COVID-19 Testing Strategies for K-12 Schools in California: A Cost-Effectiveness Analysis
Abstract
:1. Introduction
2. Materials and Methods
2.1. Overview
2.2. Decision Tree Design
2.3. Key Assumptions and Model Inputs
2.4. Epidemiologic and Health Inputs
2.5. Test Performance Inputs
2.6. Cost Inputs
2.7. Model Outputs
3. Results
3.1. Base Case
Cost-Effectiveness
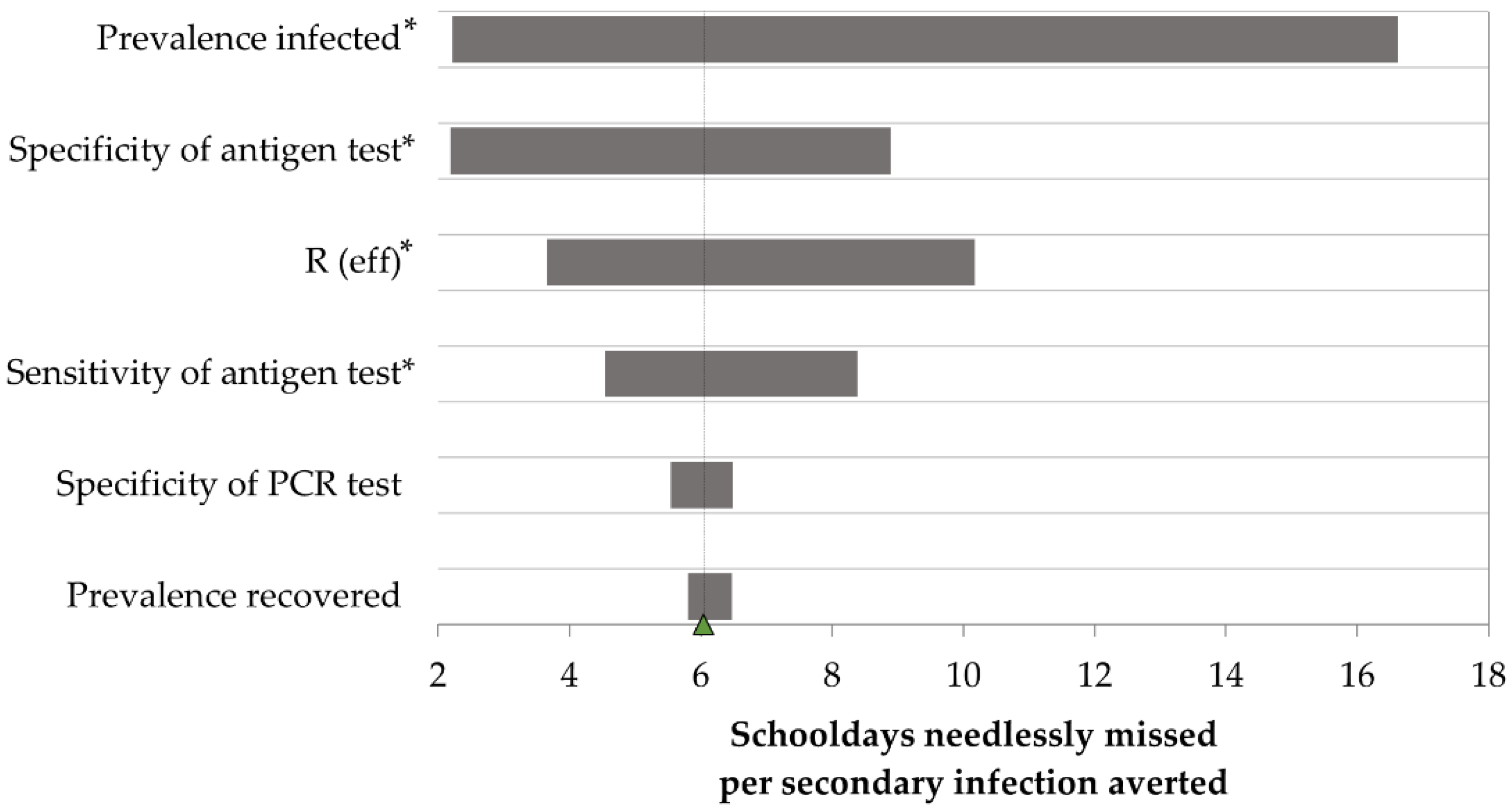
3.2. Unnecessary Days Lost versus Infections Averted
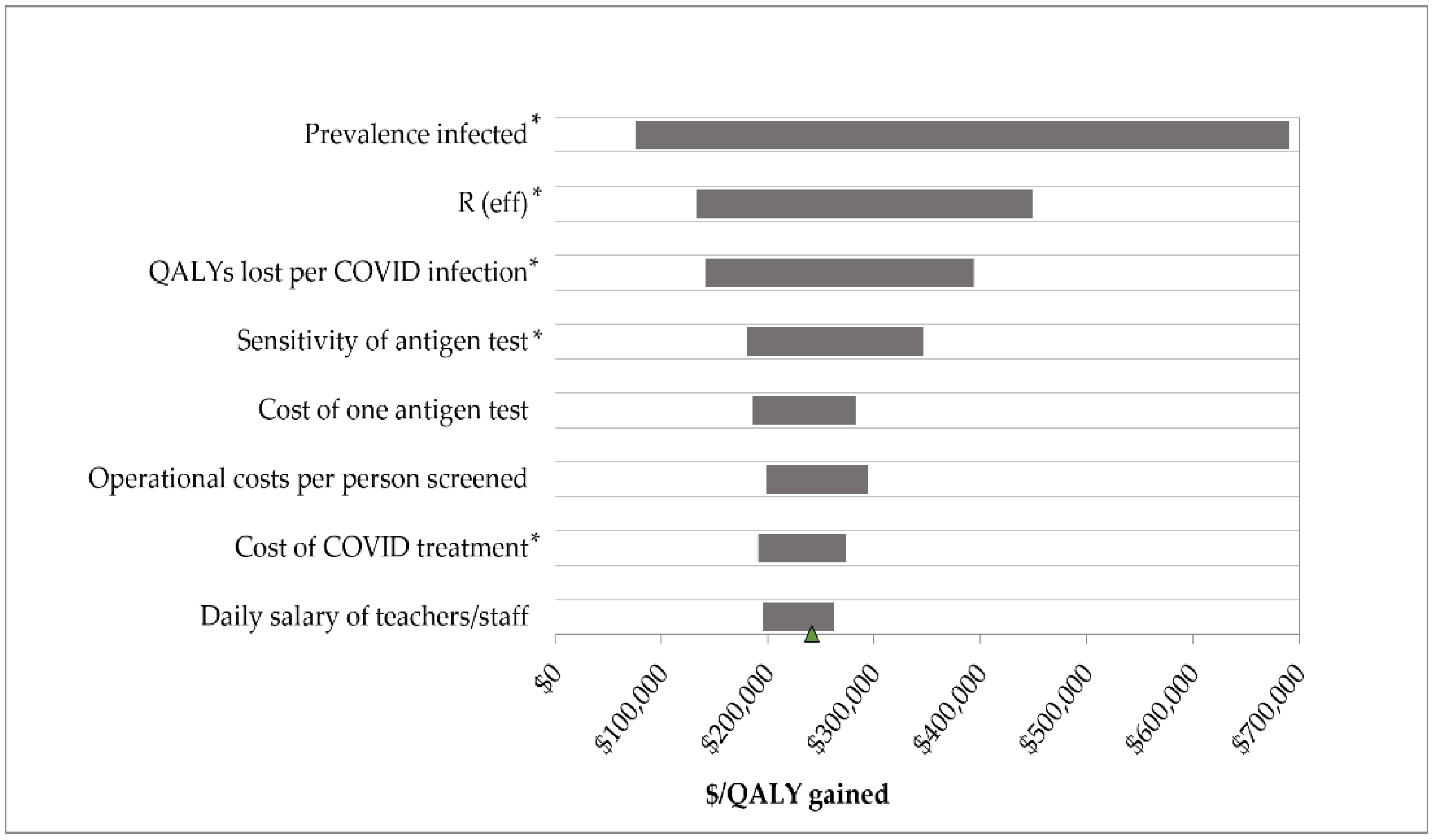
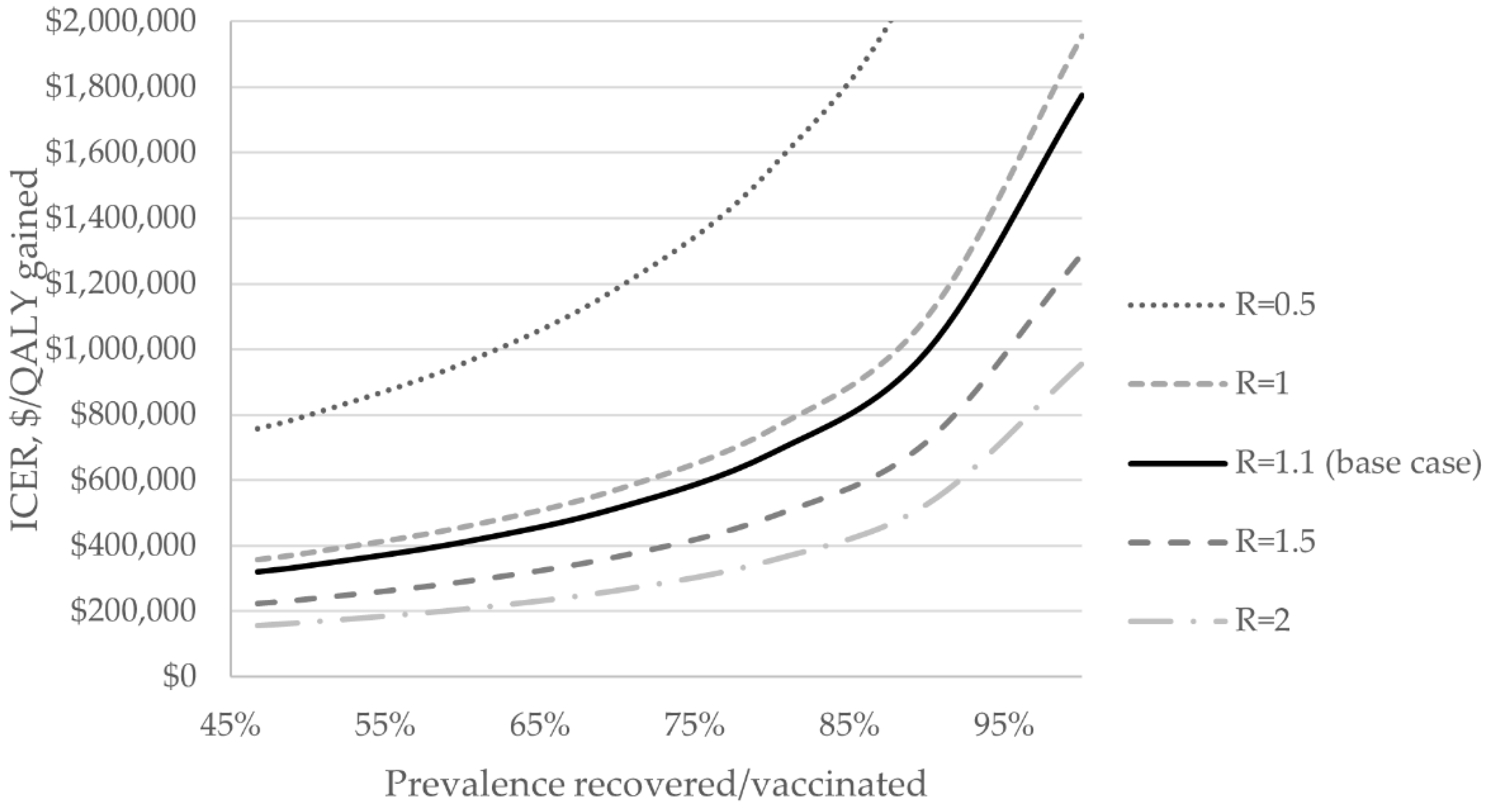
3.3. Sensitivity Analyses
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- World Health Organization. WHO Director-General’s Opening Remarks at the Media Briefing on COVID-19-11 March 2020. 11 March 2020. Available online: https://www.who.int/director-general/speeches/detail/who-director-general-s-opening-remarks-at-the-media-briefing-on-covid-19---11-march-2020 (accessed on 28 April 2021).
- New COVID-19 Cases Worldwide. Johns Hopkins University Coronavirus Resource Center. 28 April 2021. Available online: https://coronavirus.jhu.edu/data/new-cases (accessed on 28 April 2021).
- NCHS Mortality Surveillance Data. Centers for Disease Control and Prevention. 24 April 2020. Available online: https://www.cdc.gov/coronavirus/2019-ncov/covid-data/covidview/04242020/nchs-mortality-report.html (accessed on 28 April 2021).
- Kohli, S.; Blume, H. Public Schools Expected to Remain Closed for the Rest of the Academic Year, Newsom Says. Los Angeles Times. 1 April 2020. Available online: https://www.latimes.com/california/story/2020-04-01/coronavirus-school-closures-california (accessed on 28 April 2021).
- Christakis, D.A.; Van Cleve, W.; Zimmerman, F.J. Estimation of US Children’s Educational Attainment and Years of Life Lost Associated With Primary School Closures During the Coronavirus Disease 2019 Pandemic. JAMA Netw. Open 2020, 3, e2028786. [Google Scholar] [CrossRef] [PubMed]
- Meng, X.; Gregory, R. Exploring the Impact of Interrupted Education on Earnings: The Educational Cost of the Chinese Cultural Revolution. 2007. Available online: http://www.iza.org/publications/dps/ (accessed on 28 April 2021).
- Meng, X.; Gregory, R.G. The Impact of Interrupted Education on Subsequent Educational Attainment: A Cost of the Chinese Cultural Revolution. Econ. Dev. Cult. Chang. 2002, 50, 935–959. [Google Scholar] [CrossRef]
- UNESCO. Adverse Consequences of School Closures. Available online: https://en.unesco.org/covid19/educationresponse/consequences (accessed on 28 April 2021).
- Cummings, M. COVID School Closures Most Harm Students from Poorest Neighborhoods. Yale News. 5 January 2021. Available online: https://news.yale.edu/2021/01/05/covid-school-closures-most-harm-students-poorest-neighborhoods (accessed on 28 April 2021).
- California Department of Education. A Guidebook for the Safe Reopening of California’s Public Schools; California Department of Education: Sacramento, CA, USA, 2021.
- Centers for Disease Control and Prevention. Operational Strategy for K-12 Schools through Phased Prevention. 23 April 2021. Available online: https://www.cdc.gov/coronavirus/2019-ncov/community/schools-childcare/operation-strategy.html?CDC_AA_refVal=https%3A%2F%2Fwww.cdc.gov%2Fcoronavirus%2F2019-ncov%2Fcommunity%2Fschools-childcare%2Fschools.html#anchor_1616080181070 (accessed on 28 April 2021).
- Paltiel, A.D.; Zheng, A.; Walensky, R.P. Assessment of SARS-CoV-2 Screening Strategies to Permit the Safe Reopening of College Campuses in the United States. JAMA Netw. Open 2020, 3, e2016818. [Google Scholar] [CrossRef] [PubMed]
- Esbin, M.N.; Whitney, O.N.; Chong, S.; Maurer, A.; Darzacq, X.; Tjian, R. Overcoming the bottleneck to widespread testing: A rapid review of nucleic acid testing approaches for COVID-19 detection. RNA 2020, 26, 771–783. [Google Scholar] [CrossRef] [PubMed]
- Faherty, L.J.; Master, B.K.; Steiner, E.D.; Kaufman, J.H.; Predmore, Z.; Stelitano, L.; Wolfe, R. COVID-19 Testing in K-12 Schools: Insights from Early Adopters; RAND Corporation: Santa Monica, CA, USA, 2021. [Google Scholar]
- Pollock, N.R.; Jacobs, J.R.; Tran, K.; Cranston, A.E.; Smith, S.; O’Kane, C.Y.; Roady, T.J.; Moran, A.; Scarry, A.; Carroll, M.; et al. Performance and Implementation Evaluation of the Abbott BinaxNOW Rapid Antigen Test in a High-Throughput Drive-Through Community Testing Site in Massachusetts. J. Clin. Microbiol. 2021, 59, 83–104. [Google Scholar] [CrossRef] [PubMed]
- Food and Drug Administration. BinaxNOW COVID-19 Ag Card—Instructions for Use. 2020. Available online: https://www.fda.gov/media/141570/download (accessed on 28 April 2021).
- Pugh, J.; Wilkinson, D.; Savulescu, J. Sense and sensitivity: Can an inaccurate test be better than no test at all? J. Med. Ethics 2022, 48, 329–333. [Google Scholar] [CrossRef] [PubMed]
- Mina, M.J.; Peto, T.E.; García-Fiñana, M.; Semple, M.G.; Buchan, I.E. Clarifying the evidence on SARS-CoV-2 antigen rapid tests in public health responses to COVID-19. Lancet 2021, 397, 1425–1427. [Google Scholar] [CrossRef]
- California For All. Tracking COVID-19 in California. Available online: https://covid19.ca.gov/state-dashboard/ (accessed on 28 April 2021).
- California Department of Public Health. COVID-19 Seroprevalence Data. 15 April 2021. Available online: https://www.cdph.ca.gov/Programs/CID/DCDC/Pages/COVID-19/Sero-prevalence-COVID-19-Data.aspx (accessed on 28 April 2021).
- Centre for Mathematical Modelling of Infectious Diseases (CMMID). COVID-19: Estimates for California (United States). 28 April 2021. Available online: https://epiforecasts.io/covid/posts/subnational/united-states/california/ (accessed on 28 April 2021).
- Bilinski, A.; Salomon, J.A.; Giardina, J.; Ciaranello, A.; Fitzpatrick, M.C. Passing the Test: A Model-Based Analysis of Safe School-Reopening Strategies. medRxiv 2021. [Google Scholar] [CrossRef] [PubMed]
- Briggs, A. Moving Beyond “Lives-Saved” from COVID-19. Avalon Health Economics LLC. 15 May 2020. Available online: https://avalonecon.com/moving-beyond-lives-saved-from-covid-19/ (accessed on 2 July 2020).
- Briggs, A. Using Life Table Methods to Calculate QALY Losses from Deaths: With Application to COVID-19. 2020. Available online: https://avalonecon.com/wp-content/uploads/2020/04/COVID-19-QALYs-v3.pdf (accessed on 28 April 2021).
- Pilarowski, G.; Lebel, P.; Sunshine, S.; Liu, J.; Crawford, E.; Marquez, C.; Rubio, L.; Chamie, G.; Martinez, J.; Peng, J.; et al. Performance Characteristics of a Rapid Severe Acute Respiratory Syndrome Coronavirus 2 Antigen Detection Assay at a Public Plaza Testing Site in San Francisco. J. Infect. Dis. 2021, 223, 1139–1144. [Google Scholar] [CrossRef] [PubMed]
- Arevalo-Rodriguez, I.; Buitrago-Garcia, D.; Simancas-Racines, D.; Achig, P.Z.; Del Campo, R.; Ciapponi, A.; Sued, O.; Martinez-García, L.; Rutjes, A.W.; Low, N.; et al. False-negative results of initial RT-PCR assays for COVID-19: A systematic review. PLoS ONE 2020, 15, e0242958. [Google Scholar] [CrossRef]
- Foundation for Innovative New Diagnostics (FIND). SARS-CoV-2 Molecular Assay Evaluation: Results. 3 July 2020. Available online: https://www.finddx.org/covid-19/sarscov2-eval-molecular/molecular-eval-results/ (accessed on 29 April 2021).
- Elementary School Teacher Salary in California. Career Explorer. Available online: https://www.careerexplorer.com/careers/elementary-school-teacher/salary/california/ (accessed on 28 April 2021).
- Rae, M.; Claxton, G.; Kurani, N.; McDermott, D.; Cox, C. Potential Costs of COVID-19 Treatment for People with Employer Coverage. Peterson-KFF Health System Tracker. 13 March 2020. Available online: https://www.healthsystemtracker.org/brief/potential-costs-of-coronavirus-treatment-for-people-with-employer-coverage/ (accessed on 24 June 2020).
- Buonsenso, D.; Munblit, D.; De Rose, C.; Sinatti, D.; Ricchiuto, A.; Carfi, A.; Valentini, P. Preliminary evidence on long COVID in children. Acta Paediatr. 2021, 110, 2208–2211. [Google Scholar] [CrossRef]
- Office for National Statistics. Prevalence of Ongoing Symptoms Following Coronavirus (COVID-19) Infection in the UK. 2021. Available online: https://www.ons.gov.uk/peoplepopulationandcommunity/healthandsocialcare/conditionsanddiseases/bulletins/prevalenceofongoingsymptomsfollowingcoronaviruscovid19infectionintheuk/1april2021 (accessed on 14 October 2021).
- Vanness, D.J.; Lomas, J.; Ahn, M.H. A Health Opportunity Cost Threshold for Cost-Effectiveness Analysis in the United States. Ann. Intern. Med. 2021, 174, 25–32. [Google Scholar] [CrossRef] [PubMed]
- McCabe, C. Perspectives on Cost-Effectiveness Thresholds in the United States: Willingness to Pay as a Basis for a Cost-Effectiveness Threshold in the United States; Institute for Clinical and Economic Review: Boston, MA, USA, 2019. [Google Scholar]
- Head, J.R.; Andrejko, K.L.; Cheng, Q.; Collender, P.A.; Phillips, S.; Boser, A.; Heaney, A.K.; Hoover, C.M.; Wu, S.L.; Northrup, G.R.; et al. School closures reduced social mixing of children during COVID-19 with implications for transmission risk and school reopening policies. J. R. Soc. Interface 2021, 18, 20200970. [Google Scholar] [CrossRef] [PubMed]
- McCarty, M. Mina among Academicians Sparring over Role of Lateral Flow Antigen Tests in Pandemic. 18 February 2021. Available online: https://www.bioworld.com/articles/503784-mina-among-academicians-sparring-over-role-of-lateral-flow-antigen-tests-in-pandemic (accessed on 29 April 2021).
- Deeks, J.J.; Raffle, A.E. Lateral flow tests cannot rule out SARS-CoV-2 infection. BMJ 2020, 371, m4787. [Google Scholar] [CrossRef] [PubMed]
- Mahase, E. COVID-19: Innova lateral flow test is not fit for “test and release” strategy, say experts. BMJ 2020, 371, m4469. [Google Scholar] [CrossRef]
- Xiao, A.T.; Tong, Y.X.; Zhang, S. Profile of PT-PCR for SARS-CoV-2: A preliminary study from 56 COVID-19 patients. Clin. Infect. Dis. 2020, 71, 2249–2251. [Google Scholar] [CrossRef] [PubMed]
- Atkinson, B.; Petersen, E. SARS-CoV-2 shedding and infectivity. Lancet 2020, 395, 1339–1340. [Google Scholar] [CrossRef]
- Joynt, G.M.; Wu, W.K. Understanding COVID-19: What does viral RNA load really mean? Lancet Infect. Dis. 2020, 20, 635–636. [Google Scholar] [CrossRef] [Green Version]
- Slavitt, A. Omicron & Testing: Your Top 20 Questions Answered (with Michael Mina). Available online: https://lemonadamedia.com/podcast/will-we-all-get-omicron-in-2022-with-david-agus/ (accessed on 15 January 2022).
- Centers for Disease Control and Prevention. Science Brief: Transmission of SARS-CoV-2 in K-12 Schools. 19 March 2021. Available online: https://www.cdc.gov/coronavirus/2019-ncov/science/science-briefs/transmission_k_12_schools.html (accessed on 28 April 2021).
| Parameter | Base-Case (Range) | Reference |
|---|---|---|
| Epidemiologic inputs | ||
| Prevalence of currently infected * | 0.2% (0.05–0.80%) | [19] |
| Prevalence previously infected or vaccinated (“Recovered”) ** | 47% (38–55%) | [20] |
| Effective reproduction number (Reff) *** | 1.1 (0.5–2) | [21] |
| QALYs lost per infection | 0.078 (0.051–0.206) | [22,23] |
| Test performance inputs | ||
| Ag test sensitivity | 58% (37–77%) | [15,24] |
| Ag test specificity | 99.6% (99.6–100%) | [24] |
| PCR test sensitivity | 71% (53–83%) | [25,26,27] |
| PCR test specificity | 99% (97–100%) | [25,26,27] |
| Cost inputs | ||
| Cost of Ag test | USD 5 (USD 2–USD 8) | Stakeholder input |
| Cost of PCR test | USD 21 (USD 21–USD 140) | Stakeholder input |
| Operational costs per person screened (for 1× testing strategies) | USD 4.5 (USD 2.2–USD 6.8) | Stakeholder input |
| Cost of 1 day of missed schooling (students) | USD 100 (USD 50–USD 200) | Assumed |
| Cost of 1 day of missed work (teachers/staff, with full productivity loss) | USD 320 (USD 260–USD 480) | [28] |
| Option | Net Cost * | Added Cost *, ** | QALYs Lost | QALYs Gained ** | ICER (USD/QALYs) ** |
|---|---|---|---|---|---|
| No Test | USD 5264 | n/a | 0.121786 | n/a | n/a |
| 1× antigen + PCR | USD 16,439 | USD 11,175 | 0.086904 | 0.034882 | USD 320,358 |
| 1× antigen | USD 18,824 | USD 2385 | 0.086651 | 0.000253 | USD 9,428,177 |
| 2× antigen + PCR | USD 25,510 | USD 9072 | 0.070728 | 0.016176 | USD 560,830 *** |
| 2× antigen | USD 30,329 | USD 4818 | 0.070490 | 0.000238 | USD 20,243,956 |
| 1×PCR | USD 40,500 | USD 14,990 | 0.090657 | −0.019930 | Dominated |
| Option | Net Cost to Schools | Added Cost * | QALYs Lost | QALYs Gained * | ICER (USD/QALYs) * |
|---|---|---|---|---|---|
| No Test | USD 0 | n/a | 0.121786 | n/a | n/a |
| 1× antigen | USD 11,373 | USD 11,373 | 0.086651 | 0.035135 | USD 323,703 |
| 1× antigen + PCR | USD 11,453 | USD 80 | 0.086904 | −0.000253 | Dominated |
| 2× antigen | USD 20,363 | USD 8989 | 0.070490 | 0.016161 | USD 556,240 |
| 2× antigen + PCR | USD 20,483 | USD 121 | 0.070728 | −0.000238 | Dominated |
| 1×PCR | USD 28,374 | USD 8011 | 0.090657 | −0.020168 | Dominated |
| Option | Days of Needless Isolation | Added Days of Needless Isolation * | Secondary Infections | Secondary Infections Averted * | Efficiency (Days Needlessly Missed/Secondary Infection Averted) * |
|---|---|---|---|---|---|
| No test | 0.00 | n/a | 1.571 | n/a | n/a |
| 1×Ag + PCR | 4.3 | 4.3 | 1.121 | 0.450 | 9.5 |
| 2×Ag + PCR | 8.5 | 4.3 | 0.913 | 0.209 | 20.4 |
| 1×Ag | 31.9 | 23.4 | 1.118 | −0.205 | Dominated |
| 2×Ag | 63.7 | 55.2 | 0.910 | 0.003 | 17,980 |
| 1×PCR | 79.8 | 71.3 | 1.170 | −0.257 | Dominated |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Maya, S.; McCorvie, R.; Jacobson, K.; Shete, P.B.; Bardach, N.; Kahn, J.G. COVID-19 Testing Strategies for K-12 Schools in California: A Cost-Effectiveness Analysis. Int. J. Environ. Res. Public Health 2022, 19, 9371. https://doi.org/10.3390/ijerph19159371
Maya S, McCorvie R, Jacobson K, Shete PB, Bardach N, Kahn JG. COVID-19 Testing Strategies for K-12 Schools in California: A Cost-Effectiveness Analysis. International Journal of Environmental Research and Public Health. 2022; 19(15):9371. https://doi.org/10.3390/ijerph19159371
Chicago/Turabian StyleMaya, Sigal, Ryan McCorvie, Kathleen Jacobson, Priya B. Shete, Naomi Bardach, and James G. Kahn. 2022. "COVID-19 Testing Strategies for K-12 Schools in California: A Cost-Effectiveness Analysis" International Journal of Environmental Research and Public Health 19, no. 15: 9371. https://doi.org/10.3390/ijerph19159371
APA StyleMaya, S., McCorvie, R., Jacobson, K., Shete, P. B., Bardach, N., & Kahn, J. G. (2022). COVID-19 Testing Strategies for K-12 Schools in California: A Cost-Effectiveness Analysis. International Journal of Environmental Research and Public Health, 19(15), 9371. https://doi.org/10.3390/ijerph19159371






