Exposure to Secondhand Smoke: Inconsistency between Self-Response and Urine Cotinine Biomarker Based on Korean National Data during 2009–2018
Abstract
:1. Introduction
2. Materials and Methods
2.1. Korean NHANES (KNHANES)
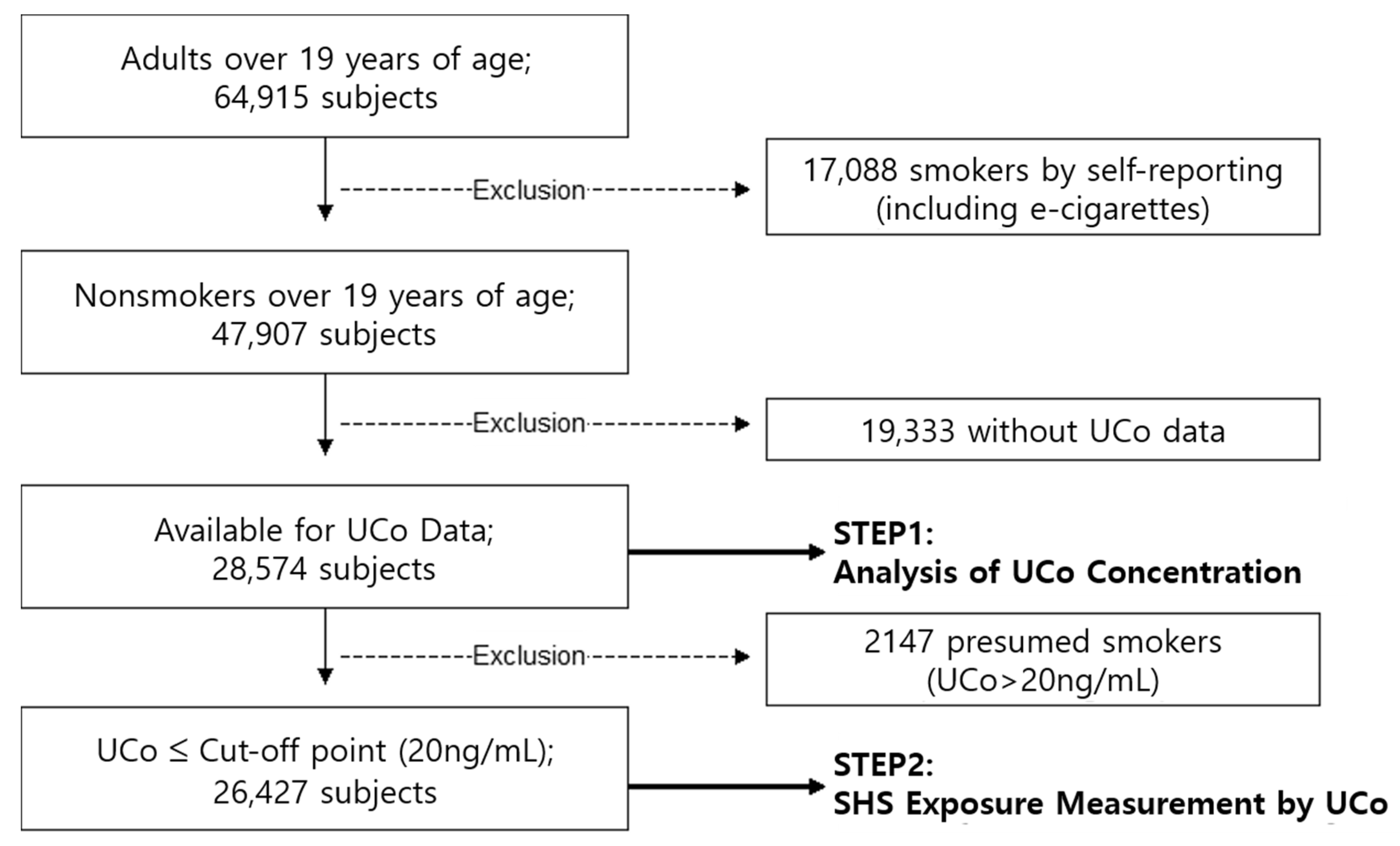
2.2. Subject Selection
2.3. Measurement of SHS Exposure Rate
2.4. Covariates
2.5. Statistical Analysis
3. Results
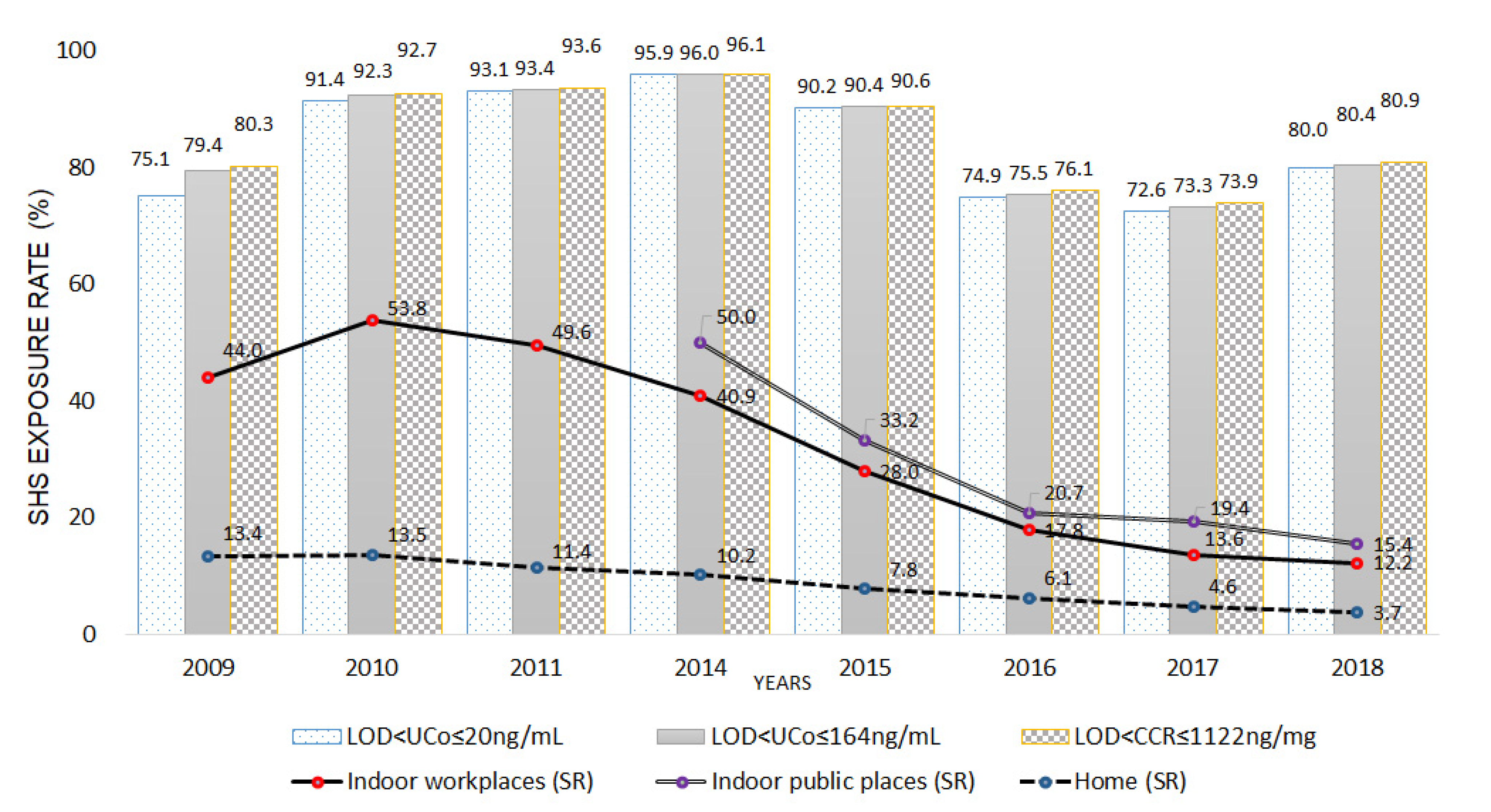
3.1. UCo Concentration and SHS Exposure Rate in Nonsmokers by Years
3.2. UCo Concentration in Nonsmokers by Subjects’ Characteristics
3.3. UCo-Measured SHS Exposure Rate by Subjects’ Characteristics
3.4. Factors Related to SHS Exposure Rates
4. Discussion
4.1. UCo Concentrations and SHS Exposure Rate
4.2. Inconsistency between Self-Reported and UCo-Measured SHS Exposure Rate
4.3. Factors Associated with SHS Exposure Measured Using Urine Cotinine Level
4.4. Policy Suggestions
4.5. Strength and Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Appendix A
References
- CDC. The Health Consequences of Smoking—50 Years of Progress: A Report of the Surgeon General; US Department of Health and Human Services, CDC, Coordinating Center for Health Promotion, National Center for Chronic Disease Prevention and Health Promotion, Office on Smoking and Health: Atlanta, GA, USA, 2014.
- Öberg, M.; Jaakkola, M.S.; Woodward, A.; Peruga, A.; Prüss-Ustün, A. Worldwide Burden of Disease from Exposure to Second-Hand Smoke: A Retrospective Analysis of Data from 192 Countries. Lancet 2011, 377, 139–146. [Google Scholar] [CrossRef]
- Makate, M.; Whetton, S.; Tait, R.J.; Dey, T.; Scollo, M.; Banks, E.; Norman, R.; Pidd, K.; Roche, A.M.; Allsop, S. Tobacco Cost of Illness Studies: A Systematic Review. Nicotine Tob. Res. 2020, 22, 458–465. [Google Scholar] [CrossRef] [PubMed]
- Oberg, M.; Woodward, A.; Jaakkola, M.S.; Peruga, A.; Prüss-Ustün, A. Global Estimate of the Burden of Disease from Second-Hand Smoke; World Health Organization: Geneva, Switzeland, 2010. [Google Scholar]
- Ino, T.; Ohtani, T.; Yoshimi, I. Urinary biomarkers for secondhand smoke. J. Clin. Lab. Anal. 2011, 25, 354–358. [Google Scholar] [CrossRef]
- Apelberg, B.J.; Hepp, L.M.; Avila-Tang, E.; Gundel, L.; Hammond, S.K.; Hovell, M.F.; Hyland, A.; Klepeis, N.E.; Madsen, C.C.; Navas-Acien, A.; et al. Environmental monitoring of secondhand smoke exposure. Tob. Control 2013, 22, 147–155. [Google Scholar] [CrossRef] [Green Version]
- Tsai, J.; Homa, D.M.; Gentzke, A.S.; Mahoney, M.; Sharapova, S.R.; Sosnoff, C.S.; Caron, K.T.; Wang, L.; Melstrom, P.C.; Trivers, K.F. Exposure to secondhand smoke among nonsmokers—United States, 1988–2014. Morb. Mortal. Wkly. Rep. 2018, 67, 1342. [Google Scholar] [CrossRef] [Green Version]
- Kwak, J.; Jeong, H.; Chun, S.; Bahk, J.H.; Park, M.; Byun, Y.; Lee, J.; Yim, H.W. Effectiveness of government anti-smoking policy on non-smoking youth in Korea: A 4-year trend analysis of national survey data. BMJ Open 2017, 7, e013984. [Google Scholar] [CrossRef] [Green Version]
- KCDC. Korea Health Statistics 2018: Korea National Health and Nutrition Examination Survey (KNHANES VII-3); KCDC: Cheongju, Korea, 2019.
- Hsieh, S.J.; Ware, L.B.; Eisner, M.D.; Yu, L.; Jacob III, P.; Havel, C.; Goniewicz, M.L.; Matthay, M.A.; Benowitz, N.L.; Calfee, C.S. Biomarkers increase detection of active smoking and secondhand smoke exposure in critically ill patients. Crit. Care Med. 2011, 39, 40. [Google Scholar] [CrossRef] [Green Version]
- Wong, S.L.; Malaison, E.; Hammond, D.; Leatherdale, S.T. Secondhand smoke exposure among Canadians: Cotinine and self-report measures from the Canadian Health Measures Survey 2007–2009. Nicotine Tob. Res. 2013, 15, 693–700. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jeong, B.Y.; Lim, M.K.; Yun, E.H.; Oh, J.K.; Park, E.Y.; Lee, D.H. Tolerance for and potential indicators of second-hand smoke exposure among nonsmokers: A comparison of self-reported and cotinine verified second-hand smoke exposure based on nationally representative data. Prev. Med. 2014, 67, 280–287. [Google Scholar] [CrossRef]
- Hackshaw, A.; Morris, J.K.; Boniface, S.; Tang, J.L.; Milenković, D. Low cigarette consumption and risk of coronary heart disease and stroke: Meta-analysis of 141 cohort studies in 55 study reports. BMJ 2018, 360, j5855. [Google Scholar] [CrossRef] [Green Version]
- Park, J.H.; Lee, C.K.; Kim, K.H.; Son, B.C.; Kim, J.H.; Suh, C.H.; Kim, S.Y.; Yu, S.D.; Choi, W.H.; Kim, D.H.; et al. Decrease in the urine cotinine concentrations of Korean non-smokers between 2009 and 2011 following implementation of stricter smoking regulations. Int. J. Hyg. Environ. Health 2016, 219, 123–128. [Google Scholar] [CrossRef]
- Jung, S.; Lee, I.S.; Kim, S.B.; Moon, C.S.; Jung, J.Y.; Kang, Y.A.; Park, M.S.; Kim, Y.S.; Kim, S.K.; Chang, J.; et al. Urine cotinine for assessing tobacco smoke exposure in Korean: Analysis of the Korea National Health and Nutrition Examination Survey (KNHANES). Tuberc. Respir. Dis. 2012, 73, 210–218. [Google Scholar] [CrossRef] [PubMed]
- Kang, Y.-H.; Lee, Y.-J.; Kim, H.-K.; Yun, Y.-H.; Jeong, S.-Y.; Lee, J.-S.; Park, J.-G. Usefulness of urinary cotinine test to distinguish smokers from nonsmokers. Ann. Lab. Med. 2003, 23, 92–97. [Google Scholar]
- KCDC. Raw data Guideline for the 7th Korea National Health and Nutrition Health Examination Survey; KCDC: Osong, Korea, 2019.
- Abdullah, A.S.; Driezen, P.; Sansone, G.; Nargis, N.; Hussain, G.A.; Quah, A.C.; Fong, G.T. Correlates of exposure to secondhand smoke (SHS) at home among non-smoking adults in Bangladesh: Findings from the ITC Bangladesh survey. BMC Pulm. Med. 2014, 14, 117. [Google Scholar] [CrossRef]
- Song, J.; Song, T.M.; Seo, D.C.; Jin, D.L.; Kim, J.S. Social big data analysis of information spread and perceived infection risk during the 2015 Middle East respiratory syndrome outbreak in South Korea. Cyberpsychol. Behav. Soc. Netw. 2017, 20, 22–29. [Google Scholar] [CrossRef] [PubMed]
- Berman, T.; Barnett-Itzhaki, Z.; Axelrod, R.; Keinan-Boker, L.; Shimony, T.; Goldsmith, R.; Göen, T.; Geva, H.; Rosen, L. Socioeconomic inequalities in exposure to environmental tobacco smoke in children in Israel. Env. Int. 2018, 121 Pt 1, 643–648. [Google Scholar] [CrossRef]
- Park, H.; Cho, S.I.; Lee, C. Second hand smoke exposure in workplace by job status and occupations. Ann. Occup. Environ. Med. 2019, 31, 3. [Google Scholar] [CrossRef]
- Vander Weg, M.W.; Cunningham, C.L.; Howren, M.B.; Cai, X. Tobacco use and exposure in rural areas: Findings from the Behavioral Risk Factor Surveillance System. Addict. Behav. 2011, 36, 231–236. [Google Scholar] [CrossRef]
- Homa, D.M.; Neff, L.J.; King, B.A.; Caraballo, R.S.; Bunnell, R.E.; Babb, S.D.; Garrett, B.E.; Sosnoff, C.S.; Wang, L. Vital signs: Disparities in nonsmokers’ exposure to secondhand smoke—United States, 1999–2012. Morb. Mortal. Wkly. Rep. 2015, 64, 103. [Google Scholar]
- Sun, L.Y.; Cheong, H.K.; Lee, E.W.; Kang, K.J.; Park, J.H. Affecting Factors of Secondhand Smoke Exposure in Korea: Focused on Different Exposure Locations. J. Korean Med. Sci. 2016, 31, 1362–1372. [Google Scholar] [CrossRef] [Green Version]
- Drobes, D.J. Concurrent alcohol and tobacco dependence: Mechanisms and treatment. Alcohol Res. Health 2002, 26, 136. [Google Scholar]
- Frazer, K.; Callinan, J.E.; McHugh, J.; van Baarsel, S.; Clarke, A.; Doherty, K.; Kelleher, C. Legislative smoking bans for reducing harms from secondhand smoke exposure, smoking prevalence and tobacco consumption. Cochrane Database Syst. Rev. 2016, 2, 2. [Google Scholar] [CrossRef] [PubMed]
- Hopkins, D.P.; Razi, S.; Leeks, K.D.; Kalra, G.P.; Chattopadhyay, S.K.; Soler, R.E.; Task Force on Community Preventive Services. Smokefree policies to reduce tobacco use: A systematic review. Am. J. Prev. Med. 2010, 38, S275–S289. [Google Scholar] [CrossRef] [PubMed]
- Siegel, M.; Albers, A.B.; Cheng, D.M.; Hamilton, W.L.; Biener, L. Local restaurant smoking regulations and the adolescent smoking initiation process: Results of a multilevel contextual analysis among Massachusetts youth. Arch. Pediatr. Adolesc. Med. 2008, 162, 477–483. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cho, H.-J. The status and future challenges of tobacco control policy in Korea. J. Prev. Med. Public Health 2014, 47, 129. [Google Scholar] [CrossRef]
- U.S. Department of Health and Human Services. The Health Consequences of Involuntary Exposure to Tobaco Smoke: A Report of the Surgeon General; U.S. Department of Health and Human Services, Centers for Disease Control and Prevention, Coordinating Center for Health Promotion, National Center for Chronic Disease Prevention and Health Promotion, Office on Smoking and Health: Atlanta, GA, USA, 2006. Available online: https://doh.dc.gov/sites/default/files/dc/sites/doh/publication/attachments/Executive_Summary_2nd_Hand_Smoke_SG_Report_2006.pdf (accessed on 17 June 2021).
- Rosen, L.J.; Lev, E.; Guttman, N.; Tillinger, E.; Rosenblat, S.; Zucker, D.M.; Myers, V. Parental perceptions and misconceptions of child tobacco smoke exposure. Nicotine Tob. Res. 2018, 20, 1369–1377. [Google Scholar] [CrossRef]
- Park, M.-B.; Choi, J.-K. Differences between the effects of conventional cigarettes, e-cigarettes and dual product use on urine cotinine levels. Tob. Induc. Dis. 2019, 17, 12. [Google Scholar] [CrossRef]
- Kim, J.; Lee, K. Characterization of urinary cotinine in non-smoking residents in smoke-free homes in the Korean National Environmental Health Survey (KoNEHS). BMC Public Health 2016, 16, 538. [Google Scholar] [CrossRef] [Green Version]
- Korea, S. Press Release: Korean Life Time Changes (1999~2014). 2016. Available online: https://kostat.go.kr/portal/korea/kor_nw/1/1/index.board?bmode=read&aSeq=352954 (accessed on 17 June 2021).
- Prochaska, J.J.; Grossman, W.; Young-Wolff, K.C.; Benowitz, N.L. Validity of self-reported adult secondhand smoke exposure. Tob. Control. 2015, 24, 48–53. [Google Scholar] [CrossRef] [Green Version]
- Boffetta, P.; Clark, S.; Shen, M.; Gislefoss, R.; Peto, R.; Andersen, A. Serum cotinine level as predictor of lung cancer risk. Cancer Epidemiol. Prev. Biomark. 2006, 15, 1184–1188. [Google Scholar] [CrossRef] [Green Version]
- Whincup, P.H.; Gilg, J.A.; Emberson, J.R.; Jarvis, M.J.; Feyerabend, C.; Bryant, A.; Walker, M.; Cook, D.G. Passive smoking and risk of coronary heart disease and stroke: Prospective study with cotinine measurement. BMJ 2004, 329, 200–205. [Google Scholar] [CrossRef] [Green Version]
- Lindsay, R.P.; Tsoh, J.Y.; Sung, H.Y.; Max, W. Secondhand smoke exposure and serum cotinine levels among current smokers in the USA. Tob. Control. 2016, 25, 224–231. [Google Scholar] [CrossRef]
- Youngmee, K.; Won-Kyung, C.; Lorraine, S.E. Effect of second-hand smoke exposure on lung function among non-smoking Korean women. Iran. J. Public Health 2013, 42, 1363. [Google Scholar]
- CDC. Secondhand Smoke: An Unequal Danger. 2015. Available online: https://www.cdc.gov/vitalsigns/tobacco/index.html (accessed on 17 June 2021).
- Lee, K.; Hwang, Y.; Hahn, E.J.; Bratset, H.; Robertson, H.; Rayens, M.K. Secondhand smoke exposure is associated with smoke-free laws but not urban/rural status. J. Air Waste Manag. Assoc. 2015, 65, 624–627. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Park, E.Y.; Yun, E.H.; Lim, M.K.; Lee, D.H.; Yang, W.; Jeong, B.Y.; Hwang, S.H. Consequences of Incomplete Smoke-free Legislation in the Republic of Korea: Results from Environmental and Biochemical Monitoring: Community based study. Cancer Res. Treat. 2016, 48, 376. [Google Scholar] [CrossRef]
- Shiels, M.S.; Rohrmann, S.; Menke, A.; Selvin, E.; Crespo, C.J.; Rifai, N.; Dobs, A.; Feinleib, M.; Guallar, E.; Platz, E.A. Association of cigarette smoking, alcohol consumption, and physical activity with sex steroid hormone levels in US men. Cancer Causes Control 2009, 20, 877–886. [Google Scholar] [CrossRef] [Green Version]
- Kim, S.; Jung, A. Optimum cutoff value of urinary cotinine distinguishing South Korean adult smokers from nonsmokers using data from the KNHANES (2008–2010). Nicotine Tob. Res. 2013, 15, 1608–1616. [Google Scholar] [CrossRef] [PubMed]
- Davis, R.; Stiles, M.F.; DeBethizy, J.D.; Reynolds, J.H. Dietary nicotine: A source of urinary cotinine. Food Chem. Toxicol. 1991, 29, 821–827. [Google Scholar] [CrossRef]
| Measures | Years | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 2009 | 2010 | 2011 | 2014 | 2015 | 2016 | 2017 | 2018 | ||
| UCo Concentration ** (Geometric meanand 95% CL, N = 28,574) | 9.28 (8.24–10.26) | 5.45 (4.85–6.13) | 3.88 (3.51–4.27) | 1.63 (1.53–1.74) | 1.49 (1.39–1.60) | 1.03 (0.97–1.10) | 1.06 (1.00–1.14) | 1.06 (1.00–1.13) | |
| SHS Exposure rate | Self-reporting, at indoor home (No. and weighted%, N = 28,569) | 449 (13.4) | 148 (13.5) | 142 (11.4) | 360 (10.2) | 321 (7.8) | 254 (6.1) | 223 (4.6) | 173 (3.7) |
| Self-reporting, at indoor public places * (No. and weighted%, N = 22,206) | - | - | - | 1701 (50.0) | 1261 (33.2) | 852 (20.7) | 808 (19.4) | 666 (15.4) | |
| Self-reporting, at indoor workplaces (No. and weighted%, N = 16,825) | 922 (44.0) | 424 (53.8) | 379 (49.6) | 788 (40.9) | 617 (28.0) | 412 (17.8) | 351 (13.6) | 337 (12.2) | |
| Cotinine measurements *** (No. and weighted%, N = 26,427) | 2115 (75.1) | 1097 (91.4) | 1159 (93.1) | 3415 (95.9) | 3639 (90.2) | 3214 (74.9) | 3234 (72.6) | 3574 (80.0) | |
| Variables | Participants (N, Weighted %) | UCo (ng/mL) | ||
|---|---|---|---|---|
| Geometric Mean | 95% CL | |||
| Sex (N = 28,574) | Male | 10,035 (40.4) | 2.0 | 1.9–2.1 |
| Female | 18,539 (59.6) | 1.7 | 1.6–1.8 | |
| Age (N = 28,574) | 65 over | 7485 (17.1) | 1.4 | 1.3–1.5 |
| 45–64 | 10,945 (37.9) | 1.6 | 1.5–1.7 | |
| 19–44 | 10,144 (45.0) | 2.2 | 2.1–2.3 | |
| Income (N = 28,436) | 4Q | 6651 (26.3) | 1.6 | 1.5–1.8 |
| 3Q | 7043 (24.7) | 1.7 | 1.6–1.8 | |
| 2Q | 7182 (24.9) | 1.8 | 1.7–1.9 | |
| 1Q | 7560 (26.3) | 2.0 | 1.9–2.2 | |
| Occupation (N = 28,574) | Blue-collar | 4535 (16.6) | 1.8 | 1.7–1.9 |
| White-collar | 9721 (38.3) | 1.8 | 1.7–1.9 | |
| Agriculture | 1520 (3.5) | 2.1 | 1.8–2.4 | |
| Unemployed | 12,799 (41.6) | 1.8 | 1.7–1.8 | |
| Region (N = 28,574) | Urban | 229,992 (84.2) | 1.8 | 1.7–1.8 |
| Rural | 5582 (15.8) | 2.0 | 1.8–2.3 | |
| House type (N = 28,574) | General | 13,715 (48.3) | 2.1 | 2.0–2.2 |
| Apartment | 14,859 (51.7) | 1.6 | 1.5–1.6 | |
| Monthly drinking (N = 28,553) | No | 14,709 (46.9) | 1.5 | 1.5–1.6 |
| Yes | 13,844 (53.1) | 2.1 | 2.0–2.2 | |
| SHS at home (N = 28,569) | No | 26,499 (92.4) | 1.7 | 1.6–1.7 |
| Yes | 2070 (7.6) | 4.4 | 3.9–4.9 | |
| SHS at indoor public places (N = 22,206) | No | 16,918 (73.2) | 1.1 | 1.1–1.2 |
| Yes | 5288 (26.8) | 1.7 | 1.6–1.9 | |
| SHS in indoor workplaces (N = 16,825) | No | 12,595 (73.1) | 1.4 | 1.4–1.5 |
| Yes | 4230 (26.9) | 3.5 | 3.2–3.8 | |
| Variables | LOD < UCo ≤ 20 ng/mL (N = 26,427) * | |
|---|---|---|
| Overall exposure | 21,447 (82.5) | |
| Sex | Male | 7789 (86.4) |
| Female | 13,658 (80.0) | |
| Age | 65 over | 5493 (78.5) |
| 45–64 | 8554 (84.4) | |
| 19–44 | 7400 (82.5) | |
| Income | 4Q | 5624 (81.5) |
| 3Q | 5441 (83.2) | |
| 2Q | 5304 (81.8) | |
| 1Q | 4980 (83.7) | |
| Occupation | Blue-collar | 3615 (87.9) |
| White-collar | 7466 (83.9) | |
| Agriculture | 1103 (81.2) | |
| Unemployed | 9263 (79.2) | |
| Region | Urban | 17,399 (82.8) |
| Rural | 4048 (80.9) | |
| House type | General | 10,597 (85.9) |
| Apartment | 10,850 (79.5) | |
| Monthly drinking | No | 10,890 (80.0) |
| Yes | 10,539 (84.9) | |
| SHS at home | No | 19,828 (81.7) |
| Yes | 1614 (93.0) | |
| SHS in indoor public places | No | 12,710 (79.6) |
| Yes | 4362 (89.3) | |
| SHS in indoor workplaces | No | 9599 (82.6) |
| Yes | 3330 (91.7) | |
| Variables | Univariate | Multiple | |
|---|---|---|---|
| LOD < UCo ≤ 20 ng/mL OR (95% CL) | |||
| Sex | Female (ref) vs. Male | 1.58 (1.46–1.72) | 1.43 (1.25–1.64) |
| Age | >65 (ref) | 1 | 1 |
| 45–64 | 1.30 (1.16–1.44) | 1.10 (0.90–1.34) | |
| 19–44 | 1.48 (1.33–1.64) | 1.21 (1.01–1.45) | |
| Income | 4Q (ref) | 1 | 1 |
| 3Q | 1.02 (0.92–1.14) | 0.94 (0.81–1.09) | |
| 2Q | 1.12 (0.99–1.27) | 1.06 (0.90–1.25) | |
| 1Q | 1.17 (1.03–1.33) | 1.07 (0.90–1.29) | |
| Occupation | Blue-collar worker (ref) | 1 | 1 |
| White-collar worker | 0.72 (0.63–0.81) | 0.77 (0.67–0.90) | |
| Agriculture | 0.60 (0.46–0.78) | 0.70 (0.53–0.93) | |
| Unemployed | 0.53 (0.47–0.59) | 1.13 (0.85–1.49) | |
| Region | Rural (ref) vs. Urban | 1.13 (0.96–1.34) | 1.29 (1.04–1.59) |
| House type | Apartment (ref) vs. General | 1.57 (1.40–1.75) | 1.55 (1.32–1.81) |
| Monthly drinking | No (ref) vs. Yes | 1.41 (1.30–1.51) | 1.17 (1.04–1.32) |
| SHS at home | No (ref) vs. exposure | 2.97 (2.33–3.79) | 3.90 (2.56–5.95) |
| SHS in indoor public places | No (ref) vs. exposure | 2.14 (1.90–2.41) | 1.51 (1.28–1.79) |
| SHS in indoor workplaces | No (ref) vs. exposure | 2.31 (1.97–2.72) | 2.06 (1.64–2.58) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sim, B.; Park, M.-B. Exposure to Secondhand Smoke: Inconsistency between Self-Response and Urine Cotinine Biomarker Based on Korean National Data during 2009–2018. Int. J. Environ. Res. Public Health 2021, 18, 9284. https://doi.org/10.3390/ijerph18179284
Sim B, Park M-B. Exposure to Secondhand Smoke: Inconsistency between Self-Response and Urine Cotinine Biomarker Based on Korean National Data during 2009–2018. International Journal of Environmental Research and Public Health. 2021; 18(17):9284. https://doi.org/10.3390/ijerph18179284
Chicago/Turabian StyleSim, Boram, and Myung-Bae Park. 2021. "Exposure to Secondhand Smoke: Inconsistency between Self-Response and Urine Cotinine Biomarker Based on Korean National Data during 2009–2018" International Journal of Environmental Research and Public Health 18, no. 17: 9284. https://doi.org/10.3390/ijerph18179284
APA StyleSim, B., & Park, M.-B. (2021). Exposure to Secondhand Smoke: Inconsistency between Self-Response and Urine Cotinine Biomarker Based on Korean National Data during 2009–2018. International Journal of Environmental Research and Public Health, 18(17), 9284. https://doi.org/10.3390/ijerph18179284







