Nicotinic Acetylcholine Receptor Involvement in Inflammatory Bowel Disease and Interactions with Gut Microbiota
Abstract
1. Nicotinic Acetylcholine Receptors
1.1. Nicotinic Acetylcholine Receptors, Composition, Subtypes, and Pharmacological Properties
1.2. Nicotinic Acetylcholine Receptor Expression by Sensory and Autonomic Ganglion Neurons that Innervate the Gut
2. The Gut-Brain Axis
2.1. Neural Communication between the Brain and the Gut
2.2. Inflammatory Control in the Gut Involves the Vagus Nerve and α7 nAChRs
2.3. Nicotinic Acetylcholine Receptor Subunits α9 and α10 are Novel Players in IBD
3. Bacterial Types in the Gastrointestinal Tract
3.1. The Gut Microbiome Plays an Important Role in Communication between the Nervous System and the Gut
3.2. Gut Dysbiosis
3.3. Effects of nAChR Stimulation by Nicotine on Intestinal Microbiota Populations
4. Potential Involvement of nAChRs in COVID-19 and Associated Dysbiosis
The Pathophysiology of COVID-19 May Involve α7 nAChRs and Inhibition of the CAP
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Gotti, C.; Zoli, M.; Clementi, F. Brain nicotinic acetylcholine receptors: Native subtypes and their relevance. Trends Pharmacol. Sci. 2006, 27, 482–491. [Google Scholar] [CrossRef]
- Skok, V.I. Nicotinic acetylcholine receptors in autonomic ganglia. Auton. Neurosci. 2002, 97, 1–11. [Google Scholar] [CrossRef]
- Albuquerque, E.X.; Pereira, E.F.R.; Alkondon, M.; Rogers, S.W. Mammalian Nicotinic Acetylcholine Receptors: From Structure to Function. Physiol. Rev. 2009, 89, 73–120. [Google Scholar] [CrossRef]
- Dani, J.A. Neuronal Nicotinic Acetylcholine Receptor Structure and Function and Response to Nicotine. Int. Rev. Neurobiol. 2015, 124, 3–19. [Google Scholar] [CrossRef] [PubMed]
- De Biasi, M. Nicotinic mechanisms in the autonomic control of organ systems. J. Neurobiol. 2002, 53, 568–579. [Google Scholar] [CrossRef] [PubMed]
- Elgoyhen, A.B.; Vetter, D.E.; Katz, E.; Rothlin, C.V.; Heinemann, S.F.; Boulter, J. 10: A determinant of nicotinic cholinergic receptor function in mammalian vestibular and cochlear mechanosensory hair cells. Proc. Natl. Acad. Sci. USA 2001, 98, 3501–3506. [Google Scholar] [CrossRef] [PubMed]
- Lustig, L.R.; Peng, H.; Hiel, H.; Yamamoto, T.; Fuchs, P.A. Molecular Cloning and Mapping of the Human Nicotinic Acetylcholine Receptor α10 (CHRNA10). Genomics 2001, 73, 272–283. [Google Scholar] [CrossRef]
- Sgard, F.; Charpantier, E.; Bertrand, S.; Walker, N.; Caput, D.; Graham, D.; Bertrand, D.; Besnard, F. A Novel Human Nicotinic Receptor Subunit, α10, That Confers Functionality to the α9-Subunit. Mol. Pharmacol. 2002, 61, 150–159. [Google Scholar] [CrossRef]
- Keyser, K.; Britto, L.; Schoepfer, R.; Whiting, P.; Cooper, J.; Conroy, W.; Prechtl, A.B.-; Karten, H.; Lindstrom, J. Three subtypes of alpha-bungarotoxin-sensitive nicotinic acetylcholine receptors are expressed in chick retina. J. Neurosci. 1993, 13, 442–454. [Google Scholar] [CrossRef]
- Hone, A.J.; Rueda-Ruzafa, L.; Gordon, T.J.; Gajewiak, J.; Christensen, S.; Dyhring, T.; Albillos, A.; McIntosh, J.M. Expression of α3β2β4 nicotinic acetylcholine receptors by rat adrenal chromaffin cells determined using novel conopeptide antagonists. J. Neurochem. 2020, 154, 158–176. [Google Scholar] [CrossRef] [PubMed]
- David, R.; Ciuraszkiewicz, A.; Simeone, X.; Orr-Urtreger, A.; Papke, R.L.; McIntosh, J.M.; Huck, S.; Scholze, P. Biochemical and functional properties of distinct nicotinic acetylcholine receptors in the superior cervical ganglion of mice with targeted deletions of nAChR subunit genes. Eur. J. Neurosci. 2010, 31, 978–993. [Google Scholar] [CrossRef] [PubMed]
- Mao, D.; Yasuda, R.P.; Fan, H.; Wolfe, B.B.; Kellar, K.J. Heterogeneity of Nicotinic Cholinergic Receptors in Rat Superior Cervical and Nodose Ganglia. Mol. Pharmacol. 2006, 70, 1693–1699. [Google Scholar] [CrossRef] [PubMed]
- Elgoyhen, A.B.; Johnson, D.S.; Boulter, J.; Vetter, D.E.; Heinemann, S. α9: An acetylcholine receptor with novel pharmacological properties expressed in rat cochlear hair cells. Cell 1994, 79, 705–715. [Google Scholar] [CrossRef]
- Seguela, P.; Wadiche, J.; Dineley-Miller, K.; Dani, J.; Patrick, J.W. Molecular cloning, functional properties, and distribution of rat brain alpha 7: A nicotinic cation channel highly permeable to calcium. J. Neurosci. 1993, 13, 596–604. [Google Scholar] [CrossRef] [PubMed]
- Gerzanich, V.; Anand, R.; Lindstrom, J. Homomers of alpha 8 and alpha 7 subunits of nicotinic receptors exhibit similar channel but contrasting binding site properties. Mol. Pharmacol. 1994, 45, 212–220. [Google Scholar] [PubMed]
- Marcovich, I.; Moglie, M.J.; Freixas, A.E.C.; Trigila, A.P.; Franchini, L.F.; Plazas, P.V.; Lipovsek, M.; Elgoyhen, A.B. Distinct Evolutionary Trajectories of Neuronal and Hair Cell Nicotinic Acetylcholine Receptors. Mol. Biol. Evol. 2020, 37, 1070–1089. [Google Scholar] [CrossRef]
- Corringer, P.-J.; Bertrand, S.; Bohler, S.; Edelstein, S.J.; Changeux, J.-P.; Bertrand, D. Critical Elements Determining Diversity in Agonist Binding and Desensitization of Neuronal Nicotinic Acetylcholine Receptors. J. Neurosci. 1998, 18, 648–657. [Google Scholar] [CrossRef]
- Ragozzino, D.; Barabino, B.; Fucile, S.; Eusebi, F. Ca2+permeability of mouse and chick nicotinic acetylcholine receptors expressed in transiently transfected human cells. J. Physiol. 1998, 507, 749–758. [Google Scholar] [CrossRef]
- Fucile, S.; Sucapane, A.; Eusebi, F. Ca2+ permeability through rat cloned α9-containing nicotinic acetylcholine receptors. Cell Calcium 2006, 39, 349–355. [Google Scholar] [CrossRef]
- Ciuraszkiewicz, A.; Schreibmayer, W.; Platzer, D.; Orr-Urtreger, A.; Scholze, P.; Huck, S. Single-channel properties of α3β4, α3β4α5 and α3β4β2 nicotinic acetylcholine receptors in mice lacking specific nicotinic acetylcholine receptor subunits. J. Physiol. 2013, 591, 3271–3288. [Google Scholar] [CrossRef][Green Version]
- Parker, M.J.; Beck, A.; Luetje, C.W. Neuronal nicotinic receptor beta2 and beta4 subunits confer large differences in agonist binding affinity. Mol. Pharmacol. 1998, 54, 1132–1139. [Google Scholar] [CrossRef] [PubMed]
- Kuryatov, A.; Olale, F.; Cooper, J.; Choi, C.; Lindstrom, J. Human α6 AChR subtypes: Subunit composition, assembly, and pharmacological responses. Neuropharmacology 2000, 39, 2570–2590. [Google Scholar] [CrossRef]
- Alkondon, M.; Pereira, E.F.R.; Cartes, W.S.; Maelicke, A.; Albuquerque, E.X. Choline is a Selective Agonist of α7 Nicotinic Acetylcholine Receptors in the Rat Brain Neurons. Eur. J. Neurosci. 1997, 9, 2734–2742. [Google Scholar] [CrossRef] [PubMed]
- Papke, R.L.; McCormack, T.J.; Jack, B.A.; Wang, D.; Bugaj-Gaweda, B.; Schiff, H.C.; Buhr, J.D.; Waber, A.J.; Stokes, C. Rhesus monkey α7 nicotinic acetylcholine receptors: Comparisons to human α7 receptors expressed in Xenopus oocytes. Eur. J. Pharmacol. 2005, 524, 11–18. [Google Scholar] [CrossRef]
- Christensen, S.B.; Hone, A.J.; Roux, I.; Kniazeff, J.; Pin, J.-P.; Upert, G.; Servent, D.; Glowatzki, E.; McIntosh, J.M. RgIA4 Potently Blocks Mouse α9α10 nAChRs and Provides Long Lasting Protection against Oxaliplatin-Induced Cold Allodynia. Front. Cell. Neurosci. 2017, 11, 219. [Google Scholar] [CrossRef]
- Hone, A.J.; Meyer, E.L.; McIntyre, M.; McIntosh, J.M. Nicotinic acetylcholine receptors in dorsal root ganglion neurons include the α6β4 subtype. FASEB J. 2011, 26, 917–926. [Google Scholar] [CrossRef]
- Genzen, J.R.; Van Cleve, W.; McGehee, D.S. Dorsal Root Ganglion Neurons Express Multiple Nicotinic Acetylcholine Receptor Subtypes. J. Neurophysiol. 2001, 86, 1773–1782. [Google Scholar] [CrossRef]
- Rau, K.K.; Johnson, R.D.; Cooper, B.Y. Nicotinic AChR in Subclassified Capsaicin-Sensitive and -Insensitive Nociceptors of the Rat DRG. J. Neurophysiol. 2005, 93, 1358–1371. [Google Scholar] [CrossRef]
- Robinson, D.R.; McNaughton, P.A.; Evans, M.L.; Hicks, G.A. Characterization of the primary spinal afferent innervation of the mouse colon using retrograde labelling. Neurogastroenterol. Motil. 2004, 16, 113–124. [Google Scholar] [CrossRef]
- Abdrakhmanova, G.R.; AlSharari, S.; Kang, M.; Damaj, M.I.; Akbarali, H.I. α7-nAChR-mediated suppression of hyperexcitability of colonic dorsal root ganglia neurons in experimental colitis. Am. J. Physiol. Liver Physiol. 2010, 299, G761–G768. [Google Scholar] [CrossRef][Green Version]
- Smith, N.J.; Hone, A.J.; Memon, T.; Bossi, S.; Smith, T.E.; McIntosh, J.M.; Olivera, B.M.; Teichert, R.W. Comparative functional expression of nAChR subtypes in rodent DRG neurons. Front. Cell. Neurosci. 2013, 7, 225. [Google Scholar] [CrossRef] [PubMed]
- Wieskopf, J.S.; Mathur, J.; Limapichat, W.; Post, M.R.; Al-Qazzaz, M.; Sorge, R.E.; Martin, L.J.; Zaykin, D.V.; Smith, S.B.; Freitas, K.; et al. The nicotinic α6 subunit gene determines variability in chronic pain sensitivity via cross-inhibition of P2X2/3 receptors. Sci. Transl. Med. 2015, 7, 225. [Google Scholar] [CrossRef] [PubMed]
- Spies, M.; Lips, K.S.; Kurzen, H.; Kummer, W.; Haberberger, R.V. Nicotinic Acetylcholine Receptors Containing Subunits α3 and α5 in Rat Nociceptive Dorsal Root Ganglion Neurons. J. Mol. Neurosci. 2006, 30, 55–56. [Google Scholar] [CrossRef]
- Genzen, J.R.; McGehee, D.S. Short- and long-term enhancement of excitatory transmission in the spinal cord dorsal horn by nicotinic acetylcholine receptors. Proc. Natl. Acad. Sci. USA 2003, 100, 6807–6812. [Google Scholar] [CrossRef] [PubMed]
- De Biasi, M.; Nigro, F.; Xu, W. Nicotinic acetylcholine receptors in the autonomic control of bladder function. Eur. J. Pharmacol. 2000, 393, 137–140. [Google Scholar] [CrossRef]
- Xu, W.; Orr-Urtreger, A.; Nigro, F.; Gelber, S.; Sutcliffe, C.B.; Armstrong, D.; Patrick, J.W.; Role, L.W.; Beaudet, A.L.; De Biasi, M. Multiorgan autonomic dysfunction in mice lacking the beta2 and the beta4 subunits of neuronal nicotinic acetylcholine receptors. J. Neurosci. 1999, 19, 9298–9305. [Google Scholar] [CrossRef]
- Zhou, X.; Ren, J.; Brown, E.; Schneider, D.A.; Caraballo-Lopez, Y.; Galligan, J.J. Pharmacological Properties of Nicotinic Acetylcholine Receptors Expressed by Guinea Pig Small Intestinal Myenteric Neurons. J. Pharmacol. Exp. Ther. 2002, 302, 889–897. [Google Scholar] [CrossRef]
- Garza, A.; Huang, L.Z.; Son, J.-H.; Winzer-Serhan, U.H. Expression of nicotinic acetylcholine receptors and subunit messenger RNAs in the enteric nervous system of the neonatal rat. Neuroscience 2009, 158, 1521–1529. [Google Scholar] [CrossRef]
- Obaid, A.; Nelson, M.E.; Lindström, J.; Salzberg, B.M. Optical studies of nicotinic acetylcholine receptor subtypes in the guinea-pig enteric nervous system. J. Exp. Biol. 2005, 208, 2981–3001. [Google Scholar] [CrossRef][Green Version]
- Foong, J.P.P.; Hirst, C.S.; Hao, M.M.; McKeown, S.J.; Boesmans, W.; Young, H.M.; Bornstein, J.C.; Berghe, P.V. Changes in Nicotinic Neurotransmission during Enteric Nervous System Development. J. Neurosci. 2015, 35, 7106–7115. [Google Scholar] [CrossRef]
- Glushakov, A.V.; Voytenko, L.P.; Skok, M.V.; Skok, V. Distribution of neuronal nicotinic acetylcholine receptors containing different alpha-subunits in the submucosal plexus of the guinea-pig. Auton. Neurosci. 2004, 110, 19–26. [Google Scholar] [CrossRef] [PubMed]
- MacEachern, S.J.; Patel, B.A.; McKay, D.M.; Sharkey, K. Nitric oxide regulation of colonic epithelial ion transport: A novel role for enteric glia in the myenteric plexus. J. Physiol. 2011, 589, 3333–3348. [Google Scholar] [CrossRef] [PubMed]
- Lai, N.Y.; Mills, K.; Chiu, I.M. Sensory neuron regulation of gastrointestinal inflammation and bacterial host defence. J. Intern. Med. 2017, 282, 5–23. [Google Scholar] [CrossRef] [PubMed]
- Keiger, C.H.; Walker, J.C. Individual variation in the expression profiles of nicotinic receptors in the olfactory bulb and trigeminal ganglion and identification of α2, α6, α9, and β3 transcripts. Biochem. Pharmacol. 2000, 59, 233–240. [Google Scholar] [CrossRef]
- Zhang, X.; Hartung, J.E.; Friedman, R.L.; Koerber, H.R.; Belfer, I.; Gold, M.S. Nicotine Evoked Currents in Human Primary Sensory Neurons. J. Pain 2019, 20, 810–818. [Google Scholar] [CrossRef] [PubMed]
- Ray, P.R.; Torck, A.; Quigley, L.; Wangzhou, A.; Neiman, M.; Rao, C.; Lam, T.; Kim, J.-Y.; Kim, T.H.; Zhang, M.Q.; et al. Comparative transcriptome profiling of the human and mouse dorsal root ganglia. Pain 2018, 159, 1325–1345. [Google Scholar] [CrossRef]
- Downs, A.; Bond, C.; Hoover, D.B. Localization of α7 nicotinic acetylcholine receptor mRNA and protein within the cholinergic anti-inflammatory pathway. Neuroscience 2014, 266, 178–185. [Google Scholar] [CrossRef]
- Mundinger, T.O.; Mei, Q.; Taborsky, G.J., Jr. Impaired activation of celiac ganglion neurons in vivo after damage to their sympathetic nerve terminals. J. Neurosci. Res. 2008, 86, 1981–1993. [Google Scholar] [CrossRef][Green Version]
- Koval, O.M.; Voitenko, L.P.; Skok, M.V.; Lykhmus, E.Y.; Tsetlin, V.; Zhmak, M.N.; Skok, V. The β-subunit composition of nicotinic acetylcholine receptors in the neurons of the guinea pig inferior mesenteric ganglion. Neurosci. Lett. 2004, 365, 143–146. [Google Scholar] [CrossRef]
- Bentley, G.A. Pharmacological studies on the hypogastric ganglion of the rat and guinea-pig. Br. J. Pharmacol. 1972, 44, 492–509. [Google Scholar] [CrossRef]
- Girard, B.M.; Merriam, L.A.; Tompkins, J.D.; Vizzard, M.A.; Parsons, R.L. Decrease in neuronal nicotinic acetylcholine receptor subunit and PSD-93 transcript levels in the male mouse MPG after cavernous nerve injury or explant culture. Am. J. Physiol. Physiol. 2013, 305, F1504–F1512. [Google Scholar] [CrossRef] [PubMed]
- Kirchgessner, A.L.; Liu, M.T. Immunohistochemical localization of nicotinic acetylcholine receptors in the guinea pig bowel and pancreas. J. Comp. Neurol. 1998, 390, 497–514. [Google Scholar] [CrossRef]
- Barrenschee, M.; Cossais, F.; Böttner, M.; Egberts, J.-H.; Becker, T.; Wedel, T. Impaired Expression of Neuregulin 1 and Nicotinic Acetylcholine Receptor β4 Subunit in Diverticular Disease. Front. Cell. Neurosci. 2019, 13, 563. [Google Scholar] [CrossRef] [PubMed]
- Vernino, S.; Low, P.A.; Fealey, R.D.; Stewart, J.D.; Farrugia, G.; Lennon, V.A. Autoantibodies to Ganglionic Acetylcholine Receptors in Autoimmune Autonomic Neuropathies. N. Engl. J. Med. 2000, 343, 847–855. [Google Scholar] [CrossRef]
- Vernino, S.; Hopkins, S.; Wang, Z. Autonomic ganglia, acetylcholine receptor antibodies, and autoimmune ganglionopathy. Auton. Neurosci. 2009, 146, 3–7. [Google Scholar] [CrossRef]
- Richardson, C.E.; Morgan, J.M.; Jasani, B.; Green, J.T.; Rhodes, J.; Williams, G.T.; Lindstrom, J.; Wonnacott, S.; Thomas, G.A.; Smith, V. Megacystis-microcolon-intestinal hypoperistalsis syndrome and the absence of the α3 nicotinic acetylcholine receptor subunit. Gastroenterology 2001, 121, 350–357. [Google Scholar] [CrossRef]
- Abdullah, N.; Defaye, M.; Altier, C. Neural control of gut homeostasis. Am. J. Physiol. Liver Physiol. 2020, 319, G718–G732. [Google Scholar] [CrossRef]
- Breit, S.; Kupferberg, A.; Rogler, G.; Hasler, G. Vagus Nerve as Modulator of the Brain–Gut Axis in Psychiatric and Inflammatory Disorders. Front. Psychiatry 2018, 9, 44. [Google Scholar] [CrossRef]
- Kruepunga, N.; Hikspoors, J.P.J.M.; Hülsman, C.J.M.; Mommen, G.M.C.; Köhler, S.E.; Lamers, W.H. Development of extrinsic innervation in the abdominal intestines of human embryos. J. Anat. 2020, 237, 655–671. [Google Scholar] [CrossRef]
- Berthoud, H.-R.; Neuhuber, W.L. Functional and chemical anatomy of the afferent vagal system. Auton. Neurosci. 2000, 85, 1–17. [Google Scholar] [CrossRef]
- Tracey, K.J. The inflammatory reflex. Nature 2002, 420, 853–859. [Google Scholar] [CrossRef] [PubMed]
- Rosas-Ballina, M.; Ochani, M.; Parrish, W.R.; Ochani, K.; Harris, Y.T.; Huston, J.M.; Chavan, S.; Tracey, K.J. Splenic nerve is required for cholinergic antiinflammatory pathway control of TNF in endotoxemia. Proc. Natl. Acad. Sci. USA 2008, 105, 11008–11013. [Google Scholar] [CrossRef] [PubMed]
- Rosas-Ballina, M.; Olofsson, P.S.; Ochani, M.; Valdés-Ferrer, S.I.; Levine, Y.A.; Reardon, C.; Tusche, M.W.; Pavlov, V.A.; Andersson, U.; Chavan, S.; et al. Acetylcholine-Synthesizing T Cells Relay Neural Signals in a Vagus Nerve Circuit. Science 2011, 334, 98–101. [Google Scholar] [CrossRef] [PubMed]
- Nezami, B.G.; Srinivasan, S. Enteric Nervous System in the Small Intestine: Pathophysiology and Clinical Implications. Curr. Gastroenterol. Rep. 2010, 12, 358–365. [Google Scholar] [CrossRef]
- Metz, C.N.; Pavlov, V.A. Vagus nerve cholinergic circuitry to the liver and the gastrointestinal tract in the neuroimmune communicatome. Am. J. Physiol. Liver Physiol. 2018, 315, G651–G658. [Google Scholar] [CrossRef]
- Arredondo, J.; Chernyavsky, A.I.; Jolkovsky, D.L.; Pinkerton, K.E.; Grando, S.A. Receptor-mediated tobacco toxicity: Cooperation of the Ras/Raf-1/MEK1/ERK and JAK-2/STAT-3 pathways downstream of a7 nicotinic receptor in oral keratinocytes. FASEB J. 2006, 20, 2093–2101. [Google Scholar] [CrossRef]
- De Jonge, W.J.; Van Der Zanden, E.P.; The, F.O.; Bijlsma, M.F.; Van Westerloo, D.J.; Bennink, R.J.; Berthoud, H.-R.; Uematsu, S.; Akira, S.; Wijngaard, R.M.V.D.; et al. Stimulation of the vagus nerve attenuates macrophage activation by activating the Jak2-STAT3 signaling pathway. Nat. Immunol. 2005, 6, 844–851. [Google Scholar] [CrossRef]
- Hoentjen, F.; Sartor, R.B.; Ozaki, M.; Jobin, C. STAT3 regulates NF-κB recruitment to the IL-12p40 promoter in dendritic cells. Blood 2005, 105, 689–696. [Google Scholar] [CrossRef]
- Yoshida, Y.; Kumar, A.; Koyama, Y.; Peng, H.; Arman, A.; Boch, J.A.; Auron, P.E. Interleukin 1 Activates STAT3/Nuclear Factor-κB Cross-talk via a Unique TRAF6- and p65-dependent Mechanism. J. Biol. Chem. 2004, 279, 1768–1776. [Google Scholar] [CrossRef]
- Tyagi, E.; Agrawal, R.; Nath, C.; Shukla, R. Cholinergic protection via α7 nicotinic acetylcholine receptors and PI3K-Akt pathway in LPS-induced neuroinflammation. Neurochem. Int. 2010, 56, 135–142. [Google Scholar] [CrossRef]
- Kim, T.-H.; Kim, S.-J.; Lee, S.-M. Stimulation of the α7 Nicotinic Acetylcholine Receptor Protects against Sepsis by Inhibiting Toll-like Receptor via Phosphoinositide 3-Kinase Activation. J. Infect. Dis. 2013, 209, 1668–1677. [Google Scholar] [CrossRef] [PubMed]
- Lu, B.; Kwan, K.; Levine, Y.A.; Olofsson, P.S.; Yang, H.; Li, J.; Joshi, S.; Wang, H.; Andersson, U.; Chavan, S.S.; et al. α7 Nicotinic Acetylcholine Receptor Signaling Inhibits Inflammasome Activation by Preventing Mitochondrial DNA Release. Mol. Med. 2014, 20, 350–358. [Google Scholar] [CrossRef] [PubMed]
- Borovikova, L.V.; Ivanova, S.; Zhang, M.; Yang, H.; Botchkina, G.I.; Watkins, L.R.; Wang, H.; Abumrad, N.N.; Eaton, J.W.; Tracey, K.J. Vagus nerve stimulation attenuates the systemic inflammatory response to endotoxin. Nat. Cell Biol. 2000, 405, 458–462. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Yu, M.; Ochani, M.; Amella, C.A.; Tanovic, M.; Susarla, S.; Li, J.H.; Wang, H.; Yang, H.; Ulloa, L.; et al. Nicotinic acetylcholine receptor α7 subunit is an essential regulator of inflammation. Nat. Cell Biol. 2002, 421, 384–388. [Google Scholar] [CrossRef]
- Parrish, W.R.; Czura, C.J.; Tracey, K.J.; Puerta, M. Experimental Therapeutic Strategies for Severe Sepsis. Ann. N. Y. Acad. Sci. 2008, 1144, 210–236. [Google Scholar] [CrossRef]
- Pavlov, V.A.; Ochani, M.; Yang, L.-H.; Gallowitsch-Puerta, M.; Ochani, K.; Lin, X.; Levi, J.; Parrish, W.R.; Rosas-Ballina, M.; Czura, C.J.; et al. Selective α7-nicotinic acetylcholine receptor agonist GTS-21 improves survival in murine endotoxemia and severe sepsis. Crit. Care Med. 2007, 35, 1139–1144. [Google Scholar] [CrossRef]
- Wang, H.; Liao, H.; Ochani, M.; Justiniani, M.; Lin, X.; Yang, L.; Al-Abed, Y.; Wang, H.; Metz, C.N.; Miller, E.J.; et al. Cholinergic agonists inhibit HMGB1 release and improve survival in experimental sepsis. Nat. Med. 2004, 10, 1216–1221. [Google Scholar] [CrossRef]
- Tsoyi, K.; Jang, H.J.; Kim, J.W.; Chang, H.K.; Lee, Y.S.; Pae, H.-O.; Kim, H.J.; Seo, H.G.; Lee, J.H.; Chung, H.-T.; et al. Stimulation of Alpha7 Nicotinic Acetylcholine Receptor by Nicotine Attenuates Inflammatory Response in Macrophages and Improves Survival in Experimental Model of Sepsis Through Heme Oxygenase-1 Induction. Antioxid. Redox Signal. 2011, 14, 2057–2070. [Google Scholar] [CrossRef]
- Olofsson, P.S.; Rosas-Ballina, M.; Levine, Y.A.; Tracey, K.J. Rethinking inflammation: Neural circuits in the regulation of immunity. Immunol. Rev. 2012, 248, 188–204. [Google Scholar] [CrossRef]
- Zhao, Y.X.; He, W.; Jing, X.H.; Liu, J.L.; Rong, P.J.; Ben, H.; Liu, K.; Zhu, B. Transcutaneous Auricular Vagus Nerve Stimulation Protects Endotoxemic Rat from Lipopolysaccharide-Induced Inflammation. Evid. Based Complement. Altern. Med. 2012, 2012, 1–10. [Google Scholar] [CrossRef]
- Maldifassi, M.C.; Martín-Sánchez, C.; Atienza, G.; Cedillo, J.L.; Arnalich, F.; Bordas, A.; Zafra, F.; Giménez, C.; Extremera, M.; Renart, J.; et al. Interaction of the α7-nicotinic subunit with its human-specific duplicated dupα7 isoform in mammalian cells: Relevance in human inflammatory responses. J. Biol. Chem. 2018, 293, 13874–13888. [Google Scholar] [CrossRef] [PubMed]
- Baird, A.; Coimbra, R.; Dang, X.; Eliceiri, B.P.; Costantini, T.W. Up-regulation of the human-specific CHRFAM7A gene in inflammatory bowel disease. BBA Clin. 2016, 5, 66–71. [Google Scholar] [CrossRef] [PubMed]
- Kessler, W.; Diedrich, S.; Menges, P.; Ebker, T.; Nielson, M.; Partecke, L.I.; Traeger, T.; Cziupka, K.; Van Der Linde, J.; Puls, R.; et al. The Role of the Vagus Nerve: Modulation of the Inflammatory Reaction in Murine Polymicrobial Sepsis. Mediat. Inflamm. 2012, 2012, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Levy, G.; Fishman, J.; Xu, D.; Chandler, B.T.J.; Feketova, E.; Dong, W.; Qin, Y.; Alli, V.; Ulloa, L.; Deitch, E.A. Parasympathetic Stimulation Via the Vagus Nerve Prevents Systemic Organ Dysfunction by Abrogating Gut Injury and Lymph Toxicity in Trauma and Hemorrhagic Shock. Shock 2013, 39, 39–44. [Google Scholar] [CrossRef]
- Li, T.; Zuo, X.; Zhou, Y.; Wang, Y.; Zhuang, H.; Zhang, L.; Zhang, H.; Xiao, X. The Vagus Nerve and Nicotinic Receptors Involve Inhibition of HMGB1 Release and Early Pro-inflammatory Cytokines Function in Collagen-Induced Arthritis. J. Clin. Immunol. 2009, 30, 213–220. [Google Scholar] [CrossRef]
- Meregnani, J.; Clarencon, D.; Vivier, M.; Peinnequin, A.; Mouret, C.; Sinniger, V.; Picq, C.; Job, A.; Canini, F.; Jacquier-Sarlin, M.; et al. Anti-inflammatory effect of vagus nerve stimulation in a rat model of inflammatory bowel disease. Auton. Neurosci. 2011, 160, 82–89. [Google Scholar] [CrossRef]
- Van Westerloo, D.J.; Giebelen, I.A.; Florquin, S.; Bruno, M.J.; LaRosa, G.J.; Ulloa, L.; Tracey, K.J.; Van Der Poll, T. The Vagus Nerve and Nicotinic Receptors Modulate Experimental Pancreatitis Severity in Mice. Gastroenterology 2006, 130, 1822–1830. [Google Scholar] [CrossRef]
- Schneider, L.; Jabrailova, B.; Soliman, H.; Hofer, S.; Strobel, O.; Hackert, T.; Büchler, M.W.; Werner, J. Pharmacological Cholinergic Stimulation as a Therapeutic Tool in Experimental Necrotizing Pancreatitis. Pancreas 2014, 43, 41–46. [Google Scholar] [CrossRef]
- Feng, X.; Li, L.; Feng, J.; He, W.; Li, N.; Shi, T.; Jie, Z.; Su, X. Vagal-α7nAChR signaling attenuates allergic asthma responses and facilitates asthma tolerance by regulating inflammatory group 2 innate lymphoid cells. Immunol. Cell Biol. 2020. [Google Scholar] [CrossRef]
- Kox, M.; Pompe, J.C.; De Gouberville, M.C.G.; Van Der Hoeven, J.G.; Hoedemaekers, C.W.E.; Pickkers, P. Effects of the α7 Nicotinic Acetylcholine Receptor Agonist Gts-21 on the Innate Immune Response in Humans. Shock 2011, 36, 5–11. [Google Scholar] [CrossRef]
- Wittebole, X.; Hahm, S.; Coyle, S.M.; Kumar, A.; Calvano, S.E.; Lowry, S.F. Nicotine exposure alters in vivo human responses to endotoxin. Clin. Exp. Immunol. 2006, 147, 28–34. [Google Scholar] [CrossRef] [PubMed]
- Cedillo, J.L.; Arnalich, F.; Martín-Sánchez, C.; Quesada, A.; Rios, J.J.; Maldifassi, M.C.; Atienza, G.; Renart, J.; Fernández, F.A.; García-Rio, F.; et al. Usefulness of α7 Nicotinic Receptor Messenger RNA Levels in Peripheral Blood Mononuclear Cells as a Marker for Cholinergic Antiinflammatory Pathway Activity in Septic Patients: Results of a Pilot Study. J. Infect. Dis. 2014, 211, 146–155. [Google Scholar] [CrossRef] [PubMed]
- Nicholson, W.; Kempf, M.-C.; Moneyham, L.; Vance, D.E. The potential role of vagus-nerve stimulation in the treatment of HIV-associated depression: A review of literature. Neuropsychiatr. Dis. Treat. 2017, 13, 1677–1689. [Google Scholar] [CrossRef] [PubMed]
- Toffa, D.H.; Touma, L.; El Meskine, T.; Bouthillier, A.; Nguyen, D.K. Learnings from 30 years of reported efficacy and safety of vagus nerve stimulation (VNS) for epilepsy treatment: A critical review. Seizure 2020, 83, 104–123. [Google Scholar] [CrossRef]
- Jiang, W.-; Zhang, C.; Wang, J.-X.; Sun, F.-H.; Xie, Y.-J.; Ou, X.; Yang, S.-B. The effect of VNS on the rehabilitation of stroke: A meta-analysis of randomized controlled studies. J. Clin. Neurosci. 2020, 81, 421–425. [Google Scholar] [CrossRef]
- Lendvai, I.S.; Maier, A.; Scheele, D.; Hurlemann, R.; Kinfe, T.M. Spotlight on cervical vagus nerve stimulation for the treatment of primary headache disorders: A review. J. Pain Res. 2018, 11, 1613–1625. [Google Scholar] [CrossRef]
- Bonaz, B.; Sinniger, V.; Hoffmann, D.; Clarençon, D.; Mathieu, N.; Dantzer, C.; Vercueil, L.; Picq, C.; Trocmé, C.; Faure, P.; et al. Chronic vagus nerve stimulation in Crohn’s disease: A 6-month follow-up pilot study. Neurogastroenterol. Motil. 2016, 28, 948–953. [Google Scholar] [CrossRef]
- Ghia, J.E.; Blennerhassett, P.; Kumar–Ondiveeran, H.; Verdu, E.F.; Collins, S.M. The Vagus Nerve: A Tonic Inhibitory Influence Associated With Inflammatory Bowel Disease in a Murine Model. Gastroenterology 2006, 131, 1122–1130. [Google Scholar] [CrossRef]
- The, F.O.; Boeckxstaens, G.E.; Snoek, S.A.; Cash, J.L.; Bennink, R.; LaRosa, G.J.; Wijngaard, R.M.V.D.; Greaves, D.R.; De Jonge, W.J. Activation of the Cholinergic Anti-Inflammatory Pathway Ameliorates Postoperative Ileus in Mice. Gastroenterology 2007, 133, 1219–1228. [Google Scholar] [CrossRef]
- Yu, Y.R.; Rodriguez, J.R. Clinical presentation of Crohn’s, ulcerative colitis, and indeterminate colitis: Symptoms, extraintestinal manifestations, and disease phenotypes. Semin. Pediatr. Surg. 2017, 26, 349–355. [Google Scholar] [CrossRef]
- Matteoli, G.; Boeckxstaens, G.E. The vagal innervation of the gut and immune homeostasis. Gut 2012, 62, 1214–1222. [Google Scholar] [CrossRef] [PubMed]
- Cailotto, C.; Gomez-Pinilla, P.J.; Costes, L.M.; Van Der Vliet, J.; Di Giovangiulio, M.; Nemethova, A.; Matteoli, G.; Boeckxstaens, G.E. Neuro-Anatomical Evidence Indicating Indirect Modulation of Macrophages by Vagal Efferents in the Intestine but Not in the Spleen. PLoS ONE 2014, 9, e87785. [Google Scholar] [CrossRef] [PubMed]
- Ahluwalia, B.; Moraes, L.; Magnusson, M.K.; Öhman, L. Immunopathogenesis of inflammatory bowel disease and mechanisms of biological therapies. Scand. J. Gastroenterol. 2018, 53, 379–389. [Google Scholar] [CrossRef] [PubMed]
- Maruta, K.; Watanabe, C.; Hozumi, H.; Kurihara, C.; Furuhashi, H.; Takajo, T.; Okada, Y.; Shirakabe, K.; Higashiyama, M.; Komoto, S.; et al. Nicotine treatment ameliorates DSS-induced colitis by suppressing MAdCAM-1 expression and leukocyte recruitment. J. Leukoc. Biol. 2018, 104, 1013–1022. [Google Scholar] [CrossRef] [PubMed]
- Abdrakhmanova, G.R.; Kang, M.; Damaj, M.I.; Akbarali, H.I. Nicotine suppresses hyperexcitability of colonic sensory neurons and visceral hypersensivity in mouse model of colonic inflammation. Am. J. Physiol. Liver Physiol. 2012, 302, G740–G747. [Google Scholar] [CrossRef] [PubMed]
- Zhou, H.; Liang, H.; Li, Z.-F.; Xiang, H.; Liu, W.; Li, J.-G. Vagus Nerve Stimulation Attenuates Intestinal Epithelial Tight Junctions Disruption in Endotoxemic Mice Through α7 Nicotinic Acetylcholine Receptors. Shock 2013, 40, 144–151. [Google Scholar] [CrossRef]
- Costantini, T.W.; Krzyzaniak, M.; Cheadle, G.A.; Putnam, J.G.; Hageny, A.-M.; Lopez, N.; Eliceiri, B.P.; Bansal, V.; Coimbra, R. Targeting α-7 Nicotinic Acetylcholine Receptor in the Enteric Nervous System. Am. J. Pathol. 2012, 181, 478–486. [Google Scholar] [CrossRef]
- Wang, R.; Li, S.; Jin, L.; Zhang, W.; Liu, N.; Wang, H.; Wang, Z.; Wei, P.; Li, F.; Yu, J.; et al. Four-week administration of nicotine moderately impacts blood metabolic profile and gut microbiota in a diet-dependent manner. Biomed. Pharmacother. 2019, 115, 108945. [Google Scholar] [CrossRef]
- Wazea, S.A.; Wadie, W.; Bahgat, A.K.; El-Abhar, H.S. Galantamine anti-colitic effect: Role of alpha-7 nicotinic acetylcholine receptor in modulating Jak/STAT3, NF-κB/HMGB1/RAGE and p-AKT/Bcl-2 pathways. Sci. Rep. 2018, 8, 5110. [Google Scholar] [CrossRef]
- Tasaka, Y.; Yasunaga, D.; Kiyoi, T.; Tanaka, M.; Tanaka, A.; Suemaru, K.; Araki, H. Involvement of stimulation of α7 nicotinic acetylcholine receptors in the suppressive effect of tropisetron on dextran sulfate sodium-induced colitis in mice. J. Pharmacol. Sci. 2015, 127, 275–283. [Google Scholar] [CrossRef][Green Version]
- Regmi, B.; Shah, M.K. Possible implications of animal models for the assessment of visceral pain. Anim. Model. Exp. Med. 2020, 3, 215–228. [Google Scholar] [CrossRef] [PubMed]
- Salaga, M.; Blomster, L.V.; Czyk, A.P.-P.; Zielinska, M.; Jacenik, D.; Cygankiewicz, A.; Krajewska, W.M.; Mikkelsen, J.D.; Fichna, J.; Piechota-Polanczyk, A. Encenicline, a 7 nicotinic acetylcholine receptor partial agonist, reduces immune cell infiltration in the colon and improves experimental colitis in mice. J. Pharmacol. Exp. Ther. 2015, 356, 157–169. [Google Scholar] [CrossRef] [PubMed]
- Da Costa, R.; Motta, E.M.; Manjavachi, M.N.; Cola, M.; Calixto, J.B. Activation of the alpha-7 nicotinic acetylcholine receptor (α7 nAchR) reverses referred mechanical hyperalgesia induced by colonic inflammation in mice. Neuropharmacology 2012, 63, 798–805. [Google Scholar] [CrossRef] [PubMed]
- Van Der Zanden, E.P.; Snoek, S.A.; Heinsbroek, S.E.; Stanisor, O.I.; Verseijden, C.; Boeckxstaens, G.E.; Peppelenbosch, M.P.; Greaves, D.R.; Gordon, S.; De Jonge, W.J. Vagus Nerve Activity Augments Intestinal Macrophage Phagocytosis via Nicotinic Acetylcholine Receptor α4β2. Gastroenterology 2009, 137, 1029–1039.e4. [Google Scholar] [CrossRef] [PubMed]
- Xiao, J.; Zhang, G.; Gao, S.; Shen, J.; Feng, H.; He, Z.; Xu, C. Combined administration of SHP2 inhibitor SHP099 and the α7nAChR agonist PNU282987 protect mice against DSS-induced colitis. Mol. Med. Rep. 2020, 22, 2235–2244. [Google Scholar] [CrossRef] [PubMed]
- AlZarea, S.; Rahman, S. Alpha-7 nicotinic receptor allosteric modulator PNU120596 prevents lipopolysaccharide-induced anxiety, cognitive deficit and depression-like behaviors in mice. Behav. Brain Res. 2019, 366, 19–28. [Google Scholar] [CrossRef]
- Engler, H.; Elsenbruch, S.; Rebernik, L.; Köcke, J.; Cramer, H.; Schöls, M.; Langhorst, J. Stress burden and neuroendocrine regulation of cytokine production in patients with ulcerative colitis in remission. Psychoneuroendocrinology 2018, 98, 101–107. [Google Scholar] [CrossRef]
- AlSharari, S.D.; Toma, W.; Mahmood, H.M.; McIntosh, J.M.; Damaj, M.I. The α9α10 nicotinic acetylcholine receptors antagonist α-conotoxin RgIA reverses colitis signs in murine dextran sodium sulfate model. Eur. J. Pharmacol. 2020, 883, 173320. [Google Scholar] [CrossRef]
- Romero, H.K.; Christensen, S.B.; Mannelli, L.D.C.; Gajewiak, J.; Ramachandra, R.; Elmslie, K.S.; Vetter, D.E.; Ghelardini, C.; Iadonato, S.P.; Mercado, J.L.; et al. Inhibition of α9α10 nicotinic acetylcholine receptors prevents chemotherapy-induced neuropathic pain. Proc. Natl. Acad. Sci. USA 2017, 114, E1825–E1832. [Google Scholar] [CrossRef]
- Mannelli, L.D.C.; Cinci, L.; Micheli, L.; Zanardelli, M.; Pacini, A.; McIntosh, M.J.; Ghelardini, C. α-Conotoxin RgIA protects against the development of nerve injury-induced chronic pain and prevents both neuronal and glial derangement. Pain 2014, 155, 1986–1995. [Google Scholar] [CrossRef]
- Huynh, P.N.; Giuvelis, D.; Christensen, S.B.; Tucker, K.L.; McIntosh, J.M. RgIA4 Accelerates Recovery from Paclitaxel-Induced Neuropathic Pain in Rats. Mar. Drugs 2019, 18, 12. [Google Scholar] [CrossRef] [PubMed]
- Pacini, A.; Micheli, L.; Maresca, M.; Branca, J.J.V.; McIntosh, J.M.; Ghelardini, C.; Mannelli, L.D.C. The α9α10 nicotinic receptor antagonist α-conotoxin RgIA prevents neuropathic pain induced by oxaliplatin treatment. Exp. Neurol. 2016, 282, 37–48. [Google Scholar] [CrossRef] [PubMed]
- Zakrzewicz, A.; Richter, K.; Agné, A.; Wilker, S.; Siebers, K.; Fink, B.; Krasteva-Christ, G.; Althaus, M.; Padberg, W.; Hone, A.J.; et al. Canonical and Novel Non-Canonical Cholinergic Agonists Inhibit ATP-Induced Release of Monocytic Interleukin-1β via Different Combinations of Nicotinic Acetylcholine Receptor Subunits α7, α9 and α10. Front. Cell. Neurosci. 2017, 11, 189. [Google Scholar] [CrossRef] [PubMed]
- Richter, K.; Mathes, V.; Fronius, M.; Althaus, M.; Hecker, A.; Krasteva-Christ, G.; Padberg, W.; Hone, A.J.; McIntosh, J.M.; Zakrzewicz, A.; et al. Phosphocholine—An agonist of metabotropic but not of ionotropic functions of α9-containing nicotinic acetylcholine receptors. Sci. Rep. 2016, 6, 28660. [Google Scholar] [CrossRef] [PubMed]
- Hecker, A.; Küllmar, M.; Wilker, S.; Richter, K.; Zakrzewicz, A.; Atanasova, S.; Mathes, V.; Timm, T.; Lerner, S.; Klein, J.; et al. Phosphocholine-Modified Macromolecules and Canonical Nicotinic Agonists Inhibit ATP-Induced IL-1β Release. J. Immunol. 2015, 195, 2325–2334. [Google Scholar] [CrossRef]
- Richter, K.; Ogiemwonyi-Schaefer, R.; Wilker, S.; Chaveiro, A.I.; Agné, A.; Hecker, M.; Reichert, M.; Amati, A.-L.; Schlüter, K.-D.; Manzini, I.; et al. Amyloid Beta Peptide (Aβ1-42) Reverses the Cholinergic Control of Monocytic IL-1β Release. J. Clin. Med. 2020, 9, 2887. [Google Scholar] [CrossRef] [PubMed]
- Cresci, A.M.G.; Izzo, C. Chapter 4—Gut Microbiome. In Adult Short Bowel Syndrome; Elsevier: Amsterdam, The Netherlands, 2019; pp. 45–54. [Google Scholar]
- Uebanso, T.; Shimohata, T.; Mawatari, K.; Takahashi, A. Functional Roles of B-Vitamins in the Gut and Gut Microbiome. Mol. Nutr. Food Res. 2020, 64, 2000426. [Google Scholar] [CrossRef]
- Gordon, C.; Behan, J.; Costello, M. Newly identified vitamin K-producing bacteria isolated from the neonatal faecal flora. Microb. Ecol. Health Dis. 2006, 18, 133–138. [Google Scholar]
- Tan, J.; McKenzie, C.; Potamitis, M.; Thorburn, A.N.; Mackay, C.R.; Macia, L. The Role of Short-Chain Fatty Acids in Health and Disease. Adv. Immunol. 2014, 121, 91–119. [Google Scholar] [CrossRef]
- Park, J.-S.; Lee, E.-J.; Lee, J.-C.; Kim, W.-K.; Kim, H.-S. Anti-inflammatory effects of short chain fatty acids in IFN-γ-stimulated RAW 264.7 murine macrophage cells: Involvement of NF-κB and ERK signaling pathways. Int. Immunopharmacol. 2007, 7, 70–77. [Google Scholar] [CrossRef]
- Rastall, R.A. Bacteria in the Gut: Friends and Foes and How to Alter the Balance. J. Nutr. 2004, 134, 2022S–2026S. [Google Scholar] [CrossRef] [PubMed]
- Marshall-Jones, Z.V.; Baillon, M.-L.A.; Croft, J.M.; Butterwick, R.F. Effects of Lactobacillus acidophilus DSM13241 as a probiotic in healthy adult cats. Am. J. Veter. Res. 2006, 67, 1005–1012. [Google Scholar] [CrossRef] [PubMed]
- Baillon, M.-L.A.; Marshall-Jones, Z.V.; Butterwick, R.F. Effects of probiotic Lactobacillus acidophilus strain DSM13241 in healthy adult dogs. Am. J. Veter. Res. 2004, 65, 338–343. [Google Scholar] [CrossRef]
- Ménard, S.; Laharie, D.; Asensio, C.; Vidal-Martinez, T.; Candalh, C.; Rullier, A.; Zerbib, F.; Mégraud, F.; Matysiak-Budnik, T.; Heyman, M. Bifidobacterium breve and Streptococcus thermophilus Secretion Products Enhance T Helper 1 Immune Response and Intestinal Barrier in Mice. Exp. Biol. Med. 2005, 230, 749–756. [Google Scholar] [CrossRef]
- Dargahi, N.; Johnson, J.; Apostolopoulos, V. Streptococcus thermophilus alters the expression of genes associated with innate and adaptive immunity in human peripheral blood mononuclear cells. PLoS ONE 2020, 15, e0228531. [Google Scholar] [CrossRef] [PubMed]
- Wrzosek, L.; Miquel, S.; Noordine, M.-L.; Bouet, S.; Chevalier-Curt, M.J.; Robert, V.; Philippe, C.; Bridonneau, C.; Cherbuy, C.; Robbe-Masselot, C.; et al. Bacteroides thetaiotaomicron and Faecalibacterium prausnitzii influence the production of mucus glycans and the development of goblet cells in the colonic epithelium of a gnotobiotic model rodent. BMC Biol. 2013, 11, 61. [Google Scholar] [CrossRef]
- Tanner, S.A.; Chassard, C.; Rigozzi, E.; Lacroix, C.; Stevens, M.J.A. Bifidobacterium thermophilum RBL67 impacts on growth and virulence gene expression of Salmonella enterica subsp. enterica serovar Typhimurium. BMC Microbiol. 2016, 16, 1–16. [Google Scholar] [CrossRef]
- Lee, J.-H.; O’Sullivan, D.J. Genomic Insights into Bifidobacteria. Microbiol. Mol. Biol. Rev. 2010, 74, 378–416. [Google Scholar] [CrossRef] [PubMed]
- Scheiman, J.; Luber, J.M.; Chavkin, T.A.; Macdonald, T.; Tung, A.; Pham, L.-D.; Wibowo, M.C.; Wurth, R.C.; Punthambaker, S.; Tierney, B.T.; et al. Meta-omics analysis of elite athletes identifies a performance-enhancing microbe that functions via lactate metabolism. Nat. Med. 2019, 25, 1104–1109. [Google Scholar] [CrossRef]
- Bunesova, V.; Lacroix, C.; Schwab, C. Mucin Cross-Feeding of Infant Bifidobacteria and Eubacterium hallii. Microb. Ecol. 2018, 75, 228–238. [Google Scholar] [CrossRef]
- Lopetuso, L.R.; Scaldaferri, F.; Petito, V.; Gasbarrini, A. Commensal clostridia: Leading players in the maintenance of gut homeostasis. Gut Pathog. 2013, 5, 1–23. [Google Scholar] [CrossRef] [PubMed]
- Drago, L.; Toscano, M.; Rodighiero, V.; De Vecchi, E.; Mogna, G. Cultivable and Pyrosequenced Fecal Microflora in Centenarians and Young Subjects. J. Clin. Gastroenterol. 2012, 46, S81–S84. [Google Scholar] [CrossRef] [PubMed]
- Pryde, S.E.; Duncan, S.H.; Hold, G.L.; Stewart, C.S.; Flint, H.J. The microbiology of butyrate formation in the human colon. FEMS Microbiol. Lett. 2002, 217, 133–139. [Google Scholar] [CrossRef]
- Wlodarska, M.; Luo, C.; Kolde, R.; D’Hennezel, E.; Annand, J.W.; Heim, C.E.; Krastel, P.; Schmitt, E.K.; Omar, A.S.; Creasey, E.A.; et al. Indoleacrylic Acid Produced by Commensal Peptostreptococcus Species Suppresses Inflammation. Cell Host Microbe 2017, 22, 25–37.e6. [Google Scholar] [CrossRef] [PubMed]
- Kho, Z.Y.; Lal, S.K. The Human Gut Microbiome—A Potential Controller of Wellness and Disease. Front. Microbiol. 2018, 9, 1835. [Google Scholar] [CrossRef] [PubMed]
- Jeffery, I.B.; O’Toole, P.W.; Öhman, L.; Claesson, M.J.; Deane, J.; Quigley, E.M.M.; Simrén, M. An irritable bowel syndrome subtype defined by species-specific alterations in faecal microbiota. Gut 2011, 61, 997–1006. [Google Scholar] [CrossRef]
- Sánchez, E.; Donat, E.; Ribes-Koninckx, C.; Fernández-Murga, M.L.; Sanz, Y. Duodenal-Mucosal Bacteria Associated with Celiac Disease in Children. Appl. Environ. Microbiol. 2013, 79, 5472–5479. [Google Scholar] [CrossRef]
- Liebregts, T.; Adam, B.; Bredack, C.; Röth, A.; Heinzel, S.; Lester, S.; Downie–Doyle, S.; Smith, E.; Drew, P.; Talley, N.J.; et al. Immune Activation in Patients with Irritable Bowel Syndrome. Gastroenterology 2007, 132, 913–920. [Google Scholar] [CrossRef]
- Kuehne, S.A.; Cartman, S.T.; Heap, J.T.; Kelly, M.L.; Cockayne, A.; Minton, N.P. The role of toxin A and toxin B in Clostridium difficile infection. Nature 2010, 467, 711–713. [Google Scholar] [CrossRef]
- Savidge, T.C.; Pan, W.-H.; Newman, P.; O’Brien, M.J.; Anton, P.M.; Pothoulakis, C. Clostridium difficile toxin B is an inflammatory enterotoxin in human intestine. Gastroenterology 2003, 125, 413–420. [Google Scholar] [CrossRef]
- Chi, L.; Mahbub, R.; Gao, B.; Bian, X.; Tu, P.; Ru, H.; Lu, K. Nicotine Alters the Gut Microbiome and Metabolites of Gut–Brain Interactions in a Sex-Specific Manner. Chem. Res. Toxicol. 2017, 30, 2110–2119. [Google Scholar] [CrossRef] [PubMed]
- Biedermann, L.; Zeitz, J.; Mwinyi, J.; Sutter-Minder, E.; Rehman, A.; Ott, S.J.; Steurer-Stey, C.; Frei, A.; Frei, P.; Scharl, M.; et al. Smoking Cessation Induces Profound Changes in the Composition of the Intestinal Microbiota in Humans. PLoS ONE 2013, 8, e59260. [Google Scholar] [CrossRef] [PubMed]
- Tomoda, K.; Kubo, K.; Asahara, T.; Andoh, A.; Nomoto, K.; Nishii, Y.; Yamamoto, Y.; Yoshikawa, M.; Kimura, H. Cigarette smoke decreases organic acids levels and population of bifidobacterium in the caecum of rats. J. Toxicol. Sci. 2011, 36, 261–266. [Google Scholar] [CrossRef] [PubMed]
- Wang, H. Side-stream smoking reduces intestinal inflammation and increases expression of tight junction proteins. World J. Gastroenterol. 2012, 18, 2180–2187. [Google Scholar] [CrossRef]
- Kaji, I.; Akiba, Y.; Konno, K.; Watanabe, M.; Kimura, S.; Iwanaga, T.; Kuri, A.; Iwamoto, K.-I.; Kuwahara, A.; Kaunitz, J.D. Neural FFA3 activation inversely regulates anion secretion evoked by nicotinic ACh receptor activation in rat proximal colon. J. Physiol. 2016, 594, 3339–3352. [Google Scholar] [CrossRef]
- Kaji, I.; Akiba, Y.; Furuyama, T.; Adelson, D.W.; Iwamoto, K.; Watanabe, M.; Kuwahara, A.; Kaunitz, J.D. Free fatty acid receptor 3 activation suppresses neurogenic motility in rat proximal colon. Neurogastroenterol. Motil. 2017, 30, e13157. [Google Scholar] [CrossRef]
- Zhu, N.; Zhang, D.; Wang, W.; Li, X.; Yang, B.; Song, J.; Zhao, X.; Huang, B.; Shi, W.; Lu, R.; et al. A Novel Coronavirus from Patients with Pneumonia in China, 2019. N. Engl. J. Med. 2020, 382, 727–733. [Google Scholar] [CrossRef]
- Zhou, P.; Yang, X.-L.; Wang, X.-G.; Hu, B.; Zhang, L.; Zhang, W.; Si, H.-R.; Zhu, Y.; Li, B.; Huang, C.-L.; et al. A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature 2020, 579, 270–273. [Google Scholar] [CrossRef]
- Walls, A.C.; Park, Y.-J.; Tortorici, M.A.; Wall, A.; McGuire, A.T.; Veesler, D. Structure, Function, and Antigenicity of the SARS-CoV-2 Spike Glycoprotein. Cell 2020, 181, 281–292.e6. [Google Scholar] [CrossRef]
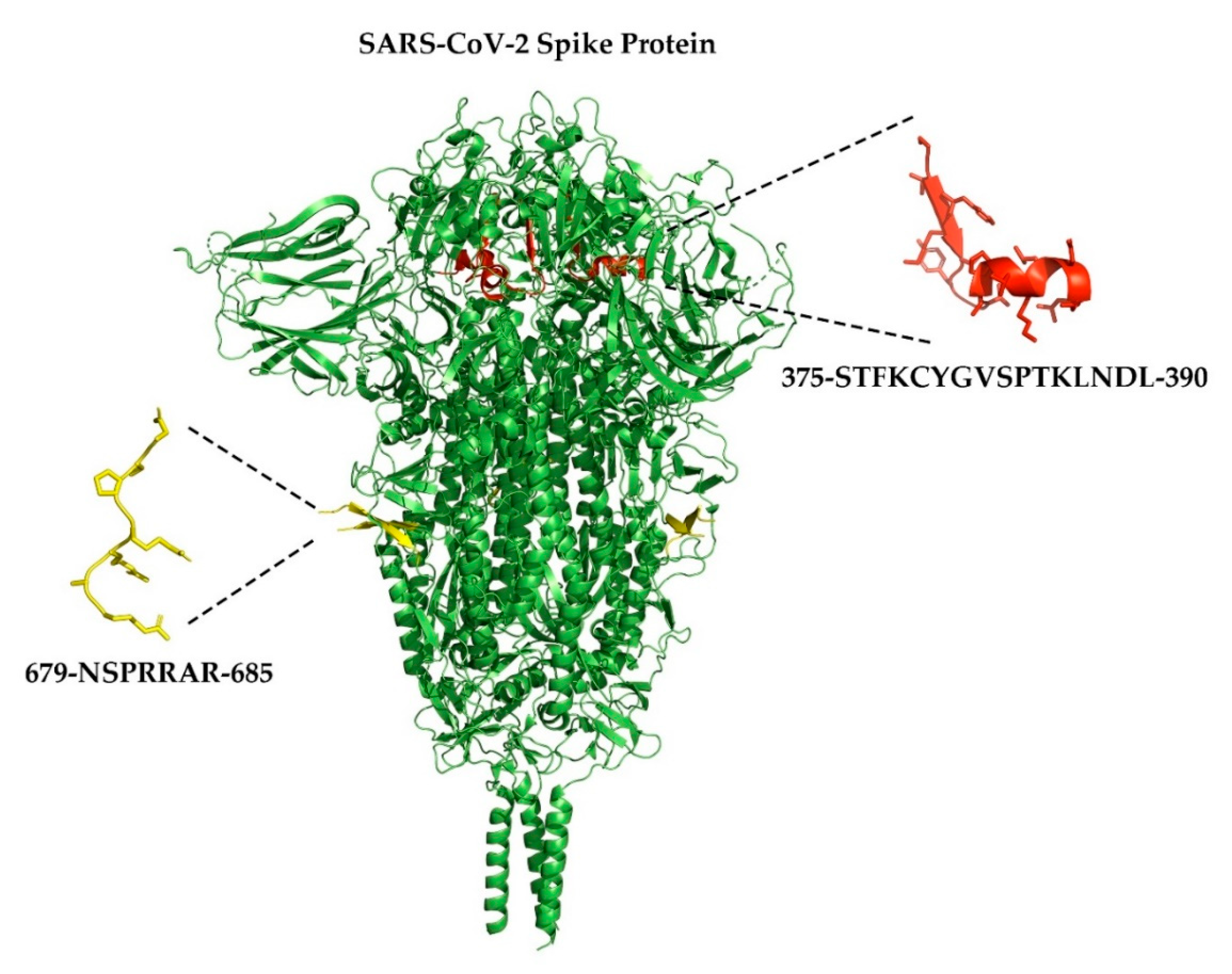
- Xia, S.; Lan, Q.; Su, S.; Wang, X.; Xu, W.; Liu, Z.; Zhu, Y.; Wang, Q.; Lu, L.; Jiang, S. The role of furin cleavage site in SARS-CoV-2 spike protein-mediated membrane fusion in the presence or absence of trypsin. Signal Transduct. Target. Ther. 2020, 5, 1–3. [Google Scholar] [CrossRef]
- Coutard, B.; Valle, C.; De Lamballerie, X.; Canard, B.; Seidah, N.; Decroly, E. The spike glycoprotein of the new coronavirus 2019-nCoV contains a furin-like cleavage site absent in CoV of the same clade. Antivir. Res. 2020, 176, 104742. [Google Scholar] [CrossRef] [PubMed]
- Changeux, J.-P.; Amoura, Z.; Rey, F.A.; Miyara, M. A nicotinic hypothesis for Covid-19 with preventive and therapeutic implications. Comptes Rendus. Biol. 2020, 343, 33–39. [Google Scholar] [CrossRef] [PubMed]
- Farsalinos, K.; Eliopoulos, E.; Leonidas, D.D.; Papadopoulos, G.E.; Tzartos, S.J.; Poulas, K. Nicotinic Cholinergic System and COVID-19: In Silico Identification of an Interaction between SARS-CoV-2 and Nicotinic Receptors with Potential Therapeutic Targeting Implications. Int. J. Mol. Sci. 2020, 21, 5807. [Google Scholar] [CrossRef]
- Farsalinos, K.; Niaura, R.; Le Houezec, J.; Barbouni, A.; Tsatsakis, A.; Kouretas, D.; Vantarakis, A.; Poulas, K. Editorial: Nicotine and SARS-CoV-2: COVID-19 may be a disease of the nicotinic cholinergic system. Toxicol. Rep. 2020, 7, 658–663. [Google Scholar] [CrossRef] [PubMed]
- Schrödinger. PyMOL Molecular Graphics System Version 2.3; Schrödinger: New York, NY, USA, 2019. [Google Scholar]
- Cai, Y.; Zhang, J.; Xiao, T.; Peng, H.; Sterling, S.M.; Jr, R.M.W.; Rawson, S.; Rits-Volloch, S.; Chen, B. Distinct conformational states of SARS-CoV-2 spike protein. Science 2020, 369, eabd4251. [Google Scholar] [CrossRef]
- Daly, J.L.; Simonetti, B.; Klein, K.; Chen, K.-E.; Williamson, M.K.; Antón-Plágaro, C.; Shoemark, D.K.; Simón-Gracia, L.; Bauer, M.; Hollandi, R.; et al. Neuropilin-1 is a host factor for SARS-CoV-2 infection. Science 2020, 370, 861–865. [Google Scholar] [CrossRef]
- Zhang, H.; Kang, Z.; Gong, H.; Xu, D.; Wang, J.; Li, Z.; Li, Z.; Cui, X.; Xiao, J.; Zhan, J.; et al. Digestive system is a potential route of COVID-19: An analysis of single-cell coexpression pattern of key proteins in viral entry process. Gut 2020, 69, 1010–1018. [Google Scholar] [CrossRef]
- Mao, R.; Qiu, Y.; He, J.-S.; Tan, J.-Y.; Li, X.-H.; Liang, J.; Shen, J.; Zhu, L.-R.; Chen, Y.; Iacucci, M.; et al. Manifestations and prognosis of gastrointestinal and liver involvement in patients with COVID-19: A systematic review and meta-analysis. Lancet Gastroenterol. Hepatol. 2020, 5, 667–678. [Google Scholar] [CrossRef]
- Guerra, I.; Algaba, A.; Jimenez, L.; Mar Aller, M.; Garza, D.; Bonillo, D.; Molina Esteban, L.M.; Bermejo, F. Incidence, Clinical Characteristics, and Evolution of SARS-CoV-2 Infection in Patients with Inflammatory Bowel Disease: A Single-Center Study in Madrid, Spain. Inflamm. Bowel Dis. 2020, 27, 25–33. [Google Scholar] [CrossRef]
- Rodríguez-Lago, I.; De La Piscina, P.R.; Elorza, A.; Merino, O.; De Zárate, J.O.; Cabriada, J.L. Characteristics and Prognosis of Patients With Inflammatory Bowel Disease During the SARS-CoV-2 Pandemic in the Basque Country (Spain). Gastroenterology 2020, 159, 781–783. [Google Scholar] [CrossRef]
- D’Amico, F.; Danese, S.; Peyrin-Biroulet, L. Systematic Review on Inflammatory Bowel Disease Patients With Coronavirus Disease 2019: It Is Time to Take Stock. Clin. Gastroenterol. Hepatol. 2020, 18, 2689–2700. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Rubio, J.; Navarro-Lopez, C.; Lopez-Najera, E.; Lopez-Najera, A.; Najera, A.; Navarro-Lopez, J.D.; Najera, A. Cytokine Release Syndrome (CRS) and Nicotine in COVID-19 Patients: Trying to Calm the Storm. Front. Immunol. 2020, 11, 1359. [Google Scholar] [CrossRef] [PubMed]
- Bonaz, B.; Sinniger, V.; Pellissier, S. Targeting the cholinergic anti-inflammatory pathway with vagus nerve stimulation in patients with Covid-19? Bioelectron. Med. 2020, 6, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, F. COVID-19 induced ARDS, and the use of galantamine to activate the cholinergic anti-inflammatory pathway. Med Hypotheses 2020, 145, 110331. [Google Scholar] [CrossRef]
- Segal, J.P.; Mak, J.W.Y.; Mullish, B.H.; Alexander, J.L.; Ng, S.C.; Marchesi, J.R. The gut microbiome: An under-recognised contributor to the COVID-19 pandemic? Ther. Adv. Gastroenterol. 2020, 13, 1756284820974914. [Google Scholar] [CrossRef]
- Din, A.U.; Mazhar, M.; Waseem, M.; Ahmad, W.; Bibi, A.; Hassan, A.; Ali, N.; Gang, W.; Qian, G.; Ullah, R.; et al. SARS-CoV-2 microbiome dysbiosis linked disorders and possible probiotics role. Biomed. Pharmacother. 2021, 133, 110947. [Google Scholar] [CrossRef]
- Gu, S.; Chen, Y.; Wu, Z.; Chen, Y.; Gao, H.; Lv, L.; Guo, F.; Zhang, X.; Luo, R.; Huang, C.; et al. Alterations of the Gut Microbiota in Patients with COVID-19 or H1N1 Influenza. Clin. Infect. Dis. 2020, 71, 2669–2678. [Google Scholar] [CrossRef]
- Zuo, T.; Liu, Q.; Zhang, F.; Lui, G.C.; Tso, E.Y.; Yeoh, Y.K.; Chen, Z.; Boon, S.S.; Chan, F.K.; Chan, P.K.; et al. Depicting SARS-CoV-2 faecal viral activity in association with gut microbiota composition in patients with COVID-19. Gut 2020, 70, 276–284. [Google Scholar] [CrossRef]
- Conte, L.; Toraldo, D.M. Targeting the gut–lung microbiota axis by means of a high-fibre diet and probiotics may have anti-inflammatory effects in COVID-19 infection. Ther. Adv. Respir. Dis. 2020, 14, 1753466620937170. [Google Scholar] [CrossRef]
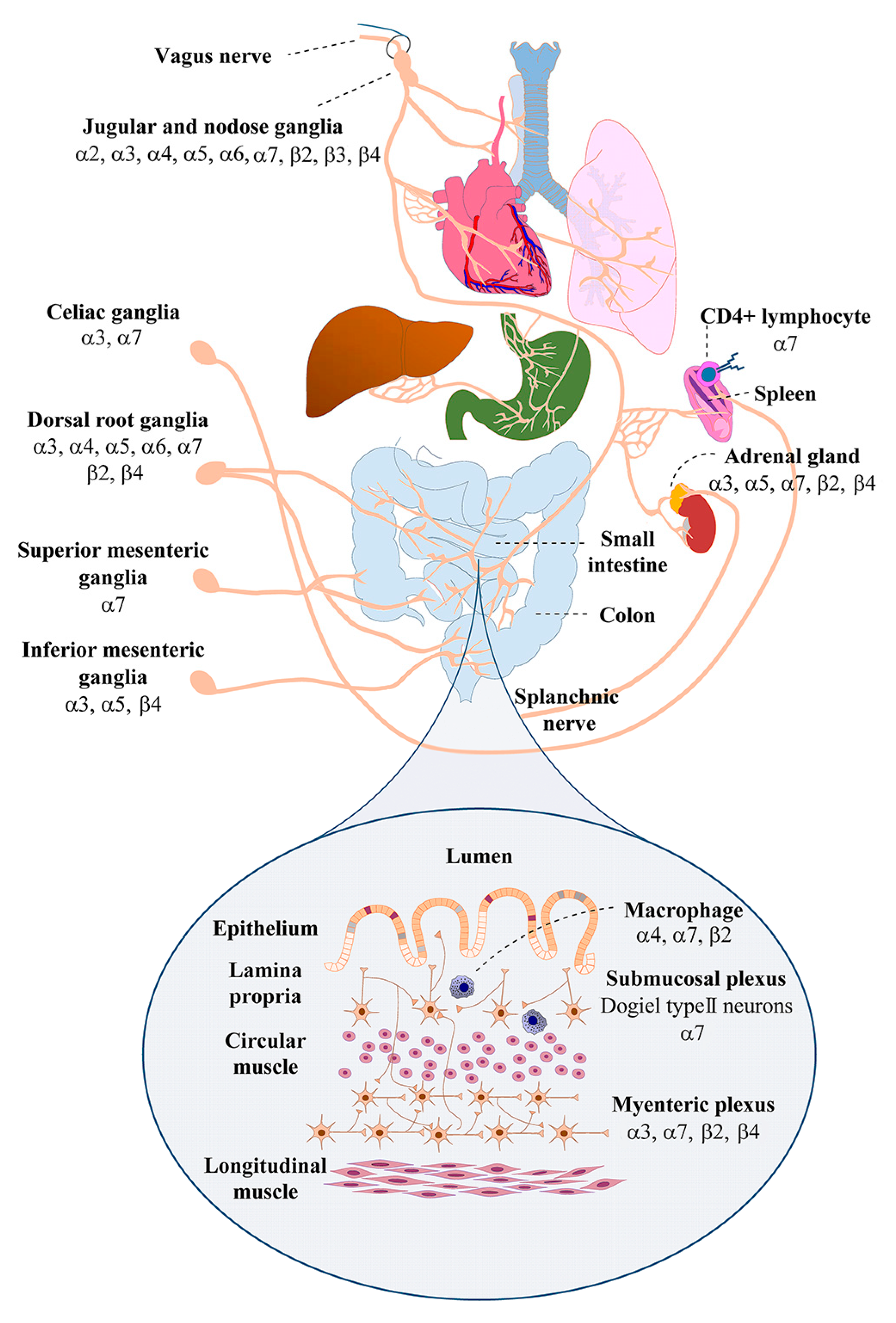
| Neural Structure | nAChR Subunits a | Functional nAChRs b | Target Organ in the Gastrointestinal Tract | Ref. |
|---|---|---|---|---|
| Nodose ganglia c | α2, α3, α4, α5, α6, α7, β2, β3, β4 | α3β4 * | Proximal small intestine and colon | [12,43,44] |
| Dorsal root ganglia c,e | α3, α4, α5, α6, α7, β2, β4 | α3β4 *, α4β2 *, α6β4 *, α7 | Small and large intestines | [26,27,43,45,46] |
| Celiac ganglia c | α3, α7 | α3 *,f, α7 f | Distal esophagus, stomach, proximal duodenum, liver, biliary system, spleen, adrenal glands | [47,48] |
| Superior mesenteric ganglia c | α7 | α7 f | Duodenum, jejunum, ileum, cecum, appendix, ascending colon, proximal transverse colon | [47] |
| Inferior mesenteric ganglia c,d | α3, α5, β4 | α3β4 * | Distal transverse, descending, and sigmoid, colon, rectum, upper anal canal | [49] |
| Inferior hypogastric plexus c,d | α3, β4, α7 | unknown | Urogential organs, pelvic viscera | [50,51] |
| Myenteric plexus c,d,e | α3, α5, α7, β2, β4 | α3β2 *, α3β4 *, α7 | Circular and longitudinal muscles of the gut wall, submucosa, epithelia, stomach, small and large intestines, colon | [37,39,52,53] |
| Ligand | Mechanism of Action | Disease-modifying Mechanisms | Effects on IBD | Ref. |
|---|---|---|---|---|
| Nicotine | Non-selective agonist | Suppression of MAdCAM-1; reduced regenerative spike action-potentials | Decreased signs and symptoms of DSS-induced colitis in mice Reduced colonic DRG neuron hyperexcitability in DSS-induced colitis in mice | [30,104] |
| Galantamine | Non-selective PAM | Reduced NF-κB, TNF-α levels, MPO, and neutrophil infiltration | Decreased signs and symptoms of TNBS-induced colitis in mice | [109] |
| PNU-282987 | α7-selective agonist | Reduced infiltration of leucocytes Reduced infiltration of macrophages, and reduced levels of IL-6, and IFN-γ | Attenuated colonic inflammation in DSS-treated mice Decreased signs and symptoms of DSS-induced colitis in mice | [110,115] |
| PNU-120596 | α7-selective PAM | Decreased IL-1β and TNF-α in LPS-treated mice | Decreased symptoms related to anxiety and depression in mice | [116] |
| GTS-21 | partial α7 agonist | Decreased TNF-α in plasma | Probable decreased colonic inflammation in patients with ulcerative colitis | [117] |
| AR-R17779 | α7 agonist | Reduced colonic infiltration of CD4+ and CD8+ lymphocytes; inhibition of macrophage activation | Decreased signs and symptoms of TNBS-induced colitis in mice Decreased signs and symptoms of postoperative ileus in mice | [99,114] |
| Encenicline | partial α7 agonist | Reduced colonic infiltration of macrophages, neutrophils, and B lymphocytes | Decreased signs and symptoms of TNBS- and DSS induced colitis in mice | [112] |
| RgIA | α9 antagonist | Reduced levels of colonic TNF-α | Decreased signs and symptoms of DSS-induced colitis in mice | [118] |
| Bacteria | Location in the GI Tract | Functional Role | Ref. |
|---|---|---|---|
| Lactobacillus | Stomach, duodenum, jejunum, ileum, colon | Improved digestion and absorption of nutrients; inhibition of the growth of pathogens by activating the immune system a,b | [132,133,134] |
| Streptococcus | Jejunum, ileum, colon | Modulation of the immune system through altered cytokine release from immune cellsc | [135,136] |
| Bacteroides | Jejunum, ileum, colon | Production of SCFAs involved in energy homeostasis and regulation of intestinal inflammationd | [137] |
| Bifidobacterium | Jejunum, ileum, colon | Inhibition of the growth of pathogens by activating the immune system; amino-acid and vitamin synthesisd | [138,139] |
| Veillonella | Colon | Production of SCFAs involved in energy homeostasisc,d | [140] |
| Eubacteria | Colon | Production of SCFAs involved in energy homeostasis and regulation of intestinal inflammationc | [141] |
| Clostridia | Colon | Participation in resistance to the colonization of pathogens; production of SCFAsc; maintenance of gut homeostasisc | [142,143,144] |
| Peptostreptococcus | Colon | Maintenance of epithelial barrier and modulation of intestinal inflammationc,d | [145] |
| Effect on Bacterial Levels | Effects on Gut Function | Ref. | |
|---|---|---|---|
| Bacteria | |||
| alpha-diversity | Increased in mice | Improvement of gut barrier function by production metabolites and antimicrobial substances | [108] |
| Lactobacillus | Increased in mice | Improvement of gut barrier function and prevent inflammation by production of SCFAs, lactate and antimicrobial substances | [108] |
| Lachnospiraceae | Increased in mice | Improvement of gut barrier function by production of beneficial metabolites such as SCFAs | [108] |
| Christensenellaceae | Decreased in female mice | Development of metabolic syndrome | [152] |
| Anaeroplasmataceae | Decreased in female mice | Alteration of the intestinal transit | [152] |
| Dehalobacteriaceae | Decreased in male mice | Development of metabolic syndrome | [152] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rueda Ruzafa, L.; Cedillo, J.L.; Hone, A.J. Nicotinic Acetylcholine Receptor Involvement in Inflammatory Bowel Disease and Interactions with Gut Microbiota. Int. J. Environ. Res. Public Health 2021, 18, 1189. https://doi.org/10.3390/ijerph18031189
Rueda Ruzafa L, Cedillo JL, Hone AJ. Nicotinic Acetylcholine Receptor Involvement in Inflammatory Bowel Disease and Interactions with Gut Microbiota. International Journal of Environmental Research and Public Health. 2021; 18(3):1189. https://doi.org/10.3390/ijerph18031189
Chicago/Turabian StyleRueda Ruzafa, Lola, José Luis Cedillo, and Arik J. Hone. 2021. "Nicotinic Acetylcholine Receptor Involvement in Inflammatory Bowel Disease and Interactions with Gut Microbiota" International Journal of Environmental Research and Public Health 18, no. 3: 1189. https://doi.org/10.3390/ijerph18031189
APA StyleRueda Ruzafa, L., Cedillo, J. L., & Hone, A. J. (2021). Nicotinic Acetylcholine Receptor Involvement in Inflammatory Bowel Disease and Interactions with Gut Microbiota. International Journal of Environmental Research and Public Health, 18(3), 1189. https://doi.org/10.3390/ijerph18031189






