Sub-Clinical Effects of Outdoor Smoke in Affected Communities
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Study Period
2.3. Health Assessments
- Blood tests, for markers of inflammation and coagulation, specifically high sensitivity C-reactive protein (CRP), fibrinogen, and a full blood examination (including hemoglobin, total and differential white cell and platelet counts) [17].
- Airway inflammation test, measuring fractional exhaled nitric oxide (FeNO) using a NiOx unit (Aerocrine AB, Solna, Sweden) [18].
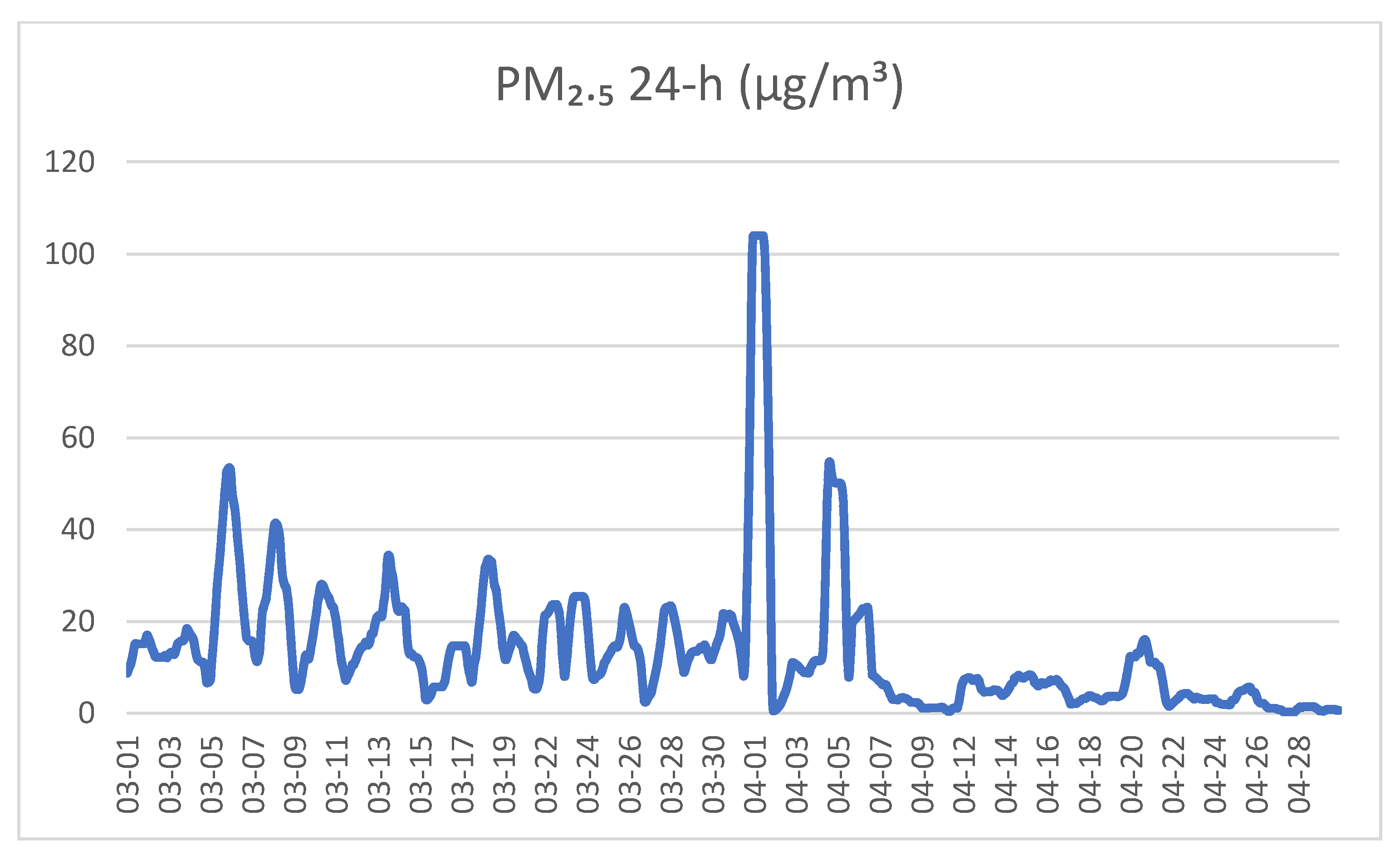
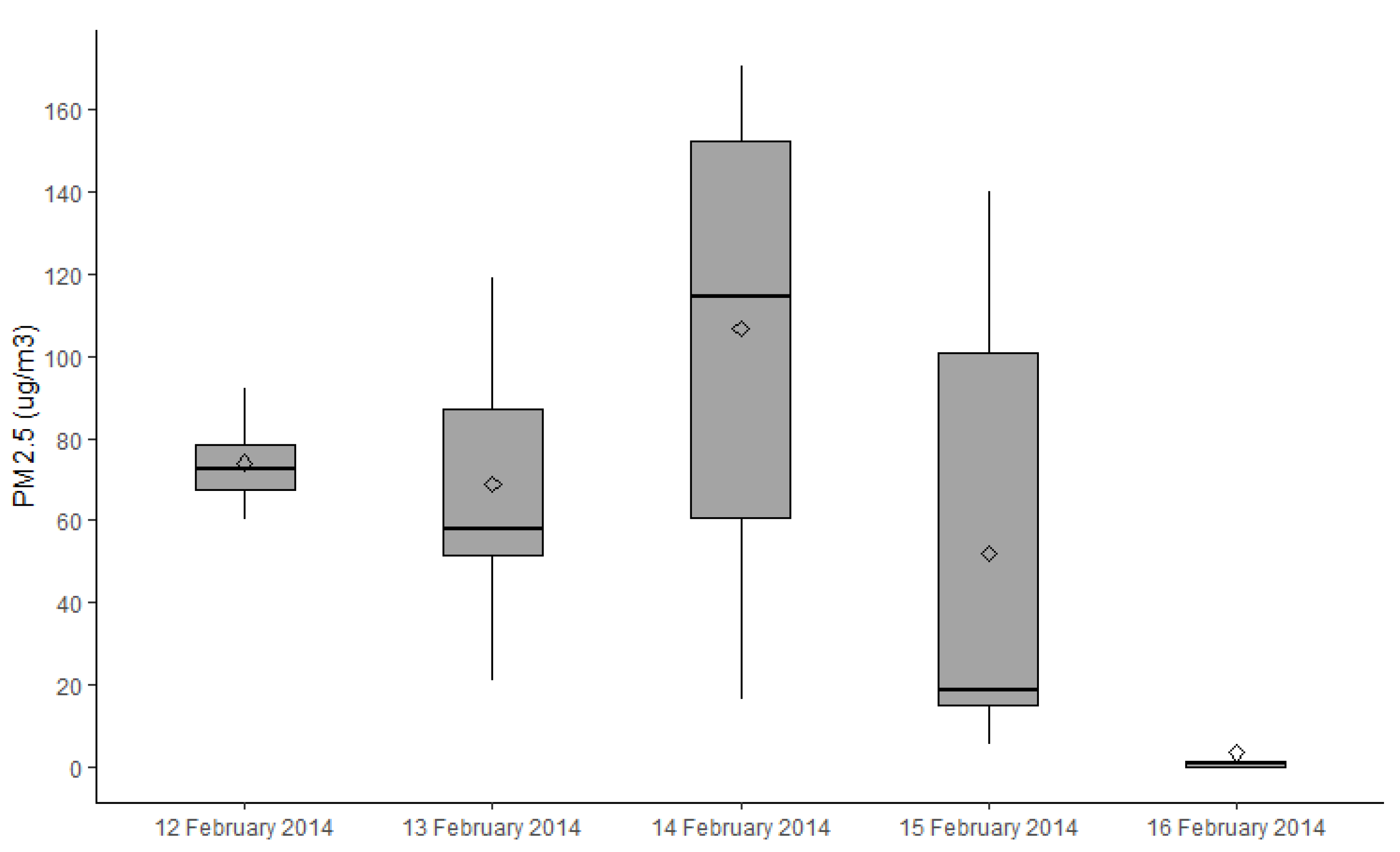
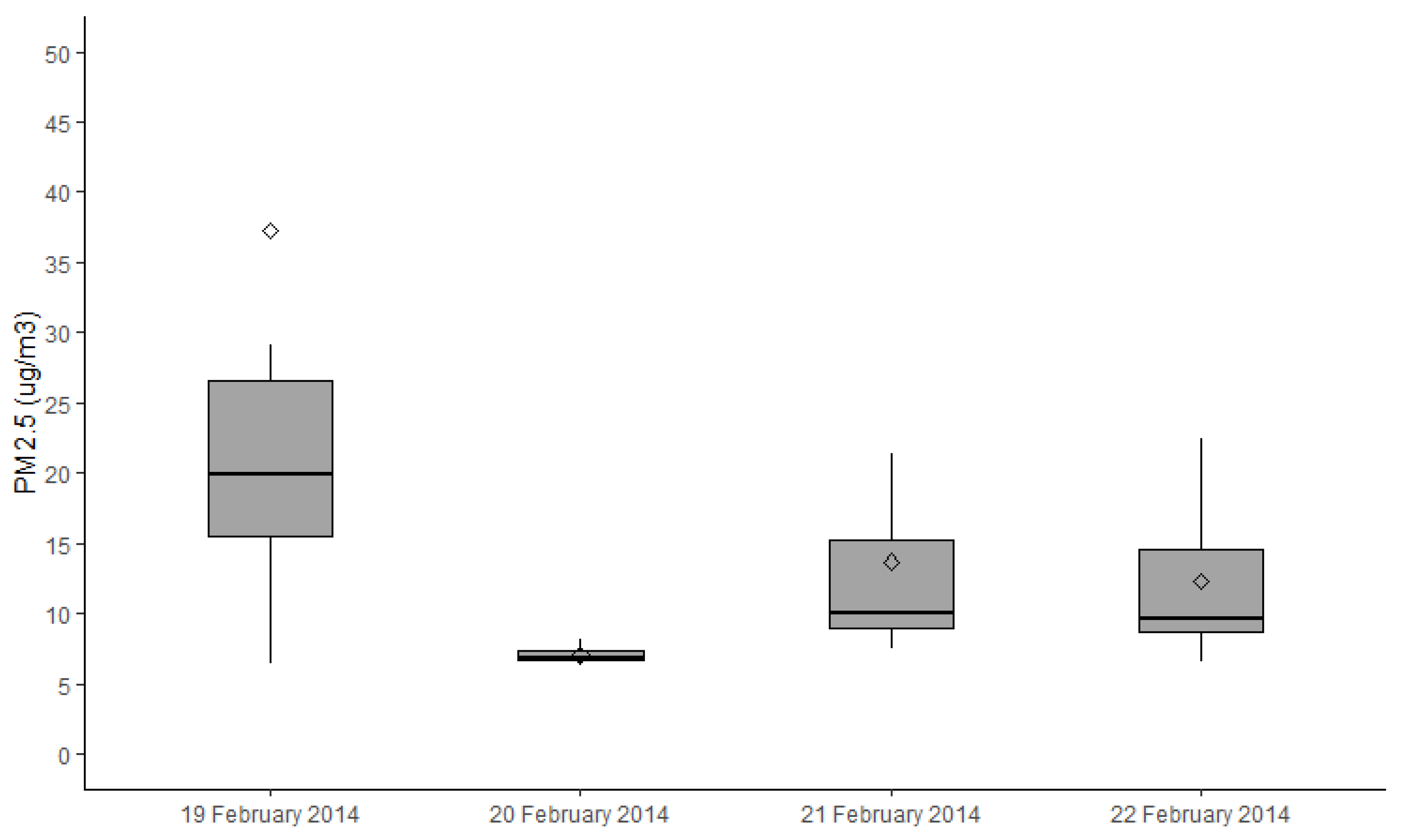
2.4. Exposure Assessment
2.5. Statistical Analysis
2.6. Ethics Approval
3. Results
4. Discussion
Strengths and Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Huang, Y.; Wu, S.; Kaplan, J.O. Sensitivity of global wildfire occurrences to various factors in the context of global change. Atmos. Environ. 2015, 121, 86–92. [Google Scholar] [CrossRef]
- Sharples, J.J.; Cary, G.J.; Fox-Hughes, P.; Mooney, S.; Evans, J.P.; Fletcher, M.-S.; Fromm, M.; Grierson, P.F.; McRae, R.; Baker, P. Natural hazards in Australia: Extreme bushfire. Clim. Chang. 2016, 139, 85–99. [Google Scholar] [CrossRef]
- Keywood, M.; Kanakidou, M.; Stohl, A.; Dentener, F.; Grassi, G.; Meyer, C.P.; Torseth, K.; Edwards, D.; Thompson, A.M.; Lohmann, U.; et al. Fire in the Air: Biomass Burning Impacts in a Changing Climate. Crit. Rev. Environ. Sci. Technol. 2011, 43, 40–83. [Google Scholar] [CrossRef]
- Keywood, M.; Cope, M.; Meyer, C.M.; Iinuma, Y.; Emmerson, K. When smoke comes to town: The impact of biomass burning smoke on air quality. Atmos. Environ. 2015, 121, 13–21. [Google Scholar] [CrossRef]
- Penman, T.D.; Christie, F.J.; Andersen, A.N.; Bradstock, R.A.; Cary, G.J.; Henderson, M.K.; Price, O.; Tran, C.; Wardle, G.M.; Williams, R.J.; et al. Prescribed burning: How can it work to conserve the things we value? Int. J. Wildland Fire 2011, 20, 721–733. [Google Scholar] [CrossRef]
- Bradstock, R.A.; Cary, G.; Davies, I.; Lindenmayer, D.; Price, O.; Williams, R. Wildfires, fuel treatment and risk mitigation in Australian eucalypt forests: Insights from landscape-scale simulation. J. Environ. Manag. 2012, 105, 66–75. [Google Scholar] [CrossRef]
- Boer, M.M.; Sadler, R.J.; Wittkuhn, R.S.; McCaw, L.; Grierson, P.F. Long-term impacts of prescribed burning on regional extent and incidence of wildfires—Evidence from 50 years of active fire management in SW Australian forests. For. Ecol. Manag. 2009, 259, 132–142. [Google Scholar] [CrossRef]
- Johnston, F.H. Bushfires and human health in a changing environment. Aust. Fam. Physician 2009, 38, 720–724. [Google Scholar]
- Alves, C.; Gonçalves, C.; Evtyugina, M.; Pio, C.; Mirante, F.; Puxbaum, H. Particulate organic compounds emitted from experimental wildland fires in a Mediterranean ecosystem. Atmos. Environ. 2010, 44, 2750–2759. [Google Scholar] [CrossRef]
- Garcia-Hurtado, E.; Pey, J.; Borrás, E.; Sánchez, P.; Vera, T.; Carratalá, A.; Alastuey, A.; Querol, X.; Vallejo, V.R. Atmospheric PM and volatile organic compounds released from Mediterranean shrubland wildfires. Atmos. Environ. 2014, 89, 85–92. [Google Scholar] [CrossRef]
- Feng, S.-L.; Gao, D.; Liao, F.; Zhou, F.; Wang, X. The health effects of ambient PM2.5 and potential mechanisms. Ecotoxicol. Environ. Saf. 2016, 128, 67–74. [Google Scholar] [CrossRef] [PubMed]
- Cohen, A.J.; Brauer, M.; Burnett, R.; Anderson, H.R.; Frostad, J.; Estep, K.; Balakrishnan, K.; Brunekreef, B.; Dandona, L.; Dandona, R.; et al. Estimates and 25-year trends of the global burden of disease attributable to ambient air pollution: An analysis of data from the Global Burden of Diseases Study 2015. Lancet 2017, 389, 1907–1918. [Google Scholar] [CrossRef]
- Haikerwal, A.; Akram, M.; Del Monaco, A.; Smith, K.; Sim, M.R.; Meyer, M.; Tonkin, A.M.; Abramson, M.J.; Dennekamp, M. Impact of Fine Particulate Matter (PM 2.5) Exposure During Wildfires on Cardiovascular Health Outcomes. J. Am. Hear. Assoc. 2015, 4, e001653. [Google Scholar] [CrossRef] [PubMed]
- Kollanus, V.; Tiittanen, P.; Niemi, J.V.; Lanki, T. Effects of long-range transported air pollution from vegetation fires on daily mortality and hospital admissions in the Helsinki metropolitan area, Finland. Environ. Res. 2016, 151, 351–358. [Google Scholar] [CrossRef]
- Sigsgaard, T.; Forsberg, B.; Annesi-Maesano, I.; Blomberg, A.; Bølling, A.; Boman, C.; Bønløkke, J.; Brauer, M.; Bruce, N.; Héroux, M.-E.; et al. Health impacts of anthropogenic biomass burning in the developed world. Eur. Respir. J. 2015, 46, 1577–1588. [Google Scholar] [CrossRef] [PubMed]
- Kollanus, V.; Prank, M.; Gens, A.; Soares, J.; Vira, J.; Kukkonen, J.; Sofiev, M.; Salonen, R.O.; Lanki, T. Mortality due to Vegetation Fire—Originated PM 2.5 Exposure in Europe—Assessment for the Years 2005 and 2008. Environ. Health Perspect. 2017, 125, 30–37. [Google Scholar] [CrossRef]
- O’Keeffe, D.; Dennekamp, M.; Straney, L.D.; Mazhar, M.; O’Dwyer, T.; Haikerwal, A.; Reisen, F.; Abramson, M.J.; Johnston, F.H. Health effects of smoke from planned burns: A study protocol. BMC Public Health 2016, 16, 186. [Google Scholar] [CrossRef]
- American Thoracic Society/European Respiratory Society recommendations for standardized procedures for the online and offline measurement of exhaled lower respiratory nitric oxide and nasal nitric oxide, 2005. Am. J. Respir. Crit. Care Med. 2005, 171, 912–930. Available online: https://www.atsjournals.org/doi/pdf/10.1164/rccm.200406-710ST (accessed on 27 January 2021). [CrossRef]
- Stebbings, J.H. Panel studies of acute health effects of air pollution: II. A methodologic study of linear regression analysis of asthma panel data. Environ. Res. 1978, 17, 10–32. [Google Scholar] [CrossRef]
- Hardin, J.W.; Hilbe, J.M. Generalized Estimating Equations; Chapman & Hall/CRC: Boca Raton, FL, USA, 2013. [Google Scholar]
- StataCorp. Stata Statistical Software: Release 14; StataCorp LP: College Station, TX, USA, 2015. [Google Scholar]
- Allen, R.W.; Mar, T.; Koenig, J.; Liu, L.-J.S.; Gould, T.; Simpson, C.; Larson, T. Changes in Lung Function and Airway Inflammation Among Asthmatic Children Residing in a Woodsmoke-Impacted Urban Area. Inhal. Toxicol. 2008, 20, 423–433. [Google Scholar] [CrossRef]
- Barregard, L.; Sallsten, G.; Andersson, L.; Almstrand, A.-C.; Gustafson, P.; Olin, A.-C. Experimental exposure to wood smoke: Effects on airway inflammation and oxidative stress. Occup. Environ. Med. 2008, 65, 319–324. [Google Scholar] [CrossRef] [PubMed]
- Stockfelt, L.; Sallsten, G.; Olin, A.-C.; Almerud, P.; Samuelsson, L.; Johannesson, S.; Molnar, P.; Strandberg, B.; Almstrand, A.-C.; Bergemalm-Rynell, K.; et al. Effects on airways of short-term exposure to two kinds of wood smoke in a chamber study of healthy humans. Inhal. Toxicol. 2011, 24, 47–59. [Google Scholar] [CrossRef] [PubMed]
- Abramson, M.J.; Wigmann, C.; Altug, H.; Schikowski, T. Ambient air pollution is associated with airway inflammation in older women: A nested cross-sectional analysis. BMJ Open Respir. Res. 2020, 7, e000549. [Google Scholar] [CrossRef] [PubMed]
- Viehmann, A.; Hertel, S.; Fuks, K.; Eisele, L.; Moebus, S.; Möhlenkamp, S.; Nonnemacher, M.; Jakobs, H.; Erbel, R.; Jöckel, K.-H.; et al. Long-term residential exposure to urban air pollution, and repeated measures of systemic blood markers of inflammation and coagulation. Occup. Environ. Med. 2015, 72, 656–663. [Google Scholar] [CrossRef]
- Bind, M.-A.; Baccarelli, A.; Zanobetti, A.; Tarantini, L.; Suh, H.; Vokonas, P.; Schwartz, J. Air pollution and markers of coagulation, inflammation, and endothelial function: Associations and epigene-environment interactions in an elderly cohort. Epidemiology 2012, 23, 332–340. [Google Scholar] [CrossRef]
- Green, R.; Broadwin, R.; Malig, B.; Basu, R.; Gold, E.B.; Qi, L.; Sternfeld, B.; Bromberger, J.T.; Greendale, G.A.; Kravitz, H.M.; et al. Long-and Short-Term Exposure to Air Pollution and Inflammatory/Hemostatic Markers in Midlife Women. Epidemiology 2015, 27, 211–220. [Google Scholar] [CrossRef]
- Kajbafzadeh, M.; Brauer, M.; Karlen, B.; Carlsten, C.; Van Eeden, S.; Allen, R.W. The impacts of traffic-related and woodsmoke particulate matter on measures of cardiovascular health: A HEPA filter intervention study. Occup. Environ. Med. 2015, 72, 394–400. [Google Scholar] [CrossRef]
- Sarnat, J.A.; Golan, R.; Greenwald, R.; Raysoni, A.U.; Kewada, P.; Winquist, A.; Sarnat, S.E.; Flanders, W.D.; Mirabelli, M.C.; Zora, J.E.; et al. Exposure to traffic pollution, acute inflammation and autonomic response in a panel of car commuters. Environ. Res. 2014, 133, 66–76. [Google Scholar] [CrossRef]
- Croft, D.P.; Cameron, S.J.; Morrell, C.N.; Lowenstein, C.J.; Ling, F.; Zareba, W.; Hopke, P.K.; Utell, M.J.; Thurston, S.W.; Thevenet-Morrison, K.; et al. Associations between ambient wood smoke and other particulate pollutants and biomarkers of systemic inflammation, coagulation and thrombosis in cardiac patients. Environ. Res. 2017, 154, 352–361. [Google Scholar] [CrossRef]
- Huttunen, K.; Siponen, T.; Salonen, I.; Yli-Tuomi, T.; Aurela, M.; Dufva, H.; Hillamo, R.; Linkola, E.; Pekkanen, J.; Pennanen, A.; et al. Low-level exposure to ambient particulate matter is associated with systemic inflammation in ischemic heart disease patients. Environ. Res. 2012, 116, 44–51. [Google Scholar] [CrossRef]
- Adetona, A.M.; Adetona, O.; Gogal, R.M.; Diaz-Sanchez, D.; Rathbun, S.L.; Naeher, L.P. Impact of Work Task-Related Acute Occupational Smoke Exposures on Select Proinflammatory Immune Parameters in Wildland Firefighters. J. Occup. Environ. Med. 2017, 59, 679–690. [Google Scholar] [CrossRef] [PubMed]
- Schwartz, J. Air Pollution and Blood Markers of Cardiovascular Risk. Environ. Health Perspect. 2001, 109, 405. [Google Scholar] [CrossRef] [PubMed]
- Ghio, A.J.; Soukup, J.M.; Case, M.; Dailey, L.A.; Richards, J.; Berntsen, J.; Devlin, R.B.; Stone, S.; Rappold, A. Exposure to wood smoke particles produces inflammation in healthy volunteers. Occup. Environ. Med. 2011, 69, 170–175. [Google Scholar] [CrossRef] [PubMed]
- Carlsten, C.; Salvi, S.; Wong, G.W.; Chung, K.F. Personal strategies to minimise effects of air pollution on respiratory health: Advice for providers, patients and the public. Eur. Respir. J. 2020, 55, 1902056. [Google Scholar] [CrossRef]

| Location and Year | Number of Assessments | Type of Smoke | Number of Assessments | Type of Smoke |
|---|---|---|---|---|
| Warburton 2013 | 14 | Planned burn | 10 | No smoke |
| Warburton 2014 | 44 | Wild fire | 39 | No smoke |
| Traralgon 2014 | 42 | Coal Mine Fire | 29 | No smoke |
| Maffra 2014 | 21 | No smoke | 14 | No smoke |
| Warburton 2015 | 7 | Planned burn | 78 | No smoke |
| Warburton 2016 | 55 | Planned burn | ||
| Total | 183 | 170 |
| Characteristic | Women Total (n = 110) | Proportions | Men Total (n = 73) | Proportions | p-Value |
|---|---|---|---|---|---|
| Age > 65 years | 40 | 36.0% | 44 | 60.3% | 0.001 |
| Current regular smoker | 15 | 13.6% | 3 | 4.1% | 0.034 |
| Smoked at all last month | 33 | 30.0% | 20 | 27.4% | 0.75 |
| Ever smoked | 41 | 37.2% | 32 | 43.8% | 0.72 |
| Asthma diagnosed | 17 | 15.5% | 12 | 16.4% | 0.86 |
| Asthma attack in the last 12 months | 6 | 5.5% | 0 | 0% | 0.028 * |
| Asthma medication | 10 | 9.1% | 8 | 11.0% | 0.97 |
| COPD | 4 | 3.6% | 4 | 5.6% | 0.80 |
| Other respiratory condition | 6 | 5.5% | 5 | 6.8% | 0.94 |
| Hypertension | 41 | 37.3% | 28 | 38.4% | 0.88 |
| Angina | 4 | 3.6% | 7 | 9.6% | 0.18 |
| High Cholesterol | 31 | 28.2% | 21 | 28.8% | 0.93 |
| Myocardial infarction or coronary event | 5 | 4.5% | 9.6% | 0.23 * | |
| Heart Failure | 1 | 0.9% | 5 | 6.8% | 0.039 * |
| Arrhythmia | 14 | 12.7% | 10 | 13.7% | 0.85 |
| Stroke or TIA | 2 | 1.8% | 5 | 6.8% | 0.12 * |
| Other heart condition | 3 | 2.7% | 8 | 11.0% | 0.028 * |
| Diabetes | 14 | 12.7% | 9 | 12.3% | 0.94 |
| Self-reported cold or flu symptoms | 18 | 16.5% | 10 | 13.7% | 0.61 |
| Immune modulators | 3 | 2.7% | 0 | 0.0% | - |
| Non-Steroidal anti-inflammatories | 2 | 1.8% | 1 | 1.4% | - |
| Anti-platelet medication | 8 | 7.2% | 8 | 10.9% | - |
| Asthma Inhalers/preventers | 10 | 9.1% | 8 | 10.9% | - |
| Antihistamines | 2 | 1.8% | 0 | 0.0% | - |
| Exposure Type | Lag Period (h) | β | 95% CI | p |
|---|---|---|---|---|
| All | 4 | 0.500 | (0.192 to 0.808) | <0.001 |
| Planned Burn Smoke | 4 | 0.335 | (−0.012 to 0.681) | 0.058 |
| Wildfire Smoke | 4 | 0.644 | (0.447 to 0.842) | <0.001 |
| Coal Mine Fire | 4 | 1.533 | (0.461 to 2.605) | 0.005 |
| All | 12 | 0.308 | (0.028 to 0.588) | 0.031 |
| Planned Burns Smoke | 12 | 0.269 | (−0.014 to 0.553) | 0.063 |
| Wildfire Smoke | 12 | 1.027 | (0.816 to 1.239) | <0.001 |
| Coal Mine Fire Smoke | 12 | 0.196 | (−1.467 to 1.859) | 0.817 |
| All | 24 | 0.381 | (−0.036 to 0.798) | 0.073 |
| Planned Burns Smoke | 24 | 0.497 | (−0.050 to 1.044) | 0.075 |
| Wildfire Smoke | 24 | 1.073 | (0.846 to 1.299) | <0.001 |
| Coal Mine Fire Smoke | 24 | 0.538 | (−1.431 to 2.506) | 0.593 |
| All | 48 | 0.344 | (−0.154 to 0.842) | 0.176 |
| Planned Burns Smoke | 48 | 0.761 | (−0.165 to 1.686) | 0.107 |
| Wildfire Smoke | 48 | 0.789 | (0.539 to 1.040) | <0.001 |
| Coal Mine Fire Smoke | 48 | −0.148 | (−2.150 to 1.854) | 0.885 |
| Exposure Type | Outcome | Exposure Period (h) | β | 95% CI | p |
|---|---|---|---|---|---|
| All | WCC | 24 | −0.088 | (−0.171 to −0.006) | 0.036 |
| Planned Burns | WCC | 24 | −0.069 | (−0.178 to 0.041) | 0.218 |
| Wildfire Smoke | WCC | 24 | −0.108 | (−0.235 to 0.018) | 0.094 |
| Coal Mine Fire | WCC | 24 | −0.203 | (−0.605 to 0.200) | 0.324 |
| All | WCC | 48 | −0.092 | (−0.192 to 0.007) | 0.070 |
| All | Neutrophils | 24 | −0.077 | (−0.144 to −0.010) | 0.024 |
| Planned Burns | Neutrophils | 24 | −0.065 | (−0.156 to 0.026) | 0.162 |
| Wildfire Smoke | Neutrophils | 24 | −0.083 | (−0.185 to 0.019) | 0.112 |
| Coal Mine Fire | Neutrophils | 24 | −0.147 | (−0.469 to 0.175) | 0.370 |
| All | Neutrophils | 48 | −0.081 | (−0.162 to −0.001) | 0.048 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
O’Dwyer, T.; Abramson, M.J.; Straney, L.; Salimi, F.; Johnston, F.; Wheeler, A.J.; O’Keeffe, D.; Haikerwal, A.; Reisen, F.; Hopper, I.; et al. Sub-Clinical Effects of Outdoor Smoke in Affected Communities. Int. J. Environ. Res. Public Health 2021, 18, 1131. https://doi.org/10.3390/ijerph18031131
O’Dwyer T, Abramson MJ, Straney L, Salimi F, Johnston F, Wheeler AJ, O’Keeffe D, Haikerwal A, Reisen F, Hopper I, et al. Sub-Clinical Effects of Outdoor Smoke in Affected Communities. International Journal of Environmental Research and Public Health. 2021; 18(3):1131. https://doi.org/10.3390/ijerph18031131
Chicago/Turabian StyleO’Dwyer, Thomas, Michael J. Abramson, Lahn Straney, Farhad Salimi, Fay Johnston, Amanda J. Wheeler, David O’Keeffe, Anjali Haikerwal, Fabienne Reisen, Ingrid Hopper, and et al. 2021. "Sub-Clinical Effects of Outdoor Smoke in Affected Communities" International Journal of Environmental Research and Public Health 18, no. 3: 1131. https://doi.org/10.3390/ijerph18031131
APA StyleO’Dwyer, T., Abramson, M. J., Straney, L., Salimi, F., Johnston, F., Wheeler, A. J., O’Keeffe, D., Haikerwal, A., Reisen, F., Hopper, I., & Dennekamp, M. (2021). Sub-Clinical Effects of Outdoor Smoke in Affected Communities. International Journal of Environmental Research and Public Health, 18(3), 1131. https://doi.org/10.3390/ijerph18031131








