Effects of Diet Control and Telemedicine-Based Resistance Exercise Intervention on Patients with Obesity and Knee Osteoarthritis: A Randomized Control Trial
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Design and Setting
2.2. Participants
2.3. Interventions
2.3.1. Diet Control Group
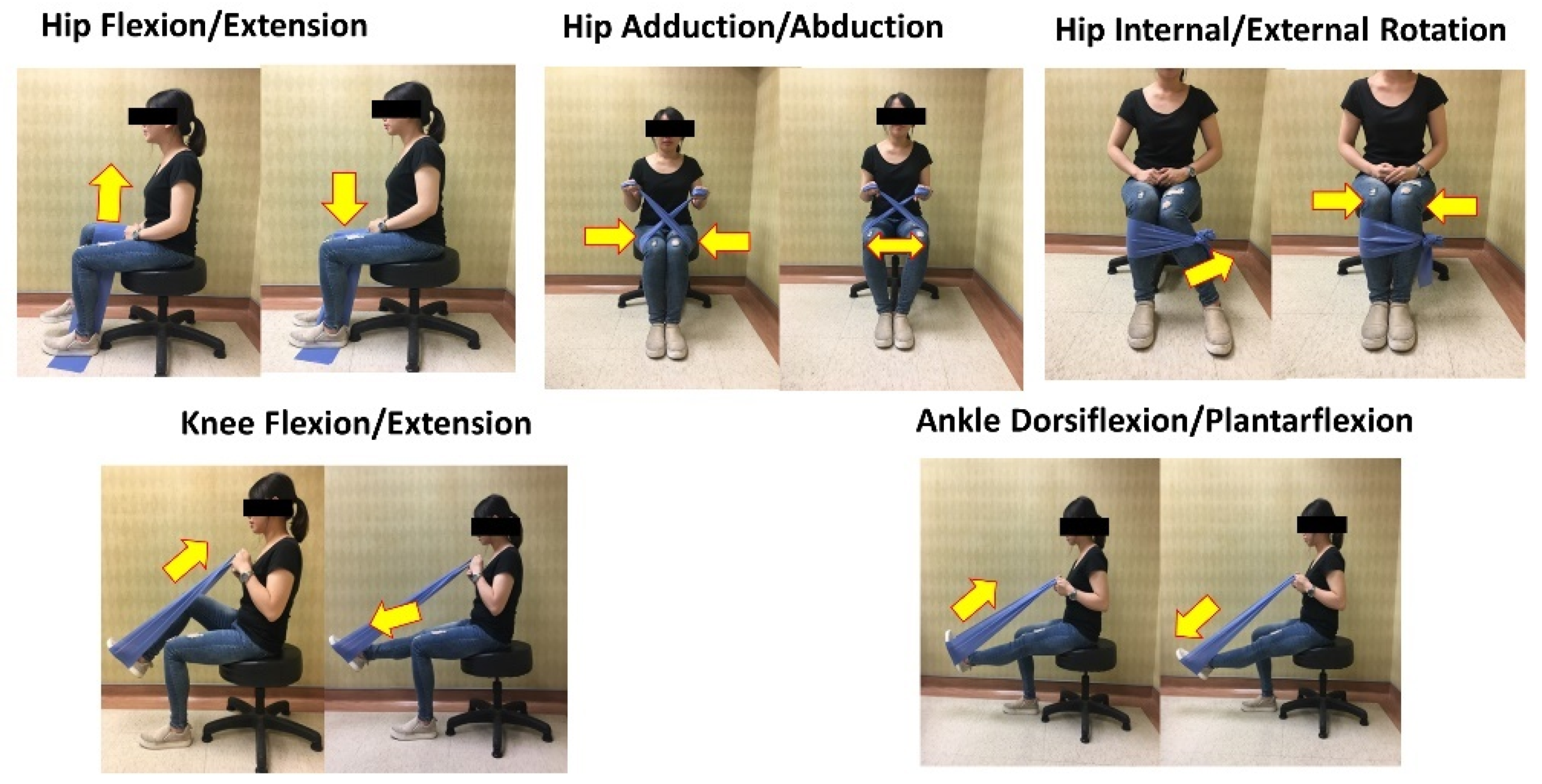
2.3.2. Elastic Band Resistance Exercise Group
2.3.3. Combined Group
2.4. Outcome Measures
2.4.1. Primary Outcomes
2.4.2. Secondary Outcomes
2.5. Statistical Analyses
3. Results
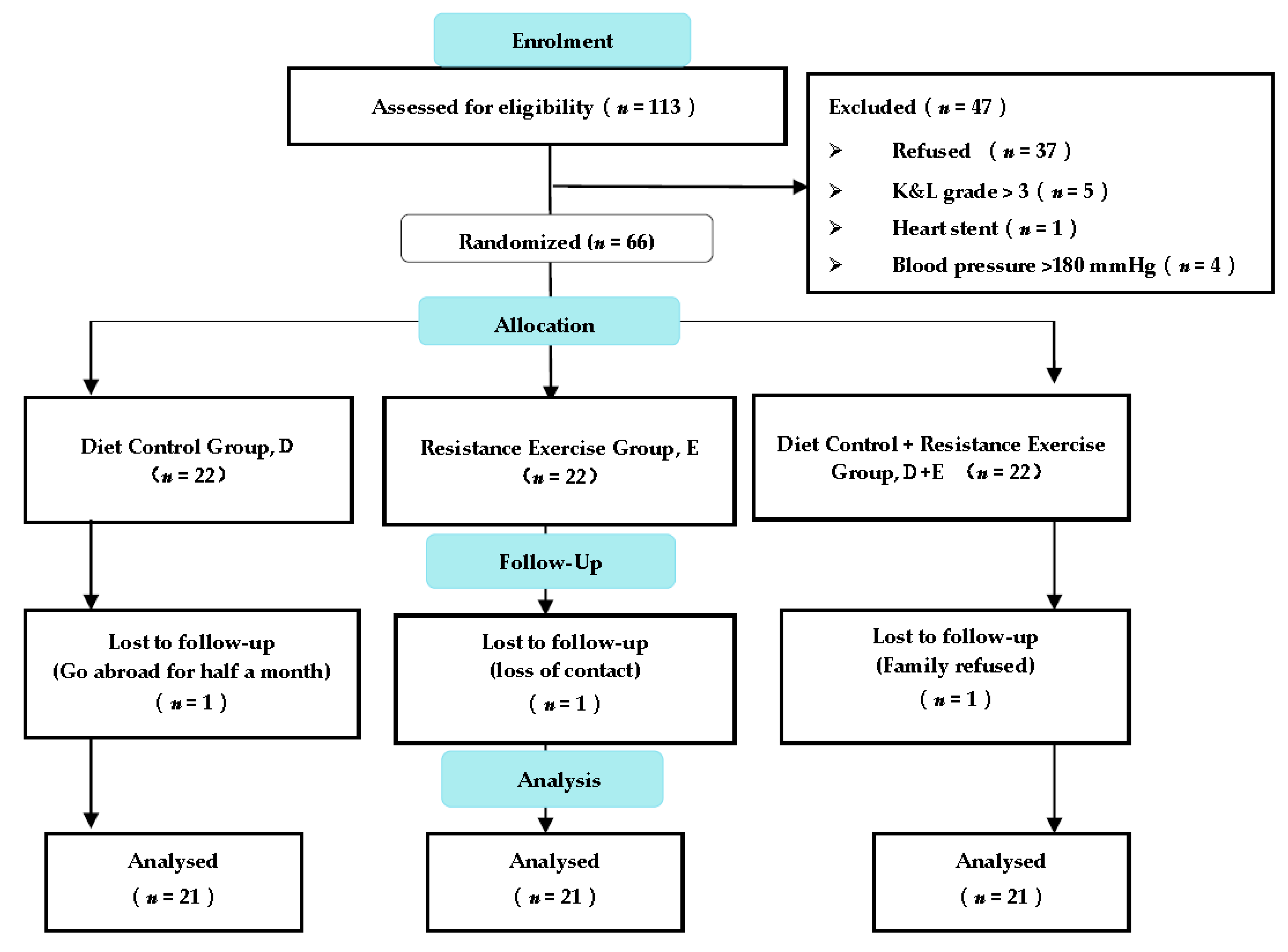
3.1. Study Flow for Participants
3.2. Primary Outcomes
3.2.1. Body Composition Analysis
3.2.2. WOMAC Analysis
3.3. Secondary Outcomes
3.3.1. Blood Biochemical Analysis
3.3.2. TUG Test
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Largo, R.; Herrero-Beaumont, G. Joint obesity as a pathogenic factor in osteoarthritis. Osteoarthr. Cartil. 2021. [Google Scholar] [CrossRef]
- Salih, S.; Sutton, P. Obesity, knee osteoarthritis and knee arthroplasty: A review. BMC Sports Sci. Med. Rehabil. 2013, 5, 1–6. [Google Scholar] [CrossRef] [Green Version]
- Lim, S.; Smith, C.A.; Costello, M.F.; MacMillan, F.; Moran, L.; Ee, C. Barriers and facilitators to weight management in overweight and obese women living in Australia with PCOS: A qualitative study. BMC Endocr. Disord. 2019, 19, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Hernandez, L.; Martinez-Arnau, F.M.; Perez-Ros, P.; Drehmer, E.; Pablos, A. Improved nutritional knowledge in the obese adult population modifies eating habits and serum and anthropometric markers. Nutrients 2020, 12, 3355. [Google Scholar] [CrossRef] [PubMed]
- Yumuk, V.; Tsigos, C.; Fried, M.; Schindler, K.; Busetto, L.; Micic, D.; Toplak, H. Obesity management task force of the european association for the study of European guidelines for obesity management in adults. Obes. Facts 2015, 8, 402–424. [Google Scholar] [CrossRef] [PubMed]
- Christensen, R.; Bartels, E.M.; Astrup, A.; Bliddal, H. Effect of weight reduction in obese patients diagnosed with knee osteoarthritis: A systematic review and meta-analysis. Ann. Rheum. Dis. 2007, 66, 433–439. [Google Scholar] [CrossRef] [PubMed]
- Christensen, P.; Bliddal, H.; Riecke, B.F.; Leeds, A.R.; Astrup, A.; Christensen, R. Comparison of a low-energy diet and a very low-energy diet in sedentary obese individuals: A pragmatic randomized controlled trial. Clin. Obes. 2011, 1, 31–40. [Google Scholar] [CrossRef] [PubMed]
- Fransen, M.; McConnell, S.; Harmer, A.R.; Van der Esch, M.; Simic, M.; Bennell, K.L. Exercise for osteoarthritis of the knee. Cochrane Database Syst. Rev. 2015, 1, CD004376. [Google Scholar] [CrossRef]
- Vincent, K.R.; Vincent, H.K. Resistance exercise for knee osteoarthritis. PM&R 2012, 4, S45–S52. [Google Scholar]
- Alrushud, A.S.; Rushton, A.B.; Kanavaki, A.M.; Greig, C.A. Effect of physical activity and dietary restriction interventions on weight loss and the musculoskeletal function of overweight and obese older adults with knee osteoarthritis: A systematic review and mixed method data synthesis. BMJ Open 2017, 7, e014537. [Google Scholar] [CrossRef] [Green Version]
- Blagojevic, M.; Jinks, C.; Jeffery, A.; Jordan, K. Risk factors for onset of osteoarthritis of the knee in older adults: A systematic review and meta-analysis. Osteoarthr. Cartil. 2010, 18, 24–33. [Google Scholar] [CrossRef] [Green Version]
- Messier, S.P.; Mihalko, S.L.; Legault, C.; Miller, G.D.; Nicklas, B.J.; DeVita, P.; Beavers, D.P.; Hunter, D.J.; Lyles, M.F.; Eckstein, F.; et al. Effects of intensive diet and exercise on knee joint loads, inflammation, and clinical outcomes among overweight and obese adults with knee osteoarthritis: The IDEA randomized clinical trial. JAMA 2013, 310, 1263–1273. [Google Scholar] [CrossRef]
- Chang, T.-F.; Liou, T.-H.; Chen, C.-H.; Huang, Y.-C.; Chang, K.-H. Effects of elastic-band exercise on lower-extremity function among female patients with osteoarthritis of the knee. Disabil. Rehabil. 2012, 34, 1727–1735. [Google Scholar] [CrossRef]
- Rogers, M.W.; Tamulevicius, N.; Semple, S.J.; Krkeljas, Z. Efficacy of home-based kinesthesia, balance & agility exercise training among persons with symptomatic knee osteoarthritis. J. Sports Sci. Med. 2012, 11, 751–758. [Google Scholar] [PubMed]
- Liao, C.-D.; Tsauo, J.-Y.; Huang, S.-W.; Ku, J.-W.; Hsiao, D.-J.; Liou, T.-H. Effects of elastic band exercise on lean mass and physical capacity in older women with sarcopenic obesity: A randomized controlled trial. Sci. Rep. 2018, 8, 2317. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Uthman, O.A.; van der Windt, D.A.; Jordan, J.L.; Dziedzic, K.S.; Healey, E.L.; Peat, G.M.; Foster, N.E. Exercise for lower limb osteoarthritis: Systematic review incorporating trial sequential analysis and network meta-analysis. BMJ 2013, 347, f5555. [Google Scholar] [CrossRef] [Green Version]
- Holden, M.A.; Button, K.; Collins, N.J.; Henrotin, Y.; Hinman, R.S.; Larsen, J.B.; Metcalf, B.; Master, H.; Skou, S.T.; Thoma, L.M.; et al. Guidance for implementing best practice therapeutic exercise for people with knee and hip osteoarthritis: What does the current evidence base tell us? Arthritis Care Res. 2020. [Google Scholar] [CrossRef] [PubMed]
- Kanavaki, A.M.; Rushton, A.; Efstathiou, N.; Alrushud, A.; Klocke, R.; Abhishek, A.; Duda, J.L. Barriers and facilitators of physical activity in knee and hip osteoarthritis: A systematic review of qualitative evidence. BMJ Open 2017, 7, e017042. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Phil, P.; Ellenbecker, T.S. Strength Band Training, 2nd ed.; Human Kinetics: Champaign, IL, USA, 2010. [Google Scholar]
- Nicolson, P.J.A.; Bennell, K.L.; Dobson, F.L.; Van Ginckel, A.; Holden, M.A.; Hinman, R.S. Interventions to increase adherence to therapeutic exercise in older adults with low back pain and/or hip/knee osteoarthritis: A systematic review and meta-analysis. Br. J. Sports Med. 2017, 51, 791–799. [Google Scholar] [CrossRef] [PubMed]
- Schafer, A.G.M.; Zalpour, C.; von Piekartz, H.; Hall, T.M.; Paelke, V. The efficacy of electronic health-supported home exercise interventions for patients with osteoarthritis of the knee: Systematic review. J. Med. Internet Res. 2018, 20, e9465. [Google Scholar] [CrossRef]
- Juhl, C.; Christensen, R.; Roos, E.M.; Zhang, W.; Lund, H. Impact of exercise type and dose on pain and disability in knee osteoarthritis: A systematic review and meta-regression analysis of randomized controlled trials. Arthritis Rheumatol. 2014, 66, 622–636. [Google Scholar] [CrossRef]
- Kolasinski, S.L.; Neogi, T.; Hochberg, M.C.; Oatis, C.; Guyatt, G.; Block, J.; Callahan, L.; Copenhaver, C.; Dodge, C.; Felson, D.; et al. 2019 American college of rheumatology/arthritis foundation guideline for the management of osteoarthritis of the hand, hip, and knee. Arthritis Rheumatol. 2020, 72, 220–233. [Google Scholar] [CrossRef]
- Chen, S.M.; Shen, F.C.; Chen, J.F.; Chang, W.D.; Chang, N.J. Effects of resistance exercise on glycated hemoglobin and functional performance in older patients with comorbid diabetes mellitus and knee osteoarthritis: A randomized trial. Int. J. Environ. Res. Public Health 2019, 17, 224. [Google Scholar] [CrossRef] [Green Version]
- Toda, Y.; Toda, T.; Takemura, S.; Wada, T.; Morimoto, T.; Ogawa, R. Change in body fat, but not body weight or metabolic correlates of obesity, is related to symptomatic relief of obese patients with knee osteoarthritis after a weight control program. J. Rheumatol. 1998, 25, 2181–2186. [Google Scholar]
- Waller, B.; Munukka, M.; Rantalainen, T.; Lammentausta, E.; Nieminen, M.T.; Kiviranta, I.; Kautiainen, H.; Hakkinen, A.; Kujala, U.M.; Heinonen, A. Effects of high intensity resistance aquatic training on body composition and walking speed in women with mild knee osteoarthritis: A 4-month RCT with 12-month follow-up. Osteoarthr. Cartil. 2017, 25, 1238–1246. [Google Scholar] [CrossRef] [Green Version]
- Wang, C.; Schmid, C.H.; Iversen, M.D.; Harvey, W.F.; Fielding, R.A.; Driban, J.B.; Price, L.L.; Wong, J.B.; Reid, K.F.; Rones, R.; et al. Comparative effectiveness of tai chi versus physical therapy for knee osteoarthritis: A randomized trial. Ann. Intern. Med. 2016, 165, 77–86. [Google Scholar] [CrossRef] [Green Version]
- Messier, S.P.; Loeser, R.F.; Miller, G.D.; Morgan, T.M.; Rejeski, W.J.; Sevick, M.A.; Ettinger, W.H., Jr.; Pahor, M.; Williamson, J.D. Exercise and dietary weight loss in overweight and obese older adults with knee osteoarthritis: The arthritis, diet, and activity promotion trial. Arthritis Rheum. 2004, 50, 1501–1510. [Google Scholar] [CrossRef] [PubMed]
- Munugoda, I.P.; Beavers, D.P.; Wirth, W.; Aitken, D.A.; Loeser, R.F.; Miller, G.D.; Lyles, M.; Carr, J.J.; Guermazi, A.; Hunter, D.J.; et al. The effect of weight loss on the progression of meniscal extrusion and size in knee osteoarthritis: A post-hoc analysis of the Intensive Diet and Exercise for Arthritis (IDEA) trial. Osteoarthr. Cartil. 2020, 28, 410–417. [Google Scholar] [CrossRef] [PubMed]
- Beavers, D.P.; Beavers, K.M.; Loeser, R.F.; Walton, N.R.; Lyles, M.F.; Nicklas, B.J.; Shapses, S.A.; Newman, J.J.; Messier, S.P. The independent and combined effects of intensive weight loss and exercise training on bone mineral density in overweight and obese older adults with osteoarthritis. Osteoarthr. Cartil. 2014, 22, 726–733. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Davis, A.M.; Perruccio, A.V.; Canizares, M.; Hawker, G.A.; Roos, E.M.; Maillefert, J.-F.; Lohmander, L.S. Comparative, validity and responsiveness of the HOOS-PS and KOOS-PS to the WOMAC physical function subscale in total joint replacement for osteoarthritis. Osteoarthr. Cartil. 2009, 17, 843–847. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dobson, F.; Hinman, R.S.; Roos, E.M.; Abbott, J.H.; Stratford, P.; Davis, A.M.; Buchbinder, R.; Snyder-Mackler, L.; Henrotin, Y.; Thumboo, J.; et al. OARSI recommended performance-based tests to assess physical function in people diagnosed with hip or knee osteoarthritis. Osteoarthr. Cartil. 2013, 21, 1042–1052. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Alghadir, A.; Anwer, S.; Brismee, J.M. The reliability and minimal detectable change of timed up and go test in individuals with grade 1-3 knee osteoarthritis. BMC Musculoskelet Disord 2015, 16, 1–7. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- World Medical Association. World medical association declaration of Helsinki: Ethical principles for medical research involving human subjects. JAMA 2013, 310, 2191–2194. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kohn, M.D.; Sassoon, A.A.; Fernando, N.D. Classifications in brief: Kellgren-lawrence classification of osteoarthritis. Clin. Orthop. Relat. Res. 2016, 474, 1886–1893. [Google Scholar] [CrossRef] [Green Version]
- Arendt-Nielsen, L.; Nie, H.; Laursen, M.B.; Laursen, B.S.; Madeleine, P.; Simonsen, O.H.; Graven-Nielsen, T. Sensitization in patients with painful knee osteoarthritis. Pain 2010, 149, 573–581. [Google Scholar] [CrossRef]
- Arab, A.; Askari, G.; Golshiri, P.; Feizi, A.; Hekmatnia, A.; Iraj, B.; Nourian, M. The effect of a lifestyle modification education on adiposity measures in overweight and obese nonalcoholic fatty liver disease patients. Int. J. Prev. Med. 2017, 8, 10. [Google Scholar] [PubMed]
- Lee, L.C.; Hsu, P.S.; Hsieh, K.C.; Chen, Y.Y.; Chu, L.P.; Lu, H.K.; Chiu, Y.C.; Li, L.; Lai, C.L. Standing 8-electrode bioelectrical impedance analysis as an alternative method to estimate visceral fat area and body fat mass in Athletes. Int. J. Gen. Med. 2021, 14, 539–548. [Google Scholar] [CrossRef]
- Escobar, A.; Quintana, J.; Bilbao, A.; Arostegui, I.; Lafuente, I.; Vidaurreta, I. Responsiveness and clinically important differences for the WOMAC and SF-36 after total knee replacement. Osteoarthr. Cartil. 2007, 15, 273–280. [Google Scholar] [CrossRef] [Green Version]
- Collins, N.J.; Misra, D.; Felson, D.T.; Crossley, K.M.; Roos, E.M. Measures of knee function: International knee documentation committee (IKDC) subjective knee evaluation form, knee injury and osteoarthritis outcome score (KOOS), knee injury and osteoarthritis outcome score physical function short form (KOOS-PS), knee outcome survey activities of daily living scale (kos-adl), lysholm knee scoring scale, oxford knee score (OKS), western ontario and McMaster universities osteoarthritis index (WOMAC), activity rating scale (ARS), and tegner activity score (TAS). Arthritis Care Res. 2011, 63, S208–S228. [Google Scholar]
- Basaran, S.; Guzel, R.; Seydaoglu, G.; Guler-Uysal, F. Validity, reliability, and comparison of the WOMAC osteoarthritis index and Lequesne algofunctional index in Turkish patients with hip or knee osteoarthritis. Clin. Rheumatol. 2010, 29, 749–756. [Google Scholar] [CrossRef]
- Wright, A.A.; Cook, C.E.; Baxter, G.D.; Dockerty, J.D.; Abbott, J.H. A comparison of 3 methodological approaches to defining major clinically important improvement of 4 performance measures in patients with hip osteoarthritis. J. Orthop. Sports Phys. Ther. 2011, 41, 319–327. [Google Scholar] [CrossRef] [PubMed]
- Mihalko, S.L.; Cox, P.; Beavers, D.P.; Miller, G.D.; Nicklas, B.J.; Lyles, M.; Hunter, D.J.; Eckstein, F.; Guermazi, A.; Loeser, R.F.; et al. Effect of intensive diet and exercise on self-efficacy in overweight and obese adults with knee osteoarthritis: The IDEA randomized clinical trial. Transl. Behav. Med. 2019, 9, 227–235. [Google Scholar] [CrossRef]
- Lee, S.; Lee, D.K. What is the proper way to apply the multiple comparison test? Korean J. Anesthesiol. 2020, 73, 572. [Google Scholar] [CrossRef]
- Lakens, D. Calculating and reporting effect sizes to facilitate cumulative science: A practical primer for t-tests and ANOVAs. Front. Psychol. 2013, 4, 863. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hunter, D.; Beavers, D.; Eckstein, F.; Guermazi, A.; Loeser, R.F.; Nicklas, B.; Mihalko, S.; Miller, G.; Lyles, M.; DeVita, P. The intensive diet and exercise for arthritis (IDEA) trial: 18-month radiographic and MRI outcomes. Osteoarthr. Cartil. 2015, 23, 1090–1098. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Christensen, P.; Henriksen, M.; Bartels, E.M.; Leeds, A.R.; Meinert Larsen, T.; Gudbergsen, H.; Riecke, B.F.; Astrup, A.; Heitmann, B.L.; Boesen, M. Long-term weight-loss maintenance in obese patients with knee osteoarthritis: A randomized trial. Am. J. Clin. Nutr. 2017, 106, 755–763. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Riecke, B.F.; Christensen, R.; Christensen, P.; Leeds, A.R.; Boesen, M.; Lohmander, L.S.; Astrup, A.; Bliddal, H. Comparing two low-energy diets for the treatment of knee osteoarthritis symptoms in obese patients: A pragmatic randomized clinical trial. Osteoarthr. Cartil. 2010, 18, 746–754. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Luppino, F.S.; de Wit, L.M.; Bouvy, P.F.; Stijnen, T.; Cuijpers, P.; Penninx, B.W.; Zitman, F.G. Overweight, obesity, and depression: A systematic review and meta-analysis of longitudinal studies. Arch. Gen. Psychiatry 2010, 67, 220–229. [Google Scholar] [CrossRef]
- Hall, M.; Castelein, B.; Wittoek, R.; Calders, P.; Van Ginckel, A. Diet-induced weight loss alone or combined with exercise in overweight or obese people with knee osteoarthritis: A systematic review and meta-analysis. Semin. Arthritis Rheum. 2019, 48, 765–777. [Google Scholar] [CrossRef]
- Fukumoto, Y.; Tateuchi, H.; Ikezoe, T.; Tsukagoshi, R.; Akiyama, H.; So, K.; Kuroda, Y.; Ichihashi, N. Effects of high-velocity resistance training on muscle function, muscle properties, and physical performance in individuals with hip osteoarthritis: A randomized controlled trial. Clin. Rehabil. 2014, 28, 48–58. [Google Scholar] [CrossRef]
- Rogers, M.W.; Tamulevicius, N.; Coetsee, M.F.; Curry, B.F.; Semple, S.J. Knee osteoarthritis and the efficacy of kinesthesia, balance & agility exercise training: A pilot study. Int. J. Exerc. Sci. 2011, 4, 124–132. [Google Scholar]
- Lacroix, A.; Kressig, R.W.; Muehlbauer, T.; Gschwind, Y.J.; Pfenninger, B.; Bruegger, O.; Granacher, U. Effects of a supervised versus an unsupervised combined balance and strength training program on balance and muscle power in healthy older adults: A randomized controlled trial. Gerontology 2016, 62, 275–288. [Google Scholar] [CrossRef] [PubMed]
- Toft-Petersen, A.P.; Tilsted, H.H.; Aarøe, J.; Rasmussen, K.; Christensen, T.; Griffin, B.A.; Aardestrup, I.V.; Andreasen, A.; Schmidt, E.B. Small dense LDL particles-a predictor of coronary artery disease evaluated by invasive and CT-based techniques: A case-control study. Lipids Health Dis. 2011, 10, 21. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Choi, H.; Shim, J.-S.; Lee, M.H.; Yoon, Y.M.; Choi, D.P.; Kim, H.C. Comparison of formulas for calculating low-density lipoprotein cholesterol in general population and high-risk patients with cardiovascular disease. Korean Circ. J. 2016, 46, 688–698. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, H.-S.; Kim, D.-G. Effect of long-term resistance exercise on body composition, blood lipid factors, and vascular compliance in the hypertensive elderly men. J. Exerc. Rehabil. 2013, 9, 271–277. [Google Scholar] [CrossRef] [PubMed]
- Sacks, F.M.; Lichtenstein, A.H.; Wu, J.H.; Appel, L.J.; Creager, M.A.; Kris-Etherton, P.M.; Miller, M.; Rimm, E.B.; Rudel, L.L.; Robinson, J.G. Dietary fats and cardiovascular disease: A presidential advisory from the American Heart Association. Circulation 2017, 136, e1–e23. [Google Scholar] [CrossRef] [PubMed]
- Du, J.Y.; Sivasundaram, L.; Trivedi, N.N.; Voos, J.E.; Victoroff, B.N. Obesity is preferentially associated with patellofemoral compartment wear: A magnetic resonance imaging assessment. J. Am. Acad. Orthop. Surg. 2021, 29, e722–e731. [Google Scholar] [CrossRef]
- Messier, S.P.; Beavers, D.P.; Mihalko, S.L.; Miller, G.D.; Lyles, M.F.; Hunter, D.J.; Carr, J.J.; Eckstein, F.; Guermazi, A.; Loeser, R.F.; et al. The effects of intensive dietary weight loss and exercise on gait in overweight and obese adults with knee osteoarthritis. The intensive diet and exercise for arthritis (IDEA) trial. J. Biomech. 2020, 98, 109477. [Google Scholar] [CrossRef]
- Coppini, L.Z.; Waitzberg, D.L.; Campos, A.C. Limitations and validation of bioelectrical impedance analysis in morbidly obese patients. Curr. Opin. Clin. Nutr. Metab. Care 2005, 8, 329–332. [Google Scholar] [CrossRef]
- Marra, M.; Sammarco, R.; De Lorenzo, A.; Iellamo, F.; Siervo, M.; Pietrobelli, A.; Donini, L.M.; Santarpia, L.; Cataldi, M.; Pasanisi, F.; et al. Assessment of body composition in health and disease using bioelectrical impedance analysis (BIA) and dual energy X-ray absorptiometry (DXA): A critical overview. Contrast Media Mol. Imaging 2019, 2019, 1–9. [Google Scholar] [CrossRef]
| Characteristics | D Group (n = 21) | E Group (n = 21) | D + E Group (n = 21) | p |
|---|---|---|---|---|
| Gender | ||||
| Male/female Age (year) | 9/12 66.0 (3.9) | 8/13 64.2 (4.1) | 6/15 65.6 (3.9) | 0.779 0.308 |
| Body height (cm) | 159.32 (5.64) | 159.77 (7.44) | 158.93 (6.82) | 0.578 |
| Body weight (kg) | 78.37 (8.50) | 79.00 (10.78) | 77.53 (10.17) | 0.891 |
| Body mass index (kg/m2) | 30.80 (2.58) | 30.84 (2.47) | 31.10 (2.60) | 0.915 |
| Body fat (%) waistline (cm) | 35.18 (6.00) 102.12 (8.10) | 34.64 (6.47) 102.07 (10.91) | 36.2 (5.27) 100.35 (7.25) | 0.690 0.764 |
| Kellgren–Lawrence grade | 1.76 (0.81) | 1.62 (0.65) | 1.81 (0.85) | 0.528 |
| I, n (%) | 10 (47.6) | 11 (50) | 10 (47.6) | |
| II, n (%) | 6 (28.6) | 8 (38.1) | 5 (23.8) | |
| III, n (%) | 5 (23.8) | 2 (9.1) | 6 (28.6) | |
| Drug, n (%) | ||||
| Antihyperlipidemic | 15 (71.4) | 13 (61.9) | 12 (57.1) | 0.719 |
| Antiarthritic | 12 (57.1) | 12 (57.1) | 10 (47.6) | 0.856 |
| VAS (baseline) | 6.19(1.03) | 6.62 (1.17) | 6.29 (1.06) | 0.399 |
| Outcomes | D | E | D + E |
|---|---|---|---|
| Body weight (kg) | |||
| Baseline | 78.37 ± 8.50 | 78.99 ± 10.78 | 77.53 ± 10.17 |
| Post-intervention | 74.96 ± 8.59 | 78.63 ± 11.00 | 74.03 ± 10.11 |
| Change | −3.41 ± 0.75 $ | −0.36 ± 1.16 | −3.50 ± 0.97 $ |
| Effect size | 0.4 | 0.03 | 0.35 |
| p | <0.001 | 0.168 | <0.001 |
| Body mass index (kg/m2) | |||
| Baseline | 30.8± 2.57 | 30.84± 2.47 | 31.10± 2.60 |
| Post-intervention | 29.45± 2.59 | 30.69± 2.59 | 29.7± 2.64 |
| Change | −1.35 ± 0.33 $ | −0.15± 0.46 | −1.41± 0.39 $ |
| Effect size | 0.52 | 0.06 | 0.53 |
| p | <0.001 | 0.161 | <0.001 |
| Body fat (%) | |||
| Baseline | 35.18 ± 6.01 | 34.64± 6.48 | 36.20± 5.27 |
| Post-intervention | 33.97 ± 6.47 | 33.48± 6.46 | 34.32± 5.50 |
| Change | −1.20 ± 0.81 | −1.17± 0.75 | −1.87± 0.78 #,$ |
| Effect size | 0.19 | 0.18 | 0.35 |
| p | <0.001 | <0.001 | <0.001 |
| Soft lean mass (kg) | |||
| Baseline | 46.29 ± 6.25 | 47.05 ± 8.41 | 45.64 ± 8.97 |
| Post-intervention | 45.05 ± 6.38 | 47.83 ± 8.53 | 44.46 ± 8.55 |
| Change | −1.23 ± 0.65 $ | 0.78 ± 0.64 | −1.18 ± 1.56 $ |
| Effect size | 0.2 | 0.1 | 0.13 |
| p | <0.001 | <0.001 | 0.002 |
| Lower-limb muscle mass (kg) | |||
| Baseline | 17.59 ± 2.62 | 17.37 ± 3.2 | 17.34 ± 3.57 |
| Post-intervention | 16.85 ± 2.60 | 17.89 ± 3.37 | 17.17 ± 3.43 |
| Change | −0.75 ± 0.40 $ | 0.52 ± 0.35 | −0.17 ± 0.55 #,$ |
| Effect size | 0.28 | 0.16 | 0.05 |
| p | <0.001 | <0.001 | <0.001 |
| Outcomes | D | E | D + E |
|---|---|---|---|
| Total Score | |||
| Baseline | 34.57 ± 7.69 | 30.24 ± 6.06 | 38.86 ± 7.98 |
| Post-intervention | 25.33 ± 6.55 | 22.62 ± 5.41 | 25.81 ± 6.61 |
| Change | −9.24 ± 3.59 | −7.62 ± 2.64 | −13.05 ± 4.09 #,$ |
| Effect size | 1.29 | 1.33 | 1.78 |
| p | <0.001 | <0.001 | <0.001 |
| Pain | |||
| Baseline | 6.48 ± 2.21 | 6.05 ± 1.99 | 7.43 ± 2.01 |
| Post-intervention | 4.33 ± 1.65 | 4.14 ± 1.46 | 4.48 ± 1.44 |
| Change | −2.14 ± 1.28 | −1.90 ± 1.48 | −2.95 ± 1.12 $ |
| Effect size | 1.1 | 1.09 | 1.69 |
| p | <0.001 | <0.001 | <0.001 |
| Stiffness | |||
| Baseline | 2.91 ± 1.09 | 1.33 ± 1.15 | 2.86 ± 1.59 |
| Post-intervention | 1.86 ± 0.66 | 0.90 ± 0.83 | 1.38 ± 0.97 |
| Change | −1.05 ± 0.81 | −0.43 ± 0.68 | −1.48 ± 1.03 $ |
| Effect size | 1.17 | 0.21 | 1.12 |
| p | <0.001 | <0.001 | <0.001 |
| Physical Function | |||
| Baseline | 25.19 ± 5.62 | 22.86 ± 4.30 | 28.57 ± 5.76 |
| Post-intervention | 19.43 ± 5.29 | 17.76 ± 3.82 | 19.95 ± 4.79 |
| Change | −5.76 ± 2.84 | −5.10 ± 1.70 | −8.62 ± 3.58 #,$ |
| Effect size | 1.06 | 1.25 | 1.63 |
| p | <0.001 | <0.001 | <0.001 |
| Outcomes | D | E | D + E |
|---|---|---|---|
| Total cholesterol (mg/dL) | |||
| Baseline | 177.17 ± 23.35 | 169.95 ± 25.06 | 177.48 ± 27.04 |
| Post-intervention | 160.10 ± 20.16 | 165.81 ± 26.89 | 151.52 ± 18.03 |
| Change | −17.57 ± 18.07 | −4.14 ± 13.40 | −25.95 ± 23.27 $ |
| Effect size | 0.78 | 0.16 | 1.13 |
| p | <0.001 | 0.175 | <0.001 |
| LDL (mg/dL) | |||
| Baseline | 99.47 ± 24.19 | 94.47 ± 24.19 | 97.57 ± 24.10 |
| Post-intervention | 84.38 ± 18.69 | 91.76 ± 24.32 | 79.52 ± 14.27 |
| Change | −15.24 ± 15.17 $ | −2.71 ± 13.94 | −18.05 ± 19.78 $ |
| Effect size | 0.7 | 0.11 | 0.91 |
| p | <0.001 | 0.325 | <0.001 |
| Triglycerides (mg/dL) | |||
| Baseline | 145.40 ± 52.95 | 128.81 ± 62.68 | 140.62 ± 54.57 |
| Post-intervention | 110.75 ± 48.77 | 118.05 ± 47.68 | 97.33 ± 35.79 |
| Change | −34.65 ± 16.17 | −10.76 ± 48.92 | −43.29 ± 26.37 $ |
| Effect size | 0.68 | 0.19 | 0.94 |
| p | <0.001 | 0.325 | <0.001 |
| Outcomes | D | E | D + E |
|---|---|---|---|
| TUG(s) | |||
| Baseline | 11.25 ± 2.15 | 10.51 ± 1.97 | 10.98 ± 1.98 |
| Post-intervention | 9.92 ± 1.68 | 9.59 ± 1.56 | 9.37 ± 1.61 |
| Change | −1.33 ± 0.59 | −0.92 ± 0.67 | −1.61 ± 0.52 $ |
| Effect size | 0.69 | 0.52 | 0.89 |
| p | <0.001 | <0.001 | < 0.001 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hsu, Y.-I.; Chen, Y.-C.; Lee, C.-L.; Chang, N.-J. Effects of Diet Control and Telemedicine-Based Resistance Exercise Intervention on Patients with Obesity and Knee Osteoarthritis: A Randomized Control Trial. Int. J. Environ. Res. Public Health 2021, 18, 7744. https://doi.org/10.3390/ijerph18157744
Hsu Y-I, Chen Y-C, Lee C-L, Chang N-J. Effects of Diet Control and Telemedicine-Based Resistance Exercise Intervention on Patients with Obesity and Knee Osteoarthritis: A Randomized Control Trial. International Journal of Environmental Research and Public Health. 2021; 18(15):7744. https://doi.org/10.3390/ijerph18157744
Chicago/Turabian StyleHsu, Yen-I, Ying-Chou Chen, Chia-Lun Lee, and Nai-Jen Chang. 2021. "Effects of Diet Control and Telemedicine-Based Resistance Exercise Intervention on Patients with Obesity and Knee Osteoarthritis: A Randomized Control Trial" International Journal of Environmental Research and Public Health 18, no. 15: 7744. https://doi.org/10.3390/ijerph18157744
APA StyleHsu, Y.-I., Chen, Y.-C., Lee, C.-L., & Chang, N.-J. (2021). Effects of Diet Control and Telemedicine-Based Resistance Exercise Intervention on Patients with Obesity and Knee Osteoarthritis: A Randomized Control Trial. International Journal of Environmental Research and Public Health, 18(15), 7744. https://doi.org/10.3390/ijerph18157744







