Microplastics in Sediments of East Surabaya, Indonesia: Regional Characteristics and Potential Risks
Abstract
:1. Introduction
2. Materials and Methods
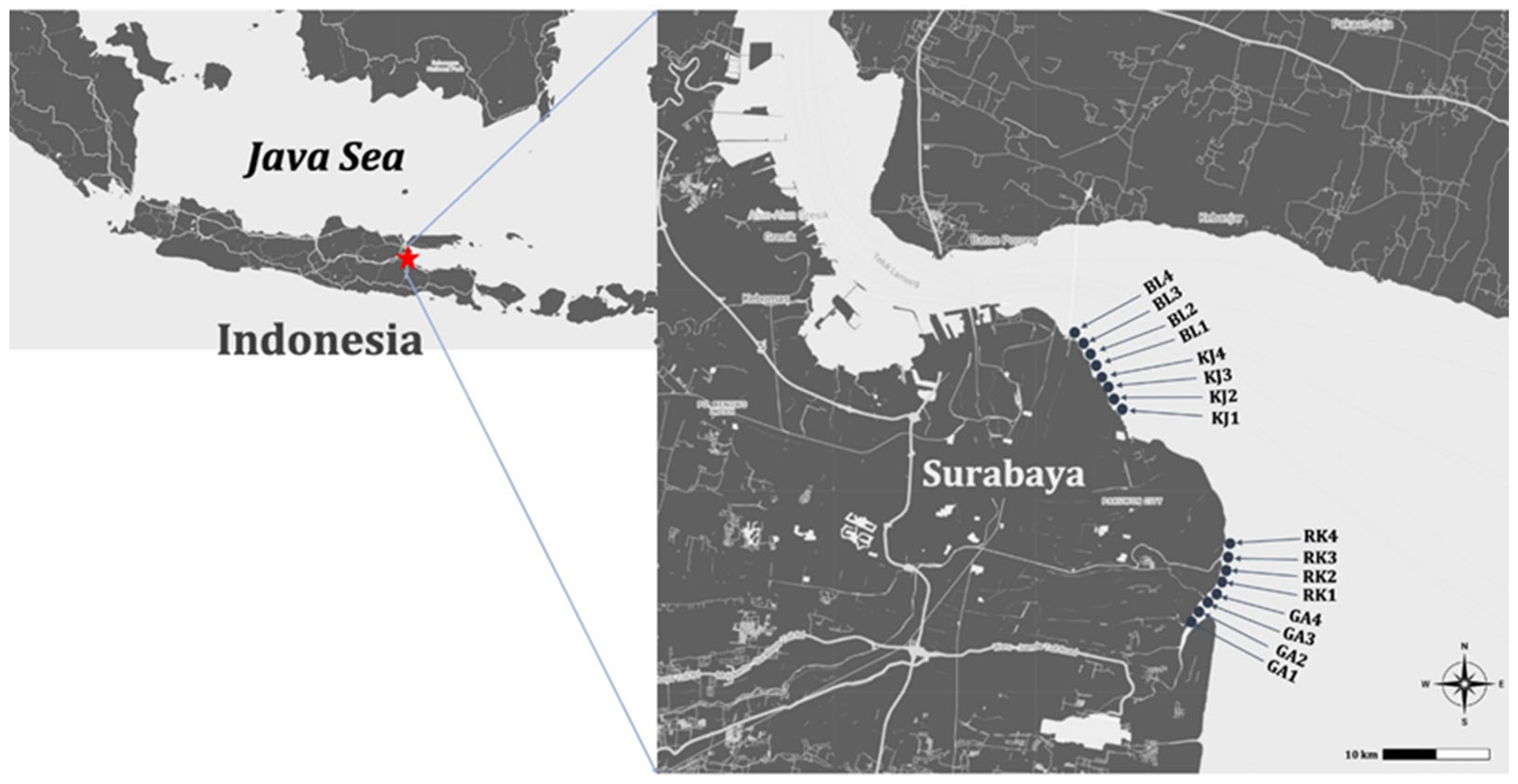
2.1. Study Area and Sample Collection
2.2. Extraction and Identification of Microplastics
2.3. Quality Assurance and Quality Control
2.4. Statistical Analysis
2.5. Risk Assessment
3. Results and Discussion
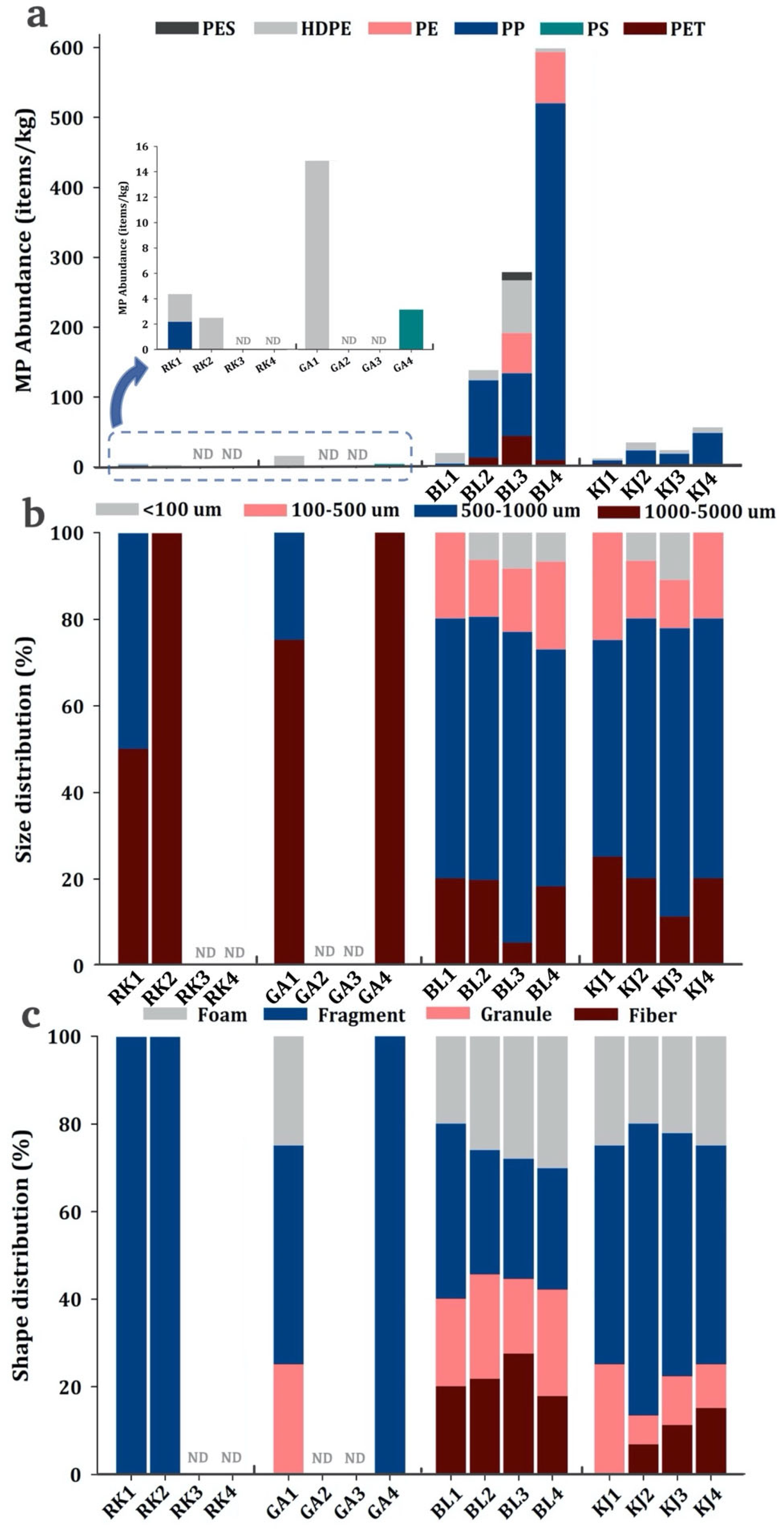
3.1. Occurrence of MPs
3.2. Characteristics of MPs
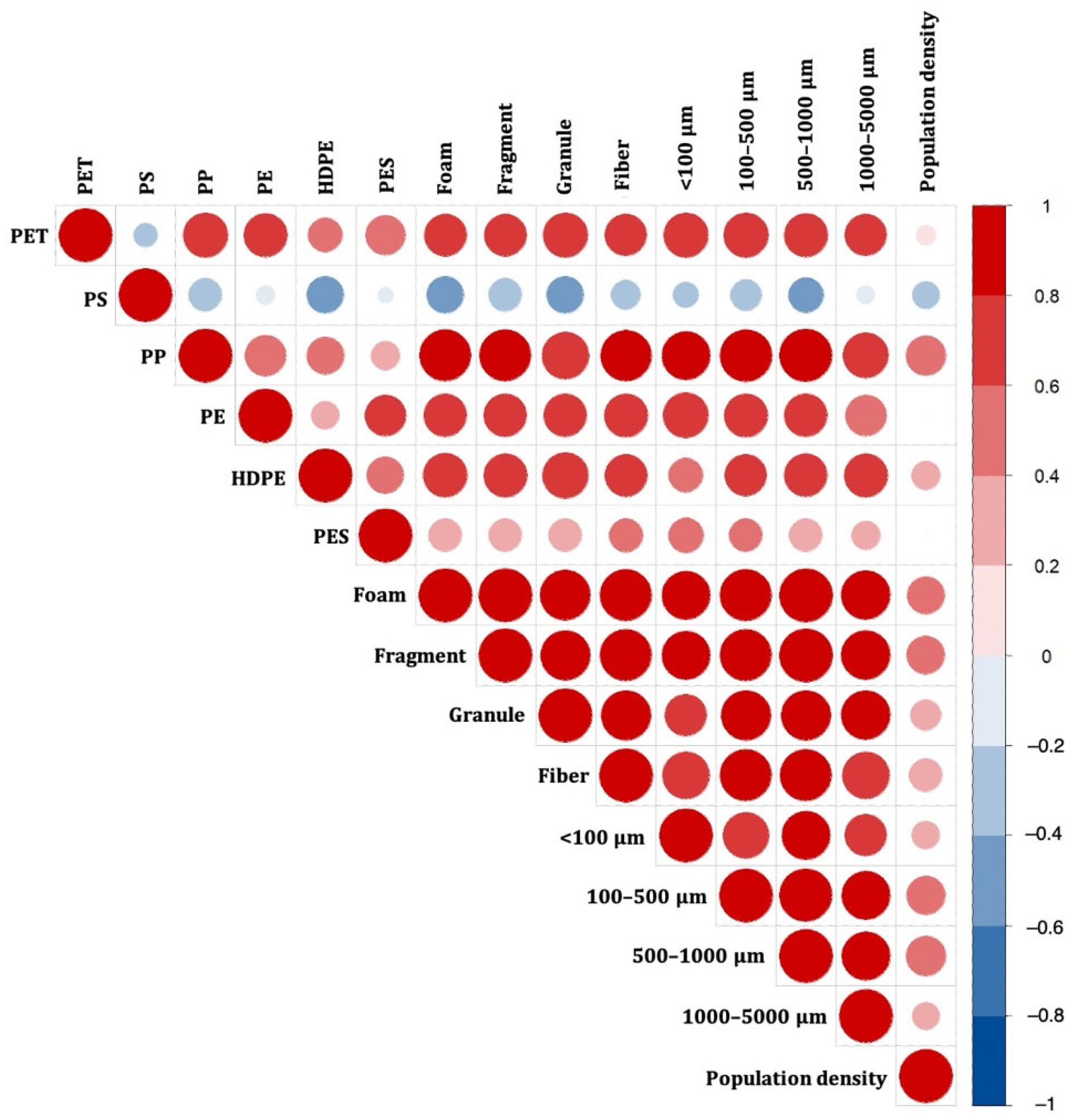
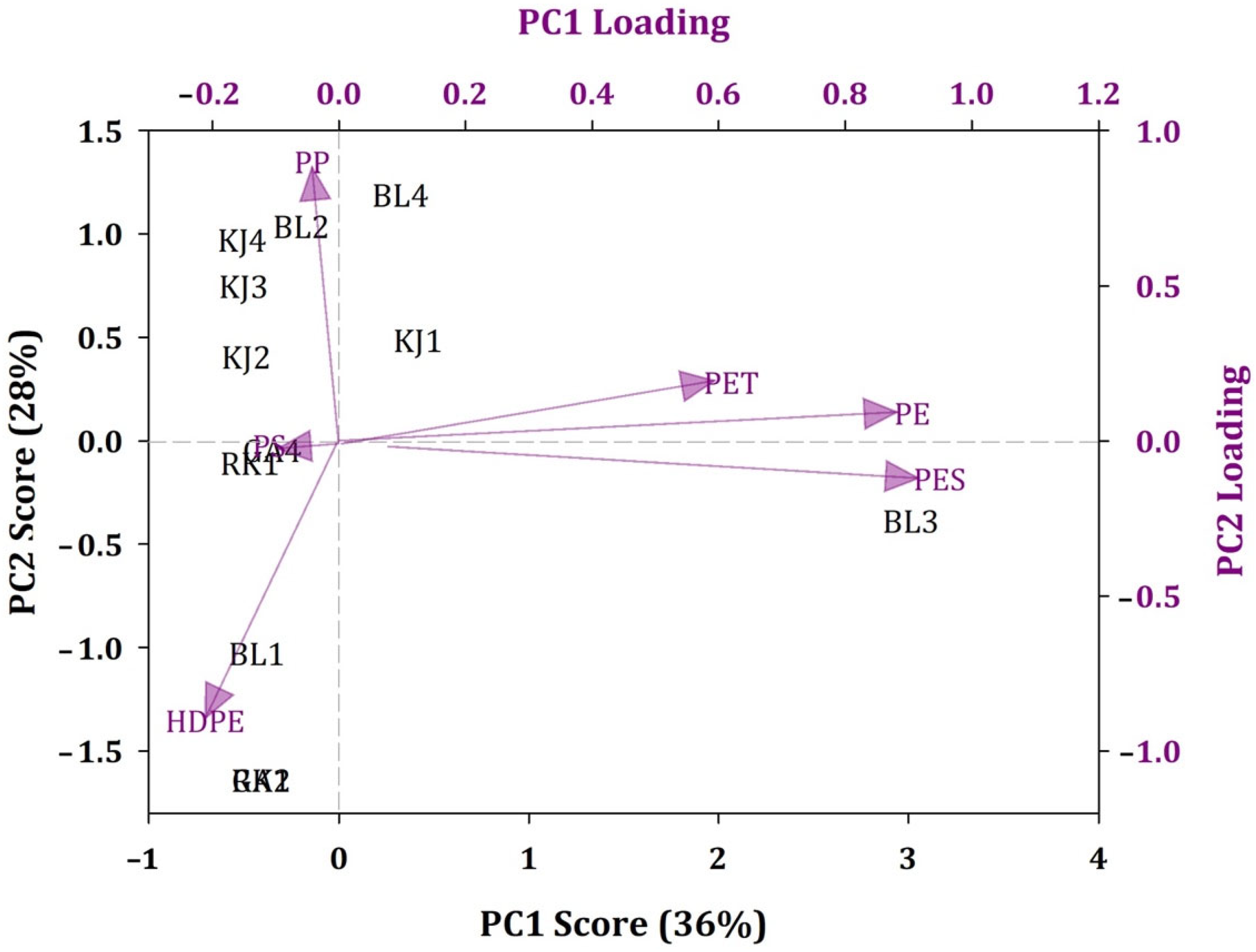
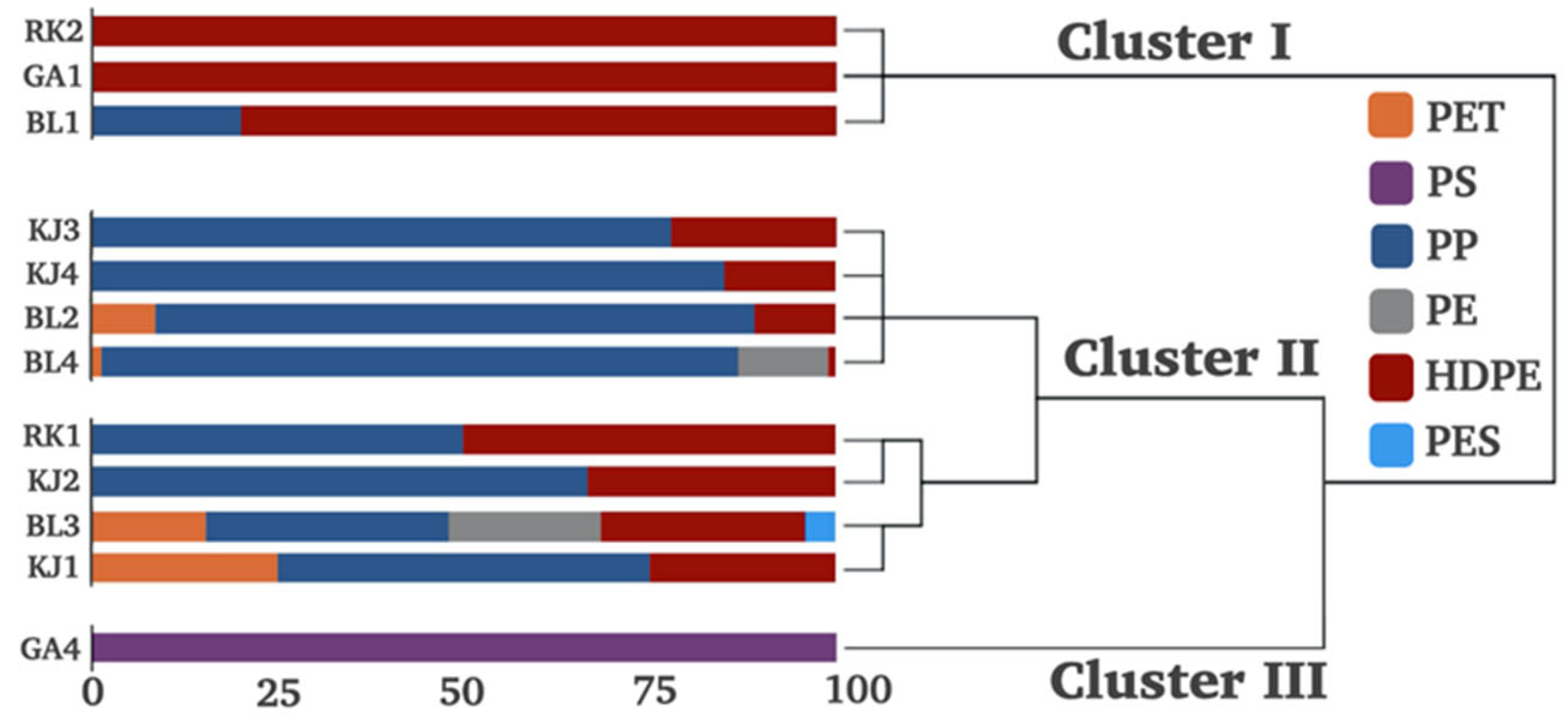
3.3. Composition Patterns and Potential Sources of MPs
3.4. Risk Assessment of MPs
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Europe, P. Plastics—The Facts 2021. 2021. Available online: https://plasticseurope.org/knowledge-hub/plastics-the-facts-2021/ (accessed on 18 July 2022).
- Akdogan, Z.; Guven, B. Microplastics in the environment: A critical review of current understanding and identification of future research needs. Environ. Pollut. 2019, 254, 113011. [Google Scholar] [CrossRef] [PubMed]
- Hanun, J.N.; Hassan, F.; Jiang, J.-J. Occurrence, fate, and sorption behavior of contaminants of emerging concern to microplastics: Influence of the weathering/aging process. J. Environ. Chem. Eng. 2021, 9, 106290. [Google Scholar] [CrossRef]
- Shiu, R.F.; Gong, G.C.; Fang, M.D.; Chow, C.H.; Chin, W.C. Marine microplastics in the surface waters of “pristine” Kuroshio. Mar. Pollut. Bull. 2021, 172, 112808. [Google Scholar] [CrossRef] [PubMed]
- Mughini-Gras, L.; van der Plaats, R.Q.J.; van der Wielen, P.; Bauerlein, P.S.; Husman, A.M.D. Riverine microplastic and microbial community compositions: A field study in the Netherlands. Water Res. 2021, 192, 116852. [Google Scholar] [CrossRef]
- Urban-Malinga, B.; Zalewski, M.; Jakubowska, A.; Wodzinowski, T.; Malinga, M.; Palys, B.; Dabrowska, A. Microplastics on sandy beaches of the southern Baltic Sea. Mar. Pollut. Bull. 2020, 155, 111170. [Google Scholar] [CrossRef]
- Tong, H.Y.; Jiang, Q.Y.; Hu, X.S.; Zhong, X.C. Occurrence and identification of microplastics in tap water from China. Chemosphere 2020, 252, 126493. [Google Scholar] [CrossRef]
- Raju, S.; Carbery, M.; Kuttykattil, A.; Senthirajah, K.; Lundmark, A.; Rogers, Z.; Suresh, S.C.B.; Evans, G.; Palanisami, T. Improved methodology to determine the fate and transport of microplastics in a secondary wastewater treatment plant. Water Res. 2020, 173, 115549. [Google Scholar] [CrossRef]
- Pohl, F.; Eggenhuisen, J.T.; Kane, I.A.; Clare, M.A. Transport and Burial of Microplastics in Deep-Marine Sediments by Turbidity Currents. Environ. Sci. Technol. 2020, 54, 4180–4189. [Google Scholar] [CrossRef]
- Khoironi, A.; Hadiyanto, H.; Anggoro, S.; Sudarno, S. Evaluation of polypropylene plastic degradation and microplastic identification in sediments at Tambak Lorok coastal area, Semarang, Indonesia. Mar. Pollut. Bull. 2020, 151, 110868. [Google Scholar] [CrossRef]
- Firdaus, M.; Trihadiningrum, Y.; Lestari, P. Microplastic pollution in the sediment of Jagir Estuary, Surabaya City, Indonesia. Mar. Pollut. Bull. 2020, 150, 110790. [Google Scholar] [CrossRef]
- Falahudin, D.; Cordova, M.R.; Sun, X.X.; Yogaswara, D.; Wulandari, I.; Hindarti, D.; Arifin, Z. The first occurrence, spatial distribution and characteristics of microplastic particles in sediments from Banten Bay, Indonesia. Sci. Total Environ. 2020, 705, 135304. [Google Scholar] [CrossRef]
- Cordova, M.R.; Purwiyanto, A.I.S.; Suteja, Y. Abundance and characteristics of microplastics in the northern coastal waters of Surabaya, Indonesia. Mar. Pollut. Bull. 2019, 142, 183–188. [Google Scholar] [CrossRef]
- Alam, F.C.; Sembiring, E.; Muntalif, B.S.; Suendo, V. Microplastic distribution in surface water and sediment river around slum and industrial area (case study: Ciwalengke River, Majalaya district, Indonesia). Chemosphere 2019, 224, 637–645. [Google Scholar] [CrossRef]
- Kwon, O.Y.; Kang, J.H.; Hong, S.H.; Shim, W.J. Spatial distribution of microplastic in the surface waters along the coast of Korea. Mar. Pollut. Bull. 2020, 155, 110729. [Google Scholar] [CrossRef]
- Kor, K.; Ghazilou, A.; Ershadifar, H. Microplastic pollution in the littoral sediments of the northern part of the Oman Sea. Mar. Pollut. Bull. 2020, 155, 111166. [Google Scholar] [CrossRef]
- Gomiero, A.; Oysaed, K.B.; Agustsson, T.; van Hoytema, N.; van Thiel, T.; Grati, F. First record of characterization, concentration and distribution of microplastics in coastal sediments of an urban fjord in south west Norway using a thermal degradation method. Chemosphere 2019, 227, 705–714. [Google Scholar] [CrossRef]
- Indonesa Marine Debris Hotspot Rapid Assessment, Synthesis Report World Bank Group. 2018. Available online: https://documents.worldbank.org/en/publication/documents-reports/documentdetail/983771527663689822/indonesia-marine-debris-hotspot-rapid-assessment-synthesis-report/ (accessed on 20 July 2022).
- Sui, Q.; Zhang, L.J.; Xia, B.; Chen, B.J.; Sun, X.M.; Zhu, L.; Wang, R.Y.; Qu, K.M. Spatiotemporal distribution, source identification and inventory of microplastics in surface sediments from Sanggou Bay, China. Sci. Total Environ. 2020, 723, 138064. [Google Scholar] [CrossRef]
- Zhao, J.M.; Ran, W.; Teng, J.; Liu, Y.L.; Liu, H.; Yin, X.N.; Cao, R.W.; Wang, Q. Microplastic pollution in sediments from the Bohai Sea and the Yellow Sea, China. Sci. Total Environ. 2018, 640, 637–645. [Google Scholar] [CrossRef]
- Li, R.; Yu, L.; Chai, M.; Wu, H.; Zhu, X. The distribution, characteristics and ecological risks of microplastics in the mangroves of Southern China. Sci. Total Environ. 2020, 708, 135025. [Google Scholar] [CrossRef]
- Lithner, D.; Larsson, Å.; Dave, G. Environmental and health hazard ranking and assessment of plastic polymers based on chemical composition. Sci. Total Environ. 2011, 409, 3309–3324. [Google Scholar] [CrossRef]
- Zhang, L.L.; Zhang, S.P.; Wang, Y.H.; Yu, K.F.; Li, R.L. The spatial distribution of microplastic in the sands of a coral reef island in the South China Sea: Comparisons of the fringing reef and atoll. Sci. Total Environ. 2019, 688, 780–786. [Google Scholar] [CrossRef]
- Kataoka, T.; Nihei, Y.; Kudou, K.; Hinata, H. Assessment of the sources and inflow processes of microplastics in the river environments of Japan. Environ. Pollut. 2019, 244, 958–965. [Google Scholar] [CrossRef]
- Cordova, M.R.; Ulumuddin, Y.I.; Purbonegoro, T.; Shiomoto, A. Characterization of microplastics in mangrove sediment of Muara Angke Wildlife Reserve, Indonesia. Mar. Pollut. Bull. 2021, 163, 112012. [Google Scholar] [CrossRef]
- Cordova, M.R.; Wahyudi, A.A.J. Microplastic in the deep-sea sediment of Southwestern Sumatera waters. Mar. Res. Indones. 2016, 41, 27–35. [Google Scholar] [CrossRef]
- Wang, C.H.; Zhao, J.; Xing, B.S. Environmental source, fate, and toxicity of microplastics. J. Hazard. Mater. 2021, 407, 17. [Google Scholar] [CrossRef]
- Wang, Y.; Zou, X.Q.; Peng, C.; Qiao, S.Q.; Wang, T.; Yu, W.W.; Khokiattiwong, O.A.; Kornkanitnan, N.R.M. Occurrence and distribution of microplastics in surface sediments from the Gulf of Thailand. Mar. Pollut. Bull. 2020, 152, 110916. [Google Scholar] [CrossRef]
- Peng, L.C.; Fu, D.D.; Qi, H.Y.; Lan, C.Q.; Yu, H.M.; Ge, C.J. Micro- and nano-plastics in marine environment: Source, distribution and threats—A review. Sci. Total Environ. 2020, 698, 134254. [Google Scholar] [CrossRef]
- Lestari, P.; Trihadiningrum, Y.; Wijaya, B.A.; Yunus, K.A.; Firdaus, M. Distribution of microplastics in Surabaya River, Indonesia. Sci. Total Environ. 2020, 726, 138560. [Google Scholar] [CrossRef] [PubMed]
- Schwarz, A.E.; Ligthart, T.N.; Boukris, E.; van Harmelen, T. Sources, transport, and accumulation of different types of plastic litter in aquatic environments: A review study. Mar. Pollut. Bull. 2019, 143, 92–100. [Google Scholar] [CrossRef] [PubMed]
- Porter, A.; Lyons, B.P.; Galloway, T.S.; Lewis, C. Role of Marine Snows in Microplastic Fate and Bioavailability. Environ. Sci. Technol. 2018, 52, 7111–7119. [Google Scholar] [CrossRef]
- Ma, H.; Pu, S.Y.; Liu, S.B.; Bai, Y.C.; Mandal, S.; Xing, B.S. Microplastics in aquatic environments: Toxicity to trigger ecological consequences. Environ. Pollut. 2020, 261, 114089. [Google Scholar] [CrossRef] [PubMed]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ni’am, A.C.; Hassan, F.; Shiu, R.-F.; Jiang, J.-J. Microplastics in Sediments of East Surabaya, Indonesia: Regional Characteristics and Potential Risks. Int. J. Environ. Res. Public Health 2022, 19, 12348. https://doi.org/10.3390/ijerph191912348
Ni’am AC, Hassan F, Shiu R-F, Jiang J-J. Microplastics in Sediments of East Surabaya, Indonesia: Regional Characteristics and Potential Risks. International Journal of Environmental Research and Public Health. 2022; 19(19):12348. https://doi.org/10.3390/ijerph191912348
Chicago/Turabian StyleNi’am, Achmad Chusnun, Fahir Hassan, Ruei-Feng Shiu, and Jheng-Jie Jiang. 2022. "Microplastics in Sediments of East Surabaya, Indonesia: Regional Characteristics and Potential Risks" International Journal of Environmental Research and Public Health 19, no. 19: 12348. https://doi.org/10.3390/ijerph191912348
APA StyleNi’am, A. C., Hassan, F., Shiu, R.-F., & Jiang, J.-J. (2022). Microplastics in Sediments of East Surabaya, Indonesia: Regional Characteristics and Potential Risks. International Journal of Environmental Research and Public Health, 19(19), 12348. https://doi.org/10.3390/ijerph191912348







