Isotherm, Kinetics and Thermodynamics of Cu(II) and Pb(II) Adsorption on Groundwater Treatment Sludge-Derived Manganese Dioxide for Wastewater Treatment Applications
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals and Materials
2.2. Mn Leaching and MnO2 Synthesis
2.3. Adsorption Experiments
2.4. Adsorbent Characterization
3. Results and Discussion
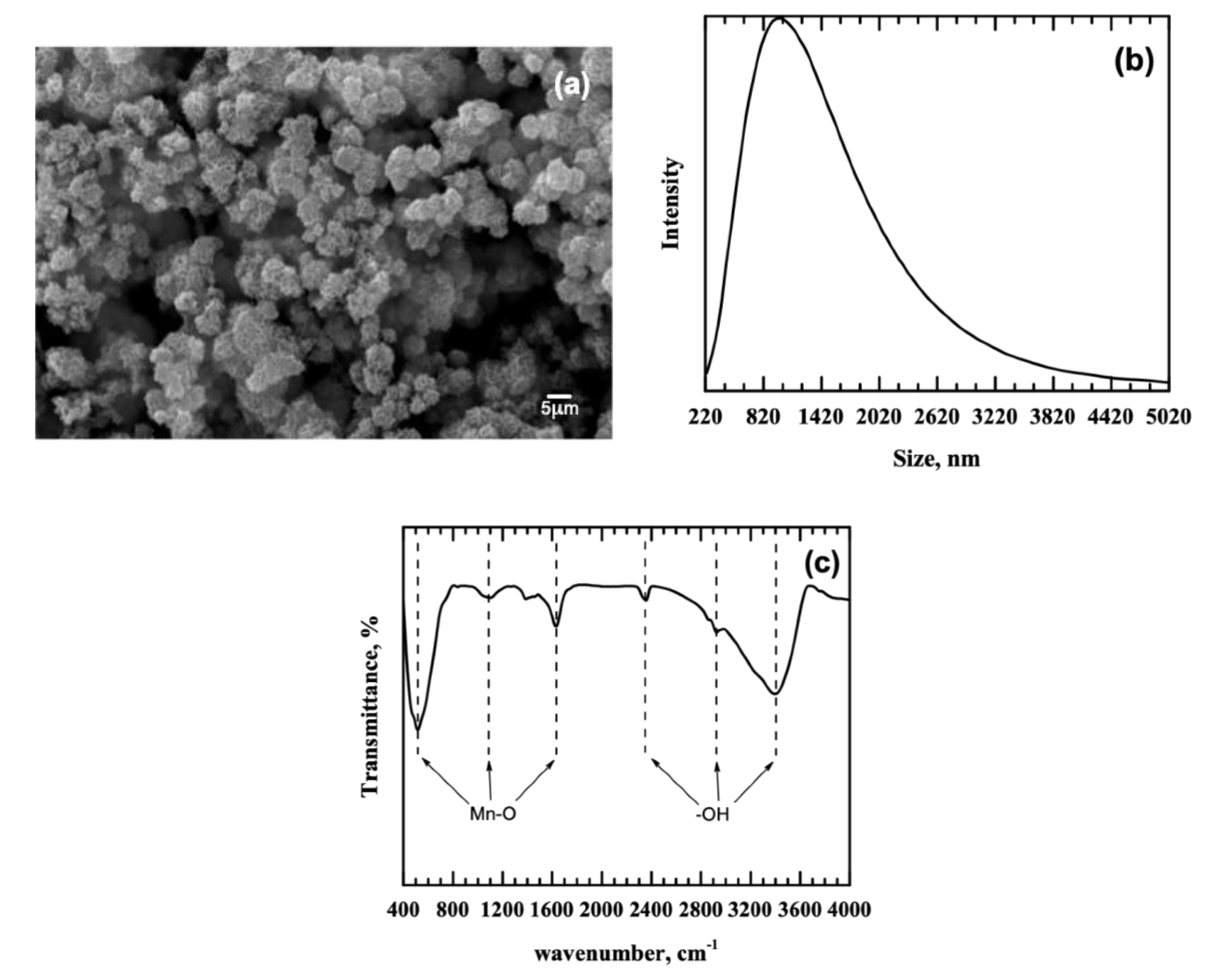
3.1. Adsorbent Properties
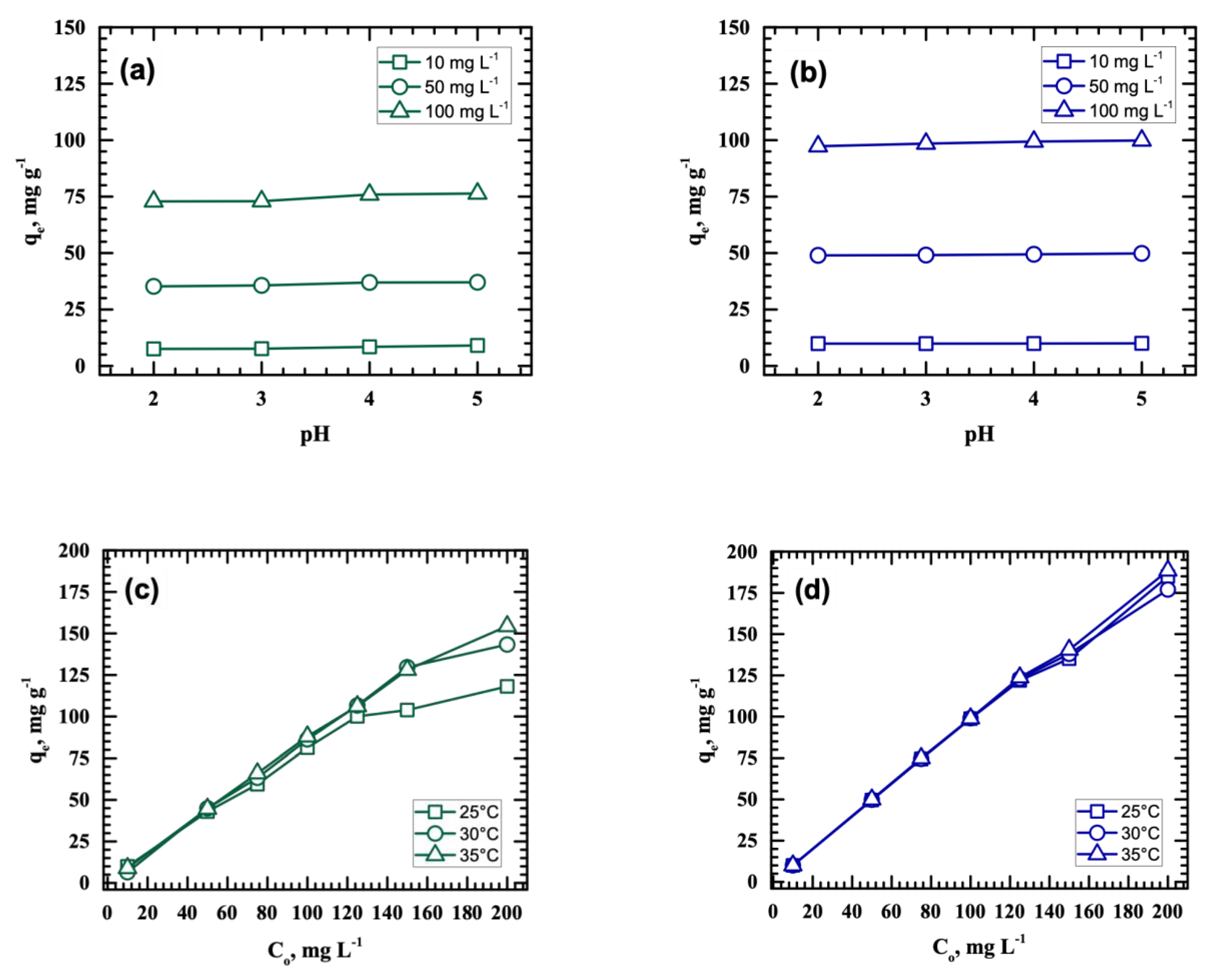
3.2. Effects of pH, Initial Metal Concentration, and Temperature
3.3. Isotherm Studies
3.4. Kinetic Studies
3.5. Thermodynamic Studies
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- de Luna, M.D.G.; Paulino, L.H.S.; Futalan, C.M.; Lu, M.C. Recovery of Zinc Granules from Synthetic Electroplating Wastewater Using Fluidized-Bed Homogeneous Crystallization Process. Int. J. Environ. Sci. Technol. 2020, 17, 129–142. [Google Scholar] [CrossRef]
- Tsai, W.-C.; de Luna, M.D.G.; Bermillo-Arriesgado, H.L.P.; Futalan, C.M.; Colades, J.I.; Wan, M.-W. Competitive Fixed-Bed Adsorption of Pb(II), Cu(II), and Ni(II) from Aqueous Solution Using Chitosan-Coated Bentonite. Int. J. Polym. Sci. 2016, 1–11. [Google Scholar] [CrossRef]
- Arida, C.V.J.; de Luna, M.D.G.; Futalan, C.M.; Wan, M.-W. Optimization of As(V) Removal Using Chitosan-Coated Bentonite from Groundwater Using Box–Behnken Design: Effects of Adsorbent Mass, Flow Rate, and Initial Concentration. Desalination Water Treat. 2016, 57, 18739–18747. [Google Scholar] [CrossRef]
- de Luna, M.D.G.; Capito, J.A.; Vilando, A.C.; Lu, M.-C. Effect of EDTA and CH2O on Copper Recovery from Simulated Electroless Copper Plating Spent Rinse Water by Unseeded Fluidized-Bed Granulation Process. Sep. Purif. Technol. 2020, 253, 117460. [Google Scholar] [CrossRef]
- O’Neal, S.L.; Zheng, W. Manganese Toxicity Upon Overexposure: A Decade in Review. Curr. Environ. Health Rep. 2015, 2, 315–328. [Google Scholar] [CrossRef] [PubMed]
- Pohanka, M. Copper and Copper Nanoparticles Toxicity and Their Impact on Basic Functions in the Body. Bratisl. Med. J. 2019, 120, 397–409. [Google Scholar] [CrossRef]
- de Luna, M.D.G.; Bellotindos, L.M.; Asiao, R.N.; Lu, M.-C. Removal and Recovery of Lead in a Fluidized-Bed Reactor by Crystallization Process. Hydrometallurgy 2015, 155, 6–12. [Google Scholar] [CrossRef]
- Yee, J.-J.; Arida, C.V.J.; Futalan, C.M.; de Luna, M.D.G.; Wan, M.-W. Treatment of Contaminated Groundwater via Arsenate Removal Using Chitosan-Coated Bentonite. Molecules 2019, 24, 2464. [Google Scholar] [CrossRef] [PubMed]
- Mitra, P.; Sharma, S.; Purohit, P.; Sharma, P. Clinical and Molecular Aspects of Lead Toxicity: An Update. Crit. Rev. Clin. Lab. Sci. 2017, 54, 506–528. [Google Scholar] [CrossRef]
- Kan, C.-C.; Sumalinog, M.J.R.; Rivera, K.K.P.; Arazo, R.O.; de Luna, M.D.G. Ultrasound-Assisted Synthesis of Adsorbents from Groundwater Treatment Residuals for Hexavalent Chromium Removal from Aqueous Solutions. Groundw. Sustain. Dev. 2017, 5, 253–260. [Google Scholar] [CrossRef]
- Ong, D.C.; Kan, C.-C.; Pingul-Ong, S.M.B.; de Luna, M.D.G. Utilization of Groundwater Treatment Plant (GWTP) Sludge for Nickel Removal from Aqueous Solutions: Isotherm and Kinetic Studies. J. Environ. Chem. Eng. 2017, 5, 5746–5753. [Google Scholar] [CrossRef]
- Nayl, A.A.; Ismail, I.M.; Aly, H.F. Recovery of Pure MnSO4∙H2O by Reductive Leaching of Manganese from Pyrolusite Ore by Sulfuric Acid and Hydrogen Peroxide. Int. J. Miner. Process. 2011, 100, 116–123. [Google Scholar] [CrossRef]
- Li, Q.; Rao, X.; Xu, B.; Yang, Y.; Liu, T.; Hu, L. Extraction of Manganese and Zinc from Their Compound Ore by Reductive Acid Leaching. Trans. Nonferrous Met. Soc. China 2017, 27, 1172–1179. [Google Scholar] [CrossRef]
- Ong, D.C.; de Luna, M.D.G.; Pingul-Ong, S.M.B.; Kan, C.-C. Manganese and Iron Recovery from Groundwater Treatment Sludge by Reductive Acid Leaching and Hydroxide Precipitation. J. Environ. Manag. 2018, 223, 723–730. [Google Scholar] [CrossRef] [PubMed]
- Ong, D.C.; Pingul-Ong, S.M.B.; Kan, C.C.; de Luna, M.D.G. Removal of Nickel Ions from Aqueous Solutions by Manganese Dioxide Derived from Groundwater Treatment Sludge. J. Clean. Prod. 2018, 190, 443–451. [Google Scholar] [CrossRef]
- Ong, D.C.; Tumampos, S.B.; Kan, C.-C.; Pingul-Ong, S.M.B.; Ensano, B.M.B.; de Luna, M.D.G. Optimization of Manganese Recovery from Groundwater Treatment Sludge for the Production of Highly-Efficient Cu(II) and Pb(II) Adsorbents. J. Environ. Chem. Eng. 2021, 9, 104705. [Google Scholar] [CrossRef]
- Pang, S.C.; Chin, S.F.; Ling, C.Y. Controlled Synthesis of Manganese Dioxide Nanostructures via a Facile Hydrothermal Route. J. Nanomater. 2012, 2012. [Google Scholar] [CrossRef]
- Kan, C.-C.; Ibe, A.H.; Rivera, K.K.P.; Arazo, R.O.; de Luna, M.D.G. Hexavalent Chromium Removal from Aqueous Solution by Adsorbents Synthesized from Groundwater Treatment Residuals. Sustain. Environ. Res. 2017, 27, 163–171. [Google Scholar] [CrossRef]
- Huang, Y.; Wu, H.; Shao, T.; Zhao, X.; Peng, H.; Gong, Y.; Wan, H. Enhanced Copper Adsorption by DTPA-Chitosan/Alginate Composite Beads: Mechanism and Application in Simulated Electroplating Wastewater. Chem. Eng. J. 2018, 339, 322–333. [Google Scholar] [CrossRef]
- Wang, Y.; Chai, L.; Chang, H.; Peng, X.; Shu, Y. Equilibrium of Hydroxyl Complex Ions in Pb2+-H2O System. Trans. Nonferrous Met. Soc. China 2009, 19, 458–462. [Google Scholar] [CrossRef]
- Muzenda, E.; Kabuba, J.; Ntuli, F.; Mollagee, M.; Mulaba-Bafubiandi, A.F. Cu (II) Removal from Synthetic Waste Water by Ion Exchange Process. In Proceedings of the World Congress on Engineering and Computer Science, San Francisco, CA, USA, 19–21 October 2011; Volume 2. [Google Scholar]
- Langmuir, I. The Adsorption of Gases on Plane Surfaces of Glass, Mica and Platinum. J. Am. Chem. Soc. 1918, 40, 1361–1403. [Google Scholar] [CrossRef]
- Freundlich, H. Über Die Adsorption in Lösungen. Z. Phys. Chem. 1906, 57, 385–470. [Google Scholar] [CrossRef]
- Temkin, M.J.; Pyzhev, V. Recent Modifications to Langmuir Isotherms. Acta Physicochim. URSS 1940, 12, 217–222. [Google Scholar]
- Dubinin, M.; Radushkevich, L. The Equation of the Characteristic Curve Ofactivated Charcoal. Proc. Acad. Sci. USSR 1947, 55, 331–333. [Google Scholar]
- Manahan, S.E. Environmental Chemistry; CRC Press: Boca Raton, FL, USA, 2017; ISBN 978-1-4987-7694-3. [Google Scholar]
- Demirkiran, N. Copper Adsorption by Natural Manganese Dioxide. Trans. Nonferrous Met. Soc. China 2015, 25, 647–653. [Google Scholar] [CrossRef]
- Sewu, D.D.; Boakye, P.; Jung, H.; Woo, S.H. Synergistic Dye Adsorption by Biochar from Co-Pyrolysis of Spent Mushroom Substrate and Saccharina Japonica. Bioresour. Technol. 2017, 244, 1142–1149. [Google Scholar] [CrossRef] [PubMed]
- Lagergren, S.Y. Zur Theorie Der Sogenannten Adsorption Gelöster Stoffe. K. Sven. Vetensk. Handl. 1898, 24, 1–39. [Google Scholar] [CrossRef]
- Ho, Y.S.; McKay, G. A Comparison of Chemisorption Kinetic Models Applied to Pollutant Removal on Various Sorbents. Process Saf. Environ. Prot. 1998, 76, 332–340. [Google Scholar] [CrossRef]
- Demiral, H.; Güngör, C. Adsorption of Copper(II) from Aqueous Solutions on Activated Carbon Prepared from Grape Bagasse. J. Clean. Prod. 2016, 124, 103–113. [Google Scholar] [CrossRef]
- Fouladgar, M.; Beheshti, M.; Sabzyan, H. Single and Binary Adsorption of Nickel and Copper from Aqueous Solutions by γ-Alumina Nanoparticles: Equilibrium and Kinetic Modeling. J. Mol. Liq. 2015, 211, 1060–1073. [Google Scholar] [CrossRef]
- Manfrin, J.; Gonçalves, A.C., Jr.; Schwantes, D.; Conradi, E., Jr.; Zimmermann, J.; Ziemer, G.L. Development of Biochar and Activated Carbon from Cigarettes Wastes and Their Applications in Pb2+ Adsorption. J. Environ. Chem. Eng. 2021, 9, 104980. [Google Scholar] [CrossRef]
- Bhat, A.; Megeri, G.B.; Thomas, C.; Bhargava, H.; Jeevitha, C.; Chandrashekar, S.; Madhu, G.M. Adsorption and Optimization Studies of Lead from Aqueous Solution Using γ-Alumina. J. Environ. Chem. Eng. 2015, 3, 30–39. [Google Scholar] [CrossRef]
- Medici, F.; Patterer, M.S.; Peluso, M.A.; Sambeth, J.E. Lead Adsorption from Aqueous Solution Using Manganese Oxides Recovered from Spent Alkaline and Zn/C Batteries. J. Solid Waste Technol. Manag. 2020, 46, 206–212. [Google Scholar] [CrossRef]
- Kamari, A.; Ngah, W.S.W. Isotherm, Kinetic and Thermodynamic Studies of Lead and Copper Uptake by H2SO4 Modified Chitosan. Colloids Surf. B Biointerfaces 2009, 73, 257–266. [Google Scholar] [CrossRef] [PubMed]
- Tran, H.N.; Lin, C.-C.; Woo, S.H.; Chao, H.-P. Efficient Removal of Copper and Lead by Mg/Al Layered Double Hydroxides Intercalated with Organic Acid Anions: Adsorption Kinetics, Isotherms, and Thermodynamics. Appl. Clay Sci. 2018, 154, 17–27. [Google Scholar] [CrossRef]
- Futalan, C.M.; Kan, C.-C.; Dalida, M.L.; Hsien, K.-J.; Pascua, C.; Wan, M.-W. Comparative and Competitive Adsorption of Copper, Lead, and Nickel Using Chitosan Immobilized on Bentonite. Carbohydr. Polym. 2011, 83, 528–536. [Google Scholar] [CrossRef]
- Futalan, C.M.; Kim, J.; Yee, J.-J. Adsorptive Treatment via Simultaneous Removal of Copper, Lead and Zinc from Soil Washing Wastewater Using Spent Coffee Grounds. Water Sci. Technol. 2019, 79, 1029–1041. [Google Scholar] [CrossRef]
- Abd El-Latif, M.M.; Ibrahim, A.M.; El-Kady, M.F. Adsorption Equilibrium, Kinetics and Thermodynamics of Methylene Blue from Aqueous Solutions Using Biopolymer Oak Sawdust Composite. J. Am. Sci. 2010, 6, 267–283. [Google Scholar]
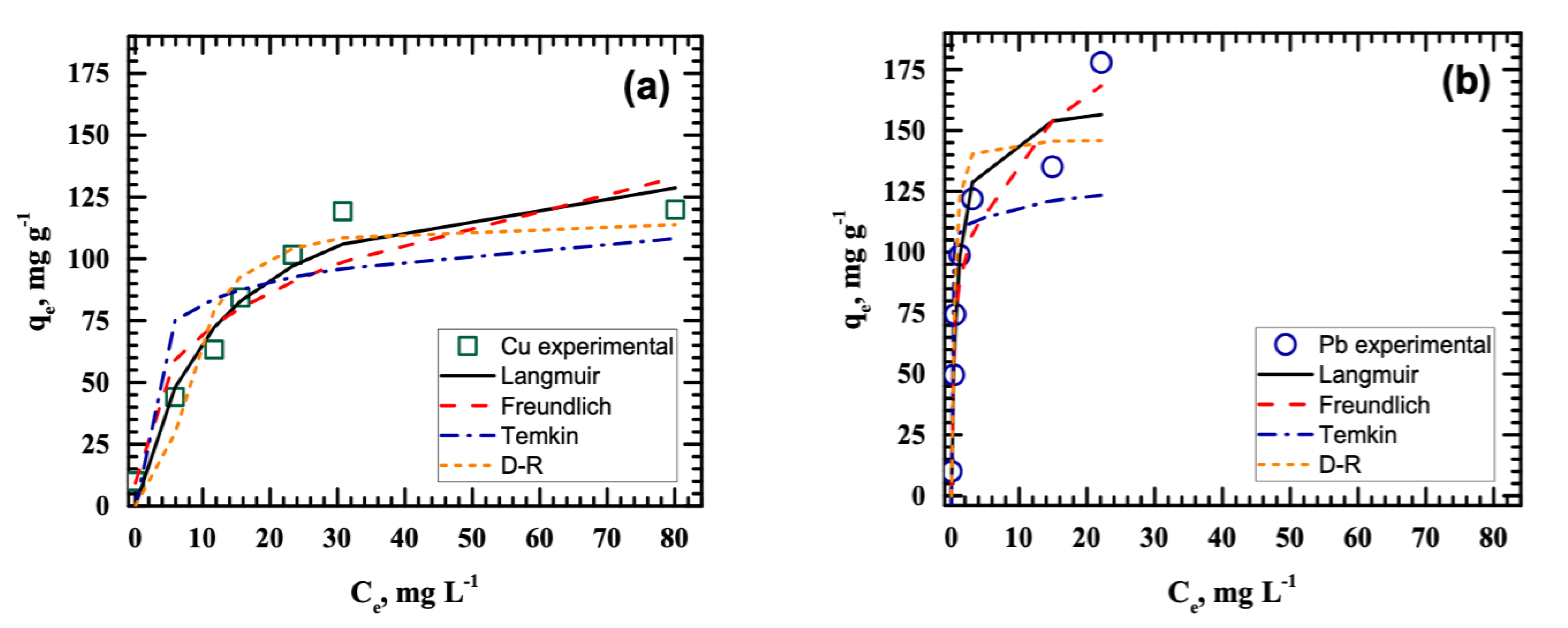
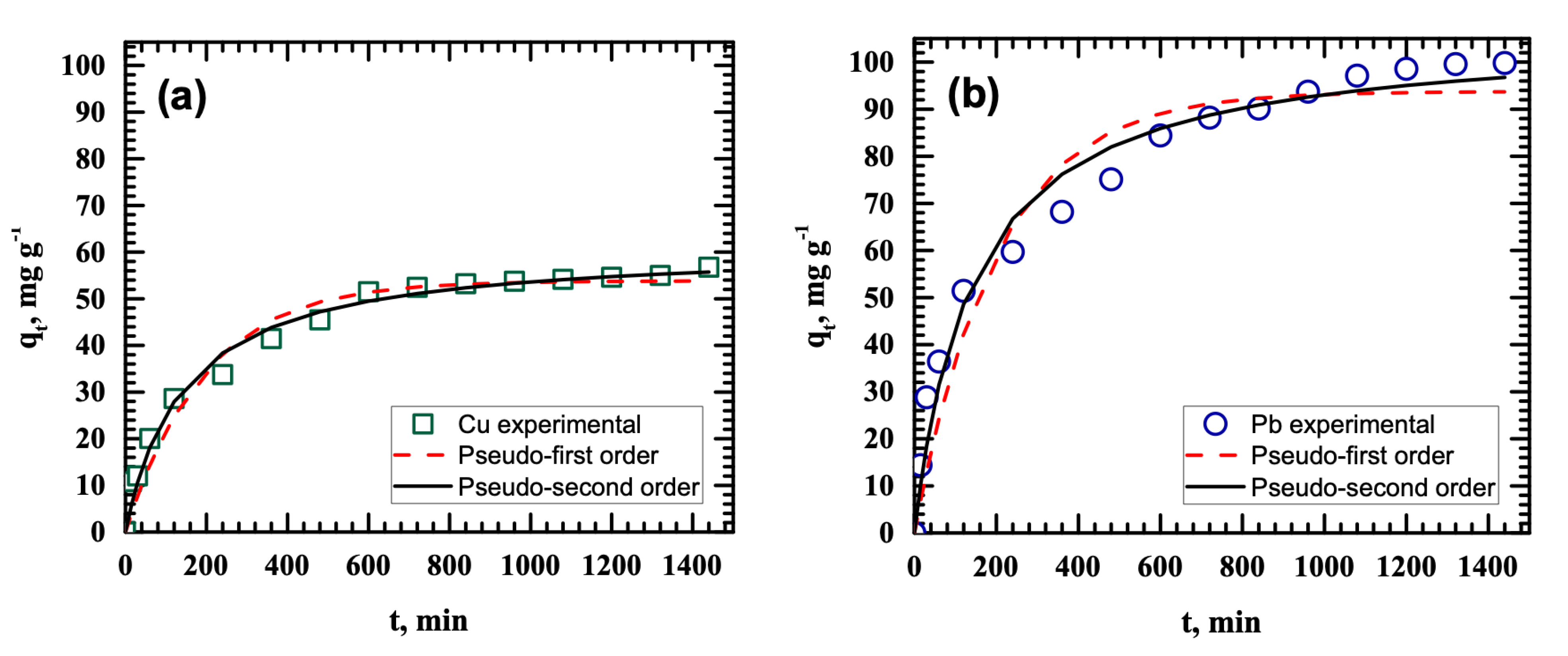
| Isotherm Model | Parameter | Cu(II) | Pb(II) |
|---|---|---|---|
| Langmuir | qm exp(mg g−1) | 119.90 | 177.89 |
| qm (mg g−1) | 148.58 | 162.19 | |
| KL (L mg−1) | 0.0808 | 1.2396 | |
| RL | 0.0583–0.5530 | 0.0040–0.0746 | |
| R2 | 0.9534 | 0.9460 | |
| Freundlich | n | 3.2331 | 4.4354 |
| KF (mg1−1/n L1/n g−1) | 34.2414 | 83.7481 | |
| R2 | 0.8952 | 0.9381 | |
| Temkin | B (J mol−1) | 12.70 | 5.3278 |
| KT (L g−1) | 62.88 | 5.1539 × 108 | |
| R2 | 0.7748 | 0.6245 | |
| Dubinin–Radushkevich | KDR (mol2 kJ−2) | 9.01 × 10−6 | 8.04 × 10−8 |
| qDR (mg g−1) | 114.77 | 145.99 | |
| R2 | 0.9207 | 0.8637 |
| Kinetic Model | Parameter | Cu(II) | Pb(II) |
|---|---|---|---|
| Pseudo-first-order | qe exp (mg·g−1) | 56.74 | 99.80 |
| qe (mg·g−1) | 53.82 | 93.74 | |
| k1 (min−1) | 0.0052 | 0.0050 | |
| R2 | 0.9679 | 0.9425 | |
| Pseudo-second-order | qe (mg·g−1) | 61.27 | 106.31 |
| k2 (g·mg−1·min−1) | 0.0001 | 6.61 × 10−5 | |
| R2 | 0.9872 | 0.9772 |
| Adsorbent | pH | Concentration (mg L−1) | Temperature (°C) | qmax (mg g−1) | References |
|---|---|---|---|---|---|
| Cu | |||||
| Activated carbon prepared from grape bagasse | 5.0 | 10 to 100 | 25 | 37.17 | [31] |
| γ-alumina nanoparticles | 5.0 | 25 to 200 | 25 | 51.30 | [32] |
| Natural manganese dioxide | 5.52–5.10 | 158.87 (2.5 mmol L−1) | 23 | 54.35 | [27] |
| Groundwater treatment sludge-derived manganese dioxide | 5.0 | 10 to 200 | 25 | 119.90 | This study |
| Pb | |||||
| Biochar and activated carbon from cigarettes wastes | 5.0 | 5 to 300 | 25 | 23.70 (biochar) 71.43 (activated carbon) | [33] |
| γ-Alumina | 5.0 | 10 to 100 | 25 | 65.67 | [34] |
| Manganese oxides recovered from spent alkaline and Zn/C batteries | 5 to 100 | 25 | 6.04 | [35] | |
| Groundwater treatment sludge-derived manganese dioxide | 5.0 | 10 to 200 | 25 | 177.89 | This study |
| Adsorbate | Temperature (°C) | ΔG° (kJ mol−1) | ΔH° (kJ mol−1) | ΔS° (kJ mol−1 K−1) | Ea (kJ mol−1) |
|---|---|---|---|---|---|
| Cu(II) | 25 | −18.030 | 65.29 | 0.2798 | 146.76 |
| 30 | −19.420 | ||||
| 35 | −20.146 | ||||
| Pb(II) | 25 | −22.588 | 37.62 | 0.2019 | 130.13 |
| 30 | −23.175 | ||||
| 35 | −23.809 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tumampos, S.B.; Ensano, B.M.B.; Pingul-Ong, S.M.B.; Ong, D.C.; Kan, C.-C.; Yee, J.-J.; de Luna, M.D.G. Isotherm, Kinetics and Thermodynamics of Cu(II) and Pb(II) Adsorption on Groundwater Treatment Sludge-Derived Manganese Dioxide for Wastewater Treatment Applications. Int. J. Environ. Res. Public Health 2021, 18, 3050. https://doi.org/10.3390/ijerph18063050
Tumampos SB, Ensano BMB, Pingul-Ong SMB, Ong DC, Kan C-C, Yee J-J, de Luna MDG. Isotherm, Kinetics and Thermodynamics of Cu(II) and Pb(II) Adsorption on Groundwater Treatment Sludge-Derived Manganese Dioxide for Wastewater Treatment Applications. International Journal of Environmental Research and Public Health. 2021; 18(6):3050. https://doi.org/10.3390/ijerph18063050
Chicago/Turabian StyleTumampos, Stephanie B., Benny Marie B. Ensano, Sheila Mae B. Pingul-Ong, Dennis C. Ong, Chi-Chuan Kan, Jurng-Jae Yee, and Mark Daniel G. de Luna. 2021. "Isotherm, Kinetics and Thermodynamics of Cu(II) and Pb(II) Adsorption on Groundwater Treatment Sludge-Derived Manganese Dioxide for Wastewater Treatment Applications" International Journal of Environmental Research and Public Health 18, no. 6: 3050. https://doi.org/10.3390/ijerph18063050
APA StyleTumampos, S. B., Ensano, B. M. B., Pingul-Ong, S. M. B., Ong, D. C., Kan, C.-C., Yee, J.-J., & de Luna, M. D. G. (2021). Isotherm, Kinetics and Thermodynamics of Cu(II) and Pb(II) Adsorption on Groundwater Treatment Sludge-Derived Manganese Dioxide for Wastewater Treatment Applications. International Journal of Environmental Research and Public Health, 18(6), 3050. https://doi.org/10.3390/ijerph18063050








