Hemodynamic Response to Three Types of Urban Spaces before and after Lockdown during the COVID-19 Pandemic
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
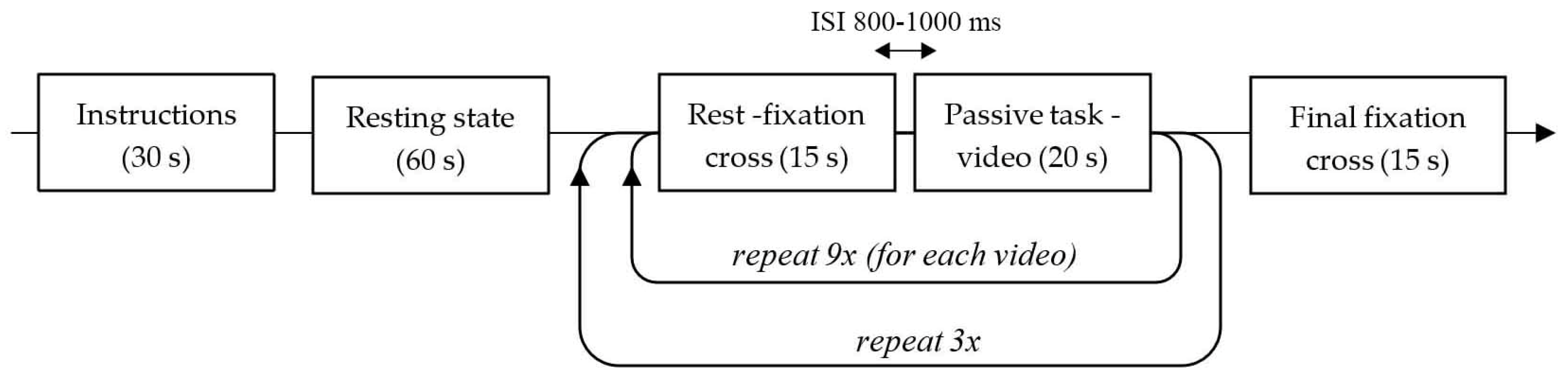
2.2. Study Protocol and Setup
2.3. Stimuli
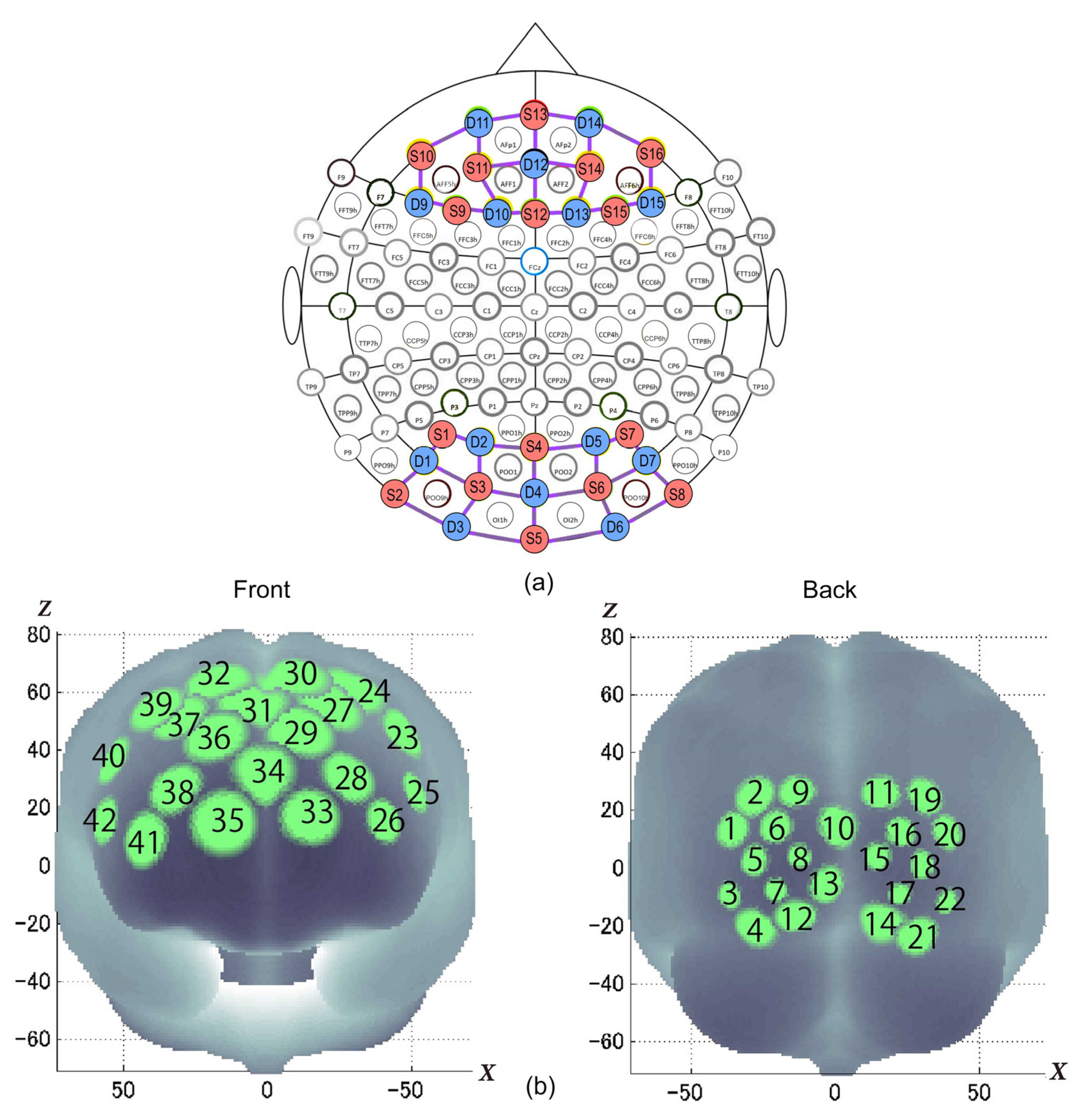
2.4. Measurement of Brain Activity
2.5. Data Processing and Analysis
3. Results
fNIRS Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Pasion, R.; Paiva, T.O.; Fernandes, C.; Barbosa, F. The AGE effect on protective behaviors during the COVID-19 outbreak: Sociodemographic, perceptions and psychological accounts. Front. Psychol. 2020, 11, 2785. [Google Scholar] [CrossRef] [PubMed]
- Ammar, A.; Chtourou, H.; Boukhris, O.; Trabelsi, K.; Masmoudi, L.; Brach, M.; Bouaziz, B.; Bentlage, E.; How, D.; Ahmed, M. Covid-19 home confinement negatively impacts social participation and life satisfaction: A worldwide multicenter study. Int. J. Environ. Res. Public Health 2020, 17, 6237. [Google Scholar] [CrossRef]
- Flanagan, E.W.; Beyl, R.A.; Fearnbach, S.N.; Altazan, A.D.; Martin, C.K.; Redman, L.M. The impact of COVID-19 stay-at-home orders on health behaviors in adults. Obesity 2021, 29, 438–445. [Google Scholar] [CrossRef] [PubMed]
- Alirol, E.; Getaz, L.; Stoll, B.; Chappuis, F.; Loutan, L. Urbanisation and infectious diseases in a globalised world. Lancet Infect. Dis. 2011, 11, 131–141. [Google Scholar] [CrossRef]
- Lai, C.Y.Y.; Ho, C.S.H.; Lim, C.R.; Ho, R.C.M. Functional near-infrared spectroscopy in psychiatry. BJPsych Adv. 2017, 23, 324–330. [Google Scholar] [CrossRef]
- Scholkmann, F.; Kleiser, S.; Metz, A.J.; Zimmermann, R.; Pavia, J.M.; Wolf, U.; Wolf, M. A review on continuous wave functional near-infrared spectroscopy and imaging instrumentation and methodology. Neuroimage 2014, 85, 6–27. [Google Scholar] [CrossRef]
- Ho, C.S.H.; Lim, L.J.H.; Lim, A.Q.; Chan, N.H.C.; Tan, R.S.; Lee, S.H.; Ho, R. Diagnostic and predictive applications of functional near-infrared spectroscopy for major depressive disorder: A systematic review. Front. Psychiatry 2020, 11, 378. [Google Scholar] [CrossRef]
- Khoe, H.C.H.; Low, J.W.; Wijerathne, S.; Ann, L.S.; Salgaonkar, H.; Lomanto, D.; Choi, J.; Baek, J.; Tam, W.W.; Pei, H.; et al. Use of prefrontal cortex activity as a measure of learning curve in surgical novices: Results of a single blind randomised controlled trial. Surg. Endosc. 2020, 34, 5604–5615. [Google Scholar] [CrossRef]
- Mangun, G.R.; Hopfinger, J.B.; Kussmaul, C.L.; Fletcher, E.M.; Heinze, H.J. Covariations in ERP and PET measures of spatial selective attention in human extrastriate visual cortex. Hum. Brain Mapp. 1997, 5, 273–279. [Google Scholar] [CrossRef]
- Heinze, H.J.; Mangun, G.R.; Burchert, W.; Hinrichs, H.; Scholz, M.; Münte, T.F.; Gös, A.; Scherg, M.; Johannes, S.; Hundeshagen, H.; et al. Combined spatial and temporal imaging of brain activity during visual selective attention in humans. Nature 1994, 372, 543–546. [Google Scholar] [CrossRef]
- Bratman, G.N.; Hamilton, J.P.; Daily, G.C. The impacts of nature experience on human cognitive function and mental health. Ann. N. Y. Acad. Sci. 2012, 1249, 118–136. [Google Scholar] [CrossRef]
- Herman, K.; Ciechanowski, L.; Przegalińska, A. Emotional well-being in urban wilderness: Assessing states of calmness and alertness in informal green spaces (IGSs) with muse—Portable EEG headband. Sustainability 2021, 13, 2212. [Google Scholar] [CrossRef]
- Lee, J. Experimental study on the health benefits of garden landscape. Int. J. Environ. Res. Public Health 2017, 14, 829. [Google Scholar] [CrossRef] [PubMed]
- Olszewska-Guizzo, A.; Sia, A.; Fogel, A.; Ho, R. Can exposure to certain urban green spaces trigger frontal alpha asymmetry in the brain?—Preliminary findings from a passive task EEG study. Int. J. Environ. Res. Public Health 2020, 17, 394. [Google Scholar] [CrossRef] [PubMed]
- Hartig, T.; Mang, M.; Evans, G.W. Restorative effects of natural environment experiences. Environ. Behav. 1991, 23, 3–26. [Google Scholar] [CrossRef]
- Berman, M.G.; Jonides, J.; Kaplan, S. The cognitive benefits of interacting with nature. Psychol. Sci. 2008, 19, 1207–1212. [Google Scholar] [CrossRef]
- Kaplan, S. Meditation, restoration, and the management of mental fatigue. Environ. Behav. 2001, 33, 480–506. [Google Scholar] [CrossRef]
- Olszewska-Guizzo, A.; Fogel, A.; Escoffier, N.; Ho, R. Effects of COVID-19-related stay-at-home order on neuropsychophysiological response to urban spaces: Beneficial role of exposure to nature? J. Environ. Psychol. 2021, 75, 101590. [Google Scholar] [CrossRef]
- Moh Steps up Precautionary Measures in Response to Increase in Cases of Novel Coronavirus Pneumonia in Wuhan. Available online: https://www.moh.gov.sg/news-highlights/details/moh-steps-up-precautionary-measures-in-response-to-increase-in-cases-of-novel-coronavirus-pneumonia-in-wuhan (accessed on 6 May 2020).
- Hart, B.M.; Schmidt, H.C.E.F.; Klein-Harmeyer, I.; Einhäuser, W. Attention in natural scenes: Contrast affects rapid visual processing and fixations alike. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2013, 368, 20130067. [Google Scholar] [CrossRef]
- Tsuzuki, D.; Jurcak, V.; Singh, A.K.; Okamoto, M.; Watanabe, E.; Dan, I. Virtual spatial registration of stand-alone fNIRS data to MNI space. NeuroImage 2007, 34, 1506–1518. [Google Scholar] [CrossRef]
- Tsuzuki, D.; Cai, D.S.; Dan, H.; Kyutoku, Y.; Fujita, A.; Watanabe, E.; Dan, I. Stable and convenient spatial registration of stand-alone NIRS data through anchor-based probabilistic registration. Neurosci. Res. 2012, 72, 163–171. [Google Scholar] [CrossRef]
- Tsuzuki, D.; Dan, I. Spatial registration for functional near-infrared spectroscopy: From channel position on the scalp to cortical location in individual and group analyses. NeuroImage 2014, 85, 92–103. [Google Scholar] [CrossRef] [PubMed]
- Rorden, C.; Brett, M. Stereotaxic display of brain lesions. Behav. Neurol. 2000, 12, 191–200. [Google Scholar] [CrossRef] [PubMed]
- Huppert, T.J.; Diamond, S.G.; Franceschini, M.A.; Boas, D.A. HomER: A review of time-series analysis methods for near-infrared spectroscopy of the brain. Appl. Opt. 2009, 48, D280–298. [Google Scholar] [CrossRef]
- Uga, M.; Dan, I.; Sano, T.; Dan, H.; Watanabe, E. Optimizing the general linear model for functional near-infrared spectroscopy: An adaptive hemodynamic response function approach. Neurophotonics 2014, 1, 015004. [Google Scholar] [CrossRef]
- Hoshi, Y.; Kobayashi, N.; Tamura, M. Interpretation of near-infrared spectroscopy signals: A study with a newly developed perfused rat brain model. J. Appl. Physiol. 2001, 90, 1657–1662. [Google Scholar] [CrossRef]
- Strangman, G.; Culver, J.P.; Thompson, J.H.; Boas, D.A. A quantitative comparison of simultaneous BOLD fMRI and NIRS recordings during functional brain activation. NeuroImage 2002, 17, 719–731. [Google Scholar] [CrossRef] [PubMed]
- Hoshi, Y. Functional near-infrared optical imaging: Utility and limitations in human brain mapping. Psychophysiology 2003, 40, 511–520. [Google Scholar] [CrossRef] [PubMed]
- Plichta, M.M.; Herrmann, M.J.; Baehne, C.G.; Ehlis, A.C.; Richter, M.M.; Pauli, P.; Fallgatter, A.J. Event-related functional near-infrared spectroscopy (fNIRS): Are the measurements reliable? NeuroImage 2006, 31, 116–124. [Google Scholar] [CrossRef]
- Jang, K.E.; Tak, S.; Jung, J.; Jang, J.; Jeong, Y.; Ye, J.C. Wavelet minimum description length detrending for near-infrared spectroscopy. J. Biomed. Opt. 2009, 14, 034004. [Google Scholar] [CrossRef] [PubMed]
- Friston, K.J.; Fletcher, P.; Josephs, O.; Holmes, A.; Rugg, M.D.; Turner, R. Event-related fMRI: Characterizing differential responses. NeuroImage 1998, 7, 30–40. [Google Scholar] [CrossRef]
- Uga, M.; Dan, I.; Dan, H.; Kyutoku, Y.; Taguchi, Y.H.; Watanabe, E. Exploring effective multiplicity in multichannel functional near-infrared spectroscopy using eigenvalues of correlation matrices. Neurophotonics 2015, 2, 015002. [Google Scholar] [CrossRef][Green Version]
- American Psychological Association. Publication Manual of the American Psychological Association; APA: Washington DC, USA, 1994. [Google Scholar]
- Olsson-Collentine, A.; Van Assen, M.A.L.M.; Hartgerink, C.H.J. The prevalence of marginally significant results in psychology over time. Psychol. Sci. 2019, 30, 576–586. [Google Scholar] [CrossRef]
- Fink, G.R.; Marshall, J.C.; Halligan, P.W.; Frith, C.D.; Frackowiak, R.S.J.; Dolan, R.J. Hemispheric specialization for global and local processing: The effect of stimulus category. In Proceedings of the Royal Society B: Biological Sciences; The Royal Society: London, UK, 1997; Volume 264, pp. 487–494. [Google Scholar]
- Olszewska-Guizzo, A.A.; Paiva, T.O.; Barbosa, F. Effects of 3D contemplative landscape videos on brain activity in a passive exposure EEG experiment. Front. Psychiatry 2018, 9, 317. [Google Scholar] [CrossRef]
- Wang, C.; Chudzicka-Czupała, A.; Grabowski, D.; Pan, R.; Adamus, K.; Wan, X.; Hetnał, M.; Tan, Y.; Olszewska-Guizzo, A.; Xu, L. The association between physical and mental health and face mask use during the COVID-19 pandemic: A comparison of two countries with different views and practices. Front. Psychiatry 2020, 11, 901. [Google Scholar] [CrossRef]
- Husain, S.F.; Yu, R.; Tang, T.-B.; Tam, W.W.; Tran, B.; Quek, T.T.; Hwang, S.-H.; Chang, C.W.; Ho, C.S.; Ho, R.C. Validating a functional near-infrared spectroscopy diagnostic paradigm for major depressive disorder. Sci. Rep. 2020, 10, 1–9. [Google Scholar] [CrossRef]
- Ward, L.M.; Aitchison, R.T.; Tawse, M.; Simmers, A.J.; Shahani, U. Reduced haemodynamic response in the ageing visual cortex measured by absolute fNIRS. PLoS ONE 2015, 10, e0125012. [Google Scholar] [CrossRef] [PubMed]
- Bakker, S.L.M.; de Leeuw, F.-E.; den Heijer, T.; Koudstaal, P.J.; Hofman, A.; Breteler, M.M.B. Cerebral haemodynamics in the elderly: The rotterdam study. Neuroepidemiology 2004, 23, 178–184. [Google Scholar] [CrossRef] [PubMed]
- Duric, V.; Duman, R.S. Depression and treatment response: Dynamic interplay of signaling pathways and altered neural processes. Cell. Mol. Life Sci. 2013, 70, 39–53. [Google Scholar] [CrossRef]
- Davidson, R.J.; Ekman, P.; Saron, C.D.; Senulis, J.A.; Friesen, W.V. Approach-withdrawal and cerebral asymmetry: Emotional expression and brain physiology: I. J. Personal. Soc. Psychol. 1990, 58, 330. [Google Scholar] [CrossRef]
- Hofer, S.M.; Berg, S.; Era, P. Evaluating the interdependence of aging-related changes in visual and auditory acuity, balance, and cognitive functioning. Psychol. Aging 2003, 18, 285. [Google Scholar] [CrossRef]
- Schmidt, S.C.E.; Anedda, B.; Burchartz, A.; Eichsteller, A.; Kolb, S.; Nigg, C.; Niessner, C.; Oriwol, D.; Worth, A.; Woll, A. Physical activity and screen time of children and adolescents before and during the COVID-19 lockdown in Germany: A natural experiment. Sci. Rep. 2020, 10, 1–12. [Google Scholar] [CrossRef]
- Wong, C.W.; Tsai, A.; Jonas, J.B.; Ohno-Matsui, K.; Chen, J.; Ang, M.; Ting, D.S.W. Digital screen time during the COVID-19 pandemic: Risk for a further myopia boom? Am. J. Ophthalmol. 2021, 223, 333. [Google Scholar] [CrossRef] [PubMed]
- Castañeda-Babarro, A.; Arbillaga-Etxarri, A.; Gutiérrez-Santamaría, B.; Coca, A. Physical activity change during COVID-19 confinement. Int. J. Environ. Res. Public Health 2020, 17, 6878. [Google Scholar] [CrossRef] [PubMed]
- Bourdas, D.I.; Zacharakis, E.D. Evolution of changes in physical activity over lockdown time: Physical activity datasets of four independent adult sample groups corresponding to each of the last four of the six COVID-19 lockdown weeks in Greece. Data Brief 2020, 32, 106301. [Google Scholar] [CrossRef]
- Iwasaki, T.; Tawara, A.; Miyake, N. Reduction of asthenopia related to accommodative relaxation by means of far point stimuli. Acta Ophthalmol. Scand. 2005, 83, 81–88. [Google Scholar] [CrossRef]
- Reddy, S.C.; Low, C.K.; Lim, Y.P.; Low, L.L.; Mardina, F.; Nursaleha, M.P. Computer vision syndrome: A study of knowledge and practices in university students. Nepal. J. Ophthalmol. 2013, 5, 161–168. [Google Scholar] [CrossRef]
- Joung, D.; Kim, G.; Choi, Y.; Lim, H.; Park, S.; Woo, J.-M.; Park, B.-J. The prefrontal cortex activity and psychological effects of viewing forest landscapes in autumn season. Int. J. Environ. Res. Public Health 2015, 12, 7235–7243. [Google Scholar] [CrossRef] [PubMed]
- Kassab, A.; Le Lan, J.; Tremblay, J.; Vannasing, P.; Dehbozorgi, M.; Pouliot, P.; Gallagher, A.; Lesage, F.; Sawan, M.; Nguyen, D.K. Multichannel wearable f NIRS-EEG system for long-term clinical monitoring. Hum. Brain Mapp. 2018, 39, 7–23. [Google Scholar] [CrossRef]

| Channel | x | y | z | SD | Anatomy | MAL(%) |
|---|---|---|---|---|---|---|
| 9 | −13.3 | −100.0 | 26.3 | 7.2 | BA18-Visual Association Cortex (V2), left | 74.8 |
| BA17-Primary Visual Cortex (V1), left | 25.2 | |||||
| 10 | 0.3 | −101.3 | 14.3 | 8.6 | BA17-Primary Visual Cortex (V1), left | 84.8 |
| BA18-Visual Association Cortex (V2), left | 15.2 | |||||
| 19 | 30.7 | −94.0 | 24.7 | 7.7 | BA18-Visual Association Cortex (V2), right | 74.7 |
| BA19-Visual Association Cortex (V3), right | 25.3 | |||||
| 27 | −21.3 | 36.3 | 55.3 | 13.6 | BA8-Frontal eye fields, left | 58.1 |
| BA9-Dorsolateral prefrontal cortex, left | 41.9 |
| Channel | Mean (mM·mm) | SD | t | p | Sig | |
|---|---|---|---|---|---|---|
| Site 1 Urban Park | 9 | −0.225 | 0.223 | −3.50 | 0.0471 | * |
| 19 | −0.164 | 0.178 | −3.19 | 0.0814 | † | |
| Site 2 Neighborhood Landscape | 9 | −0.286 | 0.287 | −3.44 | 0.0503 | † |
| 19 | −0.207 | 0.223 | −3.22 | 0.0757 | † | |
| Site 3 City Center | 10 | −0.365 | 0.388 | −3.26 | 0.0708 | † |
| 27 | −0.207 | 0.226 | −3.16 | 0.0843 | † |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Olszewska-Guizzo, A.; Mukoyama, A.; Naganawa, S.; Dan, I.; Husain, S.F.; Ho, C.S.; Ho, R. Hemodynamic Response to Three Types of Urban Spaces before and after Lockdown during the COVID-19 Pandemic. Int. J. Environ. Res. Public Health 2021, 18, 6118. https://doi.org/10.3390/ijerph18116118
Olszewska-Guizzo A, Mukoyama A, Naganawa S, Dan I, Husain SF, Ho CS, Ho R. Hemodynamic Response to Three Types of Urban Spaces before and after Lockdown during the COVID-19 Pandemic. International Journal of Environmental Research and Public Health. 2021; 18(11):6118. https://doi.org/10.3390/ijerph18116118
Chicago/Turabian StyleOlszewska-Guizzo, Agnieszka, Ayako Mukoyama, Sho Naganawa, Ippeita Dan, Syeda Fabeha Husain, Cyrus S. Ho, and Roger Ho. 2021. "Hemodynamic Response to Three Types of Urban Spaces before and after Lockdown during the COVID-19 Pandemic" International Journal of Environmental Research and Public Health 18, no. 11: 6118. https://doi.org/10.3390/ijerph18116118
APA StyleOlszewska-Guizzo, A., Mukoyama, A., Naganawa, S., Dan, I., Husain, S. F., Ho, C. S., & Ho, R. (2021). Hemodynamic Response to Three Types of Urban Spaces before and after Lockdown during the COVID-19 Pandemic. International Journal of Environmental Research and Public Health, 18(11), 6118. https://doi.org/10.3390/ijerph18116118








