The Effectiveness of Virtual Reality Interventions on Smoking, Nutrition, Alcohol, Physical Activity and/or Obesity Risk Factors: A Systematic Review
Abstract
:1. Introduction
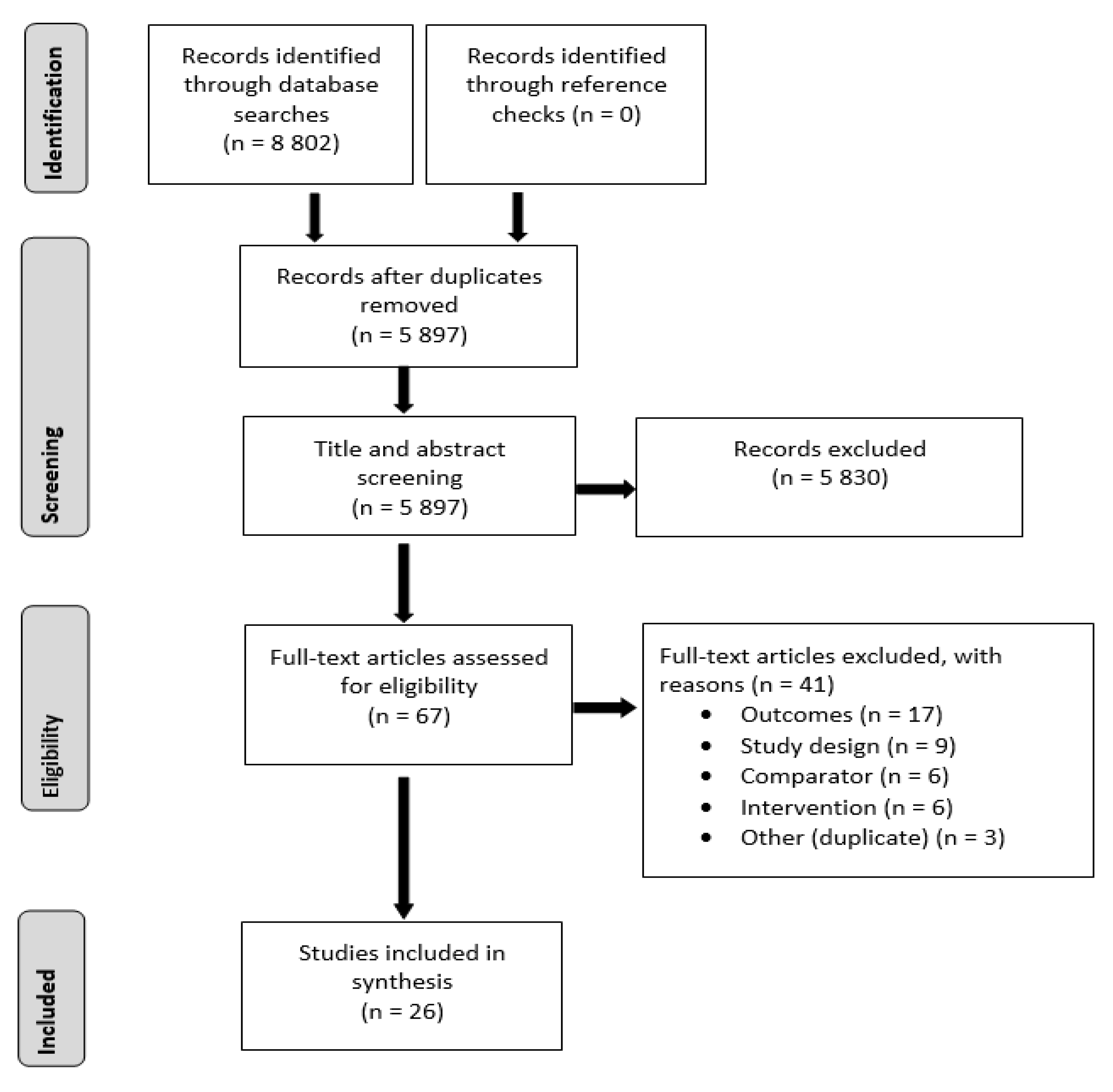
2. Materials and Methods
2.1. Design and Registration
2.2. Search Strategy
2.3. Inclusion Criteria
- Study design: Randomised controlled trials, cluster randomised controlled trials, randomised trials, and cluster randomised trials.
- Participants: People engaging in any of the following health risk factors, individually or in combination: tobacco use, inadequate nutrition, risky alcohol consumption, insufficient physical activity, and obesity; any age, any gender, from any country.
- Setting: Any setting (e.g., community, health setting).
- Intervention: Fully immersive, semi-immersive and non-immersive VR technology used to treat SNAPO risk factors. These types of VR immersion are defined as: fully immersive that required VR glasses that utilise a stereoscopic 3D environment by splitting the display between the user’s eyes; semi-immersive which provided immersion through the use of sensors, powerful displays, or replica physical setting such as an aeroplane cockpit to interact with a virtual setting; and non-immersive that consists of a virtual representation of the participant (e.g., avatar), displayed on a PC or console system, augmented reality, and related technology using computer-generated avatars in a VE.
- Comparators: No intervention or minimal support control groups, other modes of behavioural or non-behavioural interventions.
- Language: Studies published in English.
- Outcome measures:
2.4. Exclusion Criteria
2.5. Study Selection
2.6. Data Extraction
- Publication details: author(s) names, year of publication, country of study and years data collected
- Setting: population based, community or clinic-based
- Study design: randomised controlled trials, cluster randomised controlled trials, randomised trials or cluster randomised trials
- Population: demographics (age, gender, level of education, employment), sample size, recruitment methods, eligibility criteria, retention rates
- Intervention: type of virtual reality intervention
- Control/Comparators: type of control (e.g., no intervention, minimal support) or comparators (e.g., in person, telephone)
- Outcomes: smoking cessation/reduction, nutrition, alcohol use, physical activity, obesity, use, satisfaction and/or acceptability of support received and economic cost.
2.7. Quality Assessment
2.8. Data Synthesis
3. Results
3.1. Study Characteristics
3.1.1. Smoking Cessation or Reduction
3.1.2. Physical Activity
3.1.3. Physical Activity and Obesity/Weight
3.1.4. Obesity/Weight
3.1.5. Obesity/Weight and Nutrition
3.1.6. Obesity/Weight, Nutrition and Physical Activity
3.2. Primary Outcomes
3.2.1. Effectiveness of VR on Smoking Cessation or Reduction
3.2.2. Effectiveness of VR on Physical Activity
3.2.3. Effectiveness of VR on Physical Activity and Obesity/Weight
3.2.4. Effectiveness of VR on Obesity/Weight
3.2.5. Effectiveness of VR on Obesity/Weight and Nutrition
3.2.6. Effectiveness of VR on Obesity/Weight, Nutrition and Physical Activity
3.3. Secondary Outcomes
3.3.1. Secondary Outcomes for Smoking Cessation or Reduction
3.3.2. Secondary Outcomes for Physical Activity
3.3.3. Secondary Outcomes for Physical Activity and Obesity/Weight
3.3.4. Secondary Outcomes for Obesity/Weight
3.3.5. Secondary Outcomes for Obesity/Weight and Nutrition
3.3.6. Secondary Outcomes for Obesity/Weight, Nutrition and Physical Activity
3.4. Quality Assessment
4. Discussion
4.1. Principal Findings
4.2. Secondary Outcomes
4.3. Quality Assessment
4.4. Strengths and Limitations of Review
4.5. Implications for Practice across SNAPO Risks
4.6. Future VR Intervention Research across SNAPO Risks
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- World Health Organization. Noncommunicable Diseases. 2021. Available online: https://www.who.int/news-room/fact-sheets/detail/noncommunicable-diseases (accessed on 16 June 2022).
- Australian Institute of Health and Welfare. Australian Burden of Disease Study 2018—Key Findings; Australian Institute of Health and Welfare: Canberra, Australia, 2021. [Google Scholar]
- World Health Organization. Tobacco. 2022. Available online: https://www.who.int/news-room/fact-sheets/detail/tobacco (accessed on 16 June 2022).
- Afshin, A.; Sur, P.J.; Fay, K.A.; Cornaby, L.; Ferrara, G.; Salama, J.S.; Mullany, E.C.; Abate, K.H.; Abbafati, C.; Abebe, Z.; et al. Health effects of dietary risks in 195 countries, 1990–2017: A systematic analysis for the Global Burden of Disease Study 2017. Lancet 2019, 393, 1958–1972. [Google Scholar] [CrossRef]
- World Health Organization. Alcohol. 2022. Available online: https://www.who.int/news-room/fact-sheets/detail/alcohol (accessed on 16 June 2022).
- Ritchie, H.; Roser, M. Obesity. Our World in Data. 2017. Available online: https://ourworldindata.org/obesity (accessed on 16 June 2022).
- Li, L.; Yu, F.; Shi, D.; Shi, J.; Tian, Z.; Yang, J.; Wang, X.; Jiang, Q. Application of virtual reality technology in clinical medicine. Am. J. Transl. Res. 2017, 9, 3867–3880. [Google Scholar]
- Heizenrader, L.L.C. The 3 Types of Virtual Reality. 2019. Available online: https://heizenrader.com/the-3-types-of-virtual-reality/#:~:text=There%20are%203%20primary%20categories,%2C%20and%20fully%2Dimmersive%20simulations (accessed on 7 July 2022).
- Corregidor-Sánchez, A.I.; Sergura-Fragoso, A.; Rodriguez-Hernández, M.; Criado-Alvarez, J.J.; González-Gonzalez, J.; Polonio-López, B. Can exergames contribute to improving walking capacity in older adults? A systematic review and meta-analysis. Maturitas 2020, 132, 40–48. [Google Scholar] [CrossRef] [PubMed]
- Corregidor-Sánchez, A.I.; Sergura-Fragoso, A.; Rodriguez-Hernández, M.; Jiménez-Rojas, C.; Polonio-López, B.; Criado-Álvarez, J.J. Effectiveness of virtual reality technology on functional mobility of older adults: Systematic review and meta-analysis. Age Ageing 2021, 50, 370–379. [Google Scholar] [CrossRef] [PubMed]
- Durl, J.; Dietrich, T.; Pang, B.; Potter, L.; Carter, L. Utilising virtual reality in alcohol studies: A systematic review. Health Educ. J. 2018, 77, 212–225. [Google Scholar] [CrossRef]
- Felipe, F.A.; de Carvalho, F.O.; Silva, E.R.; Santos, N.G.L.; Fontes, P.A.; de Almeida, A.S.; Garção, D.C.; Nunes, P.S.; Araújo, A.A.S. Evaluation instruments for physical therapy using virtual reality in stroke patients: A systematic review. Physiotherapy 2020, 106, 194–210. [Google Scholar] [CrossRef] [PubMed]
- García-Bravo, S.; Cuesta-Gómez, A.; Campuzano-Ruiz, R.; López-Navas, M.J.; Domínguez-Paniagua, J.; Araújo-Narváez, A.; Barreñada-Copete, E.; García-Bravo, C.; Flórez-García, M.T.; Botas-Rodríguez, J.; et al. Virtual reality and video games in cardiac rehabilitation programs. A systematic review. Disabil. Rehabil. 2021, 43, 448–457. [Google Scholar] [CrossRef]
- Ghiţă, A.; Gutiérrez-Maldonado, J. Applications of virtual reality in individuals with alcohol misuse: A systematic review. Addict. Behav. 2018, 81, 1–11. [Google Scholar] [CrossRef]
- Horne, M.; Hill, A.; Murells, T.; Ugail, H.; Irving; Chinnadorai, R.; Hardy, M. Using avatars in weight management settings: A systematic review. Internet Interv. 2020, 19, 100295. [Google Scholar] [CrossRef]
- Keijsers, M.; Vega-Corredor, M.C.; Tomintz, M.; Hoermann, S. Virtual reality technology use in cigarette craving and smoking interventions (i “virtually” quit): Systematic review. J. Med. Internet Res. 2021, 23, e24307. [Google Scholar] [CrossRef]
- Laver, K.E.; George, S.; Thomas, S.; Deutsch, J.E.; Crotty, M. Virtual reality for stroke rehabilitation. Cochrane Database Syst. Rev. 2017, 2017, CD008349. [Google Scholar] [CrossRef] [Green Version]
- Miller, K.J.; Adair, B.S.; Pearce, A.J.; Said, C.M.; Ozanne, E.; Morris, M.M. Effectiveness and feasibility of virtual reality and gaming system use at home by older adults for enabling physical activity to improve health-related domains: A systematic review. Age Ageing 2014, 43, 188–195. [Google Scholar] [CrossRef]
- Peng, W.; Crouse, J.C.; Lin, J.H. Using active video games for physical activity promotion: A systematic review of the current state of research. Health Educ. Behav. 2013, 40, 171–192. [Google Scholar] [CrossRef]
- Segawa, T.; Baudry, T.; Bourla, A.; Blanc, J.; Peretti, C.; Mouchabac, S.; Ferreri, F. Virtual reality (VR) in assessment and treatment of addictive disorders: A systematic review. Front. Neurosci. 2020, 13, 1409. [Google Scholar] [CrossRef]
- Trahan, M.H.; Maynard, B.R.; Smith, K.S.; Farina, A.S.J.; Khoo, Y.M. Virtual reality exposure therapy on alcohol and nicotine: A systematic review. Res. Soc. Work. Pract. 2019, 29, 876–891. [Google Scholar] [CrossRef]
- Tsamitros, N.; Sebold, M.; Gutwinski, S.; Back, A. Virtual reality-based treatment approaches in the field of substance use disorders. Curr. Addict. Rep. 2021, 8, 399–407. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffman, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ 2021, 372, n71. [Google Scholar] [CrossRef]
- Effective Public Health Practice Project. Quality Assessment Tool for Quantitative Studies. 2010. Available online: https://www.ephpp.ca/PDF/Quality%20Assessment%20Tool_2010_2.pdf (accessed on 15 December 2021).
- Effective Public Health Practice Project. Quality Assessment Tool for Quantitative Studies Dictionary. 2009. Available online: https://www.ephpp.ca/PDF/QADictionary_dec2009.pdf (accessed on 15 December 2021).
- Bordnick, P.S.; Traylor, A.C.; Carter, B.L.; Graap, K.M. A feasibility study of virtual reality-based coping skills training for nicotine dependence. Res. Soc. Work. Pract. 2012, 22, 293–300. [Google Scholar] [CrossRef]
- Goldenhersch, E.; Thrul, J.; Ungaretti, J.; Rosencovich, N.; Waitman, C.; Ceberio, M.R. Virtual reality smartphone-based intervention for smoking cessation: Pilot randomized controlled trial on initial clinical efficacy and adherence. J. Med. Internet Res. 2020, 22, e17571. [Google Scholar] [CrossRef] [PubMed]
- Karekla, M.; Savvides, S.N.; Gloster, A. An avatar-led intervention promotes smoking cessation in young adults: A pilot randomized clinical trial. Ann. Behav. Med. 2021, 54, 747–760. [Google Scholar] [CrossRef] [PubMed]
- Pericot-Valverde, I.; Secades-Villa, R.; Gutierrez-Maldonado, J. A randomized clinical trial of cue exposure treatment through virtual reality for smoking cessation. J. Subst. Abus. Treat. 2019, 96, 26–32. [Google Scholar] [CrossRef] [PubMed]
- Woodruff, S.I.; Conway, T.L.; Edwards, C.C.; Elliott, S.P.; Crittenden, J. Evaluation of an internet virtual world chat room for adolescent smoking cessation. Addict. Behav. 2007, 32, 1769–1786. [Google Scholar] [CrossRef] [PubMed]
- Basha, M.A.; Aboelnour, N.H.; Aly, S.M.; Kamel, F.A.H. Impact of kinect-based virtual reality training on physical fitness and quality of life in severely burned children: A monocentric randomized controlled trial. Ann. Phys. Rehabil. Med. 2020, 65, 101471. [Google Scholar] [CrossRef] [PubMed]
- Chuang, T.Y.; Sung, W.H.; Chang, H.A.; Wang, R.Y. Effect of a virtual reality-enhanced exercise protocol after coronary artery bypass grafting. Phys. Ther. 2006, 86, 1369–1377. [Google Scholar] [CrossRef]
- Chuang, T.Y.; Sung, W.H.; Lin, C.Y. Application of a virtual reality-enhanced exercise protocol in patients after coronary bypass. Arch. Phys. Med. Rehabil. 2005, 86, 1929–1932. [Google Scholar] [CrossRef]
- Friederichs, S.; Bolman, C.; Oenema, A.; Guyaux, J.; Lechner, L. Motivational interviewing in a Web-based physical activity intervention with an avatar: Randomized controlled trial. J. Med. Internet Res. 2014, 16, e48. [Google Scholar] [CrossRef]
- Gulsen, C.; Soke, F.; Eldemire, K.; Apaydin, Y.; Ozkul, C.; Guclu-Gunduz, A.; Akcali, D.T. Effect of fully immersive virtual reality treatment combined with exercise in fibromyalgia patients: A randomized controlled trial. Assist. Technol. 2022, 34, 256–263. [Google Scholar] [CrossRef]
- Karssemeijer, E.G.A.; Bosser, W.J.; Aaronson, J.A.; Sanders, L.M.J.; Kessels, R.P.C.; Rikkert, M.G.M.O. Exergaming as a physical exercise strategy reduces frailty in people with dementia: A randomized controlled trial. J. Am. Med. Dir. Assoc. 2019, 20, 1502–1508.e1. [Google Scholar] [CrossRef]
- Navarro, J.; Cebolla, A.; Llorens, R.; Borrego, A.; Baños, R.M. Manipulating self-avatar body dimensions in virtual worlds to complement an internet-delivered intervention to increase physical activity in overweight women. Int. J. Environ. Res. Public Health 2020, 17, 4045. [Google Scholar] [CrossRef]
- Ruiz, J.G.; Andrade, A.D.; Anam, R.; Aguiar, R.; Sun, H.; Roos, B.A. Using anthropomorphic avatars resembling sedentary older individuals as models to enhance self-efficacy and adherence to physical activity: Psychophysiological correlates. Stud. Health Technol. Inform. 2012, 173, 405–411. [Google Scholar]
- Thomas, S.; Fazakarley, L.; Thomas, P.W.; Collyer, S.; Brenton, S.; Perring, S.; Scott, R.; Thomas, F.; Thomas, C.; Jones, K.; et al. Mii-vitaliSe: A pilot randomised controlled trial of a home gaming system (Nintendo Wii) to increase activity levels, vitality and well-being in people with multiple sclerosis. BMJ Open 2017, 7, e016966. [Google Scholar] [CrossRef]
- Ulas, K.; Semin, I. The biological and motivational effects of aerobic exercise with virtual reality. Res. Q. Exerc. Sport 2021, 92, 321–326. [Google Scholar] [CrossRef]
- Villafaina, S.; Borrega-Mouquinho, Y.; Fuentes-García, J.P.; Collado-Mateo, D.; Gusi, N. Effect of exergame training and detraining on lower-body strength, agility, and cardiorespiratory fitness in women with fibromyalgia: Single-blinded randomized controlled trial. Int. J. Environ. Res. Public Health 2020, 17, 161. [Google Scholar] [CrossRef]
- Wilzeck, V.C.; Hufschmid, J.; Bischof, L.; Hansi, C.; Nägele, M.P.; Beer, J.J.; Hufschmid, U. A significant increase in exercise test performance with virtual group motivation: A randomised open-label controlled trial. Swiss Med. Wkly. 2020, 150, w20287. [Google Scholar] [CrossRef]
- Behm-Morawitz, E.; Lewallen, J.; Choi, G. A second chance at health: How a 3D virtual world can improve health self-efficacy for weight loss management among adults. Cyberpsychology Behav. Soc. Netw. 2016, 19, 74–79. [Google Scholar] [CrossRef]
- Cesa, G.L.; Manzoni, G.M.; Bacchetta, M.; Castelnuovo, G.; Conti, S.; Gaggioli, A.; Mantovani, F.; Molinari, E.; Cárdenas-López, G.; Riva, G. Virtual reality for enhancing the cognitive behavioral treatment of obesity with binge eating disorder: Randomized controlled study with one-year follow-up. J. Med. Internet Res. 2013, 15, e113. [Google Scholar] [CrossRef]
- Ferraz, D.D.; Trippo, K.V.; Duarte, G.P.; Neto, M.G.; Santos, K.O.B.; Filho, J.O. The effects of functional training, bicycle exercise, and exergaming on walking capacity of elderly patients with Parkinson disease: A pilot randomized controlled single-blinded trial. Arch. Phys. Med. Rehabil. 2018, 99, 826–833. [Google Scholar] [CrossRef]
- Manzoni, G.M.; Cesa, G.L.; Bacchetta, M.; Castelnuovo, G.; Conti, S.; Gaggioli, A.; Mantovani, F.; Molinari, E.; Cárdenas-López, G.; Riva, G. Virtual reality-enhanced cognitive-behavioral therapy for morbid obesity: A randomized controlled study with 1 year follow-up. Cyberpsychology Behav. Soc. Netw. 2016, 19, 134–140. [Google Scholar] [CrossRef]
- Manzoni, G.M.; Pagnini, F.; Gorini, A.; Preziosa, A.; Castelnuovo, G.; Molinari, E.; Riva, G. Can relaxation training reduce emotional eating in women with obesity? An exploratory study with 3 months of follow-up. J. Am. Diet. Assoc. 2009, 109, 1427–1432. [Google Scholar] [CrossRef]
- Warburton, D.E.R.; Bredin, S.S.D.; Horita, L.T.L.; Zbogar, D.; Scott, J.M.; Esch, B.T.A.; Rhodes, R.E. The health benefits of interactive video game exercise. Appl. Physiol. Nutr. Metab. 2007, 32, 655–663. [Google Scholar] [CrossRef]
- Adamo, K.B.; Rutherford, J.A.; Goldfield, G.S. Effects of interactive video game cycling on overweight and obese adolescent health. Appl. Physiol. Nutr. Metab. 2010, 35, 805–815. [Google Scholar] [CrossRef]
- Vieira, A.S.D.S.; de Melo, M.C.D.A.; Noites, S.P.A.R.S.; Machado, J.; Gabriel, M.M. The effect of virtual reality on a home-based cardiac rehabilitation program on body composition, lipid profile and eating patterns: A randomized controlled trial. Eur. J. Integr. Med. 2017, 9, 69–78. [Google Scholar] [CrossRef]
- Sullivan, D.K.; Goetz, J.R.; Gibson, C.A.; Washburn, R.A.; Smith, B.K.; Lee, J.; Gerald, S.; Fincham, T.; Donnelly, J.E. Improving weight maintenance using virtual reality (second life). J. Nutr. Educ. Behav. 2013, 45, 264–268. [Google Scholar] [CrossRef] [PubMed]
- Brueton, V.C.; Tierney, J.F.; Stenning, S.; Meredith, S.; Harding, S.; Nazareth, I.; Rait, G. Strategies to improve retention in randomised trials: A Cochrane systematic review and meta-analysis. BMJ Open 2014, 4, e003821. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| Selection Bias | Study Design | Confounders | Blinding | Data Collection | Withdrawals and Dropouts | Global Rating | |
|---|---|---|---|---|---|---|---|
| Smoking | |||||||
| Bordnick et al. [26] | Weak | Strong | Strong | Weak | Moderate | Weak | Weak |
| Goldenhersch et al. [27] | Weak | Strong | Strong | Weak | Weak | Weak | Weak |
| Karekla et al. [28] | Weak | Strong | Strong | Weak | Weak | Weak | Weak |
| Pericot-Valverde et al. [29] | Weak | Strong | Strong | Weak | Strong | Strong | Weak |
| Woodruff et al. [30] | Weak | Strong | Weak | Weak | Weak | Moderate | Weak |
| Physical Activity | |||||||
| Basha et al. [31] | Weak | Strong | Strong | Moderate | Strong | Strong | Moderate |
| Chuang et al. [32] | Weak | Strong | Strong | Weak | Strong | Strong | Weak |
| Chuang et al. [33] | Weak | Strong | Strong | Weak | Strong | Moderate | Weak |
| Friederichs et al. [34] | Weak | Strong | Weak | Weak | Moderate | Weak | Weak |
| Gulsen et al. [35] | Weak | Strong | Strong | Moderate | Weak | Strong | Weak |
| Karssemeijer et al. [36] | Weak | Strong | Strong | Weak | Weak | Strong | Weak |
| Navarro et al. [37] | Weak | Strong | Weak | Moderate | Strong | Strong | Weak |
| Ruiz et al. [38] | Weak | Strong | Strong | Weak | Strong | Strong | Weak |
| Thomas et al. [39] | Weak | Strong | Weak | Weak | Weak | Strong | Weak |
| Ulas and Semin [40] | Weak | Strong | Strong | Weak | Strong | Weak | Weak |
| Villafaina et al. [41] | Weak | Strong | Strong | Moderate | Weak | Moderate | Weak |
| Wilzeck et al. [42] | Weak | Strong | Strong | Weak | Strong | Strong | Weak |
| Physical Activity and Obesity/Weight | |||||||
| Adamo et al. [49] | Weak | Strong | Strong | Weak | Strong | Strong | Weak |
| Obesity/Weight | |||||||
| Behm-Morawitz et al. [43] | Weak | Strong | Weak | Weak | Strong | Weak | Weak |
| Cesa et al. [44] | Weak | Strong | Strong | Moderate | Strong | Moderate | Moderate |
| Ferraz et al. [45] | Weak | Strong | Strong | Moderate | Strong | Strong | Moderate |
| Manzoni et al. [46] | Weak | Strong | Weak | Weak | Strong | Weak | Weak |
| Manzoni et al. [47] | Weak | Strong | Strong | Weak | Weak | Moderate | Weak |
| Warburton et al. [48] | Weak | Strong | Strong | Moderate | Strong | Weak | Weak |
| Obesity/Weight and Nutrition | |||||||
| Vieira et al. [50] | Weak | Strong | Strong | Weak | Moderate | Moderate | Weak |
| Obesity/Weight, Nutrition and Physical Activity | |||||||
| Sullivan et al. [51] | Weak | Strong | Strong | Weak | Weak | Weak | Weak |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tatnell, P.; Atorkey, P.; Tzelepis, F. The Effectiveness of Virtual Reality Interventions on Smoking, Nutrition, Alcohol, Physical Activity and/or Obesity Risk Factors: A Systematic Review. Int. J. Environ. Res. Public Health 2022, 19, 10821. https://doi.org/10.3390/ijerph191710821
Tatnell P, Atorkey P, Tzelepis F. The Effectiveness of Virtual Reality Interventions on Smoking, Nutrition, Alcohol, Physical Activity and/or Obesity Risk Factors: A Systematic Review. International Journal of Environmental Research and Public Health. 2022; 19(17):10821. https://doi.org/10.3390/ijerph191710821
Chicago/Turabian StyleTatnell, Peter, Prince Atorkey, and Flora Tzelepis. 2022. "The Effectiveness of Virtual Reality Interventions on Smoking, Nutrition, Alcohol, Physical Activity and/or Obesity Risk Factors: A Systematic Review" International Journal of Environmental Research and Public Health 19, no. 17: 10821. https://doi.org/10.3390/ijerph191710821
APA StyleTatnell, P., Atorkey, P., & Tzelepis, F. (2022). The Effectiveness of Virtual Reality Interventions on Smoking, Nutrition, Alcohol, Physical Activity and/or Obesity Risk Factors: A Systematic Review. International Journal of Environmental Research and Public Health, 19(17), 10821. https://doi.org/10.3390/ijerph191710821








