Experimental Evaluation of Aerosol Production after Dental Ultrasonic Instrumentation: An Analysis on Fine Particulate Matter Perturbation
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Design and Study Setting
2.2. Study Testing
2.3. Particle Count
2.4. Sample Size Estimation
2.5. Data Analysis
3. Results
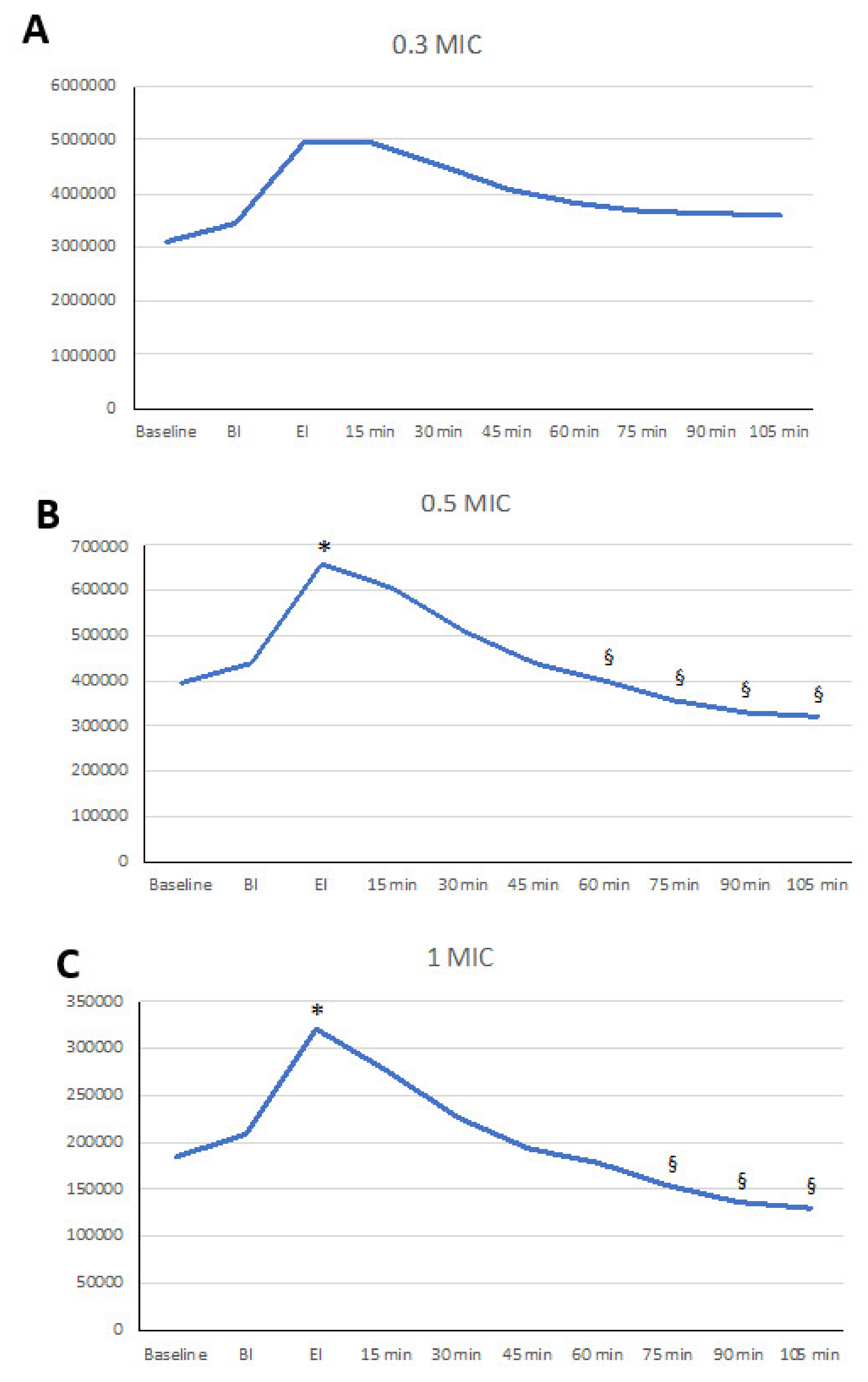
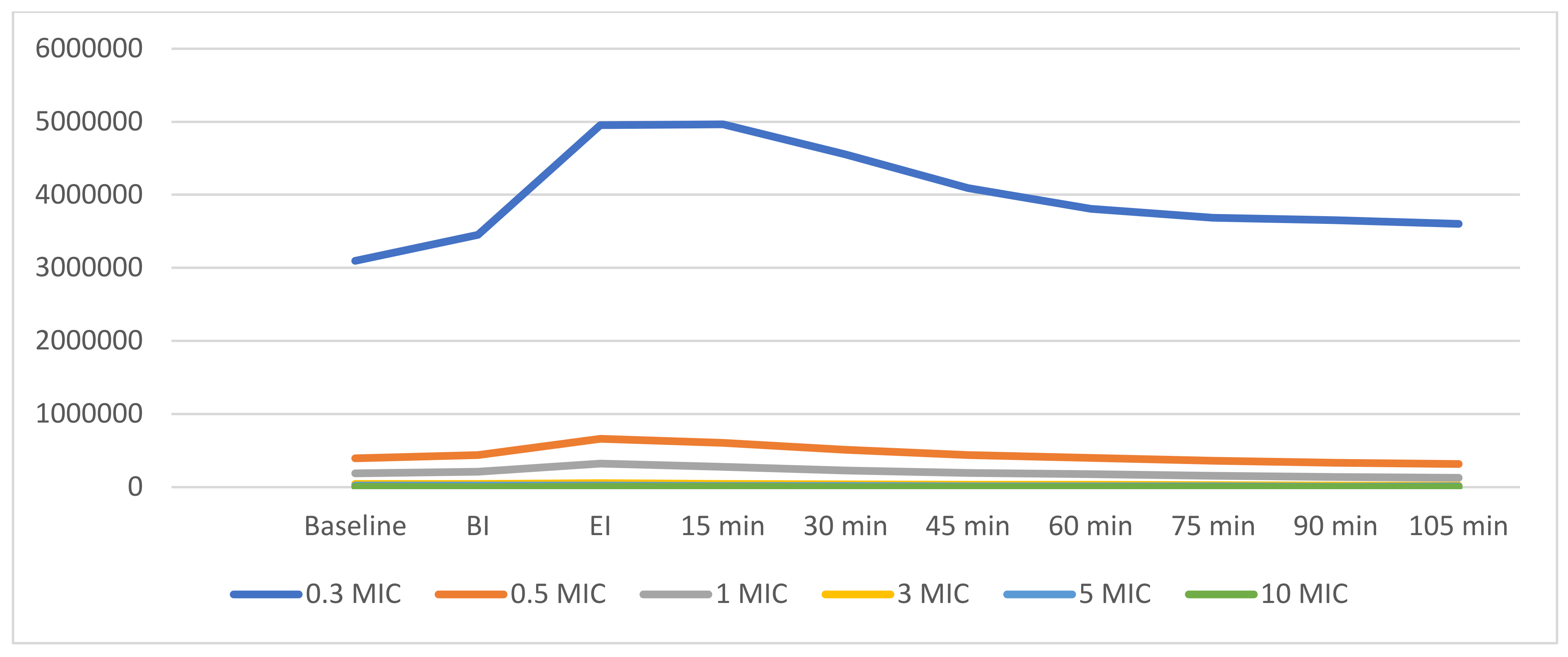
3.1. Submicron and Micro Aerosol
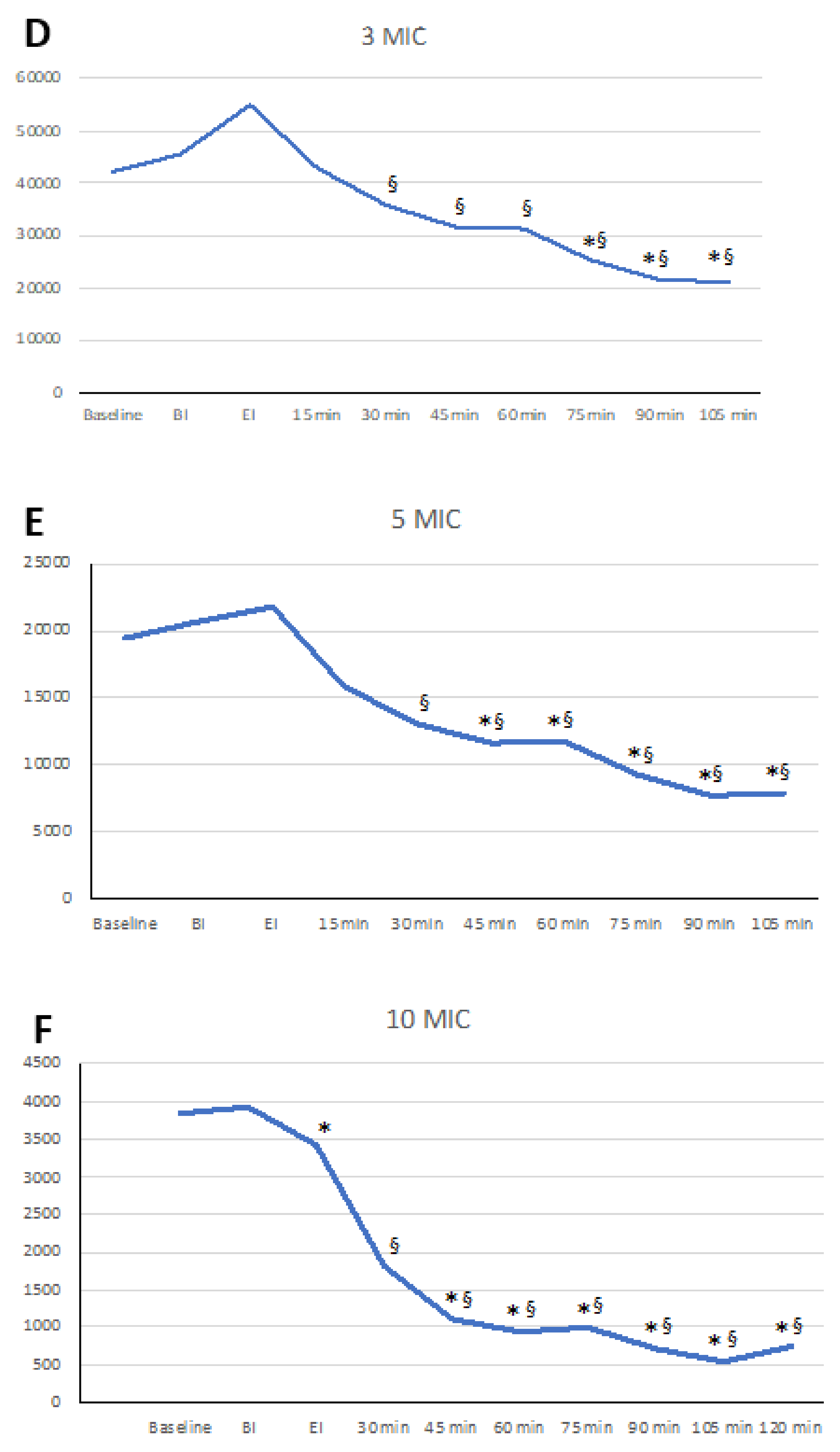
3.2. Aerosol Particles >3 µm
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Timmerman, M.F.; Menso, L.; Steinfort, J.; van Winkelhoff, A.J.; van der Weijden, G.A. Atmospheric contamination during ultra-sonic scaling. J. Clin. Periodontol. 2004, 31, 458–462. [Google Scholar] [CrossRef] [PubMed]
- Ireland, A.J.; Moreno, T.; Price, R. Airborne particles produced during enamel cleanup after removal of orthodontic appliances. Am. J. Orthod. Dentofac. Orthop. 2003, 124, 683–686. [Google Scholar] [CrossRef]
- Geisinger, M.; Iaonnidou, E. Up in the Air? Future Research Strategies to Assess Aerosols in Dentistry. JDR Clin. Transl. Res. 2021, 6, 128–131. [Google Scholar] [CrossRef] [PubMed]
- Brankston, G.; Gitterman, L.; Hirji, Z.; Lemieux, C.; Gardam, M. Transmission of influenza A in human beings. Lancet Infect. Dis. 2007, 7, 257–265. [Google Scholar] [CrossRef]
- Tellier, R. Aerosol transmission of influenza A virus: A review of new studies. J. R. Soc. Interface 2009, 6, S783–S790. [Google Scholar] [CrossRef]
- Schlosser, O. Bioaerosols and health: Current knowledge and gaps in the field of waste management. Detritus 2018, 5, 111–125. [Google Scholar] [CrossRef]
- Tang, J.W.; Wilson, P.; Shetty, N.; Noakes, C.J. Aerosol-Transmitted Infections-a New Consideration for Public Health and Infec-tion Control Teams. Curr. Treat Options Infect. Dis. 2015, 7, 176–201. [Google Scholar] [CrossRef]
- Graziani, F.; Izzetti, R.; Lardani, L.; Biancarini, M.L.; Gabriele, M. Dental Practice in the Era of Sars-Cov-2 Pandemic: A Checklist to Enhance Safety and Good Practice, 1st ed.; Edizioni Martina: Bologna, Italy, 2020. [Google Scholar]
- Gralton, J.; Tovey, E.; McLaws, M.-L.; Rawlinson, W.D. The role of particle size in aerosolised pathogen transmission: A review. J. Infect. 2011, 62, 1–13. [Google Scholar] [CrossRef]
- Seto, W.; Conly, J.; Silva, C.P.; Malik, M.; Eremin, S. Infection prevention and control measures for acute respiratory infections in healthcare settings: An update. East. Mediterr. Health J. 2013, 19, S39–S47. [Google Scholar] [CrossRef] [PubMed]
- Izzetti, R.; Nisi, M.; Gabriele, M.; Graziani, F. COVID-19 Transmission in Dental Practice: Brief Review of Preventive Measures in Italy. J. Dent. Res. 2020, 99, 1030–1038. [Google Scholar] [CrossRef]
- Pankhurst, C.L.; Coulter, W.A. Do contaminated dental unit waterlines pose a risk of infection? J. Dent. 2007, 35, 712–720. [Google Scholar] [CrossRef]
- Hallier, C.; Williams, D.W.; Potts, A.J.C.; Lewis, M.A.O. A pilot study of bioaerosol reduction using an air cleaning system during dental procedures. Br. Dent. J. 2010, 209, E14. [Google Scholar] [CrossRef]
- Laheij, A.; Kistler, J.; Belibasakis, G.; Välimaa, H.; De Soet, J. Healthcare-associated viral and bacterial infections in dentistry. J. Oral Microbiol. 2012, 4. [Google Scholar] [CrossRef]
- Volgenant, C.M.C.; Persoon, I.F.; de Ruijter, R.A.G.; de Soet, J.J. Infection control in dental health care during and after the SARS-CoV-2 outbreak. Oral Dis. 2020, 11. [Google Scholar] [CrossRef]
- Harrel, S.K.; Molinari, J. Aerosols and splatter in dentistry: A brief review of the literature and infection control implications. J. Am. Dent. Assoc. 2004, 135, 429–437. [Google Scholar] [CrossRef]
- Zemouri, C.; De Soet, H.; Crielaard, W.; Laheij, A. A scoping review on bio-aerosols in healthcare and the dental environment. PLoS ONE 2017, 12, e0178007. [Google Scholar] [CrossRef]
- Pedrosa, P.B.; Cardoso, T.A. Viral infections in workers in hospital and research laboratory settings: A comparative review of infection modes and respective biosafety aspects. Int. J. Infect. Dis. 2011, 15, e366–e376. [Google Scholar] [CrossRef] [PubMed]
- Bennett, A.M.; Fulford, M.R.; Walker, J.T.; Bradshaw, D.J.; Martin, M.V.; Marsh, P.D. Microbial aerosols in general dental practice. Br. Dent. J. 2000, 189, 664–667. [Google Scholar] [CrossRef] [PubMed]
- Leggat, P.A.; Kedjarune, U. Bacterial aerosols in the dental clinic: A review. Int. Dent. J. 2001, 51, 39–44. [Google Scholar] [CrossRef] [PubMed]
- Greco, P.M.; Lai, C.-H. A new method of assessing aerosolized bacteria generated during orthodontic debonding procedures. Am. J. Orthod. Dentofac. Orthop. 2008, 133, S79–S87. [Google Scholar] [CrossRef] [PubMed]
- Aluko, O.; Noll, K.E. Deposition and Suspension of Large, Airborne Particles. Aerosol Sci. Technol. 2006, 40, 503–513. [Google Scholar] [CrossRef]
- Nikitin, N.; Petrova, E.; Trifonova, E.; Karpova, O. Influenza Virus Aerosols in the Air and Their Infectiousness. Adv. Virol. 2014, 2014, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Dutil, S.; Mériaux, A.; De Latrémoille, M.-C.; Lazure, L.; Barbeau, J.; Duchaine, C. Measurement of Airborne Bacteria and Endotoxin Generated During Dental Cleaning. J. Occup. Environ. Hyg. 2008, 6, 121–130. [Google Scholar] [CrossRef] [PubMed]
- Baron, P.A.; Willeke, K. Aerosol Measurement Principles, Techniques and Applications, 2nd ed.; John Wiley & Sons, INC.: New York, NY, USA, 2001. [Google Scholar]
- Wang, C. Interface Science and Technology; Chapter 4 Behavior of Aerosol Particles; Elsevier Academic Press: Amsterdam, The Netherlands, 2005; Volume 5, pp. 55–78. [Google Scholar]
- Liu, Y.; Ning, Z.; Chen, Y.; Guo, M.; Liu, Y.; Gali, N.K.; Sun, L.; Duan, Y.; Cai, J.; Westerdahl, D.; et al. Aerodynamic analysis of SARS-CoV-2 in two Wuhan hospitals. Nature 2020, 582, 557–560. [Google Scholar] [CrossRef] [PubMed]
- Cagetti, M.G.; Cairoli, J.L.; Senna, A.; Guglielmo Campus. COVID-19 Outbreak in North Italy: An Overview on Dentistry. A Questionnaire Survey. Int. J. Environ. Res. Public Health 2020, 17, 3835. [Google Scholar] [CrossRef] [PubMed]
- MacIntyre, C.R.; Seale, H.; Yang, P.; Zhang, Y.; Shi, W.; Almatroudi, A.; Moa, A.; Wang, X.; Li, X.; Pang, X.; et al. Quantifying the risk of respiratory infection in healthcare workers performing high-risk procedures. Epidemiol. Infect. 2014, 142, 1802–1808. [Google Scholar] [CrossRef] [PubMed]
- Mirhoseini, S.H.; Nikaeen, M.; Khanahmd, H.; Hatamzadeh, M.; Hassanzadeh, A. Monitoring of airborne bacteria and aerosols in different wards of hospitals—Particle counting usefulness in investigation of airborne bacteria. Ann. Agric. Environ. Med. 2015, 22, 670–673. [Google Scholar] [CrossRef]
- Judson, S.D.; Munster, V.J. Nosocomial Transmission of Emerging Viruses via Aerosol-Generating Medical Procedures. Viruses 2019, 11, 940. [Google Scholar] [CrossRef]
- Cincinelli, A.; Martellini, T. Indoor Air Quality and Health. Int. J. Environ. Res. Public Health 2017, 14, 1286. [Google Scholar] [CrossRef]
- Oliver, L.C.; Shackleton, B.W. The indoor air we breathe. Public Health Rep. 1998, 113, 398–409. [Google Scholar]
- Van Doremalen, N.; Bushmaker, T.; Morris, D.H.; Holbrook, M.G.; Gamble, A.; Williamson, B.N.; Tamin, A.; Harcourt, J.L.; Thornburg, N.J.; Gerber, S.I.; et al. Aerosol and Surface Stability of SARS-CoV-2 as Compared with SARS-CoV-1. N. Engl. J. Med. 2020, 382, 1564–1567. [Google Scholar] [CrossRef] [PubMed]
| 0.3 μm | 0.5 μm | 1 μm | ||||
|---|---|---|---|---|---|---|
| Mean | Std. Deviation | Mean | Std. Deviation | Mean | Std. Deviation | |
| BASELINE | 3,099,087 | 1,874,625 | 395,197 | 198,491 | 185,647 | 98,743 |
| BI | 3,453,958 | 2,133,727 | 439,378 | 217,760 | 209,509 * | 104,464 |
| EI | 4,952,485 | 3,012,991 | 660,629 *§ | 360,456 | 321,844 *§ | 189,350 |
| 15 min | 4,968,704 | 2,720,239 | 607,357 * | 280,691 | 276,460 | 131,833 |
| 30 min | 4,551,001 | 2,439,808 | 508,893 | 217,209 | 226,759 | 102,712 |
| 45 min | 4,092,890 | 2,018,861 | 439,127 | 151,974 | 194,071 | 68,837 |
| 60 min | 3,807,691 | 1,799,915 | 400,761 # | 113,393 | 178,676 # | 58,406 |
| 75 min | 3,683,709 | 1,792,499 | 357,687 °# | 94,795 | 153,199 ##° | 44,556 |
| 90 min | 3,649,827 | 1,703,106 | 330,118 ##°° | 86,997 | 135,673 ##° | 38,993 |
| 105 min | 3,604,367 | 1,587,125 | 320,151 ##°° | 74,592 | 129,292 ##°° | 33,588 |
| 3 μm | 5 μm | 10 μm | ||||
|---|---|---|---|---|---|---|
| Mean | Std. Deviation | Mean | Std. Deviation | Mean | Std. Deviation | |
| BASELINE | 42,153 | 24,121 | 19,433 | 10,202 | 3839 | 1939 |
| BI | 45,760 | 24,418 | 20,775 | 11,565 | 3928 | 2651 |
| EI | 54,873 | 16,593 | 21,806 | 6069 | 3423 * | 1586 |
| 15 min | 43,024 | 12,333 | 15,935 | 4790 | 1821 ***§# | 979 |
| 30 min | 35,822 # | 12,094 | 13,039 # | 4136 | 1110 ***§§§### | 408 |
| 45 min | 31,508 ## | 10,067 | 11,593 §# | 4131 | 930 ***§§§###°° | 345 |
| 60 min | 31,238 ## | 15,181 | 11,732 *§§## | 6448 | 1002 ***§§§###°° | 580 |
| 75 min | 25,409 §##° | 10,438 | 9341 **§§§###°° | 4365 | 717 ***§§§###°°° | 423 |
| 90 min | 21,408 §§**###°° | 7708 | 7698 ***§§§###°° | 3068 | 546 ***§§§###°° | 272 |
| 105 min | 21,011 §§**###°° | 7359 | 7846 ***§§§###°°° | 3321 | 726 ***§§§###°°° | 450 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Graziani, F.; Izzetti, R.; Lardani, L.; Totaro, M.; Baggiani, A. Experimental Evaluation of Aerosol Production after Dental Ultrasonic Instrumentation: An Analysis on Fine Particulate Matter Perturbation. Int. J. Environ. Res. Public Health 2021, 18, 3357. https://doi.org/10.3390/ijerph18073357
Graziani F, Izzetti R, Lardani L, Totaro M, Baggiani A. Experimental Evaluation of Aerosol Production after Dental Ultrasonic Instrumentation: An Analysis on Fine Particulate Matter Perturbation. International Journal of Environmental Research and Public Health. 2021; 18(7):3357. https://doi.org/10.3390/ijerph18073357
Chicago/Turabian StyleGraziani, Filippo, Rossana Izzetti, Lisa Lardani, Michele Totaro, and Angelo Baggiani. 2021. "Experimental Evaluation of Aerosol Production after Dental Ultrasonic Instrumentation: An Analysis on Fine Particulate Matter Perturbation" International Journal of Environmental Research and Public Health 18, no. 7: 3357. https://doi.org/10.3390/ijerph18073357
APA StyleGraziani, F., Izzetti, R., Lardani, L., Totaro, M., & Baggiani, A. (2021). Experimental Evaluation of Aerosol Production after Dental Ultrasonic Instrumentation: An Analysis on Fine Particulate Matter Perturbation. International Journal of Environmental Research and Public Health, 18(7), 3357. https://doi.org/10.3390/ijerph18073357






