Air Pollution during Pregnancy and Childhood Autism Spectrum Disorder in Taiwan
Abstract
:1. Introduction
2. Materials and Methods
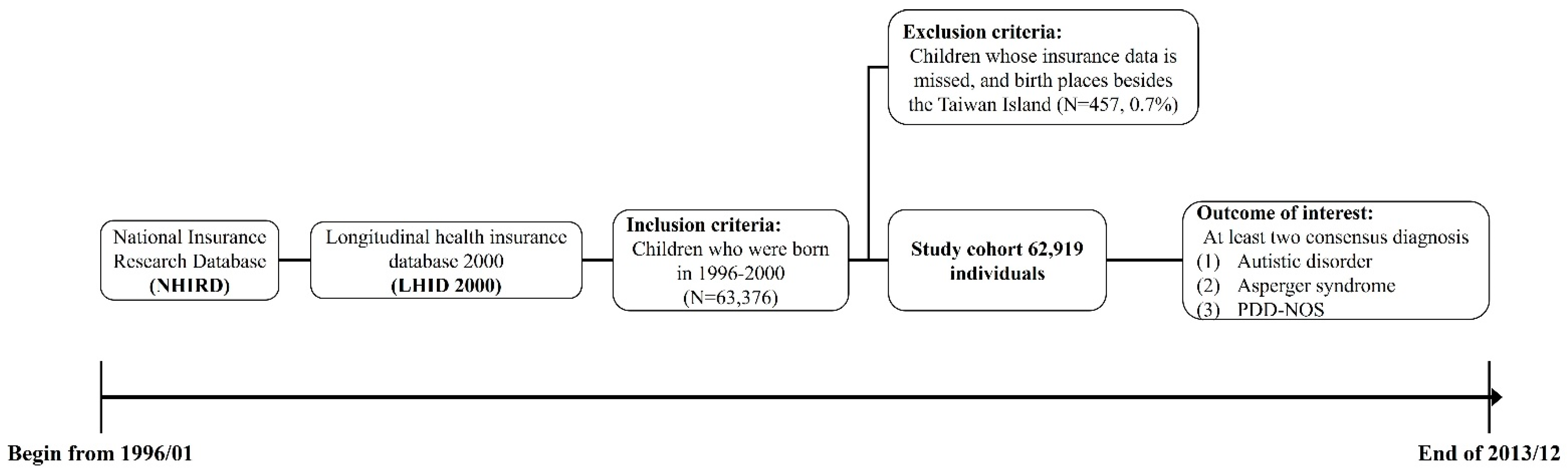
2.1. Study Cohort
2.2. Identification of Cases
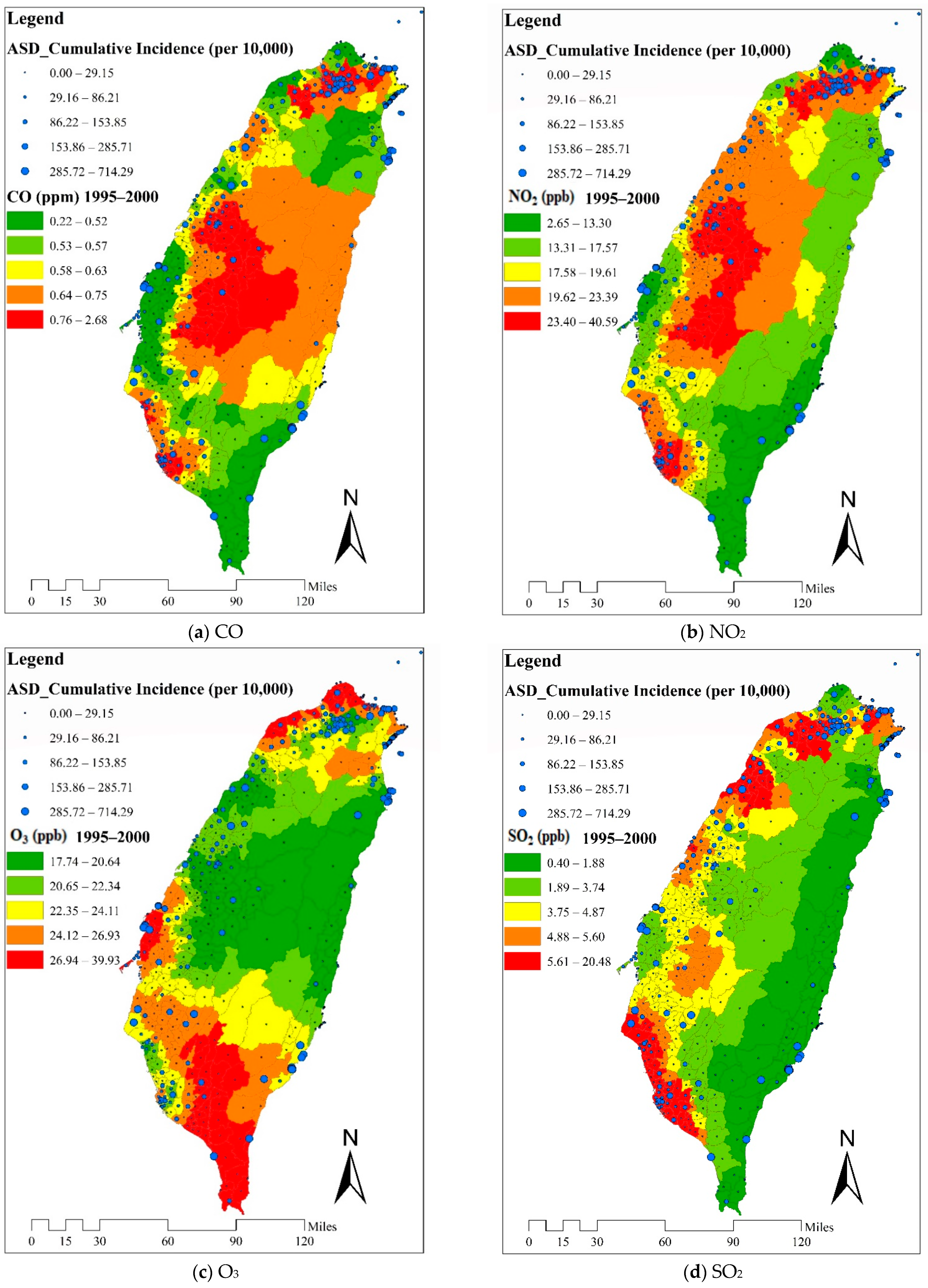
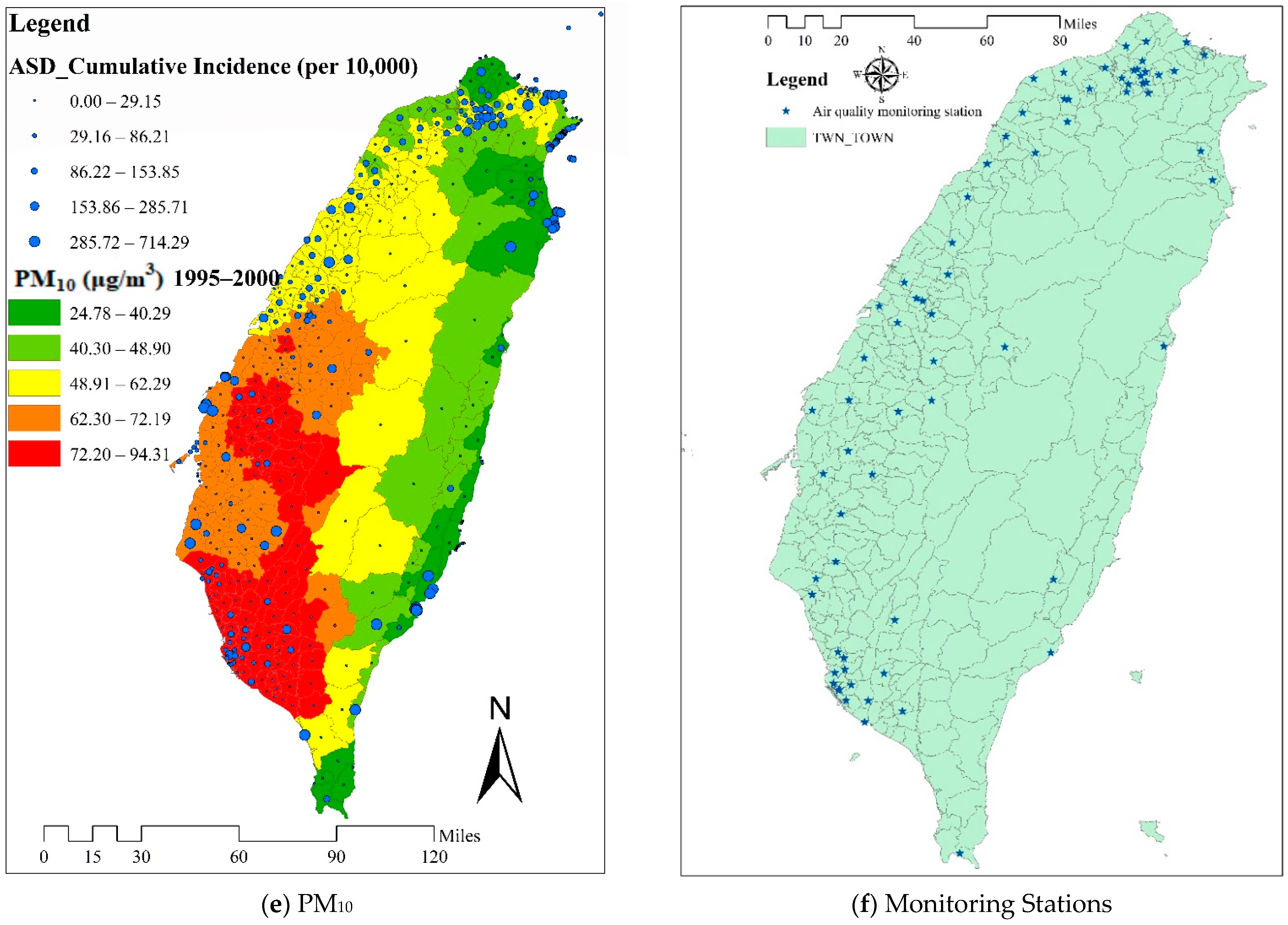
2.3. Exposure Assessment
2.4. Covariates
2.5. Statistical Analysis
3. Results
3.1. Study Sample and Distribution of Patients
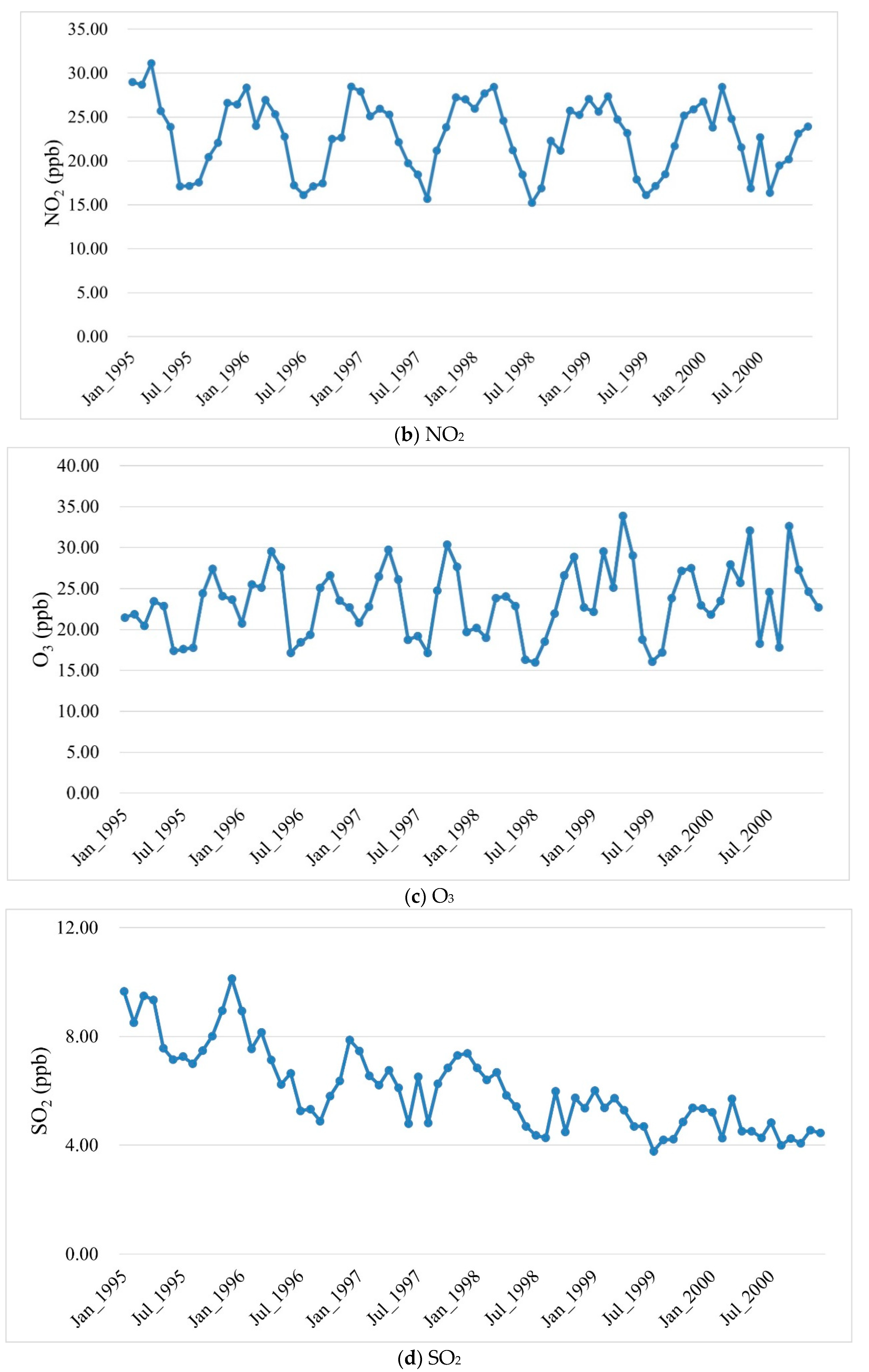
3.2. Levels of Air Pollutants
3.3. Results from Univariate Analyses of Covariates
3.4. Associations between Air Pollutants and Autism Spectrum Disorder
4. Discussion
4.1. Environmental Factors Contribute to Autism Spectrum Disorder
4.2. Associations of Air Pollution and Autism Spectrum Disorder
4.3. Trimester-Specific Associations
4.4. Strengths and Limitations of the Study
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Newschaffer, C.J.; Croen, L.A.; Daniels, J.; Giarelli, E.; Grether, J.K.; Levy, S.E.; Mandell, D.S.; Miller, L.A.; Pinto-Martin, J.; Reaven, J.; et al. The epidemiology of autism spectrum disorders. Annu. Rev. Public Health 2007, 28, 235–258. [Google Scholar] [CrossRef] [Green Version]
- Lotter, V. Epidemiology of autistic conditions in young children. Soc. Psychiatry 1966, 1, 124–135. [Google Scholar] [CrossRef]
- Folstein, S.; Rutter, M. Infantile autism a genetic study of 21 twin pairs. J. Child Psychol. Psychiatry 1977, 18, 297–321. [Google Scholar] [CrossRef]
- Matson, J.L.; Nebel-Schwalm, M.S. Comorbid psychopathology with autism spectrum disorder in children: An overview. Res. Dev. Disabil. 2007, 28, 341–352. [Google Scholar] [CrossRef] [Green Version]
- Fombonne, E. Epidemiological surveys of autism and other pervasive developmental disorders: An update. J. Autism Dev. Disord. 2003, 33, 365–382. [Google Scholar] [CrossRef] [PubMed]
- Fombonne, E. The changing epidemiology of autism. J. Appl. Res. Intellect. Disabil. 2005, 18, 281–294. [Google Scholar] [CrossRef]
- Rice, C. Prevalence of Autism Spectrum Disorders–Autism and Developmental Disabilities Monitoring Network, 14 Sites, United State, 2002; Morbidity and Mortality Weekly Report; Surveillance Summaries; MMWR: Washington, DC, USA, 2009; Volume 56, pp. 12–28. [Google Scholar]
- Haga, H.; Miyamoyo, Y. A survey on the actual state of so-called autistic children in Kyoto prefecture. Jpn. J. Child Psychiatry 1971, 12, 160–167. [Google Scholar]
- Nakai, M. Epidemiology of autistic children in Gifu-ken. Jpn. J. Child Psychiatry 1971, 12, 262–266. [Google Scholar]
- Tanino, Y. An investigation into infantile autism and autistic children in Toyama prefecture. Jpn. J. Child Psychiatry 1971, 12, 150–159. [Google Scholar]
- Yamazaki, K.; Yamashita, I.; Suwa, N.; Kuroda, T.; lwabuchi, J.; Imanmra, S.; Miyamoto, M.; Fujino, T.; lto, N.; Sugaya, K. Survey on the morbidity rate of “autistic children” in the Hokkaido district. Jpn. J. Child Psychiatry 1971, 12, 141–149. [Google Scholar]
- Sun, X.; Allison, C. A review of the prevalence of autism spectrum disorder in Asia. Res. Autism Spectr. Disord. 2010, 4, 156–167. [Google Scholar] [CrossRef]
- Sun, X.; Allison, C.; Matthews, F.E.; Sharp, S.J.; Auyeung, B.; Baron-Cohen, S.; Brayne, C. Prevalence of autism in mainland China, Hong Kong and Taiwan: A systematic review and meta-analysis. Mol. Autism 2013, 4, 7. [Google Scholar] [CrossRef] [Green Version]
- Chien, I.C.; Lin, C.H.; Chou, Y.J.; Chou, P. Prevalence and incidence of autism spectrum disorders among national health insurance enrollees in Taiwan from 1996 to 2005. J. Child Neurol. 2011, 26, 830–834. [Google Scholar] [CrossRef]
- Lai, D.C.; Tseng, Y.C.; Hou, Y.M.; Guo, H.R. Gender and geographic differences in the prevalence of autism spectrum disorders in children: Analysis of data from the national disability registry of Taiwan. Res. Dev. Disabil. 2012, 33, 909–915. [Google Scholar] [CrossRef]
- Croen, L.A.; Grether, J.K.; Hoogstrate, J.; Selvin, S. The changing prevalence of autism in California. J. Autism Dev. Disord. 2002, 32, 207–215. [Google Scholar] [CrossRef] [PubMed]
- Powell, J.; Edwards, A.; Edwards, M.; Pandit, B.; Sungum-Paliwal, S.; Whitehouse, W. Changes in the incidence of childhood autism and other autistic spectrum disorders in preschool children from two areas of the West Midlands, UK. Dev. Med. Child Neurol. 2000, 42, 624–628. [Google Scholar] [CrossRef]
- Smeeth, L.; Cook, C.; Fombonne, P.E.; Heavey, L.; Rodrigues, L.C.; Smith, P.G.; Hall, A.J. Rate of first recorded diagnosis of autism and other pervasive developmental disorders in United Kingdom general practice, 1988 to 2001. BMC Med. 2004, 2, 39. [Google Scholar] [CrossRef] [Green Version]
- Kaye, J.A.; del Mar Melero-Montes, M.; Jick, H. Mumps, measles, and rubella vaccine and the incidence of autism recorded by general practitioners: A time trend analysis. BMJ 2001, 302, 460. [Google Scholar] [CrossRef] [Green Version]
- Fombonne, E. Epidemiology of pervasive developmental disorders. Pediatr. Res. 2009, 65, 591–598. [Google Scholar] [CrossRef] [PubMed]
- Folstein, S.; Rutter, M. Genetic influences and infantile autism. Nature 1977, 265, 726–728. [Google Scholar] [CrossRef] [PubMed]
- Steffenburg, S.; Gillberg, C.; Hellgren, L.; Andersson, L.; Gillberg, I.C.; Jakobsson, G.; Bohman, M. A twin study of autism in Denmark, Finland, Iceland, Norway and Sweden. J. Child Psychol. Psychiatry 1989, 30, 405–416. [Google Scholar] [CrossRef] [PubMed]
- Hallmayer, J.; Cleveland, S.; Torres, A.; Phillips, J.; Cohen, B.; Torigoe, T.; Miller, J.; Fedele, A.; Collins, J.; Smith, K.; et al. Genetic heritability and shared environmental factors among twin pairs with autism. Arch. Gen. Psychiatry 2011, 68, 1095–1102. [Google Scholar] [CrossRef]
- Sandin, S.; Lichtenstein, P.; Kuja-Halkola, R.; Larsson, H.; Hultman, C.M.; Reichenberg, A. The familial risk of autism. JAMA 2014, 311, 1770–1777. [Google Scholar] [CrossRef]
- Vassilev, Z.P.; Robson, M.G.; Klotz, J.B. Outdoor exposure to airborne polycyclic organic matter and adverse reproductive outcomes: A pilot study. Am. J. Ind. Med. 2001, 40, 255–262. [Google Scholar] [CrossRef] [PubMed]
- Ritz, B.; Yu, F. The effect of ambient carbon monoxide on low birth weight among children born in southern California between 1989 and 1993. Environ. Health Perspect. 1999, 107, 17–25. [Google Scholar] [CrossRef]
- Currie, J.; Neidell, M.; Schmieder, J.F. Air pollution and infant health: Lessons from New Jersey. J. Health Econ. 2009, 28, 688–703. [Google Scholar] [CrossRef] [Green Version]
- Hansen, C.A.; Barnett, A.G.; Pritchard, G. The effect of ambient air pollution during early pregnancy on fetal ultrasonic measurements during mid-pregnancy. Environ. Health Perspect. 2008, 116, 362–369. [Google Scholar] [CrossRef]
- Suglia, S.F.; Gryparis, A.; Wright, R.O.; Schwartz, J.; Wright, R.J. Association of black carbon with cognition among children in a prospective birth cohort study. Am. J. Epidemiol. 2008, 167, 280–286. [Google Scholar] [CrossRef]
- Morales, E.; Julvez, J.; Torrent, M.; de Cid, R.; Guxens, M.; Bustamante, M.; Kunzli, N.; Sunyer, J. Association of early-life exposure to household gas appliances and indoor nitrogen dioxide with cognition and attention behavior in preschoolers. Am. J. Epidemiol. 2009, 169, 1327–1336. [Google Scholar] [CrossRef] [Green Version]
- Volk, H.E.; Hertz-Picciotto, I.; Delwiche, L.; Lurmann, F.; McConnell, R. Residential proximity to freeways and autism in the CHARGE study. Environ. Health Perspect. 2011, 119, 873–877. [Google Scholar] [CrossRef]
- Becerra, T.A.; Wilhelm, M.; Olsen, J.; Cockburn, M.; Ritz, B. Ambient air pollution and autism in Los Angeles County, California. Environ. Health Perspect. 2013, 121, 380–386. [Google Scholar] [CrossRef] [PubMed]
- Volk, H.E.; Lurmann, F.; Penfold, B.; Hertz-Picciotto, I.; McConnell, R. Traffic-related air pollution, particulate matter, and autism. JAMA Psychiatry 2013, 70, 71–77. [Google Scholar] [CrossRef]
- Kalkbrenner, A.E.; Windham, G.C.; Serre, M.L.; Akita, Y.; Wang, X.; Hoffman, K.; Thayer, B.P.; Daniels, J.L. Particulate matter exposure, prenatal and postnatal windows of susceptibility, and autism spectrum disorders. Epidemiology 2015, 26, 30–42. [Google Scholar] [CrossRef]
- Raz, R.; Roberts, A.L.; Lyall, K.; Hart, J.E.; Just, A.C.; Laden, F.; Weisskopf, M.G. Autism spectrum disorder and particulate matter air pollution before, during, and after pregnancy: A nested case-control analysis within the Nurses’ Health Study II Cohort. Environ. Health Perspect. 2015, 123, 264–270. [Google Scholar] [CrossRef]
- Jung, C.R.; Lin, Y.T.; Hwang, B.F. Air pollution and newly diagnostic autism spectrum disorders: A population-based cohort study in Taiwan. PLoS ONE 2013, 8, e0202996. [Google Scholar] [CrossRef] [Green Version]
- National Health Insurance Research Database, Taiwan. Available online: http://nhird.nhri.org.tw/en/index.htm (accessed on 11 February 2020).
- Liao, D.; Peuquet, D.J.; Duan, Y.; Whitsel, E.A.; Dou, J.; Smith, R.L.; Lin, H.-M.; Chen, J.-C.; Heiss, G. GIS Approaches for the estimation of residential-Level ambient PM concentrations. Environ. Health Perspect. 2006, 114, 1374–1380. [Google Scholar] [CrossRef] [PubMed]
- Environmental Systems Research Institute. How Kriging Works—Help ArcGIS for Desktop. Available online: http://desktop.arcgis.com/en/arcmap/10.3/tools/3d-analyst-toolbox/how-kriging-works.htm#GUID-94A34A70-DBCF-4B23-A198-BB50FB955DC0 (accessed on 11 February 2020).
- Pan, W.C.; Wu, C.D.; Chen, M.J.; Huang, Y.T.; Chen, C.J.; Su, H.J.; Yang, H.I. Fine particle pollution, alanine transaminase, and liver cancer: A Taiwanese prospective cohort study (REVEAL-HBV). J. Natl. Cancer Inst. 2016, 108, djv341. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chun, H.; Leung, C.; Wen, S.W.; McDonald, J.; Shin, H.H. Maternal exposure to air pollution and risk of autism in children: A systematic review and meta-analysis. Environ. Pollut. 2020, 256, 113307. [Google Scholar] [CrossRef]
- Piven, J.; Palmer, P.; Jacobi, D.; Childress, D.; Arndt, S. Broader autism phenotype: Evidence from a family history study of multiple-incidence autism families. Am. J. Psychiatry 1997, 154, 185–190. [Google Scholar]
- Bailey, A.; Palferman, S.; Heavey, L.; Couteur, A.L. Autism: The phenotype in relatives. J. Autism Dev. Disord. 1998, 28, 369–392. [Google Scholar] [CrossRef]
- Geschwind, D.H.; Levitt, P. Autism spectrum disorders: Developmental disconnection syndromes. Curr. Opin. Neurobiol. 2007, 17, 103–111. [Google Scholar] [CrossRef]
- Lyall, K.; Schmidt, R.J.; Hertz-Picciotto, I. Maternal lifestyle and environmental risk factors for autism spectrum disorders. Int. J. Epidemiol. 2014, 43, 443–464. [Google Scholar] [CrossRef]
- Calderon-Garciduenas, L.; Solt, A.C.; Henriquez-Roldan, C.; Torres-Jardon, R.; Nuse, B.; Herritt, L.; Villarreal-Calderon, R.; Osnaya, N.; Stone, I.; Garcia, R.; et al. Long-term air pollution exposure is associated with neuroinflammation, an altered innate immune response, disruption of the blood-brain barrier, ultrafine particulate deposition, and accumulation of amyloid beta-42 and alpha-Synuclein in children and young Adults. Toxicol. Pathol. 2008, 36, 289–310. [Google Scholar] [PubMed]
- Calderon-Garciduenas, L.; Mora-Tiscareno, A.; Ontiveros, E.; Gomez-Garza, G.; Barragan-Mejia, G.; Broadway, J.; Chapman, S.; Valencia-Salazar, G.; Jewells, V.; Maronpot, R.R.; et al. Air pollution, cognitive deficits and brain abnormalities: A pilot study with children and dogs. Brain Cogn. 2008, 68, 117–127. [Google Scholar] [CrossRef] [PubMed]
- Edwards, T.M.; Myers, J.P. Environmental exposures and gene regulation in disease etiology. Environ. Health Perspect. 2007, 115, 1264–1270. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Perera, F.P.; Rauh, V.; Whyatt, R.M.; Tsai, W.Y.; Tang, D.L.; Diaz, D.; Hoepner, L.; Barr, D.; Tu, Y.H.; Camann, D.; et al. Effect of prenatal exposure to airborne polycyclic aromatic hydrocarbons on neurodevelopment in the first 3 years of life among inner-city children. Environ. Health Perspect. 2006, 114, 1287–1292. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Thomas, P.; Zahorodny, W.; Peng, B.; Kim, S.; Jani, N.; Halperin, W.; Brimacombe, M. The association of autism diagnosis with socioeconomic status. Autism 2012, 16, 201–213. [Google Scholar] [CrossRef]
- Rai, D.; Lewis, G.; Lundberg, M.; Araya, R.; Svensson, A.; Dalman, C.; Carpenter, P.; Magnusson, C. Parental socioeconomic status and risk of offspring autism spectrum disorders in a Swedish population-based study. J. Am. Acad. Child Adolesc. Psychiatry 2012, 51, 467–476. [Google Scholar] [CrossRef]
- Magnusson, C.; Rai, D.; Goodman, A.; Lundberg, M.; Idring, S.; Svensson, A.; Koupil, I.; Serlachius, E.; Dalman, C. Migration and autism spectrum disorder: Population-based study. Br. J. Psychiatry 2012, 201, 109–115. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tseng, Y.C.; Guo, H.R.; Lai, D.C. Maternal nationality and developmental delays in young children: Analysis of the data from the national registry in Taiwan. Res. Dev. Disabil. 2016, 53–54, 127–134. [Google Scholar] [CrossRef] [PubMed]
- Fang, S.Y.; Wang, S.; Huang, N.; Yeh, H.H.; Chen, C.Y. Prenatal infection and autism spectrum disorders in childhood: A population-based case-control study in Taiwan. Paediatr. Perinat. Epidemiol. 2015, 29, 307–316. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.R.; Iosif, A.M.; Hansen, R.L.; Schmidt, R.J. Maternal polyunsaturated fatty acids and risk for autism spectrum disorder in the MARBLES high-risk study. Autism 2020, 24, 1191–1200. [Google Scholar] [CrossRef] [PubMed]
- Mandy, W.; Lai, M.C. Annual Research Review: The role of the environment in the developmental psychopathology of autism spectrum condition. J. Child Psychol. Psychiatry 2016, 57, 271–292. [Google Scholar] [CrossRef]
- Pinborough-Zimmerman, J.; Bilder, D.; Bakian, A.; Satterfield, R.; Carbone, P.S.; Nangle, B.E.; Randall, H.; McMahon, W.M. Sociodemographic risk factors associated with autism spectrum disorders and intellectual disability. Autism Res. 2011, 4, 438–448. [Google Scholar] [CrossRef] [PubMed]
- Rubenstein, E.; Durkin, M.S.; Harrington, R.A.; Kirby, R.S.; Schieve, L.A.; Daniels, J. Relationship between advanced maternal age and timing of first developmental evaluation in children with autism. J. Dev. Behav. Pediatr. 2018, 39, 601–609. [Google Scholar] [CrossRef] [PubMed]
- Tan, M.; Yang, T.; Zhu, J.; Li, Q.; Lai, X.; Li, Y.Y.; Tang, T.; Chen, J.; Li, T.Y. Maternal folic acid and micronutrient supplementation is associated with vitamin levels and symptoms in children with autism spectrum disorders. Reprod. Toxicol. 2020, 91, 109–115. [Google Scholar] [CrossRef]
- Tseng, P.T.; Chen, Y.W.; Stubbs, B.; Carvalho, A.F.; Whiteley, P.; Tang, C.H.; Yang, W.C.; Chen, T.Y.; Li, D.J.; Chu, C.S.; et al. Maternal breastfeeding and autism spectrum disorder in children: A systematic review and meta-analysis. Nutr. Neurosci. 2019, 22, 354–362. [Google Scholar] [CrossRef]
- Veldkamp, S.A.M.; Zondervan-Zwijnenburg, M.A.J.; van Bergen, E.; Barzeva, S.A.; Tamayo-Martinez, N.; Becht, A.I.; van Beijsterveldt, C.E.M.; Meeus, W.; Branje, S.; Hillegers, M.H.J.; et al. Parental age in relation to offspring’s neurodevelopment. J. Clin. Child Adolesc. Psychol. 2020, 18, 1–13. [Google Scholar] [CrossRef]
- Kelly, F.J. Oxidative stress: Its role in air pollution and adverse health effects. Occup. Environ. Med. 2003, 60, 612–616. [Google Scholar] [CrossRef] [Green Version]
- Kalkbrenner, A.E.; Schmidt, R.J.; Penlesky, A.C. Environmental chemical exposures and autism spectrum disorders: A review of the epidemiological evidence. Curr. Probl. Pediatr. Adolesc. Health Care 2014, 44, 277–318. [Google Scholar] [CrossRef] [Green Version]
- Croonenberghs, J.; Wauters, A.; Devreese, K.; Verkerk, R.; Scharpe, S.; Bosmans, E.; Egyed, B.; Deboutte, D.; Maes, M. Increased serum albumin, γ globulin, immunoglobulin IgG, and IgG2 and IgG4 in autism. Psychol. Med. 2002, 32, 1457–1463. [Google Scholar] [CrossRef] [PubMed]
- Molloy, C.A.; Morrow, A.L.; Meinzen-Derr, J.; Schleifer, K.; Dienger, K.; Manning-Courtney, P.; Altaye, M.; Wills-Karp, M. Elevated cytokine levels in children with autism spectrum disorder. J. Neuroimmunol. 2006, 172, 198–205. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Chauhan, A.; Sheikh, A.M.; Patil, S.; Chauhan, V.; Li, X.M.; Ji, L.; Brown, T.; Malik, M. Elevated immune response in the brain of autistic patients. J. Neuroimmunol. 2009, 207, 111–116. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zerbo, O.; Yoshida, C.; Grether, J.K.; Van de Water, J.; Ashwood, P.; Delorenze, G.N.; Hansen, R.L.; Kharrazi, M.; Croen, L.A. Neonatal cytokines and chemokines and risk of autism spectrum disorder: The early markers for autism (EMA) study: A case-control study. J. Neuroinflamm. 2014, 11, 113. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Krakowiak, P.; Goines, P.E.; Tancredi, D.J.; Ashwood, P.; Hansen, R.L.; Hertz-Picciotto, I.; Van de Water, J. Neonatal cytokine profiles associated with autism spectrum disorder. Biol. Psychiatry 2015, 81, 442–451. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Abdallah, M.W.; Larsen, N.; Grove, J.; Norgaard-Pedersen, B.; Thorsen, P.; Mortensen, E.L.; Hougaard, D.M. Amniotic fluid inflammatory cytokines: Potential markers of immunologic dysfunction in autism spectrum disorders. World J. Biol. Psychiatry 2013, 14, 528–538. [Google Scholar] [CrossRef] [PubMed]
- Jones, K.L.; Croen, L.A.; Yoshida, C.K.; Heuer, L.; Hansen, R.; Zerbo, O.; DeLorenze, G.N.; Kharrazi, M.; Yolken, R.; Ashwood, P.; et al. Autism with intellectual disability is associated with increased levels of maternal cytokines and chemokines during gestation. Mol. Psychiatry 2017, 22, 273–279. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lucchina, L.; Depino, A.M. Altered peripheral and central inflammatory responses in a mouse model of autism. Autism Res. 2014, 7, 273–289. [Google Scholar] [CrossRef]
- Zhang, J.; Piantadosi, C.A. Mitochondrial oxidative stress after carbon monoxide hypoxia in the rat brain. J. Clin. Investig. 1992, 90, 1193–1199. [Google Scholar] [CrossRef]
- Wei, T.; Chen, C.; Hou, J.; Xin, W.; Mori, A. Nitric oxide induces oxidative stress and apoptosis in neuronal cells. Biochim. Biophys. Acta (BBA) Mol. Cell Res. 2000, 1498, 72–79. [Google Scholar] [CrossRef] [Green Version]
- Longo, L.D. The biological effects of carbon monoxide on the pregnant woman, fetus, and newborn infant. Am. J. Obstet. Gynecol. 1977, 129, 69–103. [Google Scholar] [CrossRef]
- Di Cera, E.; Doyle, M.L.; Morgan, M.S.; De Cristofaro, R.; Landolfi, R.; Bizzi, B.; Castagnola, M.; Gill, S.J. Carbon monoxide and oxygen binding to human hemoglobin F0. Biochemistry 1989, 28, 2631–2638. [Google Scholar] [CrossRef] [PubMed]
- Sangalli, M.R.; McLean, A.J.; Peek, M.J.; Rivory, L.P.; Le Couteur, D.G. Carbon monoxide disposition and permeability-surface area product in the foetal circulation of the perfused term human placenta. Placenta 2003, 24, 8–11. [Google Scholar] [CrossRef] [PubMed]
- Longo, L.D. Carbon monoxide: Effects on oxygenation of the fetus in utero. Science 1976, 194, 523–525. [Google Scholar] [CrossRef] [PubMed]
- Walters, D.M.; Breysse, P.N.; Wills-Karp, M. Ambient urban Baltimore particulate-induced airway hyperresponsiveness and inflammation in mice. Am. J. Respir. Crit. Care Med. 2001, 164, 1438–1443. [Google Scholar] [CrossRef] [PubMed]
- Dexter, S.C.; Pinar, H.; Malee, M.P.; Hogan, J.; Carpenter, M.W.; Vohr, B.R. Outcome of very low birth weight infants with histopathologic chorioamnionitis. Obstet. Gynecol. 2000, 96, 172–177. [Google Scholar] [PubMed]
- Nemmar, A.; Hoet, P.H.M.; Vanquickenborne, B.; Dinsdale, D.; Thomeer, M.; Hoylaerts, M.F.; Vanbilloen, H.; Mortelmans, L.; Nemery, B. Passage of inhaled particles into the blood circulation in humans. Circulation 2002, 105, 411–414. [Google Scholar] [CrossRef] [Green Version]
- Farahani, H.; Hasan, M. Nitrogen-Dioxide Induced Changes in Level of Free Fatty-Acids, Triglyceride, Esterified fatty-acid, ganglioside and lipase activity in the Guinea-Pig Brain. J. Environ. Sci. Health Part B Pestic. Food Contam. Agric. Wastes 1992, 27, 53–71. [Google Scholar] [CrossRef] [PubMed]
- Farahani, H.; Hasan, M. Effect of NO2 on Lipids and lipid-peroxidation in the CNS of the guinea-pig. Pharmacol. Toxicol. 1990, 66, 146–149. [Google Scholar] [CrossRef]
- Li, H.Y.; Xin, X.Y. Nitrogen dioxide (NO2) pollution as a potential risk factor for developing vascular dementia and its synaptic mechanisms. Chemosphere 2013, 92, 52–58. [Google Scholar] [CrossRef]
- Calderon-Garciduenas, L.; Torres-Jardon, R.; Kulesza, R.J.; Park, S.B.; D’Angiulli, A. Air pollution and detrimental effects on children’s brain. The need for a multidisciplinary approach to the issue complexity and challenges. Front. Hum. Neurosci. 2014, 8, 613. [Google Scholar] [PubMed] [Green Version]
- Levesque, S.; Taetzsch, T.; Lull, M.E.; Kodavanti, U.; Stadler, K.; Wagner, A.; Johnson, J.A.; Duke, L.; Kodavanti, P.; Surace, M.J.; et al. Diesel exhaust activates and primes microglia: Air pollution, neuroinflammation, and regulation of dopaminergic neurotoxicity. Environ. Health Perspect. 2011, 119, 1149–1155. [Google Scholar] [CrossRef] [Green Version]
- Suzuki, T.; Oshio, S.; Iwata, M.; Saburi, H.; Odagiri, T.; Udagawa, T.; Sugawara, I.; Umezawa, M.; Takeda, K. In utero exposure to a low concentration of diesel exhaust affects spontaneous locomotor activity and monoaminergic system in male mice. Part. Fibre Toxicol. 2010, 7, 7. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Woodward, N.C.; Haghani, A.; Johnson, R.G.; Hsu, T.M.; Saffari, A.; Sioutas, C.; Kanoski, S.E.; Finch, C.E.; Morgan, T.E. Prenatal and early life exposure to air pollution induced hippocampal vascular leakage and impaired neurogenesis in association with behavioral deficits. Transl. Psychiatry 2018, 8, 261. [Google Scholar] [CrossRef] [PubMed]
- Yokota, S.; Mizuo, K.; Moriya, N.; Oshio, S.; Sugawara, I.; Takeda, K. Effect of prenatal exposure to diesel exhaust on dopaminergic system in mice. Neurosci. Lett. 2009, 449, 38–41. [Google Scholar] [CrossRef] [PubMed]
- Kim, E.; Park, H.; Hong, Y.C.; Ha, M.; Kim, Y.; Kim, B.N.; Kim, Y.; Roh, Y.M.; Lee, B.E.; Ryu, J.M.; et al. Prenatal exposure to PM10 and NO2 and children’s neurodevelopment from birth to 24 months of age: Mothers and Children’s Environmental Health (MOCEH) study. Sci. Total Environ. 2014, 481, 439–445. [Google Scholar] [CrossRef]
- Hajat, A.; Diez-Roux, A.V.; Adar, S.D.; Auchincloss, A.H.; Lovasi, G.S.; O’Neill, M.S.; Sheppard, L.; Kaufman, J.D. Air pollution and individual and neighborhood socioeconomic status: Evidence from the Multi-Ethnic Study of Atherosclerosis (MESA). Environ. Health Perspect. 2013, 121, 1325–1333. [Google Scholar] [CrossRef] [Green Version]
- Stansfeld, S.A.; Berglund, B.; Clark, C.; Lopez-Barrio, I.; Fischer, P.; Ohrstrom, E.; Haines, M.M.; Head, J.; Hygge, S.; van Kamp, I.; et al. Aircraft and road traffic noise and children’s cognition and health: A cross-national study. Lancet 2005, 365, 1942–1949. [Google Scholar] [CrossRef]
- Lin, C.C.; Yang, S.K.; Lin, K.C.; Ho, W.C.; Hsieh, W.S.; Shu, B.C.; Chen, P.C. Multilevel analysis of air pollution and early childhood neurobehavioral development. Int. J. Environ. Res. Public Health 2014, 11, 6827–6841. [Google Scholar] [CrossRef] [Green Version]
- Ritz, B.; Liew, Z.; Yan, Q.; Cui, X.; Virk, J.; Ketzel, M.; Raaschou-Nielsen, O. Air pollution and autism in Denmark. Environ. Epidemiol. 2018, 2, e028. [Google Scholar] [CrossRef]
- Guxens, M.; Ghassabian, A.; Gong, T.; Garcia-Esteban, R.; Porta, D.; Giorgis-Allemand, L.; Almqvist, C.; Aranbarri, A.; Beelen, R.; Badaloni, C.; et al. Air pollution exposure during pregnancy and childhood autistic traits in four European population-based cohort studies: The ESCAPE Project. Environ. Health Perspect. 2016, 124, 133–140. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Morales-Suarez-Varela, M.; Peraita-Costa, I.; Llopis-Gonzalez, A. Systematic review of the association between particulate matter exposure and autism spectrum disorders. Environ. Res. 2017, 153, 150–160. [Google Scholar] [CrossRef] [PubMed]
- Fordyce, T.A.; Leonhard, M.J.; Chang, E.T. A critical review of developmental exposure to particulate matter, autism spectrum disorder, and attention deficit hyperactivity disorder. J. Environ. Sci. Health Part A Toxic/Hazard. Subst. Environ. Eng. 2018, 53, 174–204. [Google Scholar] [CrossRef]
- Wegiel, J.; Kuchna, I.; Nowicki, K.; Imaki, H.; Wegiel, J.; Marchi, E.; Ma, S.Y.; Chauhan, A.; Chauhan, V.; Bobrowicz, T.W.; et al. The neuropathology of autism: Defects of neurogenesis and neuronal migration, and dysplastic changes. Acta Neuropathol. 2010, 119, 755–770. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sidman, R.L.; Rakic, P. Neuronal migration, with special reference to developing human brain: A review. Brain Res. 1973, 62, 1–35. [Google Scholar] [CrossRef]
- De Graaf-Peters, V.B.; Hadders-Algra, M. Ontogeny of the human central nervous system: What is happening when? Early Hum. Dev. 2006, 82, 257–266. [Google Scholar] [CrossRef]
- Sarnat, H.B.; Nochlin, D.; Born, D.E. Neuronal nuclear antigen (NeuN): A marker of neuronal maturation in the early human fetal nervous system1. Brain Dev. 1998, 20, 88–94. [Google Scholar] [CrossRef]
- Samuelsen, G.B.; Larsen, K.B.; Bogdanovic, N.; Laursen, H.; Græm, N.; Larsen, J.F.; Pakkenberg, B. The changing number of cells in the human fetal forebrain and its subdivisions: A stereological analysis. Cereb. Cortex 2003, 13, 115–122. [Google Scholar] [CrossRef]
- Sarnat, H.B.; Flores-Sarnat, L. Integrative classification of morphology and molecular genetics in central nervous system malformations. Am. J. Med. Genet. Part A 2004, 126A, 386–392. [Google Scholar] [CrossRef]
- Mrzljak, L.; Uylings, H.B.M.; Kostovic, I.; van Eden, C.G. Prenatal development of neurons in the human prefrontal cortex. II. A quantitative Golgi study. J. Comp. Neurol. 1992, 316, 485–496. [Google Scholar] [CrossRef]
- Rakic, P.; Cameron, R.S.; Komuro, H. Recognition, adhesion, transmembrane signaling and cell motility in guided neuronal migration. Curr. Opin. Neurobiol. 1994, 4, 63–69. [Google Scholar] [CrossRef]
- Gupta, R.K.; Hasan, K.M.; Trivedi, R.; Pradhan, M.; Das, V.; Parikh, N.A.; Narayana, P.A. Diffusion tensor imaging of the developing human cerebrum. J. Neurosci. Res. 2005, 81, 172–178. [Google Scholar] [CrossRef] [PubMed]
- Gressens, P. Mechanisms and disturbances of neuronal migration. Pediatr. Res. 2000, 48, 725–730. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rakic, P. Pre- and post-developmental neurogenesis in primates. Clin. Neurosci. Res. 2002, 2, 29–39. [Google Scholar] [CrossRef]
- Dobbing, J.; Sands, J. Timing of neuroblast multiplication in developing human brain. Nature 1970, 226, 639–640. [Google Scholar] [CrossRef] [PubMed]




| N (%) 1 | |||
|---|---|---|---|
| Variables | ASD (N = 466) | Non-ASD (N = 62,453) | p-Value |
| Sex (male/female ratio) | 4.83 | 1.08 | <0.001 |
| Male | 386 (82.83) | 32,427 (51.92) | |
| Female | 80 (17.17) | 30,016 (47.72) | |
| Birth year | 0.026 | ||
| 1996 | 78 (16.74) | 13,377 (21.42) | |
| 1997 | 90 (19.31) | 13,538 (21.68) | |
| 1998 | 101 (21.67) | 11,343 (18.16) | |
| 1999 | 101 (21.67) | 11,809 (18.91) | |
| 2000 | 96 (20.60) | 12,386 (19.83) | |
| Followed year (mean ± SD 1) | 6.76 ± 4.02 | 15.30 ± 2.20 | |
| Comorbidity | |||
| Bipolar disorder | 9 (1.93) | 61 (0.10) | <0.001 |
| Depressive disorder | 15 (3.22) | 195 (0.31) | <0.001 |
| Anxiety | 66 (14.16) | 0 (0.00) | <0.001 |
| Phobic disorder | 3 (0.64) | 60 (0.10) | 0.012 |
| Obsessive compulsive disorder | 15 (3.22) | 70 (0.11) | <0.001 |
| Intellectual disabilities | 149 (31.97) | 777 (1.24) | <0.001 |
| Preterm birth | 10 (2.15) | 514 (0.82) | 0.006 |
| Air pollutant level (mean ± SD) | |||
| CO (ppm) | 0.89 ± 0.36 | 0.83 ± 0.32 | <0.001 |
| NO2 (ppb) | 25.47 ± 6.12 | 24.34 ± 6.01 | <0.001 |
| O3 (ppb) | 21.60 ± 3.61 | 21.99 ± 3.54 | 0.019 |
| SO2 (ppb) | 5.76 ± 3.29 | 5.80 ± 3.47 | 0.783 |
| PM10 (μg/m3) | 56.83 ± 15.24 | 60.27 ± 16.20 | <0.001 |
| Covariate | Hazard Ratio | (95% CI) |
|---|---|---|
| Male | 4.45 | (3.50–5.66) * |
| Calendar year | 1.12 | (1.05–1.20) * |
| Comorbidity | ||
| Bipolar disorder | 17.66 | (9.13–34.14) * |
| Depressive disorder | 9.68 | (5.79–16.18) * |
| Phobic disorder | 6.43 | (2.07–20.03) * |
| Obsessive compulsive disorder | 25.98 | (15.54–43.44) * |
| Intellectual disabilities | 34.26 | (28.20–41.63) * |
| Preterm | 2.77 | (1.48–5.19) * |
| Crude Hazard Ratio (95%CI) | Adjusted Hazard Ratio (95%CI) | ||
|---|---|---|---|
| Model 1 1 | Model 2 2 | ||
| CO (ppm) | |||
| Gestation | 1.65 (1.32–2.07) * | 1.77 (1.41–2.22) * | 1.88 (1.49–2.36) * |
| 1st Trimester | 1.68 (1.36–2.09) * | 1.79 (1.44–2.23) * | 1.93 (1.55–2.39) * |
| 2nd Trimester | 1.56 (1.25–1.96) * | 1.66 (1.33–2.09) * | 1.77 (1.41–2.22) * |
| 3rd Trimester | 1.57 (1.25–1.97) * | 1.68 (1.34–2.11) * | 1.75 (1.39–2.21) * |
| NO2 (10 ppb) | |||
| Gestation | 1.37 (1.18–1.60) * | 1.39 (1.19–1.62) * | 1.42 (1.22–1.66) * |
| 1st Trimester | 1.32 (1.17–1.50) * | 1.34 (1.19–1.52) * | 1.39 (1.22–1.58) * |
| 2nd Trimester | 1.22 (1.07–1.38) * | 1.22 (1.07–1.38) * | 1.25 (1.10–1.42) * |
| 3rd Trimester | 1.17 (1.04–1.33) * | 1.18 (1.04–1.34) * | 1.18 (1.03–1.34) * |
| O3 (10 ppb) | |||
| Gestation | 0.73 (0.56–0.96) * | 0.69 (0.52–0.91) * | 0.74 (0.56–0.97) * |
| 1st Trimester | 0.83 (0.68–1.02) | 0.81 (0.66–0.99) * | 0.85 (0.69–1.04) |
| 2nd Trimester | 0.82 (0.67–1.00) | 0.79 (0.64–0.97) * | 0.82 (0.67–1.02) |
| 3rd Trimester | 0.86 (0.70–1.05) | 0.83 (0.67–1.01) | 0.84 (0.68–1.03) |
| SO2 (ppb) | |||
| Gestation | 0.99 (0.97–1.02) | 1.00 (0.98–1.03) | 1.01 (0.99–1.04) |
| 1st Trimester | 1.00 (0.98–1.02) | 1.01 (0.99–1.03) | 1.02 (1.00–1.04) |
| 2nd Trimester | 0.99 (0.97–1.02) | 1.00 (0.98–1.03) | 1.01 (0.98–1.03) |
| 3rd Trimester | 0.99 (0.96–1.01) | 1.00 (0.97–1.02) | 1.00 (0.98–1.03) |
| PM10 (10 μg/m3) | |||
| Gestation | 0.86 (0.81–0.92) * | 0.87 (0.82–0.92) * | 0.90 (0.85–0.96) * |
| 1st Trimester | 0.95 (0.91–0.99) * | 0.96 (0.92–1.00) | 0.98 (0.94–1.03) |
| 2nd Trimester | 0.92 (0.89–0.96) * | 0.92 (0.88–0.96) * | 0.94 (0.90–0.99) * |
| 3rd Trimester | 0.90 (0.86–0.94) * | 0.90 (0.86–0.95) * | 0.91 (0.87–0.96) * |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, S.-Y.; Cheng, Y.-Y.; Guo, H.-R.; Tseng, Y.-C. Air Pollution during Pregnancy and Childhood Autism Spectrum Disorder in Taiwan. Int. J. Environ. Res. Public Health 2021, 18, 9784. https://doi.org/10.3390/ijerph18189784
Wang S-Y, Cheng Y-Y, Guo H-R, Tseng Y-C. Air Pollution during Pregnancy and Childhood Autism Spectrum Disorder in Taiwan. International Journal of Environmental Research and Public Health. 2021; 18(18):9784. https://doi.org/10.3390/ijerph18189784
Chicago/Turabian StyleWang, Shu-Yuan, Ya-Yun Cheng, How-Ran Guo, and Yen-Cheng Tseng. 2021. "Air Pollution during Pregnancy and Childhood Autism Spectrum Disorder in Taiwan" International Journal of Environmental Research and Public Health 18, no. 18: 9784. https://doi.org/10.3390/ijerph18189784








