CrossRef]
- Australian Bureau of Statistics. Internet Activity, Australia; Australian Bureau of Statistics: Canberra, Australia, 2018. [Google Scholar]
- Pittock, A.; Hodges, L.; Lawrie, S.M. The Effectiveness of Internet-Delivered Cognitive Behavioural Therapy for Those with Bulimic Symptoms: A Systematic Review: A Review of ICBT Treatment for Bulimic Symptoms. BMC Res. Notes 2018, 11, 748. [Google Scholar] [CrossRef]
- Collins, L.M.; Murphy, S.A.; Strecher, V. The Multiphase Optimization Strategy (MOST) and the Sequential Multiple Assignment Randomized Trial (SMART): New Methods for More Potent EHealth Interventions. Am. J. Prev. Med. 2007, 32, S112–S118. [Google Scholar] [CrossRef]
- Bush, N.E.; Armstrong, C.M.; Hoyt, T.V. Smartphone Apps for Psychological Health: A Brief State of the Science Review. Psychol. Serv. 2019, 16, 188–195. [Google Scholar] [CrossRef]
- Deutsche Telekom, A.G. Smart Payments—How the Cell Phone Becomes a Wallet; 2012. Available online: http://www.studie-life.de/en/life-reports/smart-payments (accessed on 6 November 2022).
- Pew Research Center. U.S. Smartphone Use in 2015; Pew Research Center: Washington DC, USA, 2015. [Google Scholar]
- Bakker, M.; Wicherts, J.M. Outlier Removal and the Relation with Reporting Errors and Quality of Psychological Research. PLoS ONE 2014, 9, e103360. [Google Scholar] [CrossRef]
- Zabinski, M.F.; Pung, M.A.; Wilfley, D.E.; Eppstein, D.L.; Winzelberg, A.J.; Celio, A.; Taylor, C.B. Reducing Risk Factors for Eating Disorders: Targeting at-Risk Women with a Computerized Psychoeducational Program. Int. J. Eat. Disord. 2001, 29, 401–408. [Google Scholar] [CrossRef]
- Zabinski, M.F.; Wilfley, D.E.; Pung, M.A.; Winzelberg, A.J.; Eldredge, K.; Taylor, C.B. An Interactive Internet-Based Intervention for Women at Risk of Eating Disorders: A Pilot Study. Int. J. Eat. Disord. 2001, 30, 129–137. [Google Scholar] [CrossRef]
- Foran, A.-M.; O’Donnell, A.T.; Muldoon, O.T. Stigma of Eating Disorders and Recovery-Related Outcomes: A Systematic Review. Eur. Eat. Disord. Rev. 2020, 28, 385–397. [Google Scholar] [CrossRef]
- Shingleton, R.M.; Richards, L.K.; Thompson-Brenner, H. Using Technology within the Treatment of Eating Disorders: A Clinical Practice Review. Psychotherapy 2013, 50, 576–582. [Google Scholar] [CrossRef][Green Version]
- Linardon, J.; Shatte, A.; Messer, M.; Firth, J.; Fuller-Tyszkiewicz, M. E-Mental Health Interventions for the Treatment and Prevention of Eating Disorders: An Updated Systematic Review and Meta-Analysis. J. Consult. Clin. Psychol. 2020, 88, 994–1007. [Google Scholar] [CrossRef]
- Dölemeyer, R.; Tietjen, A.; Kersting, A.; Wagner, B. Internet-Based Interventions for Eating Disorders in Adults: A Systematic Review. BMC Psychiatry 2013, 13, 207. [Google Scholar] [CrossRef]
- Loucas, C.E.; Fairburn, C.G.; Whittington, C.; Pennant, M.E.; Stockton, S.; Kendall, T. E-Therapy in the Treatment and Prevention of Eating Disorders: A Systematic Review and Meta-Analysis. Behav. Res. Ther. 2014, 63, 122–131. [Google Scholar] [CrossRef]
- Schlegl, S.; Bürger, C.; Schmidt, L.; Herbst, N.; Voderholzer, U. The Potential of Technology-Based Psychological Interventions for Anorexia and Bulimia Nervosa: A Systematic Review and Recommendations for Future Research. J. Med. Internet Res. 2015, 17, e3554. [Google Scholar] [CrossRef]
- Anastasiadou, D.; Folkvord, F.; Lupiañez-Villanueva, F. A Systematic Review of MHealth Interventions for the Support of Eating Disorders. Eur. Eat. Disord. Rev. 2018, 26, 394–416. [Google Scholar] [CrossRef]
- Melioli, T.; Bauer, S.; Franko, D.L.; Moessner, M.; Ozer, F.; Chabrol, H.; Rodgers, R.F. Reducing eating disorder symptoms and risk factors using the internet: A meta-analytic review. Int. J. Eat. Disord. 2016, 49, 19–31. [Google Scholar] [CrossRef]
- Aardoom, J.J.; Dingemans, A.E.; Spinhoven, P.; van Ginkel, J.R.; de Rooij, M.; van Furth, E.F. Web-Based Fully Automated Self-Help With Different Levels of Therapist Support for Individuals With Eating Disorder Symptoms: A Randomized Controlled Trial. J. Med. Internet Res. 2016, 18, e5709. [Google Scholar] [CrossRef]
- McClay, C.-A.; Waters, L.; Schmidt, U.; Williams, C. A Survey of Attitudes towards Computerized Self-Help for Eating Disorders within a Community-Based Sample. Behav. Cogn. Psychother. 2016, 44, 65–78. [Google Scholar] [CrossRef]
- Linardon, J.; Shatte, A.; Tepper, H.; Fuller-Tyszkiewicz, M. A Survey Study of Attitudes toward, and Preferences for, e-Therapy Interventions for Eating Disorder Psychopathology. Int. J. Eat. Disord. 2020, 53, 907–916. [Google Scholar] [CrossRef]
- Sánchez-Ortiz, V.C.; Munro, C.; Stahl, D.; House, J.; Startup, H.; Treasure, J.; Williams, C.; Schmidt, U. A Randomized Controlled Trial of Internet-Based Cognitive-Behavioural Therapy for Bulimia Nervosa or Related Disorders in a Student Population. Psychol. Med. 2011, 41, 407–417. [Google Scholar] [CrossRef]
- Hay, P.P.; Bacaltchuk, J.; Stefano, S. Psychotherapy for Bulimia Nervosa and Binging. Cochrane Database Syst. Rev. 2004, CD000562. [Google Scholar] [CrossRef]
- Pretorius, N.; Arcelus, J.; Beecham, J.; Dawson, H.; Doherty, F.; Eisler, I.; Gallagher, C.; Gowers, S.; Isaacs, G.; Johnson-Sabine, E.; et al. Cognitive-Behavioural Therapy for Adolescents with Bulimic Symptomatology: The Acceptability and Effectiveness of Internet-Based Delivery. Behav. Res. Ther. 2009, 47, 729–736. [Google Scholar] [CrossRef] [PubMed]
- Ruwaard, J.; Lange, A.; Broeksteeg, J.; Renteria-Agirre, A.; Schrieken, B.; Dolan, C.V.; Emmelkamp, P. Online Cognitive–Behavioural Treatment of Bulimic Symptoms: A Randomized Controlled Trial. Clin. Psychol. Psychother. 2013, 20, 308–318. [Google Scholar] [CrossRef] [PubMed]
- Wagner, G.; Penelo, E.; Wanner, C.; Gwinner, P.; Trofaier, M.-L.; Imgart, H.; Waldherr, K.; Wöber-Bingöl, Ç.; Karwautz, A.F.K. Internet-Delivered Cognitive-Behavioural Therapy v. Conventional Guided Self-Help for Bulimia Nervosa: Long-Term Evaluation of a Randomised Controlled Trial. Br. J. Psychiatry 2013, 202, 135–141. [Google Scholar] [CrossRef]
- Andrews, S.; Ellis, D.A.; Shaw, H.; Piwek, L. Beyond Self-Report: Tools to Compare Estimated and Real-World Smartphone Use. PLoS ONE 2015, 10, e0139004. [Google Scholar] [CrossRef]
- Bakker, D.; Kazantzis, N.; Rickwood, D.; Rickard, N. Mental Health Smartphone Apps: Review and Evidence-Based Recommendations for Future Developments. JMIR Ment. Health 2016, 3, e4984. [Google Scholar] [CrossRef]
- Firth, J.; Torous, J.; Nicholas, J.; Carney, R.; Rosenbaum, S.; Sarris, J. Can Smartphone Mental Health Interventions Reduce Symptoms of Anxiety? A Meta-Analysis of Randomized Controlled Trials. J. Affect. Disord. 2017, 218, 15–22. [Google Scholar] [CrossRef]
- Firth, J.; Torous, J.; Nicholas, J.; Carney, R.; Pratap, A.; Rosenbaum, S.; Sarris, J. The Efficacy of Smartphone-Based Mental Health Interventions for Depressive Symptoms: A Meta-Analysis of Randomized Controlled Trials. World Psychiatry 2017, 16, 287–298. [Google Scholar] [CrossRef]
- Linardon, J.; Cuijpers, P.; Carlbring, P.; Messer, M.; Fuller-Tyszkiewicz, M. The Efficacy of App-Supported Smartphone Interventions for Mental Health Problems: A Meta-Analysis of Randomized Controlled Trials. World Psychiatry 2019, 18, 325–336. [Google Scholar] [CrossRef]
- O’Leary, T.; Torous, J. Smartphone Apps for Eating Disorders: An Overview of the Marketplace and Research Trends. Int. J. Eat. Disord. 2022, 55, 625–632. [Google Scholar] [CrossRef]
- Fairburn, C.G.; Rothwell, E.R. Apps and Eating Disorders: A Systematic Clinical Appraisal. Int. J. Eat. Disord. 2015, 48, 1038–1046. [Google Scholar] [CrossRef]
- Wasil, A.R.; Patel, R.; Cho, J.Y.; Shingleton, R.M.; Weisz, J.R.; DeRubeis, R.J. Smartphone Apps for Eating Disorders: A Systematic Review of Evidence-Based Content and Application of User-Adjusted Analyses. Int. J. Eat. Disord. 2021, 54, 690–700. [Google Scholar] [CrossRef]
- Barakat, S.; Maguire, S.; Surgenor, L.; Donnelly, B.; Miceska, B.; Fromholtz, K.; Russell, J.; Hay, P.; Touyz, S. The Role of Regular Eating and Self-Monitoring in the Treatment of Bulimia Nervosa: A Pilot Study of an Online Guided Self-Help CBT Program. Behav. Sci. 2017, 7, 39. [Google Scholar] [CrossRef]
- Hildebrandt, T.; Latner, J. Effect of Self-Monitoring on Binge Eating: Treatment Response or ‘Binge Drift’? Eur. Eat. Disord. Rev. 2006, 14, 17–22. [Google Scholar] [CrossRef]
- Latner, J.D.; Wilson, G.T. Self-Monitoring and the Assessment of Binge Eating. Behav. Ther. 2002, 33, 465–477. [Google Scholar] [CrossRef]
- Hildebrandt, T.; Michaeledes, A.; Mayhew, M.; Greif, R.; Sysko, R.; Toro-Ramos, T.; DeBar, L. Randomized Controlled Trial Comparing Health Coach-Delivered Smartphone-Guided Self-Help With Standard Care for Adults With Binge Eating. Am. J. Psychiatry 2020, 177, 134–142. [Google Scholar] [CrossRef]
- Tregarthen, J.P.; Lock, J.; Darcy, A.M. Development of a Smartphone Application for Eating Disorder Self-Monitoring. Int. J. Eat. Disord. 2015, 48, 972–982. [Google Scholar] [CrossRef]
- Walsh, J.C.; Groarke, J.M. Integrating Behavioral Science with Mobile (MHealth) Technology to Optimize Health Behavior Change Interventions. Eur. Psychol. 2019, 24, 38–48. [Google Scholar] [CrossRef]
- Tregarthen, J.; Kim, J.P.; Sadeh-Sharvit, S.; Neri, E.; Welch, H.; Lock, J. Comparing a Tailored Self-Help Mobile App With a Standard Self-Monitoring App for the Treatment of Eating Disorder Symptoms: Randomized Controlled Trial. JMIR Ment. Health 2019, 6, e14972. [Google Scholar] [CrossRef]
- Hildebrandt, T.; Michaelides, A.; Mackinnon, D.; Greif, R.; DeBar, L.; Sysko, R. Randomized Controlled Trial Comparing Smartphone Assisted versus Traditional Guided Self-Help for Adults with Binge Eating. Int. J. Eat. Disord. 2017, 50, 1313–1322. [Google Scholar] [CrossRef]
- Linardon, J.; Rosato, J.; Messer, M. Break Binge Eating: Reach, Engagement, and User Profile of an Internet-Based Psychoeducational and Self-Help Platform for Eating Disorders. Int. J. Eat. Disord. 2020, 53, 1719–1728. [Google Scholar] [CrossRef] [PubMed]
- Linardon, J.; Messer, M.; Shatte, A.; Skvarc, D.; Rosato, J.; Rathgen, A.; Fuller-Tyszkiewicz, M. Targeting Dietary Restraint to Reduce Binge Eating: A Randomised Controlled Trial of a Blended Internet- and Smartphone App-Based Intervention. Psychol. Med. 2021, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Christensen, H.; Griffiths, K.M.; Korten, A.E.; Brittliffe, K.; Groves, C. A Comparison of Changes in Anxiety and Depression Symptoms of Spontaneous Users and Trial Participants of a Cognitive Behavior Therapy Website. J. Med. Internet Res. 2004, 6, e46. [Google Scholar] [CrossRef] [PubMed]
- Christensen, H.; Griffiths, K.M.; Farrer, L. Adherence in Internet Interventions for Anxiety and Depression: Systematic Review. J. Med. Internet Res. 2009, 11, e1194. [Google Scholar] [CrossRef] [PubMed]
- Torous, J.; Lipschitz, J.; Ng, M.; Firth, J. Dropout Rates in Clinical Trials of Smartphone Apps for Depressive Symptoms: A Systematic Review and Meta-Analysis. J. Affect. Disord. 2020, 263, 413–419. [Google Scholar] [CrossRef] [PubMed]
- Melville, K.M.; Casey, L.M.; Kavanagh, D.J. Dropout from Internet-Based Treatment for Psychological Disorders. Br. J. Clin. Psychol. 2010, 49, 455–471. [Google Scholar] [CrossRef]
- Fleming, T.; Bavin, L.; Lucassen, M.; Stasiak, K.; Hopkins, S.; Merry, S. Beyond the Trial: Systematic Review of Real-World Uptake and Engagement With Digital Self-Help Interventions for Depression, Low Mood, or Anxiety. J. Med. Internet Res. 2018, 20, e9275. [Google Scholar] [CrossRef]
- Donkin, L.; Christensen, H.; Naismith, S.L.; Neal, B.; Hickie, I.B.; Glozier, N. A Systematic Review of the Impact of Adherence on the Effectiveness of E-Therapies. J. Med. Internet Res. 2011, 13, e52. [Google Scholar] [CrossRef]
- Beintner, I.; Jacobi, C.; Schmidt, U.H. Participation and Outcome in Manualized Self-Help for Bulimia Nervosa and Binge Eating Disorder—A Systematic Review and Metaregression Analysis. Clin. Psychol. Rev. 2014, 34, 158–176. [Google Scholar] [CrossRef]
- Fernandez, E.; Salem, D.; Swift, J.K.; Ramtahal, N. Meta-Analysis of Dropout from Cognitive Behavioral Therapy: Magnitude, Timing, and Moderators. J. Consult. Clin. Psychol. 2015, 83, 1108–1122. [Google Scholar] [CrossRef]
- Linardon, J.; Hindle, A.; Brennan, L. Dropout from Cognitive-Behavioral Therapy for Eating Disorders: A Meta-Analysis of Randomized, Controlled Trials. Int. J. Eat. Disord. 2018, 51, 381–391. [Google Scholar] [CrossRef]
- ter Huurne, E.D.; de Haan, H.A.; Postel, M.G.; van der Palen, J.; VanDerNagel, J.E.L.; DeJong, C.A.J. Web-Based Cognitive Behavioral Therapy for Female Patients With Eating Disorders: Randomized Controlled Trial. J. Med. Internet Res. 2015, 17, e152. [Google Scholar] [CrossRef] [PubMed]
- Richards, D.; Richardson, T. Computer-Based Psychological Treatments for Depression: A Systematic Review and Meta-Analysis. Clin. Psychol. Rev. 2012, 32, 329–342. [Google Scholar] [CrossRef]
- Rogers, E.M. Diffusion of Innovations; The Free Press: New York, NY, USA, 2010. [Google Scholar]
- Barak, A.; Klein, B.; Proudfoot, J.G. Defining Internet-Supported Therapeutic Interventions. Ann. Behav. Med. 2009, 38, 4–17. [Google Scholar] [CrossRef]
- Kelders, S.M.; Kok, R.N.; Ossebaard, H.C.; Gemert-Pijnen, J.E.V. Persuasive System Design Does Matter: A Systematic Review of Adherence to Web-Based Interventions. J. Med. Internet Res. 2012, 14, e2104. [Google Scholar] [CrossRef]
| DSM-5-TR Diagnostic Criteria for Bulimia Nervosa |
|---|
|
| Phase | Content |
|---|---|
| One |
|
| Two |
|
| Three |
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Barakat, S.; Maguire, S. Accessibility of Psychological Treatments for Bulimia Nervosa: A Review of Efficacy and Engagement in Online Self-Help Treatments. Int. J. Environ. Res. Public Health 2023, 20, 119. https://doi.org/10.3390/ijerph20010119
Barakat S, Maguire S. Accessibility of Psychological Treatments for Bulimia Nervosa: A Review of Efficacy and Engagement in Online Self-Help Treatments. International Journal of Environmental Research and Public Health. 2023; 20(1):119. https://doi.org/10.3390/ijerph20010119
Chicago/Turabian StyleBarakat, Sarah, and Sarah Maguire. 2023. "Accessibility of Psychological Treatments for Bulimia Nervosa: A Review of Efficacy and Engagement in Online Self-Help Treatments" International Journal of Environmental Research and Public Health 20, no. 1: 119. https://doi.org/10.3390/ijerph20010119
APA StyleBarakat, S., & Maguire, S. (2023). Accessibility of Psychological Treatments for Bulimia Nervosa: A Review of Efficacy and Engagement in Online Self-Help Treatments. International Journal of Environmental Research and Public Health, 20(1), 119. https://doi.org/10.3390/ijerph20010119





