The Effectiveness of Additional Core Stability Exercises in Improving Dynamic Sitting Balance, Gait and Functional Rehabilitation for Subacute Stroke Patients (CORE-Trial): Study Protocol for a Randomized Controlled Trial
Abstract
1. Introduction
2. Materials and Methods
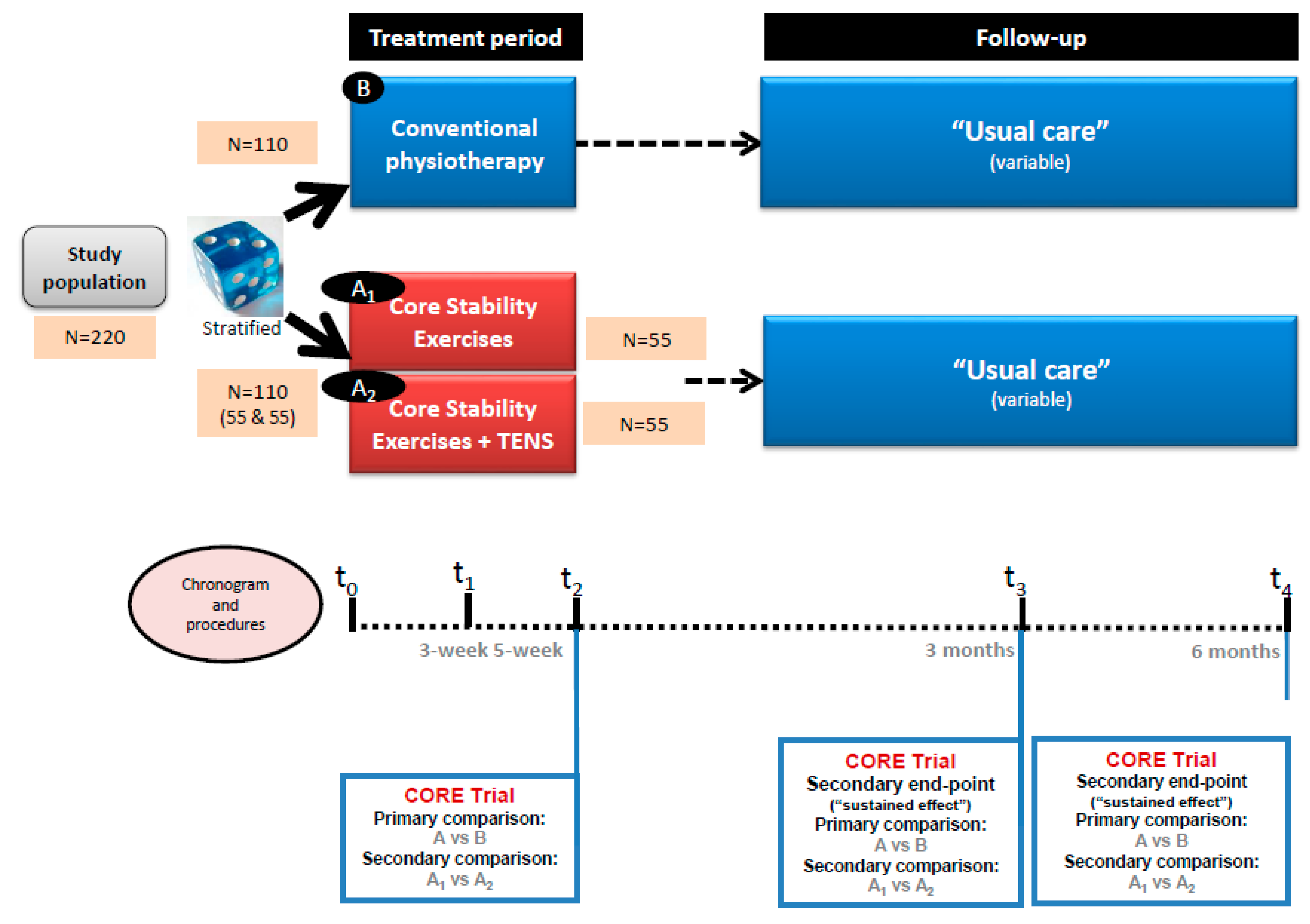
2.1. Study Design and Setting
2.2. Recruitment
Patient and Public Involvement Subsection
2.3. Blinding
2.4. Selection Criteria
- First ever-stroke ≤ 30 days (diagnostic criteria according to the World Health Organisation definition; corresponding to ICD-9 code 434) whether cortical or subcortical, and ischemic or hemorrhagic.
- Unilateral localisation of the stroke verified by computed tomography; if a patient shows previous problems, but does not have any neurological or clinical impairment, he/she would be included in the study.
- Both sexes and age ≥ 18 years old.
- Ability to understand and execute simple instructions.
- Severity of stroke by the Spanish National institute of Health Stroke Scale (S-NIHSS) [48] score ≥ 2 points.
- Modified Rankin Scale [49] > 2 points before stroke.
- Concurrent neurological disorder (e.g., Parkinson’s disease) or major orthopedic problem (e.g., amputation) that hampers sitting balance.
- Relevant psychiatric disorders that may prevent from following instructions.
- Other treatments that could influence the effects of the interventions.
- Contraindication to physical activity (e.g., heart failure).
- Use of cardiac pacemakers.
- Patients with hemorrhagic strokes that have undergone surgery for intracranial decompression.
- Patients whose stroke occurs exclusively and only in the cerebellum and brainstem. Patients whose main stroke is localised on another area and who also have a small lesion in the cerebellum and brainstem would not be excluded.
2.5. Interventions
Intervention Description
2.6. Participant Timeline
2.7. Outcomes Measures
- Dynamic sitting balance and coordination measured by S-TIS 2.0 [48]. This scale is a Spanish version of the Trunk Impairment Scale version 2.0 [55]. This scale aims to evaluate the trunk in patients who have suffered a stroke. The dynamic subscale contains items on the lateral flexion of the trunk and unilateral lifting of the hip. To assess the coordination of the trunk, the individual is asked to rotate the upper or lower part of his or her trunk six times, initiating the movements either from the shoulder girdle or from the pelvic girdle, respectively. There are two subscales; the first one has 10 items and the second one has six. The highest possible total score is consequently 16 points, which indicates an optimal dynamic sitting balance and sitting coordination. If the patient cannot maintain a sitting position for 10 s without back and arm support, with hands on thighs, feet in contact with the ground and knees bent at 90° (starting position), the total score for the scale is 0 points. This scale is utilised for inclusion criteria, and at T0, T1, T2, T3 and T4.
- Gait by stepping section of Brunel Balance Assessment (BBA) [56]. It is designed to assess functional balance for people with a wide range of abilities, and has been tested specifically for use post-stroke. There are three sections to the assessment: sitting, standing and stepping. In this study, only the stepping section is utilised. It consists of six levels to assess standing functional balance and a 5-m walk. At each level, the patient receives a score for his/her efforts. This gives an indication on whether the patient is improving within a level, even if he/she is not able to progress to the next level. The score also reflects how well the individual is functioning within that stepping section. The higher score is six points, and the individual is able to walk 5 m independently. Stepping is evaluated at T0, T1, T2, T3 and T4.
- Sitting functional balance is assessed by the Spanish version of Function in Sitting test (S-FIST) [57]. It is a bedside evaluation of sitting balance and functional sitting everyday activities that assess sensory, motor, proactive, reactive and steady balance factors. The S-FIST consists of 14 tested parameters with an ordinal scale (0–4) for each test item, with 0 indicating the lowest level of function and 4 the highest level. Each participant sat at the edge of a standard hospital bed without air mattresses, with the proximal thigh (1/2 femur length) supported by the bed. The bed height was adjusted and a step stool was used if necessary to bring the hips and knees to approximately 90° flexion, with both feet flat on the floor or stool. The higher score is 56 points. Sitting functional balance is evaluated at T0, T2, T3 and T4.
- Standing balance and risk of falling is evaluated by Berg Balance Scale (BBS) [58,59]. It provides a psychometrically sound measure of balance impairment. It is used objectively determine a patient’s ability (or inability) to safely balance during a series of predetermined tasks. It is a 14-item scale; patients must maintain positions and complete moving tasks of varying difficulty. In most items, patients must maintain a given position for a specified time. Each item consists of a 5-point ordinal scale ranging from 0 to 4, with 0 indicating the lowest level of function and 4 the highest. A score of 56 indicates functional balance. A score of < 45 indicates that individuals may be at greater risk of falling. BBS is assessed at T0, T2, T3 and T4.
- Postural control is evaluated by the Spanish version of Postural Assessment Scale for Stroke (S-PASS) [60]. It was designed specifically for patients with a stroke, regardless of postural competence. It has two subscales: mobility and balance. The first measures the patient’s ability to change position from lying, sitting and standing, and the second in maintaining stable postures in sitting and standing. The S-PASS consists of 12 items with a 4-point scale, where items are scored from 0–3. The higher score is 36 points, indicating an optimal postural control. It is evaluated at T0, T2, T3 and T4.
- Lower limb spasticity by Modified Ashworth Scale (MAS) [61]. This tool measures resistance during passive soft-tissue stretching of muscle. It is performed while the assessor moves the hip adductors, knee extensors and ankle plantar flexors in the supine and lateral position. The MAS is assessed at T0, T2, T3 and T4.
- ADL by Barthel Index (BI) [62]. This shows the degree of independence of a patient from any assistance. It covers 10 domains of function (activities): bowel and bladder control, as well as help with grooming, toilet use, feeding, transfers, walking, dressing, climbing stairs and bathing. The ADL is evaluated at T0, T2, T3 and T4.
- Health-related quality of life is measured by the Spanish-version of 5-Dimensions Questionnaire (EQ-5D-5L) [63,64]. It is a generic patient’s health-related quality of life measurement with evidence of good reliability and validity in various disease populations, including strokes. Patients chose five levels of severity (1, no problem; 2, slight problem; 3, moderate problem; 4, severe problem; and 5, unable to function/extreme problem) in five dimensions (mobility, self-care, usual activity, pain/discomfort and depression/anxiety), and rated their overall health status via the EQ-VAS. Quality of life is assessed at T0, T2, T3 and T4.
- Rate of falls is measured by a specific registry created specifically for this study. The outcome is defined as the average number of falls per patient during the intervention period and follow-up. It is recorded at T0 (falls before stroke), T2, T3 and T4.
- Gait speed is assessed by BTS G-Walk. It is a wireless system consisting of an inertial sensor composed by a triaxial accelerometer, a magnetic sensor and a triaxial gyroscope that was positioned on S1 vertebrae. From the data acquired, the system extrapolates all spatial-temporal gait. The patient walks for one minute without being aided; this variable is only performed if the patient has a 6-point stepping section of BBA.
Baseline Assessment
2.8. Statistical Analysis
3. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Trial Registration
Abbreviations
| ADL | Activities of daily living |
| BBA | Brunel Balance Assessment |
| BBS | Berg Balance Scale |
| CG | Control group |
| CP | Conventional physiotherapy |
| CSEs | Core Stability exercises |
| EG | Experimental group |
| mRS | Modified Rankin Scale |
| NIHSS | National Institute of Health Stroke Scale |
| QoL | Quality of life |
| RCT | Randomized controlled trial |
| S-FIST | Spanish version of Function in Sitting Test |
| S-PASS | Spanish version of Postural Assessment Scale for Stroke |
| S-TIS 2.0 | Spanish version of Trunk Impairment Scale |
| TENS | Transcutaneous electrical nerve stimulation |
References
- Donkor, E.S. Stroke in the 21st Century: A Snapshot of the Burden, Epidemiology, and Quality of Life. Stroke Res. Treat. 2018, 2018, 1–10. [Google Scholar] [CrossRef]
- Tyson, S.F.; Hanley, M.; Chillala, J.; Selley, A.; Tallis, R.C. Balance Disability After Stroke. Phys. Ther. 2006, 86, 30–38. [Google Scholar] [CrossRef]
- Gadidi, V.; Katz-Leurer, M.; Carmeli, E.; Bornstein, N.M. Long-Term Outcome Poststroke: Predictors of Activity Limitation and Participation Restriction. Arch. Phys. Med. Rehabil. 2011, 92, 1802–1808. [Google Scholar] [CrossRef]
- Oliveira, C.B.; Medeiros, Í.R.T.; Greters, M.G.; Frota, N.A.F.; Lucato, L.; Scaff, M.; Conforto, A. Abnormal sensory integration affects balance control in hemiparetic patients within the first year after stroke. Clinics 2011, 66, 2043–2048. [Google Scholar] [CrossRef]
- Isho, T.; Usuda, S. Association of trunk control with mobility performance and accelerometry-based gait characteristics in hemiparetic patients with subacute stroke. Gait Posture 2016, 44, 89–93. [Google Scholar] [CrossRef]
- Schmid, A.A.; Rittman, M. Consequences of Poststroke Falls: Activity Limitation, Increased Dependence, and the Development of Fear of Falling. Am. J. Occup. Ther. 2009, 63, 310–316. [Google Scholar] [CrossRef]
- Galeoto, G.; Iori, F.; De Santis, R.; Santilli, V.; Mollica, R.; Marquez, M.A.; Sansoni, J.; Berardi, A. The outcome measures for loss of functionality in the activities of daily living of adults after stroke: A systematic review. Top. Stroke Rehabil. 2019, 26, 236–245. [Google Scholar] [CrossRef] [PubMed]
- Pouwels, S.; Lalmohamed, A.; Leufkens, B.; de Boer, A.; Cooper, C.; van Staa, T.; de Vries, F. Risk of hip/femur fracture after stroke: A population-based case-control study. Stroke 2009, 40, 3281–3285. [Google Scholar] [CrossRef]
- Lozano, R.; Naghavi, M.; Foreman, K.; Lim, S.; Shibuya, K.; Aboyans, V.; Abraham, J.; Adair, T.; Aggarwal, R.; Ahn, S.Y.; et al. Global and regional mortality from 235 causes of death for 20 age groups in 1990 and 2010: A systematic analysis for the Global Burden of Disease Study 2010. Lancet 2012, 380, 2095–2128. [Google Scholar] [CrossRef]
- Park, J.; Yoo, I. Relationships of Stroke Patients’ Gait Parameters with Fear of Falling. J. Phys. Ther. Sci. 2014, 26, 1883–1884. [Google Scholar] [CrossRef][Green Version]
- Kim, E.J.; Kim, D.Y.; Kim, W.H.; Lee, K.L.; Yoon, Y.H.; Park, J.M.; Shin, J.I.; Kim, S.K.; Kim, D.G. Fear of Falling in Subacute Hemiplegic Stroke Patients: Associating Factors and Correlations with Quality of Life. Ann. Rehabil. Med. 2012, 36, 797–803. [Google Scholar] [CrossRef] [PubMed]
- Barra, J.; Marquer, A.; Joassin, R.; Reymond, C.; Metge, L.; Chauvineau, V.; Pérennou, D. Humans use internal models to construct and update a sense of verticality. Brain 2010, 133, 3552–3563. [Google Scholar] [CrossRef] [PubMed]
- Ryerson, S.; Byl, N.N.; Brown, D.A.; Wong, R.A.; Hidler, J.M. Altered Trunk Position Sense and Its Relation to Balance Functions in People Post-Stroke. J. Neurol. Phys. Ther. 2008, 32, 14–20. [Google Scholar] [CrossRef]
- van Nes, I.J.; Nienhuis, B.; Latour, H.; Geurts, A.C. Posturographic assessment of sitting balance recovery in the subacute phase of stroke. Gait Posture 2008, 28, 507–512. [Google Scholar] [CrossRef]
- Harbourne, R.T.; Lobo, M.A.; Karst, G.M.; Galloway, J.C. Sit happens: Does sitting development perturb reaching development, or vice versa? Infant Behav. Dev. 2013, 36, 438–450. [Google Scholar] [CrossRef] [PubMed]
- Hara, Y. Brain Plasticity and Rehabilitation in Stroke Patients. J. Nippon Med. Sch. 2015, 82, 4–13. [Google Scholar] [CrossRef] [PubMed]
- Coleman, E.R.; Moudgal, R.; Lang, K.; Hyacinth, H.I.; Awosika, O.O.; Kissela, B.M.; Feng, W. Early Rehabilitation After Stroke: A Narrative Review. Curr. Atheroscler. Rep. 2017, 19, 1–12. [Google Scholar] [CrossRef]
- Cassidy, J.; Cramer, S.C. Spontaneous and Therapeutic-Induced Mechanisms of Functional Recovery After Stroke. Transl. Stroke Res. 2017, 8, 33–46. [Google Scholar] [CrossRef]
- Regenhardt, R.; Takase, H.; Lo, E.H.; Lin, D.J. Translating concepts of neural repair after stroke: Structural and functional targets for recovery. Restor. Neurol. Neurosci. 2020, 38, 67–92. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.B.; Lim, S.H.; Kim, K.H.; Kim, K.J.; Kim, Y.R.; Chang, W.N.; Yeom, J.W.; Kim, Y.D.; Hwang, B.Y. Six-month functional recovery of stroke patients. Int. J. Rehabil. Res. 2015, 38, 173–180. [Google Scholar] [CrossRef] [PubMed]
- Pollock, A.; Baer, G.; Campbell, P.; Choo, P.L.; Forster, A.; Morris, J.; Pomeroy, V.M.; Langhorne, P. Physical rehabilitation approaches for the recovery of function and mobility following stroke. Cochrane Database Syst. Rev. 2014, 2014. [Google Scholar] [CrossRef]
- Cabanas-Valdés, R.; Cuchi, G.U.; Bagur-Calafat, C. Trunk training exercises approaches for improving trunk performance and functional sitting balance in patients with stroke: A systematic review. Neurorehabilitation 2013, 33, 575–592. [Google Scholar] [CrossRef]
- Van Criekinge, T.; Truijen, S.; Schröder, J.; Maebe, Z.; Blanckaert, K.; Van Der Waal, C.; Vink, M.; Saeys, W. The effectiveness of trunk training on trunk control, sitting and standing balance and mobility post-stroke: A systematic review and meta-analysis. Clin. Rehabil. 2019, 33, 992–1002. [Google Scholar] [CrossRef]
- Cabanas-Valdés, R.; Bagur-Calafat, C.; Girabent-Farrés, M.; Caballero-Gómez, F.M.; Hernández-Valiño, M.; Cuchí, G.U. The effect of additional core stability exercises on improving dynamic sitting balance and trunk control for subacute stroke patients: A randomized controlled trial. Clin. Rehabil. 2016, 30, 1024–1033. [Google Scholar] [CrossRef]
- Szafraniec, R.; Barańska, J.; Kuczyński, M. Acute effects of core stability exercises on balance control. Acta Bioeng Biomech 2018, 20, 145–151. [Google Scholar] [PubMed]
- Haruyama, K.; Kawakami, M.; Otsuka, T. Effect of Core Stability Training on Trunk Function, Standing Balance, and Mobility in Stroke Patients. Neurorehabilit. Neural Repair 2016, 31, 240–249. [Google Scholar] [CrossRef]
- Dhawale, T.; Yeole, U.; Pawar, A. Effect of trunk control exercises on balance and gait in stroke patients-randomized control trial. Age 2018, 40, 60. [Google Scholar]
- Suh, J.H.; Lee, E.C.; Kim, J.S.; Yoon, S.Y. Association between trunk core muscle thickness and functional ability in subacute hemiplegic stroke patients: An exploratory cross-sectional study. Top. Stroke Rehabil. 2021, 1–10. [Google Scholar] [CrossRef]
- Lee, N.G.; You, J.; Sung, H.; Yi, C.H.; Jeon, H.S.; Choi, B.S.; Lee, D.R.; Park, J.M.; Lee, T.H.; Ryu, I.T.; et al. Best Core Stabilization for Anticipatory Postural Adjustment and Falls in Hemiparetic Stroke. Arch. Phys. Med. Rehabil. 2018, 99, 2168–2174. [Google Scholar] [CrossRef] [PubMed]
- Cabanas-Valdés, R.; Bagur-Calafat, C.; Girabent-Farrés, M.; Caballero-Gómez, F.M.; Pontcharra-Serra, H.D.P.D.; German-Romero, A.; Urrútia, G. Long-term follow-up of a randomized controlled trial on additional core stability exercises training for improving dynamic sitting balance and trunk control in stroke patients. Clin. Rehabil. 2017, 31, 1492–1499. [Google Scholar] [CrossRef] [PubMed]
- Marzolini, S.; Robertson, A.D.; Oh, P.; Goodman, J.M.; Corbett, D.; Du, X.; MacIntosh, B.J. Aerobic Training and Mobilization Early Post-stroke: Cautions and Considerations. Front. Neurol. 2019, 10, 1187. [Google Scholar] [CrossRef]
- Akuthota, V.; Ferreiro, A.; Moore, T.; Fredericson, M. Core Stability Exercise Principles. Curr. Sports Med. Rep. 2008, 7, 39–44. [Google Scholar] [CrossRef] [PubMed]
- Akuthota, V.; Nadler, S.F. Core strengthening. Arch. Phys. Med. Rehabil. 2004, 85, 86–92. [Google Scholar] [CrossRef]
- Ben Kibler, W.; Press, J.; Sciascia, A. The Role of Core Stability in Athletic Function. Sports Med. 2006, 36, 189–198. [Google Scholar] [CrossRef] [PubMed]
- Israely, S.; Leisman, G.; Carmeli, E. Neuromuscular synergies in motor control in normal and poststroke individuals. Rev. Neurosci. 2018, 29, 593–612. [Google Scholar] [CrossRef] [PubMed]
- Chan, B.K.S.; Ng, S.S.M.; Ng, G.Y.F. A Home-Based Program of Transcutaneous Electrical Nerve Stimulation and Task-Related Trunk Training Improves Trunk Control in Patients with Stroke A Randomized Controlled Clinical Trial. Neurorehabil. Neural Repair 2015, 29, 70–79. [Google Scholar] [CrossRef]
- Ng, S.S.M.; Hui-Chan, C.W.Y. Does the use of TENS increase the effectiveness of exercise for improving walking after stroke? A randomized controlled clinical trial. Clin. Rehabil. 2009, 23, 1093–1103. [Google Scholar] [CrossRef] [PubMed]
- Sarabon, N.; Rosker, J. Effects of Fourteen-Day Bed Rest on Trunk Stabilizing Functions in Aging Adults. BioMed Res. Int. 2015, 2015, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Souza, D.C.B.; Santos, M.D.S.; Ribeiro, N.M.D.S.; Maldonado, I.L. Inpatient trunk exercises after recent stroke: An update meta-analysis of randomized controlled trials. NeuroRehabilitation 2019, 44, 369–377. [Google Scholar] [CrossRef]
- Stinear, C.M. Stroke rehabilitation research needs to be different to make a difference. F1000Research 2016, 5, 1467. [Google Scholar] [CrossRef] [PubMed]
- Crozier, J.; Roig, M.; Eng, J.J.; MacKay-Lyons, M.; Fung, J.; Ploughman, M.; Bailey, D.M.; Sweet, S.N.; Giacomantonio, N.; Thiel, A.; et al. High-Intensity Interval Training After Stroke: An Opportunity to Promote Functional Recovery, Cardiovascular Health, and Neuroplasticity. Neurorehabilit. Neural Repair 2018, 32, 543–556. [Google Scholar] [CrossRef]
- Walker, M.F.; Hoffmann, T.C.; Brady, M.C.; Dean, C.; Eng, J.J.; Farrin, A.J.; Felix, C.; Forster, A.; Langhorne, P.; Lynch, E.; et al. Improving the Development, Monitoring and Reporting of Stroke Rehabilitation Research: Consensus-Based Core Recommendations from the Stroke Recovery and Rehabilitation Roundtable. Neurorehabilit. Neural Repair 2017, 31, 877–884. [Google Scholar] [CrossRef]
- Chan, A.-W.; Tetzlaff, J.M.; Altman, D.G.; Laupacis, A.; Gøtzsche, P.C.; Krleža-Jerić, K.; Hróbjartsson, A.; Mann, H.; Dickersin, K.; Berlin, J.A.; et al. SPIRIT 2013 Statement: Defining Standard Protocol Items for Clinical Trials. Ann. Intern. Med. 2013, 158, 200–207. [Google Scholar] [CrossRef]
- Gjelsvik, B.; Breivik, K.; Verheyden, G.; Smedal, T.; Hofstad, H.; Strand, L.I. The Trunk Impairment Scale—Modified to ordinal scales in the Norwegian version. Disabil. Rehabil. 2011, 34, 1385–1395. [Google Scholar] [CrossRef][Green Version]
- Verheyden, G.; Nieuwboer, A.; Mertin, J.; Preger, R.; Kiekens, C.; Weerdt, W. De The Trunk Impairment Scale: A new tool to measure motor impairment of the trunk after stroke. Clin. Rehabil. 2004, 18, 326. [Google Scholar] [CrossRef]
- Lee, Y.; An, S.; Lee, G. Clinical utility of the modified trunk impairment scale for stroke survivors. Disabil. Rehabil. 2017, 40, 1200–1205. [Google Scholar] [CrossRef]
- Cabanas-Valdés, R.; Urrútia, G.; Bagur-Calafat, C.; Caballero-Gómez, F.M.; Germán-Romero, A.; Girabent-Farrés, M. Validation of the Spanish version of the Trunk Impairment Scale Version 2.0 (TIS 2.0) to assess dynamic sitting balance and coordination in post-stroke adult patients. Top. Stroke Rehabil. 2016, 23, 225–232. [Google Scholar] [CrossRef] [PubMed]
- Montaner, J.; Álvarez-Sabín, J. La escala de ictus del National Institute of Health (NIHSS) y su adaptación al español. Neurología 2006, 21, 192–202. [Google Scholar] [PubMed]
- Wilson, J.T.; Hareendran, A.; Grant, M.; Baird, T.; Schulz, U.G.; Muir, K.W.; Bone, I. Improving the assessment of outcomes in stroke: Use of a structured interview to assign grades on the modified Rankin Scale. Stroke 2002, 33, 2243–2246. [Google Scholar] [CrossRef] [PubMed]
- Sage, M.; Middleton, L.E.; Tang, A.; Sibley, K.M.; Brooks, D.; McIlroy, W. Validity of Rating of Perceived Exertion Ranges in Individuals in the Subacute Stage of Stroke Recovery. Top. Stroke Rehabil. 2013, 20, 519–527. [Google Scholar] [CrossRef]
- Pellegrino, L.; Giannoni, P.; Marinelli, L.; Casadio, M. Effects of continuous visual feedback during sitting balance training in chronic stroke survivors. J. Neuroeng. Rehabil. 2017, 14, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Guiu-Tula, F.X.; Cabanas-Valdés, R.; Sitjà-Rabert, M.; Urrútia, G.; Gómara-Toldrà, N. The Efficacy of the proprioceptive neuromuscular facilitation (PNF) approach in stroke rehabilitation to improve basic activities of daily living and quality of life: A systematic review and meta-analysis protocol. BMJ Open 2017, 7, e016739. [Google Scholar] [CrossRef]
- Mentiplay, B.F.; Clark, R.A.; Bower, K.J.; Williams, G.; Pua, Y.-H. Five times sit-to-stand following stroke: Relationship with strength and balance. Gait Posture 2020, 78, 35–39. [Google Scholar] [CrossRef] [PubMed]
- Hugues, A.; Di Marco, J.; Ribault, S.; Ardaillon, H.; Janiaud, P.; Xue, Y.; Zhu, J.; Pires, J.; Khademi, H.; Rubio, L.; et al. Limited evidence of physical therapy on balance after stroke: A systematic review and meta-analysis. PLoS ONE 2019, 14, e0221700. [Google Scholar] [CrossRef] [PubMed]
- Verheyden, G.; Kersten, P. Investigating the internal validity of the Trunk Impairment Scale (TIS) using Rasch analysis: The TIS 2. Disabil. Rehabil. 2010, 32, 2127–2137. [Google Scholar] [CrossRef]
- Tyson, S.F.; DeSouza, L.H. Reliability and validity of functional balance tests post stroke. Clin. Rehabil. 2004, 18, 916–923. [Google Scholar] [CrossRef] [PubMed]
- Cabanas-Valdés, R.; Bagur-Calafat, C.; Caballero-Gómez, F.M.; Cervera-Cuenca, C.; Moya-Valdés, R.; Rodríguez-Rubio, P.R.; Urrútia, G. Validation and reliability of the Spanish version of the Function in Sitting Test (S-FIST) to assess sitting balance in subacute post-stroke adult patients. Top. Stroke Rehabil. 2017, 24, 472–478. [Google Scholar] [CrossRef]
- Blum, L.; Korner-Bitensky, N. Usefulness of the Berg Balance Scale in Stroke Rehabilitation: A Systematic Review. Phys. Ther. 2008, 88, 559–566. [Google Scholar] [CrossRef]
- Berg, K. Measuring balance in the elderly: Preliminary development of an instrument. Physiother. Can. 1989, 41, 304–311. [Google Scholar] [CrossRef]
- Valdés, R.M.C.; Farrés, M.G.; Vergé, D.C.; Gómez, F.M.C.; Romero, A.G.; Calafat, C.B. Traducción y validación al español de la Postural Assessment Scale for Stroke Patients (PASS) para la valoración del equilibrio y del control postural en pacientes postictus. Rev. Neurol. 2015, 60, 151. [Google Scholar] [CrossRef]
- Ghotbi, N.; Ansari, N.N.; Naghdi, S.; Hasson, S. Measurement of lower-limb muscle spasticity: Intrarater reliability of Modified Modified Ashworth Scale. J. Rehabil. Res. Dev. 2011, 48, 83–88. [Google Scholar] [CrossRef]
- Mahoney, F.I.; Barthel, D.W. Functional Evaluation: The Barthel Index. Md. State Med. J. 1965, 14, 61–65. [Google Scholar]
- Golicki, D.; Niewada, M.; Buczek, J.; Karlińska, A.; Kobayashi, A.; Janssen, M.F.; Pickard, A.S. Validity of EQ-5D-5L in stroke. Qual. Life Res. 2015, 24, 845–850. [Google Scholar] [CrossRef] [PubMed]
- Hernandez, G.; Garin, O.; Pardo, Y.; Vilagut, G.; Pont, À.; Suárez, M.; Neira, M.; Rajmil, L.; Gorostiza, I.; Ramallo-Fariña, Y.; et al. Validity of the EQ–5D–5L and reference norms for the Spanish population. Qual. Life Res. 2018, 27, 2337–2348. [Google Scholar] [CrossRef] [PubMed]
- Halder, P.; Sterr, A.; Brem, S.; Bucher, K.; Kollias, S.; Brandeis, D. Electrophysiological evidence for cortical plasticity with movement repetition. Eur. J. Neurosci. 2005, 21, 2271–2277. [Google Scholar] [CrossRef] [PubMed]
- Kinoshita, K.; Ishida, K.; Hashimoto, M.; Nakao, H.; Shibanuma, N.; Kurosaka, M.; Otsuki, S. A vertical load applied towards the trunk unilaterally increases the bilateral abdominal muscle activities. J. Phys. Ther. Sci. 2019, 31, 273–276. [Google Scholar] [CrossRef]
- Tyson, S.; Watson, A.; Moss, S.; Troop, H.; Dean-Lofthouse, G.; Jorritsma, S.; Shannon, M.; On Behalf of the Greater Manchester Outcome Measures (GMOM) Project. Development of a framework for the evidence-based choice of outcome measures in neurological physiotherapy. Disabil. Rehabil. 2008, 30, 142–149. [Google Scholar] [CrossRef]
- Stevenson, T.J. Detecting change in patients with stroke using the Berg Balance Scale. Aust. J. Physiother. 2001, 47, 29–38. [Google Scholar] [CrossRef]
- Donoghue, D.; Stokes, E.K.; Physiotherapy Research and Older People (PROP) Group. How much change is true change? The minimum detectable change of the Berg Balance Scale in elderly people. J. Rehabil. Med. 2009, 41, 343–346. [Google Scholar] [CrossRef]
- Alzyoud, J.; Medley, A.; Thompson, M.; Csiza, L. Responsiveness, minimal detectable change, and minimal clinically important difference of the sitting balance scale and function in sitting test in people with stroke. Physiother. Theory Pract. 2020, 1–10. [Google Scholar] [CrossRef]
- Yoon, H.S.; Cha, Y.J.; You, J.; Sung, H. Effects of dynamic core-postural chain stabilization on diaphragm movement, abdominal muscle thickness, and postural control in patients with subacute stroke: A randomized control trial. Neurorehabilitation 2020, 46, 381–389. [Google Scholar] [CrossRef]
- Chien, C.-W.; Hu, M.-H.; Tang, P.-F.; Sheu, C.-F.; Hsieh, C.-L. A Comparison of Psychometric Properties of the Smart Balance Master System and the Postural Assessment Scale for Stroke in People Who Have Had Mild Stroke. Arch. Phys. Med. Rehabil. 2007, 88, 374–380. [Google Scholar] [CrossRef]
- Yoon, H.S.; Cha, Y.J.; You, J.; Sung, H. The effects of dynamic core-postural chain stabilization on respiratory function, fatigue and activities of daily living in subacute stroke patients: A randomized control trial. Neurorehabilitation 2020, 47, 471–477. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, Y.-W.; Wang, C.-H.; Wu, S.-C.; Chen, P.-C.; Sheu, C.-F.; Hsieh, C.-L. Establishing the Minimal Clinically Important Difference of the Barthel Index in Stroke Patients. Neurorehabilit. Neural Repair 2007, 21, 233–238. [Google Scholar] [CrossRef] [PubMed]
- Chen, P.; Lin, K.-C.; Liing, R.-J.; Wu, C.-Y.; Chen, C.-L.; Chang, K.-C. Validity, responsiveness, and minimal clinically important difference of EQ-5D-5L in stroke patients undergoing rehabilitation. Qual. Life Res. 2016, 25, 1585–1596. [Google Scholar] [CrossRef]
- Gamble, K.; Chiu, A.; Peiris, C. Core Stability Exercises in Addition to Usual Care Physiotherapy Improve Stability and Balance After Stroke: A Systematic Review and Meta-analysis. Arch. Phys. Med. Rehabil. 2021, 102, 762–775. [Google Scholar] [CrossRef] [PubMed]
- Van Kan, G.A.; Rolland, Y.; Andrieu, S.; Bauer, J.; Beauchet, O.; Bonnefoy, M.; Cesari, M.; Donini, L.M.; Gillette-Guyonnet, S.; Inzitari, M.; et al. Gait speed at usual pace as a predictor of adverse outcomes in community-dwelling older people an International Academy on Nutrition and Aging (IANA) Task Force. J. Nutr. Health Aging 2009, 13, 881–889. [Google Scholar] [CrossRef]
- Karthikbabu, S.; Chakrapani, M.; Ganesan, S.; Ellajosyula, R.; Solomon, J.M. Efficacy of Trunk Regimes on Balance, Mobility, Physical Function, and Community Reintegration in Chronic Stroke: A Parallel-Group Randomized Trial. J. Stroke Cerebrovasc. Dis. 2018, 27, 1003–1011. [Google Scholar] [CrossRef]
- Chen, X.; Gan, Z.; Tian, W.; Lv, Y. Effects of rehabilitation training of core muscle stability on stroke patients with hemiplegia. Pak. J. Med. Sci. 2020, 36, 461–466. [Google Scholar] [CrossRef]
- Selenitsch, N.A.; Gill, S.D. Stroke survivor activity during subacute inpatient rehabilitation: How active are patients? Int. J. Rehabil. Res. 2019, 42, 82–84. [Google Scholar] [CrossRef]
- Saumur, T.M.; Gregor, S.; Mochizuki, G.; Mansfield, A.; Mathur, S. The effect of bed rest on balance control in healthy adults: A systematic scoping review. J. Musculoskelet. Neuronal Interact 2020, 20, 101–113. [Google Scholar] [PubMed]
- Van Criekinge, T.; Hallemans, A.; Herssens, N.; Lafosse, C.; Claes, D.; De Hertogh, W.; Truijen, S.; Saeys, W. SWEAT2 Study: Effectiveness of Trunk Training on Gait and Trunk Kinematics After Stroke: A Randomized Controlled Trial. Phys. Ther. 2020, 100, 1568–1581. [Google Scholar] [CrossRef] [PubMed]
- Karthikbabu, S.; Verheyden, G. Relationship between trunk control, core muscle strength and balance confidence in community-dwelling patients with chronic stroke. Top. Stroke Rehabil. 2021, 28, 88–95. [Google Scholar] [CrossRef] [PubMed]



Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cabanas-Valdés, R.; Boix-Sala, L.; Grau-Pellicer, M.; Guzmán-Bernal, J.A.; Caballero-Gómez, F.M.; Urrútia, G. The Effectiveness of Additional Core Stability Exercises in Improving Dynamic Sitting Balance, Gait and Functional Rehabilitation for Subacute Stroke Patients (CORE-Trial): Study Protocol for a Randomized Controlled Trial. Int. J. Environ. Res. Public Health 2021, 18, 6615. https://doi.org/10.3390/ijerph18126615
Cabanas-Valdés R, Boix-Sala L, Grau-Pellicer M, Guzmán-Bernal JA, Caballero-Gómez FM, Urrútia G. The Effectiveness of Additional Core Stability Exercises in Improving Dynamic Sitting Balance, Gait and Functional Rehabilitation for Subacute Stroke Patients (CORE-Trial): Study Protocol for a Randomized Controlled Trial. International Journal of Environmental Research and Public Health. 2021; 18(12):6615. https://doi.org/10.3390/ijerph18126615
Chicago/Turabian StyleCabanas-Valdés, Rosa, Lídia Boix-Sala, Montserrat Grau-Pellicer, Juan Antonio Guzmán-Bernal, Fernanda Maria Caballero-Gómez, and Gerard Urrútia. 2021. "The Effectiveness of Additional Core Stability Exercises in Improving Dynamic Sitting Balance, Gait and Functional Rehabilitation for Subacute Stroke Patients (CORE-Trial): Study Protocol for a Randomized Controlled Trial" International Journal of Environmental Research and Public Health 18, no. 12: 6615. https://doi.org/10.3390/ijerph18126615
APA StyleCabanas-Valdés, R., Boix-Sala, L., Grau-Pellicer, M., Guzmán-Bernal, J. A., Caballero-Gómez, F. M., & Urrútia, G. (2021). The Effectiveness of Additional Core Stability Exercises in Improving Dynamic Sitting Balance, Gait and Functional Rehabilitation for Subacute Stroke Patients (CORE-Trial): Study Protocol for a Randomized Controlled Trial. International Journal of Environmental Research and Public Health, 18(12), 6615. https://doi.org/10.3390/ijerph18126615





