Who Benefits from Fermented Food Consumption? A Comparative Analysis between Psychiatrically Ill and Psychiatrically Healthy Medical Students
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Design and Ethical Considerations
2.2. Study Participants
2.3. Study Procedure and Research Instruments
2.4. Data Analysis
3. Results
3.1. Basal Charactersitics of the Study Participants
3.2. Pre-Exam Characteristics of Psychiatrically Ill Medical Students
3.3. Consumption of Fermented Food in Relation to Depressive and Anxiety Symptoms
3.4. Sensitivity and Ancillary Analyses
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Thursby, E.; Juge, N. Introduction to the Human Gut Microbiota. Biochem. J. 2017, 474, 1823–1836. [Google Scholar] [CrossRef] [PubMed]
- Mohajeri, M.H.; La Fata, G.; Steinert, R.E.; Weber, P. Relationship between the Gut Microbiome and Brain Function. Nutr. Rev. 2018, 76, 481–496. [Google Scholar] [CrossRef]
- Sarkar, A.; Harty, S.; Johnson, K.V.-A.; Moeller, A.H.; Carmody, R.N.; Lehto, S.M.; Erdman, S.E.; Dunbar, R.I.M.; Burnet, P.W.J. The Role of the Microbiome in the Neurobiology of Social Behaviour. Biol. Rev. 2020, 95, 1131–1166. [Google Scholar] [CrossRef] [PubMed]
- Silva, Y.P.; Bernardi, A.; Frozza, R.L. The Role of Short-Chain Fatty Acids from Gut Microbiota in Gut-Brain Communication. Front. Endocrinol. 2020, 11, 25. [Google Scholar] [CrossRef] [Green Version]
- Gao, K.; Mu, C.; Farzi, A.; Zhu, W. Tryptophan Metabolism: A Link Between the Gut Microbiota and Brain. Adv. Nutr. 2020, 11, 709–723. [Google Scholar] [CrossRef] [PubMed]
- Huang, R.; Ning, H.; Yang, L.; Jia, C.; Yang, F.; Xu, G.; Tan, H. Efficacy of Probiotics on Anxiety: A Meta-Analysis of Randomized Controlled Trials. Neuropsychiatry 2017, 7, 862–871. [Google Scholar] [CrossRef]
- Reis, D.J.; Ilardi, S.S.; Punt, S.E.W. The Anxiolytic Effect of Probiotics: A Systematic Review and Meta-Analysis of the Clinical and Preclinical Literature. PLoS ONE 2018, 13, e0199041. [Google Scholar] [CrossRef] [Green Version]
- Ng, Q.X.; Peters, C.; Ho, C.Y.X.; Lim, D.Y.; Yeo, W.-S. A Meta-Analysis of the Use of Probiotics to Alleviate Depressive Symptoms. J. Affect. Disord. 2018, 228, 13–19. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.T.; Walsh, R.F.L.; Sheehan, A.E. Prebiotics and Probiotics for Depression and Anxiety: A Systematic Review and Meta-Analysis of Controlled Clinical Trials. Neurosci. Biobehav. Rev. 2019, 102, 13–23. [Google Scholar] [CrossRef] [PubMed]
- Goh, K.K.; Liu, Y.-W.; Kuo, P.-H.; Chung, Y.-C.E.; Lu, M.-L.; Chen, C.-H. Effect of Probiotics on Depressive Symptoms: A Meta-Analysis of Human Studies. Psychiatry Res. 2019, 282, 112568. [Google Scholar] [CrossRef] [PubMed]
- Chao, L.; Liu, C.; Sutthawongwadee, S.; Li, Y.; Lv, W.; Chen, W.; Yu, L.; Zhou, J.; Guo, A.; Li, Z.; et al. Effects of Probiotics on Depressive or Anxiety Variables in Healthy Participants Under Stress Conditions or With a Depressive or Anxiety Diagnosis: A Meta-Analysis of Randomized Controlled Trials. Front. Neurol. 2020, 11, 421. [Google Scholar] [CrossRef]
- Cohen Kadosh, K.; Basso, M.; Knytl, P.; Johnstone, N.; Lau, J.; Gibson, G. Psychobiotic Interventions for Anxiety in Young People: A Systematic Review and Meta-Analysis, with Youth Consultation. Transl. Psychiatry 2021, 11, 352. [Google Scholar] [CrossRef]
- El Dib, R.; Periyasamy, A.G.; de Barros, J.L.; França, C.G.; Senefonte, F.L.; Vesentini, G.; Alves, M.G.O.; da Silva Rodrigues, J.V.; Gomaa, H.; Gomes Júnior, J.R.; et al. Probiotics for the Treatment of Depression and Anxiety: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Clin. Nutr. ESPEN 2021, 45, 75–90. [Google Scholar] [CrossRef] [PubMed]
- Hilimire, M.R.; DeVylder, J.E.; Forestell, C.A. Fermented Foods, Neuroticism, and Social Anxiety: An Interaction Model. Psychiatry Res. 2015, 228, 203–208. [Google Scholar] [CrossRef] [PubMed]
- Kim, C.-S.; Shin, D.-M. Probiotic Food Consumption Is Associated with Lower Severity and Prevalence of Depression: A Nationwide Cross-Sectional Study. Nutrition 2019, 63, 169–174. [Google Scholar] [CrossRef]
- Hockey, M.; McGuinness, A.J.; Marx, W.; Rocks, T.; Jacka, F.N.; Ruusunen, A. Is Dairy Consumption Associated with Depressive Symptoms or Disorders in Adults? A Systematic Review of Observational Studies. Crit. Rev. Food Sci. Nutr. 2020, 60, 3653–3668. [Google Scholar] [CrossRef]
- Karbownik, M.S.; Mokros, Ł.; Dobielska, M.; Kowalczyk, M.; Kowalczyk, E. Association between Consumption of Fermented Food and Food-Derived Prebiotics with Cognitive Performance, Depressive and Anxiety Symptoms in Psychiatrically Healthy Medical Students under Psychological Stress: A Prospective Cohort Study. Front. Nutr. 2022, 9, 850249. [Google Scholar] [CrossRef] [PubMed]
- Karbownik, M.S. Study Protocol. Association between Consumption of Fermented and Prebiotic-Containing Food and Cognitive Performance under Stress in Healthy Volunteers. OSF 4 June 2020. Available online: https://osf.io/ny2vf/ (accessed on 1 February 2022).
- Gerlitz, J.-Y.; Schupp, J. Zur Erhebung Der Big-Five-Basierten Persönlichkeitsmerkmale Im SOEP. Dokumentation der Instrumententwicklung BFI-S Auf Basis Des SOEP-Pretests 2005; Research Notes; Deutsches Institut für Wirtschaftsforschung: Berlin/Heidelberg, Germany, 2005. [Google Scholar]
- Strus, W.; Cieciuch, J. Higher-Order Factors of the Big Six—Similarities between Big Twos Identified above the Big Five and the Big Six. Personal. Individ. Differ. 2021, 171, 110544. [Google Scholar] [CrossRef]
- Paxton, A.E.; Strycker, L.A.; Toobert, D.J.; Ammerman, A.S.; Glasgow, R.E. Starting the Conversation Performance of a Brief Dietary Assessment and Intervention Tool for Health Professionals. Am. J. Prev. Med. 2011, 40, 67–71. [Google Scholar] [CrossRef] [PubMed]
- Karbownik, M.S.; Horne, R.; Paul, E.; Kowalczyk, E.; Szemraj, J. Determinants of Knowledge About Dietary Supplements Among Polish Internet Users: Nationwide Cross-Sectional Study. J. Med. Internet. Res. 2021, 23, e25228. [Google Scholar] [CrossRef] [PubMed]
- Craig, C.L.; Marshall, A.L.; Sjöström, M.; Bauman, A.E.; Booth, M.L.; Ainsworth, B.E.; Pratt, M.; Ekelund, U.; Yngve, A.; Sallis, J.F.; et al. International Physical Activity Questionnaire: 12-Country Reliability and Validity. Med. Sci. Sports Exerc. 2003, 35, 1381–1395. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Biernat, E.; Stupnicki, R.; Gajewski, A.K. International Physical Activity Questionnaire (IPAQ)—Polish version. Phys. Educ. Sport 2007, 51, 47–54. [Google Scholar]
- Kroenke, K.; Spitzer, R.L.; Williams, J.B. The PHQ-9: Validity of a Brief Depression Severity Measure. J. Gen. Intern. Med. 2001, 16, 606–613. [Google Scholar] [CrossRef] [PubMed]
- Ślusarska, B.J.; Nowicki, G.; Piasecka, H.; Zarzycka, D.; Mazur, A.; Saran, T.; Bednarek, A. Validation of the Polish Language Version of the Patient Health Questionnaire-9 in a Population of Adults Aged 35–64. Ann. Agric. Environ. Med. AAEM 2019, 26, 420–424. [Google Scholar] [CrossRef] [PubMed]
- Spitzer, R.L.; Kroenke, K.; Williams, J.B.W.; Löwe, B. A Brief Measure for Assessing Generalized Anxiety Disorder: The GAD-7. Arch. Intern. Med. 2006, 166, 1092–1097. [Google Scholar] [CrossRef] [Green Version]
- Babicki, M.; Mastalerz-Migas, A. The Occurrence of Anxiety Disorders among Poles during the COVID-19 Pandemic. Psychiatr. Pol. 2021, 55, 497–509. [Google Scholar] [CrossRef]
- Feng, Y.; Huang, W.; Tian, T.-F.; Wang, G.; Hu, C.; Chiu, H.F.K.; Ungvari, G.S.; Kilbourne, A.M.; Xiang, Y.-T. The Psychometric Properties of the Quick Inventory of Depressive Symptomatology-Self-Report (QIDS-SR) and the Patient Health Questionnaire-9 (PHQ-9) in Depressed Inpatients in China. Psychiatry Res. 2016, 243, 92–96. [Google Scholar] [CrossRef] [PubMed]
- Nowlin, R.B.; Brown, S.K. Monitoring Patient Treatment Outcomes in Inpatient Psychiatric Care with the PHQ-9. Available online: https://www.psychiatrictimes.com/view/monitoring-patient-treatment-outcomes-in-inpatient-psychiatric-care-with-the-phq-9 (accessed on 14 March 2022).
- Surono, I.S.; Hosono, A. Fermented Milks. Types and Standards of Identity. In Encyclopedia of Dairy Sciences; Academic Press: Cambridge, MA, USA, 2011; pp. 470–476. [Google Scholar] [CrossRef]
- Rezac, S.; Kok, C.R.; Heermann, M.; Hutkins, R. Fermented Foods as a Dietary Source of Live Organisms. Front. Microbiol. 2018, 9, 1785. [Google Scholar] [CrossRef]
- Liu, T.; Zhong, S.; Liao, X.; Chen, J.; He, T.; Lai, S.; Jia, Y. A Meta-Analysis of Oxidative Stress Markers in Depression. PLoS ONE 2015, 10, e0138904. [Google Scholar] [CrossRef]
- De Munter, J.; Pavlov, D.; Gorlova, A.; Sicker, M.; Proshin, A.; Kalueff, A.V.; Svistunov, A.; Kiselev, D.; Nedorubov, A.; Morozov, S.; et al. Increased Oxidative Stress in the Prefrontal Cortex as a Shared Feature of Depressive- and PTSD-Like Syndromes: Effects of a Standardized Herbal Antioxidant. Front. Nutr. 2021, 8, 122. [Google Scholar] [CrossRef]
- Lee, C.-H.; Giuliani, F. The Role of Inflammation in Depression and Fatigue. Front. Immunol. 2019, 10, 1696. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Miller, A.H.; Raison, C.L. The Role of Inflammation in Depression: From Evolutionary Imperative to Modern Treatment Target. Nat. Rev. Immunol. 2016, 16, 22–34. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cowen, P.J.; Browning, M. What Has Serotonin to Do with Depression? World Psychiatry 2015, 14, 158–160. [Google Scholar] [CrossRef] [Green Version]
- Pariante, C.M.; Lightman, S.L. The HPA Axis in Major Depression: Classical Theories and New Developments. Trends Neurosci. 2008, 31, 464–468. [Google Scholar] [CrossRef]
- Menke, A. Is the HPA Axis as Target for Depression Outdated, or Is There a New Hope? Front. Psychiatry 2019, 10, 101. [Google Scholar] [CrossRef]
- Ansorge, M.S.; Hen, R.; Gingrich, J.A. Neurodevelopmental Origins of Depressive Disorders. Curr. Opin. Pharmacol. 2007, 7, 8–17. [Google Scholar] [CrossRef]
- Chakrapani, S.; Eskander, N.; De Los Santos, L.A.; Omisore, B.A.; Mostafa, J.A. Neuroplasticity and the Biological Role of Brain Derived Neurotrophic Factor in the Pathophysiology and Management of Depression. Cureus 2020, 12, e11396. [Google Scholar] [CrossRef]
- Dimidi, E.; Cox, S.R.; Rossi, M.; Whelan, K. Fermented Foods: Definitions and Characteristics, Impact on the Gut Microbiota and Effects on Gastrointestinal Health and Disease. Nutrients 2019, 11, 1806. [Google Scholar] [CrossRef] [Green Version]
- Shandilya, S.; Kumar, S.; Kumar Jha, N.; Kumar Kesari, K.; Ruokolainen, J. Interplay of Gut Microbiota and Oxidative Stress: Perspective on Neurodegeneration and Neuroprotection. J. Adv. Res. 2021; in press. [Google Scholar] [CrossRef]
- Park, C.; Brietzke, E.; Rosenblat, J.D.; Musial, N.; Zuckerman, H.; Ragguett, R.-M.; Pan, Z.; Rong, C.; Fus, D.; McIntyre, R.S. Probiotics for the Treatment of Depressive Symptoms: An Anti-Inflammatory Mechanism? Brain Behav. Immun. 2018, 73, 115–124. [Google Scholar] [CrossRef]
- Wallace, C.J.K.; Milev, R. The Effects of Probiotics on Depressive Symptoms in Humans: A Systematic Review. Ann. Gen. Psychiatry 2017, 16, 14. [Google Scholar] [CrossRef] [Green Version]
- Ranuh, R.; Athiyyah, A.F.; Darma, A.; Risky, V.P.; Riawan, W.; Surono, I.S.; Sudarmo, S.M. Effect of the Probiotic Lactobacillus Plantarum IS-10506 on BDNF and 5HT Stimulation: Role of Intestinal Microbiota on the Gut-Brain Axis. Iran. J. Microbiol. 2019, 11, 145–150. [Google Scholar] [PubMed]
- Yong, S.J.; Tong, T.; Chew, J.; Lim, W.L. Antidepressive Mechanisms of Probiotics and Their Therapeutic Potential. Front. Neurosci. 2020, 13, 1361. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Witkamp, R.F.; van Norren, K. Let Thy Food Be Thy Medicine….When Possible. Eur. J. Pharmacol. 2018, 836, 102–114. [Google Scholar] [CrossRef] [PubMed]
- Scherr, M.; Kunz, A.; Doll, A.; Mutzenbach, J.S.; Broussalis, E.; Bergmann, H.J.; Kirschner, M.; Trinka, E.; Killer-Oberpfalzer, M. Ignoring Floor and Ceiling Effects May Underestimate the Effect of Carotid Artery Stenting on Cognitive Performance. J. Neurointerventional Surg. 2016, 8, 747–751. [Google Scholar] [CrossRef]
- Moriarity, D.P.; Ellman, L.M.; Coe, C.L.; Olino, T.M.; Alloy, L.B. A Physiometric Investigation of Inflammatory Composites: Comparison of “a Priori” Aggregates, Empirically-Identified Factors, and Individual Proteins. Brain Behav. Immun. Health 2021, 18, 100391. [Google Scholar] [CrossRef] [PubMed]
- Schlager, D.; Schwartz, J.E.; Bromet, E.J. Seasonal Variations of Current Symptoms in a Healthy Population. Br. J. Psychiatry 1993, 163, 322–326. [Google Scholar] [CrossRef] [PubMed]
- Ni, M.Y.; Kim, Y.; McDowell, I.; Wong, S.; Qiu, H.; Wong, I.O.; Galea, S.; Leung, G.M. Mental Health during and after Protests, Riots and Revolutions: A Systematic Review. Aust. N. Z. J. Psychiatry 2020, 54, 232–243. [Google Scholar] [CrossRef] [Green Version]
- Bashir, M.B.A.; Albadawy, I.M.A.H.; Cumber, S.N. Predictors and Correlates of Examination Anxiety and Depression among High School Students Taking the Sudanese National Board Examination in Khartoum State, Sudan: A Cross-Sectional Study. Pan Afr. Med. J. 2019, 33, 69. [Google Scholar] [CrossRef] [PubMed]
- Davies, M.R.; Buckman, J.E.J.; Adey, B.N.; Armour, C.; Bradley, J.R.; Curzons, S.C.B.; Davies, H.L.; Davis, K.A.S.; Goldsmith, K.A.; Hirsch, C.R.; et al. Comparison of Symptom-Based versus Self-Reported Diagnostic Measures of Anxiety and Depression Disorders in the GLAD and COPING Cohorts. J. Anxiety Disord. 2022, 85, 102491. [Google Scholar] [CrossRef]
- Leung, K.; Xu, E.; Rosic, T.; Worster, A.; Thabane, L.; Samaan, Z. Sensitivity and Specificity of Self-Reported Psychiatric Diagnoses amongst Patients Treated for Opioid Use Disorder. BMC Psychiatry 2021, 21, 520. [Google Scholar] [CrossRef]
- Jeong, H.; Yim, H.W.; Lee, S.-Y.; Lee, H.K.; Potenza, M.N.; Kwon, J.-H.; Koo, H.J.; Kweon, Y.-S.; Bhang, S.; Choi, J.-S. Discordance between Self-Report and Clinical Diagnosis of Internet Gaming Disorder in Adolescents. Sci. Rep. 2018, 8, 10084. [Google Scholar] [CrossRef]
- Suwalska, J.; Suwalska, A.; Szczygieł, M.; Łojko, D. Medical Students and Stigma of Depression. Part 2. Self-Stigma. Psychiatr. Pol. 2017, 51, 503–513. [Google Scholar] [CrossRef] [PubMed]
- Kiejna, A.; Piotrowski, P.; Adamowski, T.; Moskalewicz, J.; Wciórka, J.; Stokwiszewski, J.; Rabczenko, D.; Kessler, R. [The prevalence of common mental disorders in the population of adult Poles by sex and age structure—an EZOP Poland study]. Psychiatr. Pol. 2015, 49, 15–27. [Google Scholar] [CrossRef]
- Forycka, J.; Pawłowicz-Szlarska, E.; Burczyńska, A.; Cegielska, N.; Harendarz, K.; Nowicki, M. Polish Medical Students Facing the Pandemic—Assessment of Resilience, Well-Being and Burnout in the COVID-19 Era. PLoS ONE 2022, 17, e0261652. [Google Scholar] [CrossRef]
| Characteristics | Psychiatrically Ill (n = 25) a | Psychiatrically Healthy (n = 372) a | p-Value for Difference |
|---|---|---|---|
| Mean (Standard Deviation), Median (1st–3rd Quartiles) or Absolute Number (Frequency) | |||
| Faculty | |||
| Faculty of Medicine | 19 (76%) | 250 (67.2%) | 0.36 b |
| Faculty of Military Medicine | 6 (24%) | 122 (32.8%) | |
| Age | |||
| (years) | 22.5 (0.7) | 22.7 (1.1) | 0.79 c |
| Sex | |||
| Female | 19 (76%) | 245 (65.9%) | 0.30 b |
| Male | 6 (24%) | 127 (34.1%) | |
| Socioeconomic status | |||
| Low | 0 (0%) | 2 (0.5%) | 0.99 c |
| Middle | 18 (72%) | 264 (71.0%) | |
| High | 7 (28%) | 106 (28.5%) | |
| Number of inhabitants in a place of family residence | |||
| Below 5000 | 4 (16%) | 88 (23.6%) | 0.56 c |
| 5000–50,000 | 9 (36%) | 120 (32.3%) | |
| 50,000–500,000 | 7 (28%) | 91 (24.5%) | |
| Over 500,000 | 5 (20%) | 73 (19.6%) | |
| Anthropometry | |||
| Body-mass index (kg × m−2) | 22.3 (3.3) | 22.0 (3.1) | 0.80 c |
| Chronic diseases | |||
| Allergic | 6 (24%) | 89 (23.9%) | 0.99 b |
| Cardiological | 0 (0%) | 4 (1.1%) | 1.0 d |
| Endocrine/metabolic | 3 (12%) | 33 (8.9%) | 0.49 d |
| Gastroenterological | 4 (16%) | 21 (5.6%) | 0.063 d |
| Immune | 0 (0%) | 6 (1.6%) | 1.0 d |
| Infectious | 0 (0%) | 0 (0.0%) | N/A |
| Neoplastic | 0 (0%) | 1 (0.3%) | 1.0 d |
| Neurological | 1 (4%) | 4 (1.1%) | 0.28 d |
| Any chronic disease e | 12 (48%) | 128 (34.4%) | 0.17 b |
| Personality traits f | |||
| Neuroticism | 16 (12–18) | 12 (9–15) | 0.022 c |
| Extraversion | 12 (8–14) | 12 (9–15) | 0.95 c |
| Openness | 13 (9–20) | 15 (12–17) | 0.86 c |
| Agreeableness | 14 (13–16) | 15 (12–17) | 0.37 c |
| Conscientiousness | 15 (12–19) | 16 (14–18) | 0.71 c |
| Health-related behaviors | |||
| Current cigarette smoking/use g | 3 (12%) | 25 (6.7%) | 0.41 d |
| Physical activity h | 3 (2–4) | 3 (2–4) | 0.36 c |
| General quality of diet i | 8 (6–8) | 6 (5–8) | 0.40 c |
| Mode of eating j | 6 (5–6) | 6 (5–6) | 0.77 c |
| Mental health | |||
| Depressive symptoms k | 13 (8–20) | 7 (5–11) | <0.0001 c |
| Anxiety symptoms l | 9 (6–15) | 6 (4–10) | 0.0027 c |
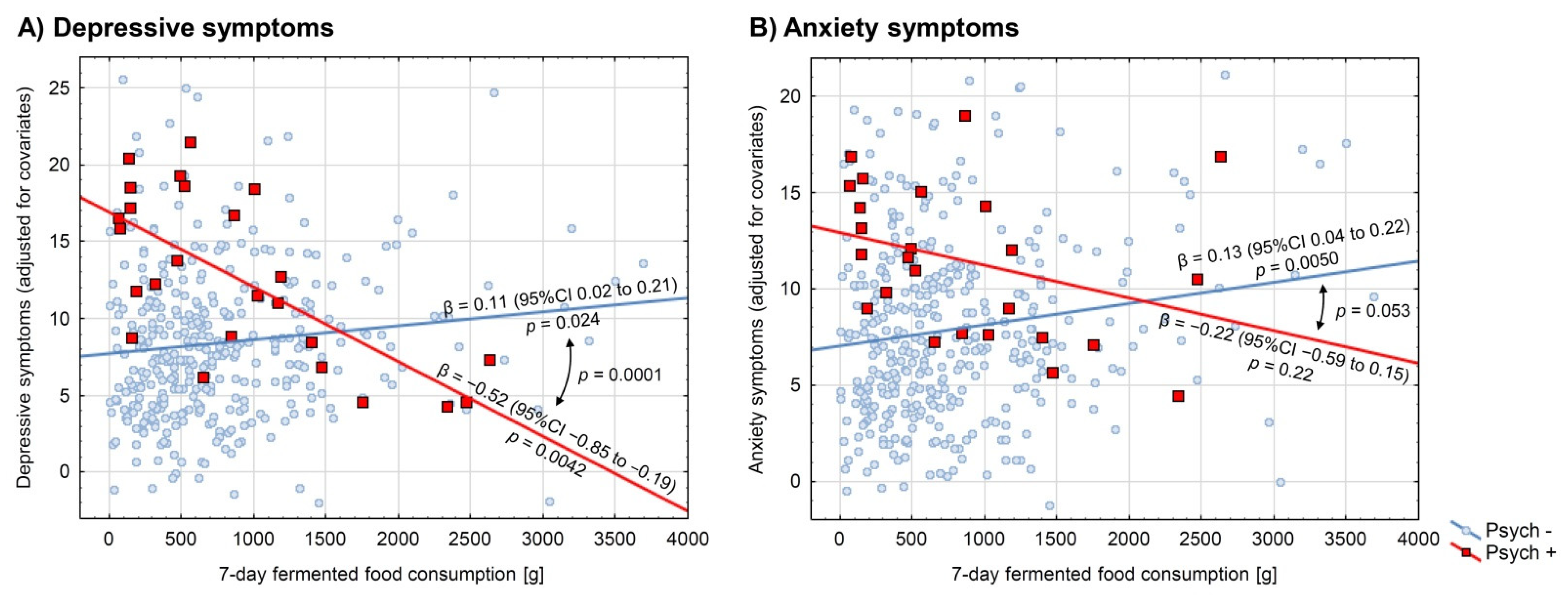
| Parameter | Slope Factor a | β Regression Coefficient | p-Value | ||
|---|---|---|---|---|---|
| Point Estimate | 95% CI | Point Estimate | 95% CI | ||
| Prediction of Depressive Symptoms (PHQ-9 score) | |||||
| Fermented food consumption | 0.9 | 0.1 to 1.7 | 0.11 | 0.01 to 0.20 | 0.027 |
| Psychiatric illness | 8.9 | 5.8 to 11.9 | 0.39 | 0.25 to 0.52 | <0.0001 |
| Fermented × Psychiatric | −5.5 | −8.2 to −2.8 | −0.28 | −0.42 to −0.15 | 0.0001 |
| Prediction of Anxiety Symptoms (GAD-7 score) | |||||
| Fermented food consumption | 1.0 | 0.3 to 1.8 | 0.12 | 0.03 to 0.21 | 0.0080 |
| Psychiatric illness | 4.8 | 1.8 to 7.7 | 0.21 | 0.08 to 0.34 | 0.0016 |
| Fermented × Psychiatric | −2.5 | −5.1 to 0.0 | −0.13 | −0.27 to 0.00 | 0.053 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Karbownik, M.S.; Mokros, Ł.; Kowalczyk, E. Who Benefits from Fermented Food Consumption? A Comparative Analysis between Psychiatrically Ill and Psychiatrically Healthy Medical Students. Int. J. Environ. Res. Public Health 2022, 19, 3861. https://doi.org/10.3390/ijerph19073861
Karbownik MS, Mokros Ł, Kowalczyk E. Who Benefits from Fermented Food Consumption? A Comparative Analysis between Psychiatrically Ill and Psychiatrically Healthy Medical Students. International Journal of Environmental Research and Public Health. 2022; 19(7):3861. https://doi.org/10.3390/ijerph19073861
Chicago/Turabian StyleKarbownik, Michał Seweryn, Łukasz Mokros, and Edward Kowalczyk. 2022. "Who Benefits from Fermented Food Consumption? A Comparative Analysis between Psychiatrically Ill and Psychiatrically Healthy Medical Students" International Journal of Environmental Research and Public Health 19, no. 7: 3861. https://doi.org/10.3390/ijerph19073861
APA StyleKarbownik, M. S., Mokros, Ł., & Kowalczyk, E. (2022). Who Benefits from Fermented Food Consumption? A Comparative Analysis between Psychiatrically Ill and Psychiatrically Healthy Medical Students. International Journal of Environmental Research and Public Health, 19(7), 3861. https://doi.org/10.3390/ijerph19073861








