The Mediating Effect of Age, Gender, and Post-Stroke Duration on the Association between Trunk and Upper Limb Recovery in Subacute Stroke Population: A Cross-Sectional Study with Mediation Analysis
Abstract
1. Introduction
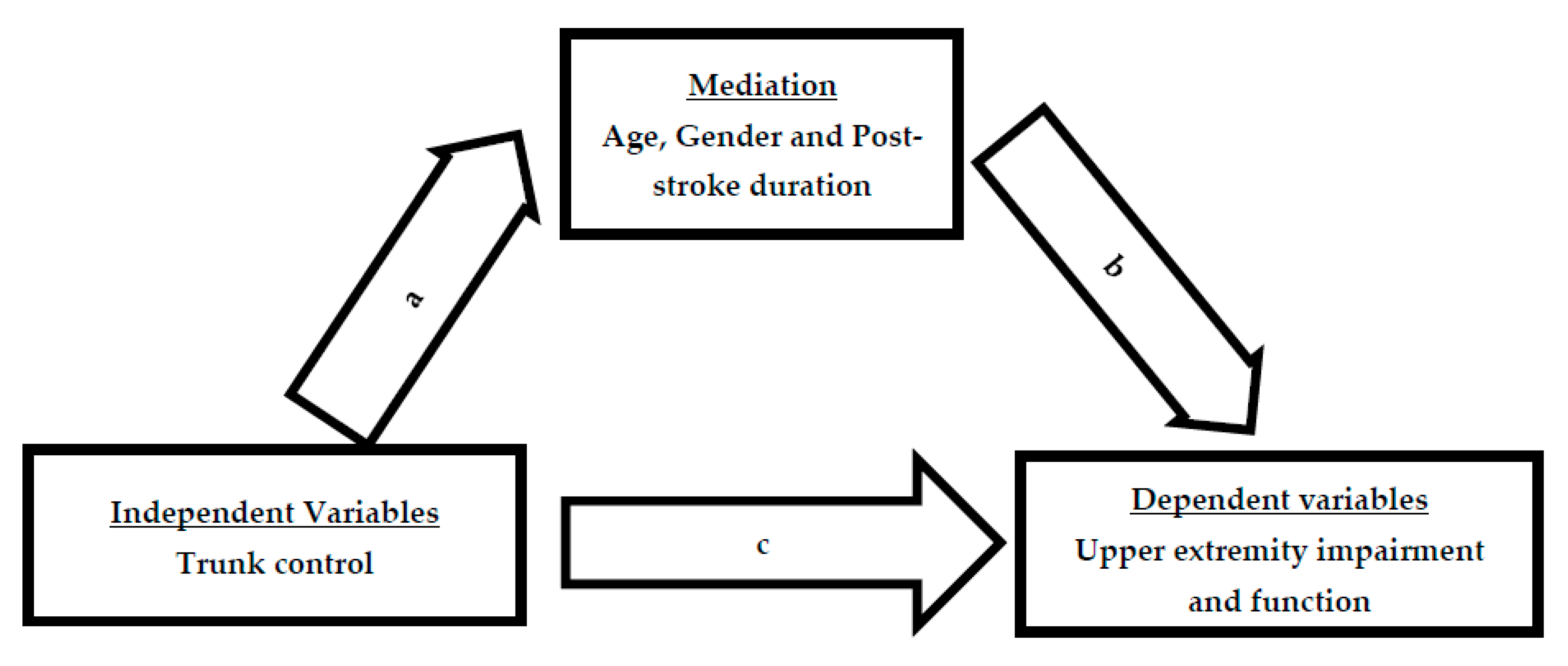
2. Materials and Methods
2.1. Study Design and Participants
2.2. Sample Size Estimation
2.3. Data Collection and Measurement Tools
2.3.1. Trunk Impairment Scale
2.3.2. The Fugl-Meyer Assessment of Upper Extremity
2.3.3. Wolf Motor Function Test
2.3.4. Procedure
2.4. Statistical Analysis
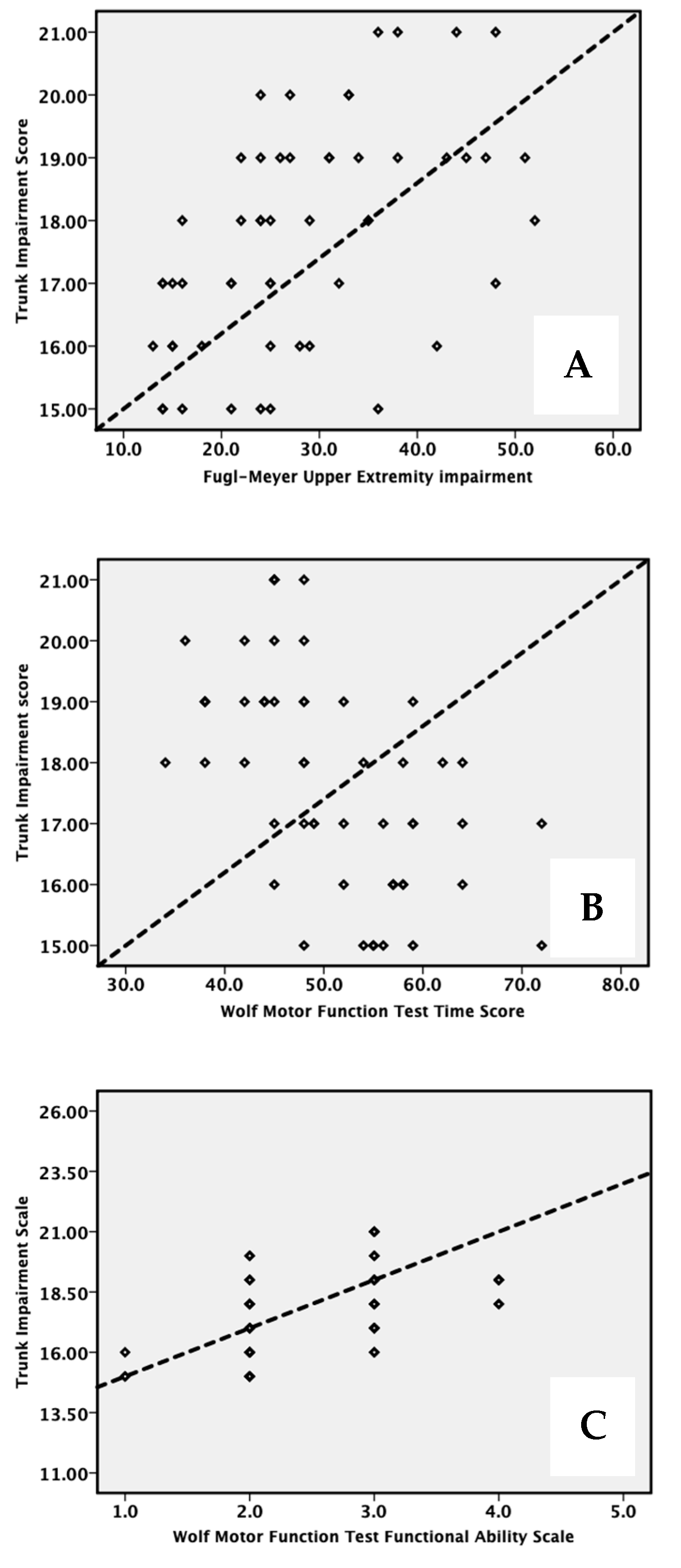
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Luft, A.; Katan, L. Global burden of stroke. Semin. Neurol. 2018, 38, 208–211. [Google Scholar]
- Feigin, V.L.; Norrving, B.; Mensah, G.A. Global burden of stroke. Circ. Res. 2017, 120, 439–448. [Google Scholar] [CrossRef] [PubMed]
- Sugiyama, M.; Ueno, Y.; Kamo, H.; Edahiro, Y.; Miyamoto, N.; Yamashiro, K.; Tanaka, R.; Shimo, Y.; Komatsu, N.; Hattori, N. Specific mechanisms of subarachnoid hemorrhage accompanied by ischemic stroke in essential thrombocythemia: Two case reports and a literature review. J. Neurol. 2019, 266, 1869–1878. [Google Scholar] [CrossRef] [PubMed]
- Guilloteau, A.; Binquet, C.; Bourredjem, A.; Fournel, I.; Lalanne-Mistrih, M.L.; Nacher, M.; Rochemont, D.; Cabie, A.; Mimeau, E.; Mislin-Tritsch, C.; et al. Social deprivation among socio-economic contrasted French areas: Using item response theory analysis to assess differential item functioning of the EPICES questionnaire in stroke patients. PLoS ONE 2020, 15, e0230661. [Google Scholar] [CrossRef]
- Opoku, S.; Gan, Y.; Fu, W.; Chen, D.; Addo-Yobo, E.; Trofimovitch, D.; Yue, W.; Yan, F.; Wang, Z.; Lu, Z. Prevalence and risk factors for dyslipidemia among adults in rural and urban China: Findings from the China National Stroke Screening and prevention project (CNSSPP). BMC Public Health 2019, 19, 1500. [Google Scholar] [CrossRef]
- Caprio, F.Z.; Sorond, F.A. Cerebrovascular disease: Primary and secondary stroke prevention. Med. Clin. 2019, 103, 295–308. [Google Scholar]
- Chinnavan, E.; Ragupathy, R.; Wah, Y.C. Effectiveness of mirror therapy on upper limb motor functions among hemiplegic patients. Bangladesh J. Med. Sci. 2020, 19, 208–213. [Google Scholar] [CrossRef]
- Yi, X.; Luo, H.; Zhou, J.; Yu, M.; Chen, X.; Tan, L.; Wei, W.; Li, J. Prevalence of stroke and stroke related risk factors: A population based cross sectional survey in southwestern China. BMC Neurol. 2020, 20, 5. [Google Scholar] [CrossRef]
- Higgins, J.; Mayo, N.E.; Desrosiers, J.; Salbach, N.M.; Ahmed, S. Upper-limb function and recovery in the acute phase poststroke. J. Rehabil. Res. Dev. 2005, 42, 65–76. [Google Scholar] [CrossRef]
- Houwink, A.; Nijland, R.H.; Geurts, A.C.; Kwakkel, G. Functional recovery of the paretic upper limb after stroke: Who regains hand capacity? Arch. Phys. Med. Rehabil. 2013, 94, 839–844. [Google Scholar] [CrossRef]
- Kantak, S.; Jax, S.; Wittenberg, G. Bimanual coordination: A missing piece of arm rehabilitation after stroke. Restor. Neurol. Neurosci. 2017, 35, 347–364. [Google Scholar] [CrossRef]
- Cauraugh, J.H.; Kang, N. Bimanual Movements and Chronic Stroke Rehabilitation: Looking Back and Looking Forward. Appl. Sci. 2021, 11, 10858. [Google Scholar] [CrossRef]
- Schambra, H.M.; Parnandi, A.; Pandit, N.G.; Uddin, J.; Wirtanen, A.; Nilsen, D.M. A taxonomy of functional upper extremity motion. Front. Neurol. 2019, 10, 857. [Google Scholar] [CrossRef]
- Messier, S.; Bourbonnais, D.; Desrosiers, J.; Roy, Y. Dynamic analysis of trunk flexion after stroke. Arch. Phys. Med. Rehabil. 2004, 85, 1619–1624. [Google Scholar] [CrossRef]
- Huang, H.; Lin, Y.; Chen, J.; Yeh, C.; Chung, K. The impact of abnormal muscle tone from hemiplegia on reclining wheelchair positioning: A sliding and pressure evaluation. Eur. J. Phys. Rehabil. Med. 2013, 49, 619–628. [Google Scholar] [PubMed]
- Tessem, S.; Hagstrøm, N.; Fallang, B. Weight distribution in standing and sitting positions, and weight transfer during reaching tasks, in seated stroke subjects and healthy subjects. Physiother. Res. Int. 2007, 12, 82–94. [Google Scholar] [CrossRef]
- Tasseel-Ponche, S.; Yelnik, A.; Bonan, I. Motor strategies of postural control after hemispheric stroke. Neurophysiol. Clin./Clin. Neurophysiol. 2015, 45, 327–333. [Google Scholar] [CrossRef] [PubMed]
- Schumacher, C.; Berry, A.; Lemus, D.; Rode, C.; Seyfarth, A.; Vallery, H. Biarticular muscles are most responsive to upper-body pitch perturbations in human standing. Sci. Rep. 2019, 9, 14492. [Google Scholar] [CrossRef]
- Hsu, W.-L.; Chen, C.P.; Nikkhoo, M.; Lin, C.-F.; Ching, C.T.-S.; Niu, C.-C.; Cheng, C.-H. Fatigue changes neck muscle control and deteriorates postural stability during arm movement perturbations in patients with chronic neck pain. Spine J. 2020, 20, 530–537. [Google Scholar] [CrossRef]
- Russo, M.; Ozeri-Engelhard, N.; Hupfeld, K.; Nettekoven, C.; Thibault, S.; Sedaghat-Nejad, E.; Buchwald, D.; Xing, D.; Zobeiri, O.; Kilteni, K.; et al. Highlights from the 30th Annual Meeting of the Society for the Neural Control of Movement. J. Neurophysiol. 2021, 126, 967–975. [Google Scholar] [CrossRef]
- Park, M.; Seok, H.; Kim, S.-H.; Noh, K.; Lee, S.Y. Comparison between neuromuscular electrical stimulation to abdominal and back muscles on postural balance in post-stroke hemiplegic patients. Ann. Rehabil. Med. 2018, 42, 652–659. [Google Scholar] [CrossRef] [PubMed]
- Malmut, L.; Lin, C.; Srdanovic, N.; Kocherginsky, M.; Harvey, R.L.; Prabhakaran, S. Arm subscore of motricity index to predict recovery of upper limb dexterity in patients with acute ischemic stroke. Am. J. Phys. Med. Rehabil. 2020, 99, 300–304. [Google Scholar] [CrossRef] [PubMed]
- Wu, D.; Ma, J.; Zhang, L.; Wang, S.; Tan, B.; Jia, G. Effect and safety of transcutaneous auricular vagus nerve stimulation on recovery of upper limb motor function in subacute ischemic stroke patients: A randomized pilot study. Neural Plast. 2020, 2020, 8841752. [Google Scholar] [CrossRef] [PubMed]
- Ballester, B.R.; Duff, A.; Maier, M.; Cameirao, M.; Bermudez, S.; Duarte, E.; Cuxart, A.; Rodríguez, S.; Verschure, P.F. Revealing an extended critical window of recovery post-stroke. bioRxiv 2018. [Google Scholar] [CrossRef]
- Meadmore, K.L.; Hallewell, E.; Freeman, C.; Hughes, A.-M. Factors affecting rehabilitation and use of upper limb after stroke: Views from healthcare professionals and stroke survivors. Top. Stroke Rehabil. 2019, 26, 94–100. [Google Scholar] [CrossRef]
- Hsieh, C.-L.; Sheu, C.-F.; Hsueh, I.-P.; Wang, C.-H. Trunk control as an early predictor of comprehensive activities of daily living function in stroke patients. Stroke 2002, 33, 2626–2630. [Google Scholar] [CrossRef]
- Franceschini, M.; Goffredo, M.; Pournajaf, S.; Paravati, S.; Agosti, M.; De Pisi, F.; Galafate, D.; Posteraro, F. Predictors of activities of daily living outcomes after upper limb robot-assisted therapy in subacute stroke patients. PLoS ONE 2018, 13, e0193235. [Google Scholar] [CrossRef]
- Verheyden, G.; Nieuwboer, A.; De Wit, L.; Feys, H.; Schuback, B.; Baert, I.; Jenni, W.; Schupp, W.; Thijs, V.; De Weerdt, W. Trunk performance after stroke: An eye catching predictor of functional outcome. J. Neurol. Neurosurg. Psychiatry 2007, 78, 694–698. [Google Scholar] [CrossRef]
- Iso, F.; Mitsunaga, W.; Yamaguchi, R.; Shimizu, N.; Ito, S.; Honda, Y.; Okubo, A.; Honda, S.; Iso, N.; Higashi, T.; et al. Relationship among trunk control, activities of daily living, and upper extremity function during the first week after stroke in patients with acute cerebral infarction. J. Phys. Ther. Sci. 2022, 34, 315–319. [Google Scholar] [CrossRef]
- Yang, S.-H.; Chung, E.-J.; Lee, J.; Lee, S.-H.; Lee, B.-H. The effect of trunk stability training based on visual feedback on trunk stability, balance, and upper limb function in stroke patients: A randomized control trial. Healthcare 2021, 9, 532. [Google Scholar] [CrossRef]
- Wee, S.; Hughes, A.; Warner, M.; Brown, S.; Cranny, A.; Mazomenos, E.; Burridge, J.; Yeo, S.D.; Kong, K.; Chan, K. Impact of trunk control on upper extremity function in subacute and chronic stroke patients and healthy controls. Physiotherapy 2015, 101, e1619. [Google Scholar] [CrossRef][Green Version]
- Lee, K.B.; Lim, S.H.; Kim, K.H.; Kim, K.J.; Kim, Y.R.; Chang, W.N.; Yeom, J.W.; Kim, Y.D.; Hwang, B.Y. Six-month functional recovery of stroke patients: A multi-time-point study. Int. J. Rehabil. Res. 2015, 38, 173. [Google Scholar] [CrossRef]
- Arslan, S.A.; Uğurlu, K.; DEMİRCİ, C.; KESKİN, D. Investigating the relation between upper extremity function and trunk control, balance and functional mobility in individuals with stroke. J. Health Sci. Med. 2021, 4, 127–131. [Google Scholar] [CrossRef]
- Roy-O’Reilly, M.; McCullough, L.D. Age and sex are critical factors in ischemic stroke pathology. Endocrinology 2018, 159, 3120–3131. [Google Scholar] [CrossRef] [PubMed]
- Yoo, J.W.; Hong, B.Y.; Jo, L.; Kim, J.-S.; Park, J.G.; Shin, B.K.; Lim, S.H. Effects of age on long-term functional recovery in patients with stroke. Medicina 2020, 56, 451. [Google Scholar] [CrossRef] [PubMed]
- Kugler, C.; Altenhöner, T.; Lochner, P.; Ferbert, A. Does age influence early recovery from ischemic stroke? J. Neurol. 2003, 250, 676–681. [Google Scholar] [CrossRef]
- Branyan, T.E.; Sohrabji, F. Sex differences in stroke co-morbidities. Exp. Neurol. 2020, 332, 113384. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.-S.; Lee, K.-B.; Roh, H.; Ahn, M.-Y.; Hwang, H.-W. Gender differences in the functional recovery after acute stroke. J. Clin. Neurol. 2010, 6, 183–188. [Google Scholar] [CrossRef]
- Verheyden, G.; Nieuwboer, A.; Mertin, J.; Preger, R.; Kiekens, C.; De Weerdt, W. The Trunk Impairment Scale: A new tool to measure motor impairment of the trunk after stroke. Clin. Rehabil. 2004, 18, 326–334. [Google Scholar] [CrossRef]
- Sag, S.; Buyukavci, R.; Sahin, F.; Sag, M.S.; Dogu, B.; Kuran, B. Assessing the validity and reliability of the Turkish version of the Trunk Impairment Scale in stroke patients. North. Clin. İstanbul 2019, 6, 156. [Google Scholar]
- Hernandez, E.D.; Galeano, C.P.; Barbosa, N.E.; Forero, S.M.; Nordin, Å.; Sunnerhagen, K.S.; Alt Murphy, M. Intra-and inter-rater reliability of Fugl-Meyer assessment of upper extremity in stroke. J. Rehabil. Med. 2019, 51, 652–659. [Google Scholar] [CrossRef]
- Lundquist, C.B.; Maribo, T. The Fugl–Meyer assessment of the upper extremity: Reliability, responsiveness and validity of the Danish version. Disabil. Rehabil. 2017, 39, 934–939. [Google Scholar] [CrossRef] [PubMed]
- Morris, D.M.; Uswatte, G.; Crago, J.E.; Cook III, E.W.; Taub, E. The reliability of the wolf motor function test for assessing upper extremity function after stroke. Arch. Phys. Med. Rehabil. 2001, 82, 750–755. [Google Scholar] [CrossRef] [PubMed]
- Fugl-Meyer, A.R.; Jääskö, L.; Leyman, I.; Olsson, S.; Steglind, S. A method for evaluation of physical performance. Scand J. Rehabil. Med. 1975, 7, 13–31. [Google Scholar] [PubMed]
- Taub, E.; Morris, D.M.; Crago, J.; King, D.K.; Bowman, M.; Bryson, C.; Bishop, S.; Pearson, S.; Shaw, S.E. Wolf Motor Function Test (WMFT) Manual; CI Therapy Research Group, University of Alabama: Birmingham, UK, 2011; pp. 1–31. [Google Scholar]
- Dickstein, R.; Shefi, S.; Marcovitz, E.; Villa, Y. Anticipatory postural adjustment in selected trunk muscles in poststroke hemiparetic patients. Arch. Phys. Med. Rehabil. 2004, 85, 261–267. [Google Scholar] [CrossRef] [PubMed]
- Callegari, B.; Saunier, G.; Duarte, M.B.; da Silva Almeida, G.C.; Amorim, C.F.; Mourey, F.; Pozzo, T.; da Silva Souza, G. Anticipatory Postural Adjustments and kinematic arm features when postural stability is manipulated. PeerJ 2018, 6, e4309. [Google Scholar] [CrossRef] [PubMed]
- Shaikh, T.; Goussev, V.; Feldman, A.G.; Levin, M.F. Arm–trunk coordination for beyond-the-reach movements in adults with stroke. Neurorehabilit. Neural Repair 2014, 28, 355–366. [Google Scholar] [CrossRef]
- Kapreli, E.; Athanasopoulos, S.; Stavridis, I.; Billis, E.; Strimpakos, N. Waterloo Footedness Questionnaire (WFQ-R): Cross-cultural adaptation and psychometric properties of Greek version. Physiotherapy 2015, 101, e721. [Google Scholar] [CrossRef][Green Version]
- Nam, H.-S.; Kim, J.-H.; Lim, Y.-J. The effect of the base of support on anticipatory postural adjustment and postural stability. J. Korean Phys. Ther. 2017, 29, 135–141. [Google Scholar] [CrossRef][Green Version]
- Roh, J.; Rymer, W.Z.; Perreault, E.J.; Yoo, S.B.; Beer, R.F. Alterations in upper limb muscle synergy structure in chronic stroke survivors. J. Neurophysiol. 2013, 109, 768–781. [Google Scholar] [CrossRef]
- Yoo, J.; Jeong, J.; Lee, W. The effect of trunk stabilization exercise using an unstable surface on the abdominal muscle structure and balance of stroke patients. J. Phys. Ther. Sci. 2014, 26, 857–859. [Google Scholar] [CrossRef]
- Rand, D.; Eng, J.J. Disparity between functional recovery and daily use of the upper and lower extremities during subacute stroke rehabilitation. Neurorehabilit. Neural Repair 2012, 26, 76–84. [Google Scholar] [CrossRef] [PubMed]
- Raghavan, P. Upper limb motor impairment after stroke. Phys. Med. Rehabil. Clin. 2015, 26, 599–610. [Google Scholar] [CrossRef] [PubMed]
- Hussain, N.; Alt Murphy, M.; Sunnerhagen, K.S. Upper limb kinematics in stroke and healthy controls using target-to-target task in virtual reality. Front. Neurol. 2018, 9, 300. [Google Scholar] [CrossRef] [PubMed]
- Tedla, J.S.; Gular, K.; Reddy, R.S.; de Sá Ferreira, A.; Rodrigues, E.C.; Kakaraparthi, V.N.; Gyer, G.; Sangadala, D.R.; Qasheesh, M.; Kovela, R.K. Effectiveness of Constraint-Induced Movement Therapy (CIMT) on Balance and Functional Mobility in the Stroke Population: A Systematic Review and Meta-Analysis. Healthcare 2022, 10, 495. [Google Scholar] [CrossRef]
- Reddy, R.S.; Gular, K.; Dixit, S.; Kandakurti, P.K.; Tedla, J.S.; Gautam, A.P.; Sangadala, D.R. Impact of Constraint-Induced Movement Therapy (CIMT) on Functional Ambulation in Stroke Patients—A Systematic Review and Meta-Analysis. Int. J. Environ. Res. Public Health 2022, 19, 12809. [Google Scholar] [CrossRef] [PubMed]
- Kimura, Y.; Ohji, S.; Ishiyama, D.; Nishio, N.; Otobe, Y.; Suzuki, M.; Ogawa, H.; Ichikawa, T.; Taguchi, R.; Shino, S.; et al. Factors associated with functional recovery in Japanese patients with convalescent stroke stratified by age: A multicenter observational study. Int. J. Rehabil. Res. 2019, 42, 249–255. [Google Scholar] [CrossRef] [PubMed]
| Variables | Values |
|---|---|
| Age in years (Mean ± SD) | 58.37 ± 6.11 |
| Gender (Male/Female) | 32/25 |
| Height in meters (Mean ± SD) | 1.66 ± 0.10 |
| Weight in Kilograms (Mean ± SD) | 72.05 ± 12.90 |
| BMI (Mean ± SD) | 26.42 ± 6.21 |
| Type of stroke (Ischemic/Hemorrhagic) | 44/10 |
| Side of brain insult (Right/Left) | 25/29 |
| Side of paralysis (Right/Left) | 29/25 |
| Hand dominance (Right/Left) | 52/2 |
| Duration of stroke (Number of days) | 144.25 ± 17.38 |
| TIS score (Mean ± SD) | 17.72 ± 1.76 |
| FMUE score | 28.33 ± 10.79 |
| WMFT | |
| 50.77 ± 8.7 |
| 2.55 ± 0.76 |
| Explanatory Variables | Direct Effect | Indirect Effect | ||||
|---|---|---|---|---|---|---|
| B | SE | p-Value | B | SE | p-Value | |
| Age × TIS × FMUE score | 3.09 | 0.72 | <0.001 | 0.29 | 0.20 | 0.167 |
| Age × TIS × WMFT TS score | −2.62 | −0.54 | <0.001 | −0.41 | −0.15 | 0.011 |
| Age × TIS × WMFT FAS score | 0.21 | 0.05 | <0.001 | 0.01 | 0.01 | 0.243 |
| Gender × TIS × FMUE score | 3.11 | 0.73 | <0.0 01 | −0.48 | 2.56 | 0.429 |
| Gender × TIS × WMFT TS score | −2.63 | 0.56 | <0.001 | 3.17 | 1.98 | 0.116 |
| Gender × TIS × WMFT FAS score | 0.21 | 0.53 | <0.001 | −0.04 | 0.18 | 0.822 |
| PSD × TIS × FMUE score | 2.17 | 0.77 | <0.001 | 5.72 | 2.08 | 0.008 |
| PSD × TIS × WMFT TS score | −2.18 | 0.63 | <0.001 | −3.19 | 1.7 | 0.006 |
| PSD × TIS × WMFT FAS score | 0.14 | 0.05 | <0.001 | 0.41 | 0.15 | 0.009 |
| Explanatory Variables | Sobel-Test | SE | p-Value |
|---|---|---|---|
| Age × TIS × FMUE score | 0.57 | 0.152 | 0.566 |
| Age × TIS × WMFT TS score | 0.61 | 0.203 | 0.540 |
| Age × TIS × WMFT FAS score | 0.62 | 0.081 | 0.530 |
| Gender × TIS × FMUE score | 0.18 | 0.947 | 0.851 |
| Gender × TIS × WMFT TS score | 1.51 | 0.742 | 0.114 |
| Gender × TIS × WMFT FAS score | 0.22 | 0.066 | 0.824 |
| PSD × TIS × FMUE score | 2.25 | 0.441 | 0.044 |
| PSD × TIS × WMFT TS score | 1.69 | 0.333 | 0.090 |
| PSD × TIS × WMFT FAS score | 2.24 | 0.32 | 0.024 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gular, K.; Sivasubramanian, V.; Reddy, R.S.; Tedla, J.S.; Dixit, S. The Mediating Effect of Age, Gender, and Post-Stroke Duration on the Association between Trunk and Upper Limb Recovery in Subacute Stroke Population: A Cross-Sectional Study with Mediation Analysis. Int. J. Environ. Res. Public Health 2022, 19, 15644. https://doi.org/10.3390/ijerph192315644
Gular K, Sivasubramanian V, Reddy RS, Tedla JS, Dixit S. The Mediating Effect of Age, Gender, and Post-Stroke Duration on the Association between Trunk and Upper Limb Recovery in Subacute Stroke Population: A Cross-Sectional Study with Mediation Analysis. International Journal of Environmental Research and Public Health. 2022; 19(23):15644. https://doi.org/10.3390/ijerph192315644
Chicago/Turabian StyleGular, Kumar, Viswanathan Sivasubramanian, Ravi Shankar Reddy, Jaya Shanker Tedla, and Snehil Dixit. 2022. "The Mediating Effect of Age, Gender, and Post-Stroke Duration on the Association between Trunk and Upper Limb Recovery in Subacute Stroke Population: A Cross-Sectional Study with Mediation Analysis" International Journal of Environmental Research and Public Health 19, no. 23: 15644. https://doi.org/10.3390/ijerph192315644
APA StyleGular, K., Sivasubramanian, V., Reddy, R. S., Tedla, J. S., & Dixit, S. (2022). The Mediating Effect of Age, Gender, and Post-Stroke Duration on the Association between Trunk and Upper Limb Recovery in Subacute Stroke Population: A Cross-Sectional Study with Mediation Analysis. International Journal of Environmental Research and Public Health, 19(23), 15644. https://doi.org/10.3390/ijerph192315644









