Multicomponent Home-Based Training Program for Chronic Kidney Disease Patients during Movement Restriction
Abstract
1. Introduction
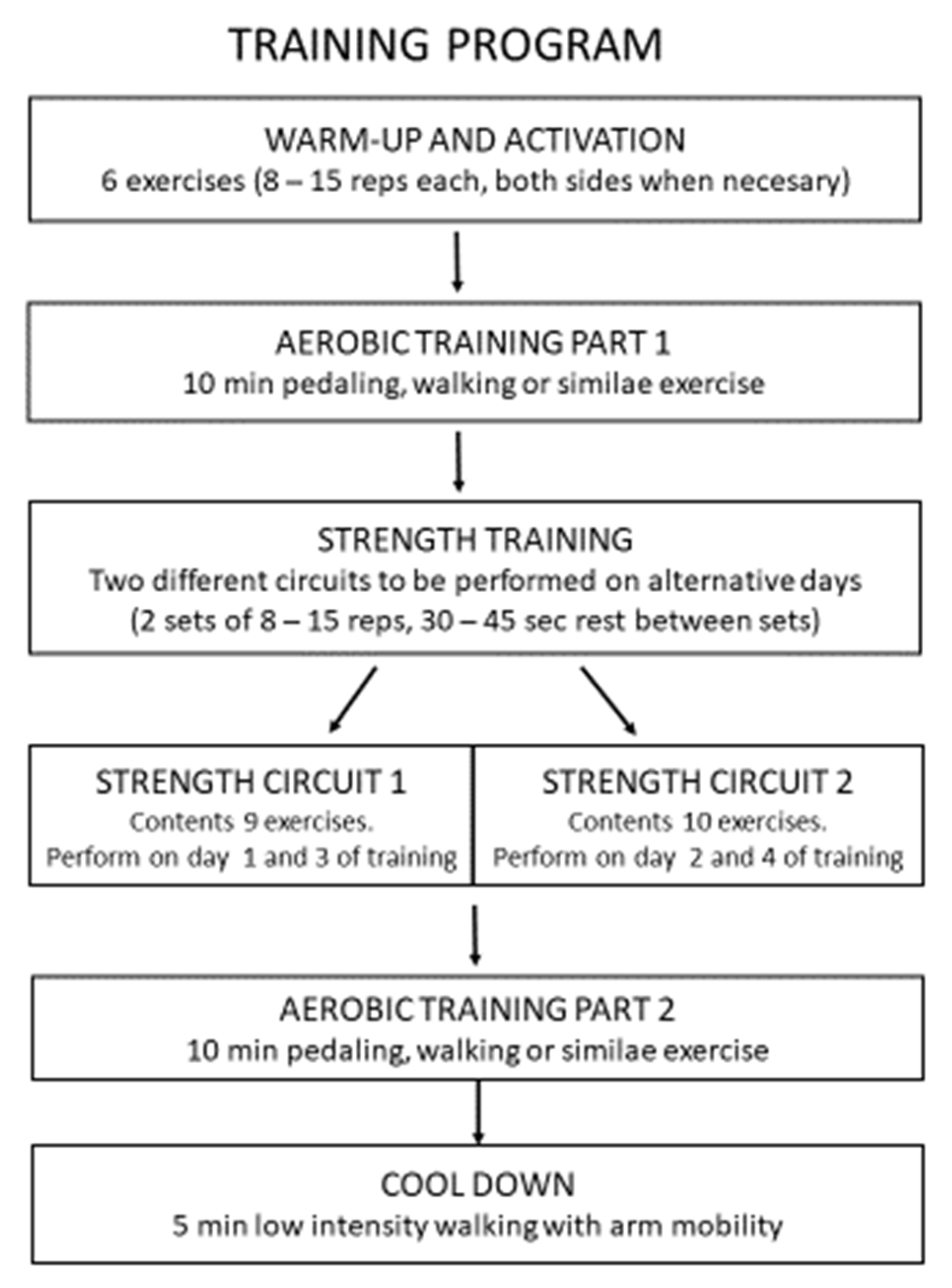
2. Method: Training Proposal
3. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Levin, A.S.; Bilous, R.W.; Coresh, J. Chapter 1: Definition and classification of CKD. Kidney Int. Suppl. 2013, 3, 19–62. [Google Scholar] [CrossRef]
- Wise, M.E.; Lovell, C. Public health surveillance in the dialysis setting: Opportunities and challenges for using electronic health records. Semin. Dial. 2013, 26, 339–406. [Google Scholar] [CrossRef] [PubMed]
- Couser, W.G.; Remuzzi, G.; Mendis, S.; Tonelli, M. The contribution of chronic kidney disease to the global burden of major noncommunicable diseases. Kidney Int. 2011, 80, 1258–1270. [Google Scholar] [CrossRef] [PubMed]
- López Chicharro, J.; López Mojares, L.M. Fisiología Clínica Del Ejercicio; Editorial Médica Panamericana S.A.: Madrid, Spain, 2008; ISBN 9788498351675. [Google Scholar]
- Parker, K. Intradialytic Exercise is Medicine for Hemodialysis Patients. Curr. Sports Med. Rep. 2016, 15, 269–275. [Google Scholar] [CrossRef]
- Tentori, F.; Elder, S.J.; Thumma, J.; Pisoni, R.L.; Bommer, J.; Fissell, R.B.; Fukuhara, S.; Jadoul, M.; Keen, M.L.; Saran, R.; et al. Physical exercise among participants in the Dialysis Outcomes and Practice Patterns Study (DOPPS): Correlates and associated outcomes. Nephrol. Dial. Transplant. 2010, 25, 3050–3062. [Google Scholar] [CrossRef]
- Young, H.M.L.; March, D.S.; Graham-Brown, M.P.M.; Jones, A.W.; Curtis, F.; Grantham, C.S.; Churchward, D.R.; Highton, P.; Smith, A.C.; Singh, S.J.; et al. Effects of intradialytic cycling exercise on exercise capacity, quality of life, physical function and cardiovascular measures in adult haemodialysis patients: A systematic review and meta-analysis. Nephrol. Dial. Transplant. 2018, 33, 1436–1445. [Google Scholar] [CrossRef]
- Cohen, S.D.; Cukor, D.; Kimmel, P.L. Anxiety in patients treated with hemodialysis. Clin. J. Am. Soc. Nephrol. 2016, 11, 2250–2255. [Google Scholar] [CrossRef]
- Koh, K.P.; Fassett, R.G.; Sharman, J.E.; Coombes, J.S.; Williams, A.D. Effect of Intradialytic Versus Home-Based Aerobic Exercise Training on Physical Function and Vascular Parameters in Hemodialysis Patients: A Randomized Pilot Study. Am. J. Kidney Dis. 2010, 55, 88–99. [Google Scholar] [CrossRef] [PubMed]
- Jayaseelan, G.; Bennett, P.N.; Bradshaw, W.; Wang, W.; Rawson, H. Exercise Benefits and Barriers: The Perceptions of People Receiving Hemodialysis. Nephrol. Nurs. J. 2018, 45, 185–219. [Google Scholar]
- Bauman, A.E.; Petersen, C.B.; Blond, K.; Rangul, V.; Hardy, L.L. The Descriptive Epidemiology of Sedentary Behaviour; Leitzmann, M., Jochem, C., Schmid, D., Eds.; Springer International Publishing AG: Cham, Switzerland, 2018; pp. 73–106. [Google Scholar] [CrossRef]
- Johansen, K.L.; Kaysen, G.A.; Young, B.S.; Hung, A.M.; Da Silva, M.; Chertow, G.M. Longitudinal study of nutritional status body composition and physical function hemodialysis patients. Am. J. Clin. Nutr. 2003, 77, 842–846. [Google Scholar] [CrossRef]
- Heiwe, S.; Jacobson, S.H. Exercise training for adults with chronic kidney disease. Cochrane Database Syst. Rev. 2011. [Google Scholar] [CrossRef]
- Manfredini, F.; Mallamaci, F.; D’Arrigo, G.; Baggetta, R.; Bolignano, D.; Torino, C.; Lamberti, N.; Bertoli, S.; Ciurlino, D.; Rocca-Rey, L.; et al. Exercise in patients on dialysis: A multicenter, randomized clinical trial. J. Am. Soc. Nephrol. 2017, 28, 1259–1268. [Google Scholar] [CrossRef] [PubMed]
- Piercy, K.L.; Troiano, R.P.; Ballard, R.M.; Carlson, S.A.; Fulton, J.E.; Galuska, D.A.; George, S.M.; Olson, R.D. The physical activity guidelines for Americans. JAMA J. Am. Med. Assoc. 2018, 320, 2020–2028. [Google Scholar] [CrossRef]
- Wilund, K.R.; Jeong, J.H.; Greenwood, S.A. Addressing myths about exercise in hemodialysis patients. Semin. Dial. 2019, 32, 297–302. [Google Scholar] [CrossRef] [PubMed]
- Smart, N.A.; Williams, A.D.; Levinger, I.; Selig, S.; Howden, E.; Coombes, J.S.; Fassett, R.G. Exercise & Sports Science Australia (ESSA) position statement on exercise and chronic kidney disease. J. Sci. Med. Sport 2013, 16, 406–411. [Google Scholar] [CrossRef] [PubMed]
- Official Journal of the International Society of Nephrology KDIGO 2012 Clinical Practice Guideline for the Evaluation and Management of Chronic Kidney Disease. Available online: www.publicationethics.org (accessed on 2 February 2021).
- WHO Guidelines on Physical Activity and Sedentary Behaviour. Available online: https://www.who.int/publications/i/item/9789240015128 (accessed on 2 February 2021).
- Dong, J.; Sundell, M.B.; Pupim, L.B.; Wu, P.; Shintani, A.; Ikizler, T.A. The Effect of Resistance Exercise to Augment Long-term Benefits of Intradialytic Oral Nutritional Supplementation in Chronic Hemodialysis Patients. J. Ren. Nutr. 2011, 21, 149–159. [Google Scholar] [CrossRef] [PubMed]
- Kopple, J.D.; Wang, H.; Casaburi, R.; Fournier, M.; Lewis, M.I.; Taylor, W.; Storer, T.W. Exercise in Maintenance Hemodialysis Patients Induces Transcriptional Changes in Genes Favoring Anabolic Muscle. J. Am. Soc. Nephrol. 2007, 18, 2975–2986. [Google Scholar] [CrossRef]
- McKenna, C.F.; Salvador, A.F.; Hendriks, F.K.; Harris, A.P.Y.; van Loon, L.J.C.; Burd, N.A. Exercising to offset muscle mass loss in hemodialysis patients: The disconnect between intention and intervention. Semin. Dial. 2019, 32, 379–385. [Google Scholar] [CrossRef]
- Fang, H.Y.; Burrows, B.T.; King, A.C.; Wilund, K.R. A Comparison of Intradialytic versus Out-of-Clinic Exercise Training Programs for Hemodialysis Patients. Blood Purif. 2020, 49, 151–157. [Google Scholar] [CrossRef]
- Delgado, C.; Johansen, K.L. Deficient Counseling on Physical Activity among Nephrologists. Nephron Clin. Pract. 2010, 116, c330–c336. [Google Scholar] [CrossRef]
- Jhamb, M.; McNulty, M.L.; Ingalsbe, G.; Childers, J.W.; Schell, J.; Conroy, M.B.; Forman, D.E.; Hergenroeder, A.; Dew, M.A. Knowledge, barriers and facilitators of exercise in dialysis patients: A qualitative study of patients, staff and nephrologists. BMC Nephrol. 2016, 17, 192. [Google Scholar] [CrossRef]
- Thompson, S.; Tonelli, M.; Klarenbach, S.; Molzahn, A. A qualitative study to explore patient and staff perceptions of intradialytic exercise. Clin. J. Am. Soc. Nephrol. 2016, 11, 1024–1033. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Wilund, K.R.; Viana, J.L.; Perez, L.M. A Critical Review of Exercise Training in Hemodialysis Patients: Personalized Activity Prescriptions Are Needed. Exerc. Sport Sci. Rev. 2020, 48, 28–39. [Google Scholar] [CrossRef] [PubMed]
- Michou, V.; Kouidi, E.; Liakopoulos, V.; Dounousi, E.; Deligiannis, A. Attitudes of hemodialysis patients, medical and nursing staff towards patients’ physical activity. Int. Urol. Nephrol. 2019, 51, 1249–1260. [Google Scholar] [CrossRef]
- Zhao, J.; Qi, Q.; Xu, S.; Shi, D. Combined aerobic resistance exercise improves dialysis adequacy and quality of life in patients on maintenance hemodialysis. Clin. Nephrol. 2020, 93, 275–282. [Google Scholar] [CrossRef] [PubMed]
- Roshanravan, B.; Gamboa, J.; Wilund, K. Exercise and CKD: Skeletal Muscle Dysfunction and Practical Application of Exercise to Prevent and Treat Physical Impairments in CKD. Am. J. Kidney Dis. 2017, 69, 837–852. [Google Scholar] [CrossRef]
- Wang, C.J.; Johansen, K.L. Are dialysis patients too frail to exercise? Semin. Dial. 2019, 32, 291–296. [Google Scholar] [CrossRef] [PubMed]
- Qiu, Z.; Zheng, K.; Zhang, H.; Feng, J.; Wang, L.; Zhou, H. Physical Exercise and Patients with Chronic Renal Failure: A Meta-Analysis. BioMed Res. Int. 2017, 2017, 1–8. [Google Scholar] [CrossRef]
- Clarkson, M.J.; Bennett, P.N.; Fraser, S.F.; Warmington, S.A. Exercise interventions for improving objective physical function in patients with end-stage kidney disease on dialysis: A systematic review and meta-analysis. Am. J. Physiol. Ren. Physiol. 2019, 316, F856–F872. [Google Scholar] [CrossRef]
- Füzéki, E.; Groneberg, D.A.; Banzer, W. Physical activity during COVID-19 induced lockdown: Recommendations. J. Occup. Med. Toxicol. 2020, 15, 25. [Google Scholar] [CrossRef]
- Capitanini, A.; Lange, S.; D’Alessandro, C.; Salotti, E.; Tavolaro, A.; Baronti, M.E.; Giannese, D.; Cupisti, A. Dialysis exercise team: The way to sustain exercise programs in hemodialysis patients. Kidney Blood Press. Res. 2014, 39, 129–133. [Google Scholar] [CrossRef] [PubMed]
- Langston, C. Managing Fluid and Electrolyte Disorders in Kidney Disease. Vet. Clin. N. Am. Small Anim. Pract. 2017, 47, 471–490. [Google Scholar] [CrossRef] [PubMed]
- Cheema, B.; Abas, H.; Smith, B.; O’Sullivan, A.; Chan, M.; Patwardhan, A.; Kelly, J.; Gillin, A.; Pang, G.; Lloyd, B.; et al. Progressive Exercise for Anabolism in Kidney Disease (PEAK): A Randomized, Controlled Trial of Resistance Training during Hemodialysis. J. Am. Soc. Nephrol. 2007, 18, 1594–1601. [Google Scholar] [CrossRef] [PubMed]
- Kopple, J.D.; Cohen, A.H.; Wang, H.; Qing, D.; Tang, Z.; Fournier, M.; Lewis, M.; Casaburi, R.; Storer, T. Effect of Exercise on mRNA Levels for Growth Factors in Skeletal Muscle of Hemodialysis Patients. J. Ren. Nutr. 2006, 16, 312–324. [Google Scholar] [CrossRef]
- Guadalupe-Grau, A.; López-Torres, O.; Martos-Bermúdez, Á.; González-Gross, M. Home-based training strategy to maintain muscle function in older adults with diabetes during COVID-19 confinement. J. Diabetes 2020, 12, 701–702. [Google Scholar] [CrossRef]
- Rikli, R.E. Reliability, validity, and methodological issues in assessing physical activity in older adults. Res. Q. Exerc. Sport 2000, 71, 89–96. [Google Scholar] [CrossRef] [PubMed]
- Jones, C.J.; Rikli, R.E.; Beam, W.C. A 30-s chair-stand test as a measure of lower body strength in community-residing older adults. Res. Q. Exerc. Sport 1999, 70, 113–119. [Google Scholar] [CrossRef]
| Physical Tests | Sex | Age Group 65–69 | Age Group 70–74 | Age Group 75–79 | Age Group 80–84 | Age Group 85–89 | Age Group 90–94 | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Low | Average | High | Low | Average | High | Low | Average | High | Low | Average | High | Low | Average | High | Low | Average | High | ||
| UGT (sec) | M | ≥6.0 | 5.9–4.3 | ≤4.2 | ≥6.3 | 6.2–4.4 | ≤4.3 | ≥7.3 | 7.2–4.6 | ≤4.5 | ≥7.7 | 7.6–5.2 | ≤5.1 | ≥9 | 8.9–5.5 | ≤5.4 | ≥10.1 | 10–6.2 | ≤6.1 |
| W | ≥6.5 | 6.4–4.8 | ≤4.7 | ≥7.2 | 7.1–4.9 | ≤4.8 | ≥7.5 | 7.4–5.2 | ≤5.1 | ≥8.8 | 8.7–5.7 | ≤5.6 | ≥9.7 | 9.6–6.2 | ≤6.1 | ≥11.6 | 11.5–7.3 | ≤7.2 | |
| STST (rep) | M | ≤11 | 12–18 | ≥19 | ≤11 | 12–17 | ≥18 | ≤10 | 11–17 | ≥18 | ≤14 | 10–15 | ≥16 | ≤7 | 8–14 | ≥15 | ≤6 | 7–12 | ≥13 |
| W | ≤10 | 11–16 | ≥17 | ≤9 | 10–15 | ≥16 | ≤9 | 10–15 | ≥16 | ≤13 | 9–14 | ≥15 | ≤7 | 8–13 | ≥14 | ≤3 | 4–11 | ≥10 | |
| 6MWT (m) | M | ≤559 | 560–700 | ≥701 | ≤544 | 545–680 | ≥681 | ≤469 | 470–640 | ≥641 | ≤444 | 445–605 | ≥606 | ≤379 | 380–570 | ≥571 | ≤304 | 305–500 | ≥501 |
| W | ≤499 | 500–635 | ≥636 | ≤479 | 480–615 | ≥616 | ≤434 | 435–585 | ≥586 | ≤384 | 385–540 | ≥541 | ≤339 | 340–510 | ≥511 | ≤274 | 275–440 | ≥441 | |
| Health Markers | Sex | Age Group | Risk Level | |||
|---|---|---|---|---|---|---|
| Low | Medium | High | Very High | |||
| BMI | ≤18.49 | 18.50–24.99 | 25–34.99 | ≥35.00 | ||
| WHR | Men | ≤0.90 | 0.91–0.93 | 0.94–1.03 | ≥1.04 | |
| Women | ≤0.76 | 0.77–0.84 | 0.85–0.9 | ≥1.00 | ||
| WC (cm) | Men | ≤94 | 94.1–102 | ≥102.1 | ||
| Women | ≤80 | 80.1–88 | ≥88.1 | |||
| WHtR | Men | ≤0.49 | 0.50–0.56 | 0.57–0.63 | ≥0.63 | |
| Women | ≤0.50 | 0.51–0.57 | 0.58–0.65 | ≥0.65 | ||
| HGST (kg) | Men | AG 60–69 | ≥40.9 | 37.1–40.8 | 26.6–37 | ≤26.5 |
| AG > 70 | ≥35.8 | 32.2–35.7 | 22.9–32.1 | ≤22.8 | ||
| Women | AG 60–69 | ≥24.7 | 21.8–24.6 | 16.7–21.7 | ≤16.6 | |
| AG > 70 | ≥20.1 | 16.9–20 | 10–16.8 | ≤9.9 | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
López-Torres, O.; Azpeitia-Martínez, C.; González-Gross, M.; Reihmane, D.; Guadalupe-Grau, A. Multicomponent Home-Based Training Program for Chronic Kidney Disease Patients during Movement Restriction. Int. J. Environ. Res. Public Health 2021, 18, 3416. https://doi.org/10.3390/ijerph18073416
López-Torres O, Azpeitia-Martínez C, González-Gross M, Reihmane D, Guadalupe-Grau A. Multicomponent Home-Based Training Program for Chronic Kidney Disease Patients during Movement Restriction. International Journal of Environmental Research and Public Health. 2021; 18(7):3416. https://doi.org/10.3390/ijerph18073416
Chicago/Turabian StyleLópez-Torres, Olga, Celia Azpeitia-Martínez, Marcela González-Gross, Dace Reihmane, and Amelia Guadalupe-Grau. 2021. "Multicomponent Home-Based Training Program for Chronic Kidney Disease Patients during Movement Restriction" International Journal of Environmental Research and Public Health 18, no. 7: 3416. https://doi.org/10.3390/ijerph18073416
APA StyleLópez-Torres, O., Azpeitia-Martínez, C., González-Gross, M., Reihmane, D., & Guadalupe-Grau, A. (2021). Multicomponent Home-Based Training Program for Chronic Kidney Disease Patients during Movement Restriction. International Journal of Environmental Research and Public Health, 18(7), 3416. https://doi.org/10.3390/ijerph18073416








