Old Tricks, New Opportunities: How Companies Violate the International Code of Marketing of Breast-Milk Substitutes and Undermine Maternal and Child Health during the COVID-19 Pandemic
Abstract
1. Introduction
Breastfeeding and Its Challenges during the COVID-19 Pandemic
2. Marketing and Breastfeeding
2.1. The International Code of Marketing of Breast-Milk Substitutes
2.2. How Do BMS Companies Promote?
2.3. Aggressive Marketing during COVID-19
3. Aim, Materials and Methods
- Are BMS companies marketing aggressively during the COVID-19 pandemic?
- If so, how are BMS companies capitalizing on the pandemic for their marketing activities?
- What are the implications for policy and programs?
3.1. Data Collection and Analysis
3.1.1. Qualitative Data
3.1.2. Desk Review with Data from the Department of Health Philippines
4. Results
4.1. Qualitative Themes
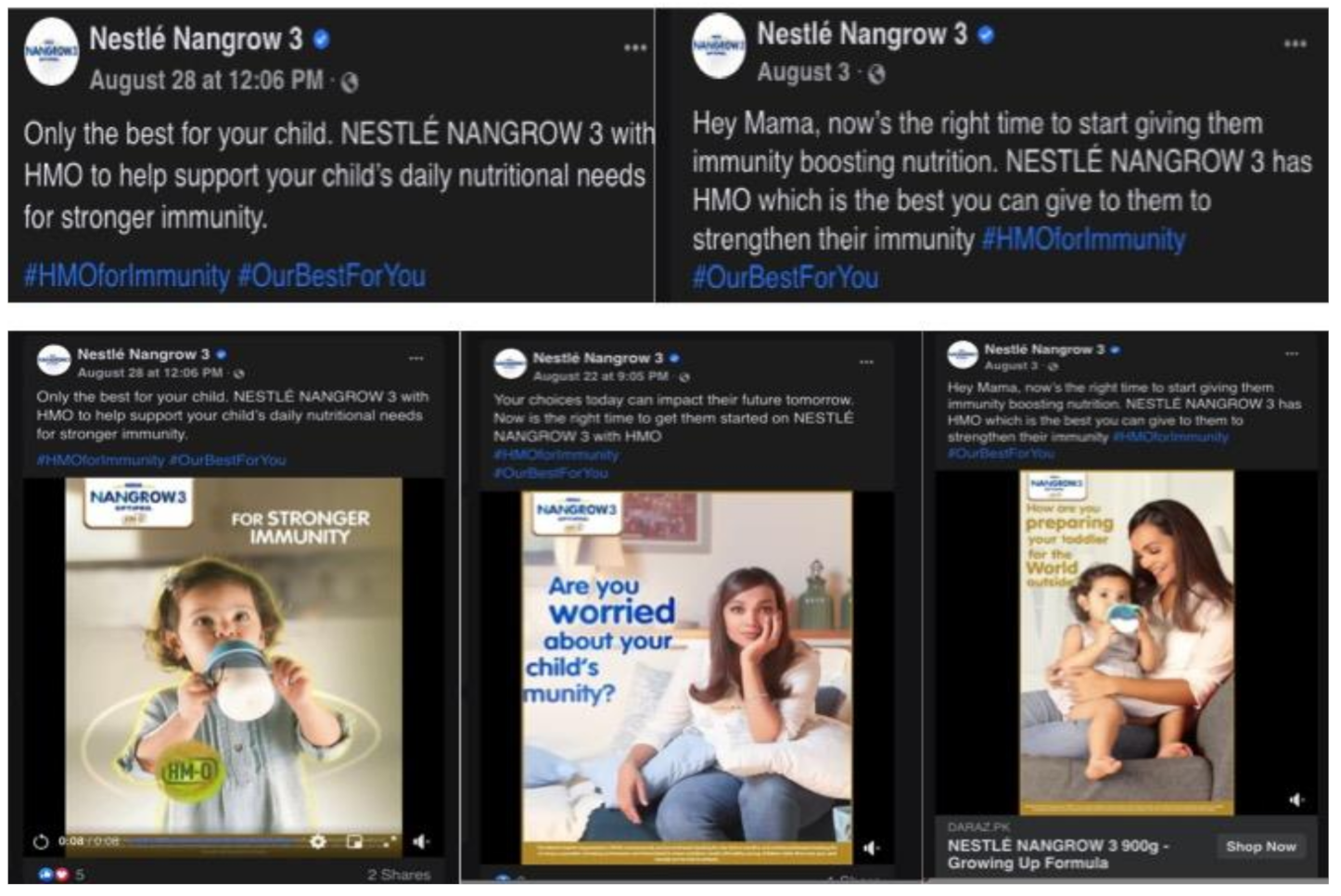
4.1.1. (i) Unfounded Health Claims on Immunity That Prompt Fear
4.1.2. (ii) Riding on Public Health Authority to Gain Legitimacy
4.1.3. (iii) Appealing to Public Sentiment on Solidarity and Hope
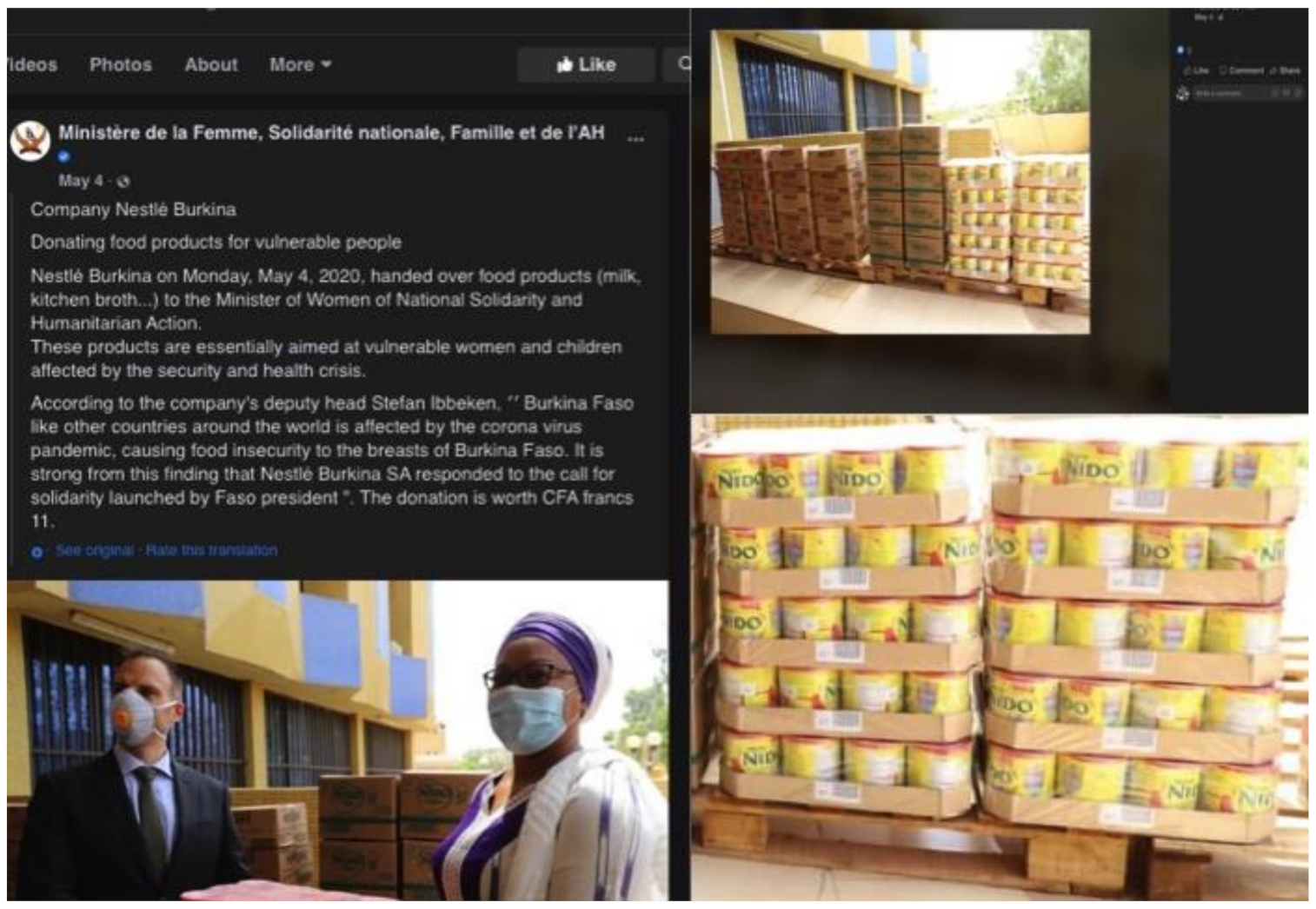
4.1.4. (iv) Influx of Donations of BMS and Supplies Related to COVID-19
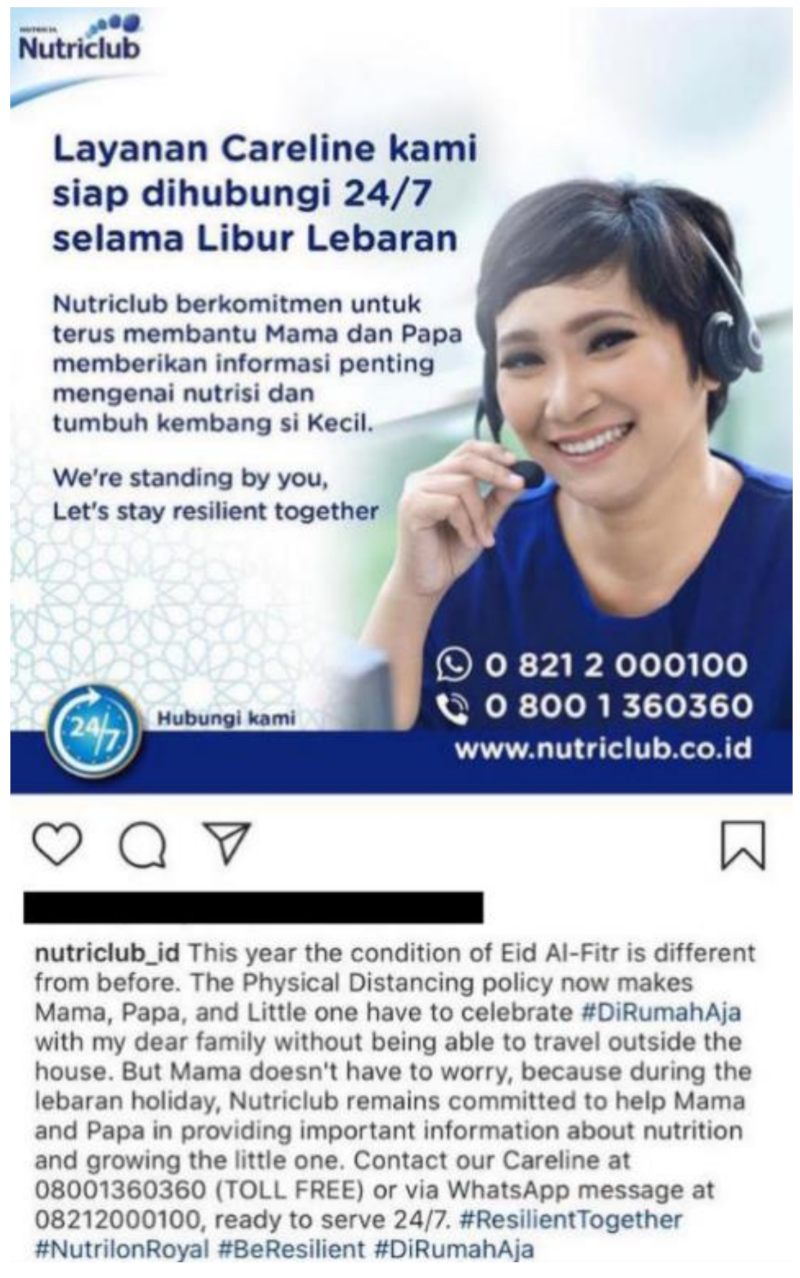
4.1.5. (v) Prominent Use of Digital Platforms to Reach Out to Parents
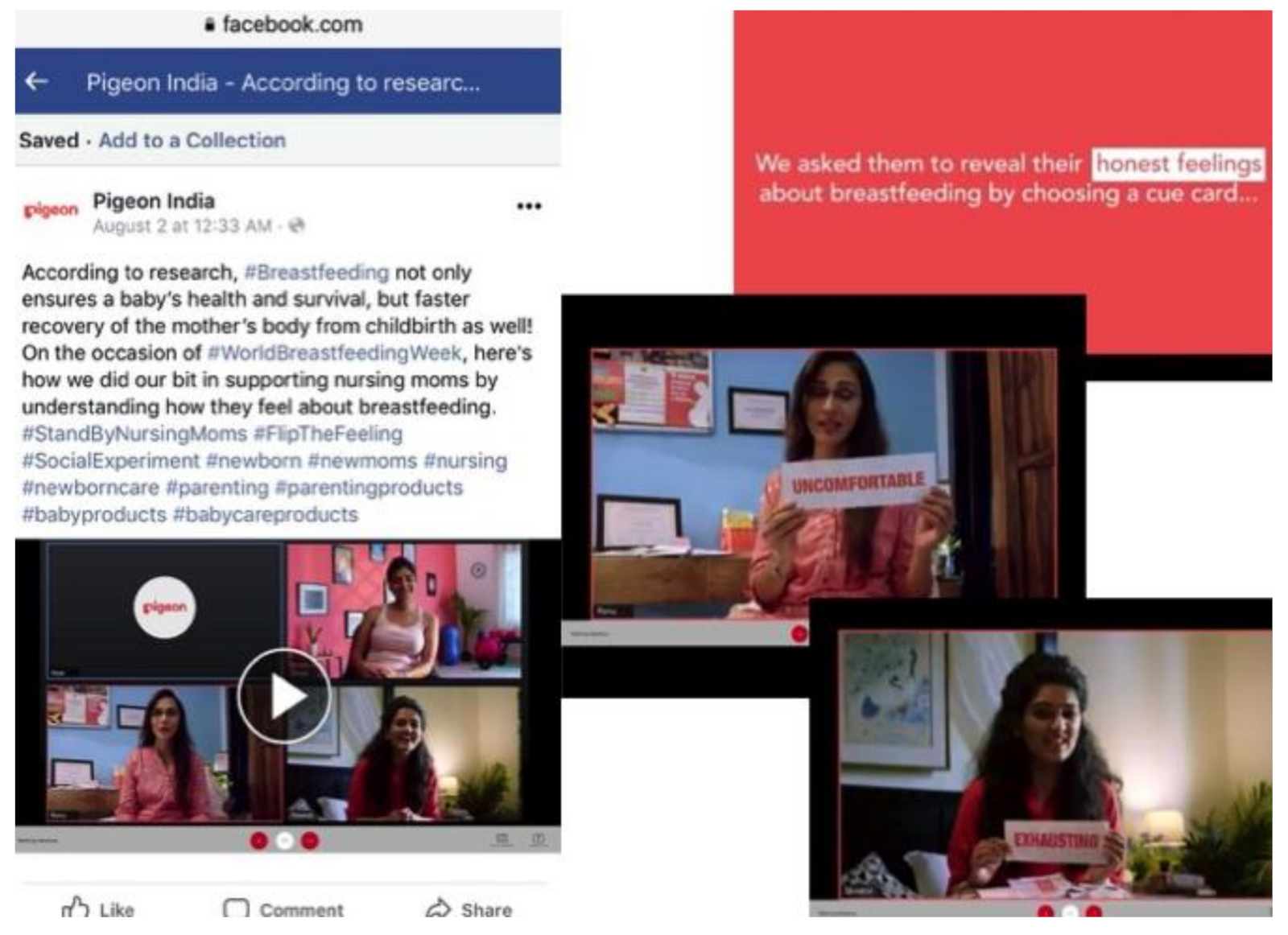
4.1.6. (vi) Promoting Uncertainty Through Endorsing Breastfeeding
4.1.7. (vii) Discounts on BMS Products That Are Linked to COVID-19
4.1.8. (viii) Reaching Out to Health Professionals through Sponsoring Educational Events on Topics Relating to COVID-19 and Infant and Young Child Feeding
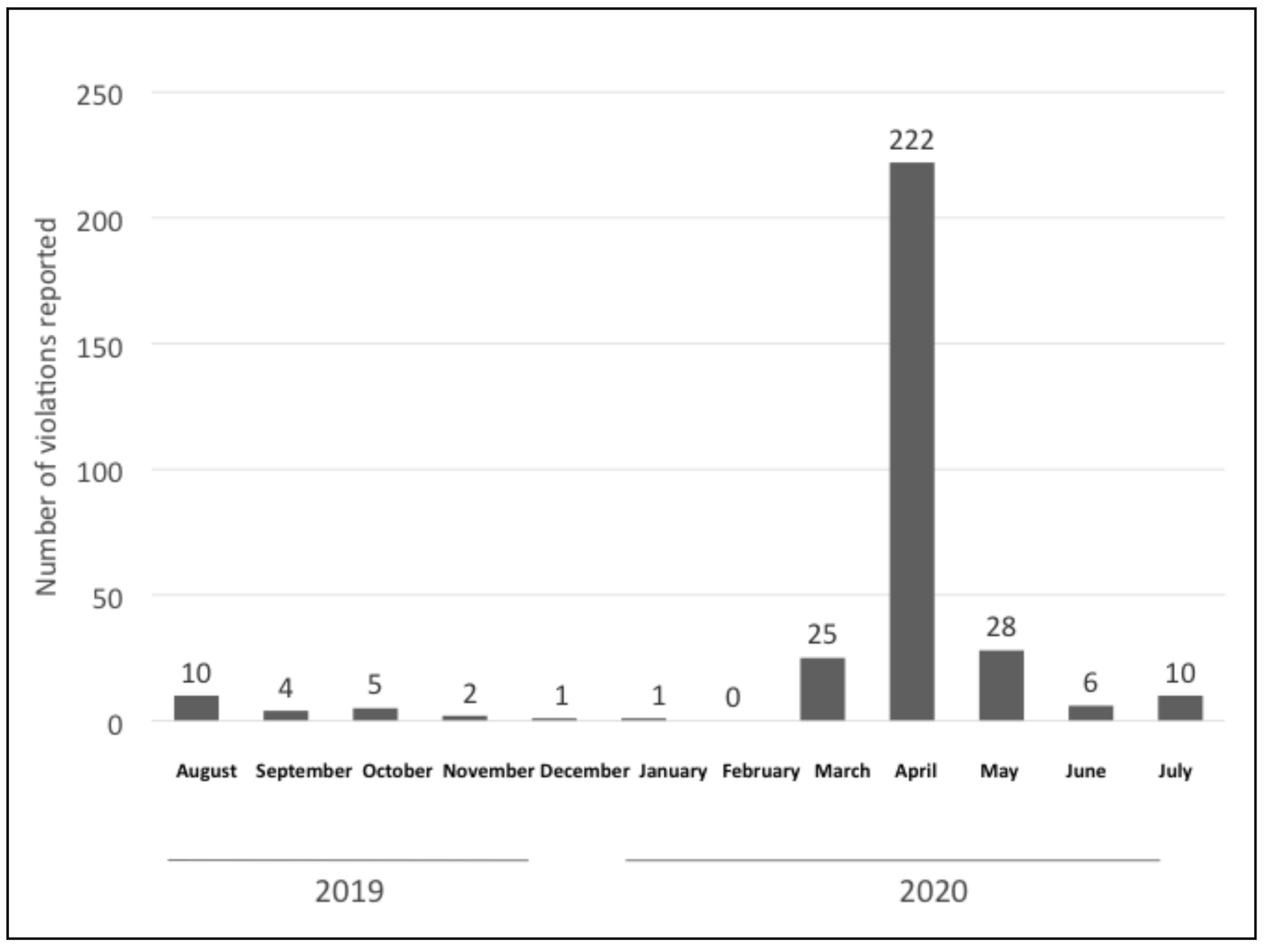
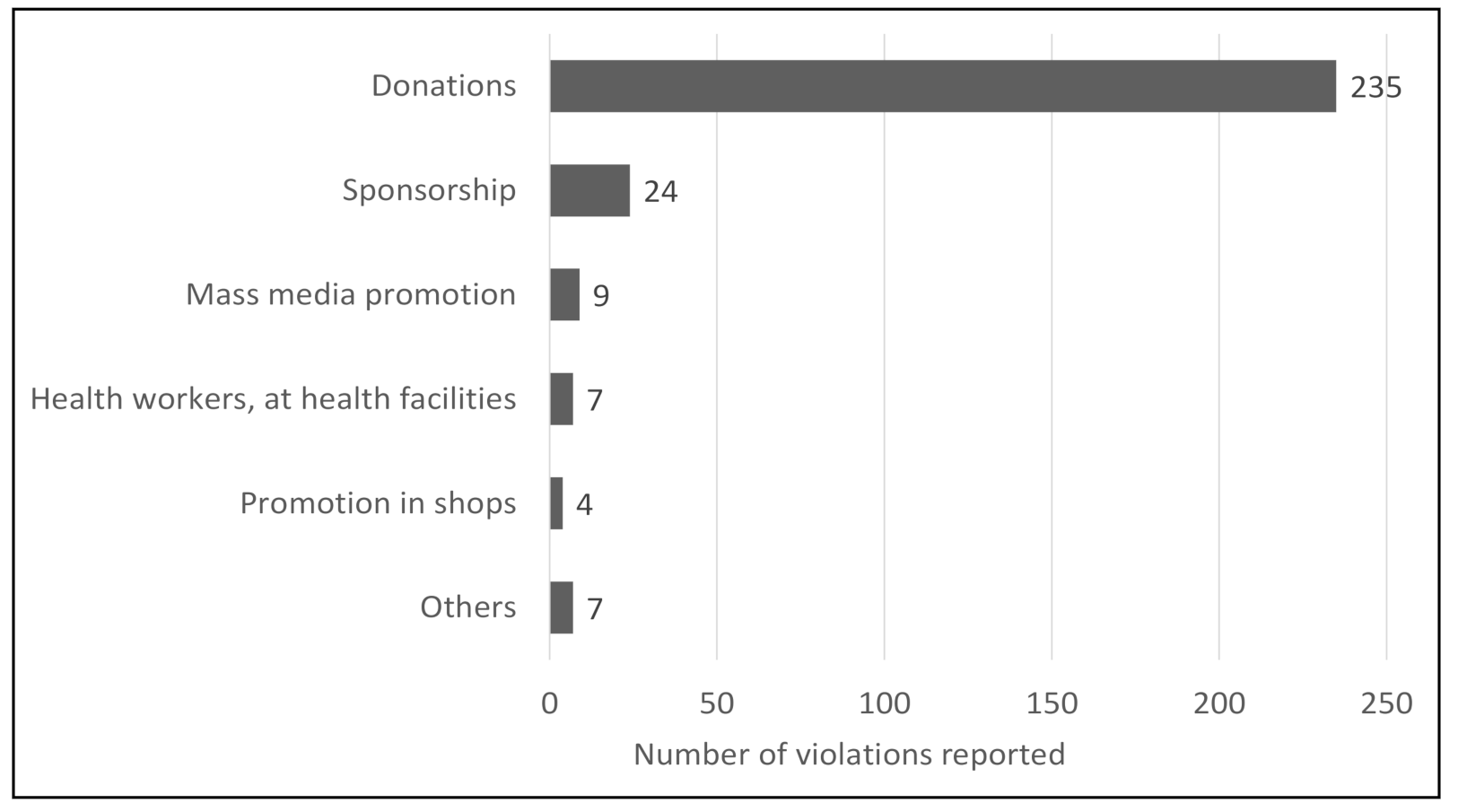
4.2. Reported Violations during COVID-19 in the Philippines
5. Discussion
5.1. Overview of Findings
5.2. Old Tricks That Have Found New Opportunities
5.3. Where Is the Accountabilty from Social Media?
5.4. Gaps and Challenges in Code Implementation
6. Limitations
7. Recommendations and Conclusions
7.1. Immediate Action for Governments and NGOs
7.1.1. (i) Monitoring Findings to Be Used to Inform Actions at the WHA
7.1.2. (ii) Targeted Enforcement Can Be Conducted with Existing Inspections
7.1.3. (iii) Breastfeeding in the Context of COVID-19 Should Be Promoted
7.1.4. (iv) Efforts Must Be Made to Prevent Spillover Effect of BMS Donations
7.1.5. (v) Code-Compliant Best Practices in Health Systems and Programs Need to Be Established as Policy
7.2. Long-Term Action on Code Implementation
7.2.1. (i) Code Awareness to Be Raised Through Public Education and Social Messages
7.2.2. (ii) Monitoring Must Be Systematic, Sustainable, and Independent
7.2.3. (iii) Social Media Platforms Need to Be Held to Account for Violating the Code
7.2.4. (iv) Laws Need to Be Reviewed (or Adopted) to Keep Up with Marketing Tactics
7.2.5. (v) Conflicts of Interest Hamper Political Will, and Thus Need to Be Tackled
7.2.6. (vi) Alternative Funding Options in Health Systems Should Be Explored
7.2.7. (vii) High-level Political Support Must Be Cultivated
7.3. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
| 1. Aim | To contribute to the provision of safe and adequate nutrition for infants by the protection and promotion of breastfeeding and the proper use of breastmilk substitutes, when these are necessary, on the basis of adequate information and through appropriate marketing and distribution. |
| 2. Scope | Applies to breastmilk substitutes * 1 or any food being marketed or otherwise represented as a partial or total replacement for breastmilk. This includes the following:
|
| 3. Promotion | No advertising or promotion of above products to the public. No nutrition or health claims on products. * ^ 2 |
| 4. Samples | No free samples to mothers, their families, or health care workers. |
| 5. Health care facilities | No promotion of products, i.e., no product displays, posters, calendars, or distribution of promotional materials. No mothercraft nurses or similar corporation-paid personnel. |
| 6. Health care workers | No gifts or samples to health care workers. Financial support and incentives should not create conflicts of interest. ^ 3 |
| 7. Supplies | No free or low-cost supplies of breastmilk substitutes to any part of the health care system. ^ 4 |
| 8. Information | Information and education materials must explain the benefits of breastfeeding, the health hazards associated with bottle-feeding, and the costs of using infant formula. Product information must be factual and scientific. Governments to avoid conflicts of interest so materials under infant and young child programs should not be sponsored by manufacturers and distributors. ^ 5 |
| 9. Labels | Product labels must clearly state the superiority of breastfeeding, the need for the advice of a health care worker, and a warning about health hazards. No pictures of infants, other pictures, or text idealizing the use of infant formula. Labels must contain the warning that powdered infant formula may contain pathogenic microorganisms and must be prepared and used appropriately. ^ 5 Labels on complementary foods should not cross-promote breastmilk substitutes, should not promote bottle-feeding, and should state the importance of continued breastfeeding. ^ 6 |
| 10. Quality | Unsuitable products, such as sweetened condensed milk, should not be promoted for babies. All products should be of a high quality (Codex Alimentarius Standards) and take account of the climatic and storage conditions of the country wherein they are used. |
Appendix B
| Year | Resolution | Key Point(s) |
|---|---|---|
| 1981 | WHA 34.22 | Stresses that adoption and adherence to the Code is a minimum requirement. Member States are urged to implement the Code into national legislation, regulations, and other suitable measures. |
| 1982 | WHA35.26 | Recognizes that commercial promotion of breastmilk substitutes contributes to an increase in artificial feeding and calls for renewed attention to implement and monitor the Code at national and international levels. |
| 1984 | WHA37.30 | Requests that the Director General work with Member States to implement and monitor the Code and to examine the promotion and use of foods unsuitable for infant and young child feeding. |
| 1986 | WHA39.28 |
|
| 1988 | WHA41.11 | Requests the Director General to provide legal and technical assistance to Member States in drafting or implementing the Code into national measures. |
| 1990 | WHA43.3 |
|
| 1994 | WHA47.5 |
|
| 1996 | WHA49.15 | Calls on Member States to ensure the following: 1. Complementary foods are not marketed for or used to undermine exclusive and sustained breastfeeding; 2. Financial support to health professionals does not create conflicts of interests; 3. Code monitoring is carried out in an independent, transparent manner free from commercial interest. |
| 2001 | WHA 54.2 | Sets global recommendation of “6 months” exclusive breastfeeding, with safe and appropriate complementary foods and continued breastfeeding for up to two years or beyond. |
| 2002 | WHA55.25 |
|
| 2005 | WHA58.32 | Asks Member States to 1. Ensure that nutrition and health claims for breastmilk substitutes are not permitted unless national/regional legislation allows; 2. Be aware of the risks of intrinsic contamination of powdered infant formulas and to ensure this information be conveyed through label warnings; 3. Ensure that financial support and other incentives for programmes and health professionals working in infant and young child health do not create conflicts of interest. |
| 2006 | WHA59.11 | Member States to make sure the response to the HIV pandemic does not include non-Code compliant donations of breastmilk substitutes or the promotion thereof. |
| 2006 | WHA59.21 | Commemorates the 25th anniversary of the adoption of the Code, welcomes the 2005 Innocenti Declaration, and asks WHO to mobilize technical support for Code implementation and monitoring. |
| 2008 | WHA61.20 | Urges Member States to 1. Scale up efforts to monitor and enforce national measures and to avoid conflicts of interest; 2. Investigate the safe use of donor milk through human milk banks for vulnerable infants, mindful of national laws and cultural and religious beliefs. |
| 2010 | WHA63.23 |
|
| 2012 | WHA65.6 |
|
| 2014 | WHA67(9) | Infant and Young Child Nutrition (MIYCN) Plan, which includes increasing the rate of exclusive breastfeeding to at least 50% by 2025 as a global target. The indicator for regulation of marketing is the number of countries with legislation or regulations fully implementing the Code and Resolutions. |
| 2016 | WHA69.9 | This Resolution welcomes the WHO Guidance on ending the inappropriate promotion of foods for infants and young children. It calls upon 1. Member States to take all necessary measures to implement the Guidance; 2. Manufacturers and distributors of foods for infants and young children to adhere to the Guidance. The Guidance clarified that follow-up milks and growing-up milks are covered by the Code and should be treated as such when implementing the Code. The Guidance also recommends that there should be no cross-promotion to promote breastmilk substitutes via the promotion of foods for infants and young children. |
| 2018 | WHA71.9 | This Resolution urges Member States to 1. Reinvigorate the Baby-friendly Hospital Initiative and the full integration of the revised 10 Steps to Successful Breastfeeding, which incorporates Code compliance in Step 1; 2. Take all necessary measures to implement recommendations to end the inappropriate promotion of foods for infants and young children. |
References
- Thorley, V. Is breastfeeding ‘normal’? Using the right language for breastfeeding. Midwifery 2019, 69, 39–44. [Google Scholar] [CrossRef]
- Rollins, N.C.; Bhandari, N.; Hajeebhoy, N.; Horton, S.; Lutter, C.K.; Martines, J.C.; Piwoz, E.G.; Richter, L.M.; Victora, C.G. Why invest, and what it will take to improve breastfeeding practices? Lancet 2016, 387, 491–504. [Google Scholar] [CrossRef]
- Victora, C.G.; Bahl, R.; Barros, A.J.D.; França, G.V.; Horton, S.; Krasevec, J.; Murch, S.; Sankar, M.J.; Walker, N.; Rollins, N.C.; et al. Breastfeeding in the 21st century: Epidemiology, mechanisms, and lifelong effect. Lancet 2016, 387, 475–490. [Google Scholar] [CrossRef]
- Walters, D.D.; Phan, L.T.H.; Mathisen, R. The cost of not breastfeeding: Global results from a new tool. Health Policy Plan. 2019, 34, 407–417. [Google Scholar] [CrossRef]
- Piwoz, E.G.; Huffman, S.L. The Impact of Marketing of Breast-Milk Substitutes on WHO-Recommended Breastfeeding Practices. Food Nutr. Bull. 2015, 36, 373–386. [Google Scholar] [CrossRef]
- Hastings, G.; Angus, K.; Eadie, D.; Hunt, K. Selling second best: How infant formula marketing works. Glob. Health 2020, 16, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Thorley, V. The Dilemma of Breastmilk Feeding. Available online: https://search.informit.org/doi/10.3316/INFORMIT.974747606578576 (accessed on 1 June 2011).
- Bhutta, Z.A.; Hauerslev, M.; Farmer, M.; Lewis-Watts, L. COVID-19, children and non-communicable diseases: Translating evidence into action. Arch. Dis. Child. 2021, 106, 141–142. [Google Scholar] [CrossRef] [PubMed]
- Pereira, A.; Cruz-Melguizo, S.; Adrien, M.; Fuentes, L.; Marin, E.; Forti, A.; Perez-Medina, T. Breastfeeding mothers with COVID-19 infection: A case series. Int. Breastfeed. J. 2020, 15, 1–8. [Google Scholar] [CrossRef]
- Roberton, T.; Carter, E.D.; Chou, V.B.; Stegmuller, A.R.; Jackson, B.D.; Tam, Y.; Sawadogo-Lewis, T.; Walker, N. Early estimates of the indirect effects of the COVID-19 pandemic on maternal and child mortality in low-income and middle-income countries: A modelling study. Lancet Glob. Health 2020, 8, e901–e908. [Google Scholar] [CrossRef]
- Altug, D.; Asri, M.; Neil, P.; Maria, M.C.; Antony, P. Early life factors and COVID-19 infection in England: A prospective analysis of UK Biobank participants. Early Hum. Dev. 2021, 105326. [Google Scholar] [CrossRef]
- Moro, G.E.; Bertino, E. Breastfeeding, Human Milk Collection and Containers, and Human Milk Banking: Hot Topics during the COVID-19 Pandemic. J. Hum. Lact. 2020, 36, 604–608. [Google Scholar] [CrossRef] [PubMed]
- Palmquist, A.E.L.; Asiodu, I.V.; Quinn, E.A. The COVID-19 liquid gold rush: Critical perspectives of human milk and SARS-CoV-2 infection. Am. J. Hum. Biol. 2020, e23481. [Google Scholar] [CrossRef]
- WHO. Breastfeeding and COVID-19. Scientific Brief. WHO. 2020. Available online: https://www.who.int/news-room/commentaries/detail/breastfeeding-and-covid-19 (accessed on 20 September 2020).
- Busch-Hallen, J.; Walters, D.; Rowe, S.; Chowdhury, A.; Arabi, M. Impact of COVID-19 on maternal and child health. Lancet Glob. Health 2020, 8, e1257. [Google Scholar] [CrossRef]
- Tomori, C.; Gribble, K.; Palmquist, A.E.; Ververs, M.; Gross, M.S. When separation is not the answer: Breastfeeding mothers and infants affected by COVID-19. Matern. Child Nutr. 2020, 16, e13033. [Google Scholar] [CrossRef] [PubMed]
- Hoang, D.V.; Cashin, J.; Gribble, K.; Marinelli, K.; Mathisen, R. Misalignment of global COVID-19 breastfeeding and newborn care guidelines with World Health Organization recommendations. BMJ Nutr. Prev. Health 2020, 3, 339–350. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Liu, C.; Dong, L.; Zhang, C.; Chen, Y.; Liu, J.; Duan, C.; Zhang, H.; Mol, B.W.; Dennis, C.-L.; et al. Coronavirus disease 2019 among pregnant Chinese women: Case series data on the safety of vaginal birth and breastfeeding. BJOG: Int. J. Obstet. Gynaecol. 2020, 127, 1109–1115. [Google Scholar] [CrossRef]
- Fan, C.; Lei, D.; Fang, C.; Li, C.; Wang, M.; Liu, Y.; Bao, Y.; Sun, Y.; Huang, J.; Guo, Y.; et al. Perinatal Transmission of 2019 Coronavirus Disease–Associated Severe Acute Respiratory Syndrome Coronavirus 2: Should We Worry? Clin. Infect. Dis. 2020. [Google Scholar] [CrossRef] [PubMed]
- Groß, R.; Conzelmann, C.; Müller, J.A.; Stenger, S.; Steinhart, K.; Kirchhoff, F.; Münch, J. Detection of SARS-CoV-2 in human breastmilk. Lancet 2020, 395, 1757–1758. [Google Scholar] [CrossRef]
- Furneaux, R. The Baby Brands Turning Indonesian Instagram into Free Formula Ads. The Bureau of Investigative Journalism. 30 July 2020. Available online: https://www.thebureauinvestigates.com/stories/2020-07-30/the-baby-brands-turning-indonesian-instagram-into-free-formula-milk-ads (accessed on 20 August 2020).
- Gribble, K.; Mathisen, R.; Ververs, M.-T.; Coutsoudis, A. Mistakes from the HIV pandemic should inform the COVID-19 response for maternal and newborn care. Int. Breastfeed. J. 2020, 15, 1–5. [Google Scholar] [CrossRef]
- How Companies Exploit the COVID-19 Crisis. Available online: http://www.babymilkaction.org/archives/24341 (accessed on 8 May 2020).
- Van Tulleken, C.; Wright, C.; Brown, A.; McCoy, D.; Costello, A. Marketing of breastmilk substitutes during the COVID-19 pandemic. Lancet 2020, 396, e58. [Google Scholar] [CrossRef]
- Dodgson, J.E. Protecting and Supporting the WHO International Code During COVID-19. J. Hum. Lact. 2020, 36, 387–389. [Google Scholar] [CrossRef] [PubMed]
- Jelliffe, D.B. Commerciogenic Malnutrition? Nutr. Rev. 2009, 30, 199–205. [Google Scholar] [CrossRef]
- WHO. International Code of Marketing of Breast-milk Substitutes. WHO. 1981. Available online: https://www.who.int/nutrition/publications/code_english.pdf (accessed on 20 September 2020).
- IBFAN-ICDC. Annotated Compilation of the International Code of Marketing of Breastmilk Substitutes and Relevant WHA Resolutions (Updated in 2018); IBFAN-ICDC: Penang, Malaysia, 2018. [Google Scholar]
- UN General Assembly. Adoption of a Convention on the Rights of the Child. New York, NY: United Nations. 17 November 1989. Available online: https://treaties.un.org/doc/Treaties/1990/09/19900902%2003-14%20AM/Ch_IV_11p.pdf (accessed on 30 August 2020).
- IBFAN-ICDC. Code Essentials 1–4, 2nd ed.; IBFAN-ICDC: Penang, Malaysia, 2018. [Google Scholar]
- IBFAN-ICDC. Breaking the Rules, Stretching the Rules 2017. Evidence of Violations of the International Code of Marketing of Breastmilk Substitutes and Subsequent Resolutions; IBFAN-ICDC: Penang, Malaysia, 2017. [Google Scholar]
- Braun, V.; Clarke, V. Using thematic analysis in psychology. Qual. Res. Psychol. 2006, 3, 77–101. [Google Scholar] [CrossRef]
- Peterson, B.L. Thematic Analysis/Interpretive Thematic Analysis. In The International Encyclopedia of Communication Research Methods; Wiley: Hoboken, NJ, USA, 2017; pp. 1–9. [Google Scholar]
- Bradley, E.H.; Curry, L.A.; Devers, K.J. Qualitative data analysis for health services research: Developing taxonomy, themes, and theory. Health Serv. Res. 2007, 42, 1758–1772. [Google Scholar] [CrossRef]
- Syed, M.; Nelson, S.C. Guidelines for Establishing Reliability When Coding Narrative Data. Emerg. Adulthood 2015, 3, 375–387. [Google Scholar] [CrossRef]
- Creswell, J.W.; Miller, D.L. Determining Validity in Qualitative Inquiry. Theory Into Pract. 2000, 39, 124–130. [Google Scholar] [CrossRef]
- Corbin, J.; Strauss, A. Basics of Qualitative Research: Techniques and Procedures for Developing Grounded Theory; Sage Publications, Inc.: Thousand Oaks, CA, USA, 2014. [Google Scholar]
- Vaismoradi, M.; Turunen, H.; Bondas, T. Content analysis and thematic analysis: Implications for conducting a qualitative descriptive study. Nurs. Health Sci. 2013, 15, 398–405. [Google Scholar] [CrossRef] [PubMed]
- Colosbaby_Vitadairy [@ColosbabyVitadairy]. Cháy Hàng Sữa Colosbaby vì Các Mẹ Tăng Đề Kháng Cho Con Trong Mùa Dịch [Colosbaby Milk is Sold Out Because Mothers Want to Build Up Resistance to Contagious Disease for Their Babies]. Facebook. 3 March 2020. Available online: https://www.facebook.com/ColosbabyVitadairy/posts/122219256017585 (accessed on 30 September 2020).
- Suabottot. (n.d.). ColosBaby Colosbaby 0 + 400g (Children from 0–12 Months). Available online: https://suabottot.com/san-pham/sua-non-colosbaby-400g-0-12-thang/ (accessed on 25 September 2020).
- Nestlé Nangrow 3 [@NestleNangrow3Pakistan]. Hey Mama, Now’s the Right Time to Start Giving Them Immunity Boosting Nutrition. NESTLÉ NANGROW 3 Has HMO Which Is the Best You Can Give to Them to Strengthen Their Immunity [Video]. Facebook. 3 August 2020. Available online: https://www.facebook.com/NestleNangrow3Pakistan/videos/725138781655439 (accessed on 20 September 2020).
- Nestlé Nangrow 3 [@NestléNangrow3Pakistan]. Only the Best for Your Child. NESTLÉ NANGROW 3 with HMO to Help Support Your Child’s Daily Nutritional Needs for Stronger Immunity [Video]. Facebook. 28 August 2020. Available online: https://www.facebook.com/NestleNangrow3Pakistan/posts/3187602367990686 (accessed on 20 September 2020).
- Nestlé Nangrow 3 [@NestléNangrow3Pakistan]. Our Biggest Breakthrough [Video]. Facebook. 22 August 2020. Available online: https://www.facebook.com/NestleNangrow3Pakistan/videos/337129764088791 (accessed on 20 September 2020).
- China Children and Teenagers’ Fund. Fángyì Bāo, Zàixiàn Yìzhěn, Gěi Yùn Mā de Zhuānshŭ Gōngyì Fúwù [Epidemic Prevention Package, Online Free Consultation, Exclusive Public Service for Pregnant Mothers]. 28 February 2020. Available online: http://www.cctf.org.cn/news/info/2020/02/28/5336.html (accessed on 1 September 2020).
- Zhāng, X. Juānzèng yíngyăng lĭbāo yăpéi quánlì chíyuán māmā fùgōng [Donate Nutrition Packages, Abbott Fully Supports Mothers Returning to Work]. Běijīng Tóutiáo. 20 March 2020. Available online: https://app.bjtitle.com/8816/newshow.php?newsid=5576384&typeid=13&uid=1&did=&mood= (accessed on 20 September 2020).
- Wŭgŭ Cáijīng. Fēi shì lán xuānbù juānzèng 500 wàn yuán, bāokuò értóng yīyòng hùlĭ kŏuzhào hé pèifāng năifěn! {Friesland Campina Announces Donation of Five Million Yuan, Including Child Medical Masks and FORMULA milk Powder!]. Sina. 31 January 2020. Available online: http://k.sina.com.cn/article_5617150275_14ecee94300100qh8s.html?cre=wappage&mod=r&loc=2&r=9&rfunc=11&tj=none (accessed on 20 September 2020).
- Danone-AQUA PR. Government to Prevent Stunting during COVID-19 Pandemic. Antara Megapolitan. 20 June 2020. Available online: https://megapolitan.antaranews.com/berita/101032/danone-sn-indonesia-supports-central-java-government-to-prevent-stunting-during-covid-19-pandemic (accessed on 20 September 2020).
- Nestlé. Nestlé Canada Provides Over $2 Million in Food Donations to Food Banks Canada to Help Families and Communi-ties Impacted by COVID-19. Nestlé Good Food Good Life. 9 April 2020. Available online: https://www.corporate.Nestle.ca/en/media/pressreleases/nestl%C3%A9-canada-provides-over-2-million-food-donations-food-banks-canada-help (accessed on 30 September 2020).
- Department of Health Philippines. Official Database of Reported Milk Code Violations; [Data file (January 2019 to July 2020)]; Department of Health: Manila, Philippines, 2020.
- Feihe. Zhuījiā 1 yì yuán năifěn! Zhōngguó fēi hè lěijì juānzèng 2 yì yuán kuăn wù. National Business Daily. 1 February 2020. Available online: https://m.nbd.com.cn/articles/2020-02-01/1404212.html (accessed on 30 August 2020).
- Danone. Danone to Support Government’s Fight against COVID-19 in Malaysia. PR Newswire. 1 April 2020. Available online: https://en.prnasia.com/releases/apac/danone-to-support-government-s-fight-against-covid-19-in-malaysia-276559.shtml (accessed on 1 September 2020).
- Ministère de la Femme, Solidarité Nationale, Famille et de l’AH. Société Nestlé Burkina. Don de Produits Alimentaires Pour les Personnes Vulnérables [Nestlé Burkina Company. Food Donation for Vulnerable people]. Facebook. 4 May 2020. Available online: https://www.facebook.com/permalink.php?story_fbid=3385142038181707&id=1346633478699250 (accessed on 30 September 2020).
- Séra, W. Nestlé Burkina: Trois Respirateurs Pour Sauver des vies. Lefaso.net. 6 August 2020. Available online: https://lefaso.net/spip.php?article98536 (accessed on 20 September 2020).
- Liu, Y. Online Medical Consultation, Free e-Books—How Companies Are Fostering Bonds with Customers Amid Coronavirus Outbreak. The South China Morning Post. 13 February 2020. Available online: https://www.scmp.com/business/china-business/article/3050015/online-medical-consultation-free-e-books-how-companies-are (accessed on 1 September 2020).
- Nutriclub Indonesia [@nutriclub_id]. Layanan Careline kami siap dihubungi 24/7 selama Libur Lebaran. Insta-gram. Retrieved 22 May 2020. Available online: https://www.instagram.com/p/CAfoFLUjQxS/ (accessed on 20 September 2020).
- OnDoctor App [@onDoctor]. Douttar kyaalhcain yu mwan k pyawwpya payy m y kalayynae ko hkan aarr aakyaungg(Episode-1). Facebook. Retrieved 21 August 2020. Available online: https://www.facebook.com/1776667909230381/videos/2840535766181481 (accessed on 20 September 2020).
- Myanmar Parenting (n.d.). About [Private Group Profile]. Facebook. Available online: https://www.facebook.com/groups/myanmarparenting (accessed on 20 October 2020).
- Illuma. Motherhood during COVID-19. How to enhance your child’s well-being and [Advertisement for Illuma Human Affinity]. Mummys Mark. Pregnancy Baby Guide 2020, 21, 18–19. [Google Scholar]
- Pigeon. Pigeon India Initiates #StandbyNursingMoms to Celebrate Breastfeeding Week. Yahoo News. 14 August 2020. Available online: https://in.news.yahoo.com/pigeon-india-initiates-standbynursingmoms-celebrate-124817054.html (accessed on 20 September 2020).
- Pigeon India [@pigeoncare]. According to Research, #Breastfeeding Not Only Ensures a baby’s Health and Survival, But Faster Recovery of the Mother’s Body from Childbirth as Well! [Video]. Facebook. 2 August 2020. Available online: https://www.facebook.com/pigeoncare/videos/2342999596004835 (accessed on 20 September 2020).
- Pigeon India [@pigeoncare]. Mothers Are Breastfeeding Superheroes. Do you agree Mommies? Facebook. 29 July 2020. Available online: https://www.facebook.com/pigeoncare/photos/3123843321003005 (accessed on 20 September 2020).
- Medela (n.d.). Supporting Breastfeeding during COVID-19: November 18, 2020 Live Webinar. Medela. Available online: https://www.medela.us/mbus/for-professionals/Education/Courses/Detail/230 (accessed on 30 October 2020).
- Medela Kenya [@MedelaKenya]. Is It Safe to #breastfeedif I Have #Coronavirus? Facebook. 25 April 2020. Available online: https://www.facebook.com/MedelaKenya/photos/118345433170885 (accessed on 20 September 2020).
- Hi-Family Club Laos [@HiFamilyClubLaos]. Aemsongsai lu bo namnom thi p am kma misi thiaetktangkan ma sek vanam nom aetla si bong bokjangaed lae mi antalai to luk nony lubo dai thangvidio lae boeng lailaoiad salaphad khuaam kangvon kiauakabkan hai nom phoemtoem dai bonni [Do You Wonder If the Milk that Comes Out of the Breast is a Different Color! Check What Each Color of Milk Indicates and Whether It Is Harmful to the Baby. Watch the Video and See More Details about Breastfeeding Concerns Here]. Facebook. Retrieved 1 September 2020. Available online: https://www.facebook.com/HiFamilyClubLaos/videos/178956873443959 (accessed on 5 June 2020).
- Gerber (n.d.). An Important Message for the Gerber Community on COVID-19. Gerber. Available online: http://news.gerber.com/news/an-important-message-for-the-gerber-community-on-covid-19 (accessed on 7 September 2020).
- Similac. In Difficult Times, Count on Similac. You Promise to Support and Nourish Your Baby. We Promise to Help. Similac. 20 March 2020. Available online: https://similac.com/count-on-similac (accessed on 20 September 2020).
- Kavle, J.; Wambu, R. How Kenya is Fighting Back to Protect Breastfeeding: Addressing Infant Formula Industry Tactics during COVID-19. Healthy Newborn Network. 1 August 2020. Available online: https://www.healthynewbornnetwork.org/blog/how-kenya-is-fighting-back-to-protect-breastfeeding-addressing-infant-formula-industry-tactics-during-covid-19/ (accessed on 20 September 2020).
- UP Med Webinars [@upmedwebinars]. Pregnancy in the Time of COVID-19. Join Our Webinar Tomorrow, April 24, at 12 NN! Facebook. 23 April 2020. Available online: https://www.facebook.com/upmedwebinars/photos/bc.AbovlJvn9Yz7phRfX5Wun1pSvEiEyvmi8sEJLeWKyx75NoRWgmVZ0sxbgW4CY4WDBVbVqC_0NUBivJBBgAcGBJ2gfQ5Dq1remEGjSt632nceO93A0ujVqGDkYmz6YL4QTeU0JOiURdXJcBQvfTpLusmG/2598700883568925/?opaqueCursor=AbpL_3mwMxMjOPpzMACdYasFy862HL8y1dcu_dJLL28mPoRisy9rSvTKiO_5m4vp1IDflv6JQQUeP8AHDghvPtLgQS1Oug_la260zMCh3PrO6JnXKYIWEfIjqxIPCYEx0u94JncI7Ke4hHMTXvdRQuPFluFaL-Au7pvTmyMDEuxt-YwZUIwi5K3C4PAF_2NL4qVeCCWnM7g37Kz_juIEil4vflu4W4IAyNlVI-M9U5BiPMSTRVPSXtuMNTS16LTkH2ihsteS6yxd9pz4HH8kKT-ozHjFg3jFgVZVRzUHn4uaIR8ROLaA7sC8HzVBju3rs-bJrWxzPEj_a1aqCo4rUQ37e3GqYm6MefBPA3jJlv5m3iQrr40gv7eAHowOwh65Sy_Ka-E1DhprC34QO7Ua6WZ0hwOi3VmzL5IVA0rYTWms_lLwR3BZk8D2m9zkocT1_VXCHfYfP1ML-bSGdqWxCNS6ttpvBQlwPr0MIoxUE8bHCVVSlnP-xhB-r2R6axJkZc8FIN29L5PTjsw1x3cTi9_sQ-3sv9RUFAIJ7oA-KJMcEw (accessed on 20 September 2020).
- Pago, A.C. Milk Code Confusion Cripples LGUs Response for Infants. Philippine Center for Investigative Journalism. 9 October 2020. Available online: https://pcij.org/article/4348/milk-and-the-pandemic-milk-code-confusion-cripples-lgus-response-for-infants (accessed on 11 October 2020).
- Hughes, H.K.; Landa, M.M.; Sharfstein, J.M. Marketing Claims for Infant Formula. JAMA Pediatr. 2017, 171, 105. [Google Scholar] [CrossRef] [PubMed]
- Pomeranz, J.L.; Palafox, M.J.R.; Harris, J.L. Toddler drinks, formulas, and milks: Labeling practices and policy implications. Prev. Med. 2018, 109, 11–16. [Google Scholar] [CrossRef]
- Munblit, D.; Crawley, H.; Hyde, R.; Boyle, R.J. Health and nutrition claims for infant formula are poorly substantiated and potentially harmful. BMJ 2020, 369, m875. [Google Scholar] [CrossRef]
- IFE (Infant and Young Child Feeding in Emergencies) Core Group. Infant and Young Child Feeding in Emergencies: Op-erational Guidance for Emergency Relief Staff and Programme Managers. Version 3.0—October 2017. Available online: www.ennonline.net/operationalguidance-v3-2017 (accessed on 15 August 2020).
- Gribble, K.; Preedy, V.R.; Watson, R.R.; Zibadi, S. 9. Formula feeding in emergencies. Hum. Health Handb. 2014, 8, 143–162. [Google Scholar] [CrossRef]
- Gribble, K.; Fernandes, C. Considerations regarding the use of infant formula products in infant and young child feeding in emergencies (IYCF-E) programs. World Nutr. 2018, 9, 261–283. [Google Scholar] [CrossRef]
- Hipgrave, D.B.; Assefa, F.; Winoto, A.; Sukotjo, S. Donated breast milk substitutes and incidence of diarrhoea among infants and young children after the May 2006 earthquake in Yogyakarta and Central Java. Public Health Nutr. 2011, 15, 307–315. [Google Scholar] [CrossRef] [PubMed]
- IBFAN-ICDC. Influx of Baby Food Supplies Swamped Central Sulawesi Emergency Camps in Indonesia. Baby Milk Action. 17 December 2018. Available online: https://www.babymilkaction.org/archives/19617 (accessed on 20 September 2020).
- Gribble, K.D. Media messages and the needs of infants and young children after Cyclone Nargis and the Wen Chuan Earthquake. Disasters 2012, 37, 80–100. [Google Scholar] [CrossRef]
- WHO, UNICEF, & IBFAN. Marketing of Breast-Milk Substitutes: National Implementation of the International Code. Status Report 2020. WHO. 2020. Available online: https://www.who.int/publications/i/item/9789240006010 (accessed on 21 August 2020).
- WHO. Guidance on Ending the Inappropriate Promotion of Foods for Infants and Young Children: Implementation Manual. WHO. 2017. Available online: https://www.who.int/nutrition/publications/infantfeeding/manual-ending-inappropriate-promotion-food/en/ (accessed on 20 September 2020).
- Brady, J.P.; Srour, L. India, Laos and South Africa Reject Sponsorship and Gifts from Formula Companies. Afr. Health Sci. 2014, 14, 211–215. [Google Scholar] [CrossRef] [PubMed]
- Wright, C.M. Relationships between paediatricians and infant formula milk companies. Arch. Dis. Child. 2006, 91, 383–385. [Google Scholar] [CrossRef]
- McFadden, A.; Kenney-Muir, N.; Whitford, H.; Renfrew, M. Breastfeeding: Policy Matter. Save the Children. 2015. Available online: https://resourcecentre.savethechildren.net/node/9442/pdf/breastfeeding_policy_matters.pdf (accessed on 1 October 2020).
- Mc Fadden, A.; Mason, F.; Baker, J.; Begin, F.; Dykes, F.; Grummer-Strawn, L.; Kenney-Muir, N.; Whitford, H.; Zehner, E.; Renfrew, M.J. Spotlight on infant formula: Coordinated global action needed. Lancet 2016, 387, 413–415. [Google Scholar] [CrossRef]
- Clark, D.L. Avoiding conflict of interest in the field of infant and young child nutrition: Better late than never. World Nutr. 2017, 8, 284–287. [Google Scholar] [CrossRef][Green Version]
- WHA (73rd). Maternal, Infant and Young Child Nutrition. Report by the Director-General (A73/4 Add.2). Geneva: WHO. 6 November 2020. Available online: https://apps.who.int/gb/ebwha/pdf_files/WHA73/A73_4Add2-en.pdf (accessed on 20 November 2020).
- Pereira-Kotze, C.; Doherty, T.; Swart, E.C. Use of social media platforms by manufacturers to market breast-milk substitutes in South Africa. BMJ Glob. Health 2020, 5, e003574. [Google Scholar] [CrossRef]
- IBFAN Asia & IBFAN-ICDC. Report on the Monitoring of the Code in 11 Countries of Asia. Breastfeeding Promotion Network in India (BPNI). 2018. Available online: https://www.bpni.org/wp-content/uploads/2018/12/Monitoring-of-the-Code-in-11-Countries-of-Asia.pdf (accessed on 30 August 2020).
- Hidayana, I.; Februhartanty, J.; Parady, V.A. Violations of the International Code of Marketing of Breast-milk Substitutes: Indonesia context. Public Health Nutr. 2017, 20, 165–173. [Google Scholar] [CrossRef] [PubMed]
- An Act Encouraging Corporate Social Responsibility, Providing Incentives Therefore, and for Other Purposes. Available online: https://www.congress.gov.ph/legisdocs/third_18/HBT6137.pdf (accessed on 12 October 2020).
- Breastmilk Substitutes Call to Action. Breastmilk Substitutes Call to Action—The Road to Code Compliance. 2020. Available online: https://www.bmscalltoaction.info/ (accessed on 27 January 2021).
- IBFAN Global Council. IBFAN’s Counter-Call to WHO and UNICEF to Stop 10-Year-Licence to Harm Children. Baby Milk Action. July 2020. Available online: http://www.babymilkaction.org/wp-content/uploads/2020/07/IBFANsCounterCall-final.pdf (accessed on 1 December 2020).
- Al-Othman, H. Food Banks’ Ban on Formula Leaves Babies to Go Hungry. The Sunday Times. 8 November 2020. Available online: https://www.thetimes.co.uk/article/food-banks-ban-on-formula-leaves-babies-to-go-hungry-tcghd25gm (accessed on 9 November 2020).
- IBFAN-ICDC. Code Monitoring Kit, 4th ed.; IBFAN-ICDC: Penang, Malaysia, 2019. [Google Scholar]






| Theme | Company | Country | Where Activity/Promotion Was Found |
|---|---|---|---|
| i. Unfounded health claims on immunity that prompt fear | Vitadairy | Vietnam | Online shopping portal |
| Nestlé | Pakistan | Social media (Facebook) | |
| ii. Association with public health authorities to gain legitimacy | Vitadairy | Vietnam | Social media (Facebook) |
| iii. Appeal to public sentiment on solidarity and hope | Reckitt Benckiser | China | Partner-NGO website |
| Abbott | China | Online news portal | |
| FrieslandCampina | China | Online news portal | |
| iv. Influx of donations of BMS products and supplies related to COVID-19 | Danone | Indonesia | Online news portal |
| Nestlé | Canada | Company website | |
| Feihe | China | Online news portal | |
| Danone | Malaysia | Company website | |
| Nestlé | Burkina Faso | Social media (Facebook) | |
| Nestlé | Burkina Faso | Online news portal | |
| v. Prominent use of digital platforms to reach out to parents | Reckitt Benckiser | China | Partner-NGO website |
| Feihe | China | Online news portal | |
| Danone | Indonesia | Social media (Instagram) | |
| Danone | Myanmar | Social media/Vlog (Facebook) | |
| Danone | Myanmar | Social media/Parenting group (Facebook) | |
| vi. Promoting uncertainty through endorsing breastfeeding | Nestlé | Singapore | Print magazine |
| Pigeon | India | Social media (Facebook) | |
| Medela | U.S. | Company website | |
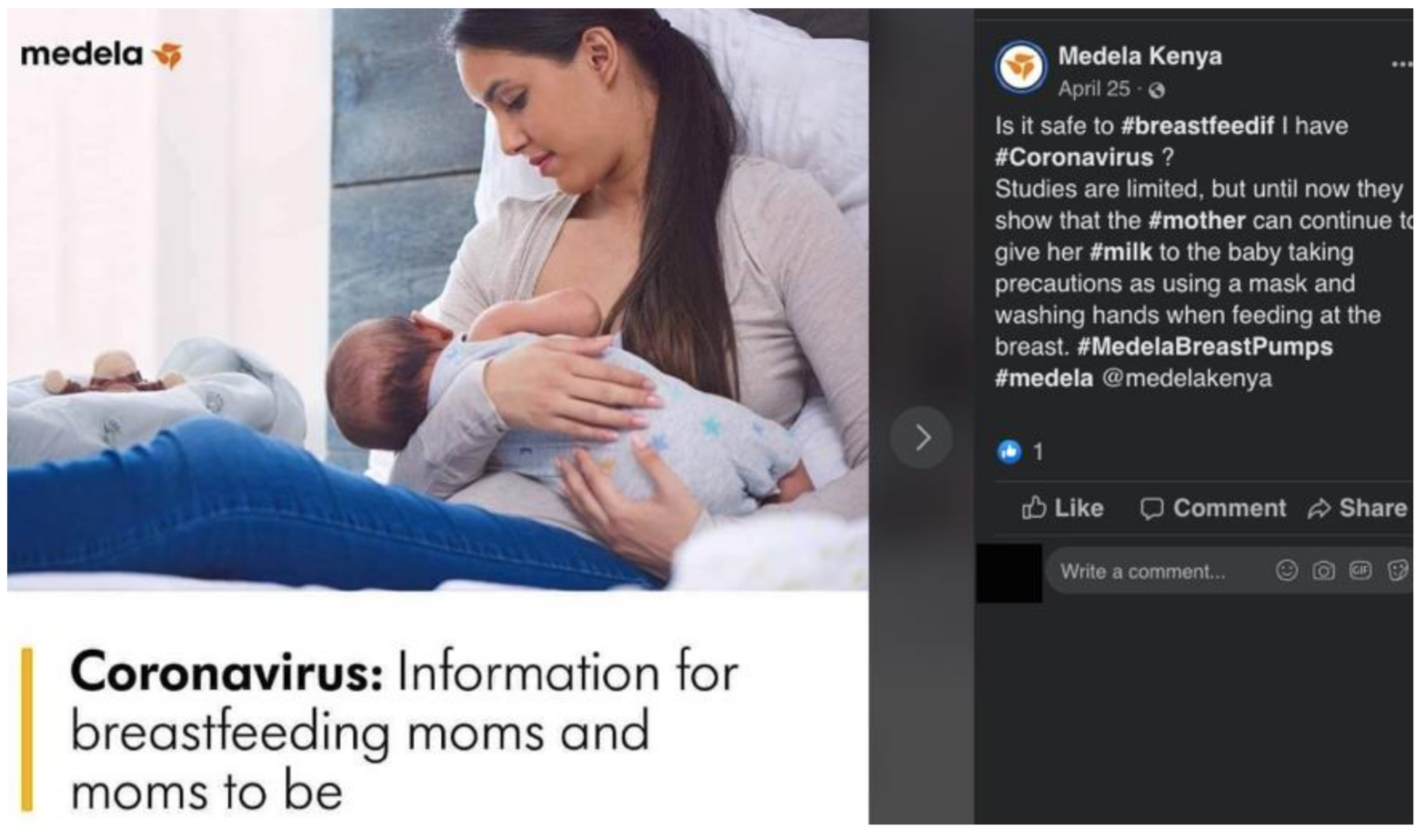
| Medela | Kenya | Social media (Facebook) | |
| Danone | Laos | Social media (Facebook) | |
| vii. Discounts on BMS products that are linked to COVID-19 | Nestlé | U.S. | Company website |
| Abbott | U.S. | Company website | |
| viii. Reaching out to health professionals through sponsoring educational events on topics relating to COVID-19 and infant and young child feeding | Nestlé | Kenya | Public health website (blog) |
| Abbott | The Philippines | Social media (Facebook) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ching, C.; Zambrano, P.; Nguyen, T.T.; Tharaney, M.; Zafimanjaka, M.G.; Mathisen, R. Old Tricks, New Opportunities: How Companies Violate the International Code of Marketing of Breast-Milk Substitutes and Undermine Maternal and Child Health during the COVID-19 Pandemic. Int. J. Environ. Res. Public Health 2021, 18, 2381. https://doi.org/10.3390/ijerph18052381
Ching C, Zambrano P, Nguyen TT, Tharaney M, Zafimanjaka MG, Mathisen R. Old Tricks, New Opportunities: How Companies Violate the International Code of Marketing of Breast-Milk Substitutes and Undermine Maternal and Child Health during the COVID-19 Pandemic. International Journal of Environmental Research and Public Health. 2021; 18(5):2381. https://doi.org/10.3390/ijerph18052381
Chicago/Turabian StyleChing, Constance, Paul Zambrano, Tuan T. Nguyen, Manisha Tharaney, Maurice Gerald Zafimanjaka, and Roger Mathisen. 2021. "Old Tricks, New Opportunities: How Companies Violate the International Code of Marketing of Breast-Milk Substitutes and Undermine Maternal and Child Health during the COVID-19 Pandemic" International Journal of Environmental Research and Public Health 18, no. 5: 2381. https://doi.org/10.3390/ijerph18052381
APA StyleChing, C., Zambrano, P., Nguyen, T. T., Tharaney, M., Zafimanjaka, M. G., & Mathisen, R. (2021). Old Tricks, New Opportunities: How Companies Violate the International Code of Marketing of Breast-Milk Substitutes and Undermine Maternal and Child Health during the COVID-19 Pandemic. International Journal of Environmental Research and Public Health, 18(5), 2381. https://doi.org/10.3390/ijerph18052381








