Psychotherapeutic Interventions to Improve Psychological Adjustment in Type 1 Diabetes: A Systematic Review
Abstract
:1. Introduction
2. Materials and Methods
2.1. Elegibility Criteria
2.1.1. Selection of Studies
2.1.2. Data Extraction
2.2. Risk of Bias in Individual Studies
3. Results
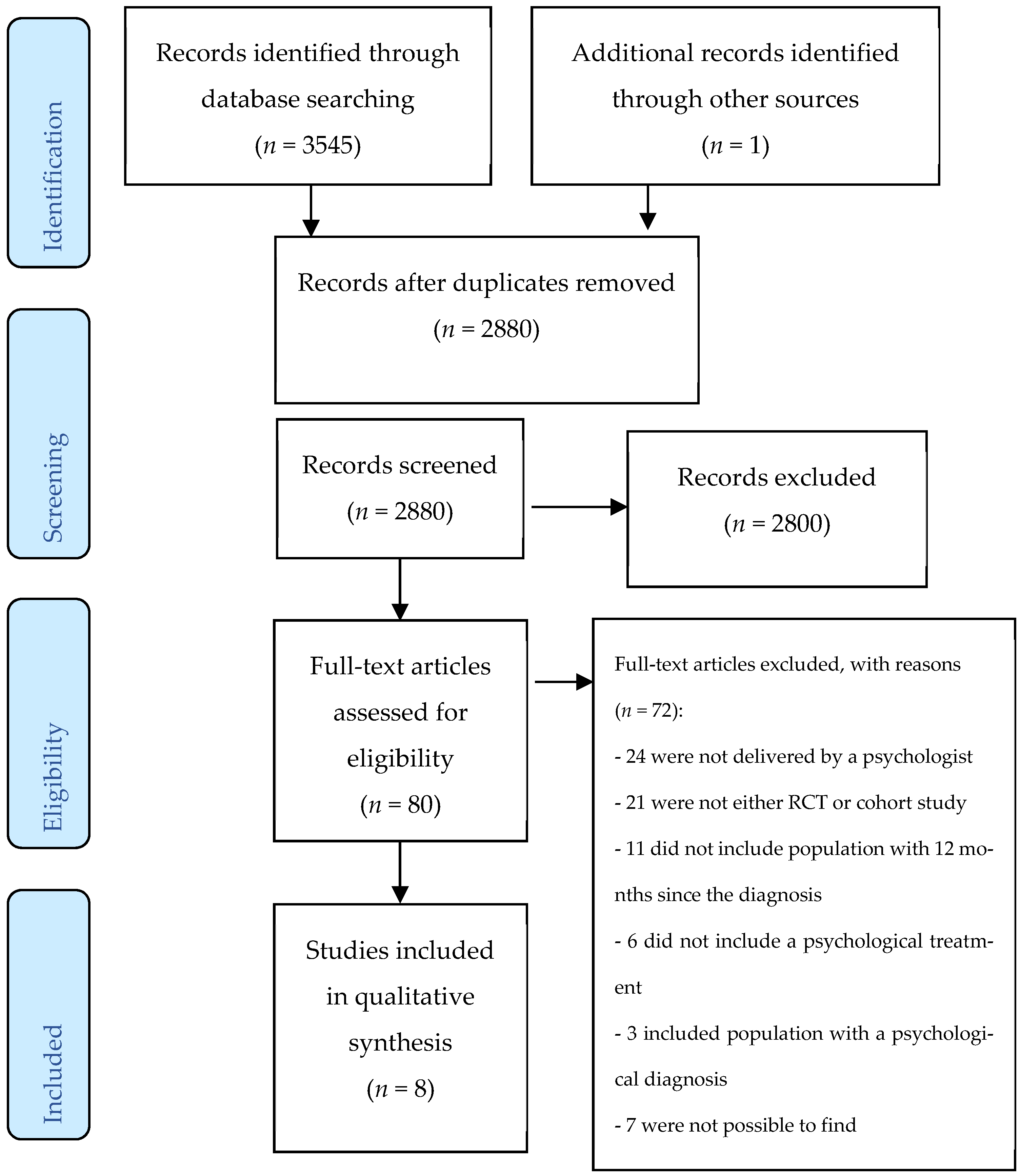
3.1. Search Results
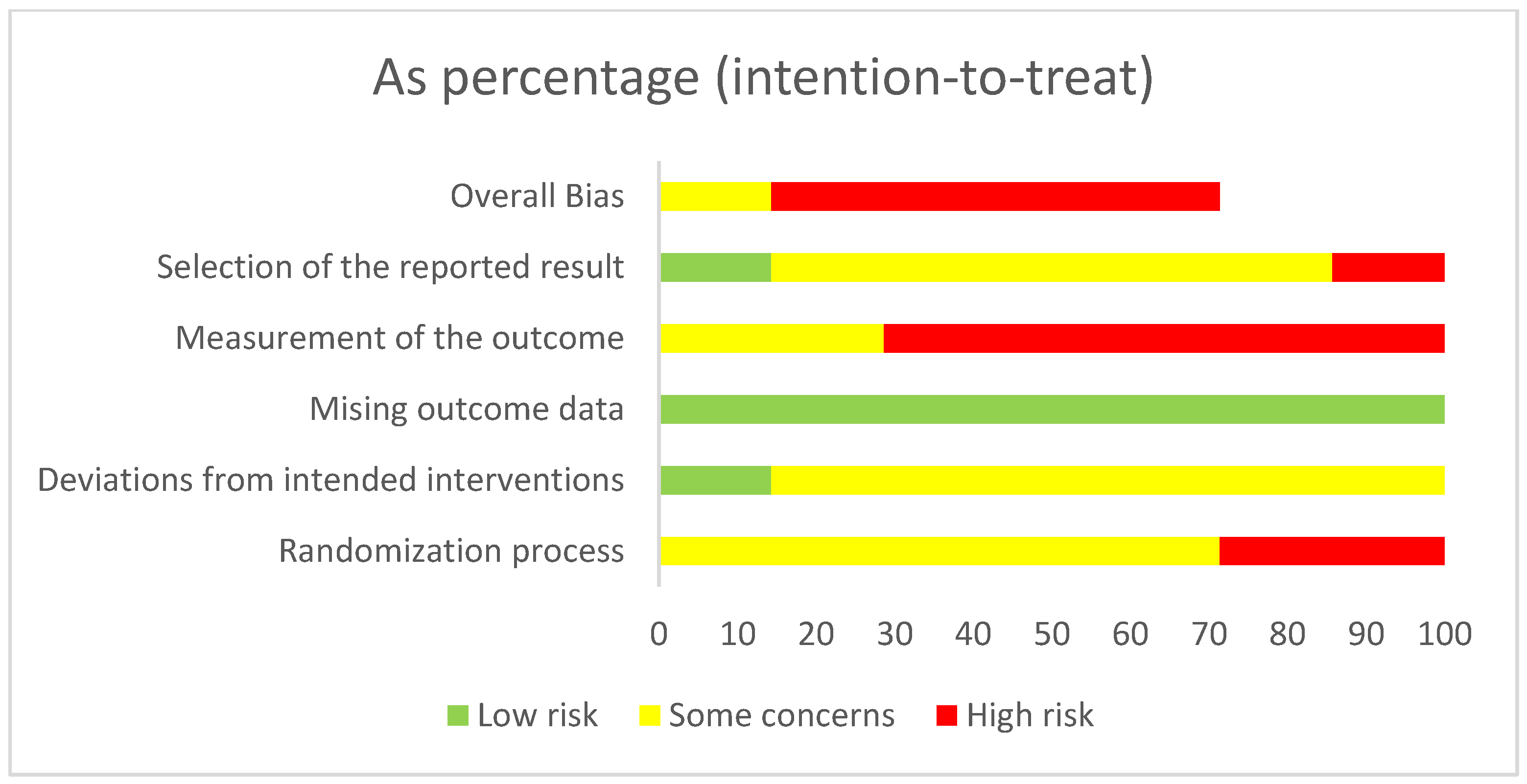
3.2. Study Quality
3.3. Synthesis of Results
3.4. Intervention
3.5. Outcomes
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Geremia, C.; Fornari, A.; Tschiedel, B. Comparison of the effect of a compact vs a conventional, long-term education program on metabolic control in children and adolescents with type 1 diabetes: A pilot, randomized clinical trial. Pediatr. Diabetes 2019, 20, 778–784. [Google Scholar] [CrossRef]
- International Diabetes Federation. IDF Diabetes Atlas, 9th ed.; International Diabetes Federation: Brussels, Belgium, 2019; Available online: https://www.diabetesatlas.org (accessed on 1 April 2019).
- Mobasseri, M.; Shirmohammadi, M.; Amiri, T.; Vahed, N.; Fard, H.H.; Ghojazadeh, M. Prevalence and incidence of type 1 diabetes in the world: A systematic review and meta-analysis. Health Promot. Perspect. 2020, 10, 98–115. [Google Scholar] [CrossRef] [PubMed]
- American Diabetes Association. Glycemic Targets: Standards of Medical Care in Diabetes. Diabetes Care 2021, 44, 73–84. [Google Scholar] [CrossRef]
- Esbitt, S.A.; Batchelder, A.W.; Tanenbaum, M.L.; Shreck, E. “Knowing That You’re Not the Only One”: Perspectives on Group-Based Cognitive-Behavioral Therapy for Adherence and Depression (CBT-AD) in Adults With Type 1 Diabetes. Cogn. Behav. Pract. 2015, 22, 393–406. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Magalhães, T.P.C.; Fóscolo, R.B.; Soares, A.N.; Reis, J.S. Type 1 diabetes mellitus: Can coaching improve health outcomes? Arch. Endocrinol. Metab. 2018, 62, 485–489. [Google Scholar] [CrossRef]
- Lansing, A.H.; Stoianova, M.; Stanger, C. Adolescent Emotional Control Moderates Benefits of a Multicomponent Intervention to Improve Type 1 Diabetes Adherence: A Pilot Randomized Controlled Trial. J. Pediatr. Psychol. 2019, 44, 126–136. [Google Scholar] [CrossRef] [PubMed]
- Ismail, K.; Maissi, E.; Thomas, S.; Chalder, T.; Schmidt, U.; Barlett, J.; Patel, A.; Dickens, C.; Creed, F.; Treasure, J. A randomised controlled trial of cognitive behaviour therapy and motivational interviewing for people with Type 1 diabetes mellitus with persistent sub-optimal glycemic control: A Diabetes and Psychological Therapies (ADaPT) study. Health Technol. Assess. 2010, 14, 1–101. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Baumeister, H.; Hutter, N.; Bengel, J.; Härter, M. Quality of life in medically ill persons with comorbid mental disorders: A systematic review and meta-analysis. Psychother. Psychosom. 2011, 80, 275–286. [Google Scholar] [CrossRef] [PubMed]
- Castellano-Guerrero, A.M.; Guerrero, R.; Relimpio, F.; Losada, F.; Mangas, M.A.; Pumar, A.; Martínez-Brocca, M.A. Prevalence and predictors of depression and anxiety in adult patients with type 1 diabetes in tertiary care setting. Acta Diabetol. 2018, 55, 943–953. [Google Scholar] [CrossRef]
- Strandberg, R.B.; Graue, M.; Wentzel-Larsen, T.; Peyrot, M.; Wahl, A.K.; Rokne, B. The relationships among fear of hypoglycaemia, diabetes-related quality of life and psychological well-being in Norwegian adults with Type 1 diabetes. Diabetes Res. Clin. Pract. 2017, 124, 11–19. [Google Scholar] [CrossRef]
- Mosaku, K.; Kolawole, B.; Mume, C.; Ikem, R. Depression, anxiety and quality of life among diabetic patients: A comparative study. J. Natl. Med. Assoc. 2008, 10, 73–78. [Google Scholar] [CrossRef]
- Martínez-Brocca, M.A.; Mayoral, E.; Irastorza, A.; Lama-Herrera, C.; Martínez-Concepción, E.; Sanz-Amores, R.; Gálvez-Moreno, M.A.; Ortega-Millán, C.; Luque-Romero, L.G.; Gómez-Huelgas, R.; et al. Plan Integral de Diabetes de Andalucía: Evaluación del II Plan Integral de Diabetes de Andalucía. Actualización 2016. Available online: https://www.repositoriosalud.es/handle/10668/2581 (accessed on 1 April 2020).
- American Diabetes Association. Standards of Medical Care in Diabetes. Abridged for Primary Care Providers. Clin. Diabetes 2018, 36, 14–37. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Serrano-Gil, M.; Jacob, S. Engaging and empowering patients to manage their type 2 diabetes, Part I: A knowledge, attitude, and practice gap? Adv. Ther. 2010, 27, 321–333. [Google Scholar] [CrossRef] [PubMed]
- Oldham-Cooper, R.; Semple, C.; Wilkinson, L.L. Reconsidering a role for attachment in eating disorder management in the context of paediatric diabetes. Clin. Child Psychol. Psychiatry 2021, 26, 669–681. [Google Scholar] [CrossRef]
- Ruiz-Aranda, D.; Zysberg, L.; García-Linares, E.; Castellano-Gerrero, A.M.; Martínez-Brocca, M.A.; Gutiérrez-Colosía, M.R. Emotional abilities and HbA1c levels in patients with type 1 diabetes. Psychoneuroendocrinology 2018, 93, 118–123. [Google Scholar] [CrossRef]
- Aljawarneh, Y.M.; Al-Qaissi, N.M.; Ghunaim, H.Y. Psychological interventions for adherence, metabolic control, and coping with stress in adolescents with type 1 diabetes: A systematic review. World J. Pediatr. 2020, 16, 456–470. [Google Scholar] [CrossRef]
- Ni, Y.X.; Ma, L.; Li, J.P. Effects of mindfulness-based intervention on glycemic control and psychological outcomes in people with diabetes: A systematic review and meta-analysis. J. Diabetes Investig. 2020, 16, 1092–1103. [Google Scholar] [CrossRef]
- Winkley, W.; Upsher, R.; Stahl, D.; Pollard, D.; Brennan, A.; Heller, S.; Ismail, K. Systematic review and meta-analysis of randomized controlled trials of psychological interventions to improve glycemic control in children and adults with type 1 diabetes. Diabet. Med. 2020, 37, 735–746. [Google Scholar] [CrossRef]
- Ayling, K.; Brierley, S.; Johnson, B.; Heller, S.; Eiser, C. Efficacy of theory-based interventions for young people with type 1 diabetes: A systematic review and meta-analysis. Br. J. Health Psychol. 2015, 20, 428–446. [Google Scholar] [CrossRef]
- Sturt, J.; Dennick, K.; Hessler, D.; Hunter, B.M.; Oliver, J.; Fisher, L. Effective interventions for reducing Diabetes Distress: Systematic review and meta-analysis. Int. Diabetes Nurs. 2015, 12, 40–55. [Google Scholar] [CrossRef] [Green Version]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; The Prisma Group. Preferred Reporting Items for Systematic Reviews and Meta-Analyses: The PRISMA Statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhong, T.; Tang, R.; Gong, S.; Li, J.; Li, X.; Zhou, Z. The remission phase in type 1 diabetes: Changing epidemiology, definitions, and emerging immuno- metabolic mechanisms. Diabetes Metab. Res. Rev. 2019, 36, e3207. [Google Scholar] [CrossRef] [PubMed]
- Sterne, J.A.C.; Savović, J.; Page, M.J.; Elbers, R.G.; Blencowe, N.S.; Boutron, I.; Cates, C.J.; Cheng, H.-Y.; Corbett, M.S.; Eldridge, S.M.; et al. RoB 2: A revised tool for assessing risk of bias in randomised trials. BMJ 2019, 366, l4898. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wells, G.; Shea, B.; O’Connell, D.; Peterson, J.; Welch, V.; Losos, M.; Tugwell, P. The Newcastle-Ottawa Scale (NOS) for Assessing the Quality of Nonrandomised Studies in Meta-Analyses. Available online: http://www.ohri.ca/programs/clinical_epidemiology/oxford.asp (accessed on 1 May 2019).
- Forlani, G.; Nuccitelli, C.; Caselli, C.; Moscatiello, S.; Mazzotti, A.; Centis, E.; Marchesini, G. A psychological support program for individuals with Type 1 diabetes. Acta Diabetol. 2013, 50, 209–216. [Google Scholar] [CrossRef]
- Amsberg, S.; Anderbro, T.; Wredling, R.; Lisspers, J.; Lins, P.-E.; Adamson, U.; Johansson, U.-B. A cognitive behavior therapy-based intervention among poorly controlled adult type 1 diabetes patients--a randomized controlled trial. Patient Educ. Couns. 2009, 77, 72–80. [Google Scholar] [CrossRef] [PubMed]
- Ellis, D.A.; Frey, M.A.; Naar-King, S.; Templin, T.; Cunningham, P.B.; Cakan, N. The effects of multisystemic therapy on diabetes stress among adolescents with chronically poorly controlled type 1 diabetes: Findings from a randomized, controlled trial. Pediatrics 2005, 116, e826–e832. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Serlachius, A.S.; Scratch, S.E.; Northam, E.A.; Frydenberg, E.; Lee, K.J.; Cameron, F.J. A randomized controlled trial of cognitive behaviour therapy to improve glycaemic control and psychosocial wellbeing in adolescents with type 1 diabetes. J. Health Psychol. 2016, 21, 1157–1169. [Google Scholar] [CrossRef] [PubMed]
- Channon, S.J.; Huws-Thomas, M.V.; Rollnick, S.; Hood, K.; Cannings-John, R.L.; Rogers, C.; Gregory, J.W. A multicenter randomized controlled trial of motivational interviewing in teenagers with diabetes. Diabetes Care 2007, 30, 1390–1395. [Google Scholar] [CrossRef] [Green Version]
- van Der Ven, N.C.; Hogenelst, M.H.; Tromp-Wever, A.M.; Twisk, J.W.R.; van Der Ploeg, H.M.; Heine, R.J.; Snoek, F.J. Short-term effects of cognitive behavioural group training (CBGT) in adult Type 1 diabetes patients in prolonged poor glycaemic control. A randomized controlled trial. Diabet. Med. 2005, 22, 1619–1623. [Google Scholar] [CrossRef]
- Fisher, L.; Hessler, D.; Polonsky, W.H.; Masharani, U.; Guzman, S.; Bowyer, V.; Strycker, L.; Ahmann, A.; Basina, M.; Blumer, I.; et al. T1-REDEEM: A Randomized Controlled Trial to Reduce Diabetes Distress Among Adults With Type 1 Diabetes. Diabetes Care 2018, 41, 1862–1869. [Google Scholar] [CrossRef] [Green Version]
- Menting, J.; Tack, C.J.; van Bon, A.C.; Jansen, H.J.; van den Bergh, J.P.; Mol, M.J.T.M.; Goedendorp, M.M.; Donders, R.; Knoop, H. Web-based cognitive behavioural therapy blended with face-to-face sessions for chronic fatigue in type 1 diabetes: A multicentre randomised controlled trial. Lancet. Diabetes Endocrinol. 2017, 5, 448–456. [Google Scholar] [CrossRef]
- Ciechanowski, P.S.; Katon, W.J.; Russo, J.E.; Walker, E.A. The patient-provider relationship: Attachment theory and adherence to treatment in diabetes. Am. J. Psychiatry 2001, 158, 29–35. [Google Scholar] [CrossRef]
- Ockene, J.K.; Ashe, K.; Peterson, K.S.; Fitzgibbon, M.; Buscemi, J.; Dulin, A. Society of Behavioral Medicine Call to Action: Include obesity/overweight management education in health professional curricula and provide coverage for behavior-based treatments of obesity/overweight most commonly provided by psychologists, dieticians, counselors, and other health care professionals and include such providers on all multidisciplinary teams treating patients who have overweight or obesity. Transl. Behav. Med. 2021, 11, 653–655. [Google Scholar] [CrossRef] [Green Version]
- Rich, K.; Murray, K.; Smith, H.; Jelbart, N. Interprofessional practice in health: A qualitative study in psychologists, exercise physiologists, and dietitians. J. Interprof. Care 2021, 35, 682–690. [Google Scholar] [CrossRef]
- García-Llana, H.; Barbero, J.; Olea, T.; Jiménez, C.; Del Peso, G.; Miguel, J.L.; Sánchez, R.; Celadilla, O.; Trocoli, F.; Argüello, M.T.; et al. Incorporación de un psicólogo en un servicio de nefrología: Criterios y proceso. [Incorporation of a psychologist into a nephrology service: Criteria and process]. Nefrologia 2010, 30, 297–303. [Google Scholar] [CrossRef]
- Coccaro, E.F.; Lazaruz, S.; Joseph, J.; Wyne, K.; Drossos, T.; Phillipson, L.; de Groot, M. Emotional regulation and diabetes distress in adults with type 1 and type 2 diabetes. Diabetes Care 2021, 44, 20–25. [Google Scholar] [CrossRef]
- Hilliard, M.E.; Yi-Frazier, J.P.; Hessler, D.; Butler, A.M.; Anderson, B.J.; Jaser, S. Stress and A1c among people with diabetes across the lifespan. Curr. Diab. Rep. 2016, 16, 67. [Google Scholar] [CrossRef] [Green Version]
- Karahan, T.F.; Yalcin, B.M. The effects of an emotional intelligence skills training program on anxiety, burnout and glycemic control in type 2 diabetes mellitus patients. Turk. Klin. J. Med. Sci. 2009, 29, 16–24. [Google Scholar]
- American Diabetes Association. Glycemic Targets: Standards of Medical Care in Diabetes. Diabetes Care 2020, 43, 66–76. [Google Scholar] [CrossRef]
| Aspects Considered | Inclusion Criteria | Exclusion Criteria |
|---|---|---|
| Population | Adolescents and adults with type 1 diabetes mellitus with at least one year since diagnosis and insulin treatment | Presenting any severe medical or psychological condition, less than a year of diabetes diagnosis or not having an insulin treatment. |
| Outcome | Quality of life and glycemic control | The study only assesses metabolic variables or other variables. The independent effect of the intervention could not be determined (e.g., combining medication and psychological intervention). |
| Design | Randomized controlled trials, prospective cohorts with control group. | Qualitative studies, systematic reviews and/or meta-analysis, protocols, clinical cases, and editors’ letters, cross-sectional studies, retrospective cohorts. |
| Type of intervention | Psychological interventions with any emotional component, delivered in group or individual format. Interventions delivered by a psychologist accompanied or not by another specialist | Interventions delivered by any specialist without a psychologist. |
| Language | All languages | None |
| Setting | All settings are included | None |
| Comparator | Waiting list, usual care, or any active control | No comparator. |
| First Author, (Year), Country | Sample (Control/Intervention) | Years Mean (SD) | Mean (SD) Diabetes Duration, Years | Study Conditions: 1. Intervention 2. Control | Intervention Duration; Format; Provider | Follow-Up Assessment | Primary Outcome Metabolic; Psychological |
|---|---|---|---|---|---|---|---|
| Ellis et al. [29], USA | 127 (63/64) | Adolescent population I: 13.4 (1.9) C: 13.1 (2.0) | I: 5.3 (3.9) C: 5.2 (4.8) | 1. Multisystemic therapy 2. Standard care | 6 months, 2–3 times per week; Home-based family sessions; Therapist | Month 7 | Month 7
|
| Van der Ven et al. [32], The Netherlands | 88 (45/43) | Adult population 37.8 (10.6) | 18.0 (10.4) | 1. Cognitive Behavior Therapy (CBT) based group training (CBGT) 2. Dutch adaptation of blood glucose awareness training (BGAT) | 6 weekly 2 h sessions; group sessions (6–8p); diabetes nurse and psychologist | Month 3 | Month 3
Self-efficacy (CIDS) Depressive symptoms (CES-D)
|
| Channon et al. [31], UK | 66 (38/28) | Adolescent population I: 15.3 (0.97) C: 15.4 (1.19) | I: 9.2 (1.96) C: 9.1 (1.47) | 1. Motivational interviewing 2. Support visits | 12 months; individual frequency, home-based; health psychologist trainee | Months 12 and 24 | Month 12
Quality of life (DQoLY)—satisfaction, impact, worries subscales Wellbeing (WBK)—depression, anxiety, positive wellbeing subscales Personal models of illness (PMDQ) Month 24
Quality of life (DQoLY)—satisfaction, impact subscales Wellbeing (WBK)—anxiety subscale Personal models of illness (PMDQ)—life worry subscale |
| Amsberg et al. [28], Sweden | 74 (36/38) | Adult population I: 41.1 (11.7) C: 41.4 (12.9) | I: 19.9 (9.4) C: 23.2 (11.8) | 1. CBT based intervention 2. Continuous glucose monitoring system | 8 weekly 2 h sessions; group (4–6p) and individual; diabetes nurse and psychologist | Months 6 and 12 | Month 6
Diabetes distress (PAID) Month 12
Diabetes distress (PAID) Wellbeing (W-BQ12) Anxiety (HAD) Depression (HAD) Perceived Stress (PSS) |
| * Forlani et al. [27], Italy | 55 (33/22) | Adult population I: 40.7 (12.0) C: 39.4 (12.7) | I: 16.6 (11.5) C: 16.6 (8.7) | 1. Psychological support program 2. Waiting list | 7 weekly 2 h sessions; groups (8–12p); psychologist | Month 6 | Month 6
Depression (BDI) Anxiety(SAS) Wellbeing (WED) |
| Serlachius et al. [30], Australia | 104 (30/74) | Adolescent population I: 14.6 (1.16) C: 14.3 (1.12) | I: 5.97 (3.12) C: 6.12 (3.80) | 1. CBT based program 2. Standard care | 5 weekly 2 h sessions; group sessions; health psychologist | Months 3 and 12 | Month 3
Self-efficacy (SED) Quality of life (DQoL) Month 12
Quality of life (DQoL)
|
| Menting et al. [34], The Netherlands | 120 (60/60) | Adult population I: 44.4 (12.1) C: 42.9 (12.5) | I: 24.2 (13.3) C: 24.1 (13.9) | 1. CBT 2. Waiting list | 5 months; 5–8 individual face-to-face sessions + web-based modules; clinical psychologists | Month 6 | Month 6
Functional impairment |
| Fisher et al. [33], USA | 301 (Knowlt 149/ Ontrack 152) | Adult population Knowlt: 47.3 (14.5) Ontrack: 42.8 (15.1) | Knowlt: 26.1 (14.0) Ontrack: 23.2 (13.3) | 1. Improving emotion regulation skills (OnTrack) 2.Education/behavior change intervention (Knowlt) | 3 months; 1-day group + 4 online videos; psychologist (OnTrack) diabetes nurse (Knowlt) | Month 9 | Month 9
Diabetes distress (T1-DDS) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Resurrección, D.M.; Navas-Campaña, D.; Gutiérrez-Colosía, M.R.; Ibáñez-Alfonso, J.A.; Ruiz-Aranda, D. Psychotherapeutic Interventions to Improve Psychological Adjustment in Type 1 Diabetes: A Systematic Review. Int. J. Environ. Res. Public Health 2021, 18, 10940. https://doi.org/10.3390/ijerph182010940
Resurrección DM, Navas-Campaña D, Gutiérrez-Colosía MR, Ibáñez-Alfonso JA, Ruiz-Aranda D. Psychotherapeutic Interventions to Improve Psychological Adjustment in Type 1 Diabetes: A Systematic Review. International Journal of Environmental Research and Public Health. 2021; 18(20):10940. https://doi.org/10.3390/ijerph182010940
Chicago/Turabian StyleResurrección, Davinia M., Desirée Navas-Campaña, Mencía R. Gutiérrez-Colosía, Joaquín A. Ibáñez-Alfonso, and Desireé Ruiz-Aranda. 2021. "Psychotherapeutic Interventions to Improve Psychological Adjustment in Type 1 Diabetes: A Systematic Review" International Journal of Environmental Research and Public Health 18, no. 20: 10940. https://doi.org/10.3390/ijerph182010940
APA StyleResurrección, D. M., Navas-Campaña, D., Gutiérrez-Colosía, M. R., Ibáñez-Alfonso, J. A., & Ruiz-Aranda, D. (2021). Psychotherapeutic Interventions to Improve Psychological Adjustment in Type 1 Diabetes: A Systematic Review. International Journal of Environmental Research and Public Health, 18(20), 10940. https://doi.org/10.3390/ijerph182010940









