Arsenic Oxidation and Removal from Water via Core–Shell MnO2@La(OH)3 Nanocomposite Adsorption
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of MnO2@La(OH)3 Nanocomposite
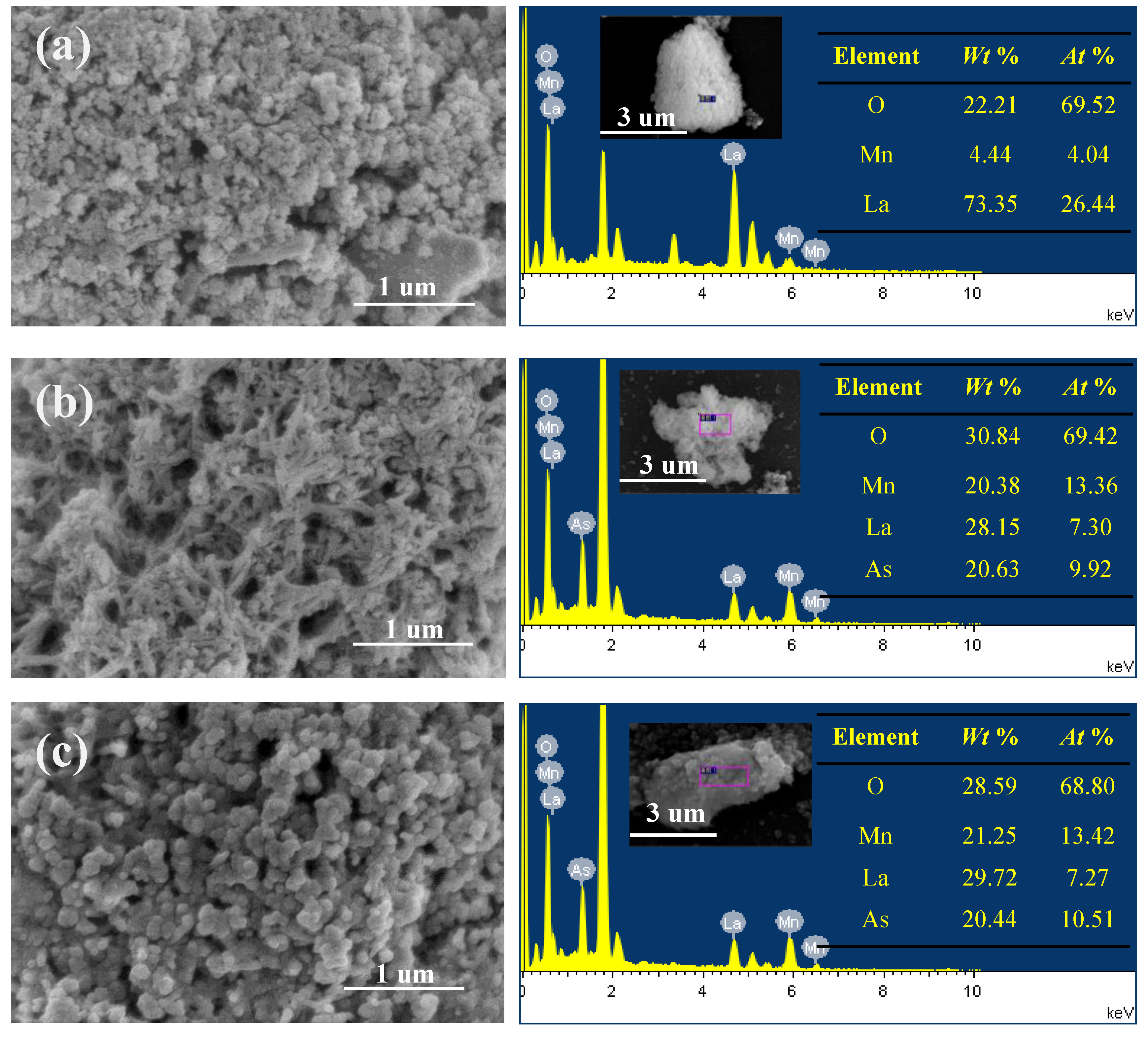
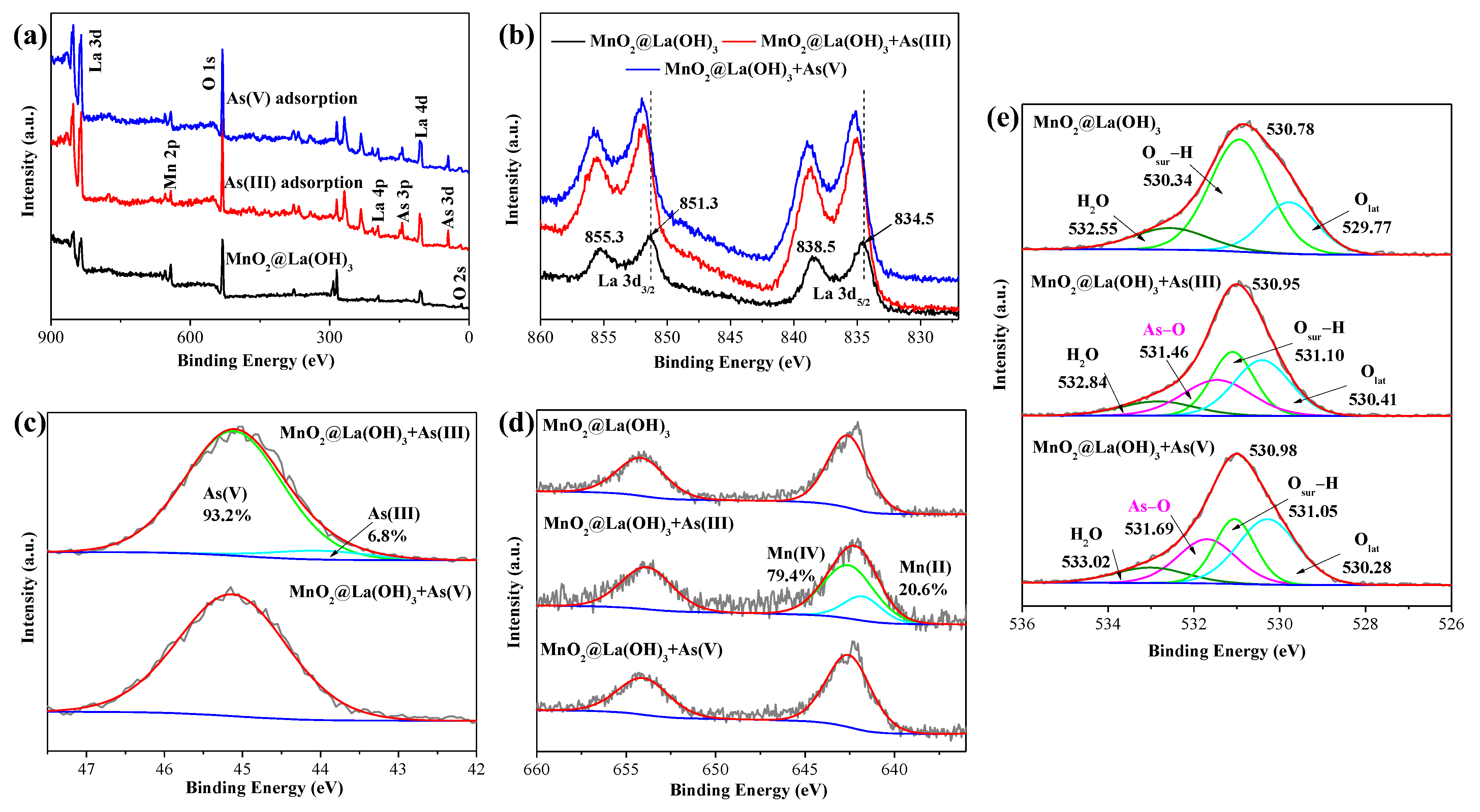
2.3. Characterization of Adsorbents before and after as Removal
2.4. Batch Adsorption Experiments
2.5. Statistical Analysis
3. Results and Discussion
3.1. Characterization of MnO2@La(OH)3 Nanocomposite
3.1.1. SEM and TEM Analysis
3.1.2. Specific Surface Area
3.1.3. Point of Zero Charge
3.2. Arsenic Adsorption Performance
3.2.1. Adsorption Kinetics
3.2.2. Effect of Adsorbent Dosage
3.2.3. Effect of Solution pH
3.2.4. Adsorption Isotherms
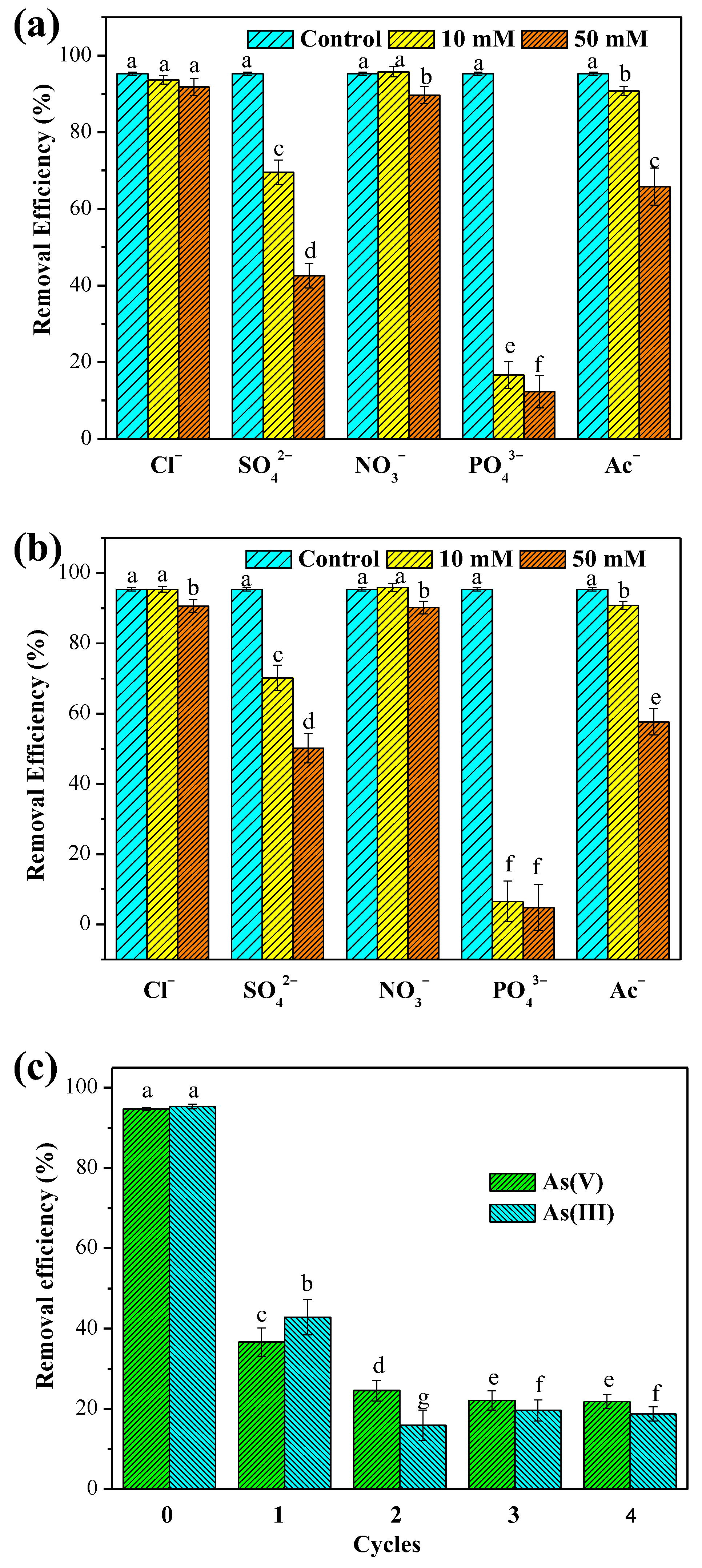
3.2.5. Effect of Co-Existing Oxyanions
3.2.6. Regeneration and Reusability
3.3. Adsorption Mechanisms
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Yang, L.; Ren, Q.; Ge, S.; Jiao, Z.; Zhan, W.; Hou, R.; Ruan, X.; Pan, Y.; Wang, Y. Metal(loid)s spatial distribution, accumulation, and potential health risk assessment in soil-wheat systems near a Pb/Zn smelter in Henan province, Central China. Int. J. Environ. Res. Public Health 2022, 19, 2527. [Google Scholar] [CrossRef] [PubMed]
- Smedley, P.L.; Kinniburgh, D.G. A review of the source, behaviour and distribution of arsenic in natural waters. Appl. Geochem. 2002, 17, 517–568. [Google Scholar] [CrossRef]
- Cui, J.; Jing, C. A review of arsenic interfacial geochemistry in groundwater and the role of organic matter. Ecotoxicol. Environ. Saf. 2019, 183, 109550. [Google Scholar] [CrossRef] [PubMed]
- Podgorski, J.; Berg, M. Global threat of arsenic in groundwater. Science 2020, 368, 845–850. [Google Scholar] [CrossRef]
- Rodriguez-Lado, L.; Sun, G.F.; Berg, M.; Zhang, Q.; Xue, H.B.; Zheng, Q.M.; Johnson, C.A. Groundwater arsenic contamination throughout China. Science 2013, 341, 866–868. [Google Scholar] [CrossRef]
- Bandaru, S.R.S.; van Genuchten, C.M.; Kumar, A.; Glade, S.; Hernandez, D.; Nahata, M.; Gadgil, A. Rapid and efficient arsenic removal by iron electrocoagulation enabled with in situ generation of hydrogen peroxide. Environ. Sci. Technol. 2020, 54, 6094–6103. [Google Scholar] [CrossRef]
- Sandoval, M.A.; Fuentes, R.; Thiam, A.; Salazar, R. Arsenic and fluoride removal by electrocoagulation process: A general review. Sci. Total Environ. 2021, 753, 142108. [Google Scholar] [CrossRef]
- Hong, J.; Zhu, Z.; Lu, H.; Qiu, Y. Synthesis and arsenic adsorption performances of ferric-based layered double hydroxide with α-alanine intercalation. Chem. Eng. J. 2014, 252, 267–274. [Google Scholar] [CrossRef]
- Ortega, A.; Oliva, I.; Contreras, K.E.; Gonzalez, I.; Cruz-Diaz, M.R.; Rivero, E.P. Arsenic removal from water by hybrid electro-regenerated anion exchange resin/electrodialysis process. Sep. Purif. Technol. 2017, 184, 319–326. [Google Scholar] [CrossRef]
- Lin, S.; Yang, H.J.; Na, Z.Y.; Lin, K.F. A novel biodegradable arsenic adsorbent by immobilization of iron oxyhydroxide (FeOOH) on the root powder of long-root Eichhornia crassipes. Chemosphere 2018, 192, 258–266. [Google Scholar] [CrossRef]
- Xue, Q.; Ran, Y.; Tan, Y.Z.; Peacock, C.L.; Du, H.H. Arsenite and arsenate binding to ferrihydrite organo-mineral coprecipitate: Implications for arsenic mobility and fate in natural environments. Chemosphere 2019, 224, 103–110. [Google Scholar] [CrossRef] [PubMed]
- Jiang, X.L.; Peng, C.J.; Fu, D.; Chen, Z.; Shen, L.; Li, Q.B.; Ouyang, T.; Wang, Y.P. Removal of arsenate by ferrihydrite via surface complexation and surface precipitation. Appl. Surf. Sci. 2015, 353, 1087–1094. [Google Scholar] [CrossRef]
- Gao, X.D.; Root, R.A.; Farrell, J.; Ela, W.; Chorover, J. Effect of silicic acid on arsenate and arsenite retention mechanisms on 6-L ferrihydrite: A spectroscopic and batch adsorption approach. Appl. Geochem. 2013, 38, 110–120. [Google Scholar] [CrossRef]
- Matsui, Y.; Shirasaki, N.; Yamaguchi, T.; Kondo, K.; Machida, K.; Fukuura, T.; Matsushita, T. Characteristics and components of poly-aluminum chloride coagulants that enhance arsenate removal by coagulation: Detailed analysis of aluminum species. Water Res. 2017, 118, 177–186. [Google Scholar] [CrossRef] [PubMed]
- Mohan, D.; Pittman, C.U. Arsenic removal from water/wastewater using adsorbents—A critical review. J. Hazard. Mater. 2007, 142, 1–53. [Google Scholar] [CrossRef] [PubMed]
- Shi, Q.; Yan, L.; Chan, T.; Jing, C. Arsenic adsorption on lanthanum-impregnated activated alumina: Spectroscopic and DFT study. Acs. Appl. Mater. Inter. 2015, 7, 26735–26741. [Google Scholar] [CrossRef] [PubMed]
- Alka, S.; Shahir, S.; Ibrahim, N.; Ndejiko, M.J.; Vo, D.-V.N.; Abd Manan, F. Arsenic removal technologies and future trends: A mini review. J. Clean. Prod. 2021, 278, 123805. [Google Scholar] [CrossRef]
- Luo, J.; Meng, X.; Crittenden, J.; Qu, J.; Hu, C.; Liu, H.; Peng, P. Arsenic adsorption on α-MnO2 nanofibers and the significance of (100) facet as compared with (110). Chem. Eng. J. 2018, 331, 492–500. [Google Scholar] [CrossRef]
- Zhang, D.; Jia, Y.; Ma, J.; Li, Z. Removal of arsenic from water by Friedel’s salt (FS: 3CaO∙Al2O3∙CaCl2∙10H2O). J. Hazard. Mater. 2011, 195, 398–404. [Google Scholar] [CrossRef]
- Mertens, J.; Rose, J.; Kaegi, R.; Chaurand, P.; Ploetze, M.; Wehrli, B.; Furrer, G. Adsorption of arsenic on polyaluminum granulate. Environ. Sci. Technol. 2012, 46, 7310–7317. [Google Scholar] [CrossRef]
- Chiu, V.Q.; Hering, J.G. Arsenic adsorption and oxidation at manganite surfaces. 1. Method for simultaneous determination of adsorbed and dissolved arsenic species. Environ. Sci. Technol. 2000, 34, 2029–2034. [Google Scholar] [CrossRef]
- Zhang, D.; Cao, R.; Wang, Y.; Wang, S.; Jia, Y. The adsorption of As(V) on poorly crystalline Fe oxyhydroxides, revisited: Effect of the reaction media and the drying treatment. J. Hazard. Mater. 2021, 416, 125863. [Google Scholar] [CrossRef] [PubMed]
- Ge, X.; Ma, Y.; Song, X.; Wang, G.; Zhang, H.; Zhang, Y.; Zhao, H. β-FeOOH nanorods/carbon foam-based hierarchically porous monolith for highly effective arsenic removal. Acs. Appl. Mater. Inter. 2017, 9, 13480–13490. [Google Scholar] [CrossRef] [PubMed]
- Pervez, M.N.; Wei, Y.; Sun, P.; Qu, G.; Naddeo, V.; Zhao, Y. α-FeOOH quantum dots impregnated graphene oxide hybrids enhanced arsenic adsorption: The mediation role of environmental organic ligands. Sci. Total Environ. 2021, 781, 146726. [Google Scholar] [CrossRef]
- Rahman, M.A.; Lamb, D.; Kunhikrishnan, A.; Rahman, M.M. Kinetics, isotherms and adsorption-desorption behavior of phosphorus from aqueous solution using zirconium-iron and iron modified biosolid biochars. Water 2021, 13, 3320. [Google Scholar] [CrossRef]
- Yang, X.; Huang, G.; An, C.; Chen, X.; Shen, J.; Yin, J.; Song, P.; Xu, Z.; Li, Y. Removal of arsenic from water through ceramic filter modified by nano-CeO2: A cost-effective approach for remote areas. Sci. Total Environ. 2021, 750, 141510. [Google Scholar] [CrossRef]
- Benis, K.Z.; Sokhansanj, A.; Norberto, J.; McPhedran, N.K.; Soltan, J. A binary oxide-biochar composite for adsorption of arsenic from aqueous solutions: Combined microwave pyrolysis and electrochemical modification. Chem. Eng. J. 2022, 446, 137024. [Google Scholar] [CrossRef]
- Zhang, W.; Fu, J.; Zhang, G.; Zhang, X. Enhanced arsenate removal by novel Fe–La composite (hydr)oxides synthesized via coprecipitation. Chem. Eng. J. 2014, 251, 69–79. [Google Scholar] [CrossRef]
- Lingamdinne, L.P.; Koduru, J.R.; Chang, Y.-Y.; Kang, S.-H.; Yang, J.-K. Facile synthesis of flowered mesoporous graphene oxide-lanthanum fluoride nanocomposite for adsorptive removal of arsenic. J. Mol. Liq. 2019, 279, 32–42. [Google Scholar] [CrossRef]
- Wang, Y.; Liu, Y.; Guo, T.; Liu, H.; Li, J.; Wang, S.; Li, X.; Wang, X.; Jia, Y. Lanthanum hydroxide: A highly efficient and selective adsorbent for arsenate removal from aqueous solution. Environ. Sci. Pollut. Res. 2020, 27, 42868–42880. [Google Scholar] [CrossRef]
- Yan, B.; Liang, T.; Yang, X.; Gadgil, A.J. Superior removal of As(III) and As(V) from water with Mn-doped β-FeOOH nanospindles on carbon foam. J. Hazard. Mater. 2021, 418, 126347. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Qiao, Q.; Tan, W.; Sun, X.; Liu, C.; Dang, Z.; Qiu, G. Arsenic detoxification by iron-manganese nodules under electrochemically controlled redox: Mechanism and application. J. Hazard. Mater. 2021, 403, 123912. [Google Scholar] [CrossRef] [PubMed]
- Xie, X.J.; Lu, C.; Xu, R.; Yang, X.Q.; Yan, L.; Su, C.L. Arsenic removal by manganese-doped mesoporous iron oxides from groundwater: Performance and mechanism. Sci. Total Environ. 2022, 806, 150615. [Google Scholar] [CrossRef] [PubMed]
- Biswas, B.K.; Inoue, K.; Ghimire, K.N.; Kawakita, H.; Ohto, K.; Harada, H. Effective removal of arsenic with lanthanum(III)- and cerium(III)-loaded orange waste gels. Sep. Sci. Technol. 2008, 43, 2144–2165. [Google Scholar] [CrossRef]
- Ding, W.; Wan, X.; Zheng, H.; Wu, Y.; Muhammad, S. Sulfite-assisted oxidation/adsorption coupled with a TiO2 supported CuO composite for rapid arsenic removal: Performance and mechanistic studies. J. Hazard. Mater. 2021, 413, 125449. [Google Scholar] [CrossRef]
- Su, J.; Lyu, T.; Cooper, M.; Mortimer, R.J.G.; Pan, G. Efficient arsenic removal by a bifunctional heterogeneous catalyst through simultaneous hydrogen peroxide (H2O2) catalytic oxidation and adsorption. J. Clean. Prod. 2021, 325, 125449. [Google Scholar] [CrossRef]
- Wei, Y.; Yang, W.; Wang, Z.; Yin, K.; Chen, T.; Liu, C. Enhanced removal of As(III) by heterogeneous catalytic oxidation of As(III) on Fe-biochar fibers with H2O2 and hydroxylamine. Chem. Eng. J. 2022, 428, 131200. [Google Scholar] [CrossRef]
- Li, X.j.; Liu, C.; Li, F.; Li, Y.; Zhang, L.; Liu, C.; Zhou, Y. The oxidative transformation of sodium arsenite at the interface of α-MnO2 and water. J. Hazard. Mater. 2010, 173, 675–681. [Google Scholar] [CrossRef]
- Su, J.; Lyu, T.; Yi, H.; Bi, L.; Pan, G. Superior arsenate adsorption and comprehensive investigation of adsorption mechanism on novel Mn-doped La2O2CO3 composites. Chem. Eng. J. 2020, 391, 123623. [Google Scholar] [CrossRef]
- Wang, Y.; Liu, H.; Wang, S.; Li, X.; Wang, X.; Jia, Y. Simultaneous removal and oxidation of arsenic from water by δ-MnO2 modified activated carbon. J. Environ. Sci. 2020, 94, 147–160. [Google Scholar] [CrossRef]
- Zhang, G.; Qu, J.; Liu, H.; Liu, R.; Wu, R. Preparation and evaluation of a novel Fe-Mn binary oxide adsorbent for effective arsenite removal. Water Res. 2007, 41, 1921–1928. [Google Scholar] [CrossRef] [PubMed]
- Zhang, G.; Zhang, D.; Yuan, Z.; Ma, X.; Lei, L.; Wu, X.; Lin, J.; Wang, X.; Wang, S.; Jia, Y. Fate of adsorbed arsenic during early stage sulfidization of nano-ferrihydrite. Environ. Sci.-Nano 2019, 6, 2228–2240. [Google Scholar] [CrossRef]
- Xu, L.; Zhao, Z.; Wang, S.; Pan, R.; Jia, Y. Transformation of arsenic in offshore sediment under the impact of anaerobic microbial activities. Water Res. 2011, 45, 6781–6788. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Xie, X.; Chen, X.; Huang, C.; Yang, S. Biochar-loaded Ce3+-enriched ultra-fine ceria nanoparticles for phosphate adsorption. J. Hazard. Mater. 2020, 396, 122626. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.; Chen, J.P. Fabrication and performance of a Mn-La metal composite for remarkable decontamination of fluoride. J. Mater. Chem. A 2014, 2, 8086–8093. [Google Scholar] [CrossRef]
- Paledi, U.; Allahkarami, E.; Rezai, B.; Aslani, M.R. Selectivity index and separation efficiency prediction in industrial magnetic separation process using a hybrid neural genetic algorithm. SN Appl. Sci. 2021, 3, 351. [Google Scholar] [CrossRef]
- Allahkarami, E.; Monfared, A.D.; Silva, L.F.O.; Dotto, G.L. Lead ferrite-activated carbon magnetic composite for efficient removal of phenol from aqueous solutions: Synthesis, characterization, and adsorption studies. Sci. Rep. 2022, 12, 10718. [Google Scholar] [CrossRef] [PubMed]
- Seoane, R.; Santaeufemia, S.; Abalde, J.; Torres, E. Efficient removal of methylene blue using living biomass of the microalga chlamydomonas moewusii: Kinetics and equilibrium studies. Int. J. Environ. Res. Public Health 2022, 19, 2653. [Google Scholar] [CrossRef]
- Yin, X.; Li, X.; Petropoulos, E.; Feng, Y.; Yang, B.; Xue, L.; Yang, L.; He, S. Phosphate removal from actual wastewater via La(OH)3-C3N4 adsorption: Performance, mechanisms and applicability. Sci. Total Environ. 2022, 814, 152971. [Google Scholar] [CrossRef]
- Zhang, H.; Wu, A.; Fu, H.; Zhang, L.; Liu, H.; Zheng, S.; Wan, H.; Xu, Z. Efficient removal of Pb(II) ions using manganese oxides: The role of crystal structure. RSC Adv. 2017, 7, 41228–41240. [Google Scholar] [CrossRef] [Green Version]
- Fang, L.; Shi, Q.; Nguyen, J.; Wu, B.; Wang, Z.; Lo, I.M.C. Removal mechanisms of phosphate by lanthanum hydroxide nanorods: Investigations using EXAFS, ATR-FTIR, DFT, and surface complexation modeling approaches. Environ. Sci. Technol. 2017, 51, 12377–12384. [Google Scholar] [CrossRef] [PubMed]
- Xu, R.; Zhang, M.; Mortimer, R.J.G.; Pan, G. Enhanced phosphorus locking by novel lanthanum/aluminum-hydroxide composite: Implications for eutrophication control. Environ. Sci. Technol. 2017, 51, 3418–3425. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Xiang, C.; Zhang, G.; Wang, H.; Ji, Q.; Qu, J. Activation of lattice oxygen in LaFe (oxy)hydroxides for efficient phosphorus removal. Environ. Sci. Technol. 2019, 53, 9073–9080. [Google Scholar] [CrossRef] [PubMed]
- Guo, S.; Sun, W.; Yang, W.; Li, Q.; Shang, J.K. Superior As(III) removal performance of hydrous MnOOH nanorods from water. RSC Adv. 2015, 5, 53280–53288. [Google Scholar] [CrossRef]
- Yu, Y.; Zhang, C.; Yang, L.; Chen, J.P. Cerium oxide modified activated carbon as an efficient and effective adsorbent for rapid uptake of arsenate and arsenite: Material development and study of performance and mechanisms. Chem. Eng. J. 2017, 315, 630–638. [Google Scholar] [CrossRef]
- Zhang, T.; Zhao, Y.; Kang, S.; Bai, H.; Song, G.; Zhang, Q. Enhanced arsenic removal from water by mechanochemical synthesis of Ca-Al-Fe ternary composites. J. Clean. Prod. 2021, 321, 128959. [Google Scholar] [CrossRef]
- Swedlund, P.J.; Holtkamp, H.; Song, Y.; Daughney, C.J. Arsenate-ferrihydrite systems from minutes to months: A macroscopic and IR spectroscopic study of an elusive equilibrium. Environ. Sci. Technol. 2014, 48, 2759–2765. [Google Scholar] [CrossRef]
- Wu, B.; Fang, L.; Fortner, J.D.; Guan, X.; Lo, I.M.C. Highly efficient and selective phosphate removal from wastewater by magnetically recoverable La(OH)3/Fe3O4 nanocomposites. Water Res. 2017, 126, 179–188. [Google Scholar] [CrossRef]
- Zhang, L.; Zhou, Q.; Liu, J.; Chang, N.; Wan, L.; Chen, J. Phosphate adsorption on lanthanum hydroxide-doped activated carbon fiber. Chem. Eng. J. 2012, 185, 160–167. [Google Scholar] [CrossRef]
- Xie, J.; Wang, Z.; Lu, S.; Wu, D.; Zhang, Z.; Kong, H. Removal and recovery of phosphate from water by lanthanum hydroxide materials. Chem. Eng. J. 2014, 254, 163–170. [Google Scholar] [CrossRef]
- Zhang, L.; Liu, Y.; Wang, Y.; Li, X.; Wang, Y. Investigation of phosphate removal mechanisms by a lanthanum hydroxide adsorbent using p-XRD, FTIR and XPS. Appl. Surf. Sci. 2021, 557, 149838. [Google Scholar] [CrossRef]
- Li, J.; Gyoten, H.; Sonoda, A.; Feng, Q.; Xue, M. Removal of trace arsenic to below drinking water standards using a Mn-Fe binary oxide. RSC Adv. 2017, 7, 1490–1497. [Google Scholar] [CrossRef]
- Yang, X.; Liu, S.; Liang, T.; Yan, X.; Zhang, Y.; Zhou, Y.; Sarkar, B.; Ok, Y.S. Ball-milled magnetite for efficient arsenic decontamination: Insights into oxidation-adsorption mechanism. J. Hazard. Mater. 2022, 427, 128117. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.; Lou, Z.; Liu, Y.; Fu, R.; Baig, S.A.; Xu, X. Adsorption behavior and removal mechanism of arsenic on graphene modified by iron-manganese binary oxide (FeMnOx/RGO) from aqueous solutions. RSC Adv. 2015, 5, 67951–67961. [Google Scholar] [CrossRef]
- Yu, L.; Ma, Y.; Ong, C.N.; Xie, J.; Liu, Y. Rapid adsorption removal of arsenate by hydrous cerium oxide-graphene composite. RSC Adv. 2015, 5, 64983–64990. [Google Scholar] [CrossRef]
- Neupane, G.; Donahoe, R.J.; Arai, Y. Kinetics of competitive adsorption/desorption of arsenate and phosphate at the ferrihydrite-water interface. Chem. Geol. 2014, 368, 31–38. [Google Scholar] [CrossRef]
- Chen, L.; Li, Y.; Sun, Y.; Chen, Y.; Qian, J. La(OH)3 loaded magnetic mesoporous nanospheres with highly efficient phosphate removal properties and superior pH stability. Chem. Eng. J. 2019, 360, 342–348. [Google Scholar] [CrossRef]
- Mueller, K.; Ciminelli, V.S.T.; Dantas, M.S.S.; Willscher, S. A comparative study of As(III) and As(V) in aqueous solutions and adsorbed on iron oxy-hydroxides by Raman spectroscopy. Water Res. 2010, 44, 5660–5672. [Google Scholar] [CrossRef]
- Yin, H.; Yang, P.; Kong, M.; Li, W. Preparation of the lanthanum-aluminum-amended attapulgite composite as a novel inactivation material to immobilize phosphorus in lake sediment. Environ. Sci. Technol. 2020, 54, 11602–11610. [Google Scholar] [CrossRef]
- Tang, L.; Feng, H.; Tang, J.; Zeng, G.; Deng, Y.; Wang, J.; Liu, Y.; Zhou, Y. Treatment of arsenic in acid wastewater and river sediment by Fe@Fe2O3 nanobunches: The effect of environmental conditions and reaction mechanism. Water Res. 2017, 117, 175–186. [Google Scholar] [CrossRef]
- Cai, G.; Tian, Y.; Li, D.; Zhang, J.; Li, L.; Wang, Q.; Sun, H.; Zhang, H.; Wang, P. Self-enhanced and efficient removal of As(III) from water using Fe-Cu-Mn composite oxide under visible-light irradiation: Synergistic oxidation and mechanisms. J. Hazard. Mater. 2022, 422, 126908. [Google Scholar] [CrossRef] [PubMed]
- Lin, L.; Qiu, W.; Wang, D.; Huang, Q.; Song, Z.; Chau, H.W. Arsenic removal in aqueous solution by a novel Fe-Mn modified biochar composite: Characterization and mechanism. Ecotoxicol. Environ. Saf. 2017, 144, 514–521. [Google Scholar] [CrossRef] [PubMed]




| Arsenic | Pseudo-First-Order Kinetic Model | Pseudo-Second-Order Kinetic Model | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| k1 (h−1) | qe (mg/g) | R2 | MSE (mg/g)2 | ARE (%) | k2 (g/(mg h)) | qe (mg/g) | R2 | MSE (mg/g)2 | ARE (%) | |
| As(V) | 0.846 | 117.33 | 0.858 | 200.77 | 18.07 | 0.00872 | 129.87 | 0.995 | 100.83 | 12.52 |
| As(III) | 0.098 | 140.54 | 0.993 | 14.79 | 11.41 | 0.00946 | 127.82 | 0.892 | 892.61 | 262.22 |
| Arsenic | Langmuir Model | Freundlich Model | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| kL (L/mg) | qmax (mg/g) | R2 | MSE (mg/g)2 | ARE (%) | kF ((mg/g)·(mg/L)−1/n) | 1/n | R2 | MSE (mg/g)2 | ARE (%) | |
| As(V) | 3.429 | 138.9 | 0.994 | 837.37 | 13.65 | 116.52 | 0.0521 | 0.990 | 1.51 | 0.80 |
| As(III) | 6.501 | 139.9 | 0.999 | 1330.3 | 33.75 | 126.23 | 0.0423 | 0.991 | 78.12 | 7.37 |
| Adsorbent | pH | qmax (mg/g) | Ref. | |
|---|---|---|---|---|
| As(V) | As(III) | |||
| MnO2@La(OH)3 | 4.0 | 138.9 | 139.9 | This study |
| Mg-Fe-Ala-LDH | 6.0 | 49.8 | 23.6 | [8] |
| Ferrihydrite | 3.0 | 142.86 | n.a. | [12] |
| Fe–Mn composite | 5.0 | 69.75 | 132.75 | [41] |
| β-FeOOH NRs/CF monolith | 6.0 | 172.9 | 103.4 | [23] |
| CF@Mn-FeOOH | 7.0 | 107.3 | 152.5 | [31] |
| Fe–Mn composite oxide | 7.0 | 31.68 | 59.44 | [33] |
| α-FeOOH QDs@GO | n.a. | 42.54 | 147.38 | [24] |
| Mn–Fe binary oxide a | – | 50 | 50 | [62] |
| Ca–Al–Fe ternary composites | n.a. | n.a. | 56.86 | [56] |
| Cerium oxide modified activated carbon | 5.0 | 43.6 | 36.8 | [55] |
| Ball-milled magnetite | n.a. | 3.2 | 5.8 | [63] |
| Cu–TiO2 | 7.5 | 19.719 | 24.244 | [35] |
| FeMnOx/RGO | 7.0 | 49.01 | 47.5 | [64] |
| Hydrous cerium oxide modified graphene | 4.0 | 62.33 | n.a. | [65] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, Y.; Guo, C.; Zhang, L.; Lu, X.; Liu, Y.; Li, X.; Wang, Y.; Wang, S. Arsenic Oxidation and Removal from Water via Core–Shell MnO2@La(OH)3 Nanocomposite Adsorption. Int. J. Environ. Res. Public Health 2022, 19, 10649. https://doi.org/10.3390/ijerph191710649
Wang Y, Guo C, Zhang L, Lu X, Liu Y, Li X, Wang Y, Wang S. Arsenic Oxidation and Removal from Water via Core–Shell MnO2@La(OH)3 Nanocomposite Adsorption. International Journal of Environmental Research and Public Health. 2022; 19(17):10649. https://doi.org/10.3390/ijerph191710649
Chicago/Turabian StyleWang, Yulong, Chen Guo, Lin Zhang, Xihao Lu, Yanhong Liu, Xuhui Li, Yangyang Wang, and Shaofeng Wang. 2022. "Arsenic Oxidation and Removal from Water via Core–Shell MnO2@La(OH)3 Nanocomposite Adsorption" International Journal of Environmental Research and Public Health 19, no. 17: 10649. https://doi.org/10.3390/ijerph191710649
APA StyleWang, Y., Guo, C., Zhang, L., Lu, X., Liu, Y., Li, X., Wang, Y., & Wang, S. (2022). Arsenic Oxidation and Removal from Water via Core–Shell MnO2@La(OH)3 Nanocomposite Adsorption. International Journal of Environmental Research and Public Health, 19(17), 10649. https://doi.org/10.3390/ijerph191710649









