Endocannabinoids and Heart Rate Variability Alterations after Exposure to Prolonged Intensive Physical Exercise of the Hellenic Navy SEALs
Abstract
:1. Introduction
2. Materials and Methods
2.1. Ethical Approval
2.2. Subjects
2.3. Exercise Training Program
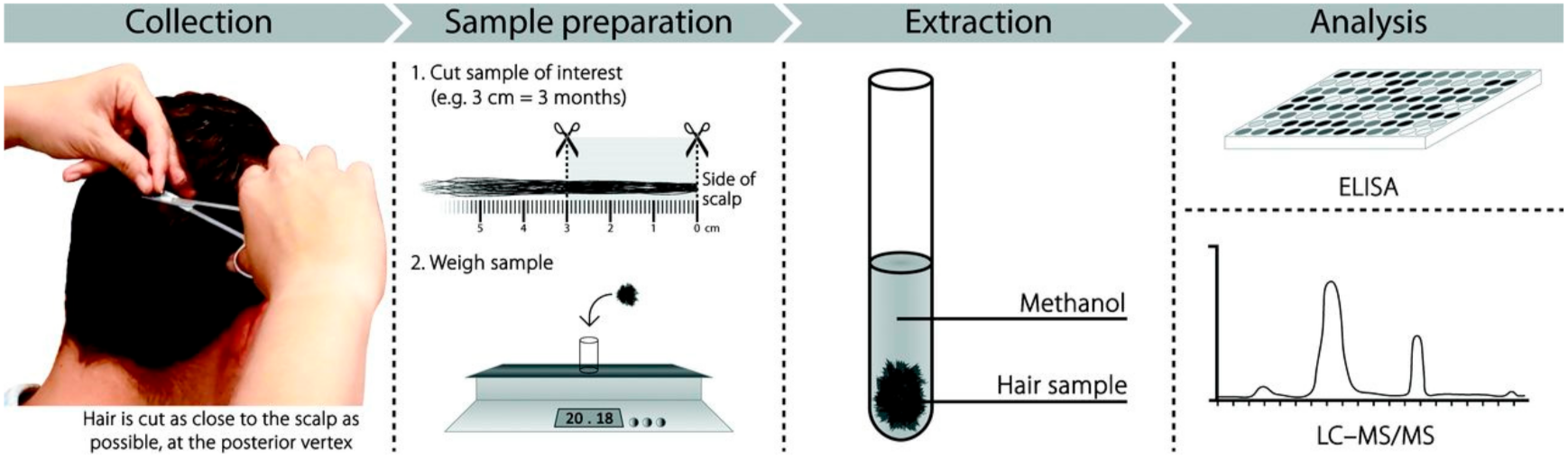
2.4. Procedure
2.5. Materials
2.5.1. Psychometric Tools
2.5.2. Psychophysiological Measurements:
2.5.3. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Galve-Roperh, I.; Chiurchiù, V.; Díaz-Alonso, J.; Bari, M.; Guzmán, M.; Maccarrone, M. Cannabinoid receptor signaling in progenitor/stem cell proliferation and differentiation. Prog. Lipid Res. 2013, 52, 633–650. [Google Scholar] [CrossRef] [PubMed]
- Wei, D.; Allsop, S.; Tye, K.; Piomelli, D. Endocannabinoid Signaling in the Control of Social Behavior. Trends Neurosci. 2017, 40, 385–396. [Google Scholar] [CrossRef] [PubMed]
- Raso, G.M.; Russo, R.; Calignano, A.; Meli, R. Palmitoylethanolamide in CNS health and disease. Pharmacol. Res. 2014, 86, 32–41. [Google Scholar] [CrossRef] [PubMed]
- Thabuis, C.; Tissot-Favre, D.; Bezelgues, J.B.; Martin, J.C.; Cruz-Hernandez, C.; Dionisi, F.; Destaillats, F. Biological functions and metabolism of oleoylethanolamide. Lipids 2008, 43, 887. [Google Scholar] [CrossRef] [PubMed]
- Ueda, N.; Tsuboi, K.; Uyama, T. Metabolism of endocannabinoids and relatedN-acylethanolamines: Canonical and alternative pathways. FEBS J. 2013, 280, 1874–1894. [Google Scholar] [CrossRef] [PubMed]
- Morena, M.; Patel, S.; Bains, J.; Hill, M.N. Neurobiological Interactions Between Stress and the Endocannabinoid System. Neuropsychopharmacology 2015, 41, 80–102. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wilker, S.; Pfeiffer, A.; Elbert, T.; Ovuga, E.; Karabatsiakis, A.; Krumbholz, A.; Thieme, D.; Schelling, G.; Kolassa, I.-T. Endocannabinoid concentrations in hair are associated with PTSD symptom severity. Psychoneuroendocrinology 2016, 67, 198–206. [Google Scholar] [CrossRef] [Green Version]
- Koenig, A.M.; Gao, W.; Umlauft, M.; Schury, K.; Reister, F.; Kirschbaum, C.; Karabatsiakis, A.; Kolassa, I.T. Altered hair endocannabinoid levels in mothers with childhood maltreatment and their newborns. Biol. Psychol. 2018, 135, 93–101. [Google Scholar] [CrossRef] [PubMed]
- Krumbholz, A.; Anielski, P.; Reisch, N.; Schelling, G.; Thieme, D. Diagnostic Value of Concentration Profiles of Glucocorticosteroids and Endocannabinoids in Hair. Ther. Drug Monit. 2013, 35, 600–607. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Škopek, M.; Heidler, J.; Hnizdil, J.; Šulc, J. The effect of cannabidiol (CBD) on simple and complex reaction times. Trends Sport Sci. 2021, 28, 147–151. [Google Scholar]
- Ho, W.S.; Kelly, M.E. Cannabinoids in the cardiovascular system. Adv. Pharmacol. 2017, 80, 329–366. [Google Scholar]
- Dugrenot, E.; Balestra, C.; Gouin, E.; L’Her, E.; Guerrero, F. Physiological effects of mixed-gas deep sea dives using a closed-circuit rebreather: A field pilot study. Eur. J. Appl. Physiol. 2021, 121, 3323–3331. [Google Scholar] [CrossRef] [PubMed]
- Bustamante-Sánchez, A.; Tornero-Aguilera, J.F.; Fernández-Elías, V.E.; Hormeño-Holgado, A.J.; Dalamitros, A.A.; Clemente-Suárez, V.J. Effect of Stress on Autonomic and Cardiovascular Systems in Military Population: A Systematic Review. Cardiol. Res. Pract. 2020, 2020, 7986249. [Google Scholar] [CrossRef] [PubMed]
- Lafère, P.; Lambrechts, K.; Germonpré, P.; Balestra, A.; Germonpré, F.L.; Marroni, A.; Cialoni, D.; Bosco, G.; Balestra, C. Heart Rate Variability During a Standard Dive: A Role for Inspired Oxygen Pressure? Front. Physiol. 2021, 12, 1060. [Google Scholar] [CrossRef] [PubMed]
- Järvelin-Pasanen, S.; Sinikallio, S.; Tarvainen, M.P. Heart rate variability and occupational stress—Systematic review. Ind. Health 2018, 56, 500–511. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Thayer, J.F.; Åhs, F.; Fredrikson, M.; Sollers, J.J., III; Wager, T.D. A meta-analysis of heart rate variability and neuroimaging studies: Implications for heart rate variability as a marker of stress and health. Neurosci. Biobehav. Rev. 2012, 36, 747–756. [Google Scholar] [CrossRef]
- Smith, R.; Thayer, J.F.; Khalsa, S.S.; Lane, R.D. The hierarchical basis of neurovisceral integration. Neurosci. Biobehav. Rev. 2017, 75, 274–296. [Google Scholar] [CrossRef]
- Donias, S.; Karastergiou, A.; Manos, N. Validation of Symptom Checklist 90 R in the Greek population. Psychiatriki 1991, 2, 42–48. [Google Scholar]
- Sauvé, B.; Koren, G.; Walsh, G.; Tokmakejian, S.; van Uum, S.H. Measurement of cortisol in human hair as a biomarker of systemic exposure. Clin. Investig. Med. 2007, 30, E183–E191. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kirschbaum, C.; Tietze, A.; Skoluda, N.; Dettenborn, L. Hair as a retrospective calendar of cortisol production—Increased cortisol incorporation into hair in the third trimester of pregnancy. Psychoneuroendocrinology 2009, 34, 32–37. [Google Scholar] [CrossRef]
- Gao, W.; Stalder, T.; Foley, P.; Rauh, M.; Deng, H.; Kirschbaum, C. Quantitative analysis of steroid hormones in human hair using a column-switching LC–APCI–MS/MS assay. J. Chromatogr. B 2013, 928, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Manenschijn, L.; Koper, J.W.; Lamberts, S.W.; van Rossum, E.F. Evaluation of a method to measure long term cortisol levels. Steroids 2011, 76, 1032–1036. [Google Scholar] [CrossRef]
- Noppe, G.; De Rijke, Y.B.; Dorst, K.; van den Akker, E.L.T.; Van Rossum, E.F.C. LC-MS/MS-based method for long-term steroid profiling in human scalp hair. Clin. Endocrinol. 2015, 83, 162–166. [Google Scholar] [CrossRef] [PubMed]
- Voegel, C.D.; Baumgartner, M.R.; Kraemer, T.; Wüst, S.; Binz, T.M. Simultaneous quantification of steroid hormones and endocannabinoids (ECs) in human hair using an automated supported liquid extraction (SLE) and LC-MS/MS—Insights into EC baseline values and correlation to steroid concentrations. Talanta 2021, 222, 121499. [Google Scholar] [CrossRef] [PubMed]
- Hill, M.N.; Miller, G.E.; Carrier, E.J.; Gorzalka, B.B.; Hillard, C.J. Circulating endocannabinoids and N-acyl ethanolamines are differentially regulated in major depression and following exposure to social stress. Psychoneuroendocrinology 2009, 34, 1257–1262. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bluett, R.J.; Gamble-George, J.C.; Hermanson, D.J.; Hartley, N.D.; Marnett, L.J.; Patel, S. Central anandamide deficiency predicts stress-induced anxiety: Behavioral reversal through endocannabinoid augmentation. Transl. Psychiatry 2014, 4, e408. [Google Scholar] [CrossRef] [Green Version]
- Patel, S.; Cravatt, B.F.; Hillard, C.J. Synergistic Interactions between Cannabinoids and Environmental Stress in the Activation of the Central Amygdala. Neuropsychopharmacology 2004, 30, 497–507. [Google Scholar] [CrossRef] [PubMed]
- Hill, M.N.; Kumar, S.A.; Filipski, S.B.; Iverson, M.; Stuhr, K.L.; Keith, J.M.; Cravatt, B.F.; Hillard, C.J.; Chattarji, S.; McEwen, B.S. Disruption of fatty acid amide hydrolase activity prevents the effects of chronic stress on anxiety and amygdala microstructure. Mol. Psychiatry 2013, 18, 1125–1135. [Google Scholar] [CrossRef] [PubMed]
- Hohmann, A.G.; Suplita, R.L.; Bolton, N.M.; Neely, M.H.; Fegley, D.; Mangieri, R.; Krey, J.F.; Walker, J.M.; Holmes, P.V.; Crystal, J.D.; et al. An endocannabinoid mechanism for stress-induced analgesia. Nature 2005, 435, 1108–1112. [Google Scholar] [CrossRef]
- Morena, M.; Roozendaal, B.; Trezza, V.; Ratano, P.; Peloso, A.; Hauer, D.; Atsak, P.; Trabace, L.; Cuomo, V.; McGaugh, J.L.; et al. Endogenous cannabinoid release within prefrontal-limbic pathways affects memory consolidation of emotional training. Proc. Natl. Acad. Sci. USA 2014, 111, 18333–18338. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gorzalka, B.B.; Hill, M.N.; Hillard, C.J. Regulation of endocannabinoid signaling by stress: Implications for stress-related affective disorders. Neurosci. Biobehav. Rev. 2008, 32, 1152–1160. [Google Scholar] [CrossRef] [PubMed]
- Katona, I.; Freund, T.F. Multiple Functions of Endocannabinoid Signaling in the Brain. Annu. Rev. Neurosci. 2012, 35, 529–558. [Google Scholar] [CrossRef] [Green Version]
- Mechoulam, R.; Parker, L.A. The Endocannabinoid System and the Brain. Annu. Rev. Psychol. 2013, 64, 21–47. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gao, W.; Walther, A.; Wekenborg, M.; Penz, M.; Kirschbaum, C. Determination of endocannabinoids and N-acylethanolamines in human hair with LC-MS/MS and their relation to symptoms of depression, burnout, and anxiety. Talanta 2020, 217, 121006. [Google Scholar] [CrossRef]
- Davis, M.P.; Behm, B.; Mehta, Z.; Fernandez, C. The Potential Benefits of Palmitoylethanolamide in Palliation: A Qualitative Systematic Review. Am. J. Hosp. Palliat. Med. 2019, 36, 1134–1154. [Google Scholar] [CrossRef] [PubMed]
- Hillard, C.J. Circulating Endocannabinoids: From Whence Do They Come and Where are They Going? Neuropsychopharmacology 2018, 43, 155–172. [Google Scholar] [CrossRef] [PubMed]
- Battistini, L.; Maccarrone, M. The Endocannabinoid System in Peripheral Lymphocytes as a Mirror of Neuroinflammatory Diseases. Curr. Pharm. Des. 2008, 14, 2370–2382. [Google Scholar] [CrossRef]
- Ori, Z.; Monir, G.; Weiss, J.; Sayhouni, X.; Singer, D.H. Heart rate variability. Frequency domain analysis. Cardiol. Clin. 1992, 10, 499–533. [Google Scholar] [CrossRef]
- Tornero-Aguilera, J.F.; Pelarigo, J.G.; Clemente-Suárez, V.J. Psychophysiological Intervention to Improve Preparedness in Military Special Operations Forces. Aerosp. Med. Hum. Perform. 2019, 90, 953–958. [Google Scholar] [CrossRef]
- Hormeño-Holgado, A.J.; Nikolaidis, P.T.; Clemente-Suárez, V.J. Psychophysiological Patterns Related to Success in a Special Operation Selection Course. Front. Physiol. 2019, 10, 867. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hormeño-Holgado, A.J.; Clemente-Suárez, V.J. Psychophysiological Monitorization in a Special Operation Selection Course. J. Med. Syst. 2019, 43, 47. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Molina, J.; Robles-Pérez, J.J.; Clemente-Suárez, V.J. Assessment of Psychophysiological Response and Specific Fine Motor Skills in Combat Units. J. Med. Syst. 2018, 42, 67. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Molina, J.; Pérez, J.J.R.; Clemente-Suárez, V.J. Effect of Parachute Jump in the Psychophysiological Response of Soldiers in Urban Combat. J. Med. Syst. 2017, 41, 99. [Google Scholar] [CrossRef] [PubMed]
- Aguilera, J.F.T.; Gil-Cabrera, J.; Clemente-Suárez, V.J. Determining the psychophysiological responses of military aircrew when exposed to acute disorientation stimuli. BMJ Mil. Health 2020. [Google Scholar] [CrossRef]
| Demographic Characteristics | HN-SEALs |
|---|---|
| Number of participants | 13 |
| Age (years) | 24.63 ± 3.81 |
| Health Problem (No) | 13 (100%) |
| Receiving medicine (No) | 13 (100%) |
| Sample Mean (±sd) | Population Means (±sd) | One Sample t-test | Effect Size Cohen’s d | |
|---|---|---|---|---|
| Somatization | 13.8 (±8.51) | 7.52 (±6.35) | 2.334 * | 0.74 |
| Interpersonal sensitivity | 3.6 (±3.86) | 7.81 (±6.04) | −3.445 ** | 1.09 |
| Hostility | 2.3 (±2.11) | 5.38 (±4.78) | −4.614 ** | 1.46 |
| Paranoid Ideation | 3.3 (±3.46) | 5.81 (±3.64) | −2.290 * | 0.72 |
| Mean (±sd) before | Mean (±sd) after | Paired Sample t-test | Effect Size Cohen’s d | |
|---|---|---|---|---|
| AEA | 3.46 (±0.46) | 3.90 (±0.59) | −4.225 ** | −1.17 |
| AG2 | 72.55 (±5.80) | 72.52 (±5.96) | 0.083 | 0.23 |
| PEA | 1453.94 (±150.72) | 1633.64 (±195.94) | −2.963 * | −0.82 |
| OEA | 1972.49 (±39.60) | 2138.19 (±137.81) | −5.115 *** | −1.42 |
| Mean HR | Mean HRV | LF HRV | |
|---|---|---|---|
| AEA | −0.756 | 0.934 * | −0.037 |
| PEA | 0.902 * | −0.056 | 0.853 ** |
| Mean HR | Mean HRV | LF HRV | HF HRV | |
|---|---|---|---|---|
| Mean difference PEA | 0.475 | −0.559 | 0.751 * | −0.709 * |
| Mean difference OEA | 0.683* | −0.640 * | 0.182 | −0.277 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mourtakos, S.; Vassiliou, G.; Kontoangelos, K.; Philippou, A.; Tzavellas, E.; Tornero-Aguilera, J.F.; Clemente-Suárez, V.J.; Papageorgiou, C.; Sidossis, L.S.; Papageorgiou, C. Endocannabinoids and Heart Rate Variability Alterations after Exposure to Prolonged Intensive Physical Exercise of the Hellenic Navy SEALs. Int. J. Environ. Res. Public Health 2022, 19, 28. https://doi.org/10.3390/ijerph19010028
Mourtakos S, Vassiliou G, Kontoangelos K, Philippou A, Tzavellas E, Tornero-Aguilera JF, Clemente-Suárez VJ, Papageorgiou C, Sidossis LS, Papageorgiou C. Endocannabinoids and Heart Rate Variability Alterations after Exposure to Prolonged Intensive Physical Exercise of the Hellenic Navy SEALs. International Journal of Environmental Research and Public Health. 2022; 19(1):28. https://doi.org/10.3390/ijerph19010028
Chicago/Turabian StyleMourtakos, Stamatis, Georgia Vassiliou, Konstantinos Kontoangelos, Anastassios Philippou, Elias Tzavellas, José Francisco Tornero-Aguilera, Vicente Javier Clemente-Suárez, Charalabos Papageorgiou, Labros S. Sidossis, and Christos Papageorgiou. 2022. "Endocannabinoids and Heart Rate Variability Alterations after Exposure to Prolonged Intensive Physical Exercise of the Hellenic Navy SEALs" International Journal of Environmental Research and Public Health 19, no. 1: 28. https://doi.org/10.3390/ijerph19010028
APA StyleMourtakos, S., Vassiliou, G., Kontoangelos, K., Philippou, A., Tzavellas, E., Tornero-Aguilera, J. F., Clemente-Suárez, V. J., Papageorgiou, C., Sidossis, L. S., & Papageorgiou, C. (2022). Endocannabinoids and Heart Rate Variability Alterations after Exposure to Prolonged Intensive Physical Exercise of the Hellenic Navy SEALs. International Journal of Environmental Research and Public Health, 19(1), 28. https://doi.org/10.3390/ijerph19010028










