Medical Cost of Cancer Care for Privately Insured Children in Chile
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Data
2.3. Sample Selection
2.4. Statistical Analysis
2.5. Patient Involvement
3. Results
3.1. General Characteristics of the Sample
3.2. Temporal Trends in Pediatric Healthcare Costs
3.3. Temporal Trends in Total Costs and Out-of-Pocket Payments by Type of Healthcare Service
3.4. Cost of Care by Type of Cancer
3.5. Regression Analysis of Childhood Cancer Medical Costs
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A. Statistical Analysis: Choice of Link Function and Distribution Family for GLMs
| Outcome | Link | Distribution | ll | df | AIC | BIC |
|---|---|---|---|---|---|---|
| Total costs: All | Log | Gamma | −41,081.8 | 30 | 82,223.6 | 82,409.6 |
| Sqrt | Gamma | −41,051.4 | 30 | 82,162.9 | 82,348.9 | |
| Log | Gaussian | −44,195.5 | 30 | 88,451.0 | 88,637.1 | |
| Sqrt | Gaussian | −44,195.3 | 30 | 88,450.7 | 88,636.7 | |
| Log | Poisson | −77,787,754.6 | 30 | 155,575,569.2 | 155,575,755.2 | |
| Sqrt | Poisson | −77,416,321.5 | 30 | 154,832,702.9 | 154,832,889.0 | |
| Total costs: Inpatient | Log | Gamma | −40,774.8 | 30 | 81,609.6 | 81,795.6 |
| Sqrt | Gamma | −40,741.3 | 30 | 81,542.6 | 81,728.7 | |
| Log | Gaussian | −44,122.9 | 30 | 88,305.7 | 88,491.8 | |
| Sqrt | Gaussian | −44,122.3 | 30 | 88,304.5 | 88,490.6 | |
| Log | Poisson | −79,744,444.3 | 30 | 159,488,948.7 | 159,489,134.7 | |
| Sqrt | Poisson | −79,364,979.6 | 30 | 158,730,019.2 | 158,730,205.3 | |
| Total costs: Outpatient | Log | Gamma | −31,644.4 | 30 | 63,348.7 | 63,534.8 |
| Sqrt | Gamma | −31,662.2 | 30 | 63,384.4 | 63,570.4 | |
| Log | Gaussian | −36,406.8 | 30 | 72,873.5 | 73,059.6 | |
| Sqrt | Gaussian | −36,432.5 (a) | 30 | 72,924.9 | 73,110.9 | |
| Log | Poisson | −6,813,615.4 | 30 | 13,627,290.9 | 13,627,476.9 | |
| Sqrt | Poisson | −6,911,786.6 | 30 | 13,823,633.1 | 13,823,819.1 | |
| OOP: All | Log | Gamma | −35,463.9 | 30 | 70,987.8 | 71,173.9 |
| Sqrt | Gamma | −35,502.3 | 30 | 71,064.5 | 71,250.5 | |
| Log | Gaussian | −40,713.7 | 30 | 81,487.3 | 81,673.4 | |
| Sqrt | Gaussian | −40,733.5 (a) | 30 | 81,527.0 | 81,713.0 | |
| Log | Poisson | −27,127,928.1 | 30 | 54,255,916.3 | 54,256,102.3 | |
| Sqrt | Poisson | −27,610,826.7 | 30 | 55,221,713.5 | 55,221,899.5 | |
| OOP: Inpatient | Log | Gamma | −34,952.4 | 30 | 69,964.8 | 70,150.8 |
| Sqrt | Gamma | −34,997.1 | 30 | 70,054.3 | 70,240.3 | |
| Log | Gaussian | −40,602.2 | 30 | 81,264.5 | 81,450.5 | |
| Sqrt | Gaussian | −40,624.4 (a) | 30 | 81,308.7 | 81,494.8 | |
| Log | Poisson | −27,718,476.6 | 30 | 55,437,013.1 | 55,437,199.2 | |
| Sqrt | Poisson | −28,244,260.8 | 30 | 56,488,581.6 | 56,488,767.6 | |
| OOP: Outpatient | Log | Gamma | −27,854.4 | 30 | 55,768.9 | 55,954.9 |
| Sqrt | Gamma | −27,864.3 | 30 | 55,788.7 | 55,974.7 | |
| Log | Gaussian | −32,617.9 (a) | 27 | 65,289.8 | 65,457.2 | |
| Sqrt | Gaussian | −33,958.5 | 30 | 67,977.0 | 68,163.1 | |
| Log | Poisson | −2,588,486.9 | 30 | 5,177,033.7 | 5,177,219.8 | |
| Sqrt | Poisson | −2,608,270.7 | 30 | 5,216,601.4 | 5,216,787.4 |
| Total Costs: All | |||
|---|---|---|---|
| Coef. | Std. Err. | p-value | |
| Lambda | −0.0388 | 0.0113 | <0.001 |
| Test H0: | Restricted log likelihood | LR test | p-value |
| lambda = −1 | −44,263.6 | 7433.5 | <0.001 |
| lambda = 0 | −40,552.8 | 11.7 | <0.001 |
| lambda = 1 | −44,194.9 | 7296.0 | <0.001 |
| Total Costs: Inpatient | |||
| Coef. | Std. Err. | p-value | |
| Lambda | 0.0432 | 0.0081 | <0.001 |
| Test H0: | Restricted log likelihood | LR test | p-value |
| lambda = −1 | −74,354.3 | 68,466.5 | <0.001 |
| lambda = 0 | −40,136.4 | 30.7 | <0.001 |
| lambda = 1 | −44,122.0 | 8001.8 | <0.001 |
| Total Costs | ||
|---|---|---|
| (1) | (2) | |
| Variables | All | Inpatient |
| Patient | ||
| Age 5–9 | −13,975.35 *** | −13,881.38 *** |
| (2425.941) | (2401.829) | |
| Age 10–14 | −11,471.39 *** | −11,991.65 *** |
| (2718.547) | (2659.666) | |
| Age 15–17 | −10,414.07 *** | −10,942.2 *** |
| (2871.567) | (2838.498) | |
| Male | 1188.13 | 1190.84 |
| (1654.738) | (1635.509) | |
| Leukemia | 28,606.03 *** | 27,148 *** |
| (2008.918) | (1990.424) | |
| Brain or nervous system | 18,961.18 *** | 17,167.58 *** |
| (2601.064) | (2530.057) | |
| Non-Hodgkin’s lymphoma | 16,238.01 *** | 15,388.69 *** |
| (3819.330) | (3733.133) | |
| Bone or articular cartilage | 17,477.19 *** | 16,587.2 *** |
| (3926.520) | (3831.486) | |
| Mesothelioma soft tissues | 8964.38 ** | 7784.35 ** |
| (3795.117) | (3569.297) | |
| GES payment | 9979.42 *** | 9396.87 *** |
| (1592.85) | (1567.691) | |
| Policyholder | ||
| Residence in Metropolitan region | 2150.75 | 2685.68 |
| (1688.46) | (1659.136) | |
| Q1 of annual income | 1022.63 | 2120.82 |
| (3096.98) | (3065.648) | |
| Q2 of annual income | −5004.19 * | −4119.12 |
| (2756.91) | (2706.885) | |
| Q3 of annual income | −3744.42 | −3308.53 |
| (2868.55) | (2826.159) | |
| Q4 of annual income | −2263.28 | −2242.98 |
| (2953.77) | (2892.623) | |
| Policyholder’s demographic controls | Yes | Yes |
| Year dummies | Yes | Yes |
| Observations | 3644 | 3644 |
| Mean of outcome | 32,124 | 29,737 |
| Outcome | Coef. | Robust Std. Err. | p-Value |
|---|---|---|---|
| Total costs: All | 1.5978 | 0.0605 | <0.001 |
| Total costs: Inpatient | 1.6209 | 0.0582 | <0.001 |
| Total costs: Outpatient | 2.5209 | 0.0828 | <0.001 |
| OOP: All | 2.2421 | 0.0675 | <0.001 |
| OOP: Inpatient | 2.1344 | 0.0626 | <0.001 |
| OOP: Outpatient | 2.2288 | 0.0770 | <0.001 |
References
- Departamento de Epidemiologia Ministerio de Salud. Primer Informe del Registro Nacional de Cáncer Infantil de Chile (Menores de 15 Años), RENCI; Departamento de Epidemiologia Ministerio de Salud: Santiago, Chile, 2018.
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef]
- Merrill, C.T.; Nagamine, M.; Hambrick, M.M. Pediatric Hospital Stays for Cancer, 2005. In Healthcare Cost and Utilization Project (HCUP) Statistical Briefs [Internet]; Agency for Healthcare Research and Quality: Rockville, MD, USA, 2007; Volume 37. Available online: http://www.hcup-us.ahrq.gov/reports/statbriefs/sb37.pdf (accessed on 20 January 2021).
- Atun, R.; Bhakta, N.; Denburg, A.; Frazier, A.L.; Friedrich, P.; Gupta, S.; Lam, C.G.; Ward, Z.J.; Yeh, J.M.; Allemani, C.; et al. Sustainable care for children with cancer: A Lancet Oncology Commission. Lancet Oncol. 2020, 21, e185–e224. [Google Scholar] [CrossRef]
- de Oliveira, C.; Bremner, K.E.; Liu, N.; Greenberg, M.L.; Nathan, P.C.; McBride, M.L.; Krahn, M.D. Costs of cancer care in children and adolescents in Ontario, Canada. Pediatr. Blood Cancer 2017, 64, e26628. [Google Scholar] [CrossRef] [PubMed]
- Russell, H.V.; Panchal, J.; VonVille, H.; Franzini, L.; Swint, J.M. Economic Evaluation of Pediatric Cancer Treatment: A Systematic Literature Review. Pediatrics 2013, 131, e273–e287. [Google Scholar] [CrossRef] [PubMed]
- Langa, K.M.; Fendrick, A.M.; Chernew, M.E.; Kabeto, M.U.; Paisley, K.L.; Hayman, J.A. Out-of-Pocket Health-Care Expenditures among Older Americans with Cancer. Value Health 2004, 7, 186–194. [Google Scholar] [CrossRef] [PubMed]
- Mariotto, A.B.; Yabroff, K.R.; Shao, Y.; Feuer, E.J.; Brown, M.L. Projections of the Cost of Cancer Care in the United States: 2010–2020. J. Natl. Cancer Inst. 2011, 103, 117–128. [Google Scholar] [CrossRef]
- Mariotto, A.B.; Enewold, L.; Zhao, J.; Zeruto, C.A.; Yabroff, K.R. Medical Care Costs Associated with Cancer Survivorship in the United States. Cancer Epidemiol. Prev. Biomark. 2020, 29, 1304–1312. [Google Scholar] [CrossRef]
- Seah, T.; Zhang, C.; Halbert, J.; Prabha, S.; Gupta, S. The magnitude and predictors of therapy abandonment in pediatric central nervous system tumors in low- and middle-income countries: Systematic review and meta-analysis. Pediatr. Blood Cáncer 2019, 66, e27692. [Google Scholar] [CrossRef]
- Friedrich, P.; Lam, C.G.; Kaur, G.; Itriago, E.; Ribeiro, C.; Arora, R.S. Determinants of Treatment Abandonment in Childhood Cancer: Results from a Global Survey. PLoS ONE 2016, 11, e0163090. [Google Scholar] [CrossRef]
- Bhakta, N.; Force, L.M.; Allemani, C.; Atun, R.; Bray, F.; Coleman, M.P.; Steliarova-foucher, E.; Frazier, A.L.; Robison, L.L.; Rodriguez-galindo, C.; et al. Review Childhood cancer burden: A review of global estimates. Lancet Oncol. 2019, 20, e42–e53. [Google Scholar] [CrossRef]
- Winkler, E.C.; Mehlis, K.; Surmann, B.; Witte, J.; Lingnau, R.; Apostolidis, L.; Walther, J.; Greiner, W. Financial toxicity in German cancer patients: How does a chronic disease impact the economic situation? Ann. Oncol. 2018, 29, viii752. [Google Scholar] [CrossRef]
- Nathan, P.C.; Henderson, T.O.; Kirchhoff, A.C.; Park, E.R.; Yabroff, K.R. Financial hardship and the economic effect of childhood cancer survivorship. J. Clin. Oncol. 2018, 36, 2198–2205. [Google Scholar] [CrossRef]
- Altice, C.K.; Banegas, M.P.; Tucker-Seeley, R.D.; Yabroff, K.R. Financial hardships experienced by cancer survivors: A systematic review. J. Natl. Cancer Inst. 2017, 109, djw205. [Google Scholar] [CrossRef] [PubMed]
- Mueller, E.L.; Hall, M.; Berry, J.G.; Carroll, A.E.; Macy, M.L. Healthcare utilization and spending by children with cancer on Medicaid. Pediatr. Blood Cáncer 2017, 64, e26569. [Google Scholar] [CrossRef] [PubMed]
- Warner, B.E.L.; Kirchhoff, A.C.; Nam, G.E.; Fluchel, M. Financial Burden of Pediatric Cancer for Patients and Their Families. J. Oncol. Pract. 2014, 11, 12–18. [Google Scholar] [CrossRef] [PubMed]
- Chae, W.; Kim, J.; Park, S.; Park, E.-C.; Jang, S.-I. The Financial Burden Associated with Medical Costs among Childhood Cancer Patients and Their Families Related to Their Socioeconomic Status: The Perspective of National Health Insurance Service. Int. J. Environ. Res. Public Health 2020, 17, 6020. [Google Scholar] [CrossRef]
- Parra-Soto, S.; Petermann-Rocha, F.; Martínez-Sanguinetti, M.A.; Leiva-Ordeñez, A.M.; Troncoso-Pantoja, C.; Ulloa, N.; Diaz-Martínez, X.; Celis-Morales, C. Cáncer en Chile y en el mundo: Una mirada actual y su futuro escenario epidemiológico. Rev. Méd. Chil. 2020, 148, 1489–1495. [Google Scholar] [CrossRef]
- Rezende, L.F.M.; Murata, E.; Giannichi, B.; Tomita, L.Y.; Wagner, G.A.; Sanchez, Z.M.; Celis-Morales, C.; Ferrari, G. Cancer cases and deaths attributable to lifestyle risk factors in Chile. BMC Cancer 2020, 20, 693. [Google Scholar] [CrossRef] [PubMed]
- Jimenez, J.; Jara, D.; Bastias, G.; Ferreccio, C.; Moscoso, C.; Sagues, S.; Cid, C.; Bronstein, E.; Herrera, C.; Nervi, B.; et al. A snapshot of cancer in Chile: Analytical frameworks for developing a cancer policy. Biol. Res. 2015, 48, 1–15. [Google Scholar]
- Santolaya, M.E.; Alvarez, A.M.; Avilés, C.L.; Becker, A.; Cofré, J.; Cumsille, M.A.; O’Ryan, M.L.; Payá, E.; Salgado, C.; Silva, P.; et al. Early hospital discharge followed by outpatient management versus continued hospitalization of children with cancer, fever, and neutropenia at low risk for invasive bacterial infection. J. Clin. Oncol. 2004, 22, 3784–3789. [Google Scholar] [CrossRef]
- Ministerio de Desarrollo Social Encuesta de Caracterización Socioeconómica Nacional (CASEN). Available online: http://observatorio.ministeriodesarrollosocial.gob.cl/encuesta-casen-2017 (accessed on 20 January 2021).
- Villalobos Dintrans, P. Out-of-pocket health expenditure differences in Chile: Insurance performance or selection? Health Policy 2018, 122, 184–191. [Google Scholar] [CrossRef] [PubMed]
- Orellana, C.; Torres, S.; Derio, L.; Prieto, M. Cancer care in Chile. Lancet Oncol. 2003, 4, 653–656. [Google Scholar] [CrossRef]
- Congreso Nacional de Chile. Ley 20015; Congreso Nacional de Chile: Santiago, Chile, 2005.
- Urriola, R. Chile: Social protection in health. Rev. Panam. Salud Publica 2006, 20, 273–286. [Google Scholar] [CrossRef][Green Version]
- DEIS—Ministerio de Salud de Chile. Bases de Datos Egresos Hospitalarios. Available online: https://deis.minsal.cl/#datosabiertos (accessed on 20 January 2021).
- Goss, P.E.; Lee, B.L.; Badovinac-Crnjevic, T.; Strasser-Weippl, K.; Chavarri-Guerra, Y.; Louis, J.S.; Villarreal-Garza, C.; Vasconcelos, C.; Cazap, E.; Vallejos, C.; et al. Planning cancer control in Latin America and the Caribbean. Lancet Oncol. 2013, 14, 391–436. [Google Scholar] [CrossRef]
- World Health Organization. ICD-10: International Statistical Classification of Diseases and Related Health Problems: Tenth Revision. Available online: https://apps.who.int/iris/handle/10665/42980 (accessed on 20 January 2021).
- Banco Central de Chile Indicadores Diarios. Available online: https://si3.bcentral.cl/Indicadoressiete/secure/Indicadoresdiarios.aspx (accessed on 20 January 2021).
- McCullagh, P.; Nelder, J.A. Generalized Linear Models; Routledge: London, UK, 2019; ISBN 0203753739. [Google Scholar]
- Manning, W.G.; Mullahy, J. Estimating log models: To transform or not to transform? J. Health Econ. 2001, 20, 461–494. [Google Scholar] [CrossRef]
- Mihaylova, B.; Briggs, A.; O’Hagan, A.; Thompson, S.G. Review of statistical methods for analysing healthcare resources and costs. Health Econ. 2011, 20, 897–916. [Google Scholar] [CrossRef] [PubMed]
- Coughlan, D.; Yeh, S.T.; O’Neill, C.; Frick, K.D. Evaluating Direct Medical Expenditures Estimation Methods of Adults Using the Medical Expenditure Panel Survey: An Example Focusing on Head and Neck Cancer. Value Health 2014, 17, 90–97. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Liu, L.; Shih, Y.-C.T.; Zhang, D.; Severini, T.A. A flexible model for correlated medical costs, with application to medical expenditure panel survey data. Stat. Med. 2016, 35, 883–894. [Google Scholar] [CrossRef] [PubMed]
- Deb, P.; Norton, E.C. Modeling Health Care Expenditures and Use. Annu. Rev. Public Health 2018, 39, 489–505. [Google Scholar] [CrossRef]
- Deb, P.; Norton, E.C.; Manning, W.G. Health Econometrics Using Stata; Stata Press: College Station, TX, USA, 2017; ISBN 978-1-59718-229-4. [Google Scholar]
- Guzman, P.C.; Cordoba, M.A.; Godoy, N.; Casta, A.; Ribeiro, K.B.; Moreno, F.; Vries, E. De Childhood cancer in Latin America: From detection to palliative care and survivorship. Cancer Epidemiol. 2021, 71, 101837. [Google Scholar] [CrossRef]
- Ministerio de Salud de Chile. Plan Nacional de Cáncer 2018–2028; Ministerio de Salud de Chile: Santiago, Chile, 2018.
- Villalobos Dintrans, P.; Hasen, F.; Izquierdo, C.; Santander, S. New challenges for health planning: Chile’s National Cancer Plan. Rev. Panam. Salud Publica 2020, 44, e6. [Google Scholar] [CrossRef]
- Russell, H.; Bernhardt, M.B. Bending the Cost Curve in Childhood Cancer. Curr. Hematol. Malig. Rep. 2016, 11, 295–302. [Google Scholar] [CrossRef]
- The World Bank World Development Indicators. Available online: https://databank.worldbank.org/source/world-development-indicators (accessed on 20 January 2021).
- Gupta, S.; Yeh, S.; Martiniuk, A.; Lam, C.G. The magnitude and predictors of abandonment of therapy in paediatric acute leukaemia in middle-income countries: A systematic review and meta-analysis. Eur. J. Cancer 2013, 49, 2555–2564. [Google Scholar] [CrossRef]
- Friedrich, P.; Lam, C.G.; Itriago, E.; Perez, R.; Ribeiro, R.C.; Arora, R.S. Magnitude of Treatment Abandonment in Childhood Cancer. PLoS ONE 2015, 10, e0135230. [Google Scholar] [CrossRef]
- Narang, A.K.; Nicholas, L.H. Out-of-pocket spending and financial burden among medicare beneficiaries with cancer. JAMA Oncol. 2017, 3, 757–765. [Google Scholar] [CrossRef] [PubMed]
- Anhang Price, R.; Stranges, E.; Elixhauser, A. Pediatric Cancer Hospitalizations, 2009. In Healthcare Cost and Utilization Project (HCUP) Statistical Briefs [Internet]; Agency for Healthcare Research and Quality: Rockville, MD, USA, 2012. Available online: http://www.hcup-us.ahrq.gov/reports/statbriefs/sb132.pdf (accessed on 20 January 2021).
- Kaul, B.S.; Barbeau, B.; Wright, J.; Fluchel, M.; Kirchhoff, A.C.; Nelson, R.E. Statewide Longitudinal Hospital Use and Charges for Pediatric and Adolescent Patients With Cancer. J. Oncol. Pract. 2015, 11, e468–e475. [Google Scholar] [CrossRef]
- Palma, J.; Mosso, C.; Paris, C.; Campbell, M.; Tong, X.; King, A.; Carrion, F.; Rivera, G.K. Establishment of a Pediatric HSCT Program in a Public Hospital in Chile. Pediatr. Blood Cáncer 2006, 46, 803–810. [Google Scholar] [CrossRef] [PubMed]
- Tsimicalis, A.; Stevens, B.; Ungar, W.J.; McKeever, P.; Greenberg, M. The cost of childhood cancer from the family’s perspective: A critical review. Pediatr. Blood Cáncer 2011, 56, 707–717. [Google Scholar] [CrossRef]
- Essue, B.M.; Iragorri, N.; Fitzgerald, N.; de Oliveira, C. The psychosocial cost burden of cancer: A systematic literature review. Psycho-Oncology 2020, 29, 1746–1760. [Google Scholar] [CrossRef] [PubMed]
- Akaike, H. Statistical predictor identification. Ann. Inst. Stat. Math. 1970, 22, 203–217. [Google Scholar] [CrossRef]
- Schwarz, G. Estimating the Dimension of a Model. Ann. Stat. 1978, 6, 461–464. [Google Scholar] [CrossRef]
- Box, G.E.P.; Cox, D.R. An Analysis of Transformations. J. R. Stat. Soc. Ser. B 1964, 26, 211–252. [Google Scholar] [CrossRef]
- Park, R.E. Estimation with Heteroscedastic Error Terms. Econometrica 1966, 34, 888. [Google Scholar] [CrossRef]
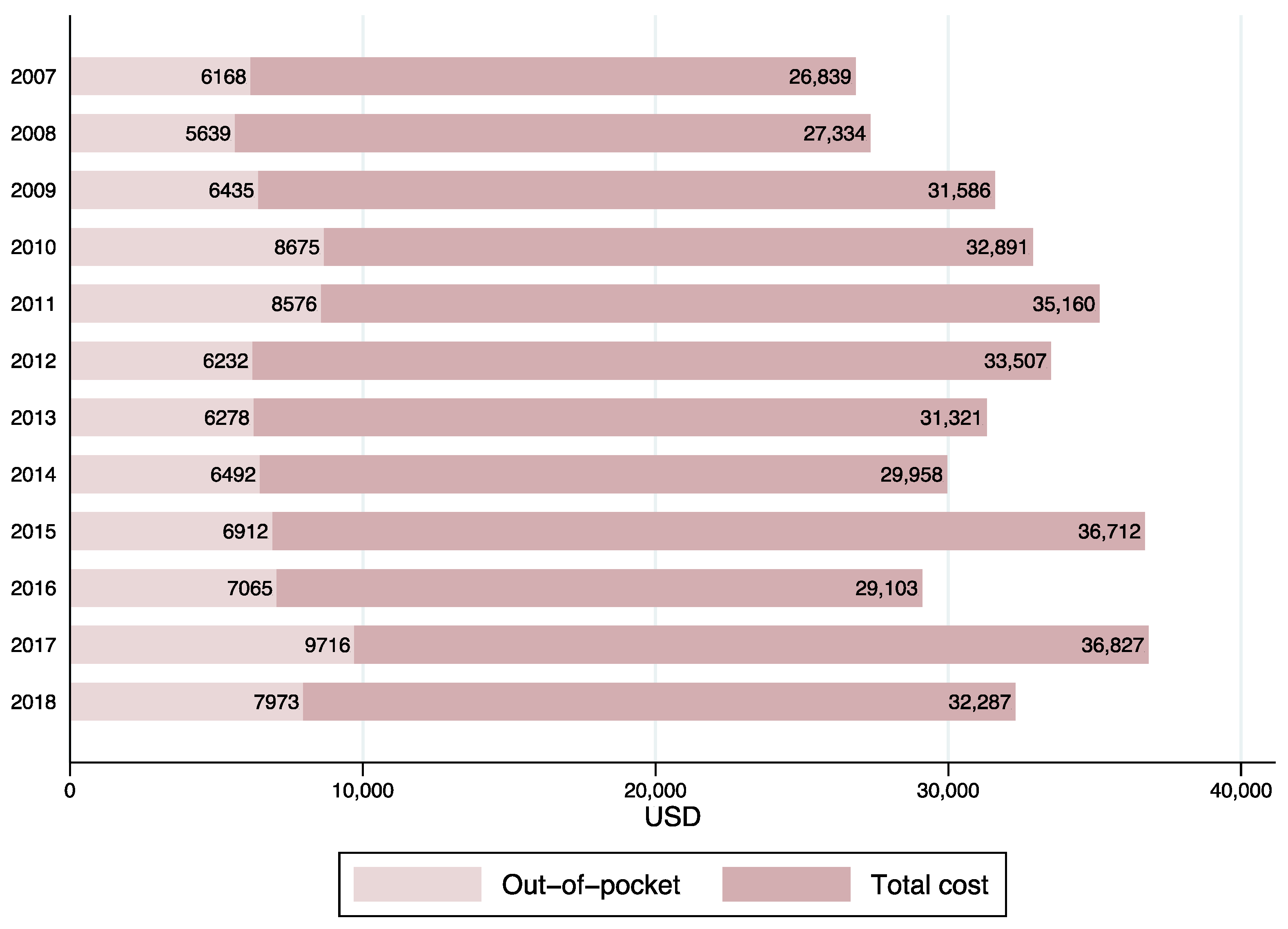

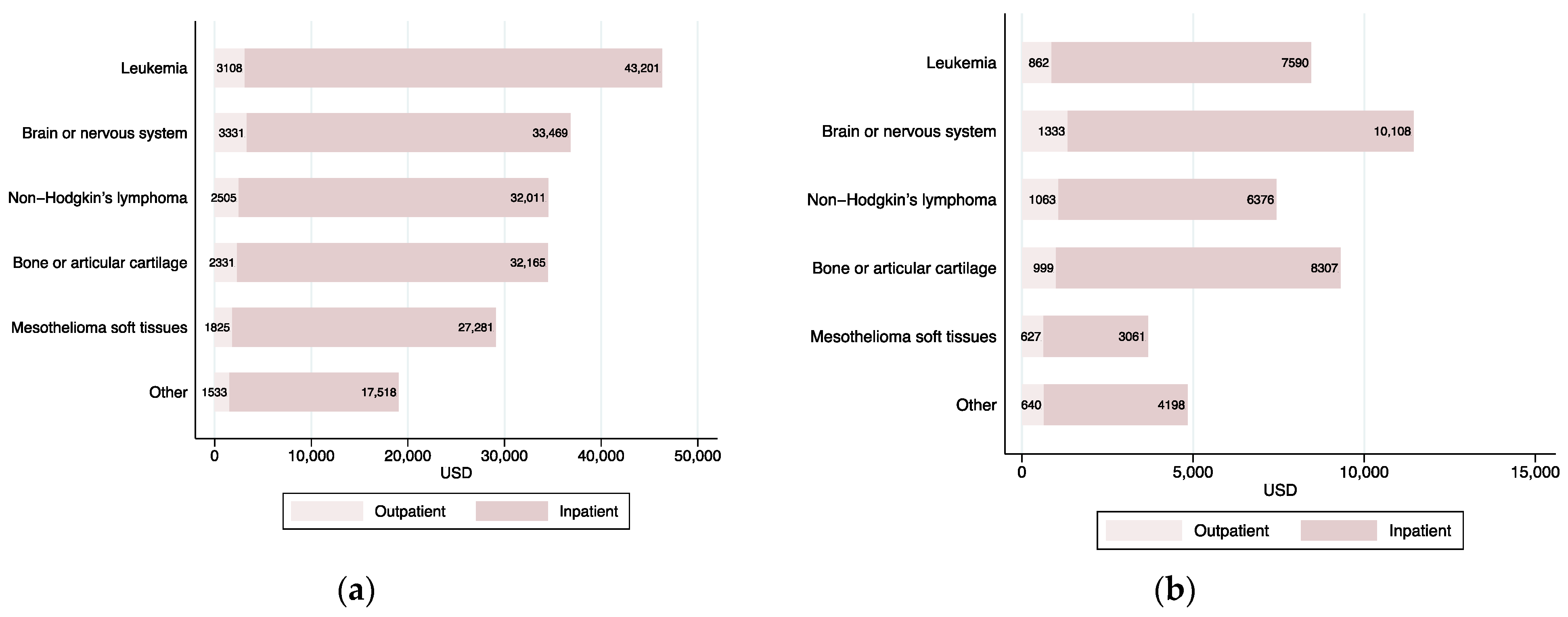
| All Years | 2007 | 2018 | Mean Difference t-Test p-Value: 2007 vs. 2018 | |
|---|---|---|---|---|
| Patient | ||||
| n | 3853 | 248 | 407 | |
| Distribution by age group (%) | ||||
| 0–4 | 26.0 | 23.8 | 22.9 | 0.783 |
| 5–9 | 30.5 | 29.4 | 34.4 | 0.185 |
| 10–14 | 24.1 | 25.4 | 22.4 | 0.379 |
| 15–17 | 19.5 | 21.4 | 20.4 | 0.766 |
| Mean age | 8.8 | 9.2 | 8.9 | 0.478 |
| % Male | 52.8 | 51.2 | 52.8 | 0.689 |
| Distribution by most frequent discharge’s diagnosis | ||||
| Leukemia | 31.8 | 30.2 | 32.2 | 0.602 |
| Brain or nervous system | 14.0 | 15.3 | 14.3 | 0.709 |
| Non-Hodgkin’s lymphoma | 5.3 | 4.8 | 4.2 | 0.695 |
| Bone or articular cartilage | 4.9 | 10.5 | 4.2 | 0.004 |
| Mesothelioma soft tissues | 3.2 | 4.0 | 3.7 | 0.824 |
| Other cancer | 40.6 | 35.1 | 41.5 | 0.099 |
| GES | 48.8 | 28.2 | 49.4 | <0.001 |
| Policyholder | ||||
| Mean age | 41.4 | 41.4 | 42.3 | 0.101 |
| % Male | 67.9 | 68.1 | 63.4 | 0.212 |
| Distribution by policyholder’s region of residence (%) | ||||
| Metropolitan | 58.9 | 57.3 | 62.7 | 0.173 |
| South | 20.7 | 25.0 | 17.7 | 0.029 |
| North | 20.4 | 17.7 | 19.7 | 0.541 |
| Mean annual income | 25,024 | 20,813 | 27,608 | <0.001 |
| Total Costs | Out-of-Pocket | |||||
|---|---|---|---|---|---|---|
| (1) | (2) | (3) | (4) | (5) | (6) | |
| Variables | All | Inpatient | Outpatient | All | Inpatient | Outpatient |
| Patient | ||||||
| Age 5–9 | −13,969.85 *** | −13,991.49 *** | 96.49 | −3214.58 *** | −3220.38 *** | −57.85 |
| (2463.942) | (2435.629) | (188.172) | (671.170) | (665.850) | (77.940) | |
| Age 10–14 | −9674.74 *** | −10,245.84 *** | 522.4 ** | −1542.35 * | −1657.94 ** | 32.56 |
| (2767.828) | (2711.179) | (243.069) | (822.560) | (790.610) | (80.320) | |
| Age 15–17 | −9661.96 *** | −10,556.51 *** | 661.95 ** | −1496.72 * | −1669.15 ** | 51.2 |
| (3041.192) | (3023.489) | (262.877) | (856.600) | (829.360) | (84.810) | |
| Male | 1363.68 | 1304.63 | 94.78 | −183.05 | −233.59 | 11.92 |
| (1773.860) | (1747.659) | (169.036) | (546.240) | (523.200) | (58.310) | |
| Leukemia | 28,304.76 *** | 26,939.25 *** | 1353.22 *** | 4369.01 *** | 4153.19 *** | 267.53 *** |
| (2105.794) | (2100.442) | (215.345) | (705.640) | (692.950) | (62.310) | |
| Brain or nervous system | 18,485.8 *** | 16,676.62 *** | 1597.48 *** | 5972.12 *** | 5171.59 *** | 688.78 *** |
| (2644.383) | (2561.675) | (340.909) | (1179.220) | (1044.750) | (195.620) | |
| Non-Hodgkin’s lymphoma | 15,833.76 *** | 15,170.36 *** | 777.33 *** | 3061.63 *** | 2697.37 *** | 427.54 *** |
| (3906.076) | (3844.151) | (287.064) | (1087.530) | (1033.340) | (154.210) | |
| Bone or articular cartilage | 17,459 *** | 16,742.74 *** | 663.52 ** | 4070.47 *** | 3909.27 *** | 268.87 ** |
| (4115.738) | (4007.618) | (303.541) | (1491.240) | (1456.600) | (107.120) | |
| Mesothelioma soft tissues | 9162.15 ** | 8511.43 ** | 432.82 | −750.11 | −787.94 | 32.02 |
| (3655.754) | (3487.766) | (400.487) | (584.050) | (548.700) | (82.530) | |
| GES payment | 8882.34 *** | 8351.97 *** | 375.9 ** | −3362.63 *** | −3127.56 *** | −241.39 *** |
| (1782.42) | (1755.309) | (163.707) | (613.62) | (593.160) | (58.040) | |
| Policyholder | ||||||
| Residence in Metropolitan region | 3210.62 * | 3926.84 ** | −539.1 *** | 894.6 | 870.76 | 67.02 |
| (1838.65) | (1814.621) | (194.623) | (583.18) | (554.330) | (60.460) | |
| Q1 of annual income | 760.89 | 1839.36 | −855.7 *** | −951.86 | −344.02 | −502.96 *** |
| (3357.45) | (3320.530) | (277.783) | (1120.34) | (1078.720) | (126.120) | |
| Q2 of annual income | −4803.83 | −3856.67 | −891.09 *** | −3007.65 *** | −2501.14 *** | −445.59 *** |
| (3029.80) | (2969.836) | (251.228) | (888.16) | (821.320) | (125.280) | |
| Q3 of annual income | −4744.52 | −4241.48 | −405.82 | −1227.25 | −917.27 | −234.05 * |
| (3111.73) | (3051.960) | (264.918) | (1036.66) | (974.580) | (130.890) | |
| Q4 of annual income | −1816.63 | −1710.82 | −163.5 | −1085.19 | −839.01 | −204.19 |
| (3217.22) | (3139.039) | (287.951) | (989.05) | (930.460) | (133.590) | |
| Policyholder’s demographic controls | Yes | Yes | Yes | Yes | Yes | Yes |
| Year dummies | Yes | Yes | Yes | Yes | Yes | Yes |
| Observations | 3644 | 3644 | 3621 | 3638 | 3212 | 3615 |
| Mean of outcome | 32,124 | 29,737 | 2387 | 7236 | 6388 | 848 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Borrescio-Higa, F.; Valdés, N. Medical Cost of Cancer Care for Privately Insured Children in Chile. Int. J. Environ. Res. Public Health 2021, 18, 6746. https://doi.org/10.3390/ijerph18136746
Borrescio-Higa F, Valdés N. Medical Cost of Cancer Care for Privately Insured Children in Chile. International Journal of Environmental Research and Public Health. 2021; 18(13):6746. https://doi.org/10.3390/ijerph18136746
Chicago/Turabian StyleBorrescio-Higa, Florencia, and Nieves Valdés. 2021. "Medical Cost of Cancer Care for Privately Insured Children in Chile" International Journal of Environmental Research and Public Health 18, no. 13: 6746. https://doi.org/10.3390/ijerph18136746
APA StyleBorrescio-Higa, F., & Valdés, N. (2021). Medical Cost of Cancer Care for Privately Insured Children in Chile. International Journal of Environmental Research and Public Health, 18(13), 6746. https://doi.org/10.3390/ijerph18136746






