Manufacturing and Application of 3D Printed Photo Fenton Reactors for Wastewater Treatment
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Lab-Scale Experiments and Analytical Procedures
- A set of primary tests with different materials is carried out to determine the chemical behavior of the pieces of the material before and after the printing process. Criteria #3, Section 3.2.2 Chemical Tests.
- Once the reactor prototypes are printed using each of the selected materials, their viability as a Fenton reactor is assessed. Thus, the same reactions are carried out without UV radiation in the reactor prototypes and, parallelly, in a Pyrex® flask used as a blank test. Criteria #6, Section 3.4.2 Chemical Tests.
- Finally, the same assays are repeated for testing the reactor prototypes under the photo-Fenton environment. This time, the assays are performed under UV irradiation and with caffeine as a contaminant. Caffeine is selected as a convenient substance for these new assays since, besides its easy availability and manageability, it is considered as an emerging contaminant (mostly due to its high water solubility and low degradability) that has been widely studied in the literature [23,24,25,26]. Criteria #6, Section 3.4.2 Chemical Tests.
2.3. Procedures and Equipment
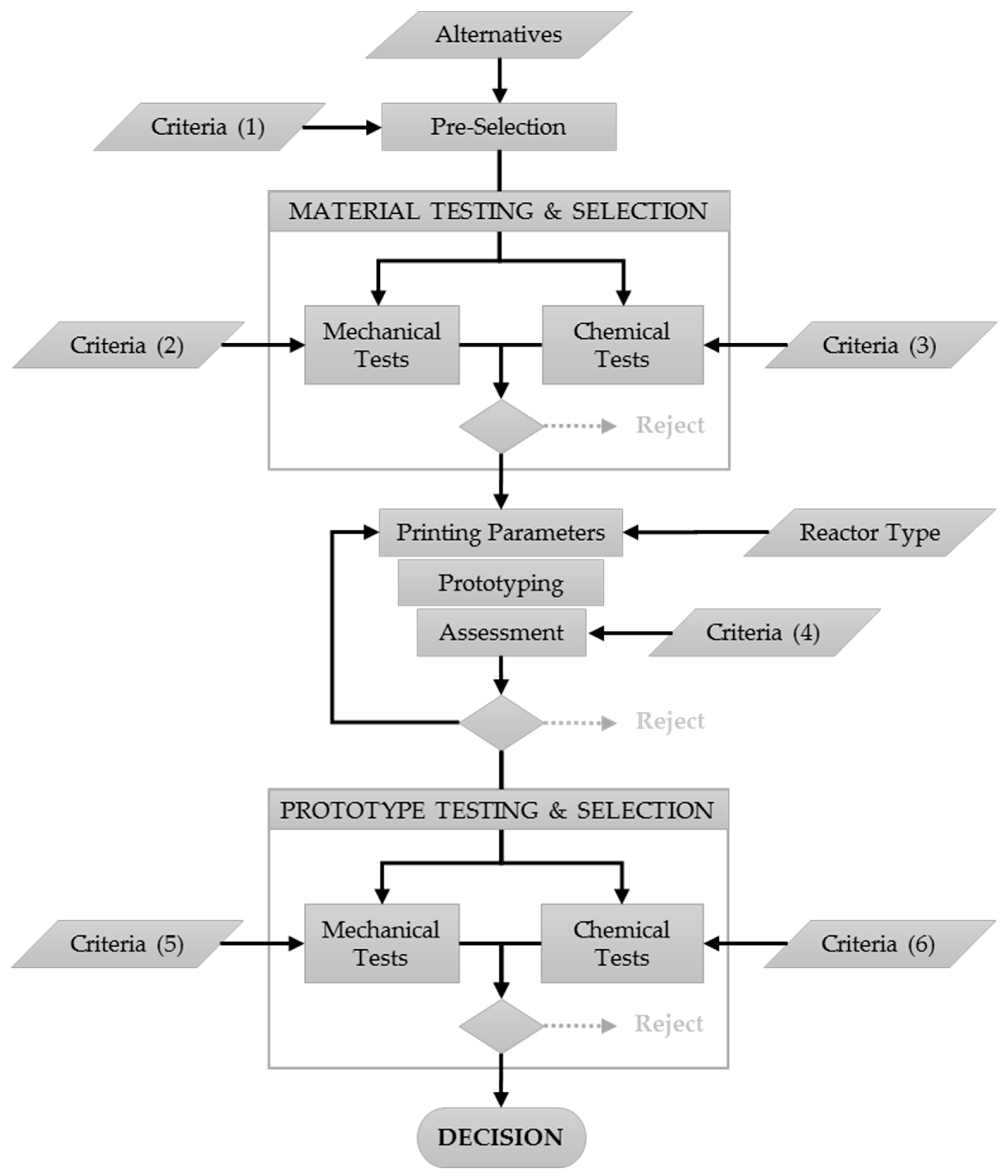
3. Results
3.1. Pre-Selection of Alternative Printing Materials
3.2. Material Testing and Selection
3.2.1. Mechanical Tests
Criterion 2.1: Printability
Criterion 2.2: Stiffness
Criterion 2.3: Heated Bed
3.2.2. Chemical Tests
Criterion 3.1: Reaction Environment
Criterion 3.2: UV Resistance
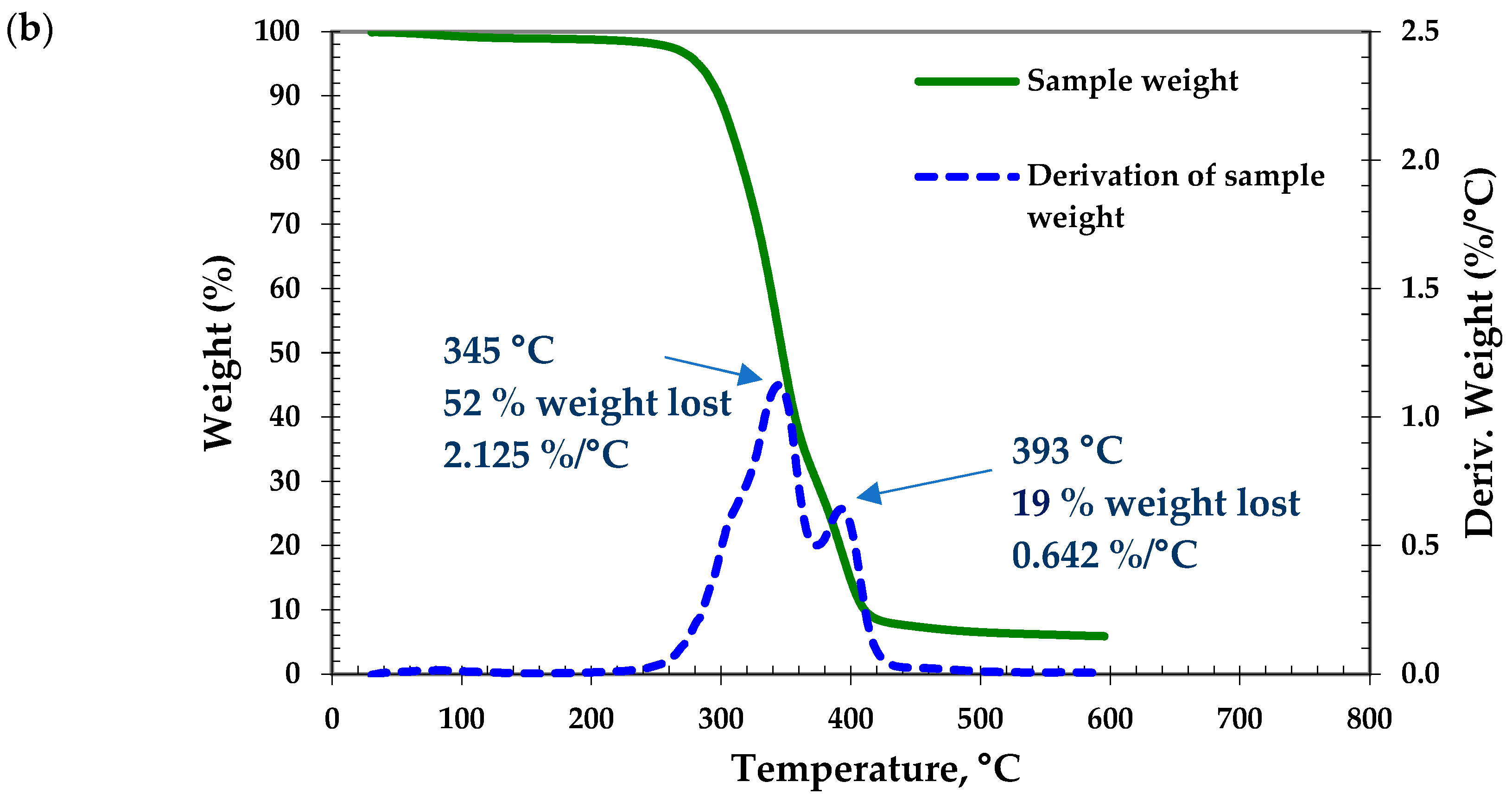
Criterion 3.3: Thermal Stability
3.3. Reactor Type, Prototyping, Printing Parameters
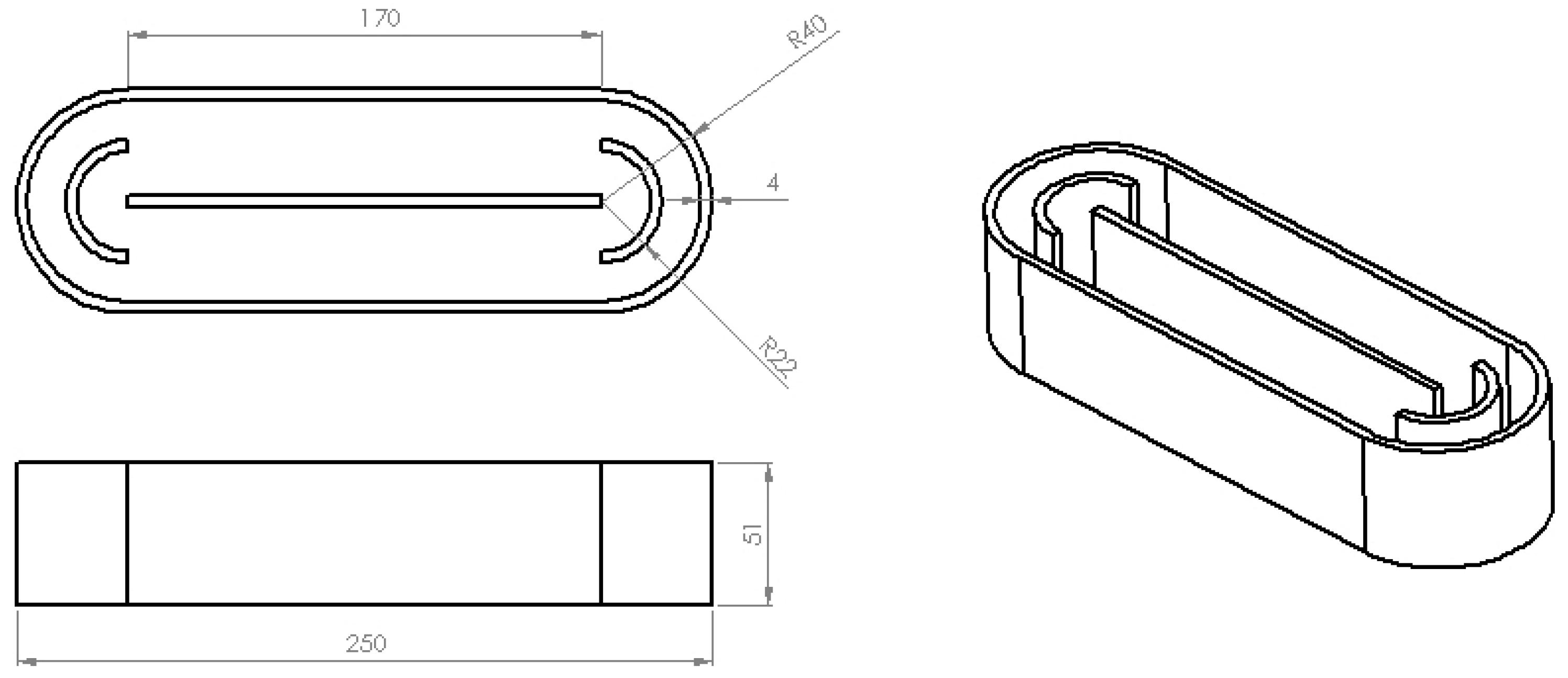
3.3.1. Prototyping
3.3.2. Printing Parameters
3.4. Prototype Testing and Selection
3.4.1. Mechanical Test
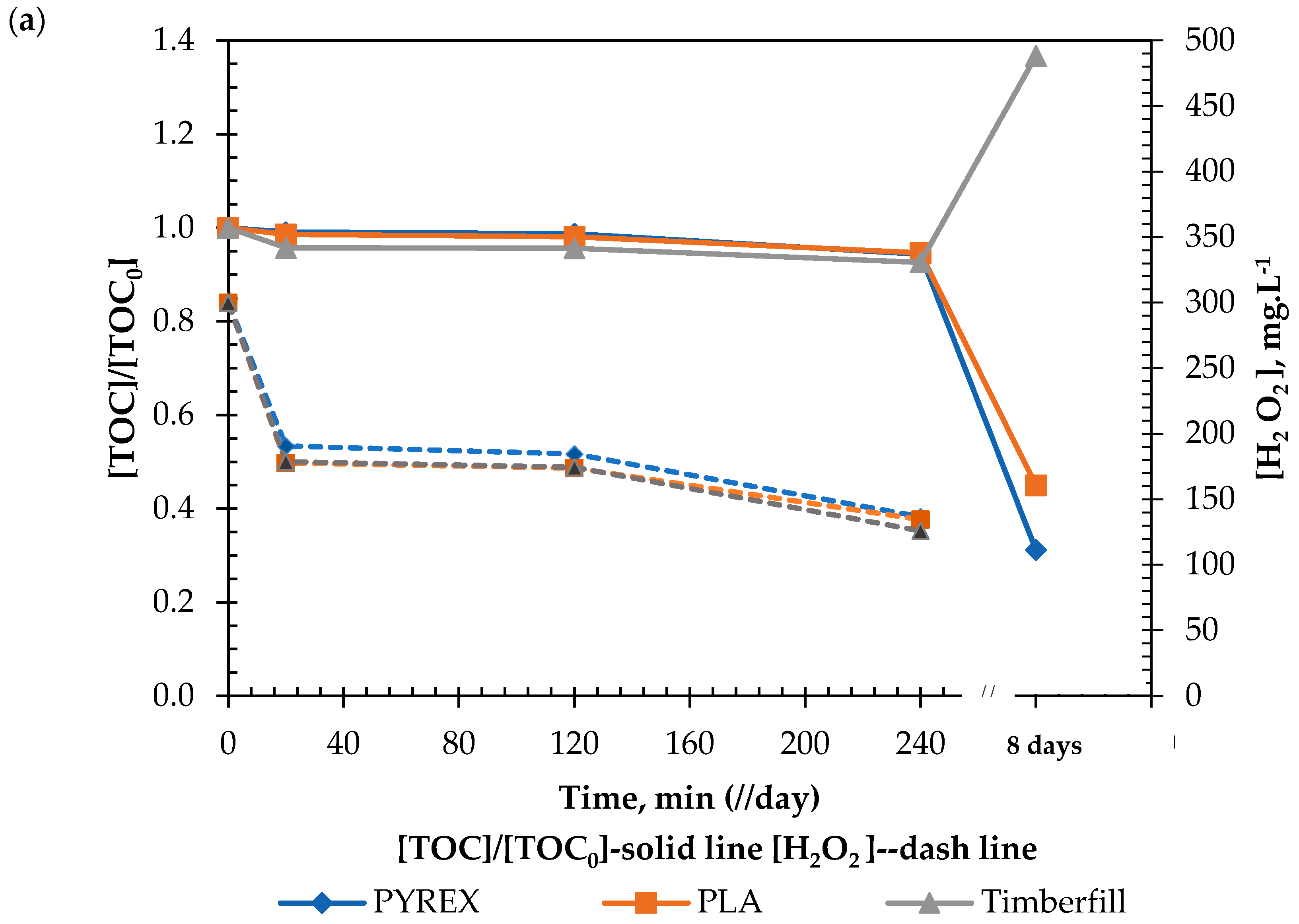
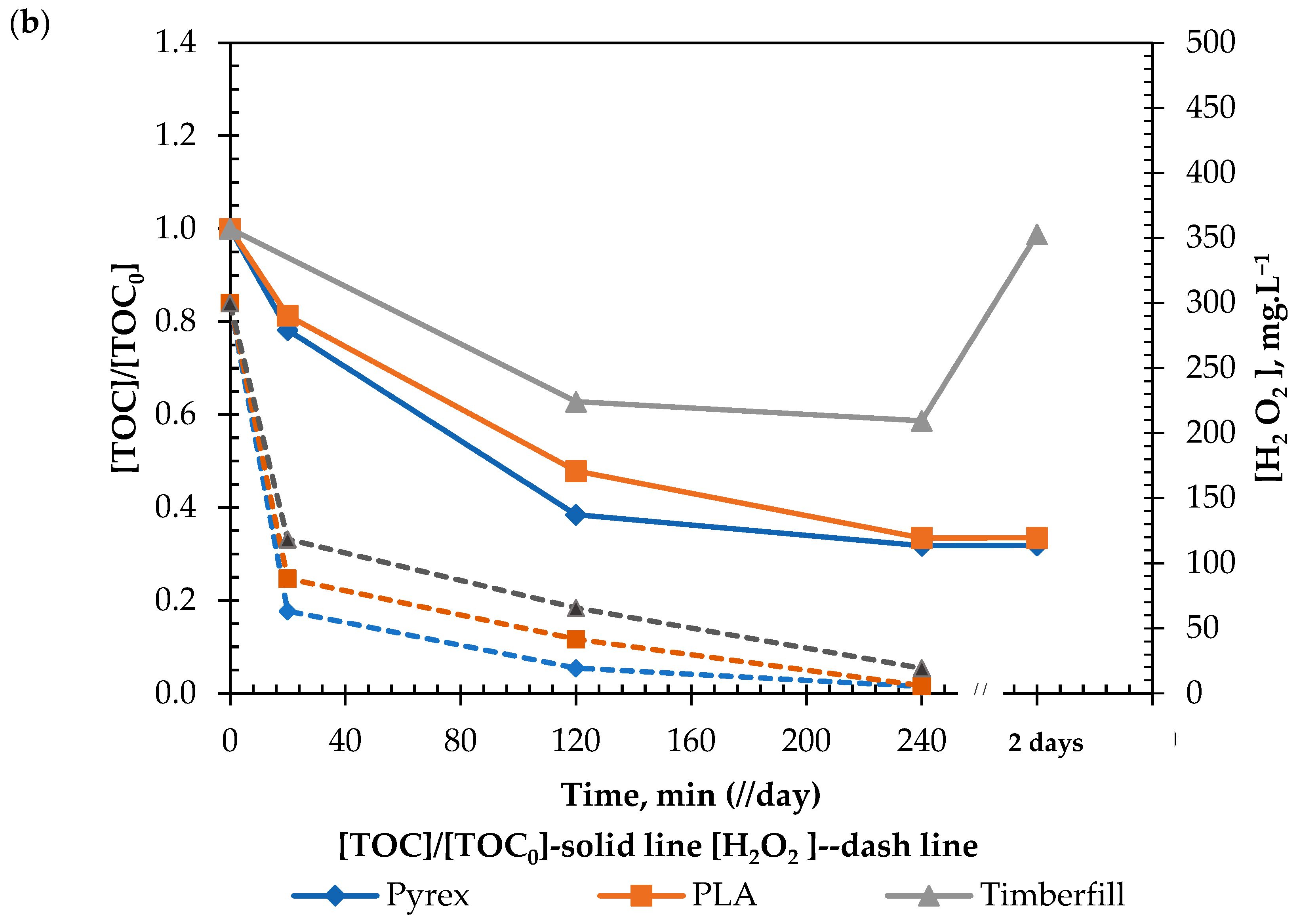
3.4.2. Chemical Test
- Criterion 6.1: Reaction viability; the capacity of the reactor to handle both Fenton and photo-Fenton reactions.
- Criterion 6.2: Material interference; the capability of the different reactors to significantly interfere with these reactions.
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ngo, T.D.; Kashani, A.; Imbalzano, G.; Nguyen, K.T.Q.; Hui, D. Additive manufacturing (3D printing): A review of materials, methods, applications and challenges. Compos. Part B Eng. 2018, 143, 172–196. [Google Scholar] [CrossRef]
- Capel, A.J.; Rimington, R.P.; Lewis, M.P.; Christie, S.D.R. 3D printing for chemical, pharmaceutical and biological applications. Nat. Rev. Chem. 2018, 2, 422–436. [Google Scholar] [CrossRef]
- Parra-Cabrera, C.; Achille, C.; Kuhn, S.; Ameloot, R. 3D printing in chemical engineering and catalytic technology: Structured catalysts, mixers and reactors. Chem. Soc. Rev. 2018, 47, 209–230. [Google Scholar] [CrossRef]
- Bettermann, S.; Kandelhard, F.; Moritz, H.-U.; Pauer, W. Digital and lean development method for 3D-printed reactors based on CAD modeling and CFD simulation. Chem. Eng. Res. Des. 2019, 152, 71–84. [Google Scholar] [CrossRef]
- Maier, M.; Lebl, R.; Sulzer, P.; Lechner, J.; Mayr, T.; Zadravec, M.; Slama, E.; Pfanner, S.; Schmölzer, C.; Pöchlauer, P.; et al. Development of customized 3D printed stainless steel reactors with inline oxygen sensors for aerobic oxidation of Grignard reagents in continuous flow. React. Chem. Eng. 2018, 4. [Google Scholar] [CrossRef]
- Okafor, O.; Robertson, K.; Goodridge, R.; Sans, V. Continuous-flow crystallisation in 3D-printed compact devices. React. Chem. Eng. 2019, 4, 1682–1688. [Google Scholar] [CrossRef]
- Bhattacharjee, N.; Urrios, A.; Kang, S.; Folch, A. The upcoming 3D-printing revolution in microfluidics. Lab Chip 2016, 16. [Google Scholar] [CrossRef]
- Ahmadian Yazdi, A.; Popma, A.; Wong, W.; Nguyen, T.; Pan, Y.; Xu, J. 3D printing: An emerging tool for novel microfluidics and lab-on-a-chip applications. Microfluid. Nanofluidics 2016, 20. [Google Scholar] [CrossRef]
- Kitson, P.; Rosnes, M.; Sans, V.; Dragone, V.; Cronin, L. Configurable 3D-Printed millifluidic and microfluidic “lab on a chip” reactionware devices. Lab Chip. 2012, 12, 3267–3271. [Google Scholar] [CrossRef]
- Hou, W.; Bubliauskas, A.; Kitson, P.J.; Francoia, J.-P.; Powell-Davies, H.; Gutierrez, J.M.P.; Frei, P.; Manzano, J.S.; Cronin, L. Automatic Generation of 3D-Printed Reactionware for Chemical Synthesis Digitization using ChemSCAD. ACS Cent. Sci. 2021, 7, 212–218. [Google Scholar] [CrossRef]
- Elliott, O.; Gray, S.; McClay, M.; Nassief, B.; Nunnelley, A.; Vogt, E.; Ekong, J.; Kardel, K.; Khoshkhoo, A.; Proaño, G.; et al. Design and Manufacturing of High Surface Area 3D-Printed Media for Moving Bed Bioreactors for Wastewater Treatment. J. Contemp. Water Res. Educ. 2017, 160, 144–156. [Google Scholar] [CrossRef]
- Proano-Pena, G.; Carrano, A.L.; Blersch, D.M. Analysis of very-high surface area 3D-printed media in a moving bed biofilm reactor for wastewater treatment. PLoS ONE 2020, 15, e0238386. [Google Scholar] [CrossRef] [PubMed]
- wan, A.; Wu, Y.; Xie, Y.; Zhao, B.; Qi, X. Experimental study and numerical simulation of moving bed biofilm reactor using 3D printed filler. Chem. Eng. Process.–Process Intensif. 2020, 157, 108146. [Google Scholar] [CrossRef]
- Desipio, M.M.; Van Bramer, S.E.; Thorpe, R.; Saha, D. Photocatalytic and photo-fenton activity of iron oxide-doped carbon nitride in 3D printed and LED driven photon concentrator. J. Hazard. Mater. 2019, 376, 178–187. [Google Scholar] [CrossRef]
- Mousset, E.; Huang Weiqi, V.; Foong Yang Kai, B.; Koh, J.S.; Tng, J.W.; Wang, Z.; Lefebvre, O. A new 3D-printed photoelectrocatalytic reactor combining the benefits of a transparent electrode and the Fenton reaction for advanced wastewater treatment. J. Mater. Chem. A 2017, 5, 24951–24964. [Google Scholar] [CrossRef]
- Carra, I.; Santos-Juanes, L.; Acien, G.; Malato, S.; Sánchez Pérez, J.A. New approach to solar photo-Fenton operation. Raceway ponds as tertiary treatment technology. J. Hazard. Mater. 2014, 279, 322–329. [Google Scholar] [CrossRef]
- Nasr Esfahani, K.; Farhadian, M.; Solaimany Nazar, A.R. Interaction effects of various reaction parameters on the treatment of sulfidic spent caustic through electro-photo-Fenton. Int. J. Environ. Sci. Technol. 2019, 16, 7165–7174. [Google Scholar] [CrossRef]
- Obra, I.; Ponce Robles, L.; Miralles-Cuevas, S.; Oller, I.; Malato, S.; Sánchez Pérez, J.A. Microcontaminant removal in secondary effluents by solar photo-Fenton at circumneutral pH in raceway pond reactors. Catal. Today 2016, 287. [Google Scholar] [CrossRef]
- Keenan, C.R.; Sedlak, D.L. Ligand-Enhanced Reactive Oxidant Generation by Nanoparticulate Zero-Valent Iron and Oxygen. Environ. Sci. Technol. 2008, 42, 6936–6941. [Google Scholar] [CrossRef]
- Falyouna, O.; Maamoun, I.; Bensaida, K.; Sugihara, Y.; Eljamal, O. Removal of Ciprofloxacin from Aqueous Solutions by Nanoscale Zerovalent Iron-Based Materials: A Mini Review. In Proceedings of the International Exchange and Innovation Conference on Engineering & Sciences (IEICES), Fukuoka, Japan, 22–23 October 2020; Volume 6, pp. 179–185. [Google Scholar] [CrossRef]
- Lee, C.; Keenan, C.R.; Sedlak, D.L. Polyoxometalate-Enhanced Oxidation of Organic Compounds by Nanoparticulate Zero-Valent Iron and Ferrous Ion in the Presence of Oxygen. Environ. Sci. Technol. 2008, 42, 4921–4926. [Google Scholar] [CrossRef]
- Erokhin, K.S.; Gordeev, E.G.; Ananikov, V.P. Revealing interactions of layered polymeric materials at solid-liquid interface for building solvent compatibility charts for 3D printing applications. Sci. Rep. 2019, 9, 20177. [Google Scholar] [CrossRef]
- Rodríguez-Gil, J.L.; Catalá, M.; Alonso, S.G.; Maroto, R.R.; Valcárcel, Y.; Segura, Y.; Molina, R.; Melero, J.A.; Martínez, F. Heterogeneous photo-Fenton treatment for the reduction of pharmaceutical contamination in Madrid rivers and ecotoxicological evaluation by a miniaturized fern spores bioassay. Chemosphere 2010, 80, 381–388. [Google Scholar] [CrossRef]
- Gómez, M.J.; Martínez Bueno, M.J.; Lacorte, S.; Fernández-Alba, A.R.; Agüera, A. Pilot survey monitoring pharmaceuticals and related compounds in a sewage treatment plant located on the Mediterranean coast. Chemosphere 2007, 66, 993–1002. [Google Scholar] [CrossRef]
- Tokumura, M.; Ohta, A.; Znad, H.T.; Kawase, Y. UV light assisted decolorization of dark brown colored coffee effluent by photo-Fenton reaction. Water Res. 2006, 40, 3775–3784. [Google Scholar] [CrossRef]
- Tokumura, M.; Znad, H.T.; Kawase, Y. Decolorization of dark brown colored coffee effluent by solar photo-Fenton reaction: Effect of solar light dose on decolorization kinetics. Water Res. 2008, 42, 4665–4673. [Google Scholar] [CrossRef]
- Nogueira, R.F.P.; Oliveira, M.C.; Paterlini, W.C. Simple and fast spectrophotometric determination of H2O2 in photo-Fenton reactions using metavanadate. Talanta 2005, 66, 86–91. [Google Scholar] [CrossRef] [PubMed]
- ASTM D638-14. Standard Test Method for Tensile Properties of Plastics; ASTM International: West Conshohocken, PA, USA, 2014. [Google Scholar]
- Singh, S.; Ramakrishna, S.; Singh, R. Material issues in additive manufacturing: A review. J. Manuf. Process. 2017, 25, 185–200. [Google Scholar] [CrossRef]
- Gyak, K.; Vishwakarma, N.; Hwang, Y.-H.; Kim, J.; Yun, H.; Kim, D. 3D-printed Monolithic SiCN Ceramic Microreactors from a Photocurable Preceramic Resin for High Temperature Ammonia Cracking Process. React. Chem. Eng. 2019, 4. [Google Scholar] [CrossRef]
- Gross, B.C.; Erkal, J.L.; Lockwood, S.Y.; Chen, C.; Spence, D.M. Evaluation of 3D Printing and Its Potential Impact on Biotechnology and the Chemical Sciences. Anal. Chem. 2014, 86, 3240–3253. [Google Scholar] [CrossRef] [PubMed]
- Bogdan, E.; Michorczyk, P. 3D Printing in Heterogeneous Catalysis—The State of the Art. Materials 2020, 13, 4534. [Google Scholar] [CrossRef]
- Tang, X.; Alavi, S. Recent advances in starch, polyvinyl alcohol based polymer blends, nanocomposites and their biodegradability. Carbohydr. Polym. 2011, 85, 7–16. [Google Scholar] [CrossRef]
- Rohde, S.; Cantrell, J.; Jerez, A.; Krese, C.; Damiani, D.; Gurnani, R.; DiSandro, L.; Anton, J.; Young, A.; Steinbach, D.; et al. Experimental Characterization of the Shear Properties of 3D–Printed ABS and Polycarbonate Parts. Exp. Mech. 2018, 58, 871–884. [Google Scholar] [CrossRef]
- Fries, J.; Durna, A. Recycling of Used Filament from 3D Printing. In Proceedings of the 18th International Multidisciplinary Scientific GeoConference SGEM, Vienna, Austria, 3–6 December 2018; pp. 153–160. [Google Scholar]
- Zandi, M.D.; Jerez-Mesa, R.; Lluma-Fuentes, J.; Jorba-Peiro, J.; Travieso-Rodriguez, J.A. Study of the manufacturing process effects of fused filament fabrication and injection molding on tensile properties of composite PLA-wood parts. Int. J. Adv. Manuf. Technol. 2020, 108, 1725–1735. [Google Scholar] [CrossRef]
- Jiao, Y.; Fish, J. Is an additive decomposition of a rate of deformation and objective stress rates passé? Comput. Methods Appl. Mech. Eng. 2017, 327, 196–225. [Google Scholar] [CrossRef]
- Xiao, H.; Bruhns, O.T.; Meyers, A. Elastoplasticity beyond small deformations. Acta Mech. 2006, 182, 31–111. [Google Scholar] [CrossRef]
- Ward, I.M.; Sweeney, J. An Introduction to the Mechanical Properties of Solid Polymers; Wiley: Hoboken, NJ, USA, 2004; ISBN 978-0-470-02037-1. [Google Scholar]
- Mackay, M.; Swain, Z.; Banbury, C.; Phan, D.; Edwards, D. The performance of the hot end in a plasticating 3D printer. J. Rheol. (N. Y.) 2017, 61, 229–236. [Google Scholar] [CrossRef]
- Prajapati, H.; Ravoori, D.; Jain, A. Measurement and modeling of filament temperature distribution in the standoff gap between nozzle and bed in polymer-based additive manufacturing. Addit. Manuf. 2018, 24, 224–231. [Google Scholar] [CrossRef]
- Bellini, A.; Güçeri, S.; Bertoldi, M. Liquefier Dynamics in Fused Deposition. J. Manuf. Sci. Eng. Asme 2004, 126. [Google Scholar] [CrossRef]
- Peng, F.; Vogt, B.D.; Cakmak, M. Complex flow and temperature history during melt extrusion in material extrusion additive manufacturing. Addit. Manuf. 2018, 22, 197–206. [Google Scholar] [CrossRef]
- Ajinjeru, C.; Kishore, V.; Liu, P.; Lindahl, J.; Hassen, A.A.; Kunc, V.; Post, B.; Love, L.; Duty, C. Determination of melt processing conditions for high performance amorphous thermoplastics for large format additive manufacturing. Addit. Manuf. 2018, 21, 125–132. [Google Scholar] [CrossRef]
- Pigeonneau, F.; Xu, D.; Vincent, M.; Agassant, J.-F. Heating and flow computations of an amorphous polymer in the liquefier of a material extrusion 3D printer. Addit. Manuf. 2020, 32, 101001. [Google Scholar] [CrossRef]
- Baumgart, E. Stiffness—An unknown world of mechanical science? Injury 2000, 31 (Suppl. S2). [Google Scholar] [PubMed]
- Zandi, M.D.; Jerez-Mesa, R.; Llumà, J.; Roa, J.; Travieso-Rodriguez, J.A. Experimental analysis of manufacturing parameters’ effect on the flexural properties of wood-PLA composite parts built through FFF. Int. J. Adv. Manuf. Technol. 2020, 106. [Google Scholar] [CrossRef]
- Travieso-Rodriguez, J.A.; Jerez-Mesa, R.; Llumà, J.; Gómez-Gras, G.; Casadesús-Farràs, O. Comparative study of the flexural properties of ABS, PLA and a PLA-wood composite manufactured through fused filament fabrication. Rapid Prototyp. J. 2020, 27. [Google Scholar] [CrossRef]
- Kotz, F.; Risch, P.; Helmer, D.; Rapp, B.E. High-Performance Materials for 3D Printing in Chemical Synthesis Applications. Adv. Mater. 2019, 31, 1805982. [Google Scholar] [CrossRef] [PubMed]
- Santos, R.M.; Botelho, G.L.; Machado, A. V Artificial and natural weathering of ABS. J. Appl. Polym. Sci. 2010, 116, 2005–2014. [Google Scholar] [CrossRef]
- Rao, Z.X.; Patel, B.; Monaco, A.; Cao, Z.J.; Barniol-Xicota, M.; Pichon, E.; Ladlow, M.; Hilton, S.T. 3D-Printed Polypropylene Continuous-Flow Column Reactors: Exploration of Reactor Utility in SNAr Reactions and the Synthesis of Bicyclic and Tetracyclic Heterocycles. European J. Org. Chem. 2017, 2017, 6499–6504. [Google Scholar] [CrossRef]
- Rossi, S.; Porta, R.; Brenna, D.; Puglisi, A.; Benaglia, M. Stereoselective Catalytic Synthesis of Active Pharmaceutical Ingredients in Homemade 3D-Printed Mesoreactors. Angew. Chemie Int. Ed. 2017, 56, 4290–4294. [Google Scholar] [CrossRef]
- Neumaier, J.M.; Madani, A.; Klein, T.; Ziegler, T. Low-budget 3D-printed equipment for continuous flow reactions. Beilstein J. Org. Chem. 2019, 15, 558–566. [Google Scholar] [CrossRef]
- Sans, V. Emerging trends in flow chemistry enabled by 3D printing: Robust reactors, biocatalysis and electrochemistry. Curr. Opin. Green Sustain. Chem. 2020, 25, 100367. [Google Scholar] [CrossRef]
- Pignatello, J.; Oliveros, E.; MacKay, A. Advanced Oxidation Processes for Organic Contaminant Destruction Based on Fenton Reaction and Related Chemistry. Crit. Rev. Environ. Sci. Technol. 2006, 36, 1–84. [Google Scholar] [CrossRef]
- Rozas, O.; Contreras, D.; Mondaca, M.A.; Pérez-Moya, M.; Mansilla, H.D. Experimental design of Fenton and photo-Fenton reactions for the treatment of ampicillin solutions. J. Hazard. Mater. 2010, 177, 1025–1030. [Google Scholar] [CrossRef]
- Yamal-Turbay, E.; Graells, M.; Pérez-Moya, M. Systematic Assessment of the Influence of Hydrogen Peroxide Dosage on Caffeine Degradation by the Photo-Fenton Process. Ind. Eng. Chem. Res. 2012, 51, 4770–4778. [Google Scholar] [CrossRef]
- DOGC Reglament dels serveis públics de sanejament. D. Of. la General. Catalunya 2003, 11143–11158.
- Romero, V.; Acevedo, S.; Pilar, M.; Giménez, J.; Esplugas, S. Enhancement of Fenton and photo-Fenton processes at initial circumneutral pH for the degradation of the β-blocker Metoprolol. Water Res. 2015, 88, 449–457. [Google Scholar] [CrossRef]
- Audino, F.; Campanyà, G.; Pérez-Moya, M.; Espuña, A.; Graells, M. Systematic optimization approach for the efficient management of the photo-Fenton treatment process. Sci. Total Environ. 2019, 646, 902–913. [Google Scholar] [CrossRef] [PubMed]
- Afshar, A.; Mihut, D. Enhancing durability of 3D printed polymer structures by metallization. J. Mater. Sci. Technol. 2020, 53, 185–191. [Google Scholar] [CrossRef]
- Zhang, S.-U. Degradation Classification of 3D Printing Thermoplastics Using Fourier Transform Infrared Spectroscopy and Artificial Neural Networks. Appl. Sci. 2018, 8, 1224. [Google Scholar] [CrossRef]
- Wu, G.; Liu, S.; Jia, H.; Dai, J. Preparation and properties of heat resistant polylactic acid (PLA)/Nano-SiO2 composite filament. J. Wuhan Univ. Technol. Sci. Ed. 2016, 31, 164–171. [Google Scholar] [CrossRef]
- Zazo, J.A.; Pliego, G.; Blasco, S.; Casas, J.A.; Rodriguez, J.J. Intensification of the Fenton Process by Increasing the Temperature. Ind. Eng. Chem. Res. 2011, 50, 866–870. [Google Scholar] [CrossRef]
- Giwa, A.-R.A.; Bello, I.A.; Olabintan, A.B.; Bello, O.S.; Saleh, T.A. Kinetic and thermodynamic studies of fenton oxidative decolorization of methylene blue. Heliyon 2020, 6, e04454. [Google Scholar] [CrossRef]
- Ng, H.M.; Mohamad Saidi, N.; Omar, F.S.; Kasi, R.; T subramaniam, R.; Baig, S. Thermogravimetric Analysis of Polymers. Encycl. Polym. Sci. Technol. 2018, 1–29. [Google Scholar] [CrossRef]
- Wojtyła, S.; Klama, P.; Baran, T. Is 3D printing safe? Analysis of the thermal treatment of thermoplastics: ABS, PLA, PET, and nylon. J. Occup. Environ. Hyg. 2017, 14. [Google Scholar] [CrossRef] [PubMed]
- Cabrera-Reina, A.; Miralles-Cuevas, S.; Rivas, G.; Sánchez Pérez, J.A. Comparison of different detoxification pilot plants for the treatment of industrial wastewater by solar photo-Fenton: Are raceway pond reactors a feasible option? Sci. Total Environ. 2019, 648, 601–608. [Google Scholar] [CrossRef] [PubMed]
- Morales-Planas, S.; Minguella-Canela, J.; Lluma-Fuentes, J.; Travieso-Rodriguez, J.A.; García-Granada, A.-A. Multi Jet Fusion PA12 Manufacturing Parameters for Watertightness, Strength and Tolerances. Materials 2018, 11, 1472. [Google Scholar] [CrossRef]
- De Ciurana, J.; Serenóa, L.; Vallès, È. Selecting Process Parameters in RepRap Additive Manufacturing System for PLA Scaffolds Manufacture. Procedia CIRP. 2013, 5, 152–157. [Google Scholar] [CrossRef]
- Afrose, M.F.; Masood, S.H.; Iovenitti, P.; Nikzad, M.; Sbarski, I. Effects of part build orientations on fatigue behaviour of FDM-processed PLA material. Prog. Addit. Manuf. 2016, 1, 21–28. [Google Scholar] [CrossRef]
- ASTM D6272. Standard Test Method for Flexural Properties of Unreinforced and Reinforced Plastics And electrical Insulating Materials by Four-Point Bending; ASTM International: West Conshohocken, PA, USA, 2002. [Google Scholar]
- Travieso-Rodriguez, J.A.; Jerez-Mesa, R.; Llumà, J.; Traver-Ramos, O.; Gomez-Gras, G.; Roa Rovira, J.J. Mechanical Properties of 3D-Printing Polylactic Acid Parts subjected to Bending Stress and Fatigue Testing. Materials 2019, 12, 3859. [Google Scholar] [CrossRef]




| Metallic | Ceramic | Polymeric Base |
|---|---|---|
| Titanium | Alumina | Nylon |
| Aluminum | Zircon dioxide | Polycarbonate (PC) |
| Stainless steel | Hydroxyapatite | Polyvinyl alcohol (PVA) |
| Copper | Titanium oxide | Acrylonitrile butadiene styrene (ABS) |
| Inconel | Tri-calcium phosphate | Polylactic acid (PLA) |
| Gold/Platinum | Bio-glass | Composite PLA–wood fibers (Timberfill®) |
| Criteria #1 | Criterion 1.1 Chemical Properties | Criterion 1.2 Mechanical Properties | Criterion 1.3 Manufacturing Cost | Decision |
|---|---|---|---|---|
| Metallic | Passed | Passed | Failed | Rejected |
| Ceramic | Passed | Passed | Failed | Rejected |
| Polymeric | Passed | Passed | Passed | Selected |
| Composite | Passed | Passed | Passed | Selected |
| Criteria #1 | Criterion 1.4 Cost | Criterion 1.5 Heat Resistance | Criterion 1.6 Mechanical Strength | Criterion 1.7 Sustainability | Criterion 1.8 Water Solubility | Decision |
|---|---|---|---|---|---|---|
| Nylon | Failed | Failed | Passed | Non-biodegradable | Passed | Rejected |
| PC | Failed | Passed | Passed | Biodegradable | Passed | Rejected |
| PVA | Failed | Passed | Passed | Biodegradable | Failed | Rejected |
| ABS | Passed | Passed | Failed | Non-biodegradable | Passed | Selected |
| PLA | Passed | Passed | Passed | Biodegradable | Passed | Selected |
| Timberfill® | Passed | Passed | Failed | Biodegradable | Passed | Selected |
| Criteria #2 | Criterion 2.1 Printability | Criterion 2.2 Stiffness | Criterion 2.3 Heated Bed | Decision |
|---|---|---|---|---|
| PLA | Passed | Passed | Not Required | Selected |
| Timberfill® | Passed | Passed | Not Required | Selected |
| ABS | Passed | Failed | Required | Rejected |
| Criteria #3 | Criterion 3.1 & 3.2 Reaction Environment & Light Resistance | Criterion 3.3 Thermal Stability | Decision |
|---|---|---|---|
| PLA | Passed | Passed | Selected |
| Timberfill® | Passed | Passed | Selected |
| ABS | Failed | - | Rejected |
| Cost | Efficiency (Common Polluted Wastewater) | Efficiency (High Polluted Wastewater) | Treatment Capacity | Accumulated Energy | Decision | |
|---|---|---|---|---|---|---|
| RPR | Passed | Passed | Passed | Passed | Passed | Selected |
| CPC | Failed | Passed | Passed | Passed | Passed | Rejected |
| FP | Failed | Passed | Passed | Passed | Passed | Rejected |
| Thickness (mm) | σ von-Mises (MPa) | Maximum Design Stress (Safety Factor 1.5 Mpa) | Timberfill® Maximum Stress (Mpa) | PLA Maximum Stress (Mpa) |
|---|---|---|---|---|
| 40 | 38.4 | 57.5 | ||
| 45 | 28.9 | 43.3 | 47.26 ± 0.86 | 109.50 ± 4.70 |
| 50 | 22.1 | 33.1 |
| Criteria #4 | Criterion 4.1 Maximum Stress | Decision |
|---|---|---|
| Timberfill® | Passed | Worst option |
| PLA | Passed | Best option |
| Printing Parameters | |||
| Parameter | Value | Parameter | Value |
| Contour width | 1.2 mm | Brim | 5 mm |
| Solid upper layers width | 1.2 mm | Overlap/contour intersection | 15% |
| Solid lower layers width | 1.2 mm | Support material | No |
| Extra contour | Required | Space between filaments | 1.5 mm |
| Combine filling every | 2 layers | Raft (base layer) | No |
| Flow ratios | 1 | Speed trips in vacuum | 130 mm/s |
| Extruder Parameters | |||
| Retraction length | 2 mm | Extra length when reprinting | 0 mm |
| Raise in Z | 0 mm | Minimum distance for shrinkage | 2 mm |
| Speed retraction | 40 mm/s | Infill Pattern | Honeycomb |
| Layer height (mm) | 0.2 | Density (%) | 75 |
| Nozzle diameter (mm) | 0.6 for PLA, 0.7 for Timberfill® | ||
| Printing velocity (mm/s) | 40 for PLA, 30 for Timberfill® | ||
| Criteria #5 | Criterion 5.1 Leakage | Decision |
|---|---|---|
| Timberfill® | Passed | Selected |
| PLA | Passed | Selected |
| Criteria #6 | Criterion 6.1 Reaction Viability | Criterion 6.2 Material Interference | Decision |
|---|---|---|---|
| PLA | Passed | Not observed | Selected |
| Timberfill® | Passed | Observed | Rejected |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nasr Esfahani, K.; Zandi, M.D.; Travieso-Rodriguez, J.A.; Graells, M.; Pérez-Moya, M. Manufacturing and Application of 3D Printed Photo Fenton Reactors for Wastewater Treatment. Int. J. Environ. Res. Public Health 2021, 18, 4885. https://doi.org/10.3390/ijerph18094885
Nasr Esfahani K, Zandi MD, Travieso-Rodriguez JA, Graells M, Pérez-Moya M. Manufacturing and Application of 3D Printed Photo Fenton Reactors for Wastewater Treatment. International Journal of Environmental Research and Public Health. 2021; 18(9):4885. https://doi.org/10.3390/ijerph18094885
Chicago/Turabian StyleNasr Esfahani, Kourosh, Mohammad Damous Zandi, J. Antonio Travieso-Rodriguez, Moisès Graells, and Montserrat Pérez-Moya. 2021. "Manufacturing and Application of 3D Printed Photo Fenton Reactors for Wastewater Treatment" International Journal of Environmental Research and Public Health 18, no. 9: 4885. https://doi.org/10.3390/ijerph18094885
APA StyleNasr Esfahani, K., Zandi, M. D., Travieso-Rodriguez, J. A., Graells, M., & Pérez-Moya, M. (2021). Manufacturing and Application of 3D Printed Photo Fenton Reactors for Wastewater Treatment. International Journal of Environmental Research and Public Health, 18(9), 4885. https://doi.org/10.3390/ijerph18094885










