Weather Variability and COVID-19 Transmission: A Review of Recent Research
Abstract
1. Introduction
2. Materials and Methods
2.1. Search Strategy
2.2. Inclusion and Exclusion Criteria
2.3. Quality Assessment and Data Analysis
3. Results
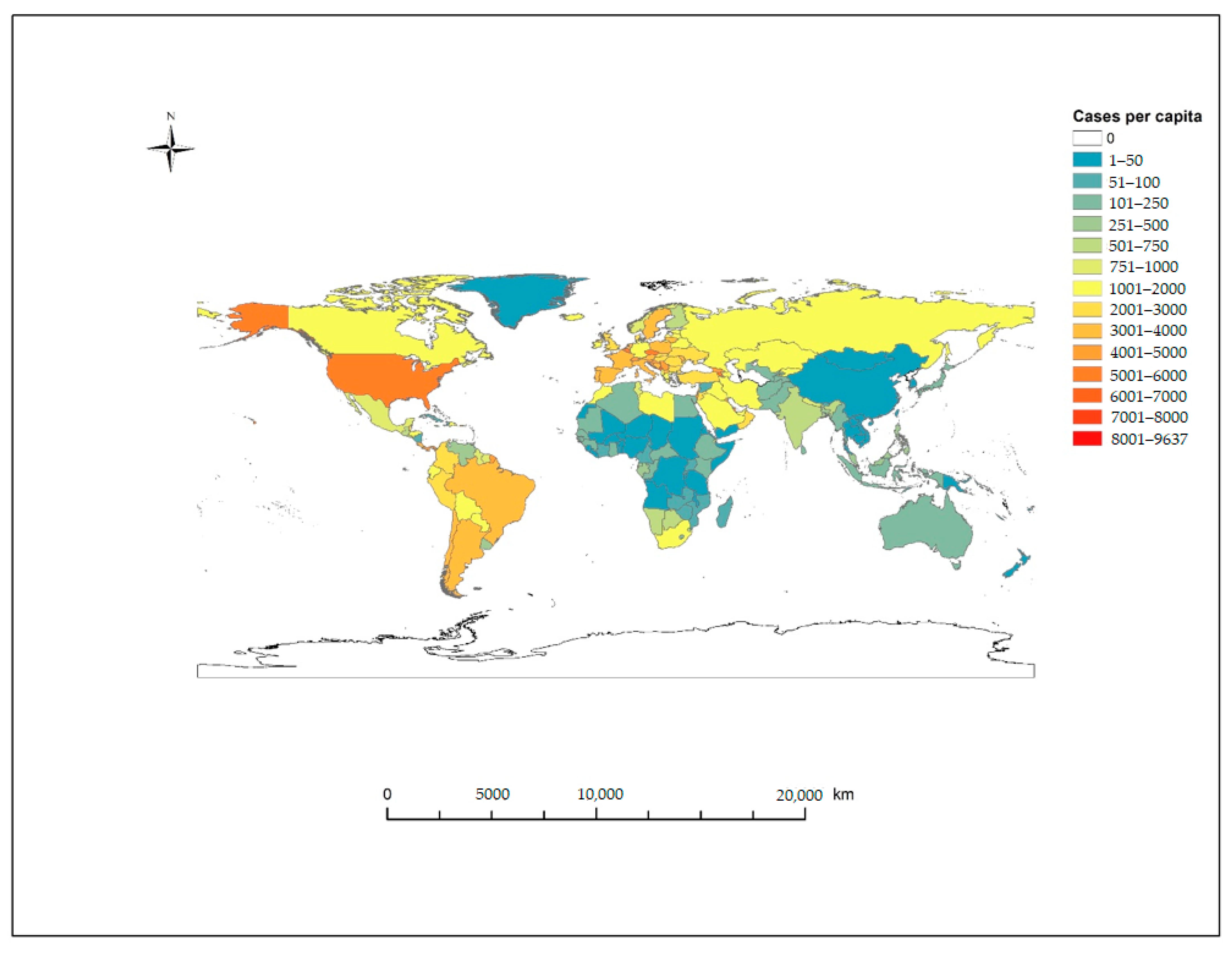
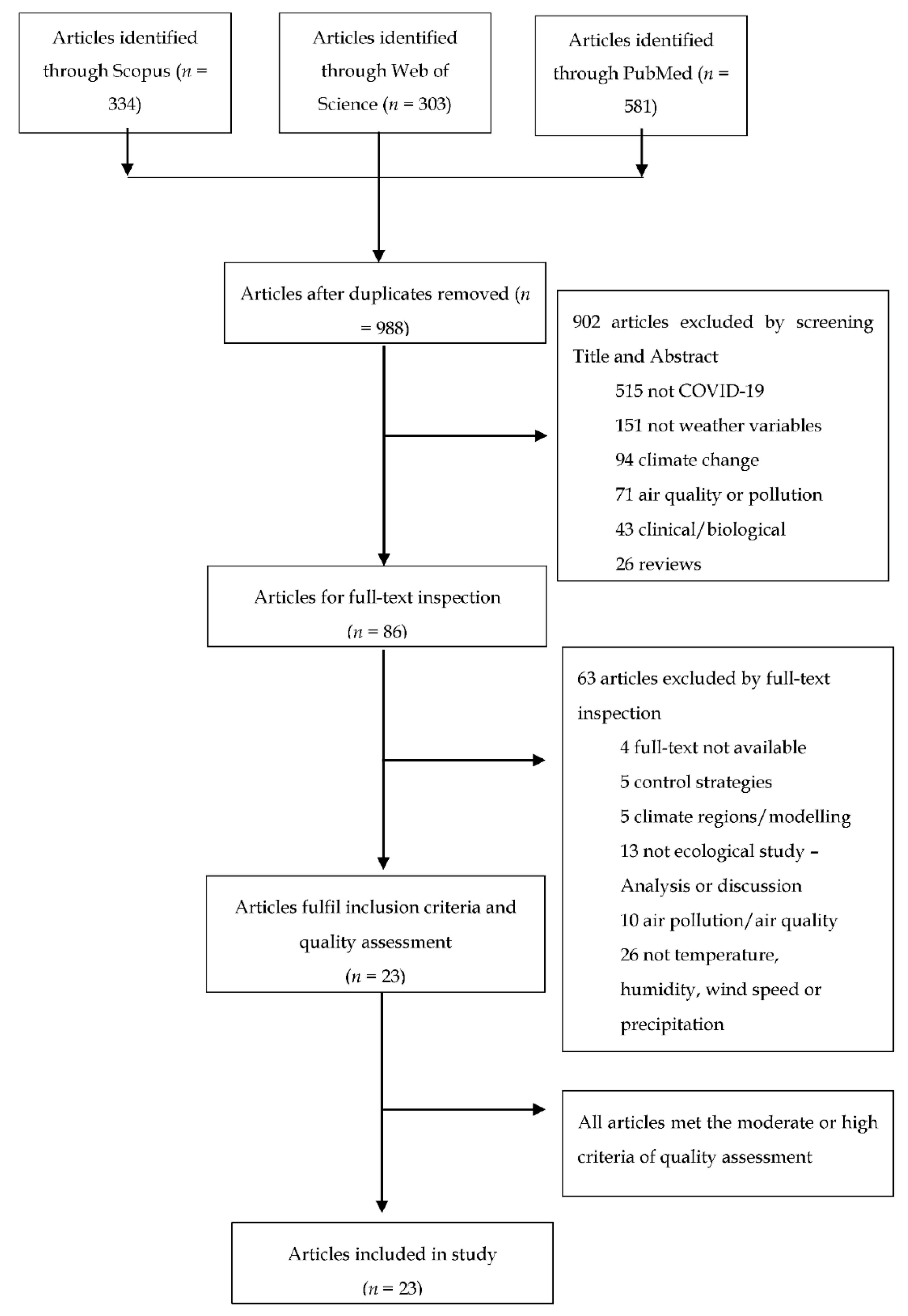
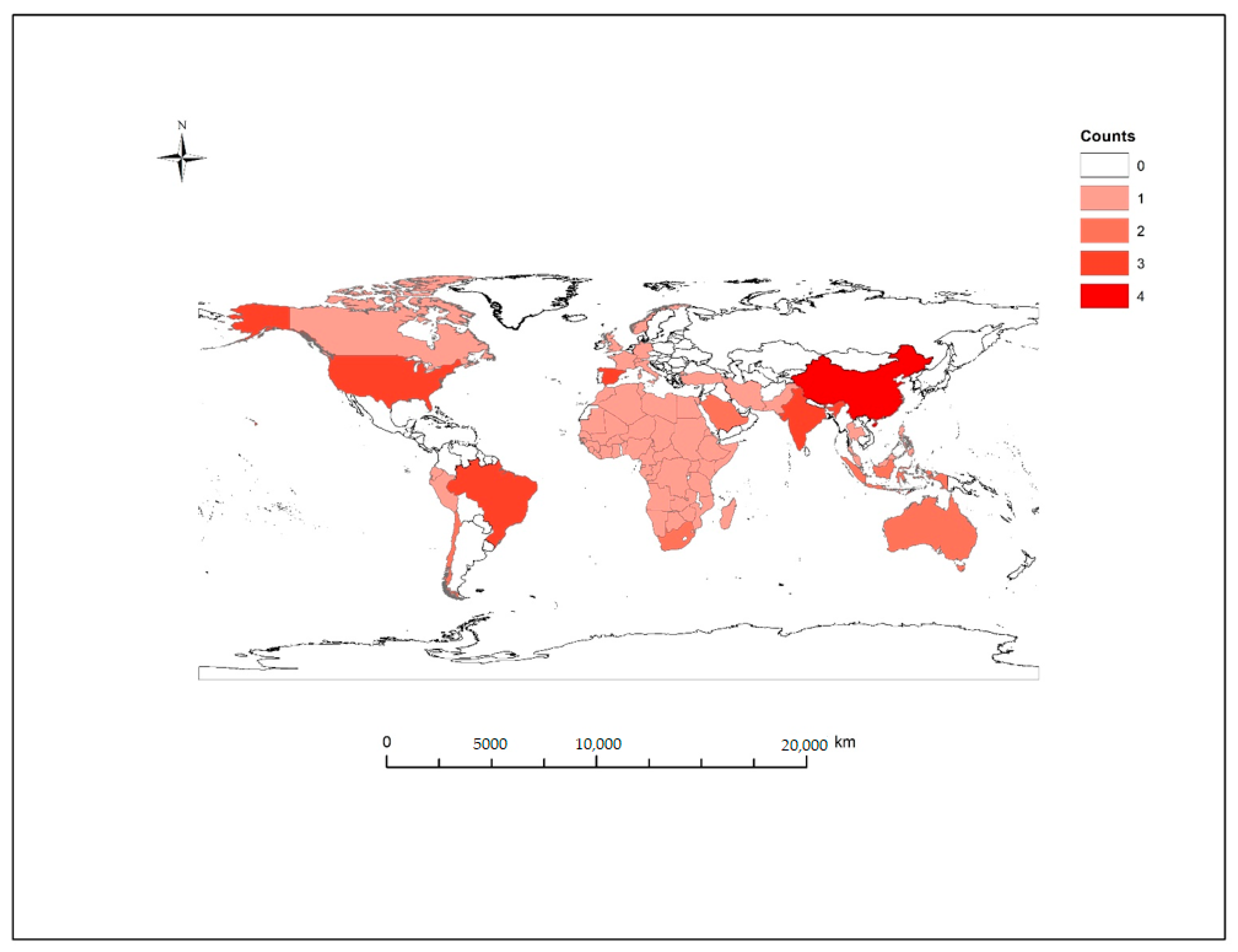
3.1. Literature Search
3.2. Study Characteristics
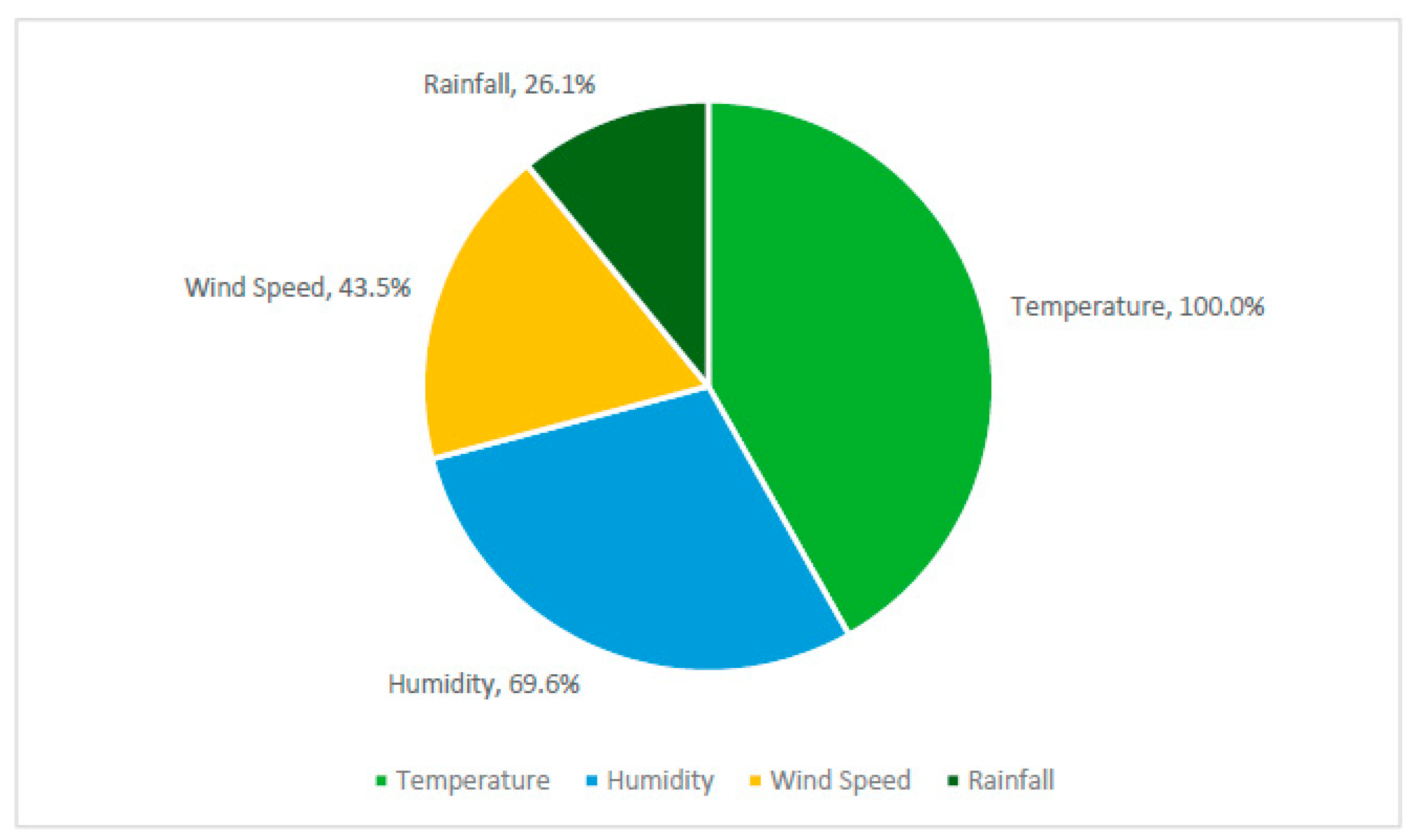
3.3. Weather Variables and COVID-19
3.3.1. Temperature
3.3.2. Humidity
3.3.3. Wind Speed
3.3.4. Precipitation
4. Discussion
4.1. Weather Variables and COVID-19
4.2. Strengths and Limitations
4.3. Recommendations for Future Research
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Allam, Z. The First 50° days of COVID-19: A Detailed Chronological Timeline and Extensive Review of Literature Documenting the Pandemic. In Surveying the Covid-19 Pandemic and its Implications; Elsevier: Amsterdam, The Netherlands, 2020; pp. 1–7. [Google Scholar] [CrossRef]
- World Health Organization. Timeline of WHO’s Response to COVID-19. Available online: https://www.who.int/news/item/29-06-2020-covidtimeline (accessed on 10 October 2020).
- Dong, E.; Du, H.; Gardner, L. An interactive web-based dashboard to track COVID-19 in real time. Lancet. Infect. Dis. 2020, 20, 533–534. [Google Scholar] [CrossRef]
- Sohrabi, C.; Alsafi, Z.; O’Neill, N.; Khan, M.; Kerwan, A.; Al-Jabir, A.; Iosifidis, C.; Agha, R. World Health Organization declares global emergency: A review of the 2019 novel coronavirus (COVID-19). Int. J. Surg. 2020, 76, 71–76. [Google Scholar] [CrossRef] [PubMed]
- Lan, J.; Ge, J.; Yu, J.; Shan, S.; Zhou, H.; Fan, S.; Zhang, Q.; Shi, X.; Wang, Q.; Zhang, L.; et al. Structure of the SARS-CoV-2 spike receptor-binding domain bound to the ACE2 receptor. Nature 2020, 581, 215–220. [Google Scholar] [CrossRef] [PubMed]
- Keni, R.; Alexander, A.; Nayak, P.G.; Mudgal, J.; Nandakumar, K. COVID-19: Emergence, Spread, Possible Treatments, and Global Burden. Front. Public Health 2020, 8, 216. [Google Scholar] [CrossRef]
- Carotti, M.; Salaffi, F.; Sarzi-Puttini, P.; Agostini, A.; Borgheresi, A.; Minorati, D.; Galli, M.; Marotto, D.; Giovagnoni, A. Chest CT features of coronavirus disease 2019 (COVID-19) pneumonia: Key points for radiologists. Radiol. Med. 2020, 125, 636–646. [Google Scholar] [CrossRef] [PubMed]
- Ge, H.; Wang, X.; Yuan, X.; Xiao, G.; Wang, C.; Deng, T.; Yuan, Q.; Xiao, X. The epidemiology and clinical information about COVID-19. Eur. J. Clin. Microbiol. Infect. Dis. 2020, 39, 1011–1019. [Google Scholar] [CrossRef]
- Lee, E.Y.P.; Ng, M.-Y.; Khong, P.-L. COVID-19 pneumonia: What has CT taught us? Lancet Infect. Dis. 2020, 20, 384–385. [Google Scholar] [CrossRef]
- Clark, A.; Jit, M.; Warren-Gash, C.; Guthrie, B.; Wang, H.H.; Mercer, S.W.; Sanderson, C.; McKee, M.; Troeger, C.; Ong, K.L. Global, regional, and national estimates of the population at increased risk of severe COVID-19 due to underlying health conditions in 2020: A modelling study. Lancet Glob. Health 2020, 8, e1003–e1017. [Google Scholar] [CrossRef]
- Sobral, M.F.F.; Duarte, G.B.; da Penha Sobral, A.I.G.; Marinho, M.L.M.; de Souza Melo, A. Association between climate variables and global transmission oF SARS-CoV-2. Sci. Total Environ. 2020, 729, 138997. [Google Scholar] [CrossRef]
- Xu, Z.W.; Li, Z.J.; Hu, W.B. Global dynamic spatiotemporal pattern of seasonal influenza since 2009 influenza pandemic. Infect. Dis. Poverty 2020, 9, 2. [Google Scholar] [CrossRef]
- Smit, A.J.; Fitchett, J.M.; Engelbrecht, F.A.; Scholes, R.J.; Dzhivhuho, G.; Sweijd, N.A. Winter Is Coming: A Southern Hemisphere Perspective of the Environmental Drivers of SARS-CoV-2 and the Potential Seasonality of COVID-19. Int. J. Environ. Res. Public Health 2020, 17, 5634. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Ye, C.; Yu, J.; Zhu, W.; Wang, Y.; Li, Z.; Xu, Z.; Cheng, J.; Wang, N.; Hao, L.; et al. The complex associations of climate variability with seasonal influenza A and B virus transmission in subtropical Shanghai, China. Sci. Total Environ. 2020, 701, 134607. [Google Scholar] [CrossRef] [PubMed]
- Taubenberger, J.K. The origin and virulence of the 1918 “Spanish” influenza virus. Proc. Am. Philos. Soc. 2006, 150, 86–112. [Google Scholar]
- Taubenberger, J.K.; Morens, D.M. 1918 Influenza: The mother of all pandemics. Emerg. Infect. Dis. 2006, 12, 15–22. [Google Scholar] [CrossRef] [PubMed]
- More, A.F.; Loveluck, C.P.; Clifford, H.; Handley, M.J.; Korotkikh, E.V.; Kurbatov, A.V.; McCormick, M.; Mayewski, P.A. The Impact of a Six-Year Climate Anomaly on the “Spanish Flu” Pandemic and WWI. GeoHealth 2020, 4, e2020GH000277. [Google Scholar] [CrossRef]
- Park, J.E.; Son, W.S.; Ryu, Y.; Choi, S.B.; Kwon, O.; Ahn, I. Effects of temperature, humidity, and diurnal temperature range on influenza incidence in a temperate region. Influenza Other Respir. Viruses 2020, 14, 11–18. [Google Scholar] [CrossRef]
- Tamerius, J.; Nelson, M.I.; Zhou, S.Z.; Viboud, C.; Miller, M.A.; Alonso, W.J. Global influenza seasonality: Reconciling patterns across temperate and tropical regions. Environ. Health Perspect. 2011, 119, 439–445. [Google Scholar] [CrossRef]
- Shaman, J.; Kohn, M. Absolute humidity modulates influenza survival, transmission, and seasonality. Proc. Natl. Acad. Sci. USA 2009, 106, 3243–3248. [Google Scholar] [CrossRef]
- Liu, H.; Zhang, Y.; Tian, Y.; Zheng, Y.; Gou, F.; Yang, X.; He, J.; Liu, X.; Meng, L.; Hu, W. Epidemic features of seasonal influenza transmission among eight different climate zones in Gansu, China. Environ. Res. 2020, 183, 109189. [Google Scholar] [CrossRef]
- Roussel, M.; Pontier, D.; Cohen, J.-M.; Lina, B.; Fouchet, D. Quantifying the role of weather on seasonal influenza. BMC Public Health 2016, 16, 441. [Google Scholar] [CrossRef]
- Towers, S.; Chowell, G.; Hameed, R.; Jastrebski, M.; Khan, M.; Meeks, J.; Mubayi, A.; Harris, G. Climate change and influenza: The likelihood of early and severe influenza seasons following warmer than average winters. PLoS Curr. 2013, 5. [Google Scholar] [CrossRef] [PubMed]
- Simonsen, L.; Gog, J.R.; Olson, D.; Viboud, C. Infectious Disease Surveillance in the Big Data Era: Towards Faster and Locally Relevant Systems. J. Infect. Dis. 2016, 214, S380–S385. [Google Scholar] [CrossRef]
- Zhang, Y.; Yakob, L.; Bonsall, M.B.; Hu, W. Predicting seasonal influenza epidemics using cross-hemisphere influenza surveillance data and local internet query data. Sci. Rep. 2019, 9, 3262. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Bambrick, H.; Mengersen, K.; Tong, S.; Hu, W. Using Google Trends and ambient temperature to predict seasonal influenza outbreaks. Environ. Int. 2018, 117, 284–291. [Google Scholar] [CrossRef] [PubMed]
- Bansal, S.; Chowell, G.; Simonsen, L.; Vespignani, A.; Viboud, C. Big Data for Infectious Disease Surveillance and Modeling. J. Infect. Dis. 2016, 214, S375–S379. [Google Scholar] [CrossRef]
- Wang, N.; Mengersen, K.; Kimlin, M.; Zhou, M.; Tong, S.; Fang, L.; Wang, B.; Hu, W. Lung cancer and particulate pollution: A critical review of spatial and temporal analysis evidence. Environ. Res. 2018, 164, 585–596. [Google Scholar] [CrossRef]
- Bukhari, Q.; Massaro, J.M.; D’Agostino, R.B., Sr.; Khan, S. Effects of Weather on Coronavirus Pandemic. Int. J. Environ. Res. Public Health 2020, 17, 5399. [Google Scholar] [CrossRef]
- Huang, Z.; Huang, J.; Gu, Q.; Du, P.; Liang, H.; Dong, Q. Optimal temperature zone for the dispersal of COVID-19. Sci. Total Environ. 2020, 736, 139487. [Google Scholar] [CrossRef]
- Wu, Y.; Jing, W.; Liu, J.; Ma, Q.; Yuan, J.; Wang, Y.; Du, M.; Liu, M. Effects of temperature and humidity on the daily new cases and new deaths of COVID-19 in 166 countries. Sci. Total Environ. 2020, 729, 139051. [Google Scholar] [CrossRef]
- Meyer, A.; Sadler, R.; Faverjon, C.; Cameron, A.R.; Bannister-Tyrrell, M. Evidence That Higher Temperatures Are Associated With a Marginally Lower Incidence of COVID-19 Cases. Front. Public Health 2020, 8, 367. [Google Scholar] [CrossRef]
- Bashir, M.F.; Ma, B.; Bilal; Komal, B.; Bashir, M.A.; Tan, D.; Bashir, M. Correlation between climate indicators and COVID-19 pandemic in New York, USA. Sci. Total Environ. 2020, 728, 138835. [Google Scholar] [CrossRef] [PubMed]
- To, T.; Zhang, K.; Maguire, B.; Terebessy, E.; Fong, I.; Parikh, S.; Zhu, J. Correlation of ambient temperature and COVID-19 incidence in Canada. Sci. Total Environ. 2020, 750, 141484. [Google Scholar] [CrossRef] [PubMed]
- Chien, L.C.; Chen, L.W. Meteorological impacts on the incidence of COVID-19 in the U.S. Stoch. Environ. Res. Risk Assess. 2020, 34, 1675–1680. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Zhou, J.; Yao, J.; Zhang, X.; Li, L.; Xu, X.; He, X.; Wang, B.; Fu, S.; Niu, T.; et al. Impact of meteorological factors on the COVID-19 transmission: A multi-city study in China. Sci. Total Environ. 2020, 726, 138513. [Google Scholar] [CrossRef]
- Xie, J.; Zhu, Y. Association between ambient temperature and COVID-19 infection in 122 cities from China. Sci. Total Environ. 2020, 724, 138201. [Google Scholar] [CrossRef]
- Qi, H.; Xiao, S.; Shi, R.; Ward, M.P.; Chen, Y.; Tu, W.; Su, Q.; Wang, W.; Wang, X.; Zhang, Z. COVID-19 transmission in Mainland China is associated with temperature and humidity: A time-series analysis. Sci. Total Environ. 2020, 728, 138778. [Google Scholar] [CrossRef]
- Shi, P.; Dong, Y.; Yan, H.; Zhao, C.; Li, X.; Liu, W.; He, M.; Tang, S.; Xi, S. Impact of temperature on the dynamics of the COVID-19 outbreak in China. Sci. Total Environ. 2020, 728, 138890. [Google Scholar] [CrossRef]
- Zhu, L.; Liu, X.; Huang, H.; Avellán-Llaguno, R.D.; Lazo, M.M.L.; Gaggero, A.; Rifo, R.S.; Patiño, L.; Valencia-Avellan, M.; Diringer, B.; et al. Meteorological impact on the COVID-19 pandemic: A study across eight severely affected regions in South America. Sci. Total Environ. 2020, 744, 140881. [Google Scholar] [CrossRef]
- Prata, D.N.; Rodrigues, W.; Bermejo, P.H. Temperature significantly changes COVID-19 transmission in (sub)tropical cities of Brazil. Sci. Total Environ. 2020, 729, 138862. [Google Scholar] [CrossRef]
- Ward, M.P.; Xiao, S.; Zhang, Z. The role of climate during the COVID-19 epidemic in New South Wales, Australia. Transbound Emerg. Dis. 2020, 67, 2313–2317. [Google Scholar] [CrossRef]
- Adekunle, I.A.; Tella, S.A.; Oyesiku, K.O.; Oseni, I.O. Spatio-temporal analysis of meteorological factors in abating the spread of COVID-19 in Africa. Heliyon 2020, 6, e04749. [Google Scholar] [CrossRef] [PubMed]
- Alkhowailed, M.; Shariq, A.; Alqossayir, F.; Alzahrani, O.A.; Rasheed, Z.; Al Abdulmonem, W. Impact of meteorological parameters on COVID-19 pandemic: A comprehensive study from Saudi Arabia. Inform. Med. Unlocked 2020, 20, 100418. [Google Scholar] [CrossRef] [PubMed]
- Goswami, K.; Bharali, S.; Hazarika, J. Projections for COVID-19 pandemic in India and effect of temperature and humidity. Diabetes Metab. Syndr. 2020, 14, 801–805. [Google Scholar] [CrossRef] [PubMed]
- Meraj, G.; Farooq, M.; Singh, S.K.; Romshoo, S.A.; Sudhanshu; Nathawat, M.S.; Kanga, S. Coronavirus pandemic versus temperature in the context of Indian subcontinent: A preliminary statistical analysis. Environ. Dev. Sustain. 2020, 1–11. [Google Scholar] [CrossRef]
- Paez, A.; Lopez, F.A.; Menezes, T.; Cavalcanti, R.; Pitta, M. A Spatio-Temporal Analysis of the Environmental Correlates of COVID-19 Incidence in Spain. Geogr. Anal. 2020. [Google Scholar] [CrossRef]
- Briz-Redón, Á.; Serrano-Aroca, Á. A spatio-temporal analysis for exploring the effect of temperature on COVID-19 early evolution in Spain. Sci. Total Environ. 2020, 728, 138811. [Google Scholar] [CrossRef]
- Tosepu, R.; Gunawan, J.; Effendy, D.S.; Ahmad, O.A.I.; Lestari, H.; Bahar, H.; Asfian, P. Correlation between weather and Covid-19 pandemic in Jakarta, Indonesia. Sci. Total Environ. 2020, 725, 138436. [Google Scholar] [CrossRef]
- Pani, S.K.; Lin, N.H.; RavindraBabu, S. Association of COVID-19 pandemic with meteorological parameters over Singapore. Sci. Total Environ. 2020, 740, 140112. [Google Scholar] [CrossRef]
- Menebo, M.M. Temperature and precipitation associate with Covid-19 new daily cases: A correlation study between weather and Covid-19 pandemic in Oslo, Norway. Sci. Total Environ. 2020, 737, 139659. [Google Scholar] [CrossRef]
- Paynter, S. Humidity and respiratory virus transmission in tropical and temperate settings. Epidemiol. Infect. 2015, 143, 1110–1118. [Google Scholar] [CrossRef]
- Casanova, L.M.; Jeon, S.; Rutala, W.A.; Weber, D.J.; Sobsey, M.D. Effects of air temperature and relative humidity on coronavirus survival on surfaces. Appl. Environ. Microbiol. 2010, 76, 2712–2717. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization—Indonesia. Coronavirus Disease 2019 (COVID-19) Situation Report-19; 2020. Available online: https://www.who.int/docs/default-source/searo/indonesia/covid19/who-situation-report-19.pdf?sfvrsn=531a8fe6_2 (accessed on 14 December 2020).
- Aboubakr, H.A.; Sharafeldin, T.A.; Goyal, S.M. Stability of SARS-CoV-2 and other coronaviruses in the environment and on common touch surfaces and the influence of climatic conditions: A review. Transbound. Emerg. Dis. 2020. [Google Scholar] [CrossRef] [PubMed]
- Chan, K.H.; Peiris, J.S.; Lam, S.Y.; Poon, L.L.; Yuen, K.Y.; Seto, W.H. The Effects of Temperature and Relative Humidity on the Viability of the SARS Coronavirus. Adv. Virol. 2011, 2011, 734690. [Google Scholar] [CrossRef]
- Riddell, S.; Goldie, S.; Hill, A.; Eagles, D.; Drew, T.W. The effect of temperature on persistence of SARS-CoV-2 on common surfaces. Virol. J. 2020, 17, 145. [Google Scholar] [CrossRef]
- Lowen, A.C.; Mubareka, S.; Steel, J.; Palese, P. Influenza virus transmission is dependent on relative humidity and temperature. PLoS Pathog. 2007, 3, 1470–1476. [Google Scholar] [CrossRef] [PubMed]
- García, L.F. Immune Response, Inflammation, and the Clinical Spectrum of COVID-19. Front. Immunol. 2020, 11, 1441. [Google Scholar] [CrossRef]
- Dzien, A.; Dzien-Bischinger, C.; Lechleitner, M.; Winner, H.; Weiss, G. Will the COVID-19 pandemic slow down in the Northern hemisphere by the onset of summer? An epidemiological hypothesis. Infection 2020, 48, 627–629. [Google Scholar] [CrossRef]
- Lofgren, E.; Fefferman, N.H.; Naumov, Y.N.; Gorski, J.; Naumova, E.N. Influenza Seasonality: Underlying Causes and Modeling Theories. J. Virol. 2007, 81, 5429. [Google Scholar] [CrossRef]
- Méndez-Arriaga, F. The temperature and regional climate effects on communitarian COVID-19 contagion in Mexico throughout phase 1. Sci. Total Environ. 2020, 735, 139560. [Google Scholar] [CrossRef]
- Dai, H.; Zhao, B. Association of the infection probability of COVID-19 with ventilation rates in confined spaces. Build Simul. 2020, 13, 1321–1327. [Google Scholar] [CrossRef]
- Soleimani, Z.; Teymouri, P.; Darvishi Boloorani, A.; Mesdaghinia, A.; Middleton, N.; Griffin, D.W. An overview of bioaerosol load and health impacts associated with dust storms: A focus on the Middle East. Atmos. Environ. 2020, 223, 117187. [Google Scholar] [CrossRef]
- Domingo, J.L.; Rovira, J. Effects of air pollutants on the transmission and severity of respiratory viral infections. Environ. Res. 2020, 187, 109650. [Google Scholar] [CrossRef] [PubMed]
- Setti, L.; Passarini, F.; De Gennaro, G.; Barbieri, P.; Perrone, M.G.; Borelli, M.; Palmisani, J.; Di Gilio, A.; Torboli, V.; Fontana, F.; et al. SARS-Cov-2RNA found on particulate matter of Bergamo in Northern Italy: First evidence. Environ. Res. 2020, 188, 109754. [Google Scholar] [CrossRef] [PubMed]
- Pan, M.; Lednicky, J.A.; Wu, C.Y. Collection, particle sizing and detection of airborne viruses. J. Appl. Microbiol. 2019, 127, 1596–1611. [Google Scholar] [CrossRef] [PubMed]
- Fisman, D.N. Seasonality of Infectious Diseases. Annu. Rev. Public Health 2007, 28, 127–143. [Google Scholar] [CrossRef] [PubMed]
- Martinez, M.E. The calendar of epidemics: Seasonal cycles of infectious diseases. PLoS Pathog. 2018, 14, e1007327. [Google Scholar] [CrossRef]
| Record | Journal | Site | Study Period | Source of Case Data | Weather Variables | Study Design | Main Findings | Strengths | Limitations and Confounders |
|---|---|---|---|---|---|---|---|---|---|
| Adekunle et al. 2020 [43] | Heliyon | Africa (urban–rural) | 30 March 2020–29 April 2020 (31 days) | Daily confirmed cases from WHO daily situation reports | Daily Temperature (mean), wind speed, relative humidity | Ecological–time series analysis (GAM) | Mean temperature negatively correlated with COVID-19 cases, with 1 °C increase, cases decreased by 13.53% (95% CI: 1.53–4.63) in the range of −2.42 °C–33.37 °C. Average wind speed positively correlated, 1% increase in average wind speed (m/s) associated with 11.21% (95%CI: 0.51–1.19) increase in confirmed cases. Relative humidity (%) not significant. | P values and 95% CI reported. GAM for lag/incubation period effects and non-linear relationship | Confounders not identified. Regional variations in testing and reporting. |
| Alkhowailed et al. 2020 [44] | Inform Med Unlocked | Saudi Arabia (urban) | 5 March 2020–7 June 2020 (95 days) | Daily new cases from Saudi Arabia Ministry of Health dashboard | Temperature, Dew point, Relative Humidity, Wind speed, atmospheric pressure | Ecological study–Spearman’s correlation | Temperature and humidity (weak) negative correlation with new daily cases. Wind speed also negatively correlated with new daily cases. | P values and 95% CI reported Longer time-period | Incubation period/lag not accounted for. Testing limitations. Population density may confound results |
| Bashir et al. 2020 [33] | Sci Total Environ | New York City, USA (urban/metropolitan) | 1 March 2020–12 April 2020 (43 days) | Total confirmed cases and daily new cases from the New York City health department COVID-19 data archive | Temperature, Relative Humidity, Wind speed, Precipitation/rainfall | Ecological study–Spearman’s correlation | Average temperature, minimum temperature and air quality significant correlated with new cases and mortality. Temperature range −3.37 °C–25 °C | A large number of climate variables analyzed in the model | Air quality (confounder) Potentially limited testing and reporting. Short time-period (~one month of data) No accounting for onset/incubation period lag |
| Briz-Redón & Serrano-Aroca, 2020 [48] | Sci Total Environ | Spain (urban) | 25 February 2020–28 March 2020 (33 days) | Daily cumulative cases from online resource | Temperature | Ecological study–spatio-temporal | No consistent evidence for association between temperature and cumulative cases in the temperature range −3.19–29.26 | Onset/incubation period lag included in model. Population density, age distribution, travelers and companies by province. | Short study period–2 weeks during lockdown period. Early in pandemic–limited testing and reporting ability Only assessed temperature |
| Bukhari et al. 2020 [29] | Int J Environ Res Public Health | Global (urban) | 20 January 2020–1 May 2020 (103 days) | Confirmed cases from John Hopkins University Coronavirus Resource Centre repository | Temperature, relative humidity, absolute humidity, wind speed. | Ecological study—correlation | For the time-period, the majority of new cases were reported in regions with a mean temperature between 0 °C–17 °C and absolute humidity between 1–9 g/m³. In regions >17 °C and AH >9 g/m³ reported cases much lower for the same time-period. | Large global data set–data at country or state level | Testing and under-reporting of cases, population density, community structures, socioeconomic factors Lag/incubation period not accounted for |
| Chien & Chen, 2020 [35] | Stoch Environ Res Risk Assess | USA (urban–rural) | 16 March–22 April 2020 (38 days) | Confirmed cases from Johns Hopkins Coronavirus Resource Centre | Temperature, Relative Humidity, Precipitation/Rainfall | Ecological study—GAM | Temperature and precipitation significant negative correlation with COVID-19 cases. RR% −0.21 (95%CI: −0.26, −0.15), in further modelling a threshold of 15.3 °C was identified, where RR% switched from negative to positive above this threshold and peaking at 20.25 °C and decreasing towards 29.2 °C. Relative humidity significant positive correlation with COVID-19 cases | GAM–modelling for spatial and temporal factors including lag/incubation period. Included confounders in model | Confounding variables–county-specific population, age, gender, racial composition and poverty level in the model. Limited testing and reporting. Short study period |
| Goswami et al. 2020 [45] | Diabetes Metab Syndr | India (urban–rural) | 1 April 2020–10 May 2020 (40 days) | Confirmed cases from official reports of the Ministry of Health and Family Welfare of India | Temperature, relative humidity | Ecological study—GAM | Statistically significant relationship between cases as a result of interaction between AT and ARH on COVID-19 incidence. Not consistent across regions of India. | Used 3 day moving average to account for lag. | Large geographical area and varying climate range, early strict lockdowns Limited testing |
| Huang et al. 2020 [30] | Sci Total Environ | Global (urban) | 21 January 2020–6 May 2020 (107 days) | Johns Hopkins University | Temperature, Relative humidity, Absolute humidity | Ecological–time series analysis | Significant association between temperature and AH—60% of cases occurred in the temperature range 5–15 °C, cases peaked at 11.54 °C. 73.8% of confirmed cases in regions with AH of 3–10 g/m³. | Large data set with cases from 185 countries/regions. Data from a longer time-period (~3 months) | Correlation between temperature and cases as a distribution rather than assessing association. Doesn’t account for lag or imported cases. Control measures and public health measures |
| Liu et al. 2020 [36] | Sci Total Environ | China (urban/metropolitan) | 20 January 2020–2 March 2020 (43 days) | Daily confirmed cases from Health Commissions per city | Temperature, humidity, diurnal temperature range | Ecological study—GLM | AT, AH and DTR negatively associated with transmission in pooled results. AT increase of 1 °C correlated to decrease in daily case counts RR = 0.80 (95% CI: 0.75–0.85) in the range −20 °C–20 °C. 1% increase in DTR associated with decrease in lag cases RR = 0.90 (95% CI: 0.86–0.95). increase in AH of 1 g/m³ associated with decrease in cases RR = 0.72 (95% CI: 0.59–0.89) and 0.33 (95% CI: 0.21–0.54) based on lag period. | Accounted for lag using 0, 3, 7 and 14-day intervals. | Migration Scale Index (MSI) |
| Menebo, 2020 [51] | Sci Total Environ | Oslo, Norway (urban/metropolitan) | 27 February 2020–2 May 2020 (66 days) | Daily cases from the Norwegian public health institute | Temperature, Precipitation/rainfall, Wind speed | Ecological study–Spearman’s rank correlation | Tmax (r = 0.347; p = 005) and Tavg (r = 0.293 p = 0.019) significant positive correlation with COVID-19 cases in the temperature range −0.5 °C–21.9 °C. Precipitation significant negative correlation with COVID-19 cases. | Lag analysis of 5,6,14 days for weather variables and onset | Humidity not included in model Public health measures–lockdowns, sanitization, testing capabilities not included in analysis |
| Meraj et al. 2020 [46] | Environ Dev Sustain. | India (urban–rural) | 9 March 2020–27 May 2020 (80 days) | Cumulative cases from official region web sites for Maharashtra, Kashmir and Rajasthan | Temperature | Ecological study–Pearson’s correlation | Significant positive correlation with COVID-19 cases in Rajasthan (25 °C–45 °C) and Kashmir (10 °C–32 °C). No significant association between temperature and COVID-19 in Maharashtra (29 °C–38 °C). | Comparison between provinces with varying climates and ecologies | Only temperature included for three provinces. Limited testing rates initially (increased over time). |
| Meyer et al. 2020 [32] | Front public Health | Global (100 countries)(Urban) | 29 December 2020–17 March 2020 (80 days) | Daily cases from WHO daily reports | Temperature, Humidity | Population–cohort study GLMM | Statistically significant association with temperature and COVID-19 incidence in temperature range −33.9–34.3 °C). Small effect size. | Lag delay between 3 and 20 days in model. Cases identified as local or imported. | Limitations in COVID-19 testing and reporting early in pandemic. |
| Paez et al. 2020 [47] | Geogr Anal. | Spain (urban) | 13 March 2020–11 April 2020 (30 days) | Daily new cases at provincial level from Centro de Datos COVID-19 | Temperature, Humidity, Daily Sunshine | Ecological study–Spatio-temporal | Temperature significant negative correlation with COVID-19 incidence in the range 1 °C–23.2 °C, humidity significant negative correlation when accounting for control variables. | Confounders included in SUR model as controls Spatial and temporal modelling. Lag/incubation period included | Confounding variables–GDP, age, population density, province area. Human behavioral changes as a result of lockdown, hours of sunshine and population density |
| Pani et al. 2020 [50] | Sci Total Environ | Singapore (urban/metropolitan) | 23 January 2020–31 May 2020 (59 days) | Daily cases of new infections and deaths from the Ministry of Health (MOH) | Temperature, Relative humidity, Absolute humidity, Surface pressure, Dew point, Wind speed, Water vapor | Ecological study–Spearman’s rank correlation | Temperature significant positive association with daily and cumulative cases in the range 24 °C–32 °C. TRH significant for transmission, along with AH and WV. WS not significant. | Large range of weather variables included in model | Meteorological data limited to one site. Public health measures and personal hygiene. Testing limitations. |
| Prata et al. 2020 [41] | Sci Total Environ | Brazil (urban) | 27 February 2020–1 April 2020 (35 days) | Daily cumulative cases reported by the Ministry of Health of Brazil | Temperature | Ecological study—GAM | For each 1 °C rise in temperature, daily COVID-19 cases decreased by 4.9% when temperature below 25.8 °C, in the range of 16.8 °C–27.4 °C. | Climate and temperature zones, GAM and GLM analysis | Testing limitations, only temperature included |
| Qi et al. 2020 [38] | Sci Total Environ | China (urban) | 1 December 2019–11 February 2020 (73 days) | Daily confirmed cases from National Health Commission | Temperature, Relative Humidity | Ecological study–time series analysis GAM | Temperature and Humidity have a significant negative correlation with COVID-19 incidence in the range 1.5 °C–11.42 °C. | Data from early outbreak in China prior to travel restrictions. GAM with internet search results for health-seeking behaviors. 14-day onset lag/incubation period | Short study period early in outbreak Confounders not included in model–socioeconomic status, intervention measures, weather is at city, not province level |
| Shi et al. 2020 [39] | Sci Total Environ | China (urban) | 20 January 2020–29 February 2020 (41 days) | Confirmed cases from China National health Commission (CNHC) | Temperature | Ecological–spatio-temporalSEIR model | Temperature had a significant effect on COVID-19 incidence in the range −22 °C–26 °C. Increasing temperature associated with decreased infection rate. RR = 0.96 (95%CI: 0.93–0.99) | Dynamic transmission model with lag/incubation period delay | Only temperature included. Limited study period early in outbreak. Limited testing and reporting, change in diagnostic criteria–healthy patients excluded. |
| To et al. 2020 [34] | Sci Total Environ | Canada (urban–rural) | 25 January 2020–18 May 2020 (115 days) | Case data for Alberta, British Columbia, Ontario and Quebec | Temperature, Precipitation/rainfall, Wind speed | Ecological study–linear regression model | No significant association for Temperature and COVID-19 incidence when adjusted for wind speed, precipitation and province in the temperature range −6.83 °C–7.94 °C | A large geographical area with variations in temperature across the country, 2-week lag delay for climate variables | Model adjusted for wind speed, precipitation and province to evaluate temperature Linear regression model. Local public health policies, testing rates and urbanization varies across regions |
| Tosepu et al. 2020 [49] | Sci Total Environ | Indonesia (urban/metropolitan) | January 2020–29 March 2020 (~29 days) | Daily COVID-19 cases from the Ministry of Health of Republic of Indonesia | Temperature (min, max, avg), Humidity, Precipitation/Rainfall | Ecological study–Spearman’s rank correlation | Average temperature correlated with COVID-19 cases (26.1 °C–28.6 °C). Tmin, Tmax, humidity and rainfall not significantly correlated. | Large range of weather variables | High mobility and population density in Jakarta Small data set/limited testing, no accounting for lag/incubation period |
| Ward et al. 2020 [42] | Transbound Emerg Dis | Australia (urban) | 12 February 2020–30 March 2020 (35 days) | NSW Government Case Reports | Temperature, Relative humidity, Precipitation/Rainfall | Ecological study–time series analysis GAM | Negative significant relationship between relative humidity and COVID-19 cases (p = 0.0304) where a 1% decrease in morning humidity associated with up to 6.11% increase in cases. Temperature (18.4 °C–25.5 °C) and rainfall were not significant in time series analysis. | Good quality data set available, high testing rate and reporting. Accounted for 14-day lag/incubation period | Limited case numbers available, mostly imported cases rather than local transmission |
| Wu et al. 2020 [31] | Sci Total Environ | Global (166 countries excluding China) (urban) | Up to 27 March 2020 (~75 days) | Daily cases and deaths from WHO COVID-19 daily situation reports | Temperature, Dew point, Wind speed, Relative humidity | Ecological study–time series analysis GAM | Temperature and RH negatively correlated to daily cases. 1 °C increase in temperature associated with 3.08% (95% CI: 1.53–4.63%) reduction in cases in the range of −5.28 °C–34.3 °C. 1% increase in RH associated with 0.85% (95% CI: 0.51–1.19%) reduction in new daily cases. | Global data set with daily new cases and deaths. Included confounders in modelling–economic level, additional health conditions, age and density. Daily lag/incubation period included in model | Confounders—Wind speed, median age, Global Health Security Index, Human Development Index, population density |
| Xie & Zhu, 2020 [37] | Sci Total Environ | China (urban) | 23 January 2020–29 February 2020 (38 days) | Daily cases from official websites of each city heath commission. | Temperature, Relative Humidity, Air Pressure, Wind speed | Ecological study– exposure response GAM | Exposure response positive linear when temperature below 3 °C and flat above 3 °C. Each 1 °C rise in temperature was associated with 4.861% (95%CI:3.209–6.513) increase in COVID-19 cases average daily temperature range −33.8 °C–26.9 °C | Large number of variables included in model. Lag/incubation period from 0–7, 0–14, 0–21 days | Case data from early in outbreak, limited testing and diagnostic capabilities. Short study period |
| Zhu et al. 2020 [40] | Sci Total Environ | South America (urban/metropolitan) | 23 February 2020–6 May 2020 (74 days) | Data from National health departments or secondary websites | Temperature. Wind speed, Relative Humidity | Ecological study–Spearman’s rank correlation | Large variation in correlation by region. Daily average temperature and absolute humidity were most strongly correlated with COVID-19 cases averaged across regions. (Temperature range 3.9 °C–35 °C) Wind speed not significantly correlated with daily cases. | Accounted for lag using daily incubated cases (median ~4 days prior to positive test) | Demographic, geographical and socioeconomic factors, healthcare infrastructure, governmental and social policies, testing and reporting. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
McClymont, H.; Hu, W. Weather Variability and COVID-19 Transmission: A Review of Recent Research. Int. J. Environ. Res. Public Health 2021, 18, 396. https://doi.org/10.3390/ijerph18020396
McClymont H, Hu W. Weather Variability and COVID-19 Transmission: A Review of Recent Research. International Journal of Environmental Research and Public Health. 2021; 18(2):396. https://doi.org/10.3390/ijerph18020396
Chicago/Turabian StyleMcClymont, Hannah, and Wenbiao Hu. 2021. "Weather Variability and COVID-19 Transmission: A Review of Recent Research" International Journal of Environmental Research and Public Health 18, no. 2: 396. https://doi.org/10.3390/ijerph18020396
APA StyleMcClymont, H., & Hu, W. (2021). Weather Variability and COVID-19 Transmission: A Review of Recent Research. International Journal of Environmental Research and Public Health, 18(2), 396. https://doi.org/10.3390/ijerph18020396






