Exercise Fat Oxidation Is Positively Associated with Body Fatness in Men with Obesity: Defying the Metabolic Flexibility Paradigm
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
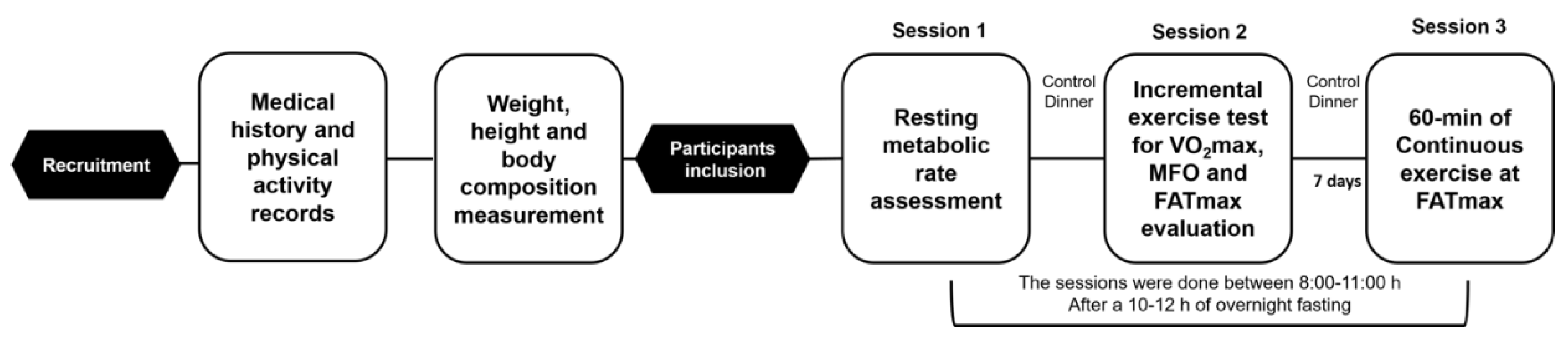
2.2. Study Design
2.3. Anthropometric Measurements
2.4. Metabolic Oxidative Measurements
- -
- Fat oxidation:
- -
- Carbohydrate oxidation:
2.4.1. Resting Metabolic Rate Procedure
2.4.2. Incremental Exercise Test Procedure
2.5. 60 min Treadmill Walking at FATMax
2.6. Blood Lactate Assay
2.7. Statistical Analysis
3. Results
3.1. Participants Physical Fitness and FATMax
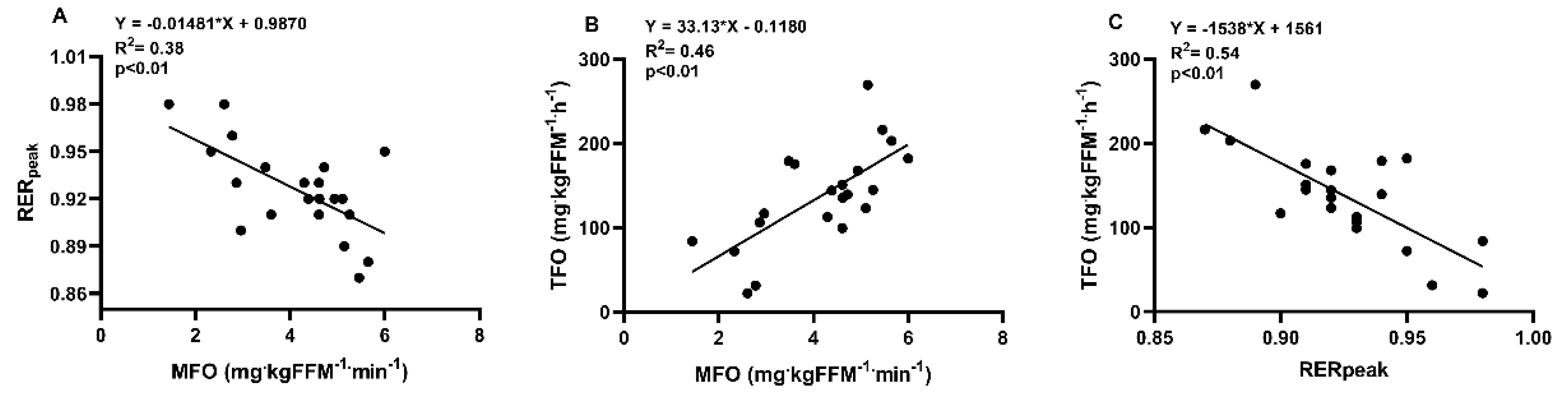
3.2. Metflex Markers
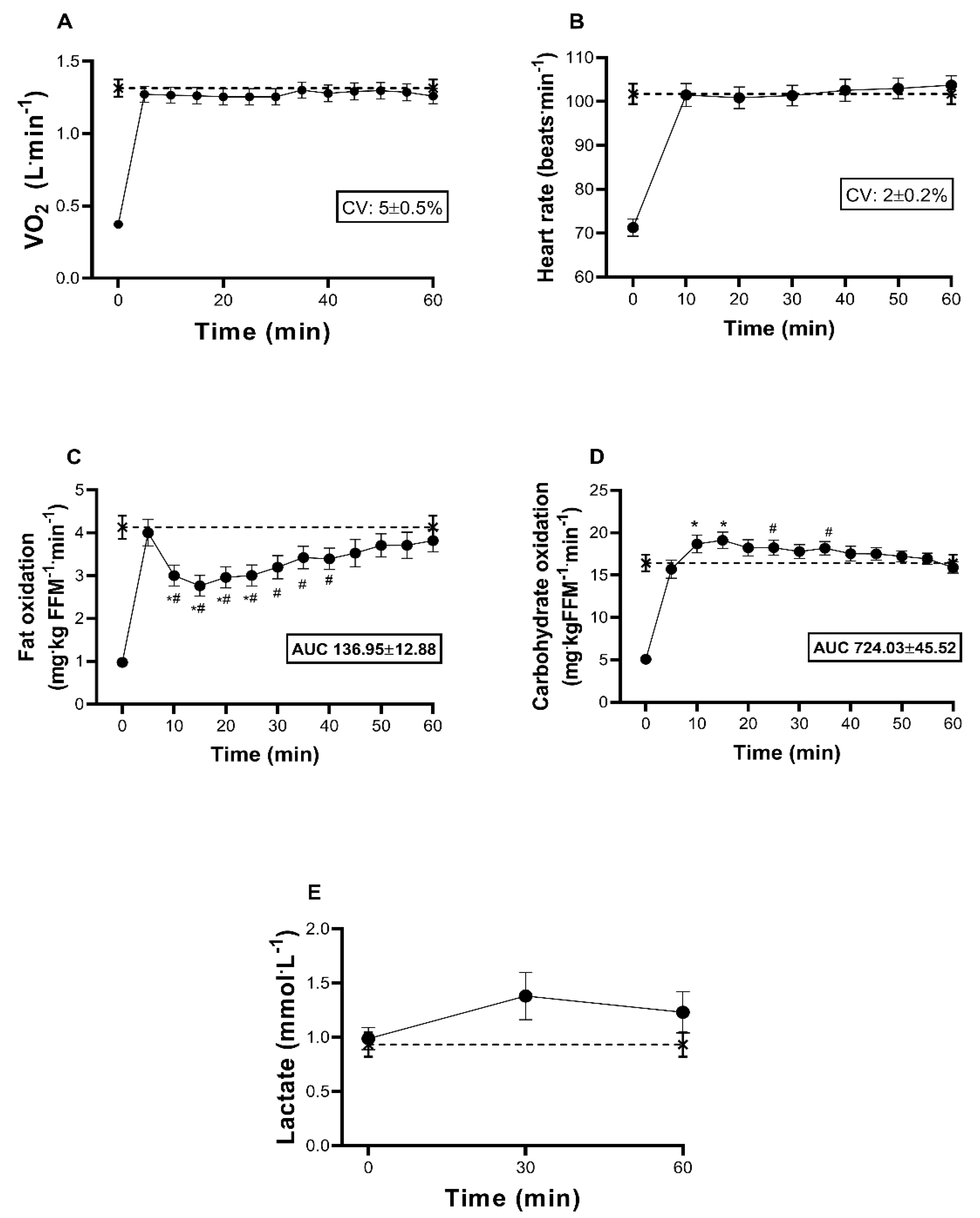
3.3. Substrate Oxidation at FATmax (Test 2)
4. Discussion
4.1. Correlation of Metflex Markers
4.2. Obesity and Metflex
4.3. Kinetics of Macronutrient Oxidation during FATmax
4.4. Study Strengths
4.5. Study Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Abdelaal, M.; Le Roux, C.W.; Docherty, N. Morbidity and mortality associated with obesity. Ann. Transl. Med. 2017, 5, 161. [Google Scholar] [CrossRef]
- Goodpaster, B.H.; Sparks, L.M. Metabolic Flexibility in Health and Disease. Cell Metab. 2017, 25, 1027–1036. [Google Scholar] [CrossRef]
- Houmard, J.A. Intramuscular lipid oxidation and obesity. Am. J. Physiol. Integr. Comp. Physiol. 2008, 294, R1111–R1116. [Google Scholar] [CrossRef] [PubMed]
- Galgani, J.E.; Fernández-Verdejo, R. Pathophysiological role of metabolic flexibility on metabolic health. Obes. Rev. 2021, 22, e13131. [Google Scholar] [CrossRef] [PubMed]
- Park, S.S.; Seo, Y.-K. Excess Accumulation of Lipid Impairs Insulin Sensitivity in Skeletal Muscle. Int. J. Mol. Sci. 2020, 21, 1949. [Google Scholar] [CrossRef] [PubMed]
- Arad, A.D.; Basile, A.J.; Albu, J.; DiMenna, F.J. No Influence of Overweight/Obesity on Exercise Lipid Oxidation: A Systematic Review. Int. J. Mol. Sci. 2020, 21, 1614. [Google Scholar] [CrossRef]
- Amaro-Gahete, F.J.; Sanchez-Delgado, G.; Jurado-Fasoli, L.; De-La-O, A.; Castillo, M.J.; Helge, J.; Ruiz, J.R. Assessment of maximal fat oxidation during exercise: A systematic review. Scand. J. Med. Sci. Sports 2019, 29, 910–921. [Google Scholar] [CrossRef]
- San-Millán, I.; Brooks, G.A. Assessment of Metabolic Flexibility by Means of Measuring Blood Lactate, Fat, and Carbohydrate Oxidation Responses to Exercise in Professional Endurance Athletes and Less-Fit Individuals. Sports Med. 2018, 48, 467–479. [Google Scholar] [CrossRef]
- Takagi, S.; Sakamoto, S.; Midorikawa, T.; Konishi, M.; Katsumura, T. Determination of the exercise intensity that elicits maximal fat oxidation in short-time testing. J. Sports Sci. 2014, 32, 175–182. [Google Scholar] [CrossRef]
- Özdemir, Ç.; Özgünen, K.; Günaştı, Ö.; Eryılmaz, S.K.; Kılcı, A.; Kurdak, S.S. Changes in substrate utilization rates during 40 min of walking within the Fatmax range. Physiol. Int. 2019, 106, 294–304. [Google Scholar] [CrossRef]
- Hargreaves, M.; Spriet, L.L. Skeletal muscle energy metabolism during exercise. Nat. Metab. 2020, 2, 817–828. [Google Scholar] [CrossRef]
- Maunder, E.; Plews, D.J.; Kilding, A.E. Contextualising Maximal Fat Oxidation during Exercise: Determinants and Normative Values. Front. Physiol. 2018, 9, 599. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Verdejo, R.; Bajpeyi, S.; Ravussin, E.; Galgani, J.E. Metabolic flexibility to lipid availability during exercise is enhanced in individuals with high insulin sensitivity. Am. J. Physiol. Endocrinol. Metab. 2018, 315, E715–E722. [Google Scholar] [CrossRef] [PubMed]
- Flores, P.H.; Rodriguez, J.; Yang, J.; Cotter, J.A.; Harris, M.B. Using Dynamic Metabolic Adjustments during Exercise to Quantify Metabolic Flexibility. FASEB J. 2020, 34, 1. [Google Scholar] [CrossRef]
- Blaize, A.N.; Potteiger, J.A.; Claytor, R.P.; Noe, D.A. Body Fat has No Effect on the Maximal Fat Oxidation Rate in Young, Normal, and Overweight Women. J. Strength Cond. Res. 2014, 28, 2121–2126. [Google Scholar] [CrossRef] [PubMed]
- Robinson, S.L.; Chambers, E.S.; Fletcher, G.; Wallis, G.A. Lipolytic Markers, Insulin and Resting Fat Oxidation are Associated with Maximal Fat Oxidation. Int. J. Sports Med. 2016, 37, 607–613. [Google Scholar] [CrossRef]
- Croci, I.; Hickman, I.; Wood, R.; Borrani, F.; Macdonald, G.A.; Byrne, N. Fat oxidation over a range of exercise intensities: Fitness versus fatness. Appl. Physiol. Nutr. Metab. 2014, 39, 1352–1359. [Google Scholar] [CrossRef]
- LaMonte, M.J.; Barlow, C.E.; Jurca, R.; Kampert, J.B.; Church, T.S.; Blair, S.N. Cardiorespiratory Fitness Is Inversely Associated With the Incidence of Metabolic Syndrome: A prospective study of men and women. Circulation 2005, 112, 505–512. [Google Scholar] [CrossRef]
- Siri, W.E. Body composition from fluid spaces and density: Analysis of methods. 1961. Nutrition 1993, 9, 480–492. [Google Scholar]
- Weir, J.B.D.V. New methods for calculating metabolic rate with special reference to protein metabolism. J. Physiol. 1949, 109, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Frayn, K.N. Calculation of substrate oxidation rates in vivo from gaseous exchange. J. Appl. Physiol. Respir. Environ. Exerc. Physiol. 1983, 55, 628–634. [Google Scholar] [CrossRef]
- Dumke, C.L. Health-related physical fitness and testing prescription. In ACSM’s Guidelines for Exercise Testing and Prescription, 10th ed.; Diebe, D., Ehrman, J.K., Liguori, G., Magal, M., Eds.; Wolters Kluwer: Beijing, China, 2018; Chapter 4. [Google Scholar]
- Borg, G.A. Psychophysical bases of perceived exertion. Med. Sci. Sports Exerc. 1982, 14, 377–381. [Google Scholar] [CrossRef] [PubMed]
- Ramírez, J.C.; Sanchez, J.S.; Foncea, H.Z.; Gonzalez, M.M.; Dinamarca, B.L.; Rojas, L.G. Cardiorespiratory fitness and fat oxidation during exercise as protective factors for insulin resistance in sedentary women with overweight or obesity. Nutr. Hosp. 2018, 35, 312–317. [Google Scholar] [CrossRef]
- Emerenziani, G.P.; Ferrari, D.; Marocco, C.; Greco, E.A.; Migliaccio, S.; Lenzi, A.; Baldari, C.; Guidetti, L. Relationship between individual ventilatory threshold and maximal fat oxidation (MFO) over different obesity classes in women. PLoS ONE 2019, 14, e0215307. [Google Scholar] [CrossRef] [PubMed]
- Ara, I.; Larsen, S.; Stallknecht, B.M.; Guerra, B.; Morales-Alamo, D.; Andersen, J.L.; Ponce-González, J.G.; Guadalupe-Grau, A.; Galbo, H.; Calbet, J.A.; et al. Normal mitochondrial function and increased fat oxidation capacity in leg and arm muscles in obese humans. Int. J. Obes. 2011, 35, 99–108. [Google Scholar] [CrossRef]
- Goodpaster, B.H.; Wolfe, R.R.; Kelley, D.E. Effects of Obesity on Substrate Utilization during Exercise. Obes. Res. 2002, 10, 575–584. [Google Scholar] [CrossRef]
- Horowitz, J.F.; Klein, S. Oxidation of nonplasma fatty acids during exercise is increased in women with abdominal obesity. J. Appl. Physiol. 2000, 89, 2276–2282. [Google Scholar] [CrossRef]
- Isacco, L.; Duche, P.; Thivel, D.; Meddahi-Pelle, A.; Lemoine-Morel, S.; Duclos, M.; Boisseau, N. Fat Mass Localization Alters Fuel Oxidation during Exercise in Normal Weight Women. Med. Sci. Sports Exerc. 2013, 45, 1887–1896. [Google Scholar] [CrossRef]
- Rosenkilde, M.; Nordby, P.; Nielsen, L.B.; Stallknecht, B.M.; Helge, J. Fat oxidation at rest predicts peak fat oxidation during exercise and metabolic phenotype in overweight men. Int. J. Obes. 2010, 34, 871–877. [Google Scholar] [CrossRef]
- Schutz, Y.; Tremblay, A.; Weinsier, R.L.; Nelson, K.M. Role of fat oxidation in the long-term stabilization of body weight in obese women. Am. J. Clin. Nutr. 1992, 55, 670–674. [Google Scholar] [CrossRef]
- Peterson, C.M.; Zhang, B.; Johannsen, D.L.; Ravussin, E. Eight weeks of overfeeding alters substrate partitioning without affecting metabolic flexibility in men. Int. J. Obes. 2017, 41, 887–893. [Google Scholar] [CrossRef][Green Version]
- Ludwig, D.S.; Ebbeling, C.B. The Carbohydrate-Insulin Model of Obesity: Beyond “Calories In, Calories Out”. JAMA Intern. Med. 2018, 178, 1098–1103. [Google Scholar] [CrossRef]
- Muoio, D.M. Metabolic Inflexibility: When Mitochondrial Indecision Leads to Metabolic Gridlock. Cell 2014, 159, 1253–1262. [Google Scholar] [CrossRef] [PubMed]
- Samuel, V.T.; Shulman, G.I. Mechanisms for Insulin Resistance: Common Threads and Missing Links. Cell 2012, 148, 852–871. [Google Scholar] [CrossRef] [PubMed]
- Spriet, L.L. New Insights into the Interaction of Carbohydrate and Fat Metabolism During Exercise. Sports Med. 2014, 44 (Suppl. 1), S87–S96. [Google Scholar] [CrossRef]
- Sahlin, K. Control of lipid oxidation at the mitochondrial level. Appl. Physiol. Nutr. Metab. 2009, 34, 382–388. [Google Scholar] [CrossRef] [PubMed]
- Yu, S.S.; Lefkowitz, R.J.; Hausdorff, W.P. Beta-adrenergic receptor sequestration. A potential mechanism of receptor resen-sitization. J. Biol. Chem. 1993, 268, 337–341. [Google Scholar] [CrossRef]
- Holloway, G.P.; Bezaire, V.; Heigenhauser, G.J.F.; Tandon, N.N.; Glatz, J.F.C.; Luiken, J.J.F.P.; Bonen, A.; Spriet, L.L. Mitochondrial long chain fatty acid oxidation, fatty acid translocase/CD36 content and carnitine palmitoyltransferase I activity in human skeletal muscle during aerobic exercise. J. Physiol. 2006, 571 Pt 1, 201–210. [Google Scholar] [CrossRef]
- Adolfsson, P.; Nilsson, S.; Albertsson-Wikland, K.; Lindblad, B. Hormonal response during physical exercise of different in-tensities in adolescents with type 1 diabetes and healthy controls. Pediatr. Diabetes 2012, 13, 587–596. [Google Scholar] [CrossRef] [PubMed]
- Watt, M.J.; Heigenhauser, G.J.F.; Dyck, D.J.; Spriet, L.L. Intramuscular triacylglycerol, glycogen and acetyl group metabolism during 4 h of moderate exercise in man. J. Physiol. 2002, 541 Pt 3, 969–978. [Google Scholar] [CrossRef]
- Jayewardene, A.F.; Gwinn, T.; Hancock, D.P.; Mavros, Y.; Rooney, K.B. The associations between polymorphisms in the CD36 gene, fat oxidation and cardiovascular disease risk factors in a young adult Australian population: A pilot study. Obes. Res. Clin. Pract. 2014, 8, e618–e621. [Google Scholar] [CrossRef]
- De Jesus, Í.C.; Alle, L.F.; Munhoz, E.C.; Da Silva, L.R.; Lopes, W.A.; Tureck, L.V.; Purim, K.S.M.; Titski, A.C.K.; Leite, N. Trp64Arg polymorphism of the ADRB3 gene associated with maximal fat oxidation and LDL-C levels in non-obese adolescents. J. Pediatr. 2018, 94, 425–431. [Google Scholar] [CrossRef] [PubMed]
- Montes-De-Oca-García, A.; Perez-Bey, A.; Velázquez-Díaz, D.; Corral-Pérez, J.; Opazo-Díaz, E.; Rebollo-Ramos, M.; Gómez-Gallego, F.; Cuenca-García, M.; Casals, C.; Ponce-González, J. Influence of ACE Gene I/D Polymorphism on Cardiometabolic Risk, Maximal Fat Oxidation, Cardiorespiratory Fitness, Diet and Physical Activity in Young Adults. Int. J. Environ. Res. Public Health 2021, 18, 3443. [Google Scholar] [CrossRef]
- Chrzanowski-Smith, O.J.; Edinburgh, R.M.; Smith, E.; Thomas, M.P.; Walhin, J.; Koumanov, F.; Williams, S.; Betts, J.A.; Gonzalez, J.T. Resting skeletal muscle PNPLA2 (ATGL) and CPT1B are associated with peak fat oxidation rates in men and women but do not explain observed sex differences. Exp. Physiol. 2021, 106, 1208–1223. [Google Scholar] [CrossRef]
- Ventura-Clapier, R.; Piquereau, J.; Veksler, V.; Garnier, A. Estrogens, Estrogen Receptors Effects on Cardiac and Skeletal Muscle Mitochondria. Front. Endocrinol. 2019, 10, 557. [Google Scholar] [CrossRef] [PubMed]
- Riddell, M.C.; Jamnik, V.K.; Iscoe, K.E.; Timmons, B.W.; Gledhill, N. Fat oxidation rate and the exercise intensity that elicits maximal fat oxidation decreases with pubertal status in young male subjects. J. Appl. Physiol. 2008, 105, 742–748. [Google Scholar] [CrossRef] [PubMed]
- Achten, J.; Venables, M.; Jeukendrup, A.E. Fat oxidation rates are higher during running compared with cycling over a wide range of intensities. Metabolism 2003, 52, 747–752. [Google Scholar] [CrossRef]
- Egan, B.; Ashley, D.T.; Kennedy, E.; O’Connor, P.L.; O’Gorman, D.J. Higher rate of fat oxidation during rowing compared with cycling ergometer exercise across a range of exercise intensities. Scand. J. Med. Sci. Sports 2016, 26, 630–637. [Google Scholar] [CrossRef]
- Davies, C.T.M.; Few, J.; Foster, K.G.; Sargeant, A.J. Plasma catecholamine concentration during dynamic exercise involving different muscle groups. Eur. J. Appl. Physiol. Occup. Physiol. 1974, 32, 195–206. [Google Scholar] [CrossRef]

| Variable | Mean (95% CI) |
|---|---|
| Age (years) | 27 (24−30) |
| Height (m) | 1.75 (1.72−1.79) |
| Body mass (kg) | 100.3 (93.8−106.7) |
| Body mass index (BMI; kg∙m−2) | 32.6 (30.6−35.2) |
| Fat mass (FM; %) | 35.4 (32.0−39.5) |
| Fat mass index (FMI; kg∙m−2) | 11.8 (10.1−13.9) |
| Fat-free mass (FFM; %) | 64.6 (60.5−67.5) |
| Peak oxygen uptake (VO2peak; mL∙kg−1∙min−1) | 39.9 (36.1−41.7) |
| Maximum heart rate (HRmax; beats∙min−1) | 183 (175−187) |
| Maximum blood lactate (LAb; mmol∙L−1) a | 6.0 (5.0−7.0) |
| Maximal fat oxidation (MFO; mg∙kgFFM−1∙min−1) | 4.1 (3.6−4.7) |
| VO2 (L∙min−1) * | 1.3 (1.2−1.4) |
| % VO2peak (mL∙kg−1∙min−1) * | 35.9 (33.1−39.2) |
| HR (beats∙min−1) * | 102 (96−107) |
| LAb (mM) * | 0.9 (0.7−1.2) |
| Self-perceived exertion (1 to 10) * | 1 (0−2) |
| Energy expenditure (EE; kcal∙min−1) * | 6.6 (5.8−7.1) |
| Body Mass Index (kg∙m−2) | Fat Mass (g∙100 g−1) | |
|---|---|---|
| MFO (mg∙kgFFM−1∙min−1) | 0.49 (0.07, 0.76) * | 0.64 (0.29, 0.84) ** |
| FATmax (%VO2peak) | 0.01 (−0.42, 0.44) | 0.09 (−0.36, 0.50) |
| TFO (mg∙kgFFM∙min−1) | 0.38 (−0.06, 0.70) | 0.63 (0.27, 0.83) ** |
| RERpeak (VCO2∙VO2−1) | −0.48 (−0.76, −0.06) * | −0.67 (−0.85, −0.33) ** |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chávez-Guevara, I.A.; Hernández-Torres, R.P.; Trejo-Trejo, M.; González-Rodríguez, E.; Moreno-Brito, V.; Wall-Medrano, A.; Pérez-León, J.A.; Ramos-Jiménez, A. Exercise Fat Oxidation Is Positively Associated with Body Fatness in Men with Obesity: Defying the Metabolic Flexibility Paradigm. Int. J. Environ. Res. Public Health 2021, 18, 6945. https://doi.org/10.3390/ijerph18136945
Chávez-Guevara IA, Hernández-Torres RP, Trejo-Trejo M, González-Rodríguez E, Moreno-Brito V, Wall-Medrano A, Pérez-León JA, Ramos-Jiménez A. Exercise Fat Oxidation Is Positively Associated with Body Fatness in Men with Obesity: Defying the Metabolic Flexibility Paradigm. International Journal of Environmental Research and Public Health. 2021; 18(13):6945. https://doi.org/10.3390/ijerph18136945
Chicago/Turabian StyleChávez-Guevara, Isaac A., Rosa P. Hernández-Torres, Marina Trejo-Trejo, Everardo González-Rodríguez, Verónica Moreno-Brito, Abraham Wall-Medrano, Jorge A. Pérez-León, and Arnulfo Ramos-Jiménez. 2021. "Exercise Fat Oxidation Is Positively Associated with Body Fatness in Men with Obesity: Defying the Metabolic Flexibility Paradigm" International Journal of Environmental Research and Public Health 18, no. 13: 6945. https://doi.org/10.3390/ijerph18136945
APA StyleChávez-Guevara, I. A., Hernández-Torres, R. P., Trejo-Trejo, M., González-Rodríguez, E., Moreno-Brito, V., Wall-Medrano, A., Pérez-León, J. A., & Ramos-Jiménez, A. (2021). Exercise Fat Oxidation Is Positively Associated with Body Fatness in Men with Obesity: Defying the Metabolic Flexibility Paradigm. International Journal of Environmental Research and Public Health, 18(13), 6945. https://doi.org/10.3390/ijerph18136945








