Influence of a Training Academy on the Parasympathetic Nervous System Reactivation of Firefighter Recruits—An Observational Cohort Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Study Design
Firefighter Academy Training Program
2.3. Procedures
2.3.1. Body Composition Data
2.3.2. Heart Rate Data
2.4. Statistical Analyses
3. Results
4. Discussion
Strengths and Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Smith, D.L.; Barr, A.D.; Kales, S.N. Extreme sacrifice: Sudden cardiac death in the US Fire Service. Extreme Physiol. Med. 2013, 2, 6. [Google Scholar] [CrossRef] [PubMed]
- Smith, D.L.; Deblois, J.P.; Kales, S.N.; Horn, G.P. Cardiovascular Strain of Firefighting and the Risk of Sudden Cardiac Events. Exerc. Sport Sci. Rev. 2016, 44, 90–97. [Google Scholar] [CrossRef] [PubMed]
- Sothmann, M.S.; Saupe, K.; Jasenof, D.; Blaney, J. Heart Rate Response of Firefighters to Actual Emergencies. J. Occup. Environ. Med. 1992, 34, 797–800. [Google Scholar] [CrossRef] [PubMed]
- Bugajska, J.; Zuzewicz, K.; Szmauz-Dybko, M.; Konarska, M. Cardiovascular Stress, Energy Expenditure and Subjective Perceived Ratings of Fire Fighters During Typical Fire Suppression and Rescue Tasks. Int. J. Occup. Saf. Ergon. 2007, 13, 323–331. [Google Scholar] [CrossRef] [PubMed]
- Fahy, R.F.; Molis, J.L. Firefighter Fatalities in the United States–2018; National Fire Protection Association (NFPA): Quincy, MA, USA, 2019. [Google Scholar]
- Campbell, R.; Evarts, B.; Molis, J.L. US Firefighter Injuries–2018; National Fire Protection Association (NFPA): Quicy, MA, USA, 2019. [Google Scholar]
- Haller, J.M.; Smith, D.L. Examination of Strenuous Activity Preceding Cardiac Death during Firefighting Duties. Safety 2019, 5, 50. [Google Scholar] [CrossRef]
- Kales, S.N.; Soteriades, E.S.; Christophi, C.A.; Christiani, D.C. Emergency Duties and Deaths from Heart Disease among Firefighters in the United States. N. Engl. J. Med. 2007, 356, 1207–1215. [Google Scholar] [CrossRef]
- Peçanha, T.; Bartels, R.; Brito, L.C.; Paula-Ribeiro, M.; Oliveira, R.S.; Goldberger, J.J. Methods of assessment of the post-exercise cardiac autonomic recovery: A methodological review. Int. J. Cardiol. 2017, 227, 795–802. [Google Scholar] [CrossRef]
- Borresen, J.; Lambert, M.I. Autonomic Control of Heart Rate during and after Exercise. Sports Med. 2008, 38, 633–646. [Google Scholar] [CrossRef]
- Peçanha, T.; Silva-Júnior, N.D.; Forjaz, C.L.D.M. Heart rate recovery: Autonomic determinants, methods of assessment and association with mortality and cardiovascular diseases. Clin. Physiol. Funct. Imaging 2014, 34, 327–339. [Google Scholar] [CrossRef]
- Nishime, E.O.; Cole, C.R.; Blackstone, E.H.; Pashkow, F.J.; Lauer, M.S. Heart Rate Recovery and Treadmill Exercise Score as Predictors of Mortality in Patients Referred for Exercise ECG. JAMA 2000, 284, 1392–1398. [Google Scholar] [CrossRef]
- Vivekananthan, D.P.; Blackstone, E.H.; E Pothier, C.; Lauer, M.S. Heart rate recovery after exercise is apredictor of mortality, independent of the angiographic severity of coronary disease. J. Am. Coll. Cardiol. 2003, 42, 831–838. [Google Scholar] [CrossRef]
- Cole, C.R.; Foody, J.M.; Blackstone, E.H.; Lauer, M.S. Heart rate recovery after submaximal exercise testing as a predictor of mortality in a cardiovascularly healthy cohort. Ann. Intern. Med. 2000, 132, 552–555. [Google Scholar] [CrossRef] [PubMed]
- Jouven, X.; Empana, J.-P.; Schwartz, P.J.; Desnos, M.; Courbon, D.; Ducimetière, P. Heart-Rate Profile during Exercise as a Predictor of Sudden Death. N. Engl. J. Med. 2005, 352, 1951–1958. [Google Scholar] [CrossRef] [PubMed]
- Kannankeril, P.J.; Goldberger, J.J. Parasympathetic effects on cardiac electrophysiology during exercise and recovery. Am. J. Physiol. Circ. Physiol. 2002, 282, H2091–H2098. [Google Scholar] [CrossRef] [PubMed]
- Kales, S.N.; Smith, D.L. Firefighting and the Heart. Circulation 2017, 135, 1296–1299. [Google Scholar] [CrossRef] [PubMed]
- Cornell, D.J.; Gnacinski, S.L.; Meyer, B.B.; Ebersole, K.T. Changes in Health and Fitness in Firefighter Recruits. Med. Sci. Sports Exerc. 2017, 49, 2223–2233. [Google Scholar] [CrossRef]
- Gnacinski, S.L.; Ebersole, K.T.; Cornell, D.J.; Mims, J.; Zamzow, A.; Meyer, B.B. Firefighters’ cardiovascular health and fitness: An observation of adaptations that occur during firefighter training academies. Work 2016, 54, 43–50. [Google Scholar] [CrossRef]
- Roberts, M.A.; O’Dea, J.; Boyce, A.; Mannix, E.T. Fitness levels of firefighter recruits before and after a supervised exercise training program. J. Strength Cond. Res. 2002, 16, 271–277. [Google Scholar]
- Poplin, G.S.; Roe, D.J.; Peate, W.; Harris, R.B.; BurgessMD, J.L. The Association of Aerobic Fitness With Injuries in the Fire Service. Am. J. Epidemiol. 2014, 179, 149–155. [Google Scholar] [CrossRef]
- International Association of Fire Fighters; International Association of Fire Chiefs (IAFF/IAFC). The Fire Service Joint Labor Management Wellness-Fitness Initiative, 4th ed.; International Association of Fire Fighters; International Association of Fire Chiefs (IAFF/IAFC): Washington, DC, USA, 2018. [Google Scholar]
- Griffin, S.C.; Regan, T.L.; Harber, P.; Lutz, A.E.; Hu, C.; Peate, W.F.; Burgess, J.L. Evaluation of a fitness intervention for new firefighters: Injury reduction and economic benefits. Inj. Prev. 2016, 22, 181–188. [Google Scholar] [CrossRef]
- Cornell, D.J.; Gnacinski, S.L.; Zamzow, A.; Mims, J.; Ebersole, K.T. Influence of body mass index on movement efficiency among firefighter recruits. Work 2016, 54, 679–687. [Google Scholar] [CrossRef] [PubMed]
- Riebe, D.C.; Ehrman, J.K.; Liguori, G.; Magal, M. Health-related physical fitness testing and interpretation. In ACSM’s Guidelines for Exercise Testing and Prescription, 10th ed.; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2018. [Google Scholar]
- Jackson, A.S.; Pollock, M.L. Practical Assessment of Body Composition. Physician Sportsmed. 1985, 13, 76–90. [Google Scholar] [CrossRef] [PubMed]
- Siri, W.E. The Gross Composition of the Body. Adv. Biol. Med Phys. 1956, 4, 239–280. [Google Scholar] [CrossRef] [PubMed]
- Sharkey, B.J. Fitness and Work Capacity; U.S. Department of Agriculture: Washington, DC, USA, 1977; p. 81.
- Cornell, D.J.; Gnacinski, S.L.; Zamzow, A.; Mims, J.; Ebersole, K.T. Measures of health, fitness, and functional movement among firefighter recruits. Int. J. Occup. Saf. Ergon. 2017, 23, 198–204. [Google Scholar] [CrossRef]
- Bouts, A.M.; Brackman, L.; Martin, E.; Subasic, A.M.; Potkanowicz, E.S. The accuracy and validity of iOS-based heart rate apps during moderate to high intensity exercise. Int. J. Exerc. Sci. 2018, 11, 533–540. [Google Scholar]
- Montes, J.; Navalta, J.W. Reliability of the Polar T31 Uncoded Heart Rate Monitor in Free Motion and Treadmill Activities. Int. J. Exerc. Sci. 2019, 12, 69–76. [Google Scholar]
- Bosquet, L.; Gamelin, F.-X.; Berthoin, S. Reliability of Postexercise Heart Rate Recovery. Int. J. Sports Med. 2008, 29, 238–243. [Google Scholar] [CrossRef]
- Tanaka, H.; Monahan, K.D.; Seals, D.R. Age-predicted maximal heart rate revisited. J. Am. Coll. Cardiol. 2001, 37, 153–156. [Google Scholar] [CrossRef]
- Dupuy, O.; Mekary, S.; Berryman, N.; Bherer, L.; Audiffren, M.; Bosquet, L. Reliability of heart rate measures used to assess post-exercise parasympathetic reactivation. Clin. Physiol. Funct. Imaging 2012, 32, 296–304. [Google Scholar] [CrossRef]
- Al Haddad, H.; Laursen, P.B.; Chollet, D.; Ahmaidi, S.; Buchheit, M. Reliability of Resting and Postexercise Heart Rate Measures. Int. J. Sports Med. 2011, 32, 598–605. [Google Scholar] [CrossRef]
- Dimkpa, U.; Ibhazehiebo, K. Assessment of the influence of age on the rate of heart rate decline after maximal exercise in non-athletic adult males. Clin. Physiol. Funct. Imaging 2009, 29, 68–73. [Google Scholar] [CrossRef] [PubMed]
- Esco, M.R.; Williford, H.N.; Olson, M.S. Skinfold Thickness is Related to Cardiovascular Autonomic Control as Assessed by Heart Rate Variability and Heart Rate Recovery. J. Strength Cond. Res. 2011, 25, 2304–2310. [Google Scholar] [CrossRef] [PubMed]
- Blomqvist, C.G.; Saltin, B. Cardiovascular Adaptations to Physical Training. Annu. Rev. Physiol. 1983, 45, 169–189. [Google Scholar] [CrossRef] [PubMed]
- Yu, C.C.; Au, C.T.; Lee, F.Y.; So, R.C.; Wong, J.P.; Mak, G.Y.; Chien, E.P.; McManus, A.M. Association Between Leisure Time Physical Activity, Cardiopulmonary Fitness, Cardiovascular Risk Factors, and Cardiovascular Workload at Work in Firefighters. Saf. Health Work. 2015, 6, 192–199. [Google Scholar] [CrossRef] [PubMed]
- Kodama, S.; Saito, K.; Tanaka, S.; Maki, M.; Yachi, Y.; Asumi, M.; Sugawara, A.; Totsuka, K.; Shimano, H.; Ohashi, Y.; et al. Cardiorespiratory Fitness as a Quantitative Predictor of All-Cause Mortality and Cardiovascular Events in Healthy Men and Women. JAMA 2009, 301, 2024–2035. [Google Scholar] [CrossRef] [PubMed]
- Ebersole, K.T.; Cornell, D.J.; Flees, R.J.; Shemelya, C.M.; Noel, S.E. Contribution of the Autonomic Nervous System to Recovery in Firefighters. J. Athl. Train. 2020, 55, 1001–1008. [Google Scholar] [CrossRef] [PubMed]
- National Fire Protection Association (NFPA). NFPA 1584: Standard on the Rehabilitation Process for Members During Emergency Operations and Training Exercises; National Fire Protection Association (NFPA): Quincy, MA, USA, 2015. [Google Scholar]
- Marcel-Millet, P.; Ravier, G.; Esco, M.R.; Groslambert, A. Does firefighters’ physical fitness influence their cardiac parasympathetic reactivation? Analysis with post-exercise heart rate variability and ultra-short-term measures. Int. J. Occup. Saf. Ergon. 2020, 10, 1–9. [Google Scholar] [CrossRef]
| Variables | Mean (SD) | Range |
|---|---|---|
| Age, yrs | 31.6 (4.4) | 25–45 |
| Height, cm | 178.5 (5.2) | 168.9–189.2 |
| Body Mass, kg | 89.3 (11.4) | 70.8–114.8 |
| BMI, kg·m−2 | 28.0 (3.4) | 21.9–36.6 |
| BF, % | 14.8 (3.0) | 10.9–21.6 |
| Variable | W1 | W6 | W15 |
|---|---|---|---|
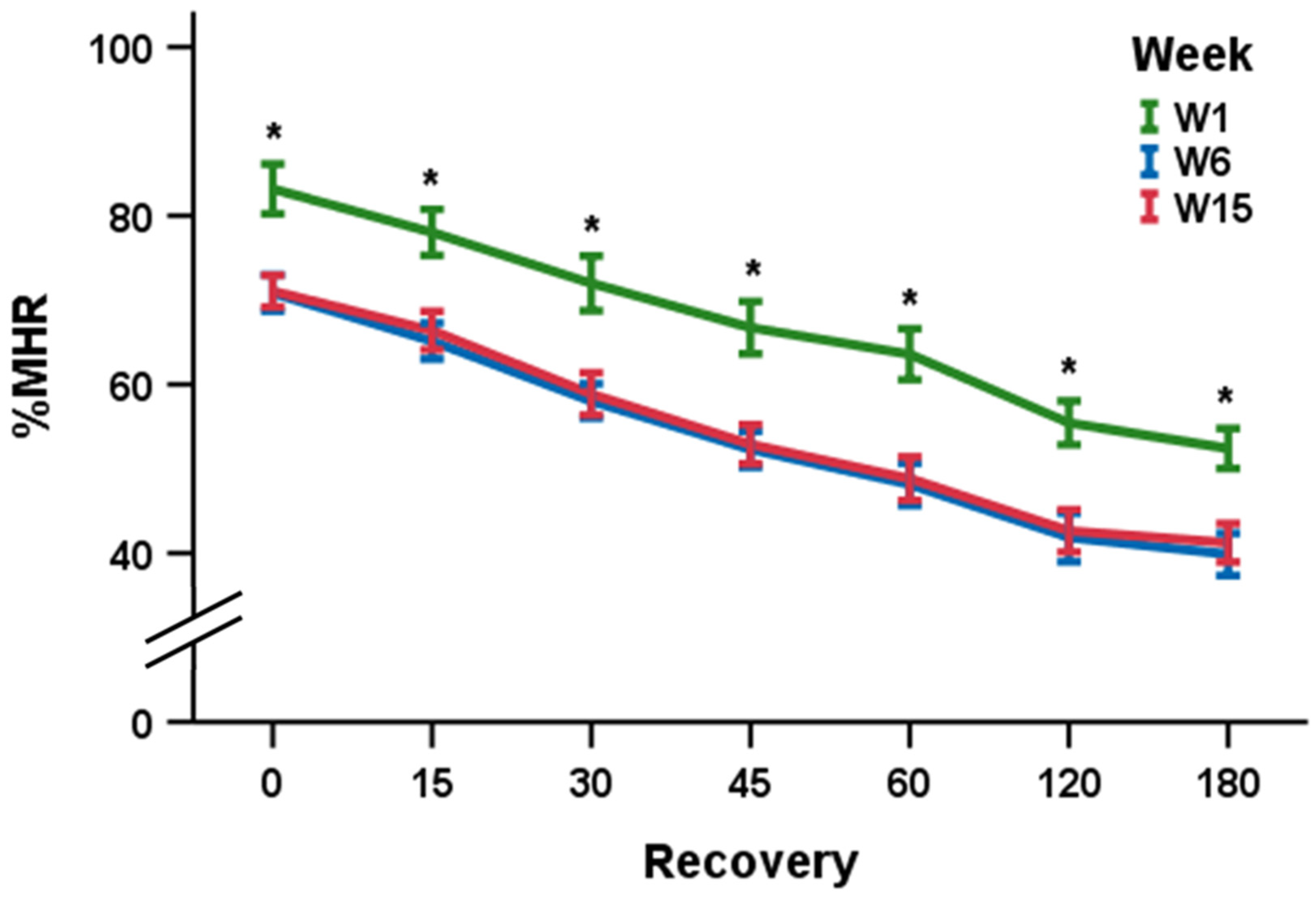
| %MHR0 | 83.2 (7.2) * | 70.8 (5.2) | 71.1 (4.6) |
| %MHR15 | 78.0 (6.6) * | 65.2 (5.2) | 66.4 (5.5) |
| %MHR30 | 72.0 (7.8) * | 58.1 (4.9) | 58.9 (6.1) |
| %MHR45 | 66.7 (7.5) * | 52.3 (5.6) | 52.9 (5.6) |
| %MHR60 | 63.6 (7.3) * | 48.2 (6.0) | 48.8 (6.3) |
| %MHR120 | 55.5 (6.3) * | 41.9 (7.0) | 42.7 (6.1) |
| %MHR180 | 52.4 (5.7) * | 39.8 (6.1) | 41.2 (5.5) |
| Variable | W1 | W6 | W15 |
|---|---|---|---|
| ΔHRR30, b·min−1 | 20.8 (6.3) | 23.8 (9.4) | 22.7 (6.2) |
| ΔHRR60, b·min−1 | 36.4 (6.9) | 42.2 (9.8) | 41.4 (8.7) |
| ΔHRR120, b·min−1 | 51.6 (8.7) | 53.9 (11.8) | 52.8 (8.8) |
| ΔHRR180, b·min−1 | 57.2 (10.1) | 57.6 (10.8) | 55.5 (8.5) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cornell, D.J.; Noel, S.E.; Zhang, X.; Ebersole, K.T. Influence of a Training Academy on the Parasympathetic Nervous System Reactivation of Firefighter Recruits—An Observational Cohort Study. Int. J. Environ. Res. Public Health 2021, 18, 109. https://doi.org/10.3390/ijerph18010109
Cornell DJ, Noel SE, Zhang X, Ebersole KT. Influence of a Training Academy on the Parasympathetic Nervous System Reactivation of Firefighter Recruits—An Observational Cohort Study. International Journal of Environmental Research and Public Health. 2021; 18(1):109. https://doi.org/10.3390/ijerph18010109
Chicago/Turabian StyleCornell, David J., Sabrina E. Noel, Xiyuan Zhang, and Kyle T. Ebersole. 2021. "Influence of a Training Academy on the Parasympathetic Nervous System Reactivation of Firefighter Recruits—An Observational Cohort Study" International Journal of Environmental Research and Public Health 18, no. 1: 109. https://doi.org/10.3390/ijerph18010109
APA StyleCornell, D. J., Noel, S. E., Zhang, X., & Ebersole, K. T. (2021). Influence of a Training Academy on the Parasympathetic Nervous System Reactivation of Firefighter Recruits—An Observational Cohort Study. International Journal of Environmental Research and Public Health, 18(1), 109. https://doi.org/10.3390/ijerph18010109





