Perinatal Health Inequalities in the Industrial Region of Estonia: A Birth Registry-Based Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population and Variables
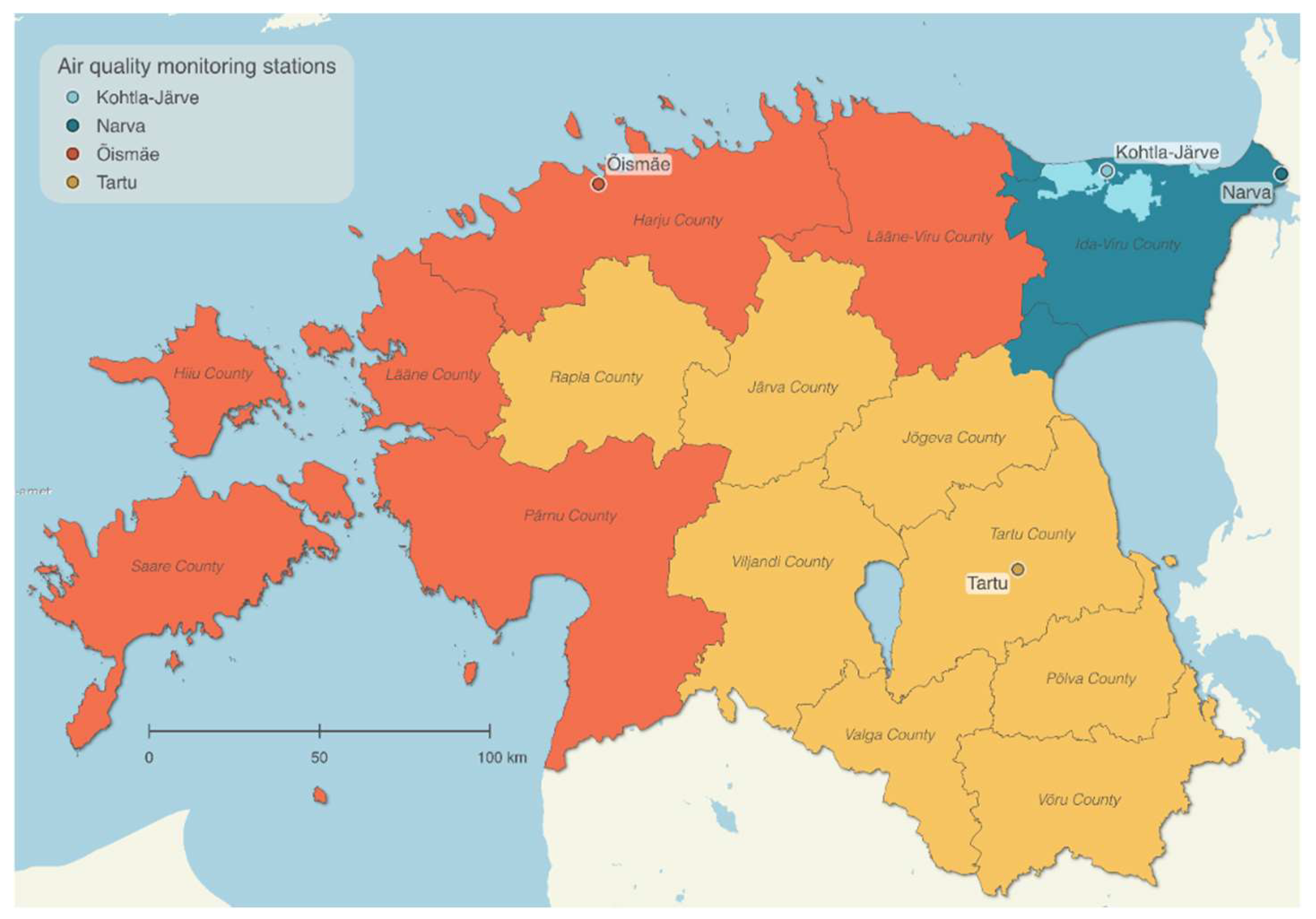
2.2. Air Pollution Exposure Assessment
2.3. Statistical Analysis
3. Results
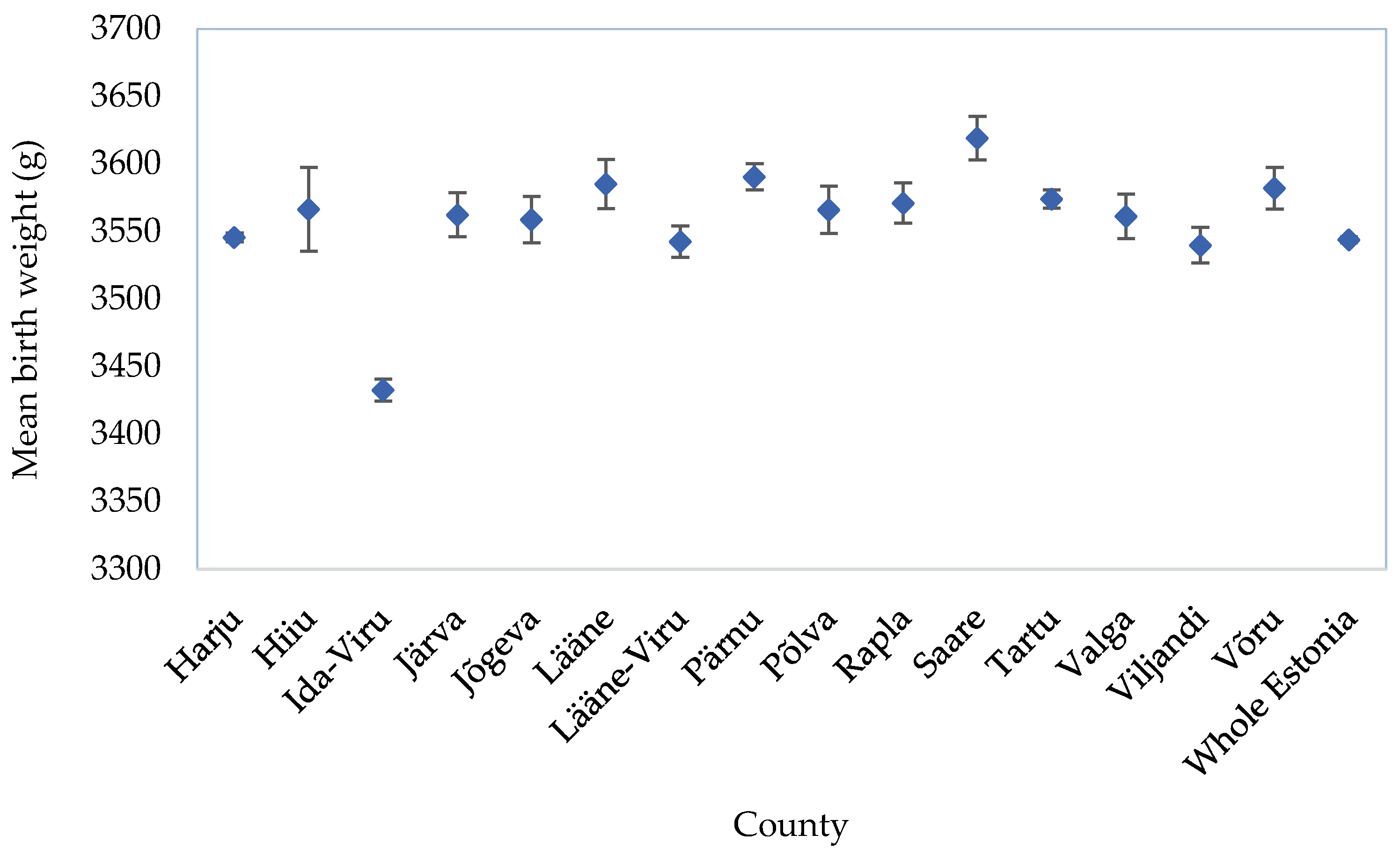
3.1. Distribution of Adverse Birth Outcomes of Singleton Births in Ida-Viru County and Estonia
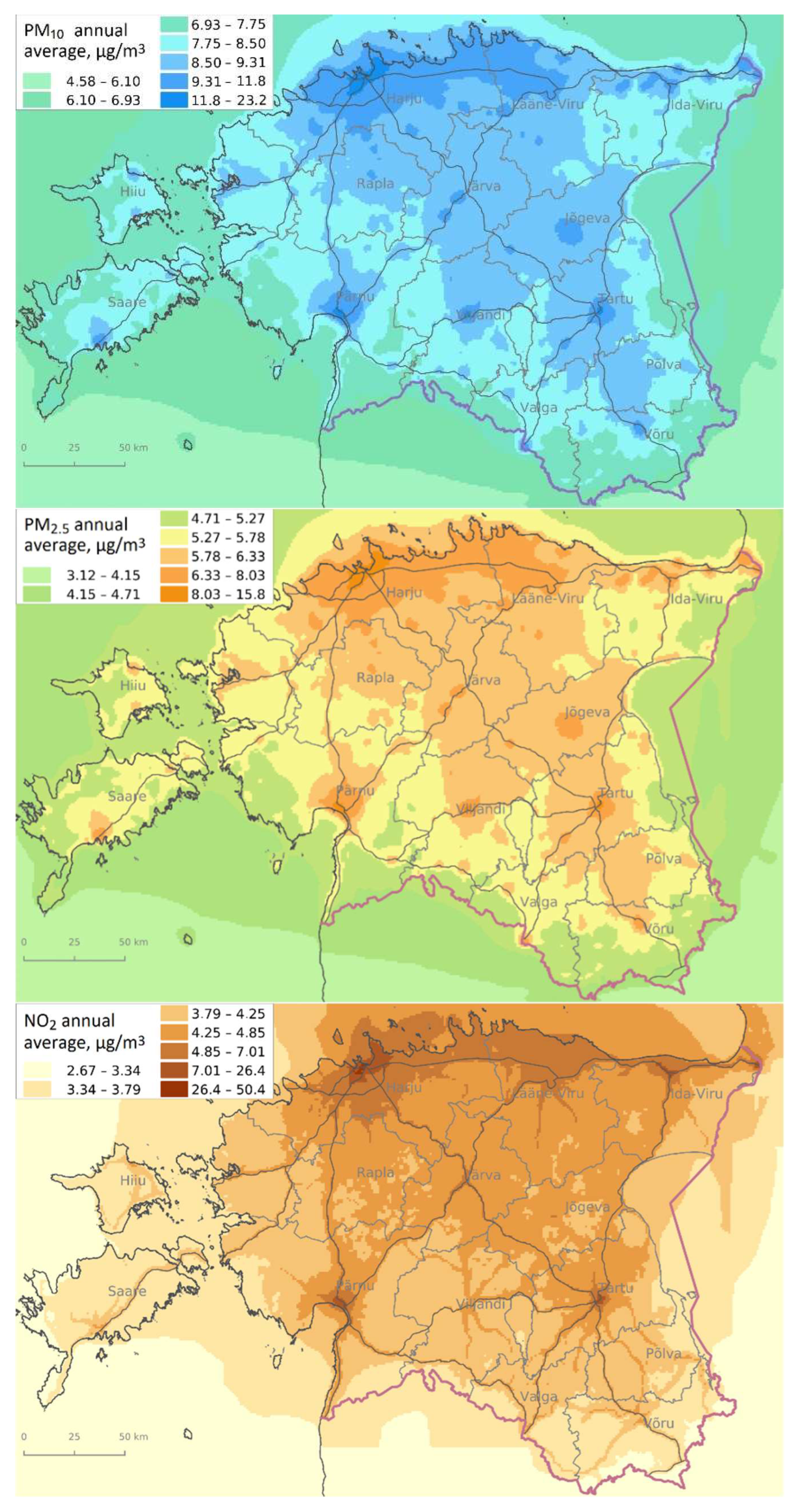
3.2. Air Pollution Exposure during Pregnancy
3.3. Impact of Air Pollution, Residential Proximity, and Sociodemographic Factors on Adverse Birth Outcomes
4. Discussion
4.1. Limitations and Strengths
4.2. Future Research Perspectives
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Dahlgren, G.; Whitehead, M. European Strategies for Tackling Social Inequities in Health: Levelling up Part 2; WHO Regional Office for Europe: Copenhagen, Denmark, 2006. [Google Scholar]
- Solar, O.; Irwin, A. A conceptual Framework for Action on the Social Determinants of Health; Social Determinants of Health: Discussion Paper 2 (Policy and Practice); World Health Organization: Geneva, Switzerland, 2010.
- Evans, L.; Engelman, M.; Mikulas, A.; Malecki, K. How are social determinants of health integrated into epigenetic research? A systematic review. Soc. Sci. Med. 2021, 273, 113738. [Google Scholar] [CrossRef] [PubMed]
- De Sario, M.; Pasetto, R.; Vecchi, S.; Zeka, A.; Hoek, G.; Michelozzi, P.; Iavarone, I.; Fletcher, T.; Bauleo, L.; Ancona, C. A scoping review of the epidemiological methods used to investigate the health effects of industrially contaminated sites. Epidemiol. Prev. 2018, 42, 59–68. [Google Scholar] [CrossRef]
- Dunn, A.M.; Burns, C.; Sattler, B. Environmental health of children. J. Pediatr. Health Care 2003, 17, 223–231. [Google Scholar] [CrossRef]
- Lee, S.; Hong, Y.-C.; Park, H.; Kim, Y.; Ha, M.; Ha, E. Combined effects of multiple prenatal exposure to pollutants on birth weight: The Mothers and Children’s Environmental Health (MOCEH) study. Environ. Res. 2020, 181, 108832. [Google Scholar] [CrossRef] [PubMed]
- Poursafa, P.; Kelishadi, R. What health professionals should know about the health effects of air pollution and climate change on children and pregnant mothers. Iran. J. Nurs. Midwifery Res. 2011, 16, 257–264. [Google Scholar] [PubMed]
- Barker, D.J.P.; Godfrey, K.M.; Gluckman, P.D.; Harding, J.E.; Owens, J.A.; Robinson, J.S. Fetal nutrition and cardiovascular disease in adult life. Lancet 1993, 341, 938–941. [Google Scholar] [CrossRef]
- Gluckman, P.D.; Hanson, M.A.; Cooper, C.; Thornburg, K.L. Effect of in utero and early-life conditions on adult health and disease. N. Engl. J. Med. 2008, 359, 61–73. [Google Scholar] [CrossRef]
- Kajantie, E.; Osmond, C.; Barker, D.J.; Forsén, T.; Phillips, D.I.; Eriksson, J.G. Size at birth as a predictor of mortality in adulthood: A follow-up of 350 000 person-years. Int. J. Epidemiol. 2005, 34, 655–663. [Google Scholar] [CrossRef]
- Martín-Calvo, N.; Goni, L.; Tur, J.A.; Martínez, J.A. Low birth weight and small for gestational age are associated with complications of childhood and adolescence obesity: Systematic review and meta-analysis. Obes. Rev. 2022, 23, e13380. [Google Scholar] [CrossRef]
- Swanson, J.M.; Entringer, S.; Buss, C.; Wadhwa, P.D. Developmental origins of health and disease: Environmental exposures. Semin. Reprod. Med. 2009, 27, 391–402. [Google Scholar] [CrossRef]
- Zanetti, D.; Tikkanen, E.; Gustafsson, S.; Priest, J.R.; Burgess, S.; Ingelsson, E. Birthweight, Type 2 Diabetes Mellitus, and Cardiovascular Disease. Circ. Genom. Precis. Med. 2018, 11, e002054. [Google Scholar] [CrossRef] [PubMed]
- Blumenshine, P.; Egerter, S.; Barclay, C.J.; Cubbin, C.; Braveman, P.A. Socioeconomic Disparities in Adverse Birth Outcomes: A Systematic Review. Am. J. Prev. Med. 2010, 39, 263–272. [Google Scholar] [CrossRef] [PubMed]
- McHale, P.; Maudsley, G.; Pennington, A.; Schlüter, D.K.; Barr, B.; Paranjothy, S.; Taylor-Robinson, D. Mediators of socioeconomic inequalities in preterm birth: A systematic review. BMC Public Health 2022, 22, 1134. [Google Scholar] [CrossRef] [PubMed]
- Albert-Ballestar, S.; García-Altés, A. Measuring health inequalities: A systematic review of widely used indicators and topics. Int. J. Equity Health 2021, 20, 73. [Google Scholar] [CrossRef] [PubMed]
- ECHI-European Core Health Indicators. Available online: https://health.ec.europa.eu/indicators-and-data/european-core-health-indicators-echi/echi-european-core-health-indicators_en (accessed on 13 July 2022).
- Melody, S.M.; Ford, J.; Wills, K.; Venn, A.; Johnston, F.H. Maternal exposure to short-to medium-term outdoor air pollution and obstetric and neonatal outcomes: A systematic review. Environ. Pollut. 2019, 244, 915–925. [Google Scholar] [CrossRef]
- Stieb, D.M.; Chen, L.; Eshoul, M.; Judek, S. Ambient air pollution, birth weight and preterm birth: A systematic review and meta-analysis. Environ. Res. 2012, 117, 100–111. [Google Scholar] [CrossRef]
- Casey, J.A.; Karasek, D.; Ogburn, E.L.; Goin, D.E.; Dang, K.; Braveman, P.A.; Morello-Frosch, R. Retirements of Coal and Oil Power Plants in California: Association With Reduced Preterm Birth Among Populations Nearby. Am. J. Epidemiol. 2018, 187, 1586–1594. [Google Scholar] [CrossRef]
- DeCicca, P.; Malak, N. When good fences aren’t enough: The impact of neighboring air pollution on infant health. J. Environ. Econ. Manag. 2020, 102, 102324. [Google Scholar] [CrossRef]
- Ha, S.; Hu, H.; Roth, J.; Kan, H.; Xu, X. Associations Between Residential Proximity to Power Plants and Adverse Birth Outcomes. Am. J. Epidemiol. 2015, 182, 215–224. [Google Scholar] [CrossRef]
- Tsai, S.-S.; Yu, H.-S.; Chang, C.-C.; Chuang, H.-Y.; Yang, C.-Y. Increased Risk of Preterm Delivery in Women Residing near Thermal Power Plants in Taiwan. Arch. Environ. Health Int. J. 2004, 59, 478–483. [Google Scholar] [CrossRef]
- Yang, M.; Bhatta, R.A.; Chou, S.-Y.; Hsieh, C.-I. The Impact of Prenatal Exposure to Power Plant Emissions on Birth Weight: Evidence from a Pennsylvania Power Plant Located Upwind of New Jersey. J. Policy Anal. Manag. 2017, 36, 557–583. [Google Scholar] [CrossRef] [PubMed]
- Gray, S.C.; Edwards, S.E.; Schultz, B.D.; Miranda, M.L. Assessing the impact of race, social factors and air pollution on birth outcomes: A population-based study. Environ. Health 2014, 13, 4. [Google Scholar] [CrossRef]
- Veber, T.; Dahal, U.; Lang, K.; Orru, K.; Orru, H. Industrial Air Pollution Leads to Adverse Birth Outcomes: A Systematized Review of Different Exposure Metrics and Health Effects in Newborns. Public Health Rev. 2022, 43, 1604775. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, C.C.; Amrhein, C.G.; Serrano-Lomelin, J.A.; Osornio-Vargas, A.R. Geographic information assessment of maternal ambient health hazards and adverse birth outcomes in Canada. Sci. Total Environ. 2019, 696, 134091. [Google Scholar] [CrossRef]
- Svechkina, A.; Dubnov, J.; Portnov, B.A. Environmental risk factors associated with low birth weight: The case study of the Haifa Bay Area in Israel. Environ. Res. 2018, 165, 337–348. [Google Scholar] [CrossRef]
- Serrano-Lomelin, J.; Nielsen, C.C.; Jabbar, M.S.M.; Wine, O.; Bellinger, C.; Villeneuve, P.J.; Stieb, D.; Aelicks, N.; Aziz, K.; Buka, I.; et al. Interdisciplinary-driven hypotheses on spatial associations of mixtures of industrial air pollutants with adverse birth outcomes. Environ. Int. 2019, 131, 104972. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Sun, J.; Liu, Y.; Liang, H.; Wang, M.; Wang, C.; Shi, T. Different exposure levels of fine particulate matter and preterm birth: A meta-analysis based on cohort studies. Environ. Sci. Pollut. Res. 2017, 24, 17976–17984. [Google Scholar] [CrossRef]
- Bergstra, A.D.; Brunekreef, B.; Burdorf, A. The influence of industry-related air pollution on birth outcomes in an industrialized area. Environ. Pollut. 2021, 269, 115741. [Google Scholar] [CrossRef]
- Lin, C.-M.; Li, C.-Y.; Mao, I.-F. Increased Risks of Term Low-Birth-Weight Infants in a Petrochemical Industrial City with High Air Pollution Levels. Arch. Environ. Health Int. J. 2004, 59, 663–668. [Google Scholar] [CrossRef]
- dos Reis, M.M.; Guimarães, M.T.; Braga, A.L.F.; Martins, L.C.; Pereira, L.A.A. Air pollution and low birth weight in an industrialized city in Southeastern Brazil, 2003–2006. Rev. Bras. Epidemiol. 2017, 20, 189–199. [Google Scholar] [CrossRef]
- Rogers, J.F.; Dunlop, A.L. Air Pollution and Very Low Birth Weight Infants: A Target Population? Pediatrics 2006, 118, 156–164. [Google Scholar] [CrossRef] [PubMed]
- Cassidy-Bushrow, A.E.; Burmeister, C.; Lamerato, L.; Lemke, L.D.; Mathieu, M.; O’Leary, B.F.; Sperone, F.G.; Straughen, J.K.; Reiners, J.J. Prenatal airshed pollutants and preterm birth in an observational birth cohort study in Detroit, Michigan, USA. Environ. Res. 2020, 189, 109845. [Google Scholar] [CrossRef] [PubMed]
- Mohorovic, L. First two months of pregnancy—critical time for preterm delivery and low birthweight caused by adverse effects of coal combustion toxics. Early Hum. Dev. 2004, 80, 115–123. [Google Scholar] [CrossRef] [PubMed]
- Yang, M.; Chou, S.-Y. The impact of environmental regulation on fetal health: Evidence from the shutdown of a coal-fired power plant located upwind of New Jersey. J. Environ. Econ. Manag. 2018, 90, 269–293. [Google Scholar] [CrossRef]
- Hill, E.L. Shale gas development and infant health: Evidence from Pennsylvania. J. Health Econ. 2018, 61, 134–150. [Google Scholar] [CrossRef]
- Lin, M.-C.; Chiu, H.-F.; Yu, H.-S.; Tsai, S.-S.; Cheng, B.-H.; Wu, T.-N.; Sung, F.-C.; Yang, C.-Y. Increased Risk of Preterm Delivery in Areas with Air Pollution from a Petroleum Refinery Plant in Taiwan. J. Toxicol. Environ. Health A 2001, 64, 637–644. [Google Scholar] [CrossRef]
- Yang, C.-Y.; Chang, C.-C.; Chuang, H.-Y.; Ho, C.-K.; Wu, T.-N.; Chang, P.-Y. Increased risk of preterm delivery among people living near the three oil refineries in Taiwan. Environ. Int. 2004, 30, 337–342. [Google Scholar] [CrossRef]
- Lin, M.-C.; Yu, H.-S.; Tsai, S.-S.; Cheng, B.-H.; Hsu, T.-Y.; Wu, T.-N.; Yang, C.-Y. Adverse Pregnancy Outcome in a Petrochemical Polluted Area in Taiwan. J. Toxicol. Environ. Health A 2001, 63, 565–574. [Google Scholar] [CrossRef]
- Yang, C.-Y.; Cheng, B.-H.; Hsu, T.-Y.; Chuang, H.-Y.; Wu, T.-N.; Chen, P.-C. Association between Petrochemical Air Pollution and Adverse Pregnancy Outcomes in Taiwan. Arch. Environ. Health Int. J. 2002, 57, 461–465. [Google Scholar] [CrossRef]
- Yang, C.-Y.; Chiu, H.-F.; Tsai, S.-S.; Chang, C.-C.; Chuang, H.-Y. Increased Risk of Preterm Delivery in Areas with Cancer Mortality Problems from Petrochemical Complexes. Environ. Res. 2002, 89, 195–200. [Google Scholar] [CrossRef]
- Porter, T.R.; Kent, S.T.; Su, W.; Beck, H.M.; Gohlke, J.M. Spatiotemporal association between birth outcomes and coke production and steel making facilities in Alabama, USA: A cross-sectional study. Environ. Health 2014, 13, 85. [Google Scholar] [CrossRef] [PubMed]
- Currie, J.; Davis, L.; Greenstone, M.; Walker, R. Environmental Health Risks and Housing Values: Evidence from 1,600 Toxic Plant Openings and Closings. Am. Econ. Rev. 2015, 105, 678–709. [Google Scholar] [CrossRef] [PubMed]
- Pasetto, R.; Mattioli, B.; Marsili, D. Environmental justice in industrially contaminated sites. A review of scientific evidence in the WHO European Region. Int. J. Environ. Res. Public Health 2019, 16, 998. [Google Scholar] [CrossRef]
- Perlin, S.A.; Wong, D.; Sexton, K. Residential Proximity to Industrial Sources of Air Pollution: Interrelationships among Race, Poverty, and Age. J. Air Waste Manag. Assoc. 2001, 51, 406–421. [Google Scholar] [CrossRef]
- Auger, N.; Giraud, J.; Daniel, M. The joint influence of area income, income inequality, and immigrant density on adverse birth outcomes: A population-based study. BMC Public Health 2009, 9, 237. [Google Scholar] [CrossRef]
- de Graaf, J.P.; Steegers, E.A.P.; Bonsel, G.J. Inequalities in perinatal and maternal health. Curr. Opin. Obstet. Gynecol. 2013, 25, 98–108. [Google Scholar] [CrossRef]
- Malley, C.S.; Kuylenstierna, J.C.I.; Vallack, H.W.; Henze, D.K.; Blencowe, H.; Ashmore, M.R. Preterm birth associated with maternal fine particulate matter exposure: A global, regional and national assessment. Environ. Int. 2017, 101, 173–182. [Google Scholar] [CrossRef]
- Messer, L.C.; Vinikoor, L.C.; Laraia, B.A.; Kaufman, J.S.; Eyster, J.; Holzman, C.; Culhane, J.; Elo, I.; Burke, J.G.; O’Campo, P. Socioeconomic domains and associations with preterm birth. Soc. Sci. Med. 2008, 67, 1247–1257. [Google Scholar] [CrossRef]
- Lavigne, E.; Yasseen, A.S.; Stieb, D.M.; Hystad, P.; van Donkelaar, A.; Martin, R.V.; Brook, J.R.; Crouse, D.L.; Burnett, R.T.; Chen, H.; et al. Ambient air pollution and adverse birth outcomes: Differences by maternal comorbidities. Environ. Res. 2016, 148, 457–466. [Google Scholar] [CrossRef]
- Melody, S.; Wills, K.; Knibbs, L.D.; Ford, J.; Venn, A.; Johnston, F. Adverse birth outcomes in Victoria, Australia in association with maternal exposure to low levels of ambient air pollution. Environ. Res. 2020, 188, 109784. [Google Scholar] [CrossRef]
- Koo, Y.-J.; Ryu, H.-M.; Yang, J.-H.; Lim, J.-H.; Lee, J.-E.; Kim, M.-Y.; Chung, J.-H. Pregnancy outcomes according to increasing maternal age. Taiwan J. Obstet. Gynecol. 2012, 51, 60–65. [Google Scholar] [CrossRef] [PubMed]
- Ogawa, K.; Urayama, K.Y.; Tanigaki, S.; Sago, H.; Sato, S.; Saito, S.; Morisaki, N. Association between very advanced maternal age and adverse pregnancy outcomes: A cross sectional Japanese study. BMC Pregnancy Childbirth 2017, 17, 349. [Google Scholar] [CrossRef] [PubMed]
- Martuzzi, M.; Pasetto, R.; Martin-Olmedo, P. Industrially Contaminated Sites and Health. J. Environ. Public Health 2014, 2014, e198574. [Google Scholar] [CrossRef] [PubMed]
- Keskkonnaagentuur Environmental Monitoring|Environment Agency, Estonia. Available online: https://keskkonnaagentuur.ee/keskkonnaseire-ja-analuusid/keskkonnaseire (accessed on 11 July 2022).
- OECD Greenhouse Gas Emissions. Available online: https://stats.oecd.org/Index.aspx?QueryId=51342# (accessed on 11 July 2022).
- OECD. OECD Environmental Performance Reviews: Estonia 2017; OECD Environmental Performance Reviews; OECD: Paris, France, 2017; Available online: https://www.oecd-ilibrary.org/environment/oecd-environmental-performance-reviews-estonia-2017_9789264268241-en (accessed on 27 July 2022).
- Global Footprint Network Ecological Footprint of the Countries. Available online: https://data.footprintnetwork.org/#/compareCountries?type=EFCpc&cn=all&yr=2018 (accessed on 11 July 2022).
- Orru, H.; Idavain, J.; Pindus, M.; Orru, K.; Kesanurm, K.; Lang, A.; Tomasova, J. Residents’ Self-Reported Health Effects and Annoyance in Relation to Air Pollution Exposure in an Industrial Area in Eastern-Estonia. Int. J. Environ. Res. Public Health 2018, 15, 252. [Google Scholar] [CrossRef]
- Idavain, J.; Julge, K.; Rebane, T.; Lang, A.; Orru, H. Respiratory symptoms, asthma and levels of fractional exhaled nitric oxide in schoolchildren in the industrial areas of Estonia. Sci. Total Environ. 2019, 650, 65–72. [Google Scholar] [CrossRef]
- Idavain, J.; Lang, K.; Tomasova, J.; Lang, A.; Orru, H. Cancer incidence trends in the oil shale industrial region in Estonia. Int. J. Environ. Res. Public Health 2020, 17, 3833. [Google Scholar] [CrossRef]
- Kanger, L.; Sovacool, B.K. Towards a multi-scalar and multi-horizon framework of energy injustice: A whole systems analysis of Estonian energy transition. Polit. Geogr. 2022, 93, 102544. [Google Scholar] [CrossRef]
- Republic of Estonia Government. Strategy “Estonia 2035” (Adopted by the Riigikogu on 12 May 2021). Available online: https://www.valitsus.ee/en/node/31 (accessed on 5 January 2022).
- Muiznieks, N.; Rozenvalds, J.; Birka, I. Ethnicity and social cohesion in the post-Soviet Baltic states. Patterns Prejud. 2013, 47, 288–308. [Google Scholar] [CrossRef]
- Michelson, A.; Koppel, K.; Melesk, K.; Arrak, K.; Laurimäe, M.; Murasov, M.; Paat-Ahi, G.; Piwek, A. Adaptation of Ida-Virumaa County’s Economy and Labor Market to the Reduction of Oil Shale Industry; Poliitikauuringute Keskus Praxis: Tallinn, Estonia, 2020; Available online: http://www.praxis.ee/en/works/ida-viru-oil-shale-industry (accessed on 27 July 2022).
- Pihor, K.; Kralik, S.; Aolaid-Aas, A.; Jürgenson, A.; Rell, M.; Paat-Ahi, G.; Batueva, V. Assessing the Socio-Economic Impact of Oil Shale Mining and Processing; Poliitikauuringute Keskus Praxis: Tallinn, Estonia, 2013; Available online: http://www.praxis.ee/en/works/assessing-the-socio-economic-impact-of-oil-shale-mining-and-processing (accessed on 27 July 2022).
- Airviro|Apertum. Available online: http://airviro.com/airviro/ (accessed on 5 January 2022).
- Geoportal: Republic of Estonia, Land Board Address Data Geocoding. Available online: https://geoportaal.maaamet.ee/eng/Services/index.php?lang_id=2&page_id=873 (accessed on 12 July 2022).
- World Health Organization. WHO global Air Quality Guidelines: Particulate Matter (PM2.5 and PM10), Ozone, Nitrogen Dioxide, Sulfur Dioxide and Carbon Monoxide; World Health Organization: Geneva, Switzerland, 2021; ISBN 978-92-4-003422-8.
- Sapkota, A.; Chelikowsky, A.P.; Nachman, K.E.; Cohen, A.J.; Ritz, B. Exposure to particulate matter and adverse birth outcomes: A comprehensive review and meta-analysis. Air Qual. Atmos. Health 2012, 5, 369–381. [Google Scholar] [CrossRef]
- Klepac, P.; Locatelli, I.; Korošec, S.; Künzli, N.; Kukec, A. Ambient air pollution and pregnancy outcomes: A comprehensive review and identification of environmental public health challenges. Environ. Res. 2018, 167, 144–159. [Google Scholar] [CrossRef]
- Ji, Y.; Song, F.; Xu, B.; Zhu, Y.; Lu, C.; Xia, Y. Association between exposure to particulate matter during pregnancy and birthweight: A systematic review and a meta-analysis of birth cohort studies. J. Biomed. Res. 2019, 33, 56–68. [Google Scholar] [CrossRef]
- Jacobs, M.; Zhang, G.; Chen, S.; Mullins, B.; Bell, M.; Jin, L.; Guo, Y.; Huxley, R.; Pereira, G. The association between ambient air pollution and selected adverse pregnancy outcomes in China: A systematic review. Sci. Total Environ. 2017, 579, 1179–1192. [Google Scholar] [CrossRef] [PubMed]
- Pirrus, J.; Kljavin, K.; Derlõš, M. Kahaneva Rahvastikuga Piirkondades Probleemsete Korterelamute Elanike Ümberasustamine ja Kasutusest Väljalangenud Korterelamute Lammutamine; I tööetapi RAPORT; Ministry of Finance: Tallinn, Estonia, 2020.
- Aizer, A.; Currie, J. The intergenerational transmission of inequality: Maternal disadvantage and health at birth. Science 2014, 344, 856–861. [Google Scholar] [CrossRef] [PubMed]
- Williams, D.R.; Mohammed, S.A. Racism and Health I: Pathways and Scientific Evidence. Am. Behav. Sci. 2013, 57, 1152–1173. [Google Scholar] [CrossRef]
- Keenan-Devlin, L.S.; Smart, B.P.; Grobman, W.; Adam, E.K.; Freedman, A.; Buss, C.; Entringer, S.; Miller, G.E.; Borders, A.E.B. The intersection of race and socioeconomic status is associated with inflammation patterns during pregnancy and adverse pregnancy outcomes. Am. J. Reprod. Immunol. 2022, 87, e13489. [Google Scholar] [CrossRef]
- Kramer, M.S.; Séguin, L.; Lydon, J.; Goulet, L. Socio-economic disparities in pregnancy outcome: Why do the poor fare so poorly? Paediatr. Perinat. Epidemiol. 2000, 14, 194–210. [Google Scholar] [CrossRef]
- Bonzini, M.; Coggon, D.; Palmer, K.T. Risk of prematurity, low birthweight and pre-eclampsia in relation to working hours and physical activities: A systematic review. Occup. Environ. Med. 2007, 64, 228–243. [Google Scholar] [CrossRef]
- WHO. How Health Systems Can Address Health Inequities Linked to Migration and Ethnicity; WHO: Geneva, Switzerland, 2010.
- Palloni, A. Reproducing inequalities: Luck, wallets, and the enduring effects of childhood health. Demography 2006, 43, 587–615. [Google Scholar] [CrossRef]
- Kane, J.B.; Harris, K.M.; Siega-Riz, A.M. Intergenerational pathways linking maternal early life adversity to offspring birthweight. Soc. Sci. Med. 2018, 207, 89–96. [Google Scholar] [CrossRef]
- Kuh, D.; Shlomo, Y.B. Life course approaches to socioeconomic differentials in health. In A Life Course Approach to Chronic Disease Epidemiology; Kuh, D., Ben Shlomo, Y., Ezra, S., Eds.; Oxford University Press: Oxford, UK, 2004; pp. 77–115. ISBN 978-0-19-857815-4. [Google Scholar]
- Gorber, S.C.; Schofield-Hurwitz, S.; Hardt, J.; Levasseur, G.; Tremblay, M. The accuracy of self-reported smoking: A systematic review of the relationship between self-reported and cotinine-assessed smoking status. Nicotine Tob. Res. 2009, 11, 12–24. [Google Scholar] [CrossRef]
- Reile, R.; Veideman, T. Eesti Täiskasvanud Rahvastiku Tervisekäitumise Uuring 2020; National Institute for Health Development: Tallinn, Estonia, 2021; Available online: https://www.tai.ee/sites/default/files/2021-04/TKU2020_kogumik.pdf (accessed on 27 July 2022).
- Lynch, C.D.; Zhang, J. The research implications of the selection of a gestational age estimation method. Paediatr. Perinat. Epidemiol. 2007, 21, 86–96. [Google Scholar] [CrossRef] [PubMed]
- Sheppard, L.; Burnett, R.T.; Szpiro, A.A.; Kim, S.-Y.; Jerrett, M.; Pope, C.A.; Brunekreef, B. Confounding and exposure measurement error in air pollution epidemiology. Air Qual. Atmos. Health 2012, 5, 203–216. [Google Scholar] [CrossRef] [PubMed]
- Zeger, S.L.; Thomas, D.; Dominici, F.; Samet, J.M.; Schwartz, J.; Dockery, D.; Cohen, A. Exposure measurement error in time-series studies of air pollution: Concepts and consequences. Environ. Health Perspect. 2000, 108, 419–426. [Google Scholar] [CrossRef] [PubMed]
- Marmot, M.; Bell, R. Social inequalities in health: A proper concern of epidemiology. Ann. Epidemiol. 2016, 26, 238–240. [Google Scholar] [CrossRef] [PubMed]
- Sarigiannis, D.A. The exposome paradigm in environmental health. In Environmental Exposures and Human Health Challenges; IGI Global: Hershey, PA, USA, 2019; pp. 1–29. [Google Scholar]

| Ida-Viru County | Estonia | |||||
|---|---|---|---|---|---|---|
| All Births | LBW 1 | PTB 2 | All Births | LBW | PTB | |
| Infant gender, % (n) | ||||||
| Male | 51.3 (9552) | 4.4 (421) | 4.4 (423) | 51.4 (107,013) | 3.0 (3217) | 3.5 (3724) |
| Female | 48.7 (9074) | 5.2 (473) | 4.1 (372) | 48.6 (101,300) | 3.6 (3628) | 3.0 (3053) |
| Mother’s ethnicity, % (n) | ||||||
| Estonian | 16 (2988) | 4.1 (122) | 4.1 (122) | 72.3 (150,514) | 3.0 (4512) | 3.1 (4711) |
| Russian | 80.1 (14,906) | 5.0 (739) | 4.3 (644) | 25.1 (52,270) | 4.0 (2109) | 3.6 (1867) |
| Others | 3.9 (723) | 4.4 (32) | 3.9 (28) | 2.6 (5310) | 4.1 (217) | 3.6 (192) |
| Mother’s education, % (n) | ||||||
| Basic | 0.7 (125) | 10.4 (13) | 7.2 (9) | 1.0 (2176) | 6.7 (145) | 6.0 (131) |
| Secondary | 13.8 (2573) | 9.0 (231) | 7.3 (188) | 13.2 (27,476) | 4.8 (1312) | 4.2 (1161) |
| Higher secondary | 25.1 (4682) | 4.4 (208) | 3.8 (179) | 24.0 (49,979) | 3.3 (1673) | 3.4 (1683) |
| Applied education | 42.3 (7870) | 4.2 (333) | 3.9 (305) | 30.6 (63,753) | 3.2 (2021) | 3.2 (2021) |
| Higher education | 18.1 (3370) | 3.2 (108) | 3.4 (113) | 31.1 (64,840) | 2.6 (1688) | 2.7 (1774) |
| Mother’s age in years, % (n) | ||||||
| ≤20 | 5.5 (1025) | 6.5 (67) | 6.0 (61) | 3.7 (7632) | 5.1 (392) | 4.7 (361) |
| 20–24 | 24.6 (4589) | 4.5 (206) | 3.4 (154) | 17.8 (37,126) | 3.3 (1232) | 3 (1115) |
| 25–29 | 30.5 (5681) | 4.4 (248) | 3.9 (221) | 31.8 (66,307) | 2.9 (1908) | 2.8 (1889) |
| 30–34 | 23.4 (4362) | 4.7 (203) | 4.1 (181) | 27.7 (57,760) | 3.0 (1721) | 3.1 (1795) |
| 35–39 | 12.7 (2368) | 5.3 (125) | 5.5 (130) | 14.9 (30,990) | 3.7 (1162) | 3.8 (1190) |
| >40 | 3.2 (601) | 7.5 (45) | 8.0 (48) | 4.1(8496) | 5.1 (430) | 5.0 (427) |
| Proximity to industrial areas 3, % (n) | ||||||
| ≤3 km | 10.1 (1791) | 5.4 (96) | 4.8 (86) | 0.99 (1816) | 5.3 (97) | 4.8 (88) |
| ≤5 km | 43.4 (7699) | 4.9 (375) | 4.2 (321) | 4.2 (7724) | 4.9 (376) | 4.2 (323) |
| ≤10 km | 61.9 (10,992) | 4.6 (508) | 4.0 (444) | 6.0 (11,033) | 4.6 (509) | 4.0 (446) |
| Ida-Viru County | Estonia | ||||
|---|---|---|---|---|---|
| PM10 | PM2.5 | PM10 | PM2.5 | NO2 | |
| OR (95% CI) | |||||
| Preterm birth (PTB) | |||||
| Adjusted model 2 | |||||
| I trimester | 1.10 (0.98–1.24) | 1.01 (0.73–1.39) | 1.07 (1.02–1.13) | 1.09 1 (1.00–1.20) | 1.02 (0.94–1.10) |
| III trimester | 1.09 (0.97–1.23) | 1.26 (0.92–1.72) | 1.03 (0.97–1.09) | 1.01 (0.92–1.11) | 0.91 (0.84–0.99) |
| Fully–adjusted model 3 | |||||
| I trimester | 1.12 (0.99–1.26) | 0.99 (0.70–1.37) | 1.05 (0.99–1.11) | 1.12 (1.02–1.23) | 1.04 (0.96–1.13) |
| III trimester | 1.12 (0.98–1.26) | 1.30 (0.95–1.79) | 1.03 (0.97–1.09) | 1.03 (0.94–1.13) | 0.90 (0.83–0.98) |
| Low birth weight (LBW) | |||||
| Adjusted model | |||||
| I trimester | 1.10 (0.98–1.23) | 0.99 (0.73–1.35) | 1.05 (0.99–1.11) | 1.05 (0.96–1.15) | 0.96 (0.88–1.03) |
| III trimester | 1.04 (0.93–1.17) | 1.50 (1.12–1.99) | 1.05 (0.93–1.17) | 1.02 (0.93–1.13) | 0.93 (0.85–1.01) |
| Fully–adjusted model | |||||
| I trimester | 1.12 (0.99–1.25) | 0.96 (0.70–1.33) | 1.02 (0.97–1.08) | 1.06 (0.97–1.17) | 0.98 (0.91–1.06) |
| III trimester | 1.06 (0.94–1.19) | 1.56 (1.16–2.08) | 1.06 (0.94–1.19) | 1.03 (0.94–1.14) | 0.92 (0.85–1.00) |
| Residential Proximity | ≤3 km 1 | ≤5 km 1 | ≤10 km 1 |
|---|---|---|---|
| OR (95% CI) | |||
| Preterm birth (PTB) | |||
| Adjusted model 3 | 1.54 2 (1.23–1.90) | 1.38 (1.22–1.56) | 1.35 (1.22–1.50) |
| Fully–adjusted model 4 | 1.58 (1.26–1.98) | 1.07 (0.94–1.22) | 1.01 (0.89–1.14) |
| Low birth weight (LBW) | |||
| Adjusted model | 1.55 (1.26–1.91) | 1.48 (1.32–1.66) | 1.42 (1.29–1.57) |
| Fully–adjusted model | 1.51 (1.21–1.88) | 1.18 (1.04–1.33) | 1.09 (0.97–1.23) |
| Ida-Viru County | Estonia | |||
|---|---|---|---|---|
| Mother’s Ethnicity | Russians 1 | Other Non-Estonians 1 | Russians | Other Non-Estonians |
| OR (95% CI) | ||||
| Preterm birth (PTB) | ||||
| Adjusted model 3 | 1.09 (0.89–1.33) | 0.92 (0.60–1.39) | 1.15 2 (1.09–1.22) | 1.14 (0.99–1.33) |
| Fully–Adjusted model 4 | 1.08 (0.88–1.33) | 0.93 (0.60–1.41) | 1.05 (0.99–1.11) | 1.10 (0.94–1.28) |
| Low birth weight (LBW) | ||||
| Adjusted model | 1.26 (1.04–1.54) | 1.09 (0.72–1.61) | 1.36 (1.29–1.43) | 1.37 (1.19–1.57) |
| Fully–Adjusted model | 1.29 (1.05–1.57) | 1.11 (0.74–1.67) | 1.29 (1.22–1.36) | 1.34 (1.15–1.55) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dahal, U.; Veber, T.; Åström, D.O.; Tamm, T.; Albreht, L.; Teinemaa, E.; Orru, K.; Orru, H. Perinatal Health Inequalities in the Industrial Region of Estonia: A Birth Registry-Based Study. Int. J. Environ. Res. Public Health 2022, 19, 11559. https://doi.org/10.3390/ijerph191811559
Dahal U, Veber T, Åström DO, Tamm T, Albreht L, Teinemaa E, Orru K, Orru H. Perinatal Health Inequalities in the Industrial Region of Estonia: A Birth Registry-Based Study. International Journal of Environmental Research and Public Health. 2022; 19(18):11559. https://doi.org/10.3390/ijerph191811559
Chicago/Turabian StyleDahal, Usha, Triin Veber, Daniel Oudin Åström, Tanel Tamm, Leena Albreht, Erik Teinemaa, Kati Orru, and Hans Orru. 2022. "Perinatal Health Inequalities in the Industrial Region of Estonia: A Birth Registry-Based Study" International Journal of Environmental Research and Public Health 19, no. 18: 11559. https://doi.org/10.3390/ijerph191811559
APA StyleDahal, U., Veber, T., Åström, D. O., Tamm, T., Albreht, L., Teinemaa, E., Orru, K., & Orru, H. (2022). Perinatal Health Inequalities in the Industrial Region of Estonia: A Birth Registry-Based Study. International Journal of Environmental Research and Public Health, 19(18), 11559. https://doi.org/10.3390/ijerph191811559








