Projecting the Impact of SARS-CoV-2 Variants and the Vaccination Program on the Fourth Wave of the COVID-19 Pandemic in South Korea
Abstract
:1. Introduction
2. Materials and Methods
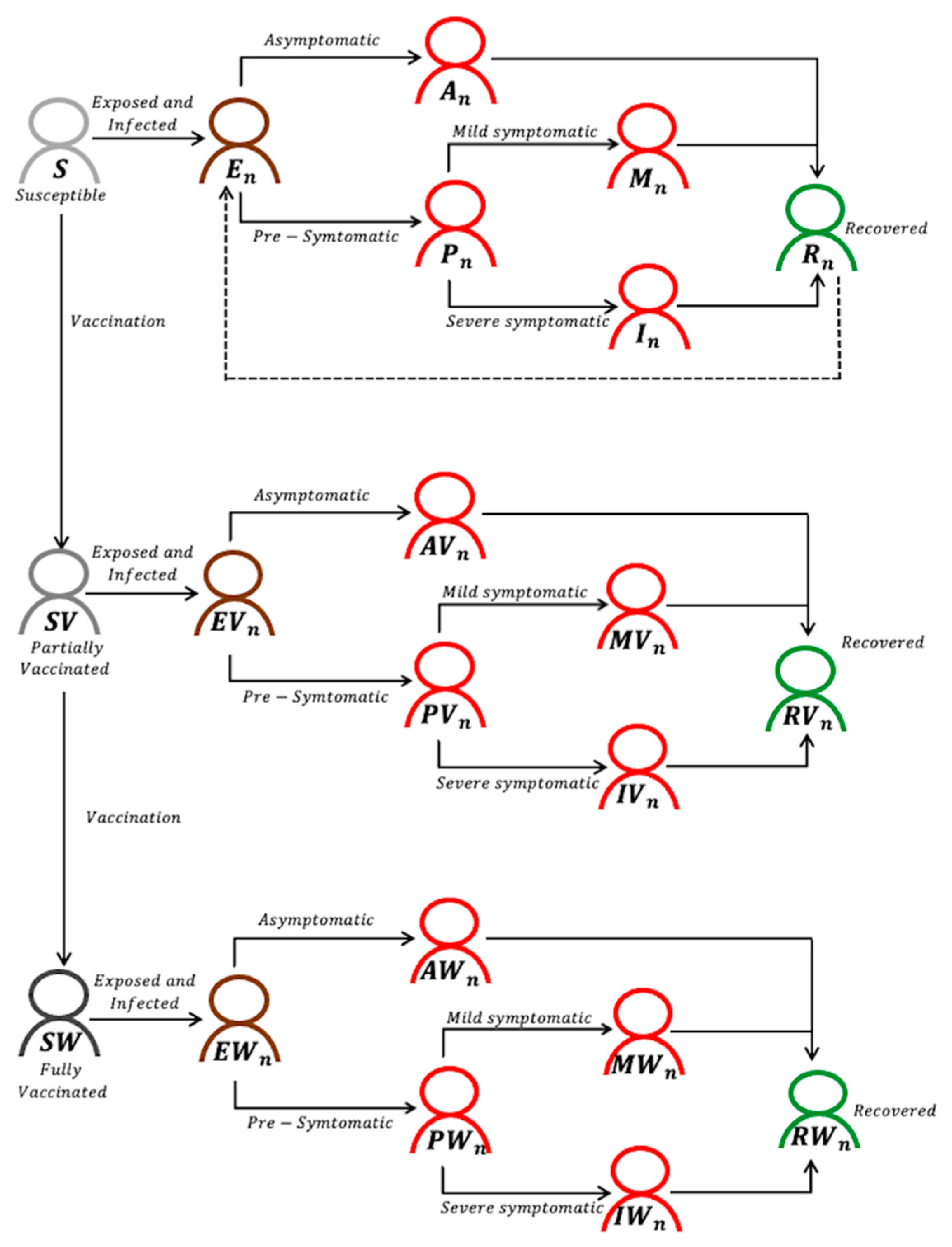
2.1. Model Structure
2.2. Disease Dynamics
2.3. Vaccination
| Notation | Description | Estimates | References |
|---|---|---|---|
| Incubation period (days) | 5.2 days | [18] | |
| Asymptomatic period (days) | 5.0 days | [16,20,21] | |
| Pre-symptomatic period (days) | 2.3 days | [14,16,19] | |
| Infectious period from onset of mild symptoms (days) | 3.2 days | [16,20] | |
| Infectious period from onset of severe symptoms (days) | 3.2 days | [16,20] | |
| Reduction of infection by variant strain due to cross immunity | 0.7 | [32] | |
| Relative transmission potential of strain 2 compared to strain 1 | 2 | [10] | |
| Relative infectivity of asymptomatic individuals compared to the pre-symptomatic stage | 0.26 | [15] | |
| Relative infectivity of mildly symptomatic individuals compared to the pre-symptomatic stage, considering isolation | 0.22 | [14,17,33] | |
| Relative infectivity of severely symptomatic individuals compared to the pre-symptomatic stage, considering isolation | 0.44 | [14,17,33] | |
| Proportion of infections that are symptomatic | 0.29 for k = 1; 0.21 for k = 2; 0.27 for k = 3; 0.33 for k = 4; 0.40 for k = 5; 0.49 for k = 6; 0.63 for k = 7; 0.69 for k = 8 | [13] | |
| Proportion of symptomatic cases that exhibit mild symptoms | 0.90 for k = 1 and 2; 0.85 for k = 3, 4, and 5; 0.60 for k = 6 and 7; 0.20 for k = 8 | [34,35] | |
| Vaccine efficacy, after the first dose to before the second dose, against infection by strain 1 among the age group k | 0.53 for k = 1, 2, …, 6; 0.37 for k = 7 and 8 | [22,23,24,31] | |
| Vaccine efficacy, after the first dose to before the second dose, against infection by strain 2 among the age group k | 0.21 for k = 1, 2, …, 6; 0.14 for k = 7 and 8 | [10,29,36,37] | |
| Vaccine efficacy, after the first dose to before the second dose, against symptomatic disease by strain 1 among the age group k | 0.58 for k = 1, 2, …, 6; 0.41 for k = 7 and 8 | [22,23,24,31] | |
| Vaccine efficacy, after the first dose to before the second dose, against symptomatic disease by strain 2 among the age group k | 0.14 for k = 1, 2, …, 6; 0.10 for k = 7 and 8 | [10,29,36,37] | |
| Vaccine efficacy, after the second dose, against infection by strain 1 among the age group k | 0.64 for k = 1, 2, …, 6; 0.37 for k = 7 and 8 | [38,39] | |
| Vaccine efficacy, after the second dose, against infection by strain 2 among the age group k | 0.48 for k = 1, 2, …, 6; 0.33 for k = 7 and 8 | [8,9] | |
| Vaccine efficacy, after the second dose, against symptomatic disease by strain 1 among the age group k | 0.37 for k = 1, 2, …, 6; 0.26 for k = 7 and 8 | [22,23,24,31] | |
| Vaccine efficacy, after the second dose, against symptomatic disease by strain 2 among the age group k | 0.33 for k = 1, 2, …, 6; 0.23 for k = 7 and 8 | [8,9] |
2.4. Model Implementation
| Partially Vaccinated (%) | Fully Vaccinated (%) | ||
|---|---|---|---|
| Total | 19.5 | 10.4 | |
| Age group | 20–29 | 6.4 | 4.1 |
| 30–39 | 16.4 | 14.4 | |
| 40–49 | 9.0 | 4.7 | |
| 50–59 | 8.6 | 3.6 | |
| 60–69 | 79.9 | 3.3 | |
| 70 and above | 33.6 | 51.1 | |
3. Results
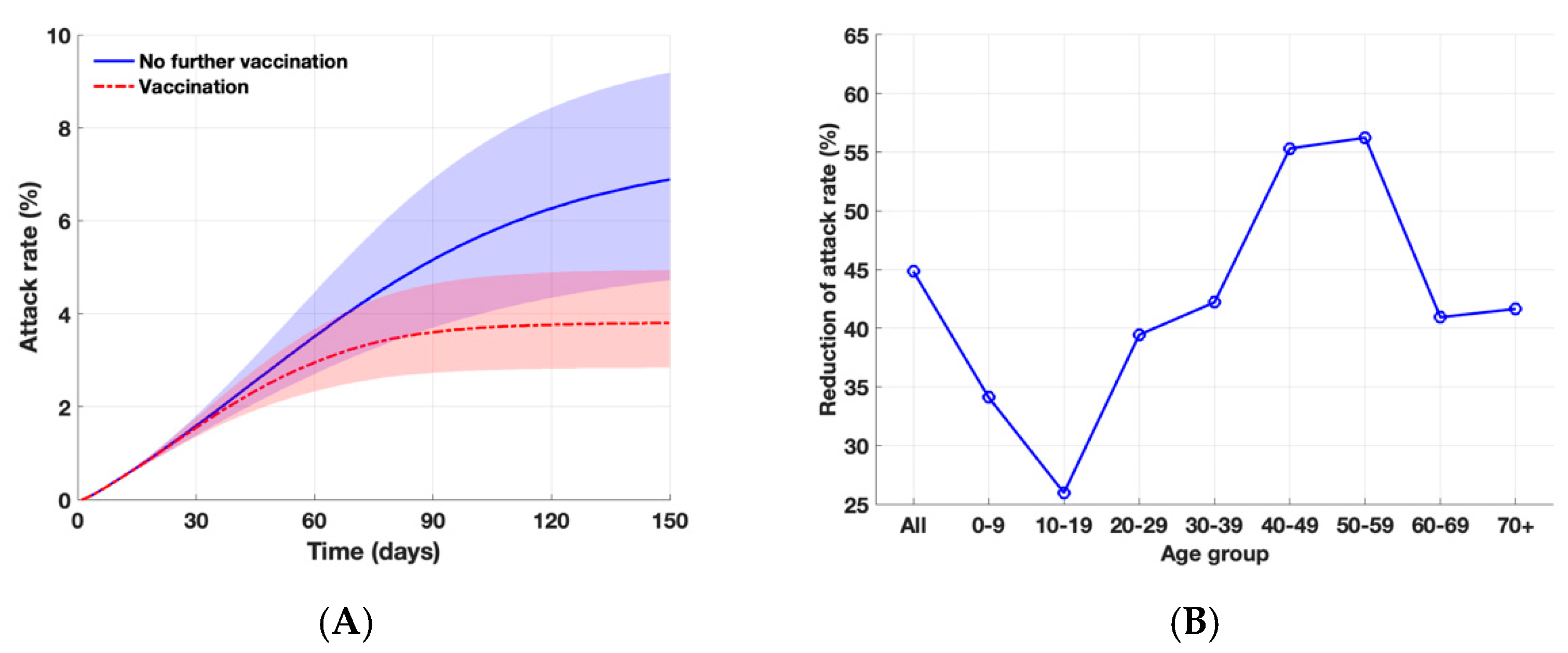
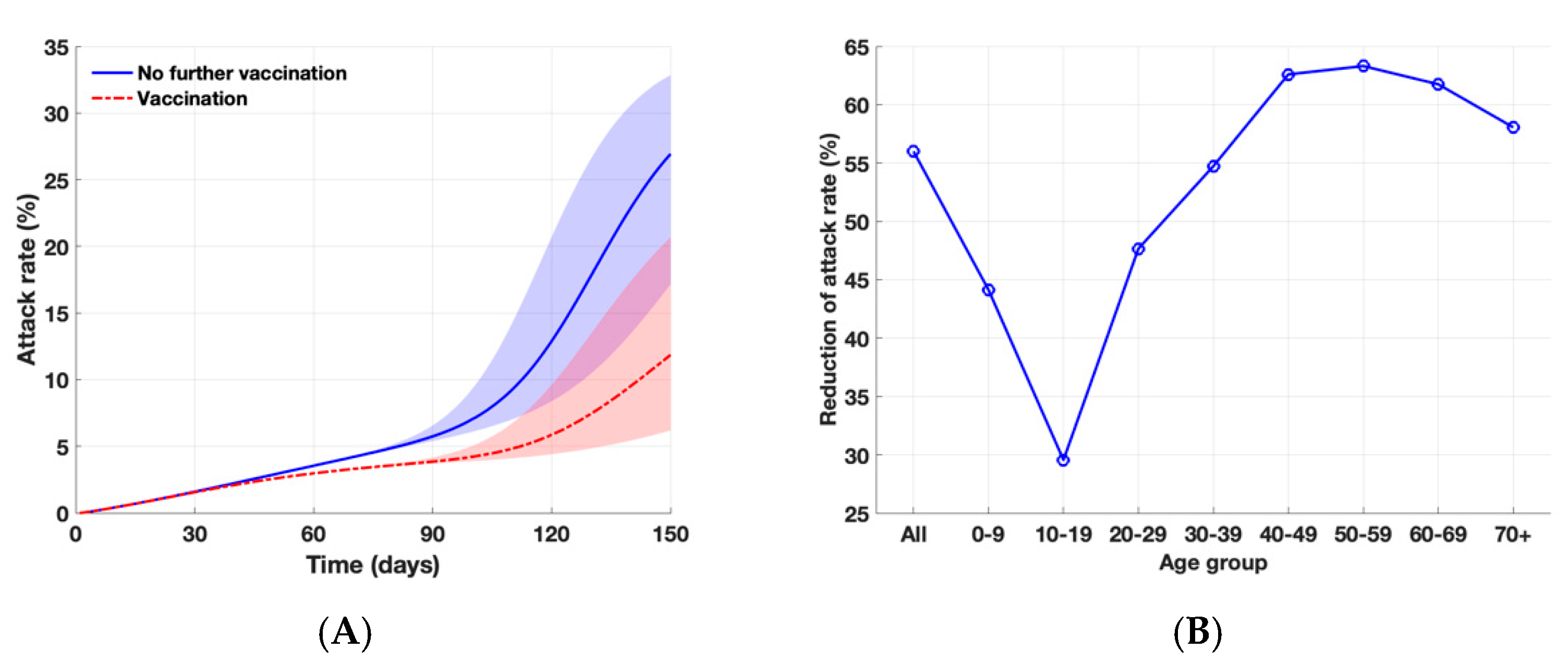
3.1. Attack Rates
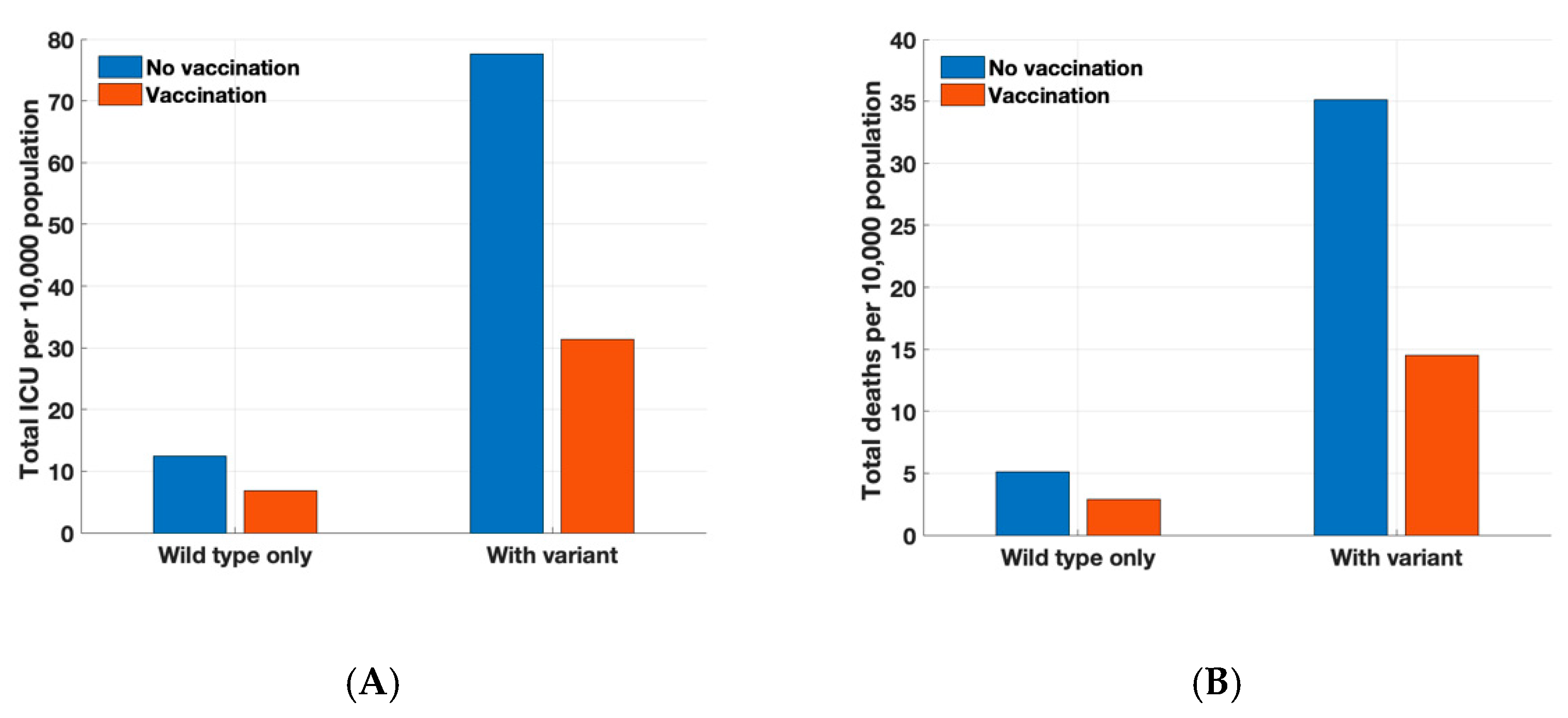
3.2. Hospitalization and Deaths
4. Discussion
5. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- WHO COVID-19 Weekly Epidemiological Update. Available online: https://www.who.int/emergencies/diseases/novel-coronavirus-2019/situation-reports (accessed on 5 July 2021).
- Christie, A.; Mbaeyi, S.A.; Walensky, R.P. CDC Interim Recommendations for Fully Vaccinated People: An Important First Step. JAMA J. Am. Med. Assoc. 2021, 325, 1501–1502. [Google Scholar] [CrossRef]
- Galloway, S.E.; Paul, P.; MacCannell, D.R.; Johansson, M.A.; Brooks, J.T.; MacNeil, A.; Slayton, R.B.; Tong, S.; Silk, B.J.; Armstrong, G.L. Emergence of SARS-CoV-2 B.1.1.7 Lineage—United States, December 29, 2020-January 12, 2021. Mmwr. Morb. Mortal. Wkly. Rep. 2021, 70, 95–99. [Google Scholar] [CrossRef]
- Davies, N.G.; Abbott, S.; Barnard, R.C.; Jarvis, C.I.; Kucharski, A.J.; Munday, J.D.; Pearson, C.A.B.; Russell, T.W.; Tully, D.C.; Washburne, A.D. Estimated transmissibility and impact of SARS-CoV-2 lineage B.1.1.7 in England. Science 2021, 372, eabg3055. [Google Scholar] [CrossRef]
- Challen, R.; Brooks-Pollock, E.; Read, J.M.; Dyson, L.; Tsaneva-Atanasova, K.; Danon, L. Risk of mortality in patients infected with SARS-CoV-2 variant of concern 202012/1: Matched cohort study. BMJ 2021, 372, n579. [Google Scholar] [CrossRef]
- Gómez, C.E.; Perdiguero, B.; Esteban, M. Emerging SARS-CoV-2 Variants and Impact in Global Vaccination Programs against SARS-CoV-2/COVID-19. Vaccines 2021, 9, 243. [Google Scholar] [CrossRef] [PubMed]
- Several Local Gov’ts Waging All-out Battle Against Coronavirus Variants. Available online: https://en.yna.co.kr/view/AEN20210511008900315?section=search (accessed on 11 May 2021).
- Mahase, E. Covid-19: Where are we on vaccines and variants? BMJ 2021, 372, n597. [Google Scholar] [CrossRef] [PubMed]
- Sheikh, A.; McMenamin, J.; Taylor, B.; Robertson, C. SARS-CoV-2 Delta VOC in Scotland: Demographics, risk of hospital admission, and vaccine effectiveness. Lancet 2021, 397, 2461–2462. [Google Scholar] [CrossRef]
- Callaway, E. Delta coronavirus variant: Scientists brace for impact. Nature 2021, 595, 17–18. [Google Scholar] [CrossRef] [PubMed]
- Prem, K.; Cook, A.R.; Jit, M. Projecting social contact matrices in 152 countries using contact surveys and demographic data. PLoS Comput. Biol. 2017, 13, e1005697. [Google Scholar] [CrossRef]
- Mossong, J.; Hens, N.; Jit, M.; Beutels, P.; Auranen, K.; Mikolajczyk, R.; Massari, M.; Salmaso, S.; Tomba, G.S.; Wallinga, J. Social contacts and mixing patterns relevant to the spread of infectious diseases. PLoS Med. 2008, 5, e74. [Google Scholar] [CrossRef] [PubMed]
- Davies, N.G.; Klepac, P.; Liu, Y.; Prem, K.; Jit, M.; Eggo, R.M. Age-dependent effects in the transmission and control of COVID-19 epidemics. Nat. Med. 2020, 26, 1205–1211. [Google Scholar] [CrossRef]
- Moghadas, S.M.; Fitzpatrick, M.C.; Sah, P.; Pandey, A.; Shoukat, A.; Singer, B.H.; Galvani, A.P. The implications of silent transmission for the control of COVID-19 outbreaks. Proc. Natl. Acad. Sci. USA 2020, 117, 17513–17515. [Google Scholar] [CrossRef] [PubMed]
- Sayampanathan, A.A.; Heng, C.S.; Pin, P.H.; Pang, J.; Leong, T.Y.; Lee, V.J. Infectivity of asymptomatic versus symptomatic COVID-19. Lancet 2021, 397, 93–94. [Google Scholar] [CrossRef]
- Moghadas, S.M.; Vilches, T.N.; Zhang, K.; Nourbakhsh, S.; Sah, P.; Fitzpatrick, M.C.; Galvani, A.P. Evaluation of COVID-19 vaccination strategies with a delayed second dose. PLoS Biol. 2021, 19, e3001211. [Google Scholar] [CrossRef] [PubMed]
- Jarvis, C.I.; Van Zandvoort, K.; Gimma, A.; Prem, K.; Klepac, P.; Rubin, G.J.; Edmunds, W.J. Quantifying the impact of physical distance measures on the transmission of COVID-19 in the UK. BMC Med. 2020, 18, 124. [Google Scholar] [CrossRef]
- Li, Q.; Guan, X.; Wu, P.; Wang, X.; Zhou, L.; Tong, Y.; Ren, R.; Leung, K.S.M.; Lau, E.H.Y.; Wong, J.Y. Early Transmission Dynamics in Wuhan, China, of Novel Coronavirus-Infected Pneumonia. N. Engl. J. Med. 2020, 382, 1199–1207. [Google Scholar] [CrossRef]
- He, X.; Lau, E.H.Y.; Wu, P.; Deng, X.; Wang, J.; Hao, X.; Lau, Y.C.; Wong, J.Y.; Guan, Y.; Tan, X. Temporal dynamics in viral shedding and transmissibility of COVID-19. Nat. Med. 2020, 26, 672–675. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, R.; Pei, S.; Chen, B.; Song, Y.; Zhang, T.; Yang, W.; Shaman, J. Substantial undocumented infection facilitates the rapid dissemination of novel coronavirus (COVID-19). Science 2020, 368, 489–493. [Google Scholar] [CrossRef] [Green Version]
- Gatto, M.; Bertuzzo, E.; Mari, L.; Miccoli, S.; Carraro, L.; Casagrandi, R.; Rinaldo, A. Spread and dynamics of the COVID-19 epidemic in Italy: Effects of emergency containment measures. Proc. Natl. Acad. Sci. USA 2020, 117, 10484–10491. [Google Scholar] [CrossRef] [Green Version]
- Baden, L.R.; El Sahly, H.M.; Essink, B.; Kotloff, K.; Frey, S.; Novak, R.; Diemert, D.; Spector, S.A.; Rouphael, N.; Creech, C.B. Efficacy and Safety of the mRNA-1273 SARS-CoV-2 Vaccine. N. Engl. J. Med. 2020, 384, 403–416. [Google Scholar] [CrossRef] [PubMed]
- Polack, F.P.; Thomas, S.J.; Kitchin, N.; Absalon, J.; Gurtman, A.; Lockhart, S.; Perez, J.L.; Perez Marc, G.; Moreira, E.D.; Zerbini, C. Safety and Efficacy of the BNT162b2 mRNA Covid-19 Vaccine. N. Engl. J. Med. 2020, 383, 2603–2615. [Google Scholar] [CrossRef] [PubMed]
- Voysey, M.; Clemens, S.A.C.; Madhi, S.A.; Weckx, L.Y.; Folegatti, P.M.; Aley, P.K.; Angus, B.; Baillie, V.L.; Barnabas, S.L.; Bhorat, Q.E. Safety and efficacy of the ChAdOx1 nCoV-19 vaccine (AZD1222) against SARS-CoV-2: An interim analysis of four randomised controlled trials in Brazil, South Africa, and the UK. Lancet 2021, 397, 99–111. [Google Scholar] [CrossRef]
- KCDC. The Updates of COVID-19 in Republic of Korea. In Centers for Disease Control and Prevention Korea: 2021. Available online: http://www.kdca.go.kr/board/board.es?mid=a20501010000&bid=0015 (accessed on 5 July 2021).
- Kim, A. Korea to vaccinate 12 million against COVID-19 by June: Minister. The Korea Herald. 15 March 2021. [Google Scholar]
- Matrajt, L.; Eaton, J.; Leung, T.; Dimitrov, D.; Schiffer, J.T.; Swan, D.A.; Janes, H. Optimizing vaccine allocation for COVID-19 vaccines shows the potential role of single-dose vaccination. Nat. Commun. 2021, 12, 3449. [Google Scholar] [CrossRef]
- Lumley, S.F.; Rodger, G.; Constantinides, B.; Sanderson, N.; Chau, K.K.; Street, T.L.; O’Donnell, D.; Howarth, A.; Hatch, S.B.; Marsden, B.D.; et al. An observational cohort study on the incidence of SARS-CoV-2 infection and B.1.1.7 variant infection in healthcare workers by antibody and vaccination status. Clin. Infect. Dis. 2021, ciab608. [Google Scholar] [CrossRef] [PubMed]
- Bernal, J.; Andrews, N.; Gower, C.; Stowe, J.; Robertson, C.; Tessier, E.; Simmons, R.; Cottrell, S.; Roberts, R.; O’Doherty, M. Early Effectiveness of COVID-19 Vaccination with BNT162b2 mRNA Vaccine and ChAdOx1 Adenovirus Vector Vaccine on Symptomatic Disease, Hospitalisations and Mortality in Older Adults in England. Available online: https://www.medrxiv.org/content/10.1101/2021.03.01.21252652v1 (accessed on 30 March 2021).
- Iacobucci, G. Covid-19: Infections fell by 65% after first dose of AstraZeneca or Pfizer vaccine, data show. BMJ 2021, 373, n1068. [Google Scholar] [CrossRef]
- Soiza, R.L.; Scicluna, C.; Thomson, E.C. Efficacy and safety of COVID-19 vaccines in older people. Age Ageing 2021, 50, 279–283. [Google Scholar] [CrossRef]
- Kissler, S.M.; Tedijanto, C.; Goldstein, E.; Grad, Y.H.; Lipsitch, M. Projecting the transmission dynamics of SARS-CoV-2 through the postpandemic period. Science 2020, 368, 860–868. [Google Scholar] [CrossRef] [PubMed]
- Ferretti, L.; Wymant, C.; Kendall, M.; Zhao, L.; Nurtay, A.; Abeler-Dorner, L.; Parker, M.; Bonsall, D.; Fraser, C. Quantifying SARS-CoV-2 transmission suggests epidemic control with digital contact tracing. Science 2020, 368, 6491. [Google Scholar] [CrossRef] [Green Version]
- Shoukat, A.; Wells, C.R.; Langley, J.M.; Singer, B.H.; Galvani, A.P.; Moghadas, S.M. Projecting demand for critical care beds during COVID-19 outbreaks in Canada. CMAJ 2020, 192, E489–E496. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moghadas, S.M.; Shoukat, A.; Fitzpatrick, M.C.; Wells, C.R.; Sah, P.; Pandey, A.; Sachs, J.D.; Wang, Z.; Meyers, L.A.; Singer, B.H. Projecting hospital utilization during the COVID-19 outbreaks in the United States. Proc. Natl. Acad. Sci. USA 2020, 117, 9122–9126. [Google Scholar] [CrossRef] [Green Version]
- Dagan, N.; Barda, N.; Kepten, E.; Miron, O.; Perchik, S.; Katz, M.A.; Hernan, M.A.; Lipsitch, M.; Reis, B.; Balicer, R.D. BNT162b2 mRNA Covid-19 Vaccine in a Nationwide Mass Vaccination Setting. N. Engl. J. Med. 2021, 384, 1412–1423. [Google Scholar] [CrossRef]
- Hall, V.J.; Foulkes, S.; Saei, A.; Andrews, N.; Oguti, B.; Charlett, A.; Wellington, E.; Stowe, J.; Gillson, N.; Atti, A.; et al. COVID-19 vaccine coverage in health-care workers in England and effectiveness of BNT162b2 mRNA vaccine against infection (SIREN): A prospective, multicentre, cohort study. Lancet 2021, 397, 1725–1735. [Google Scholar] [CrossRef]
- Pawlowski, C.; Lenehan, P.; Puranik, A.; Agarwal, V.; Venkatakrishnan, A.J.; Niesen, M.J.M.; O’Horo, J.C.; Virk, A.; Swift, M.D.; Badley, A.D. FDA-authorized mRNA COVID-19 vaccines are effective per real-world evidence synthesized across a multi-state health system. Med. (N. Y.) 2021. [Google Scholar] [CrossRef]
- Shrotri, M.; Krutikov, M.; Palmer, T.; Giddings, R.; Azmi, B.; Subbarao, S.; Fuller, C.; Irwin-Singer, A.; Davies, D.; Tut, G. Vaccine effectiveness of the first dose of ChAdOx1 nCoV-19 and BNT162b2 against SARS-CoV-2 infection in residents of long-term care facilities in England (VIVALDI): A prospective cohort study. Lancet Infect. Dis. 2021. [Google Scholar] [CrossRef]
- Moghadas, S.M.; Vilches, T.N.; Zhang, K.; Wells, C.R.; Shoukat, A.; Singer, B.H.; Meyers, L.A.; Neuzil, K.M.; Langley, J.M.; Fitzpatrick, M.C. The impact of vaccination on COVID-19 outbreaks in the United States. Clin. Infect. Dis. Off. Publ. Infect. Dis. Soc. Am. 2021. [Google Scholar] [CrossRef]
- Parpia, A.S.; Martinez, I.; El-Sayed, A.M.; Wells, C.R.; Myers, L.; Duncan, J.; Collins, J.; Fitzpatrick, M.C.; Galvani, A.P.; Pandey, A. Racial disparities in COVID-19 mortality across Michigan, United States. EClinicalMedicine 2021, 33, 100761. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shim, E. Projecting the Impact of SARS-CoV-2 Variants and the Vaccination Program on the Fourth Wave of the COVID-19 Pandemic in South Korea. Int. J. Environ. Res. Public Health 2021, 18, 7578. https://doi.org/10.3390/ijerph18147578
Shim E. Projecting the Impact of SARS-CoV-2 Variants and the Vaccination Program on the Fourth Wave of the COVID-19 Pandemic in South Korea. International Journal of Environmental Research and Public Health. 2021; 18(14):7578. https://doi.org/10.3390/ijerph18147578
Chicago/Turabian StyleShim, Eunha. 2021. "Projecting the Impact of SARS-CoV-2 Variants and the Vaccination Program on the Fourth Wave of the COVID-19 Pandemic in South Korea" International Journal of Environmental Research and Public Health 18, no. 14: 7578. https://doi.org/10.3390/ijerph18147578
APA StyleShim, E. (2021). Projecting the Impact of SARS-CoV-2 Variants and the Vaccination Program on the Fourth Wave of the COVID-19 Pandemic in South Korea. International Journal of Environmental Research and Public Health, 18(14), 7578. https://doi.org/10.3390/ijerph18147578






