Effects of Physical Exercise on Executive Function in Adults with Depression: A Systematic Review and Meta-Analysis
Abstract
1. Introduction
2. Materials and Methods
2.1. Search Strategy
2.2. Eligibility Criteria
2.3. Data Management
2.4. Data Extraction
2.5. Risk of Bias (Quality) Assessment
2.6. Strategy for Data Synthesis
3. Results
3.1. Study Selection
3.2. Study Characteristics
3.3. Methodological Quality
3.4. Meta-Analysis Results of the Effects of Interventions on Working Memory
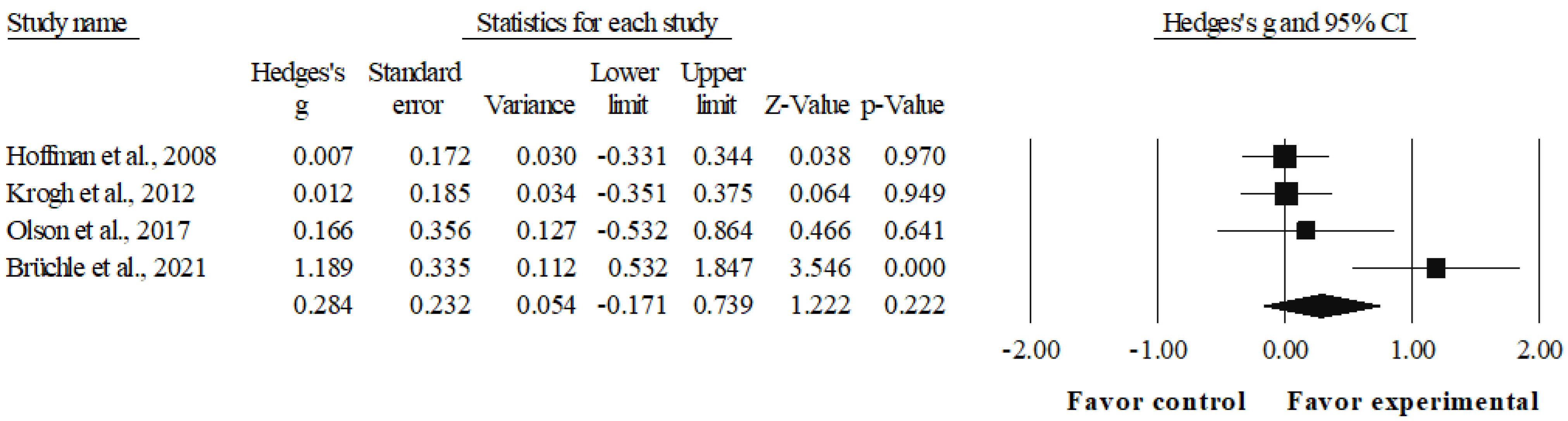
3.5. Meta-Analysis Results of the Effects of Interventions on Inhibition
3.6. Meta-Analysis Results of the Effects of Interventions on Cognitive Flexibility
3.7. Adverse Effects
4. Discussion
4.1. Working Memory
4.2. Inhibition
4.3. Cognitive Flexibility
4.4. Potential Limitations and Suggestions for Future Research
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Appendix A
| Database | Search Strategy |
| EBSCO | TX (“executive functions” OR “cognitive function” OR cognition OR “inhibitory control” OR inhibition OR “working memory” OR “executive functioning” OR “cognitive flexibility”) AND TX (sport OR “modified sport” OR fitness OR exercise OR “physical activity” OR athletics OR “sport practice”) AND TX (depression OR “depressive disorder” OR “mood disorders” OR “major depressive disorder”) |
| PubMed | (((((((((“executive function”[Title/Abstract]) OR (“cognitive function”[Title/Abstract])) OR (cognition)) OR (“inhibitory control”[Title/Abstract])) OR (inhibition[Title/Abstract])) OR (“working memory”[Title/Abstract])) OR (“executive functioning”[Title/Abstract])) OR (“cognitive flexibility”[Title/Abstract])) AND (((((((sport[Title/Abstract]) OR (“modified sport”[Title/Abstract])) OR (fitness[Title/Abstract])) OR (exercise[Title/Abstract])) OR (“physical activity”[Title/Abstract])) OR (athletics[Title/Abstract])) OR (“sport practice”[Title/Abstract]))) AND ((((depression[Title/Abstract]) OR (“depressive disorder”[Title/Abstract])) OR (“mood disorders”[Title/Abstract])) OR (“major depressive disorder”[Title/Abstract])) |
| Scopus | ((TITLE-ABS-KEY (depression)) OR (TITLE-ABS-KEY (“depressive disorder”)) OR (TITLE-ABS-KEY (“mood disorders”)) OR (TITLE-ABS-KEY (“major depressive disorder”))) AND ((TITLE-ABS-KEY (sport)) OR (TITLE-ABS-KEY (“modified sport”)) OR (TITLE-ABS-KEY (fitness)) OR (TITLE-ABS-KEY (exercise)) OR (TITLE-ABS-KEY (“physical activity”)) OR (TITLE-ABS-KEY (athletics)) OR (TITLE-ABS-KEY (“sport practice”))) AND ((TITLE-ABS-KEY (“executive function”)) OR (TITLE-ABS-KEY (“cognitive function”)) OR (TITLE-ABS-KEY (cognition)) OR (TITLE-ABS-KEY (“inhibitory control”)) OR (TITLE-ABS-KEY (inhibition)) OR (TITLE-ABS-KEY (“working memory”)) OR (TITLE-ABS-KEY (“executive functioning”)) OR (TITLE-ABS-KEY (“cognitive flexibility”))) |
| Web of Science | ((TS = (“executive function” OR “cognitive function” OR cognition OR “inhibitory control” OR inhibition OR “working memory” OR “executive functioning” OR “cognitive flexibility”)) AND TS = (sport OR “modified sport” OR fitness OR exercise OR “physical activity” OR athletics OR “sport practice”)) AND TS = (depression OR “depressive disorder” OR “mood disorders” OR “major depressive disorder”) |
References
- Global Burden of 369 Diseases and Injuries in 204 Countries and Territories, 1990–2019: A Systematic Analysis for the Global Burden of Disease Study 2019. Lancet 2020, 396, 1204–1222. [CrossRef]
- Croatto, G.; Vancampfort, D.; Miola, A.; Olivola, M.; Fiedorowicz, J.G.; Firth, J.; Alexinschi, O.; Gaina, M.A.; Makkai, V.; Soares, F.C.; et al. The Impact of Pharmacological and Non-Pharmacological Interventions on Physical Health Outcomes in People with Mood Disorders across the Lifespan: An Umbrella Review of the Evidence from Randomised Controlled Trials. Mol. Psychiatry 2022. [Google Scholar] [CrossRef] [PubMed]
- Wittenborn, A.K.; Woods, S.B.; Priest, J.B.; Morgan, P.C.; Tseng, C.-F.; Huerta, P.; Edwards, C. Couple and Family Interventions for Depressive and Bipolar Disorders: Evidence Base Update (2010–2019). J. Marital Fam. Ther. 2022, 48, 129–153. [Google Scholar] [CrossRef] [PubMed]
- Stewart, W.F.; Ricci, J.A.; Chee, E.; Hahn, S.R.; Morganstein, D. Cost of Lost Productive Work Time among US Workers with Depression. JAMA 2003, 289, 3135–3144. [Google Scholar] [CrossRef] [PubMed]
- Keshavarz, K.; Hedayati, A.; Rezaei, M.; Goudarzi, Z.; Moghimi, E.; Rezaee, M.; Lotfi, F. Economic Burden of Major Depressive Disorder: A Case Study in Southern Iran. BMC Psychiatry 2022, 22, 577. [Google Scholar] [CrossRef] [PubMed]
- Mathers, C.D.; Loncar, D. Projections of Global Mortality and Burden of Disease from 2002 to 2030. PLoS Med. 2006, 3, e442. [Google Scholar] [CrossRef] [PubMed]
- Smith, K. Mental Health: A World of Depression. Nature 2014, 515, 181. [Google Scholar] [CrossRef]
- Semkovska, M.; Quinlivan, L.; O’Grady, T.; Johnson, R.; Collins, A.; O’Connor, J.; Knittle, H.; Ahern, E.; Gload, T. Cognitive Function Following a Major Depressive Episode: A Systematic Review and Meta-Analysis. Lancet Psychiatry 2019, 6, 851–861. [Google Scholar] [CrossRef]
- Rock, P.L.; Roiser, J.P.; Riedel, W.J.; Blackwell, A.D. Cognitive Impairment in Depression: A Systematic Review and Meta-Analysis. Psychol. Med. 2014, 44, 2029–2040. [Google Scholar] [CrossRef]
- Henry, J.; Crawford, J.R. A Meta-Analytic Review of Verbal Fluency Deficits in Depression. J. Clin. Exp. Neuropsychol. 2005, 27, 78–101. [Google Scholar] [CrossRef] [PubMed]
- Stegenga, B.T.; Kamphuis, M.H.; King, M.; Nazareth, I.; Geerlings, M.I. The Natural Course and Outcome of Major Depressive Disorder in Primary Care: The PREDICT-NL Study. Soc. Psychiatry Psychiatr. Epidemiol. 2012, 47, 87–95. [Google Scholar] [CrossRef] [PubMed]
- Knight, M.J.; Air, T.; Baune, B.T. The Role of Cognitive Impairment in Psychosocial Functioning in Remitted Depression. J. Affect. Disord. 2018, 235, 129–134. [Google Scholar] [CrossRef] [PubMed]
- Mcintyre, R.S.; Soczynska, J.Z.; Woldeyohannes, H.O.; Alsuwaidan, M.T.; Cha, D.S.; Carvalho, A.F.; Jerrell, J.M.; Dale, R.M.; Gallaugher, L.A.; Muzina, D.J.; et al. ScienceDirect The Impact of Cognitive Impairment on Perceived Workforce Performance: Results from the International Mood Disorders Collaborative Project. Compr. Psychiatry 2015, 56, 279–282. [Google Scholar] [CrossRef] [PubMed]
- Baune, B.T.; Miller, R.; Mcafoose, J.; Johnson, M.; Quirk, F.; Mitchell, D. The Role of Cognitive Impairment in General Functioning in Major Depression. Psychiatry Res. 2010, 176, 183–189. [Google Scholar] [CrossRef]
- Bortolato, B.; Carvalho, A.F.; McIntyre, R.S. Cognitive Dysfunction in Major Depressive Disorder: A State-of-the-Art Clinical Review. CNS Neurol. Disord. Drug Targets 2014, 13, 1804–1818. [Google Scholar] [CrossRef]
- Friedman, N.; Miyake, A. Unity and Diversity of Executive Functions: Individual Differences as a Window on Cognitive Structure. Cortex 2017, 86, 186–204. [Google Scholar] [CrossRef]
- Lehto, J.; Juujärvi, P.; Kooistra, L.; Pulkkinen, L. Dimensions of Executive Functioning: Evidence from Children. Br. J. Dev. Psychol. 2003, 21, 59–80. [Google Scholar] [CrossRef]
- Miyake, A.; Friedman, N.P.; Emerson, M.J.; Witzki, A.H.; Howerter, A.; Wager, T.D. The Unity and Diversity of Executive Functions and Their Contributions to Complex “Frontal Lobe” Tasks: A Latent Variable Analysis. Cogn. Psychol. 2000, 41, 49–100. [Google Scholar] [CrossRef]
- Zuckerman, H.; Pan, Z.; Park, C.; Brietzke, E.; Musial, N.; Shariq, A.S.; Iacobucci, M.; Yim, S.J.; Lui, L.M.W.; Rong, C.; et al. Recognition and Treatment of Cognitive Dysfunction in Major Depressive Disorder. Front. Psychiatry 2018, 9, 655. [Google Scholar] [CrossRef]
- Gonda, X.; Pompili, M.; Serafini, G.; Carvalho, A.F.; Rihmer, Z.; Dome, P. The Role of Cognitive Dysfunction in the Symptoms and Remission from Depression. Ann. Gen. Psychiatry 2015, 14, 27. [Google Scholar] [CrossRef]
- Culpepper, L.; Lam, R.W.; McIntyre, R.S. Cognitive Impairment in Patients With Depression: Awareness, Assessment, and Management. J. Clin. Psychiatry 2017, 78, 1383–1394. [Google Scholar] [CrossRef] [PubMed]
- Sun, M.; Lanctot, K.; Herrmann, N.; Gallagher, D. Exercise for Cognitive Symptoms in Depression: A Systematic Review of Interventional Studies. Can. J. Psychiatry. 2018, 63, 115–128. [Google Scholar] [CrossRef] [PubMed]
- Guo, S.; Liu, F.; Shen, J.; Wei, M.; Yang, Y. Comparative Efficacy of Seven Exercise Interventions for Symptoms of Depression in College Students: A Network of Meta-Analysis. Medicine 2020, 99, e23058. [Google Scholar] [CrossRef]
- Xie, Y.; Wu, Z.; Sun, L.; Zhou, L.; Wang, G.; Xiao, L.; Wang, H. The Effects and Mechanisms of Exercise on the Treatment of Depression. Front. Psychiatry 2021, 12, 705559. [Google Scholar] [CrossRef] [PubMed]
- Knight, M.J.; Mills, N.T.; Baune, B.T. Contemporary Methods of Improving Cognitive Dysfunction in Clinical Depression. Expert Rev. Neurother. 2019, 19, 431–443. [Google Scholar] [CrossRef]
- Euteneuer, F.; Dannehl, K.; Del Rey, A.; Engler, H.; Schedlowski, M.; Rief, W. Immunological Effects of Behavioral Activation with Exercise in Major Depression: An Exploratory Randomized Controlled Trial. Transl. Psychiatry 2017, 7, e1132. [Google Scholar] [CrossRef] [PubMed]
- Micheli, L.; Ceccarelli, M.; D’Andrea, G.; Tirone, F. Depression and Adult Neurogenesis: Positive Effects of the Antidepressant Fluoxetine and of Physical Exercise. Brain Res. Bull. 2018, 143, 181–193. [Google Scholar] [CrossRef]
- Chen, C.; Nakagawa, S.; An, Y.; Ito, K.; Kitaichi, Y.; Kusumi, I. The Exercise-Glucocorticoid Paradox: How Exercise Is Beneficial to Cognition, Mood, and the Brain While Increasing Glucocorticoid Levels. Front. Neuroendocrinol. 2017, 44, 83–102. [Google Scholar] [CrossRef]
- Zhao, J.-L.; Jiang, W.-T.; Wang, X.; Cai, Z.-D.; Liu, Z.-H.; Liu, G.-R. Exercise, Brain Plasticity, and Depression. CNS Neurosci. Ther. 2020, 26, 885–895. [Google Scholar] [CrossRef]
- Brüchle, W.; Schwarzer, C.; Berns, C.; Scho, S.; Schneefeld, J.; Koester, D.; Schack, T.; Schneider, U.; Rosenkranz, K. Physical Activity Reduces Clinical Symptoms and Restores Neuroplasticity in Major Depression. Front. Psychiatry 2021, 12, 660642. [Google Scholar] [CrossRef]
- Caspersen, C.J.; Powell, K.E.; Christenson, G.M. Physical Activity, Exercise, and Physical Fitness: Definitions and Distinctions for Health-Related Research. Public Health Rep. 1985, 100, 126–131. [Google Scholar] [PubMed]
- Kubesch, S.; Bretschneider, V.; Freudenmann, R.; Weidenhammer, N.; Lehmann, M.; Spitzer, M.; Grön, G. Aerobic Endurance Exercise Improves Executive Functions in Depressed Patients. J. Clin. Psychiatry 2003, 64, 1005–1012. [Google Scholar] [CrossRef] [PubMed]
- Olson, R.L.; Brush, C.J.; Ehmann, P.J.; Alderman, B.L. A Randomized Trial of Aerobic Exercise on Cognitive Control in Major Depression. Clin. Neurophysiol. 2017, 128, 903–913. [Google Scholar] [CrossRef] [PubMed]
- Krogh, J.; Videbech, P.; Thomsen, C.; Gluud, C.; Nordentoft, M. DEMO-II Trial. Aerobic Exercise versus Stretching Exercise in Patients with Major Depression-a Randomised Clinical Trial. PLoS ONE 2012, 7, e48316. [Google Scholar] [CrossRef]
- Buschert, V.; Prochazka, D.; Bartl, H.; Diemer, J.; Malchow, B.; Zwanzger, P.; Brunnauer, A. Effects of Physical Activity on Cognitive Performance: A Controlled Clinical Study in Depressive Patients. Eur. Arch. Psychiatry Clin. Neurosci. 2019, 269, 555–563. [Google Scholar] [CrossRef]
- Krogh, J.; Saltin, B.; Gluud, C.; Nordentoft, M. The DEMO Trial: A Randomized, Parallel-Group, Observer-Blinded Clinical Trial of Strength versus Aerobic versus Relaxation Training for Patients with Mild to Moderate Depression. J. Clin. Psychiatry 2009, 70, 790–800. [Google Scholar] [CrossRef]
- Zhang, J.; Chen, T. Effect of Aerobic Exercise on Cognitive Function and Symptoms in Patients with Depression. Natl. Acad. Sci. Lett. 2019, 42, 419–421. [Google Scholar] [CrossRef]
- Hoffman, B.M.; Blumenthal, J.A.; Babyak, M.A.; Smith, P.J.; Rogers, S.D.; Doraiswamy, P.M.; Sherwood, A. Exercise Fails to Improve Neurocognition in Depressed Middle-Aged and Older Adults. Med. Sci. Sport. Exerc. 2008, 40, 1344–1352. [Google Scholar] [CrossRef]
- Chen, C.; Shan, W. Pharmacological and Non-Pharmacological Treatments for Major Depressive Disorder in Adults: A Systematic Review and Network Meta-Analysis. Psychiatry Res. 2019, 281, 112595. [Google Scholar] [CrossRef]
- Carneiro, L.; Afonso, J.; Ramirez-Campillo, R.; Murawska-Ciałowciz, E.; Marques, A.; Clemente, F.M. The Effects of Exclusively Resistance Training-Based Supervised Programs in People with Depression: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Int. J. Environ. Res. Public Health 2020, 17, 6715. [Google Scholar] [CrossRef]
- Carneiro, L.; Rosenbaum, S.; Ward, P.B.; Clemente, F.M.; Ramirez-Campillo, R.; Monteiro-Júnior, R.S.; Martins, A.; Afonso, J. Web-Based Exercise Interventions for Patients with Depressive and Anxiety Disorders: A Systematic Review of Randomized Controlled Trials. Rev. Bras. Psiquiatr. 2022, 44, 331–341. [Google Scholar] [CrossRef] [PubMed]
- Schuch, F.B.; Vancampfort, D.; Richards, J.; Rosenbaum, S.; Ward, P.B.; Stubbs, B. Exercise as a Treatment for Depression: A Meta-Analysis Adjusting for Publication Bias. J. Psychiatr. Res. 2016, 77, 42–51. [Google Scholar] [CrossRef] [PubMed]
- Brondino, N.; Rocchetti, M.; Fusar-Poli, L.; Codrons, E.; Correale, L.; Vandoni, M.; Barbui, C.; Politi, P. A Systematic Review of Cognitive Effects of Exercise in Depression. Acta Psychiatr. Scand. 2017, 135, 285–295. [Google Scholar] [CrossRef] [PubMed]
- Nuechterlein, K.H.; Green, M.F.; Kern, R.S.; Baade, L.E.; Barch, D.M.; Cohen, J.D.; Essock, S.; Fenton, W.S.; Frese, F.J., 3rd; Gold, J.M.; et al. The MATRICS Consensus Cognitive Battery, Part 1: Test Selection, Reliability, and Validity. Am. J. Psychiatry 2008, 165, 203–213. [Google Scholar] [CrossRef] [PubMed]
- Kriesche, D.; Woll, C.F.J.; Tschentscher, N.; Engel, R.R.; Karch, S. Neurocognitive Deficits in Depression: A Systematic Review of Cognitive Impairment in the Acute and Remitted State. Eur. Arch. Psychiatry Clin. Neurosci. 2022, 1, 1–14. [Google Scholar] [CrossRef]
- Jaeger, J.; Berns, S.; Uzelac, S.; Davis-Conway, S. Neurocognitive Deficits and Disability in Major Depressive Disorder. Psychiatry Res. 2006, 145, 39–48. [Google Scholar] [CrossRef]
- Withall, A.; Harris, L.M.; Cumming, S.R. The Relationship between Cognitive Function and Clinical and Functional Outcomes in Major Depressive Disorder. Psychol. Med. 2009, 39, 393–402. [Google Scholar] [CrossRef]
- Fossati, P.; Ergis, A.M.; Allilaire, J.F. [Executive functioning in unipolar depression: A review]. Encephale 2002, 28, 97–107. [Google Scholar]
- Sharma, V.K.; Das, S.; Mondal, S.; Goswami, U.; Gandhi, A. Effect of Sahaj Yoga on Neuro-Cognitive Functions in Patients Suffering from Major Depression. Indian J. Physiol. Pharmacol. 2006, 50, 375–383. [Google Scholar]
- Shojania, K.G.; Sampson, M.; Ansari, M.T.; Ji, J.; Doucette, S.; Moher, D. How Quickly Do Systematic Reviews Go out of Date? A Survival Analysis. Ann. Intern. Med. 2007, 147, 224–233. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 Statement: An Updated Guideline for Reporting Systematic Reviews. BMJ 2021, 10, 89. [Google Scholar] [CrossRef]
- Considine, J.; Shaban, R.Z.; Fry, M.; Curtis, K. Evidence Based Emergency Nursing: Designing a Research Question and Searching the Literature. Int. Emerg. Nurs. 2017, 32, 78–82. [Google Scholar] [CrossRef] [PubMed]
- Frost, A.; Moussaoui, S.; Kaur, J.; Aziz, S.; Fukuda, K.; Niemeier, M. Is the N-Back Task a Measure of Unstructured Working Memory Capacity? Towards Understanding Its Connection to Other Working Memory Tasks. Acta Psychol. 2021, 219, 103398. [Google Scholar] [CrossRef] [PubMed]
- Barzykowski, K.; Wereszczyński, M.; Hajdas, S.; Radel, R. Cognitive Inhibition Behavioral Tasks in Online and Laboratory Settings: Data from Stroop, SART and Eriksen Flanker Tasks. Data Br. 2022, 43, 108398. [Google Scholar] [CrossRef]
- Suzuki, H.; Sakuma, N.; Kobayashi, M.; Ogawa, S.; Inagaki, H.; Edahiro, A.; Ura, C.; Sugiyama, M.; Miyamae, F.; Watanabe, Y.; et al. Normative Data of the Trail Making Test Among Urban Community-Dwelling Older Adults in Japan. Front. Aging Neurosci. 2022, 14, 832158. [Google Scholar] [CrossRef]
- Thompson, P.D.; Arena, R.; Riebe, D.; Pescatello, L.S. ACSM’s New Preparticipation Health Screening Recommendations from ACSM’s Guidelines for Exercise Testing and Prescription, Ninth Edition. Curr. Sports Med. Rep. 2013, 12, 215–217. [Google Scholar] [CrossRef]
- Contreras-Osorio, F.; Ramirez-Campillo, R.; Cerda-Vega, E.; Campos-Jara, R.; Martínez-Salazar, C.; Arellano-Roco, C.; Campos-Jara, C. Effects of Sport-Based Interventions on Executive Function in Older Adults: A Systematic Review and Meta-Analysis Protocol. Brain Sci. 2022, 12, 1142. [Google Scholar] [CrossRef]
- de Morton, N.A. The PEDro Scale Is a Valid Measure of the Methodological Quality of Clinical Trials: A Demographic Study. Aust. J. Physiother. 2009, 55, 129–133. [Google Scholar] [CrossRef]
- Maher, C.G.; Sherrington, C.; Herbert, R.D.; Moseley, A.M.; Elkins, M. Reliability of the PEDro Scale for Rating Quality of Randomized Controlled Trials. Phys. Ther. 2003, 83, 713–721. [Google Scholar] [CrossRef]
- Yamato, T.P.; Maher, C.; Koes, B.; Moseley, A. The PEDro Scale Had Acceptably High Convergent Validity, Construct Validity, and Interrater Reliability in Evaluating Methodological Quality of Pharmaceutical Trials. J. Clin. Epidemiol. 2017, 86, 176–181. [Google Scholar] [CrossRef]
- Ramirez-Campillo, R.; Castillo, D.; Raya-González, J.; Moran, J.; de Villarreal, E.S.; Lloyd, R.S. Effects of Plyometric Jump Training on Jump and Sprint Performance in Young Male Soccer Players: A Systematic Review and Meta-Analysis. Sports Med. 2020, 50, 2125–2143. [Google Scholar] [CrossRef] [PubMed]
- Ramirez-Campillo, R.; Sánchez, J.; Romero-Moraleda, B.; Javier, Y.; García-Hermoso, A.; Clemente, F. Effects of Plyometric Jump Training in Female Soccer Player’s Vertical Jump Height: A Systematic Review with Meta-Analysis. J. Sports Sci. 2020, 38, 1475–1487. [Google Scholar] [CrossRef] [PubMed]
- Stojanović, E.; Ristić, V.; McMaster, D.T.; Milanović, Z. Effect of Plyometric Training on Vertical Jump Performance in Female Athletes: A Systematic Review and Meta-Analysis. Sports Med. 2017, 47, 975–986. [Google Scholar] [CrossRef] [PubMed]
- García-Hermoso, A.; Ramírez-Campillo, R.; Izquierdo, M. Is Muscular Fitness Associated with Future Health Benefits in Children and Adolescents? A Systematic Review and Meta-Analysis of Longitudinal Studies. Sports Med. 2019, 49, 1079–1094. [Google Scholar] [CrossRef]
- Moran, J.; Ramirez-Campillo, R.; Granacher, U. Effects of Jumping Exercise on Muscular Power in Older Adults: A Meta-Analysis. Sports Med. 2018, 48, 2843–2857. [Google Scholar] [CrossRef]
- Hopkins, W.G.; Marshall, S.W.; Batterham, A.M.; Hanin, J. Progressive Statistics for Studies in Sports Medicine and Exercise Science. Med. Sci. Sports Exerc. 2009, 41, 3–13. [Google Scholar] [CrossRef]
- Higgins, J.P.T.; Thompson, S.G. Quantifying Heterogeneity in a Meta-Analysis. Stat. Med. 2002, 21, 1539–1558. [Google Scholar] [CrossRef]
- Baddeley, A. Working Memory. Science 1992, 255, 556–559. [Google Scholar] [CrossRef]
- Baddeley, A. Working Memory: Theories, Models, and Controversies. Annu. Rev. Psychol. 2012, 63, 1–29. [Google Scholar] [CrossRef]
- Smith, P.J.; Blumenthal, J.A.; Hoffman, B.M.; Cooper, H.; Strauman, T.A.; Welsh-Bohmer, K.; Browndyke, J.N.; Sherwood, A. Aerobic Exercise and Neurocognitive Performance: A Meta-Analytic Review of Randomized Controlled Trials. Psychosom. Med. 2010, 72, 239–252. [Google Scholar] [CrossRef]
- Greer, T.L.; Grannemann, B.D.; Chansard, M.; Karim, A.I.; Trivedi, M.H. Dose-Dependent Changes in Cognitive Function with Exercise Augmentation for Major Depression: Results from the TREAD Study. Eur. Neuropsychopharmacol. 2015, 25, 248–256. [Google Scholar] [CrossRef] [PubMed]
- Falkai, P.; Schmitt, A.; Rosenbeiger, C.P.; Maurus, I.; Hattenkofer, L.; Hasan, A.; Malchow, B.; Heim-Ohmayer, P.; Halle, M.; Heitkamp, M. Aerobic Exercise in Severe Mental Illness: Requirements from the Perspective of Sports Medicine. Eur. Arch. Psychiatry Clin. Neurosci. 2022, 272, 643–677. [Google Scholar] [CrossRef] [PubMed]
- Garber, C.E.; Blissmer, B.; Deschenes, M.R.; Franklin, B.A.; Lamonte, M.J.; Lee, I.-M.; Nieman, D.C.; Swain, D.P. American College of Sports Medicine Position Stand. Quantity and Quality of Exercise for Developing and Maintaining Cardiorespiratory, Musculoskeletal, and Neuromotor Fitness in Apparently Healthy Adults: Guidance for Prescribing Exercise. Med. Sci. Sports Exerc. 2011, 43, 1334–1359. [Google Scholar] [CrossRef]
- Friedman, N.P.; Miyake, A. The Relations among Inhibition and Interference Control Functions: A Latent-Variable Analysis. J. Exp. Psychol. Gen. 2004, 133, 101–135. [Google Scholar] [CrossRef] [PubMed]
- Dregan, A.; Gulliford, M.C. Leisure-Time Physical Activity over the Life Course and Cognitive Functioning in Late Mid-Adult Years: A Cohort-Based Investigation. Psychol. Med. 2013, 43, 2447–2458. [Google Scholar] [CrossRef] [PubMed]
- Gaertner, B.; Buttery, A.K.; Finger, J.D.; Wolfsgruber, S.; Wagner, M.; Busch, M.A. Physical Exercise and Cognitive Function across the Life Span: Results of a Nationwide Population-Based Study. J. Sci. Med. Sport 2018, 21, 489–494. [Google Scholar] [CrossRef]
- Monteiro, F.C.; Schuch, F.B.; Deslandes, A.C.; Mosqueiro, B.P.; Caldieraro, M.A.; Fleck, M.P.D.A. Factors Associated with Adherence to Sports and Exercise among Outpatients with Major Depressive Disorder. Trends Psychiatry Psychother. 2021, 43, 108–115. [Google Scholar] [CrossRef]
- Ranjbar, E.; Memari, A.H.; Hafizi, S.; Shayestehfar, M.; Mirfazeli, F.S.; Eshghi, M.A. Depression and Exercise: A Clinical Review and Management Guideline. Asian J. Sports Med. 2015, 6, e24055. [Google Scholar] [CrossRef]
- Schuch, F.B.; Vasconcelos-Moreno, M.P.; Borowsky, C.; Zimmermann, A.B.; Rocha, N.S.; Fleck, M.P. Exercise and Severe Major Depression: Effect on Symptom Severity and Quality of Life at Discharge in an Inpatient Cohort. J. Psychiatr. Res. 2015, 61, 25–32. [Google Scholar] [CrossRef]
- Monnart, A.; Kornreich, C.; Verbanck, P.; Campanella, S. Just Swap Out of Negative Vibes? Rumination and Inhibition Deficits in Major Depressive Disorder: Data from Event-Related Potentials Studies. Front. Psychol. 2016, 7, 1019. [Google Scholar] [CrossRef]
- Diamond, A. Activities and Programs That Improve Children’s Executive Functions. Curr. Dir. Psychol. Sci. 2012, 21, 335–341. [Google Scholar] [CrossRef] [PubMed]
- Pesce, C. Shifting the Focus From Quantitative to Qualitative Exercise Characteristics in Exercise and Cognition Research. J. Sport Exerc. Psychol. 2012, 34, 766–786. [Google Scholar] [CrossRef] [PubMed]
- Hu, M.X.; Turner, D.; Generaal, E.; Bos, D.; Ikram, M.K.; Ikram, M.A.; Cuijpers, P.; Penninx, B.W.J.H. Exercise Interventions for the Prevention of Depression: A Systematic Review of Meta-Analyses. BMC Public Health 2020, 20, 1255. [Google Scholar] [CrossRef] [PubMed]
- Fossati, C.; Torre, G.; Vasta, S.; Giombini, A.; Quaranta, F.; Papalia, R.; Pigozzi, F. Physical Exercise and Mental Health: The Routes of a Reciprocal Relation. Int. J. Environ. Res. Public Health 2021, 18, 12364. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Tong, W.-X.; Zhang, M.; Wang, G.-X.; Zhang, Y.-S.; Meng, S.-Q.; Li, Y.-X.; Cui, Z.-L.; Zhang, J.-Y.; Ye, Y.-P.; et al. Epidemiological Study of Physical Activity, Negative Moods, and Their Correlations among College Students. Int. J. Environ. Res. Public Health 2022, 19, 11748. [Google Scholar] [CrossRef]



| PICOS | Inclusion Criteria | Exclusion Criteria |
|---|---|---|
| 1. Population | 1.1. Adults (age, 18–65 years) diagnosed with major depression or unipolar depression according to the criteria of a validated instrument, such as the Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition (DSM-IV), or the International Classification of Diseases 10th Revision (ICD-10), without restrictions based on sex or fitness level. | 1.1. Children, adolescents, or older adults. 1.2. Presence of another primary neurological or psychiatric diagnosis, such as dementia, bipolar disorder, or psychosis. 1.3. Medical comorbidities that limit participation in physical exercise activities (e.g., significant musculoskeletal difficulties). 1.4. Active drug or alcohol abuse or dependence. 1.5. Pregnant or lactating women. 1.6. Intellectual disability. |
| 2. Intervention | 2.1. Chronic exercise intervention programs (with a minimum duration of 3 weeks) as independent therapy or complementary to usual psychiatric treatment (e.g., pharmacological treatment). 2.2. The interventions should involve aerobic training or strength training or programs combining different types of exercises (e.g., coordination, endurance, or strength training). | 2.1. Acute interventions. 2.2. Chronic exercise intervention programs combined with meditation. 2.3. Chronic interventions that are not related to physical exercise. |
| 3. Comparator | 3.1. A group made up of adults diagnosed with depression not exposed to physical exercise intervention. 3.2. The control condition may be active (e.g., relaxation techniques) or passive (e.g., placebo). | 3.1. Absence of control group. |
| 4. Outcome | 4.1. Pre–post-intervention values for one or more direct assessment measures for the executive functions of working memory, inhibition, or cognitive flexibility. | 4.1. Indirect measures of executive functions (e.g., questionnaire). |
| 5. Study design | 5.1. Longitudinal studies with at least one experimental group and one control group that include pre- and post-intervention measurements. | 5.1. Cross-sectional studies. |
| References | N | Sex (M/F) | Age (Years) | Diagnosis | Diagnostic Instruments | Baseline Depressive Symptom Severity | Baseline Cognitive Status | Comorbidity |
|---|---|---|---|---|---|---|---|---|
| Hoffman et al., 2008 [38] | 153; EG: 104, CG: 49 | 37/116 | EG: 51 ± 7, CG: 51.2 ± 7.8 | Major depressive disorder | DSM-IV-TR criteria; BDI-II (≥12); HAM-D | HAM-D: 16.8 ± 4.3 (mild-to-moderate) | No cognitive impairment | Hypertension EG: 27, CG: 14; diabetes EG: 7, CG: 4; smoking EG: 15, CG: 8; alcohol (≥3 alcoholic drinks per week) EG: 7, CG: 5 |
| Krogh et al., 2009 [36] | 165; EG1: 55, EG2: 55, CG: 55 | 43/122 | EG1: 41.9 ± 8.7, EG2: 38.1 ± 9.0, CG: 36.7 ± 8.7 | Unipolar depression | ICD-10 and DSM-IV criteria; Major Depression Inventory | HAM-D-17: 17.8 ± 3.8 (mild-to-moderate) | Not reported | Not reported |
| Krogh et al., 2012 [34] | 115; EG: 56, CG: 59 | 38/77 | EG: 39.7 ± 11.3, CG: 43.4 ± 11.2 | Major depression | DSM-IV criteria; Danish version of the Mini-International Neuropsychiatric Interview; HAM-D17 (>12) | HAM-D-17: 18.9 (95%CI 13–28) (moderate) | Not reported | Generalized anxiety EG: 33, CG: 34; Hypertension EG: 11, CG: 12 |
| Olson et al., 2017 [33] | 30; EG: 15, CG; 15 | 6/24 | EG: 21.0 ± 1.9, CG; 21.2 ± 2.2 | Major depressive disorder | ICD-10, DSM-IV and DSM-V criteria; MINI | BDI-II EG: 24.5 ± 11.5, CG: 24.3 ± 11.9 (moderate) | Not reported | Anxiety (~18%) |
| Zhang and Chen, 2019 [37] | 125; EG: 63, CG: 62 | 44/81 | EG: 31.4 ± 7.2, CG: 32.2 ± 7.6 | Single phase depression | DSM-IV criteria | HAM-D EG: 10.3 ± 3.4, CG: 10.0 ± 3.8 (mild) | Decreased processing speed, attention, memory, verbal fluency and executive function (at least 1 SD lower than the standardized value) | Hypertension, diabetes |
| Brüchle et al., 2021 [30] | 41; EG: 23, CG: 18 | 23/18 | EG: 33.3 ± 3.06, CG: 40.11 ± 3.63 | Major depressive disorder | ICD-10 criteria; BDI-II ≥ 10; HAM-D-17 ≥ 9 | HAM-D-17 EG: 19.17 ± 0.78, CG: 17.83 ± 0.75; BDI-II EG: 27.74 ± 1.44, CG: 26.11 ± 1.77 (moderate) | No cognitive impairment | Not reported |
| Buschert et al., 2019 [35] | 30; EG: 15, CG: 15 | 11/19 | EG: 47.27 ± 6.84, CG:47.47 ± 8.47 | Unipolar depression | ICD-10 criteria | BDI-II EG: 22.40 ± 8.53, CG: 18.27 ± 11.56; HAM-D-7 EG: 11.00 ± 3.42, CG: 9.67 ± 4.21 (moderate) | Not reported | Other mood disorders (EG: n = 1), neurotic, stress-related and somatoform disorders (EG: n = 1; CG n = 3), and disorders of adult personality and behavior (EG: n = 1; CG: n = 1) |
| References | Exercise Program | Control Condition | Compliance with the Intervention Program | Length of Intervention | Weekly Frequency | Length of Sessions | Intensity | Other Treatments | Executive Function Tasks |
|---|---|---|---|---|---|---|---|---|---|
| Hoffman et al., 2008 [38] | Supervised exercise: (1) 10 min walking warm-up exercise, (2) 30 min walking or jogging on a treadmill at an intensity that would maintain their heart rate within the assigned training range, (3) 5 min cool-down exercises. Home-based exercise: Participants in a home-based exercise program received the same exercise prescription. Telephone follow-up and follow-up through scheduled visits during the intervention period. | Placebo pill | 68% of home exercisers completed at least 75% of the 48 scheduled sessions compared with 67% of supervised exercisers. Treatment completers attended an average of 38.8 exercise sessions. | 16 weeks | 3/week | 45 min | 70%–85% of HRR. | CG: for insomnia, use of a hypnotic (zolpidem) no more than four doses during treatment. | Animal naming, COWAT, digit symbol subtest (WAIS-R), digits backward, Ruff 2 & 7 test, Stroop color and word test, TMT B-A. |
| Krogh et al., 2009 [36] | Strength group: Circuit-training program with six exercises on machines involving large muscle groups: leg extension, leg press, total abdominal, lower back, chest press, and vertical traction. As a supplement to this, free weights and sandbags were used for exercising the calf muscles, the arm abductors, the triceps muscles, and the hip abductors. Aerobic group: 10 different aerobic exercises using large muscle groups. Machines were used for cycling, running, stepping, abdominal exercises, and rowing. Additional exercises were sliding movements on small carpets, trampoline, step bench, jump rope, and Ski Fitter. | Relaxation training | The mean participation was strength group: 18.0 (56.2%), aerobic group: 16.2 (50.6%), and control group: 10.5 (32.8%) sessions of the 32 sessions. | 4 months | 2/week | 90 min | Strength group: 50%–75% of RM. Aerobic group: 70%–89% of HRmax. Relaxation group: <12 on the Borg scale. | Antidepressant medication EG1: 39, EG2: 37, CG: 38; psychotherapy EG1: 24, EG2: 28, CG: 25 | Digit span test, subtracting serial sevens, digit symbol test, verbal fluency S and animals, TMT—Part B. |
| Krogh et al., 2012 [34] | Aerobic training: (1) 10 min general low-intensity warm-up, (2) 30 min aerobic exercise on a stationary cycle ergometer, (3) 5 min low-intensity cool-down period. | Stretching exercises | Average attendance: 36%. Mean attendance EG: 13.5 sessions (range 0–34 and SD 9.4), CG: 12.5 sessions (range 0–34 and SD 9.3) of a planned total of 36 sessions. | 3 months | 3/week | 45 min | 65%–80% of HRmax. | Antidepressant medication EG: 4, CG: 8 | Digits backward, subtracting serial sevens, Stroop test, TMT—Part B, digit symbol subtest (WAIS-R), verbal fluency S and animals. |
| Olson et al., 2017 [33] | 45 min of continuous steady-state exercise performed on a treadmill or cycle ergometer at a prescribed moderate intensity. | Stretching exercises | 75% retention rate. | 8 weeks | 3/week | 30–45 min | 40%–65% of HRR. | Antidepressant medication (∼14%). | Modified flanker task. |
| Zhang and Chen, 2019 [37] | Participants were asked to jog 3 km each time. | Routine treatment (drug therapy) | Four cases were lost in the experimental group. | 8 weeks | 3/week | Not defined | Target rate: 170-age. If the patient had high blood pressure, diabetes or other physical body disease, a heart rate of 110 bpm was indicated. | Routine treatment (drug therapy) | Wechsler Memory Scale Revised in China digit span test (backward), VF test, TMT-B. |
| Brüchle et al., 2021 [30] | Exercise program: Each session lasted 60 min (without breaks) and focused on one of the three exercise types once a week, namely, coordination, endurance, or strength training. These three exercise sessions were repeated every week. Each session began with a 10 min warm-up which combined physical and cognitive tasks, by coding certain movements (e.g., circling of arms, lifting knees up) with colors (colored cards held up by the instructor). The color-movement associations changed randomly in every session. | Cooperation group games with other participants. | Out of 50 recruited patients, 23 (of 25) in the experimental group and 18 (of 25) in the control group completed the study. Only results of patients who participated in all intervention sessions during the 3-week intervention period are reported. | 3 weeks | 3/week | 60 min | Mean heart rate EG: 126.84 ± 2.83 bpm, CG: 83.02 ± 3.08 bpm. | Medication (e.g., with antidepressants) was not changed during the intervention period. Benzodiazepine <1 mg/day lorazepam equivalent. Excluded: anticonvulsive medication, lithium, and antipsychotics. | Work performance series, N-back verbal test (2-back), TMT-B, response inhibition task, Stroop interference test. |
| Buschert et al., 2019 [35] | Endurance training: The sessions comprised outdoor walking, Nordic walking, or running in groups of up to five patients. In adverse weather conditions, patients exercised indoors on stationary bicycles. | Occupational therapy in a group | Overall dropout rate: 21%. Number of treatment sessions received in this study EG: mean = 10.00 (SD = 2.95); CG: mean = 14.27 (SD = 8.18). | 3–4 weeks | 2–3/week | 30 min | 85% of HRmax, calculated as 200 minus age in years was taken as the upper limit. | Groups did not differ over the course of the entire treatment regarding psychopharmacological treatment (no antidepressive mediation, tri-/tetracylics, selective antidepressants, or changes in the medication). | Digit span backward, computer-assisted cardsorting procedure (Computergestütztes Kartensortierverfahren, CKV). |
| Study | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | Total |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Brüchle et al., 2021 [30] | 1 | 0 | 0 | 1 | 0 | 0 | 1 | 0 | 1 | 1 | 1 | 5 |
| Olson et al., 2017 [33] | 1 | 1 | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 1 | 1 | 4 |
| Krogh et al., 2012 [34] | 1 | 1 | 1 | 1 | 0 | 0 | 1 | 1 | 1 | 1 | 1 | 8 |
| Buschert et al., 2019 [35] | 1 | 1 | 0 | 1 | 0 | 0 | 0 | 0 | 1 | 1 | 1 | 5 |
| Krogh et al., 2009 [36] | 1 | 1 | 1 | 1 | 0 | 0 | 1 | 0 | 1 | 1 | 1 | 7 |
| Zhang and Chen, 2019 [37] | 1 | 0 | 0 | 1 | 0 | 0 | 1 | 0 | 1 | 1 | 1 | 5 |
| Hoffman et al., 2008 [38] | 0 | 1 | 0 | 1 | 0 | 0 | 1 | 0 | 1 | 1 | 1 | 6 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Contreras-Osorio, F.; Ramirez-Campillo, R.; Cerda-Vega, E.; Campos-Jara, R.; Martínez-Salazar, C.; Reigal, R.E.; Hernández-Mendo, A.; Carneiro, L.; Campos-Jara, C. Effects of Physical Exercise on Executive Function in Adults with Depression: A Systematic Review and Meta-Analysis. Int. J. Environ. Res. Public Health 2022, 19, 15270. https://doi.org/10.3390/ijerph192215270
Contreras-Osorio F, Ramirez-Campillo R, Cerda-Vega E, Campos-Jara R, Martínez-Salazar C, Reigal RE, Hernández-Mendo A, Carneiro L, Campos-Jara C. Effects of Physical Exercise on Executive Function in Adults with Depression: A Systematic Review and Meta-Analysis. International Journal of Environmental Research and Public Health. 2022; 19(22):15270. https://doi.org/10.3390/ijerph192215270
Chicago/Turabian StyleContreras-Osorio, Falonn, Rodrigo Ramirez-Campillo, Enrique Cerda-Vega, Rodrigo Campos-Jara, Cristian Martínez-Salazar, Rafael E. Reigal, Antonio Hernández-Mendo, Lara Carneiro, and Christian Campos-Jara. 2022. "Effects of Physical Exercise on Executive Function in Adults with Depression: A Systematic Review and Meta-Analysis" International Journal of Environmental Research and Public Health 19, no. 22: 15270. https://doi.org/10.3390/ijerph192215270
APA StyleContreras-Osorio, F., Ramirez-Campillo, R., Cerda-Vega, E., Campos-Jara, R., Martínez-Salazar, C., Reigal, R. E., Hernández-Mendo, A., Carneiro, L., & Campos-Jara, C. (2022). Effects of Physical Exercise on Executive Function in Adults with Depression: A Systematic Review and Meta-Analysis. International Journal of Environmental Research and Public Health, 19(22), 15270. https://doi.org/10.3390/ijerph192215270








