Potential Improvement in Rehabilitation Quality of 2019 Novel Coronavirus by Isometric Training System; Is There “Muscle-Lung Cross-Talk”?
Abstract
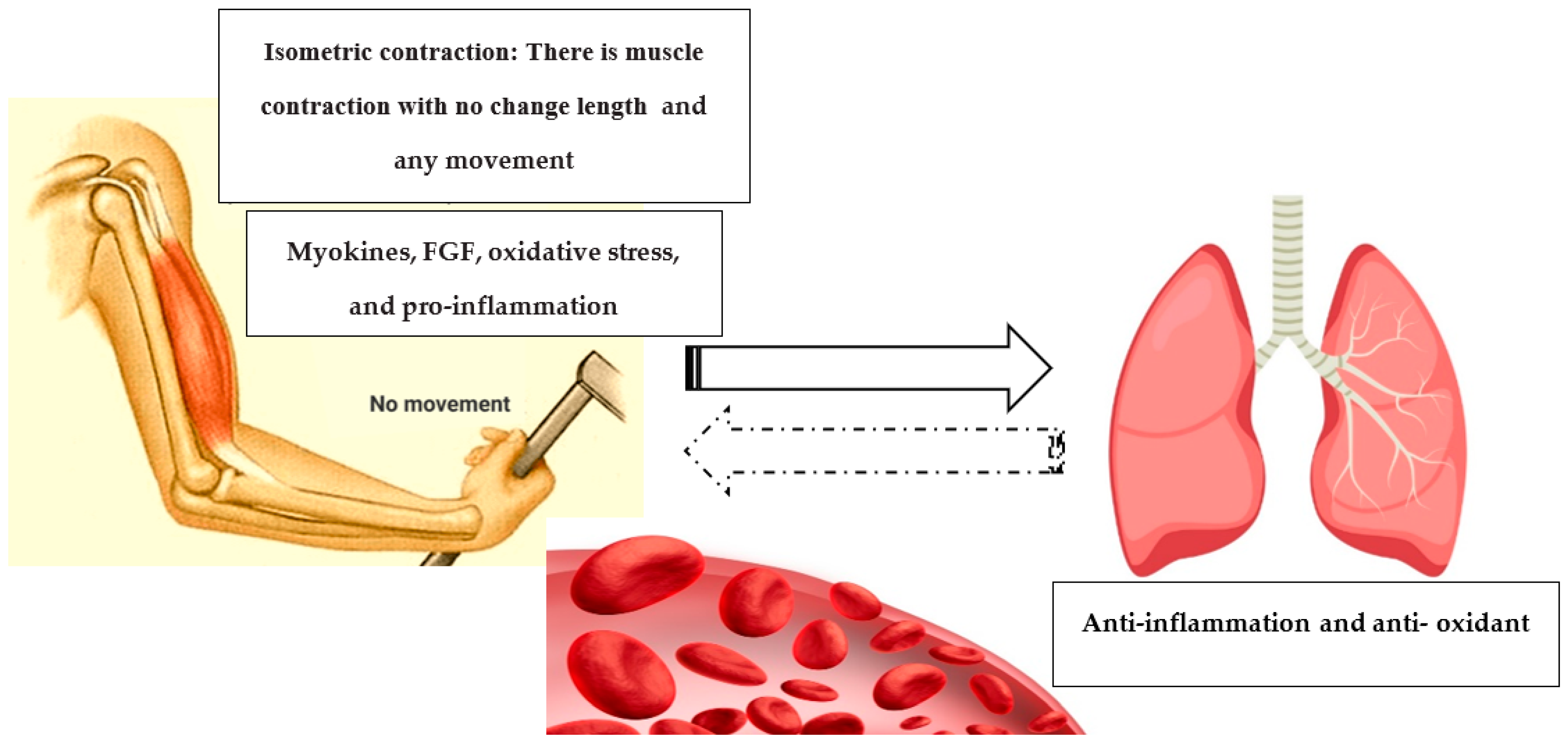
:Dear Editor
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Rothan, H.A.; Byrareddy, S.N. The epidemiology and pathogenesis of coronavirus disease (COVID-19) outbreak. J. Autoimmun. 2020, 109, 102433. [Google Scholar] [CrossRef]
- Zinovkin, R.A.; Grebenchikov, O.A. Transcription Factor Nrf2 as a Potential Therapeutic Target for Prevention of Cytokine Storm in COVID-19 Patients. Biochemistry 2020, 85, 833–837. [Google Scholar] [CrossRef]
- Violi, F.; Oliva, A.; Cangemi, R.; Ceccarelli, G.; Pignatelli, P.; Carnevale, R.; Cammisotto, V.; Lichtner, M.; Alessandri, F.; De Angelis, M.; et al. Nox2 activation in Covid-19. Redox Biol. 2020, 36, 101655. [Google Scholar] [CrossRef]
- Nobari, H.; Kargarfard, M.; Minasian, V.; Cholewa, J.M.; Pérez-Gómez, J. The effects of 14-week betaine supplementation on endocrine markers, body composition and anthropometrics in professional youth soccer players: A double blind, randomized, placebo-controlled trial. J. Int. Soc. Sports Nutr. 2021, 18, 20. [Google Scholar] [CrossRef]
- Delgado-Roche, L.; Mesta, F. Oxidative stress as key player in severe acute respiratory syndrome coronavirus (SARS-CoV) infection. Arch. Med. Res. 2020, 51, 384–387. [Google Scholar] [CrossRef]
- Nobari, H.; Nejad, H.A.; Kargarfard, M.; Mohseni, S.; Suzuki, K.; Carmelo Adsuar, J.; Pérez-Gómez, J. The Effect of Acute Intense Exercise on Activity of Antioxidant Enzymes in Smokers and Non-smokers. Biomolecules 2021, 11, 171. [Google Scholar] [CrossRef]
- Palomino, D.C.T.; Marti, L.C. Chemokines and immunity. Einstein 2015, 13, 469–473. [Google Scholar] [CrossRef] [Green Version]
- Silva, J.R.; Rumpf, M.C.; Hertzog, M.; Castagna, C.; Farooq, A.; Girard, O.; Hader, K. Acute and Residual Soccer Match-Related Fatigue: A Systematic Review and Meta-analysis. Sports Med. 2018, 48, 539–583. [Google Scholar] [CrossRef]
- Lu, Y.; Mak, K.-K.; Van Bever, H.P.S.; Ng, T.P.; Mak, A.; Ho, R.C.-M. Prevalence of anxiety and depressive symptoms in adolescents with asthma: A meta-analysis and meta-regression. Pediatr. Allergy Immunol. 2012, 23, 707–715. [Google Scholar] [CrossRef]
- Samadi, M.; Shirvani, H.; Rahmati-Ahmadabad, S. A study of possible role of exercise and some antioxidant supplements against coronavirus disease 2019 (COVID-19): A cytokines related perspective. Apunt. Sports Med. 2020, 55, 115–117. [Google Scholar] [CrossRef]
- Ruhee, R.T.; Suzuki, K. The Integrative Role of Sulforaphane in Preventing Inflammation, Oxidative Stress and Fatigue: A Review of a Potential Protective Phytochemical. Antioxidants 2020, 9, 521. [Google Scholar] [CrossRef] [PubMed]
- Vargas-Mendoza, N.; Morales-González, Á.; Madrigal-Santillán, E.O.; Madrigal-Bujaidar, E.; Álvarez-González, I.; García-Melo, L.F.; Anguiano-Robledo, L.; Fregoso-Aguilar, T.; Morales-Gonzalez, J.A. Antioxidant and Adaptative Response Mediated by Nrf2 during Physical Exercise. Antioxidants 2019, 8, 196. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fashi, M.; Agha-Alinejad, H.; Mahabadi, H.A.; Rezaei, B.; Pakrad, B.B.; Rezaei, S. The effects of aerobic exercise on NF-κB and TNF-α in lung tissue of male rat. Nov. Biomed. 2015, 3, 131–134. [Google Scholar]
- Wang, P.; Li, C.G.; Qi, Z.; Cui, D.; Ding, S. Acute exercise stress promotes Ref1/Nrf2 signalling and increases mitochondrial antioxidant activity in skeletal muscle. Exp. Physiol. 2016, 101, 410–420. [Google Scholar] [CrossRef]
- Suzuki, K.; Hayashida, H. Effect of Exercise Intensity on Cell-Mediated Immunity. Sports 2021, 9, 8. [Google Scholar] [CrossRef] [PubMed]
- Nobari, H.; Fashi, M.; Eskandari, A.; Villafaina, S.; Murillo-Garcia, Á.; Pérez-Gómez, J. Effect of COVID-19 on Health-Related Quality of Life in Adolescents and Children: A Systematic Review. Int. J. Environ. Res. Public Health 2021, 18, 4563. [Google Scholar] [CrossRef]
- Pescatello, L.S.; Riebe, D.; Thompson, P.D. ACSM’s Guidelines for Exercise Testing and Prescription; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2014. [Google Scholar]
- Mehrabani, J.; Sedighianrad, M.; Ramezani, N.; Nobari, H.; Bagheri, R. Effect of endurance training on plasma levels of AGRP and HOMA-IR in diabetic rats. Turk. J. Kinesiol. 2017, 3, 43–48. [Google Scholar]
- Zhu, W. Should, and how can, exercise be done during a coronavirus outbreak? An interview with Dr. Jeffrey A. Woods. J. Sport Health Sci. 2020, 9, 105–107. [Google Scholar] [CrossRef]
- Khoramipour, K.; Basereh, A.; Hekmatikar, A.A.; Castell, L.; Ruhee, R.T.; Suzuki, K. Physical activity and nutrition guidelines to help with the fight against COVID-19. J. Sports Sci. 2021, 39, 101–107. [Google Scholar] [CrossRef] [PubMed]
- Oranchuk, D.J.; Storey, A.G.; Nelson, A.R.; Cronin, J.B. Isometric training and long-term adaptations: Effects of muscle length, intensity, and intent: A systematic review. Scand. J. Med. Sci. Sports 2018, 29, 484–503. [Google Scholar] [CrossRef]
- Fashi, M.; Ahmadizad, S.; Nobari, H.; Pérez-Gómez, J.; Oliveira, R.; Carlos-Vivas, J.; Ardigò, L. Effect of Acute Ramadan Fasting on Muscle Function and Buffering System of Male Athletes. Healthcare 2021, 9, 397. [Google Scholar] [CrossRef] [PubMed]
- Aghababa, A.; Badicu, G.; Fathirezaie, Z.; Rohani, H.; Nabilpour, M.; Sani, S.Z.; Khodadadeh, E. Different Effects of the COVID-19 Pandemic on Exercise Indexes and Mood States Based on Sport Types, Exercise Dependency and Individual Characteristics. Children 2021, 8, 438. [Google Scholar] [CrossRef] [PubMed]
- Garatachea, N.; García-López, D.; Bernal, A.; Almar, M.; Gonzalez-Gallego, J. Oxidative stress response to isometric exercise in women: Effect of age and exercise intensity. Int. Sportmed. J. 2012, 13, 85–95. [Google Scholar]
- Pasini, A.F.; Stranieri, C.; Cominacini, L.; Mozzini, C. Potential Role of Antioxidant and Anti-Inflammatory Therapies to Prevent Severe SARS-Cov-2 Complications. Antioxidants 2021, 10, 272. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, K.; Tominaga, T.; Ruhee, R.T.; Ma, S. Characterization and Modulation of Systemic Inflammatory Response to Exhaustive Exercise in Relation to Oxidative Stress. Antioxidants 2020, 9, 401. [Google Scholar] [CrossRef]
- Nobari, H.; Cholewa, J.M.; Pérez-Gómez, J.; Castillo-Rodríguez, A. Effects of 14-weeks betaine supplementation on pro-inflammatory cytokines and hematology status in professional youth soccer players during a competition season: A double blind, randomized, placebo-controlled trial. J. Int. Soc. Sports Nutr. 2021, 18, 42. [Google Scholar] [CrossRef] [PubMed]
- Sugiyama, Y.; Asai, K.; Yamada, K.; Kureya, Y.; Ijiri, N.; Watanabe, T.; Kanazawa, H.; Hirata, K. Decreased levels of irisin, a skeletal muscle cell-derived myokine, are related to emphysema associated with chronic obstructive pulmonary disease. Int. J. Chronic Obstr. Pulm. Dis. 2017, 12, 765–772. [Google Scholar] [CrossRef] [Green Version]
- Khalafi, M.; Alamdari, K.A.; Symonds, M.E.; Nobari, H.; Carlos-Vivas, J. Impact of acute exercise on immediate and following early post-exercise FGF-21 concentration in adults: Systematic review and meta-analysis. Hormones 2021, 20, 23–33. [Google Scholar] [CrossRef]
- Cuevas-Ramos, D.; Mehta, R.; Aguilar-Salinas, C.A. Fibroblast Growth Factor 21 and Browning of White Adipose Tissue. Front. Physiol. 2019, 10, 37. [Google Scholar] [CrossRef] [Green Version]
- Hondermarck, H.; Bartlett, N.W.; Nurcombe, V. The role of growth factor receptors in viral infections: An opportunity for drug repurposing against emerging viral diseases such as COVID-19? FASEB BioAdv. 2020, 2, 296–303. [Google Scholar] [CrossRef]
- Grigoletto, I.; Cavalheri, V.; de Lima, F.F.; Ramos, E.M.C. Recovery after COVID-19: The potential role of pulmonary rehabilitation. Braz. J. Phys. Ther. 2020, 24, 463–464. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nobari, H.; Fashi, M.; Eskandari, A.; Pérez-Gómez, J.; Suzuki, K. Potential Improvement in Rehabilitation Quality of 2019 Novel Coronavirus by Isometric Training System; Is There “Muscle-Lung Cross-Talk”? Int. J. Environ. Res. Public Health 2021, 18, 6304. https://doi.org/10.3390/ijerph18126304
Nobari H, Fashi M, Eskandari A, Pérez-Gómez J, Suzuki K. Potential Improvement in Rehabilitation Quality of 2019 Novel Coronavirus by Isometric Training System; Is There “Muscle-Lung Cross-Talk”? International Journal of Environmental Research and Public Health. 2021; 18(12):6304. https://doi.org/10.3390/ijerph18126304
Chicago/Turabian StyleNobari, Hadi, Mohamad Fashi, Arezoo Eskandari, Jorge Pérez-Gómez, and Katsuhiko Suzuki. 2021. "Potential Improvement in Rehabilitation Quality of 2019 Novel Coronavirus by Isometric Training System; Is There “Muscle-Lung Cross-Talk”?" International Journal of Environmental Research and Public Health 18, no. 12: 6304. https://doi.org/10.3390/ijerph18126304
APA StyleNobari, H., Fashi, M., Eskandari, A., Pérez-Gómez, J., & Suzuki, K. (2021). Potential Improvement in Rehabilitation Quality of 2019 Novel Coronavirus by Isometric Training System; Is There “Muscle-Lung Cross-Talk”? International Journal of Environmental Research and Public Health, 18(12), 6304. https://doi.org/10.3390/ijerph18126304








