WtsWrng Interim Comparative Effectiveness Evaluation and Description of the Challenges to Develop, Assess, and Introduce This Novel Digital Application in a Traditional Health System
Abstract
1. Introduction
1.1. Context Analysis
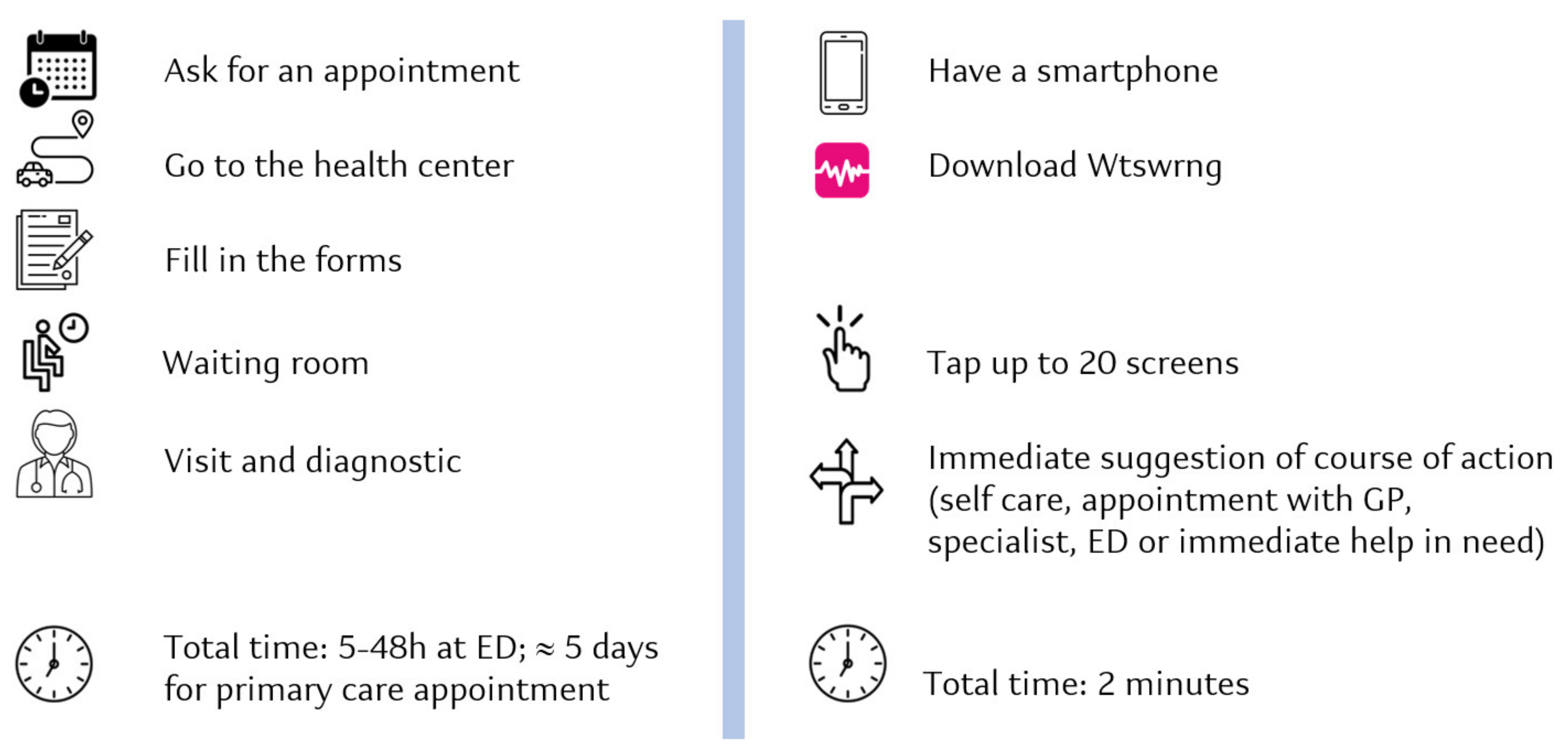
1.2. WtsWrng (WW) DA: Why and for What Purpose
2. Methods: WtsWrng Development and Evaluation
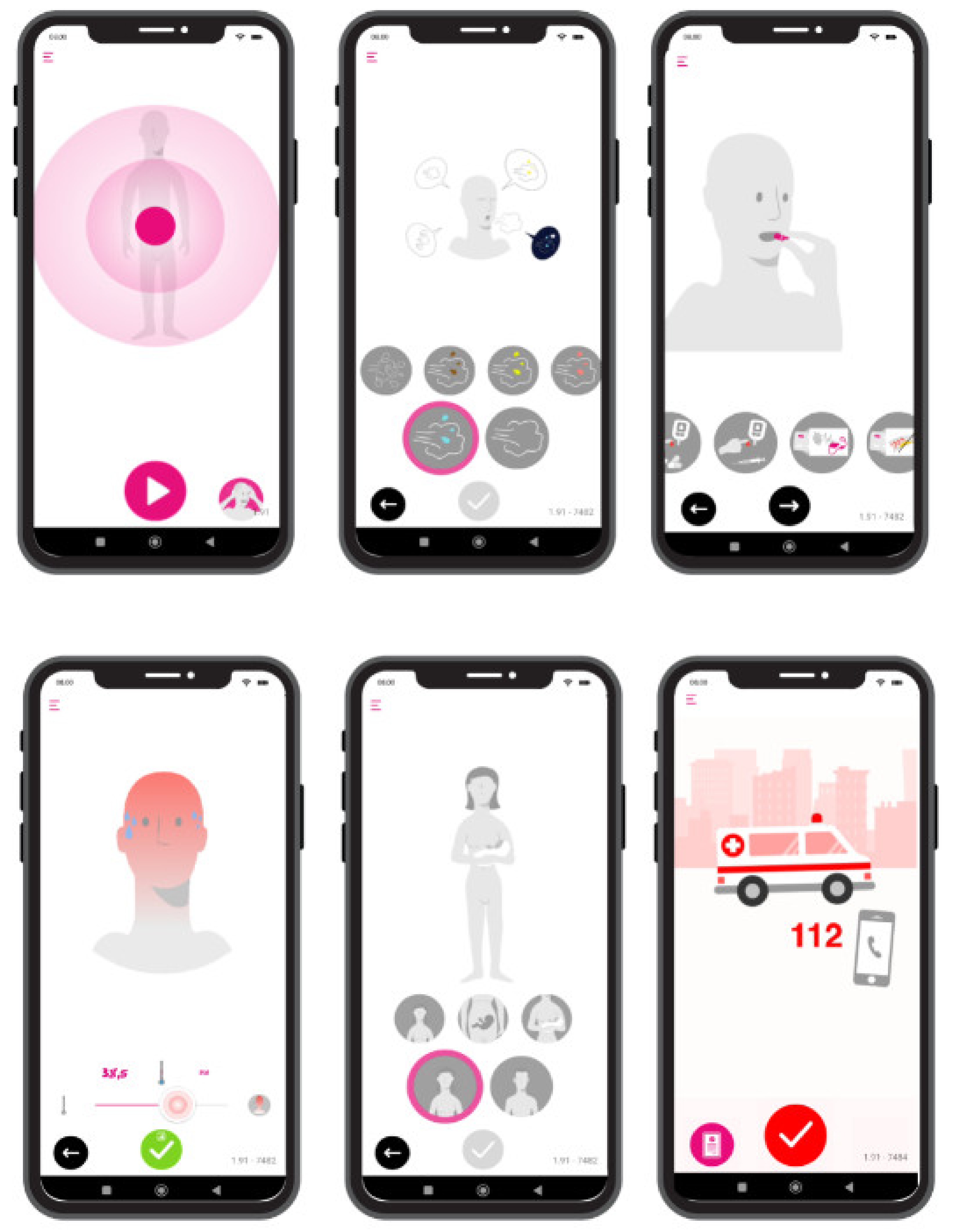
2.1. Building the First Prototype: Ensuring the Accuracy of WtsWrng Decision Trees and the Subsequent Algorithms and Drawing Design
2.2. Usability and User Experience
2.3. Clinical Validation of WtsWrng: From Fast-Track Early Scientific Advice from NICE on WW Evidence Generation Plan to the Study Design, Agile Iterative Development, and Agile Iterative Interim Analysis
2.4. Protection of WW
3. Results
3.1. Usability and User Experience
3.2. Clinical Validation: Agile Iterative Interim Analysis
3.3. Protection of WW
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Jones, R. Access to primary care: Creative solutions are needed. Br. J. Gen. Pract. 2015, 65, 620–621. [Google Scholar] [CrossRef] [PubMed][Green Version]
- World Health Organization. World Bank and WHO: Half the World Lacks Access to Essential Health Services, 100 Million Still Pushed into Extreme Poverty Because of Health Expenses. Available online: https://www.who.int/news/item/13-12-2017-world-bank-and-who-half-the-world-lacks-access-to-essential-health-services-100-million-still-pushed-into-extreme-poverty-because-of-health-expenses (accessed on 17 August 2022).
- World Health Organization. Tracking Universal Health Coverage: 2017 Global Monitoring Report. 2017. Available online: https://apps.who.int/iris/bitstream/handle/10665/259817/9789241513555-eng.pdf (accessed on 17 August 2022).
- Bresnick, J. How Many Emergency Department Visits Are Really Avoidable? 2017. Available online: https://healthitanalytics.com/news/how-many-emergency-department-visits-are-really-avoidable (accessed on 17 August 2022).
- Weinick, R.M.; Burns, R.M.; Mehrotra, A. Many Emergency Department Visits Could be Managed at Urgent Care Centers and Retail Clinics. Health Aff. 2010, 29, 1630–1636. [Google Scholar] [CrossRef] [PubMed]
- Renfrow, J. UnitedHealth: Healthcare Could Save $32B Annually by Diverting Avoidable ED Visits|Fierce Healthcare. 2019. Available online: https://www.fiercehealthcare.com/payer/two-thirds-ed-visits-avoidable (accessed on 31 August 2022).
- Zane, R. The Hardest Time in the History of Emergency Medicine. NEJM Catal. Innov. Care Deliv. 2022, 3. Available online: https://catalyst.nejm.org/doi/full/10.1056/CAT.22.0009 (accessed on 29 August 2022). [CrossRef]
- Schwartz, E. The Global Health Care Worker Shortage: 10 Numbers to Note. Available online: https://www.projecthope.org/the-global-health-worker-shortage-10-numbers-to-note/04/2022/ (accessed on 23 August 2022).
- Sonis, J.D.; Aaronson, E.L.; Lee, R.Y.; Philpotts, L.L.; White, B.A. Emergency Department Patient Experience: A Systematic Review of the Literature. J. Patient Exp. 2018, 5, 101–106. [Google Scholar] [CrossRef] [PubMed]
- Soler, W.; Gómez Muñoz, M.; Bragulat, E.; Álvarez, A. El triaje: Herramienta fundamental en urgencias y emergencias. An. Sist. Sanit. Navar. 2010, 33, 55–68. [Google Scholar] [CrossRef][Green Version]
- Bullard, M.J.; Musgrave, E.; Warren, D.; Unger, B.; Skeldon, T.; Grierson, R.; van der Linde, E.; Swain, J. Revisions to the Canadian Emergency Department Triage and Acuity Scale (CTAS) Guidelines 2016. CJEM 2017, 19, S18–S27. [Google Scholar] [CrossRef]
- Robertson-Steel, I. Evolution of triage systems. Emerg. Med. J. 2006, 23, 154–155. [Google Scholar] [CrossRef]
- Joint Commission International. Communicating Clearly and Effectively to Patients How to Overcome Common Communication Challenges in Health Care. 2018. Available online: https://store.jointcommissioninternational.org/assets/3/7/jci-wp-communicating-clearly-final_(1).pdf (accessed on 30 August 2022).
- Allar, B.; Ortega, G. Our Health System Is Failing Patients with Limited English—Scientific American. Scientific American. 2021. Available online: https://www.scientificamerican.com/article/our-health-system-is-failing-patients-with-limited-english/ (accessed on 29 August 2022).
- Allen, S. 2020 Global Health Care Outlook. Laying a Foundation for the Future. Deloitte Insights. 2020. Available online: https://www2.deloitte.com/content/dam/insights/us/articles/GLOB22843-Global-HC-Outlook/DI-Global-HC-Outlook-Report.pdf (accessed on 23 January 2020).
- Kirk, A. One in Four Self-Diagnose on the Internet Instead of Visiting the Doctor. 2015. Available online: https://www.telegraph.co.uk/news/health/news/11760658/One-in-four-self-diagnose-on-the-internet-instead-of-visiting-the-doctor.html (accessed on 29 August 2022).
- Brucher, L.; Pontet, K.; Duprel, C.; Mosel, K.; Moujahid, S.; Georges, A. A Journey towards Smart Health: The Impact of Digitalization on Patient Experience. 2018. Available online: https://www2.deloitte.com/content/dam/Deloitte/lu/Documents/life-sciences-health-care/lu_journey-smart-health-digitalisation.pdf (accessed on 13 January 2020).
- Meskó, B.; Dhunnoo, P. Digital Health and the Fight against the COVID-19 Pandemic. 2020. Available online: https://www.matrc.org/wp-content/uploads/2020/04/Digital-Health-and-COVID19.pdf (accessed on 29 August 2022).
- Mehrotra, A.; Ray, K.; Brockmeyer, D.M.; Barnett, M.L.; Bender, J.A. Rapidly Converting to “Virtual Practices”: Outpatient Care in the Era of COVID-19. 2020. Available online: https://catalyst.nejm.org/doi/full/10.1056/CAT.20.0091 (accessed on 29 August 2022).
- Guo, C.; Ashrafian, H.; Ghafur, S.; Fontana, G.; Gardner, C.; Prime, M. Challenges for the evaluation of digital health solutions—A call for innovative evidence generation approaches. NPJ Digit. Med. 2020, 3, 110. [Google Scholar] [CrossRef]
- Greaves, F.; Joshi, I.; Campbell, M.; Roberts, S.; Patel, N.; Powell, J. What is an Appropriate Level of Evidence for a Digital Health Intervention? Lancet 2018, 392, 2665–2667. [Google Scholar] [CrossRef]
- Chambers, D.; Cantrell, A.J.; Johnson, M.; Preston, L.; Baxter, S.K.; Booth, A.; Turner, J. Digital and online Symptom Checkers and Health Assessment/Triage Services for Urgent Health Problems: Systematic Review. BMJ Open 2019, 9, e027743. [Google Scholar] [CrossRef]
- Atluri, V.; Rao, S.; Rajah, T.; Schneider, J.; Thibaut, M.; Varanasi, S.; Velamoor, S. Unlocking Digital Health: Opportunities for the Mobile Value Chain; McKinsey & Company: Atlanta, GA, USA, 2015; p. 8. Available online: https://www.mckinsey.com/industries/healthcare-systems-and-services/our-insights/unlocking-digital-health-opportunities-for-the-mobile-value-chain (accessed on 9 December 2019).
- Yu, S.W.Y.; Ma, A.; Tsang, V.H.M.; Chung, L.S.W.; Leung, S.C.; Leung, L.P. Triage Accuracy of Online Symptom Checkers for Accident and Emergency Department Patients. Hong Kong J. Emerg. Med. 2019, 27, 217–222. [Google Scholar] [CrossRef]
- Verzantvoort, N.C.M.; Teunis, T.; Verheij, T.J.M.; van der Velden, A.W. Self-triage for acute primary care via a smartphone application: Practical, safe and efficient? PLoS ONE 2018, 13, e0199284. [Google Scholar] [CrossRef]
- Christensen, C.M.; Bohmer, R.M.J.; Kenagy, J. Will Disruptive Innovations Cure Health Care? Harv. Bus. Rev. 2000, 78, 102–112. [Google Scholar]
- European Union Medical Device Regulation. The European Union Medical Device Regulation—Regulation (EU) 2017/745 (EU MDR). 2022. Available online: https://eumdr.com/ (accessed on 19 August 2022).
- O’Rourke, B.; Oortwijn, W.; Schuller, T. The New Definition of Health Technology Assessment: A Milestone in International Collaboration. Int. J. Technol. Assess. Health Care 2020, 36, 187–190. [Google Scholar] [CrossRef]
- EUnetHTA. HTA Core Model. 2016. Available online: https://www.eunethta.eu/hta-core-model/ (accessed on 7 September 2022).
- Millenson, M.L.; Baldwin, J.L.; Zipperer, L.; Singh, H. Beyond Dr. Google: The Evidence on Consumer-Facing Digital Tools for Diagnosis. Diagnosis 2018, 5, 95–105. [Google Scholar] [CrossRef]
- Sole, M.L.; Stuart, P.L.; Deichen, M. Web-Based Triage in a College Health Setting. J. Am. Coll. Health 2010, 54, 289–294. [Google Scholar] [CrossRef]
- Salisbury, C.; Procter, S.; Stewart, K.; Bowen, L.; Purdy, S.; Ridd, M.; Valderas, J.; Blakeman, T.; Reeves, D. The Content of General Practice Consultations: Cross-Sectional Study based on Video Recordings. Br. J. Gen. Pract. 2013, 63, e751–e759. [Google Scholar] [CrossRef] [PubMed]
- Duckitt, R.; Palsson, R.; Bosanska, L.; Dagna, L.; Mine, D.T.; Vardi, M. Common diagnoses in Internal Medicine in Europe 2009: A pan-European, Multi-Centre Survey. Eur. J. Intern. Med. 2010, 21, 449–452. [Google Scholar]
- Walls, R.M.; Hockberger, R.S.; Gausche-Hill, M. Rosen’s Emergency Medicine: Concepts and Clinical Practice, 10th ed.; Elsevier: Amsterdam, The Netherlands, 2022; 148p. [Google Scholar]
- Stern, S.D.C.; Cifu, A.S.; Altkorn, D. Symptom to Diagnosis: An Evidence-Based Guide, 4th ed.; McGraw Hill Medical: New York, NY, USA, 2019; Available online: https://accessmedicine.mhmedical.com/book.aspx?bookID=2715 (accessed on 18 August 2022).
- Guillén, B.C. Manual de Urgencias, 3rd ed.; Coordinadores y SANED: Madrid, Spain, 2018; pp. 1–1653. [Google Scholar]
- Stanford Anesthesia Cognitive Aid Program. Emergency Manual. 2022. Available online: http://web.stanford.edu/dept/anesthesia/em/SEM_printing.pdf (accessed on 19 August 2022).
- Family Doctor. Symptom Checker. Available online: https://familydoctor.org/your-health-resources/health-tools/symptom-checker/ (accessed on 18 August 2022).
- Simon, C.; O’Reilly, K.; Proctor, R.; Buckmaster, J. Emergencies in Primary Care; Oxford University Press: Oxford, UK, 2007; Available online: https://academic.oup.com/book/31748 (accessed on 18 August 2022).
- Campo, T.M.; Lafferty, K.A. Essential Procedures for Emergency, Urgent, and Primary Care Settings, 3rd ed.; Springer Publishing Company: New York, NY, USA, 2021. [Google Scholar]
- Rare Diseases Registry Program. Single Group Study. Available online: https://registries.ncats.nih.gov/glossary/single-group-study/ (accessed on 6 October 2022).
- Clinicaltrials.gov. Transversal, Controlled and Not Randomized Study, Laying on the Desire of Children and Eating Disorders in a Population of Infertile Women. Available online: https://clinicaltrials.gov/ct2/show/NCT02029365 (accessed on 6 October 2022).
- Health Research Authority. Fast Track Review Guidance for COVID-19 Studies. 2022. Available online: https://www.hra.nhs.uk/covid-19-research/fast-track-review-guidance-covid-19-studies/ (accessed on 30 August 2022).
- Jiménez, J.G. Modelo Andorrano De Triaje (Model Andorrà De Triatge: Mat). 2003. Available online: https://docplayer.es/20789909-Modelo-andorrano-de-triaje-model-model-andorra-de-triatge-mat.html (accessed on 18 August 2022).
- Emergency Nurses Association. Emergency Severity Index: A Triage Tool for Emergency Department Care; Emergency Nurses Association: Schaumburg, IL, USA, 2020. [Google Scholar]
- Carrasco, L.J.; Jover, L. Métodos Estadísticos Para Evaluar la Concordancia. Med. Clin. 2004, 122, 28–34. [Google Scholar] [CrossRef]
- Shankar, V.; Bangdiwala, S.I. Behavior of Agreement Measures in the Presence of Zero Cells and Biased Marginal Distributions. J. Appl. Stat. 2008, 35, 445–464. [Google Scholar] [CrossRef]
- Jansen-Kosterink, S.; Broekhuis, M.; van Velsen, L. Time to Act Mature—Gearing eHealth Evaluations towards Technology Readiness Levels. Digit. Health 2022, 8, 205520762211133. [Google Scholar] [CrossRef] [PubMed]
- Martinez, B.; Hall-Clifford, R.; Coyote, E.; Stroux, L.; Valderrama, C.E.; Aaron, C.; Francis, A.; Hendren, C.; Rohloff, P.; Clifford, G.D. Agile Development of a Smartphone App for Perinatal Monitoring in a Resource-Constrained Setting. J. Health Inform. Dev. Ctries 2017, 11. Available online: http://www.jhidc.org/index.php/jhidc/article/view/158/212 (accessed on 30 August 2022).
- StataCorp. Stata Statistical Software: Release 13; StataCorp LP: College Station, TX, USA, 2013. [Google Scholar]
- IPA4SME Consortium. IPA4SME. 2019. Available online: https://www.ipa4sme.eu/ (accessed on 30 August 2022).
- IBM. What is Supervised Learning? IBM Cloud Education. 2020. Available online: https://www.ibm.com/cloud/learn/supervised-learning (accessed on 31 August 2022).
- Christensen, C.; Waldeck, A.; Fogg, R. How Disruptive Innovation Can Finally Revolutionize Healthcare: A Plan for Incumbents and Startups to Build a Future of Better Health and Lower Costs. Industry Horizons. 2017. Available online: https://www.christenseninstitute.org/wp-content/uploads/2017/05/How-Disruption-Can-Finally-Revolutionize-Healthcare-final.pdf (accessed on 29 August 2022).
- Statista. Smartphone Subscriptions Worldwide 2027. 2022. Available online: https://www.statista.com/statistics/330695/number-of-smartphone-users-worldwide/ (accessed on 7 September 2022).
- Miller, J. Checking Up on Symptom Checkers. Harvard Medical School. 2015. Available online: https://hms.harvard.edu/news/checking-symptom-checkers (accessed on 29 August 2022).
- Ipsos, M.; York Health Economics Consortium; Salisbury, C. Evaluation of Babylon GP at Hand Final Evaluation Report. 2019. Available online: http://www.ipsos-mori.com/terms (accessed on 29 August 2022).
- Wallace, W.; Chan, C.; Chidambaram, S.; Hanna, L.; Iqbal, F.M.; Acharya, A.; Normahani, P.; Ashrafian, H.; Markar, S.R.; Sounderajah, V.; et al. The Diagnostic and Triage Accuracy of Digital and Online Symptom Checker Tools: A Systematic Review. NPJ Digit. Med. 2022, 5, 1–9. [Google Scholar] [CrossRef]
- Ceney, A.; Tolond, S.; Glowinski, A.; Marks, B.; Swift, S.; Palser, T. Accuracy of Online Symptom Checkers and the Potential Impact on Service Utilisation. PLoS ONE 2021, 16, e0254088. [Google Scholar] [CrossRef]
- Eisenhauer, T. Top 10 Business Communication Trends for 2022—Axero Solutions. Available online: https://axerosolutions.com/blog/top-10-business-communication-trends-improve-communication-in-the-workplace (accessed on 24 August 2022).
- Statista. Video Games—Worldwide|Statista Market Forecast. 2022. Available online: https://www.statista.com/outlook/dmo/digital-media/video-games/worldwide (accessed on 30 August 2022).
- Cheung, M.M.Y.; Saini, B.; Smith, L. Using drawings to explore patients’ perceptions of their illness: A scoping review. J. Multidiscip. Healthc. 2016, 9, 631. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5125999/ (accessed on 6 October 2022). [CrossRef]
- Delamerced, A. The Importance of Medical Illustration in Patient Communication. In-Training. 2018. Available online: https://in-training.org/importance-medical-illustration-patient-communication-15851 (accessed on 6 October 2022).
- Deloitte Centre for Health Solutions. Connected Health—How Digital Technology Is Transforming Health and Social Care. Deloitte. 2015, p. 40. Available online: https://www2.deloitte.com/content/dam/Deloitte/uk/Documents/life-sciences-health-care/deloitte-uk-connected-health.pdf (accessed on 5 December 2019).
- Moshi, M.R.; Parsons, J.; Tooher, R.; Merlin, T. Evaluation of Mobile Health Applications: Is Regulatory Policy Up to the Challenge? Int. J. Technol. Assess Health Care 2019, 35, 351–360. [Google Scholar] [CrossRef]
- Moshi, M.R.; Tooher, R.; Merlin, T. Suitability of current evaluation frameworks for use in the health technology assessment of mobile medical applications: A systematic review. Int. J. Technol. Assess Health Care 2018, 34, 464–475. [Google Scholar] [CrossRef]
- Vis, C.; Bührmann, L.; Riper, H.; Ossebaard, H.C. Health technology assessment frameworks for eHealth: A systematic review. Int. J. Technol. Assess Health Care 2020, 36, 204–216. [Google Scholar] [CrossRef]
- Principles behind the Agile Manifesto. Available online: http://agilemanifesto.org/principles.html (accessed on 30 August 2022).
- Oortwijn, W.; Sampietro-Colom, L.; Trowman, R. How to Deal with the Inevitable: Generating Real-World Data and Using Real-World Evidence for HTA Purposes—From Theory to Action. Int. J. Technol. Assess Health Care 2019, 35, 346–350. [Google Scholar] [CrossRef]
- Gwet, K. Kappa Statistic Is Not Satisfactory for Assessing the Extent of Agreement between Raters. Statistical Methods for Inter-Rater Reliability Assessment. 2002, pp. 1–16. Available online: https://www.scirp.org/(S(lz5mqp453edsnp55rrgjct55.))/reference/referencespapers.aspx?referenceid=2208179 (accessed on 17 August 2022).
- Shankar, V.; Bangdiwala, S.I. Observer agreement paradoxes in 2 × 2 tables: Comparison of agreement measures. BMC Med. Res. Methodol. 2014, 14, 100. [Google Scholar] [CrossRef]
- Delgado, R.; Tibau, X.A. Why Cohen’s Kappa should be avoided as performance measure in classification. PLoS ONE 2019, 14, e0222916. [Google Scholar] [CrossRef]
- Dettori, J.R.; Norvell, D.C. Kappa and Beyond: Is There Agreement? Glob. Spine J. 2020, 10, 499–501. [Google Scholar] [CrossRef]
- Semigran, H.L.; Linder, J.A.; Gidengil, C.; Mehrotra, A. Evaluation of symptom checkers for self diagnosis and triage: Audit study. BMJ 2015, 351, h3480. [Google Scholar] [CrossRef]
- IQVIA. Digital Health Trends. 2021. Available online: https://www.iqvia.com/insights/the-iqvia-institute/reports/digital-health-trends-2021 (accessed on 30 August 2022).
- EIT Health Germany. DiGAs—A Model for Europe? Possible Options for Achieving a European System. 2021. Available online: https://eithealth.eu/wp-content/uploads/2022/03/The-DiGAs-framework-a-model-for-Europe.pdf (accessed on 29 August 2022).
- Jungman, S.; Kraft, D. European Healthtech Needs Stronger Medicine Than Germany’s Weak Digital Law. 2022. Available online: https://sifted.eu/articles/europe-germany-digital-health/ (accessed on 30 August 2022).
- MedTech Europe. Recognising the Value of Digital Health Apps: An Assessment of Five European Healthcare Systems. 2021. Available online: www.medtecheurope.orgwww.medtecheurope.org (accessed on 30 August 2022).
- National Institute for Health and Care Excellence. Evidence Standards Framework for Digital Health Technologies Contents; National Institute for Health and Care Excellence: London, UK, 2019. [Google Scholar]
- Zane, R.D.; Wiler, J.L. Embracing Technology to Save Primary Care. N. Engl. J. Med. Catal. 2018. Available online: https://catalyst.nejm.org/doi/full/10.1056/CAT.18.0101 (accessed on 30 August 2022).
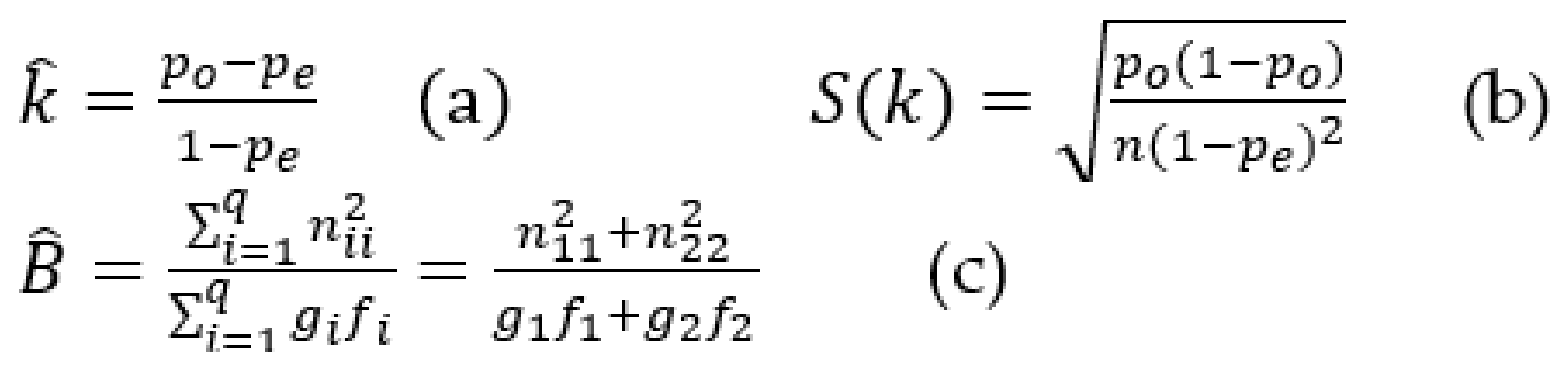
| WtsWrng | ||||
|---|---|---|---|---|
| NO | YES | Total | ||
| Hospital | NO | |||
| YES | ||||
| Total | ||||
| n (%) | |
|---|---|
| Gender | |
| Female | 179 (47) |
| Male | 203 (53) |
| Age | |
| 18–29 | 43 (11) |
| 30–39 | 37 (10) |
| 40–49 | 35 (9) |
| 50–59 | 67 (18) |
| 60–69 | 78 (20) |
| 70–79 | 80 (21) |
| 80–99 | 42 (11) |
| Nationality | |
| Spain | 312 (82) |
| Other European countries | 14 (4) |
| Latin American countries | 47 (12) |
| Others | 9 (2) |
| Employment status | |
| Housekeeper | 2 (1) |
| Self-employed | 6 (2) |
| Student | 13 (3) |
| Employed | 128 (34) |
| Unemployed | 37 (10) |
| Sick leave | 19 (5) |
| Retired | 177 (46) |
| Higher education | |
| Without studies | 9 (2) |
| Primary school | 73 (19) |
| Secondary school | 34 (9) |
| High school | 135 (35) |
| Bachelor’s degree | 103 (27) |
| Master’s degree | 18 (5) |
| PhD | 10 (3) |
| User of technologies (Mobile, Tablet, etc.) | |
| YES | 360 (94) |
| NO | 22 (6) |
| Use of DA | |
| YES | 248 (65) |
| NO | 134 (35) |
| Use of health DA | |
| YES | 165 (43) |
| NO | 217 (57) |
| Sens. | Spec. | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Chronic kidney diseases | 371 | 10 | 1 | 0 | 381 | 1 | 372 | 10 | 0.97 | 0.00 | 0.97 | 0.000 | 0.997 |
| Lung cancer | 368 | 5 | 7 | 2 | 373 | 9 | 375 | 7 | 0.97 | 0.23 | 0.97 | 0.286 | 0.981 |
| Cellulitis | 366 | 3 | 10 | 3 | 369 | 13 | 376 | 6 | 0.97 | 0.30 | 0.97 | 0.500 | 0.973 |
| Gastroenteritis | 354 | 5 | 16 | 7 | 359 | 23 | 370 | 12 | 0.95 | 0.37 | 0.94 | 0.583 | 0.957 |
| Heart failure | 343 | 21 | 16 | 2 | 364 | 18 | 359 | 23 | 0.9 | 0.05 | 0.90 | 0.087 | 0.955 |
| Urinary infection | 332 | 3 | 40 | 7 | 372 | 10 | 335 | 47 | 0.89 | 0.21 | 0.88 | 0.700 | 0.892 |
| Asthma | 333 | 4 | 42 | 3 | 337 | 45 | 375 | 7 | 0.88 | 0.09 | 0.88 | 0.429 | 0.888 |
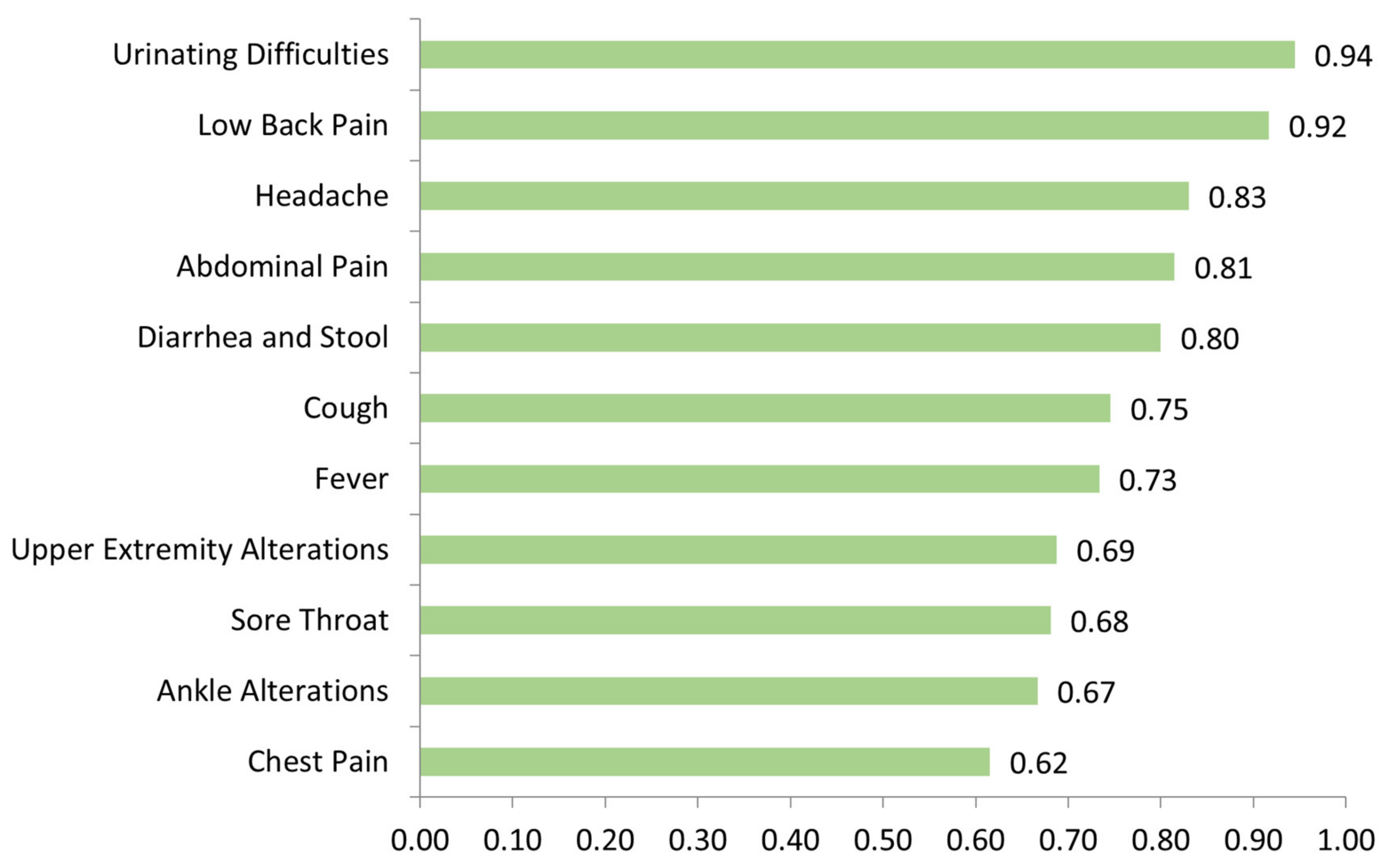
| Low back pain | 309 | 2 | 66 | 5 | 311 | 71 | 375 | 7 | 0.82 | 0.10 | 0.82 | 0.714 | 0.824 |
| Renal colic | 292 | 3 | 80 | 7 | 295 | 87 | 372 | 10 | 0.78 | 0.10 | 0.77 | 0.700 | 0.785 |
| COVID-19 | 136 | 10 | 162 | 74 | 146 | 236 | 298 | 84 | 0.55 | 0.20 | 0.38 | 0.881 | 0.456 |
| Other respiratory disorders | 144 | 2 | 227 | 9 | 146 | 236 | 371 | 11 | 0.4 | 0.02 | 0.37 | 0.818 | 0.388 |
| COPD | 141 | 1 | 225 | 15 | 142 | 240 | 366 | 16 | 0.41 | 0.04 | 0.36 | 0.938 | 0.385 |
| Results (N = 382) | |
|---|---|
| Observed concordance () | 0.4–0.97 (0.8 in 8 diagnostics) * |
| Kappa | −0.005–0.37 |
| B statistic | 0.36–0.97 (0.8 in 8 diagnostics) * |
| Sensitivity | 0.09–0.94 (0.8 in 3 diagnostics) * |
| Specificity | 0.39–0.98 (0.8 in 8 diagnostics) * |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sampietro-Colom, L.; Fernandez-Barcelo, C.; Abbas, I.; Valdasquin, B.; Rabasseda, N.; García-Lorenzo, B.; Sanchez, M.; Sans, M.; Garcia, N.; Granados, A. WtsWrng Interim Comparative Effectiveness Evaluation and Description of the Challenges to Develop, Assess, and Introduce This Novel Digital Application in a Traditional Health System. Int. J. Environ. Res. Public Health 2022, 19, 13873. https://doi.org/10.3390/ijerph192113873
Sampietro-Colom L, Fernandez-Barcelo C, Abbas I, Valdasquin B, Rabasseda N, García-Lorenzo B, Sanchez M, Sans M, Garcia N, Granados A. WtsWrng Interim Comparative Effectiveness Evaluation and Description of the Challenges to Develop, Assess, and Introduce This Novel Digital Application in a Traditional Health System. International Journal of Environmental Research and Public Health. 2022; 19(21):13873. https://doi.org/10.3390/ijerph192113873
Chicago/Turabian StyleSampietro-Colom, Laura, Carla Fernandez-Barcelo, Ismail Abbas, Blanca Valdasquin, Nicolau Rabasseda, Borja García-Lorenzo, Miquel Sanchez, Mireia Sans, Noemi Garcia, and Alicia Granados. 2022. "WtsWrng Interim Comparative Effectiveness Evaluation and Description of the Challenges to Develop, Assess, and Introduce This Novel Digital Application in a Traditional Health System" International Journal of Environmental Research and Public Health 19, no. 21: 13873. https://doi.org/10.3390/ijerph192113873
APA StyleSampietro-Colom, L., Fernandez-Barcelo, C., Abbas, I., Valdasquin, B., Rabasseda, N., García-Lorenzo, B., Sanchez, M., Sans, M., Garcia, N., & Granados, A. (2022). WtsWrng Interim Comparative Effectiveness Evaluation and Description of the Challenges to Develop, Assess, and Introduce This Novel Digital Application in a Traditional Health System. International Journal of Environmental Research and Public Health, 19(21), 13873. https://doi.org/10.3390/ijerph192113873





