Prevalence and Risk Factors of Infection in the Representative COVID-19 Cohort Munich
Abstract
1. Introduction
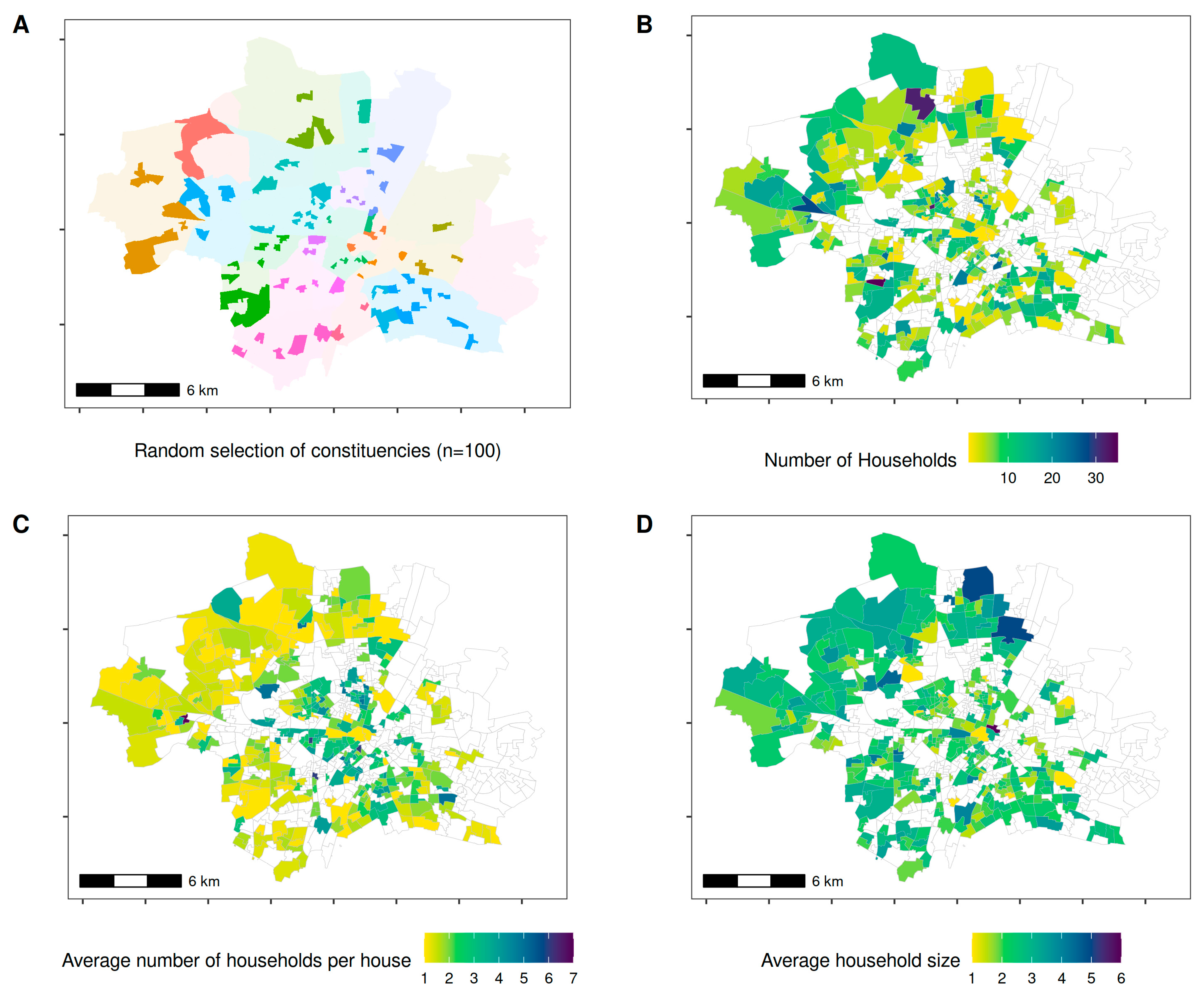
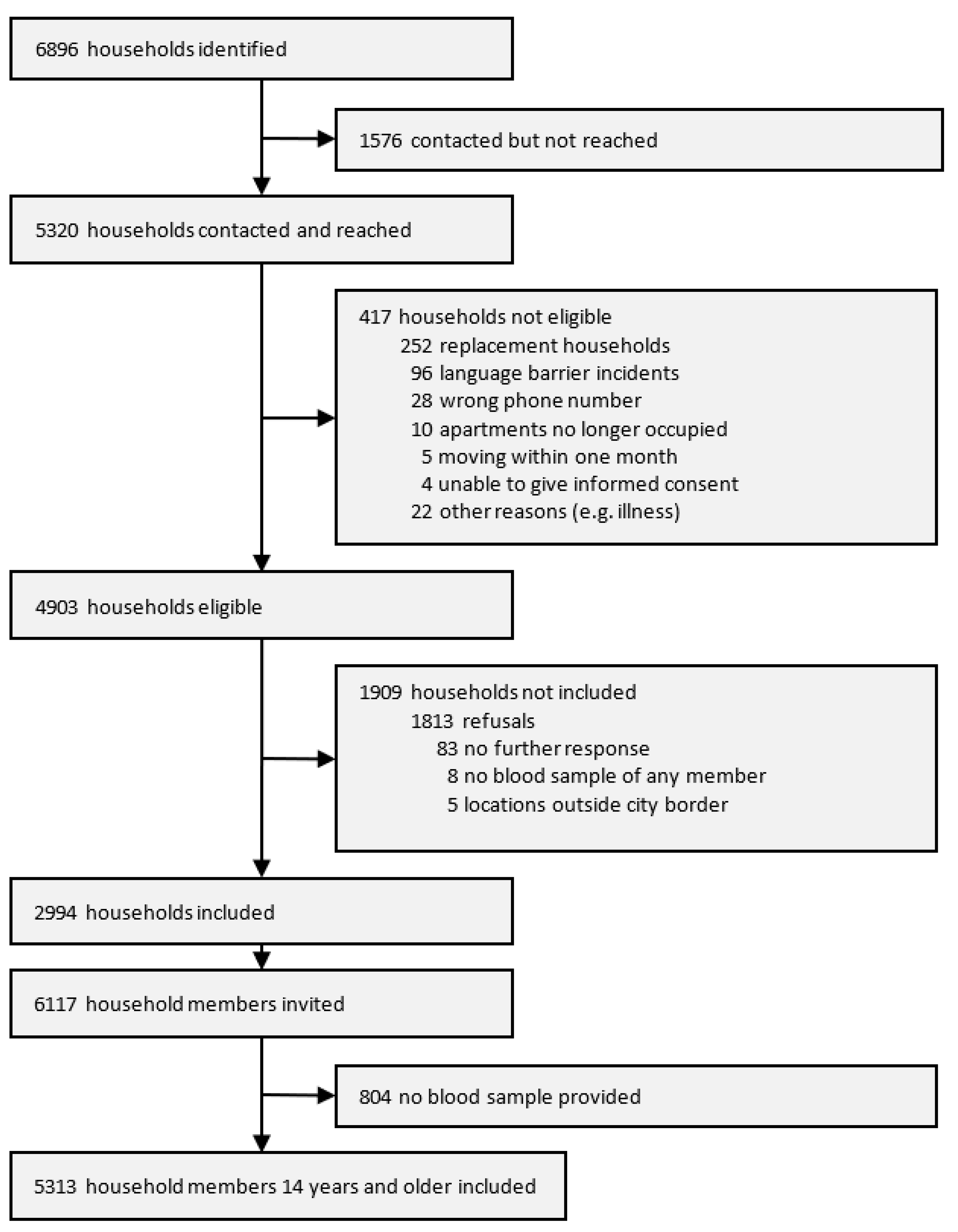
2. Methods
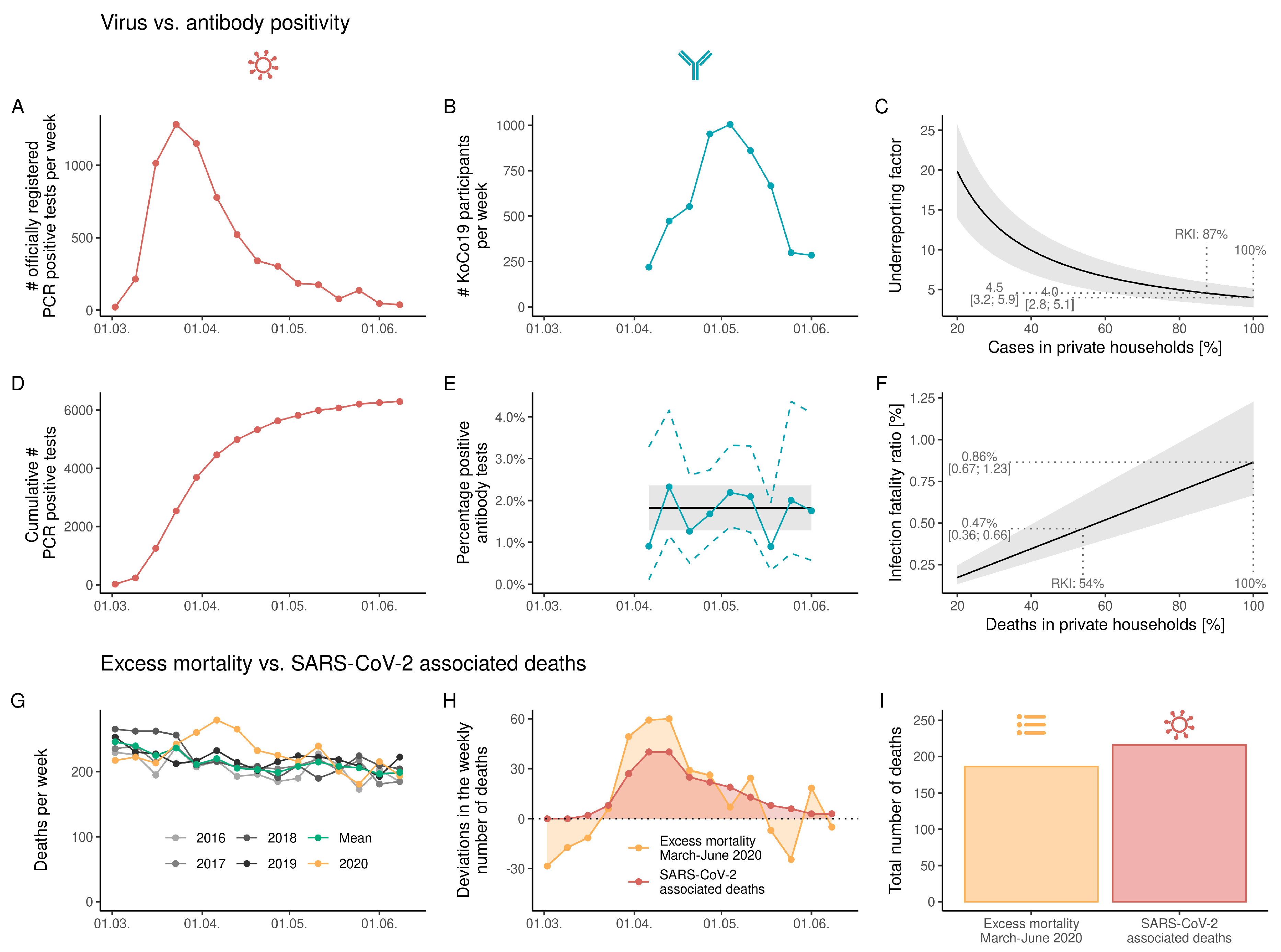
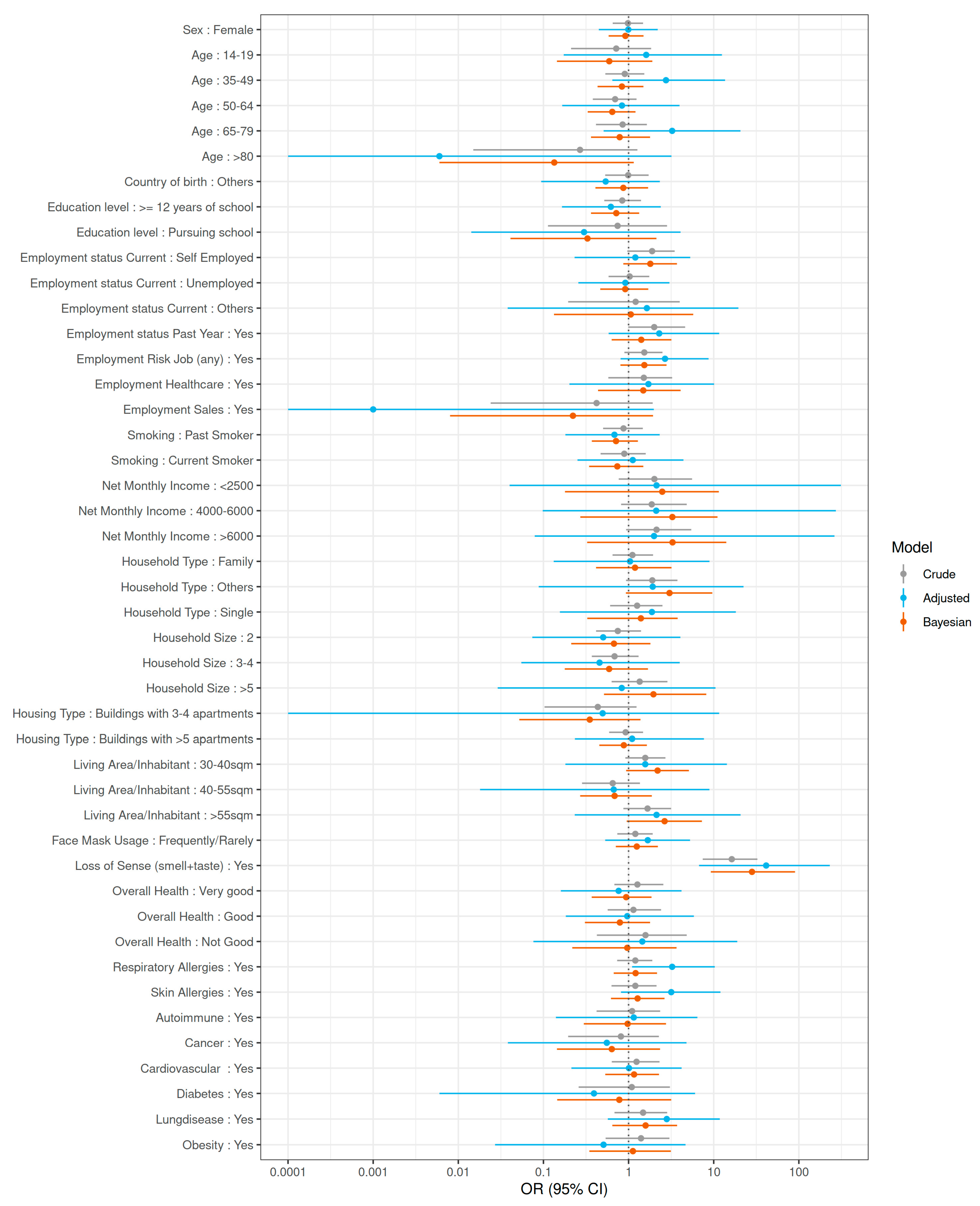
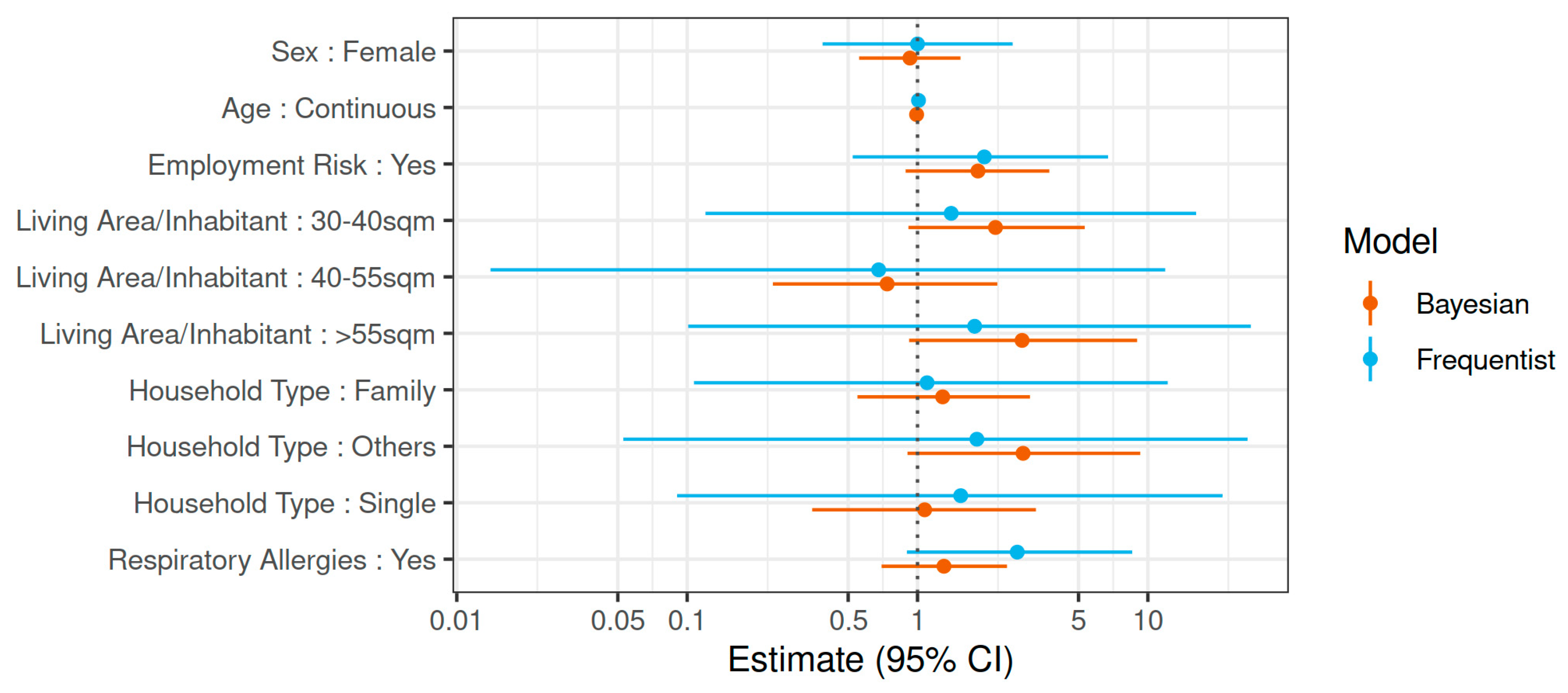
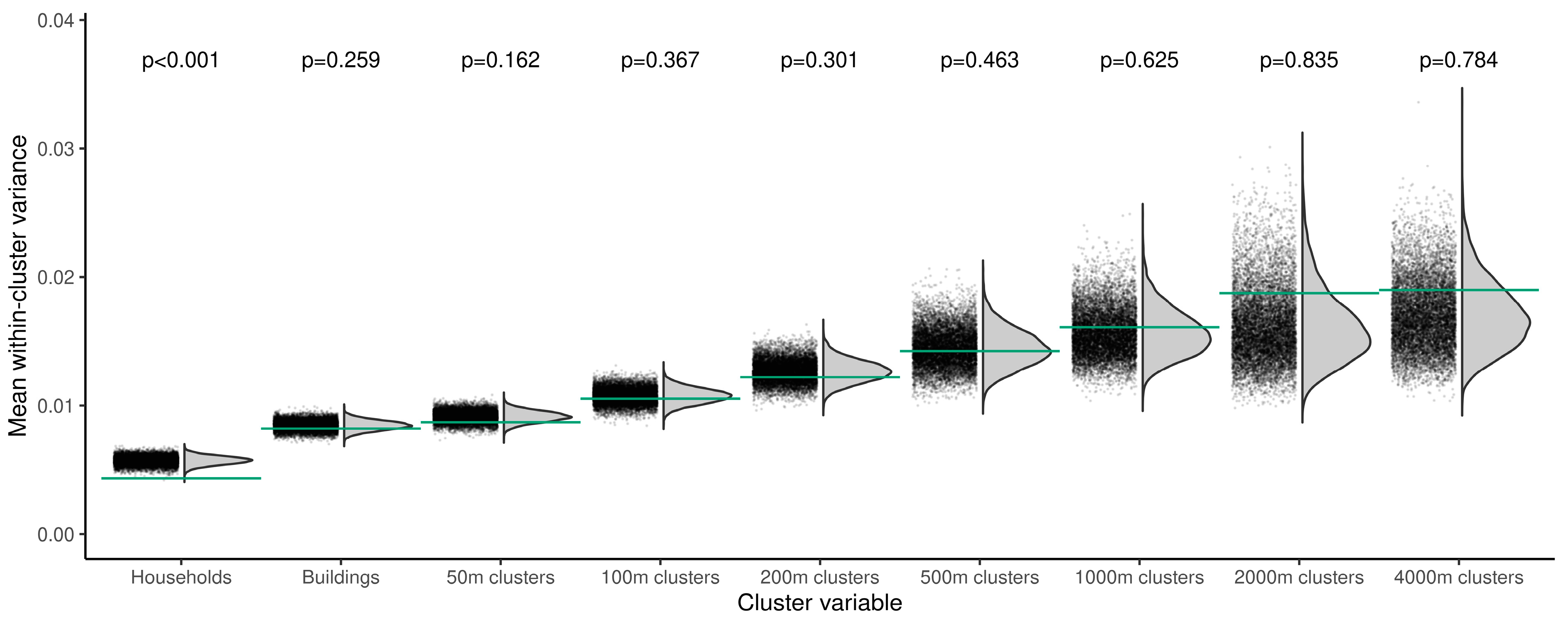
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Rothe, C.; Schunk, M.; Sothmann, P.; Bretzel, G.; Froeschl, G.; Wallrauch, C.; Zimmer, T.; Thiel, V.; Janke, C.; Guggemos, W.; et al. Transmission of 2019-nCoV Infection from an Asymptomatic Contact in Germany. N. Engl. J. Med. 2020, 382, 970–971. [Google Scholar] [CrossRef]
- Coronavirus in München: Die Aktuelle Entwicklung [Internet]. 2020. Available online: https://www.muenchen.de/aktuell/2020-03/coronavirus-muenchen-infektion-aktueller-stand.html (accessed on 9 October 2020).
- Bundesministerium für Gesundheit: Coronavirus SARS-CoV-2: Chronik der Bisherigen Maßnahmen [Internet]. 2021. Available online: https://www.bundesgesundheitsministerium.de/coronavirus/chronik-coronavirus.html (accessed on 17 March 2021).
- Pearce, N.; Vandenbroucke, J.P.; VanderWeele, T.J.; Greenland, S. Accurate Statistics on COVID-19 Are Essential for Policy Guidance and Decisions. Am. J. Public Health 2020, 110, 949–951. [Google Scholar] [CrossRef] [PubMed]
- Meyerowitz-Katz, G.; Merone, L. A systematic review and meta-analysis of published research data on COVID-19 infection fatality rates. Int. J. Infect. Dis. 2020, 101, 138–148. [Google Scholar] [CrossRef]
- Gudbjartsson, D.F.; Norddahl, G.L.; Melsted, P.; Gunnarsdottir, K.; Holm, H.; Eythorsson, E.; Arnthorsson, A.O.; Helgason, D.; Bjarnadottir, K.; Ingvarsson, R.F.; et al. Humoral Immune Response to SARS-CoV-2 in Iceland. N. Engl. J. Med. 2020, 383, 1724–1734. [Google Scholar] [CrossRef]
- Shakiba, M.; Hashemi Nazari, S.S.; Mehrabian, F.; Rezvani, S.M.; Ghasempour, Z.; Heidarzadeh, A. Seroprevalence of COVID-19 Virus Infection in Guilan Province, Iran [Internet]. Infectious Diseases (except HIV/AIDS). 2020. Available online: http://medrxiv.org/lookup/doi/10.1101/2020.04.26.20079244 (accessed on 5 August 2020).
- Streeck, H.; Schulte, B.; Kümmerer, B.M.; Richter, E.; Höller, T.; Fuhrmann, C.; Arnthorsson, A.O.; Helgason, D.; Bjarnadottir, K.; Ingvarsson, R.F.; et al. Infection fatality rate of SARS-CoV2 in a su-per-spreading event in Germany. Nat. Commun. 2020, 11, 1–12. [Google Scholar] [CrossRef]
- Sempos, C.T.; Tian, L. Adjusting Coronavirus Prevalence Estimates for Laboratory Test Kit Error. Am. J. Epidemiol. 2021, 190, 109–115. [Google Scholar] [CrossRef]
- The KoCo19 Collaboration Group; Radon, K.; Saathoff, E.; Pritsch, M.; Noller, J.M.G.; Kroidl, I.; Olbrich, L.; Thiel, V.; Diefenbach, M.; Riess, F.; et al. Protocol of a population-based prospective COVID-19 cohort study Munich, Germany (KoCo19). BMC Public Health 2020, 20, 1–9. [Google Scholar]
- Olbrich, L.; Castelletti, N.; Schälte, Y.; Garí, M.; Pütz, P.; Bakuli, A.; Pritsch, M.; Kroidl, I.; Saathoff, E.; Noller, J.M.G.; et al. A Serology Strategy for Epidemiological Studies Based on the Comparison of the Performance of Seven Different Test Systems—The Representative COVID-19 Cohort Munich. medRxiv 2021. [Google Scholar] [CrossRef]
- Haselmann, V.; Özçürümez, M.K.; Klawonn, F.; Ast, V.; Gerhards, C.; Eichner, R.L.; Costina, V.; Dobler, G.; Geilenkeuser, W.-J.; Wölfel, R.; et al. Results of the first pilot external quality assessment (EQA) scheme for anti-SARS-CoV2-antibody testing. Clin. Chem. Lab. Med. 2020, 58, 2121–2130. [Google Scholar] [CrossRef]
- Robert Koch Institut. Täglicher Lagebericht (13.06.2020) des RKI zur Coronavirus-Krankheit-2019 (COVID-19)—Aktualisierter Stand für Deutschland [Internet]. 2020 [cited 2020 Dec 2]. Available online: https://www.rki.de/DE/Content/InfAZ/N/Neuartiges_Coronavirus/Situationsberichte/2020-06-13-de.pdf;jsessionid=2A2033699409CB8BA030EB0F22EA0BD4.internet062?__blob=publicationFile (accessed on 2 December 2020).
- Molenberghs, G.; Verbeke, G. Models for Discrete Longitudinal Data; Springer: Berlin/Heidelberg, Germany, 2005; p. 683. [Google Scholar]
- Erler, N.S.; Rizopoulos, D.; Lesaffre, E.M.E.H. JointAI: Joint Analysis and Imputation of Incomplete Data in R. arXiv 2020, arXiv:190710867. [Google Scholar]
- Nichols, T.E.; Holmes, A.P. Nonparametric permutation tests for functional neuroimaging: A primer with examples. Hum. Brain Mapp. 2002, 15, 1–25. [Google Scholar] [CrossRef]
- Lee, E.C.; Wada, N.I.; Grabowski, M.K.; Gurley, E.S.; Lessler, J. The engines of SARS-CoV-2 spread. Science 2020, 370, 406–407. [Google Scholar] [CrossRef]
- Snoeck, C.J.; Vaillant, M.; Abdelrahman, T.; Satagopam, V.P.; Turner, J.D.; Beaumont, K.; Gomes, C.P.C.; Fritz, J.V.; Schröder, V.E.; Kaysen, A.; et al. Prevalence of SARS-CoV-2 Infection in the Luxembourgish Population: The CON-VINCE Study. [Internet]. Infectious Diseases (except HIV/AIDS). 2020. Available online: http://medrxiv.org/lookup/doi/10.1101/2020.05.11.20092916 (accessed on 5 August 2020).
- Pollán, M.; Pérez-Gómez, B.; Pastor-Barriuso, R.; Oteo, J.; Hernán, M.A.; Pérez-Olmeda, M.; Eng, J.L.S.M.; Fernández-García, A.; Cruz, I.; de Larrea, N.F.; et al. Prevalence of SARS-CoV-2 in Spain (ENE-COVID): A nationwide, population-based seroepidemiological study. Lancet 2020, 396, 535–544. [Google Scholar] [CrossRef]
- Stringhini, S.; Wisniak, A.; Piumatti, G.; Azman, A.S.; Lauer, S.A.; Baysson, H.; De Ridder, D.; Petrovic, D.; Schrempft, S.; Marcus, K.; et al. Seroprevalence of anti-SARS-CoV-2 IgG antibodies in Geneva, Switzerland (SEROCoV-POP): A population-based study. Lancet 2020, 396, 313–319. [Google Scholar] [CrossRef]
- Hippich, M.; Holthaus, L.; Assfalg, R.; Zapardiel-Gonzalo, J.; Kapfelsperger, H.; Heigermoser, M.; Haupt, F.; Ewald, D.A.; Welzhofer, T.C.; Marcus, B.A.; et al. A Public Health Antibody Screening Indicates a 6-Fold Higher SARS-CoV-2 Exposure Rate than Reported Cases in Children. Med 2021, 2, 149–163. [Google Scholar] [CrossRef] [PubMed]
- Pastor-Barriuso, R.; Perez-Gomez, B.; Hernan, M.A.; Perez-Olmeda, M.; Yotti, R.; Oteo, J.; Sanmartín, J.L.; León-Gómez, I.; Fernández-García, A.; Fernández-Navarro, P.; et al. Infection fatality risk for SARS-CoV-2: A nationwide seroepidemiological study in the non-institutionalized population of Spain [Internet]. Epidemiology 2020. Available online: http://medrxiv.org/lookup/doi/10.1101/2020.08.06.20169722 (accessed on 2 December 2020).
- Perez-Saez, J.; Lauer, S.A.; Kaiser, L.; Regard, S.; Delaporte, E.; Guessous, I.; Stringhini, S.; Azman, A.S.; Serocov-POP Study Group. Serology-informed estimates of SARS-CoV-2 in-fection fatality risk in Geneva, Switzerland. Lancet Infect. Dis. 2021, 21, 69–70. [Google Scholar] [CrossRef]
- McDade, T.W.; McNally, E.M.; Zelikovich, A.S.; D’Aquila, R.; Mustanski, B.; Miller, A.; Vaught, L.A.; Reiser, N.L.; Bogdanovic, E.; Fallon, K.S.; et al. High seroprevalence for SARS-CoV-2 among household members of essential workers detected using a dried blood spot assay. PLoS ONE 2020, 15, e0237833. [Google Scholar] [CrossRef]
- WHO. Keep Health Workers Safe to Keep Patients Safe: WHO [Internet]. 2020. Available online: https://www.who.int/news/item/17-09-2020-keep-health-workers-safe-to-keep-patients-safe-who (accessed on 28 March 2021).
- Yao, Y.; Wang, H.; Liu, Z. Expression of ACE2 in airways: Implication for COVID-19 risk and disease management in patients with chronic inflammatory respiratory diseases. Clin. Exp. Allergy 2020, 50, 1313–1324. [Google Scholar] [CrossRef] [PubMed]
- Ibekwe, T.S.; Fasunla, A.J.; Orimadegun, A.E. Systematic Review and Meta-analysis of Smell and Taste Disorders in COVID-19. OTO Open 2020, 4. [Google Scholar] [CrossRef]
- Tsang, T.K.; Lau, L.L.; Cauchemez, S.; Cowling, B.J. Household Transmission of Influenza Virus. Trends Microbiol. 2016, 24, 123–133. [Google Scholar] [CrossRef]
- Laxminarayan, R.; Wahl, B.; Dudala, S.R.; Gopal, K.; Mohan B., C.; Neelima, S.; Reddy, K.S.J.; Radhakrishnan, J.; Lewnard, J.A. Epidemiology and transmission dynamics of COVID-19 in two Indian states. Science 2020, 370, 691–697. [Google Scholar] [CrossRef] [PubMed]
- Rach, S.; Günther, K.; Hadeler, B. Participants who were difficult to recruit at baseline are less likely to complete a follow-up questionnaire—Results from the German National Cohort. BMC Med. Res. Methodol. 2020, 20, 1–7. [Google Scholar] [CrossRef] [PubMed]
| Characteristics | Munich Population | KoCo19 Study Participants | |||
|---|---|---|---|---|---|
| Individual Characteristics | |||||
| N | 1,561,720 | 5313 | |||
| n | % | nmissing | n | % | |
| Sex | 0 | ||||
| Female | 789,437 | 50.1 | 2766 | 52.1 | |
| Age (years) | 0 | ||||
| 0–19 | 263,053 | 16.8 | 267 | 5.0 | |
| 20–34 | 390,382 | 25.0 | 1346 | 25.3 | |
| 35–49 | 348,651 | 22.3 | 1542 | 29.0 | |
| 50–64 | 291,562 | 18.7 | 1306 | 24.6 | |
| 65–79 | 184,764 | 11.8 | 676 | 12.7 | |
| 80+ | 83,308 | 5.3 | 176 | 3.3 | |
| Country of birth | 465 | ||||
| Outside Germany | 476,575 | 30.5 | 849 | 17.5 | |
| Level of education | NA | NA | 701 | ||
| Still in school | 100 | 2.2 | |||
| <12 y | 1386 | 30.1 | |||
| ≥12 y | 3126 | 67.8 | |||
| Employment status | NA | NA | 576 | ||
| Employed | 2911 | 61.5 | |||
| Self-employed | 471 | 9.9 | |||
| Not working 1 | 1258 | 26.6 | |||
| Others 2 | 97 | 2.0 | |||
| Risk employment 3 | NA | NA | 470 | ||
| Yes | 851 | 17.6 | |||
| Household characteristics | |||||
| N | 2994 | ||||
| Housing type: building with | 148,607 | 100 | 0 | ||
| 1–2 apartments | 82,119 | 55.3 | 661 | 22.1 | |
| 3–4 apartments | 10,938 | 7.4 | 192 | 6.4 | |
| ≥5 apartments | 50,339 | 33.9 | 2137 | 71.4 | |
| Others 4 | 5211 | 3.5 | 4 | 0.1 | |
| Household type | 854,288 | 100 | 307 | ||
| Single | 468,937 | 54.9 | 680 | 25.3 | |
| Couple | 160,339 | 18.8 | 922 | 34.3 | |
| Family | 185,752 | 21.7 | 875 | 32.6 | |
| Others 5 | 39,260 | 4.6 | 210 | 7.8 | |
| No. of household members | 854,288 | 100 | 1 | ||
| 1 | 468,937 | 54·9 | 784 | 26.2 | |
| 2 | 193,376 | 22·6 | 1171 | 39.1 | |
| 3–4 | 106,074 | 12·4 | 880 | 29.4 | |
| 5+ | 85,901 | 10.1 | 158 | 5.3 | |
| Living area per inhabitant | NA | NA | 319 | ||
| ≤30 m2 | 800 | 29.9 | |||
| 30–40 m2 | 634 | 23.7 | |||
| 40–55 m2 | 579 | 21.6 | |||
| >55 m2 | 662 | 24.7 | |||
| Net family income | NA | NA | 924 | ||
| EUR ≤ 2500 | 445 | 21.5 | |||
| EUR 2500–4000 | 502 | 24.3 | |||
| EUR 4000–6000 | 607 | 29.3 | |||
| EUR > 6000+ | 516 | 24.9 | |||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pritsch, M.; Radon, K.; Bakuli, A.; Le Gleut, R.; Olbrich, L.; Guggenbüehl Noller, J.M.; Saathoff, E.; Castelletti, N.; Garí, M.; Pütz, P.; et al. Prevalence and Risk Factors of Infection in the Representative COVID-19 Cohort Munich. Int. J. Environ. Res. Public Health 2021, 18, 3572. https://doi.org/10.3390/ijerph18073572
Pritsch M, Radon K, Bakuli A, Le Gleut R, Olbrich L, Guggenbüehl Noller JM, Saathoff E, Castelletti N, Garí M, Pütz P, et al. Prevalence and Risk Factors of Infection in the Representative COVID-19 Cohort Munich. International Journal of Environmental Research and Public Health. 2021; 18(7):3572. https://doi.org/10.3390/ijerph18073572
Chicago/Turabian StylePritsch, Michael, Katja Radon, Abhishek Bakuli, Ronan Le Gleut, Laura Olbrich, Jessica Michelle Guggenbüehl Noller, Elmar Saathoff, Noemi Castelletti, Mercè Garí, Peter Pütz, and et al. 2021. "Prevalence and Risk Factors of Infection in the Representative COVID-19 Cohort Munich" International Journal of Environmental Research and Public Health 18, no. 7: 3572. https://doi.org/10.3390/ijerph18073572
APA StylePritsch, M., Radon, K., Bakuli, A., Le Gleut, R., Olbrich, L., Guggenbüehl Noller, J. M., Saathoff, E., Castelletti, N., Garí, M., Pütz, P., Schälte, Y., Frahnow, T., Wölfel, R., Rothe, C., Pletschette, M., Metaxa, D., Forster, F., Thiel, V., Rieß, F., ... on behalf of the KoCo19 study group. (2021). Prevalence and Risk Factors of Infection in the Representative COVID-19 Cohort Munich. International Journal of Environmental Research and Public Health, 18(7), 3572. https://doi.org/10.3390/ijerph18073572







