Full-Face Mask Use during SCUBA Diving Counters Related Oxidative Stress and Endothelial Dysfunction
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Protocol
2.2. Flow Mediated Dilation (FMD)
2.3. Venous Blood Samples and Urine
2.4. Blood Sample Analysis
2.4.1. ROS Production
2.4.2. 8-Isoprostane
2.4.3. Nitric Oxide Metabolites (NOx)
2.4.4. Inducible Nitric Oxide Synthase (iNOS)
2.4.5. Nitrotyrosine (3-NT)
2.5. Statistical Analysis
3. Results
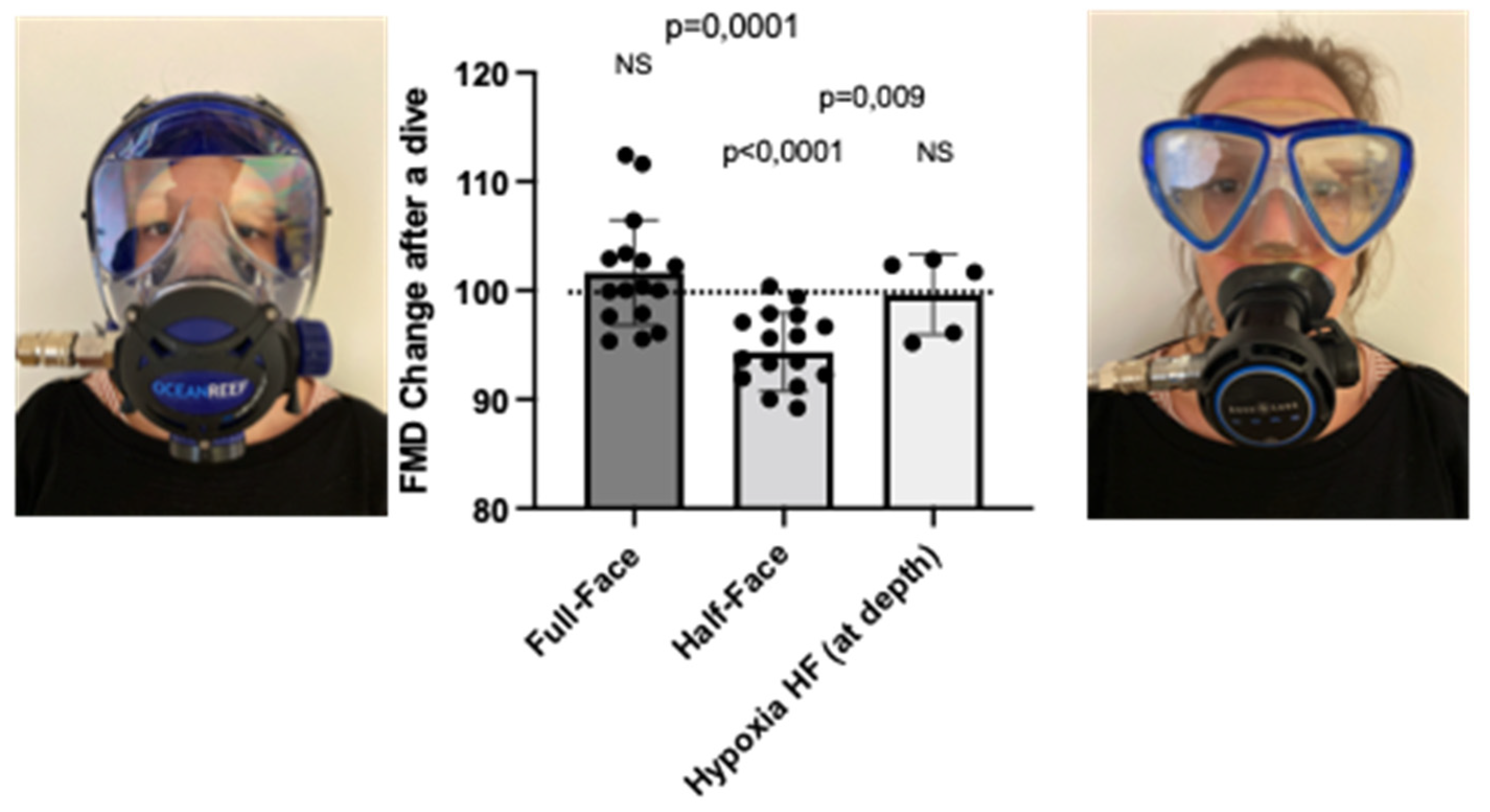
3.1. Nasal–Oral Breathing by Wearing a Full-Face Mask and Breathing a Hypoxic Air Mixture Prevents FMD Reduction after a Shallow Dive, in Comparison to a Half-Face Mask
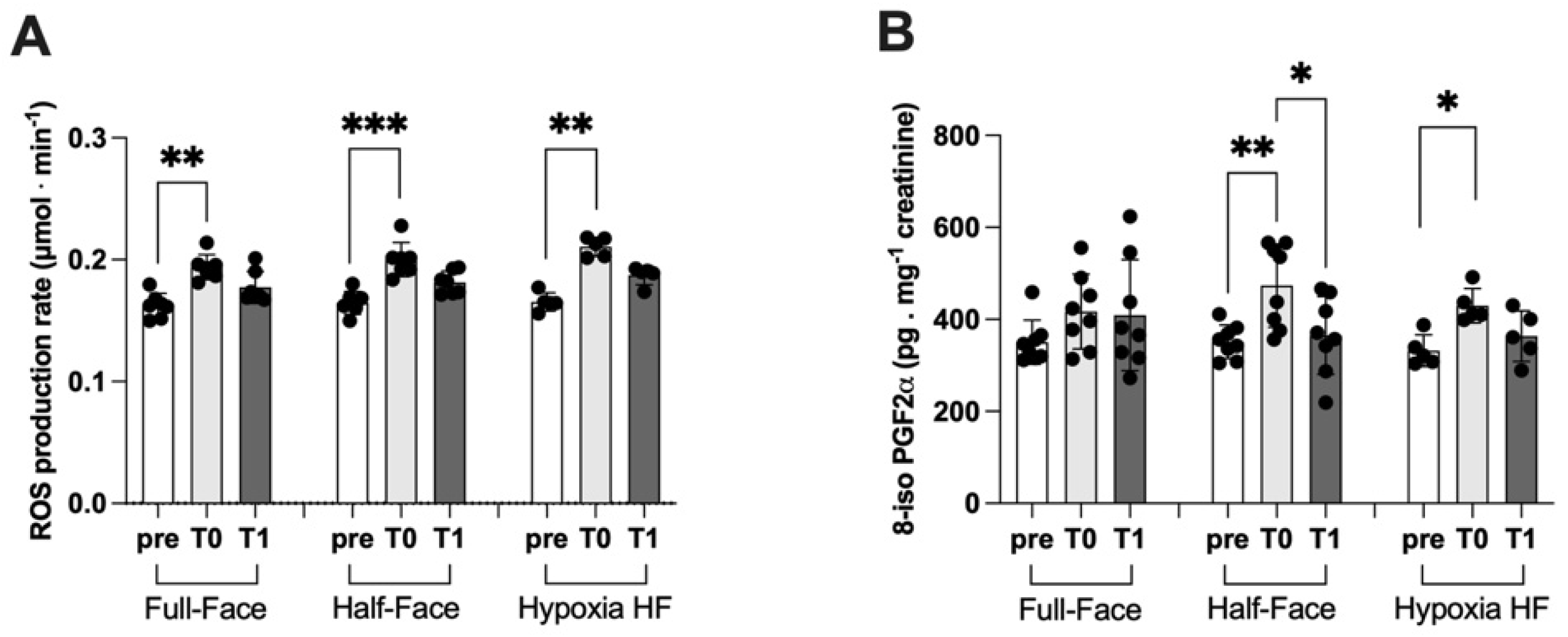
3.2. ROS Production and Lipid Peroxidation
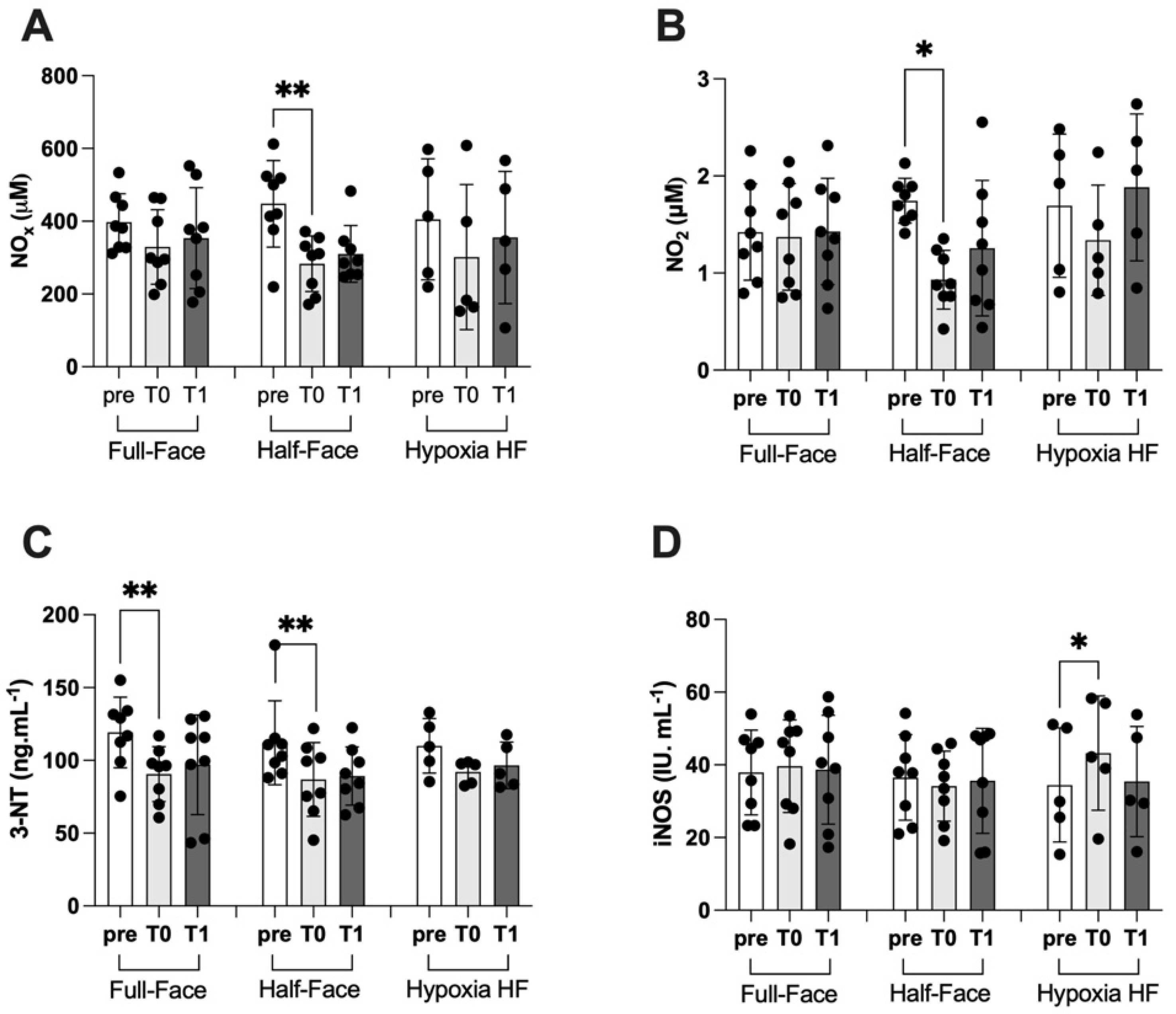
3.3. Nitric Oxide Synthase and Nitric Oxide Metabolites
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| EPO | Erythropoietin |
| EPR | Electron paramagnetic resonance |
| FMD | Flow mediated dilation |
| GSH | Intracellular reduced glutathione |
| HPLC | High-performance liquid chromatography |
| iNOS | Inducible nitric oxide synthase |
| 8-iso PGF2-a | 8-Isoprostane |
| MPPs | Matrix metallopeptidases |
| NF-κB | Nuclear factor kappa-light-chain-enhancer of activated B cells |
| NRF2 | Nuclear factor (erythroid-derived 2)-like 2 |
| NO | Nitric oxide |
| NO2 | Nitrite |
| NOx | NO metabolites (NO2+NO3) |
| 3-NT | Nitrotyrosine |
| PO2 | Oxygen partial pressure |
| ROS | Reactive oxygen species |
References
- Papadopoulou, V.; Tang, M.X.; Balestra, C.; Eckersley, R.J.; Karapantsios, T.D. Circulatory bubble dynamics: From physical to biological aspects. Adv. Colloid Interface Sci. 2014, 206, 239–249. [Google Scholar] [CrossRef] [PubMed]
- Lambrechts, K.; Pontier, J.M.; Balestra, C.; Mazur, A.; Wang, Q.; Buzzacott, P.; Theron, M.; Mansourati, J.; Guerrero, F. Effect of a single, open-sea, air scuba dive on human micro- and macrovascular function. Eur. J. Appl. Physiol. 2013, 113, 2637–2645. [Google Scholar] [CrossRef]
- Marinovic, J.; Ljubkovic, M.; Breskovic, T.; Gunjaca, G.; Obad, A.; Modun, D.; Bilopavlovic, N.; Tsikas, D.; Dujic, Z. Effects of successive air and nitrox dives on human vascular function. Eur. J. Appl. Physiol. 2012, 112, 2131–2137. [Google Scholar] [CrossRef] [PubMed]
- Brubakk, A.O.; Duplancic, D.; Valic, Z.; Palada, I.; Obad, A.; Bakovic, D.; Wisloff, U.; Dujic, Z. A single air dive reduces arterial endothelial function in man. J. Physiol. 2005, 566, 901–906. [Google Scholar] [CrossRef] [PubMed]
- Lambrechts, K.; Pontier, J.M.; Mazur, A.; Buzzacott, P.; Morin, J.; Wang, Q.; Theron, M.; Guerrero, F. Effect of decompression-induced bubble formation on highly trained divers microvascular function. Physiol. Rep. 2013, 1, e00142. [Google Scholar] [CrossRef] [PubMed]
- Obad, A.; Valic, Z.; Palada, I.; Brubakk, A.O.; Modun, D.; Dujic, Z. Antioxidant pretreatment and reduced arterial endothelial dysfunction after diving. Aviat. Space Environ. Med. 2007, 78, 1114–1120. [Google Scholar] [CrossRef]
- Obad, A.; Palada, I.; Valic, Z.; Ivancev, V.; Bakovic, D.; Wisloff, U.; Brubakk, A.O.; Dujic, Z. The effects of acute oral antioxidants on diving-induced alterations in human cardiovascular function. J. Physiol. 2007, 578, 859–870. [Google Scholar] [CrossRef] [PubMed]
- Balestra, C.; Cimino, F.; Theunissen, S.; Snoeck, T.; Provyn, S.; Canali, R.; Bonina, A.; Virgili, F. A red orange extract modulates the vascular response to a recreational dive: A pilot study on the effect of anthocyanins on the physiological consequences of scuba diving. Nat. Prod. Res. 2016, 30, 2101–2106. [Google Scholar] [CrossRef]
- Theunissen, S.; Guerrero, F.; Sponsiello, N.; Cialoni, D.; Pieri, M.; Germonpre, P.; Obeid, G.; Tillmans, F.; Papadopoulou, V.; Hemelryck, W.; et al. Nitric oxide-related endothelial changes in breath-hold and scuba divers. Undersea Hyperb. Med. 2013, 40, 135–144. [Google Scholar]
- Theunissen, S.; Schumacker, J.; Guerrero, F.; Tillmans, F.; Boutros, A.; Lambrechts, K.; Mazur, A.; Pieri, M.; Germonpre, P.; Balestra, C. Dark chocolate reduces endothelial dysfunction after successive breath-hold dives in cool water. Eur. J. Appl. Physiol. 2013, 113, 2967–2975. [Google Scholar] [CrossRef] [PubMed]
- Bosco, G.; Paganini, M.; Giacon, T.A.; Oppio, A.; Vezzoli, A.; Dellanoce, C.; Moro, T.; Paoli, A.; Zanotti, F.; Zavan, B.; et al. Oxidative Stress and Inflammation, MicroRNA, and Hemoglobin Variations after Administration of Oxygen at Different Pressures and Concentrations: A Randomized Trial. Int. J. Environ. Res. Public Health 2021, 18, 9755. [Google Scholar] [CrossRef]
- Nossum, V.; Hjelde, A.; Brubakk, A.O. Small amounts of venous gas embolism cause delayed impairment of endothelial function and increase polymorphonuclear neutrophil infiltration. Eur. J. Appl. Physiol. 2002, 86, 209–214. [Google Scholar] [CrossRef] [PubMed]
- Sobolewski, P.; Kandel, J.; Klinger, A.L.; Eckmann, D.M. Air bubble contact with endothelial cells in vitro induces calcium influx and IP3-dependent release of calcium stores. Am. J. Physiol. Cell Physiol. 2011, 301, C679–C686. [Google Scholar] [CrossRef] [PubMed]
- Klinger, A.L.; Pichette, B.; Sobolewski, P.; Eckmann, D.M. Mechanotransductional basis of endothelial cell response to intravascular bubbles. Integr. Biol. 2011, 3, 1033–1042. [Google Scholar] [CrossRef]
- Lambrechts, K.; Pontier, J.M.; Mazur, A.; Theron, M.; Buzzacott, P.; Wang, Q.; Belhomme, M.; Guerrero, F. Mechanism of action of antiplatelet drugs on decompression sickness in rats: A protective effect of anti-GPIIbIIIa therapy. J. Appl. Physiol. 2015, 118, 1234–1239. [Google Scholar] [CrossRef]
- Pontier, J.M.; Vallee, N.; Bourdon, L. Bubble-induced platelet aggregation in a rat model of decompression sickness. J. Appl. Physiol. 2009, 107, 1825–1829. [Google Scholar] [CrossRef]
- Pontier, J.M.; Blatteau, J.E.; Vallee, N. Blood platelet count and severity of decompression sickness in rats after a provocative dive. Aviat. Space Environ. Med. 2008, 79, 761–764. [Google Scholar] [CrossRef]
- Yang, M.; Barak, O.F.; Dujic, Z.; Madden, D.; Bhopale, V.M.; Bhullar, J.; Thom, S.R. Ascorbic acid supplementation diminishes microparticle elevations and neutrophil activation following SCUBA diving. Am. J. Physiol. 2015, 309, R338–R344. [Google Scholar] [CrossRef]
- Ersson, A.; Walles, M.; Ohlsson, K.; Ekholm, A. Chronic hyperbaric exposure activates proinflammatory mediators in humans. J. Appl. Physiol. 2002, 92, 2375–2380. [Google Scholar] [CrossRef] [PubMed]
- Thom, S.R.; Yang, M.; Bhopale, V.M.; Huang, S.; Milovanova, T.N. Microparticles initiate decompression-induced neutrophil activation and subsequent vascular injuries. J. Appl. Physiol. 2011, 110, 340–351. [Google Scholar] [CrossRef]
- Germonpre, P.; Balestra, C. Preconditioning to Reduce Decompression Stress in Scuba Divers. Aerosp. Med. Hum. Perform. 2017, 88, 114–120. [Google Scholar] [CrossRef]
- Theunissen, S.; Balestra, C.; Boutros, A.; De Bels, D.; Guerrero, F.; Germonpre, P. The effect of pre-dive ingestion of dark chocolate on endothelial function after a scuba dive. Diving Hyperb. Med. 2015, 45, 4–9. [Google Scholar] [PubMed]
- Lundberg, J.O.; Weitzberg, E. Nasal nitric oxide in man. Thorax 1999, 54, 947–952. [Google Scholar] [CrossRef] [PubMed]
- Maniaci, A.; Merlino, F.; Cocuzza, S.; Iannella, G.; Vicini, C.; Cammaroto, G.; Lechien, J.R.; Calvo-Henriquez, C.; La Mantia, I. Endoscopic surgical treatment for rhinogenic contact point headache: Systematic review and meta-analysis. Eur. Arch. Otorhinolaryngol. 2021, 278, 1743–1753. [Google Scholar] [CrossRef] [PubMed]
- Knowles, M.R.; Zariwala, M.; Leigh, M. Primary Ciliary Dyskinesia. Clin. Chest Med. 2016, 37, 449–461. [Google Scholar] [CrossRef] [PubMed]
- World Medical, A. World Medical Association Declaration of Helsinki: Ethical principles for medical research involving human subjects. JAMA 2013, 310, 2191–2194. [Google Scholar] [CrossRef]
- Balestra, C.; Theunissen, S.; Papadopoulou, V.; Le Mener, C.; Germonpre, P.; Guerrero, F.; Lafere, P. Pre-dive Whole-Body Vibration Better Reduces Decompression-Induced Vascular Gas Emboli than Oxygenation or a Combination of Both. Front. Physiol. 2016, 7, 586. [Google Scholar] [CrossRef] [PubMed]
- Pyke, K.E.; Tschakovsky, M.E. The relationship between shear stress and flow-mediated dilatation: Implications for the assessment of endothelial function. J. Physiol. 2005, 568, 357–369. [Google Scholar] [CrossRef] [PubMed]
- Corretti, M.C.; Anderson, T.J.; Benjamin, E.J.; Celermajer, D.; Charbonneau, F.; Creager, M.A.; Deanfield, J.; Drexler, H.; Gerhard-Herman, M.; Herrington, D.; et al. Guidelines for the ultrasound assessment of endothelial-dependent flow-mediated vasodilation of the brachial artery: A report of the International Brachial Artery Reactivity Task Force. J. Am. Coll. Cardiol. 2002, 39, 257–265. [Google Scholar] [CrossRef]
- Areas, G.P.T.; Mazzuco, A.; Caruso, F.R.; Jaenisch, R.B.; Cabiddu, R.; Phillips, S.A.; Arena, R.; Borghi-Silva, A. Flow-mediated dilation and heart failure: A review with implications to physical rehabilitation. Heart Fail. Rev. 2019, 24, 69–80. [Google Scholar] [CrossRef]
- Mrakic-Sposta, S.; Gussoni, M.; Dellanoce, C.; Marzorati, M.; Montorsi, M.; Rasica, L.; Pratali, L.; D’Angelo, G.; Martinelli, M.; Bastiani, L.; et al. Effects of acute and sub-acute hypobaric hypoxia on oxidative stress: A field study in the Alps. Eur. J. Appl. Physiol. 2021, 121, 297–306. [Google Scholar] [CrossRef]
- Mrakic-Sposta, S.; Gussoni, M.; Montorsi, M.; Porcelli, S.; Vezzoli, A. A quantitative method to monitor reactive oxygen species production by electron paramagnetic resonance in physiological and pathological conditions. Oxid. Med. Cell. Longev. 2014, 2014, 306179. [Google Scholar] [CrossRef]
- Mrakic-Sposta, S.; Vezzoli, A.; Rizzato, A.; Della Noce, C.; Malacrida, S.; Montorsi, M.; Paganini, M.; Cancellara, P.; Bosco, G. Oxidative stress assessment in breath-hold diving. Eur. J. Appl. Physiol. 2019, 119, 2449–2456. [Google Scholar] [CrossRef] [PubMed]
- Strapazzon, G.; Malacrida, S.; Vezzoli, A.; Dal Cappello, T.; Falla, M.; Lochner, P.; Moretti, S.; Procter, E.; Brugger, H.; Mrakic-Sposta, S. Oxidative stress response to acute hypobaric hypoxia and its association with indirect measurement of increased intracranial pressure: A field study. Sci. Rep. 2016, 6, 32426. [Google Scholar] [CrossRef] [PubMed]
- Barak, O.F.; Janjic, N.; Drvis, I.; Mijacika, T.; Mudnic, I.; Coombs, G.B.; Thom, S.R.; Madic, D.; Dujic, Z. Vascular dysfunction following breath-hold diving. Can. J. Physiol. Pharm. 2020, 98, 124–130. [Google Scholar] [CrossRef] [PubMed]
- Theunissen, S.; Sponsiello, N.; Rozloznik, M.; Germonpre, P.; Guerrero, F.; Cialoni, D.; Balestra, C. Oxidative stress in breath-hold divers after repetitive dives. Diving Hyperb. Med. 2013, 43, 63–66. [Google Scholar]
- Ciccone, M.M.; Bilianou, E.; Balbarini, A.; Gesualdo, M.; Ghiadoni, L.; Metra, M.; Palmiero, P.; Pedrinelli, R.; Salvetti, M.; Scicchitano, P.; et al. Task force on: ‘Early markers of atherosclerosis: Influence of age and sex’. J. Cardiovasc. Med. 2013, 14, 757–766. [Google Scholar] [CrossRef]
- Moens, A.L.; Goovaerts, I.; Claeys, M.J.; Vrints, C.J. Flow-mediated vasodilation: A diagnostic instrument, or an experimental tool? Chest 2005, 127, 2254–2263. [Google Scholar] [CrossRef]
- Wink, D.A.; Miranda, K.M.; Espey, M.G.; Pluta, R.M.; Hewett, S.J.; Colton, C.; Vitek, M.; Feelisch, M.; Grisham, M.B. Mechanisms of the antioxidant effects of nitric oxide. Antioxid. Redox Signal. 2001, 3, 203–213. [Google Scholar] [CrossRef]
- Craighead, D.H.; Heinbockel, T.C.; Freeberg, K.A.; Rossman, M.J.; Jackman, R.A.; Jankowski, L.R.; Hamilton, M.N.; Ziemba, B.P.; Reisz, J.A.; D’Alessandro, A.; et al. Time-Efficient Inspiratory Muscle Strength Training Lowers Blood Pressure and Improves Endothelial Function, NO Bioavailability, and Oxidative Stress in Midlife/Older Adults With Above-Normal Blood Pressure. J. Am. Heart Assoc. 2021, 10, e020980. [Google Scholar] [CrossRef]
- Nguyen, P.K.; Katikireddy, C.K.; McConnell, M.V.; Kushida, C.; Yang, P.C. Nasal continuous positive airway pressure improves myocardial perfusion reserve and endothelial-dependent vasodilation in patients with obstructive sleep apnea. J. Cardiovasc. Magn. Reson. 2010, 12, 50. [Google Scholar] [CrossRef] [PubMed]
- Balestra, C.; Lambrechts, K.; Mrakic-Sposta, S.; Vezzoli, A.; Levenez, M.; Germonpre, P.; Virgili, F.; Bosco, G.; Lafere, P. Hypoxic and Hyperoxic Breathing as a Complement to Low-Intensity Physical Exercise Programs: A Proof-of-Principle Study. Int. J. Mol. Sci. 2021, 22, 9600. [Google Scholar] [CrossRef]
- Gorlach, A.; Dimova, E.Y.; Petry, A.; Martinez-Ruiz, A.; Hernansanz-Agustin, P.; Rolo, A.P.; Palmeira, C.M.; Kietzmann, T. Reactive oxygen species, nutrition, hypoxia and diseases: Problems solved? Redox Biol. 2015, 6, 372–385. [Google Scholar] [CrossRef]
- Uusijärvi, J.; Eriksson, K.; Larsson, A.C.; Nihlén, C.; Schiffer, T.; Lindholm, P.; Weitzberg, E. Effects of hyperbaric oxygen on nitric oxide generation in humans. Nitric Oxide 2015, 44, 88–97. [Google Scholar] [CrossRef]
- Jiang, Z.L.; Fletcher, N.M.; Diamond, M.P.; Abu-Soud, H.M.; Saed, G.M. Hypoxia regulates iNOS expression in human normal peritoneal and adhesion fibroblasts through nuclear factor kappa B activation mechanism. Fertil. Steril. 2009, 91, 616–621. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Moro, M.A.; De Alba, J.; Leza, J.C.; Lorenzo, P.; Fernández, A.P.; Bentura, M.L.; Boscá, L.; Rodrigo, J.; Lizasoain, I. Neuronal expression of inducible nitric oxide synthase after oxygen and glucose deprivation in rat forebrain slices. Eur. J. Neurosci. 1998, 10, 445–456. [Google Scholar] [CrossRef] [PubMed]
- Ji, L.L.; Gomez-Cabrera, M.C.; Vina, J. Exercise and hormesis: Activation of cellular antioxidant signaling pathway. Ann. N. Y. Acad. Sci. 2006, 1067, 425–435. [Google Scholar] [CrossRef]
- Pierini, D.; Bryan, N.S. Nitric oxide availability as a marker of oxidative stress. Methods Mol. Biol. 2015, 1208, 63–71. [Google Scholar] [CrossRef]
- Fratantonio, D.; Virgili, F.; Zucchi, A.; Lambrechts, K.; Latronico, T.; Lafere, P.; Germonpre, P.; Balestra, C. Increasing Oxygen Partial Pressures Induce a Distinct Transcriptional Response in Human PBMC: A Pilot Study on the “Normobaric Oxygen Paradox”. Int. J. Mol. Sci. 2021, 22, 458. [Google Scholar] [CrossRef]
- Anavi, S.; Tirosh, O. iNOS as a metabolic enzyme under stress conditions. Free Radic. Biol. Med. 2020, 146, 16–35. [Google Scholar] [CrossRef]
- Korbecki, J.; Simińska, D.; Gąssowska-Dobrowolska, M.; Listos, J.; Gutowska, I.; Chlubek, D.; Baranowska-Bosiacka, I. Chronic and Cycling Hypoxia: Drivers of Cancer Chronic Inflammation through HIF-1 and NF-κB Activation: A Review of the Molecular Mechanisms. Int. J. Mol. Sci. 2021, 22, 10701. [Google Scholar] [CrossRef]
- Chao, J.; Guo, Y.; Li, P.; Chao, L. Opposing Effects of Oxygen Regulation on Kallistatin Expression: Kallistatin as a Novel Mediator of Oxygen-Induced HIF-1-eNOS-NO Pathway. Oxid. Med. Cell. Longev. 2017, 2017, 5262958. [Google Scholar] [CrossRef]
- Ohshima, H.; Friesen, M.; Brouet, I.; Bartsch, H. Nitrotyrosine as a new marker for endogenous nitrosation and nitration of proteins. Food Chem. Toxicol. 1990, 28, 647–652. [Google Scholar] [CrossRef]
- Ali, S.S.; Hsiao, M.; Zhao, H.W.; Dugan, L.L.; Haddad, G.G.; Zhou, D. Hypoxia-adaptation involves mitochondrial metabolic depression and decreased ROS leakage. PLoS ONE 2012, 7, e36801. [Google Scholar] [CrossRef]
- Cialoni, D.; Brizzolari, A.; Samaja, M.; Pieri, M.; Marroni, A. Altered Venous Blood Nitric Oxide Levels at Depth and Related Bubble Formation During Scuba Diving. Front. Physiol. 2019, 10, 57. [Google Scholar] [CrossRef]
- Cucchiaro, G.; Tatum, A.H.; Brown, M.C.; Camporesi, E.M.; Daucher, J.W.; Hakim, T.S. Inducible nitric oxide synthase in the lung and exhaled nitric oxide after hyperoxia. Am. J. Physiol. 1999, 277, L636–L644. [Google Scholar] [CrossRef]
- Attaye, I.; Smulders, Y.M.; de Waard, M.C.; Oudemans-van Straaten, H.M.; Smit, B.; Van Wijhe, M.H.; Musters, R.J.; Koolwijk, P.; Spoelstra-de Man, A.M.E. The effects of hyperoxia on microvascular endothelial cell proliferation and production of vaso-active substances. Intensive Care Med. Exp. 2017, 5, 22. [Google Scholar] [CrossRef]
- Zhilyaev, S.Y.; Moskvin, A.N.; Platonova, T.F.; Gutsaeva, D.R.; Churilina, I.V.; Demchenko, I.T. Hyperoxic vasoconstriction in the brain is mediated by inactivation of nitric oxide by superoxide anions. Neurosci. Behav. Physiol. 2003, 33, 783–787. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Levenez, M.; Lambrechts, K.; Mrakic-Sposta, S.; Vezzoli, A.; Germonpré, P.; Pique, H.; Virgili, F.; Bosco, G.; Lafère, P.; Balestra, C. Full-Face Mask Use during SCUBA Diving Counters Related Oxidative Stress and Endothelial Dysfunction. Int. J. Environ. Res. Public Health 2022, 19, 965. https://doi.org/10.3390/ijerph19020965
Levenez M, Lambrechts K, Mrakic-Sposta S, Vezzoli A, Germonpré P, Pique H, Virgili F, Bosco G, Lafère P, Balestra C. Full-Face Mask Use during SCUBA Diving Counters Related Oxidative Stress and Endothelial Dysfunction. International Journal of Environmental Research and Public Health. 2022; 19(2):965. https://doi.org/10.3390/ijerph19020965
Chicago/Turabian StyleLevenez, Morgan, Kate Lambrechts, Simona Mrakic-Sposta, Alessandra Vezzoli, Peter Germonpré, Hadrien Pique, Fabio Virgili, Gerardo Bosco, Pierre Lafère, and Costantino Balestra. 2022. "Full-Face Mask Use during SCUBA Diving Counters Related Oxidative Stress and Endothelial Dysfunction" International Journal of Environmental Research and Public Health 19, no. 2: 965. https://doi.org/10.3390/ijerph19020965
APA StyleLevenez, M., Lambrechts, K., Mrakic-Sposta, S., Vezzoli, A., Germonpré, P., Pique, H., Virgili, F., Bosco, G., Lafère, P., & Balestra, C. (2022). Full-Face Mask Use during SCUBA Diving Counters Related Oxidative Stress and Endothelial Dysfunction. International Journal of Environmental Research and Public Health, 19(2), 965. https://doi.org/10.3390/ijerph19020965









