Neural Responses to Infant Emotions and Emotional Self-Awareness in Mothers and Fathers during Pregnancy
Abstract
1. Introduction
1.1. Parental Sensitivity, Affect Regulation, and Emotional Self-Awareness
1.2. Research on Neural Correlates of Parental Responsiveness to Infant Cues
1.3. Study Aims
2. Materials and Methods
2.1. Participants and Procedures
2.2. Psychopathological Screening
2.3. Psychological Measures: Emotional Self-Awareness
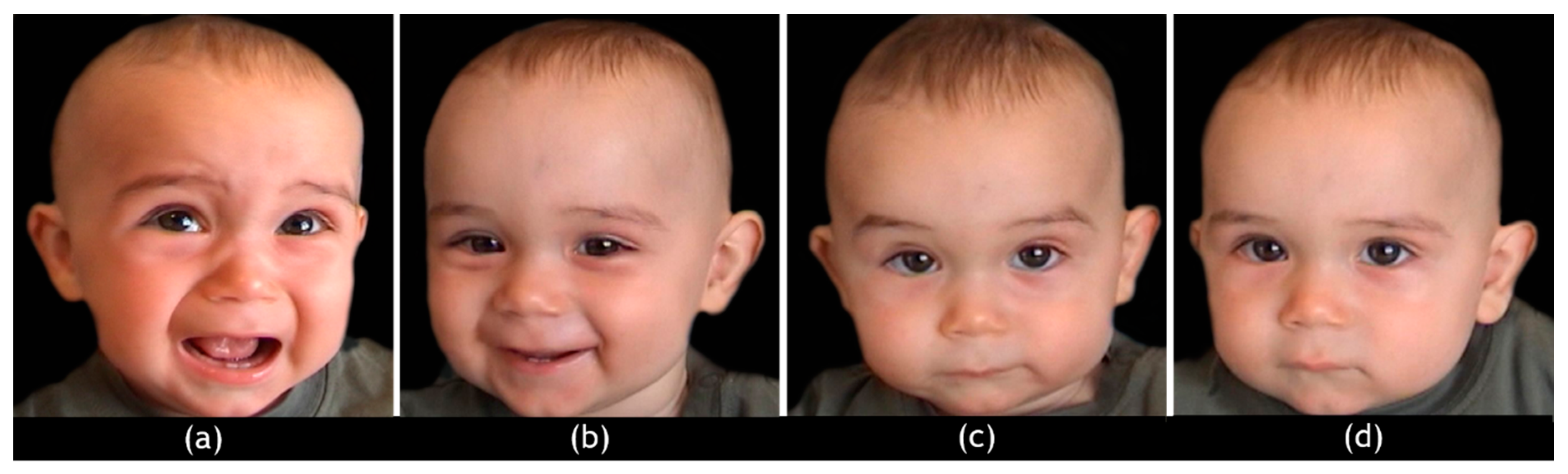
2.4. hdEEG Stimuli
2.5. hdEEG Recordings
2.6. Data Analyses
2.6.1. Psychological Measures
2.6.2. hdEEG Processing and Analysis
3. Results
3.1. Psychological Data
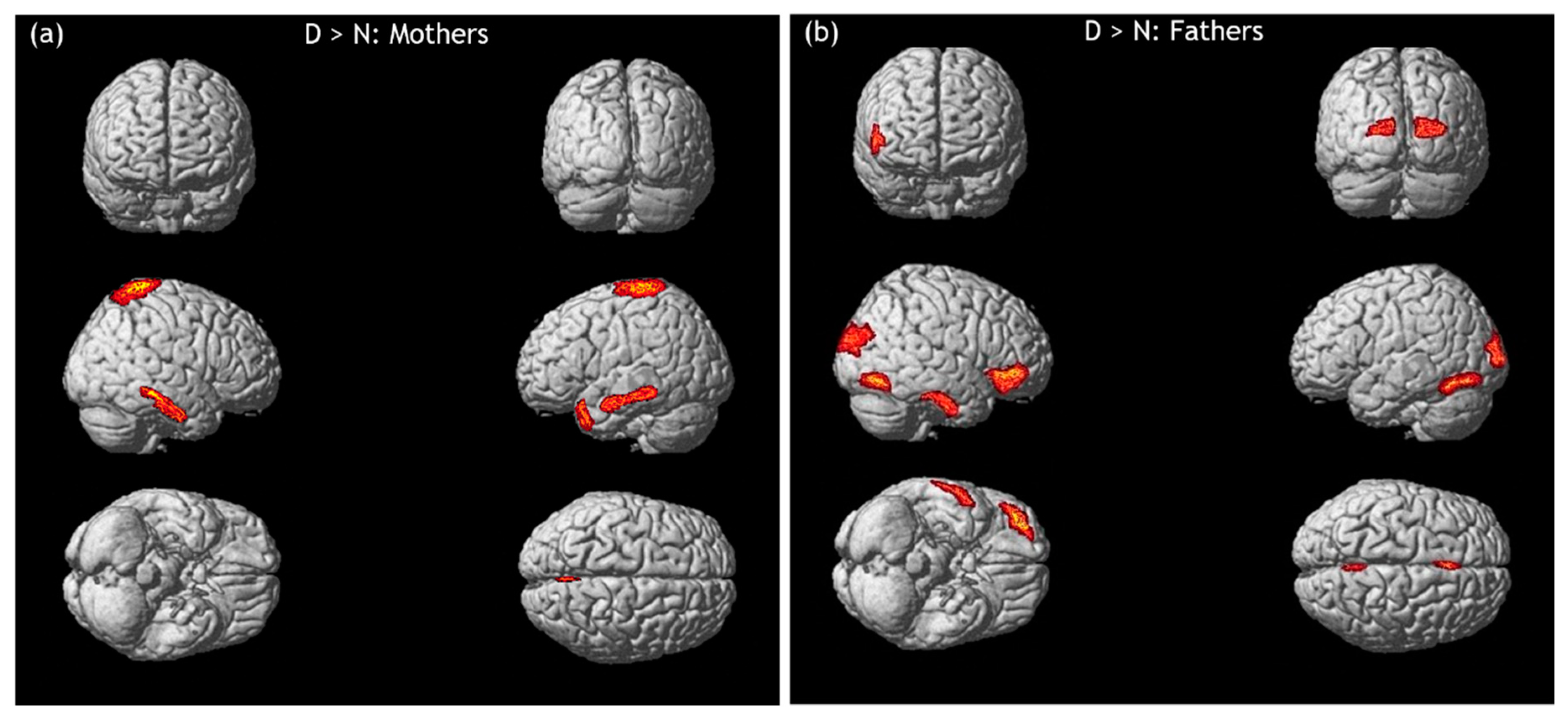
3.2. hdEEG Data: Infant Emotional Faces to Neutral Faces
3.2.1. Infant Distressed Faces versus Infant Neutral Faces
3.2.2. Infant Happy Faces versus Infant Neutral Faces
3.2.3. Infant Ambiguous Faces versus Infant Neutral Faces
3.3. Correlations between Psychological Data and hdEEG Data
4. Discussion
Neural Responses to Infant Emotions Vary According to Emotional Self-Awareness, in Mothers and Fathers
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Trevarthen, C.; Aitken, K.J. Infant Intersubjectivity: Research, Theory, and Clinical Applications. J. Child Psychol. Psychiatry 2001, 42, 3–48. [Google Scholar] [CrossRef]
- Trevarthen, C. First Things First: Infants Make Good Use of the Sympathetic Rhythm of Imitation, without Reason or Language. J. Child Psychother. 2005, 31, 91–113. [Google Scholar] [CrossRef]
- Trevarthen, C. What Is It like to Be a Person Who Knows Nothing? Defining the Active Intersubjective Mind of a Newborn Human Being. Infant Child Dev. 2011, 20, 119–135. [Google Scholar] [CrossRef]
- Papoušek, H.; Papoušek, M. Biological Basis of Social Interactions: Implications of Research for an Understanding of Behavioural Deviance. J. Child Psychol. Psychiatry 1983, 24, 117–129. [Google Scholar] [CrossRef] [PubMed]
- Stern, D.N. The Interpersonal World of the Infant; Basic Books: New York, NY, USA, 1985. [Google Scholar]
- Koester, L.S.; Papoušek, H.; Papoušek, M. Psychobiological Models of Infant Development: Influences on the Concept of Intuitive Parenting. Adv. Psychol. 1987, 46, 275–287. [Google Scholar] [CrossRef]
- Keren, M.; Feldman, R.; Eidelman, A.I.; Sirota, L.; Lester, B. Clinical Interview for High-Risk Parents of Premature Infants (CLIP) as a Predictor of Early Disruptions in the Mother-Infant Relationship at the Nursery. Infant Ment. Health J. 2003, 24, 93–110. [Google Scholar] [CrossRef]
- Feldman, R.; Eidelman, A.I. Maternal Postpartum Behavior and the Emergence of Infant–Mother and Infant–Father Synchrony in Preterm and Full-Term Infants: The Role of Neonatal Vagal Tone. Dev. Psychobiol. 2007, 49, 290–302. [Google Scholar] [CrossRef]
- Harding, C.; Levin, A.; Crossley, S.-L.; Murphy, R.; van den Engel-Hoek, L. Effects of Early Communication Intervention on Speech and Communication Skills of Preterm Infants in the Neonatal Intensive Care Unit (NICU): A Systematic Review. J. Neonatal Nurs. 2019, 25, 177–188. [Google Scholar] [CrossRef]
- Goldberg, S.; Perrotta, M.; Minde, K.; Corter, C. Maternal Behavior and Attachment in Low-Birth-Weight Twins and Singletons. Child Dev. 1986, 57, 34. [Google Scholar] [CrossRef] [PubMed]
- Bornstein, M.H. Handbook of Parenting; Lawrence Erlbaum Associates: Mahwah, NJ, USA, 2002; Volume 1–5. [Google Scholar]
- Bowlby, J. Attachment and Loss: Attachment; Basic Books: New York, NY, USA, 1969; Volume 1. [Google Scholar]
- Lorenz, K. Die Angeborenen Formen Möglicher Erfahrung. Z. Für Tierpsychol. 1943, 5, 235–409. [Google Scholar] [CrossRef]
- Lorenz, K. Studies in Animal and Human Behavior; Methuen: London, UK, 1971; Volume 2. [Google Scholar]
- Caria, A.; de Falco, S.; Venuti, P.; Lee, S.; Esposito, G.; Rigo, P.; Birbaumer, N.; Bornstein, M.H. Species-Specific Response to Human Infant Faces in the Premotor Cortex. NeuroImage 2012, 60, 884–893. [Google Scholar] [CrossRef] [PubMed]
- Brosch, T.; Sander, D.; Scherer, K.R. That Baby Caught My Eye... Attention Capture by Infant Faces. Emotion 2007, 7, 685–689. [Google Scholar] [CrossRef] [PubMed]
- Glocker, M.L.; Langleben, D.D.; Ruparel, K.; Loughead, J.W.; Valdez, J.N.; Griffin, M.D.; Sachser, N.; Gur, R.C. Baby Schema Modulates the Brain Reward System in Nulliparous Women. Proc. Natl. Acad. Sci. USA 2009, 106, 9115–9119. [Google Scholar] [CrossRef] [PubMed]
- Kringelbach, M.L.; Lehtonen, A.; Squire, S.; Harvey, A.G.; Craske, M.G.; Holliday, I.E.; Green, A.L.; Aziz, T.Z.; Hansen, P.C.; Cornelissen, P.L.; et al. A Specific and Rapid Neural Signature for Parental Instinct. PLoS ONE 2008, 3, e1664. [Google Scholar] [CrossRef]
- Seligman, M.E.P. Phobias and Preparedness. Behav. Ther. 1971, 2, 307–320. [Google Scholar] [CrossRef]
- Hodsoll, J.; Quinn, K.A.; Hodsoll, S. Attentional Prioritization of Infant Faces Is Limited to Own-Race Infants. PLoS ONE 2010, 5, e12509. [Google Scholar] [CrossRef]
- Proverbio, A.M.; Gabriele, V.D.; Manfredi, M.; Adorni, R. No Race Effect (ORE) in the Automatic Orienting toward Baby Faces: When Ethnic Group Does Not Matter. Psychology 2011, 2, 931–935. [Google Scholar] [CrossRef]
- Thompson-Booth, C.; Viding, E.; Mayes, L.C.; Rutherford, H.J.V.; Hodsoll, S.; McCrory, E.J. Here’s Looking at You, Kid: Attention to Infant Emotional Faces in Mothers and Non-Mothers. Dev. Sci. 2014, 17, 35–46. [Google Scholar] [CrossRef]
- Grasso, D.J.; Moser, J.S.; Dozier, M.; Simons, R. ERP Correlates of Attention Allocation in Mothers Processing Faces of Their Children. Biol. Psychol. 2009, 81, 95–102. [Google Scholar] [CrossRef]
- Proverbio, A.M.; Brignone, V.; Matarazzo, S.; Del Zotto, M.; Zani, A. Gender and Parental Status Affect the Visual Cortical Response to Infant Facial Expression. Neuropsychologia 2006, 44, 2987–2999. [Google Scholar] [CrossRef]
- Pearson, R.M.; Lightman, S.L.; Evans, J. Attentional Processing of Infant Emotion during Late Pregnancy and Mother–Infant Relations after Birth. Arch. Womens Ment. Health 2011, 14, 23–31. [Google Scholar] [CrossRef] [PubMed]
- Ainsworth, M.D.S. Infancy in Uganda: Infant Care and the Growth of Love; Johns Hopkins University Press: Baltimore, MD, USA, 1967. [Google Scholar]
- Ammaniti, M.; Trentini, C. How New Knowledge About Parenting Reveals the Neurobiological Implications of Intersubjectivity: A Conceptual Synthesis of Recent Research. Psychoanal. Dialogues 2009, 19, 537–555. [Google Scholar] [CrossRef]
- Condon, J.T.; Corkindale, C.J.; Boyce, P. Assessment of Postnatal Paternal–Infant Attachment: Development of a Questionnaire Instrument. J. Reprod. Infant Psychol. 2008, 26, 195–210. [Google Scholar] [CrossRef]
- Emde, R.N. Embodiment and Our Immersion With Others: Commentary On Fonagy and Target. J. Am. Psychoanal. Assoc. 2007, 55, 485–492. [Google Scholar] [CrossRef]
- Habib, C.; Lancaster, S. Changes in Identity and Paternal–Foetal Attachment across a First Pregnancy. J. Reprod. Infant Psychol. 2010, 28, 128–142. [Google Scholar] [CrossRef]
- Righetti, P.L.; Dell’Avanzo, M.; Grigio, M.; Nicolini, U. Maternal/Paternal Antenatal Attachment and Fourth-Dimensional Ultrasound Technique: A Preliminary Report. Br. J. Psychol. 2005, 96, 129–137. [Google Scholar] [CrossRef]
- Tambelli, R.; Trentini, C.; Trovato, A.; Volpi, B. Role of Psychosocial Risk Factors in Predicting Maternal and Paternal Depressive Symptomatology during Pregnancy. Infant Ment. Health J. 2019, 40, 541–556. [Google Scholar] [CrossRef]
- Belsky, J.; Fearon, R.M.P. Early Attachment Security, Subsequent Maternal Sensitivity, and Later Child Development: Does Continuity in Development Depend upon Continuity of Caregiving? Attach. Hum. Dev. 2002, 4, 361–387. [Google Scholar] [CrossRef]
- Bigelow, A.E.; MacLean, K.; Proctor, J.; Myatt, T.; Gillis, R.; Power, M. Maternal Sensitivity throughout Infancy: Continuity and Relation to Attachment Security. Infant Behav. Dev. 2010, 33, 50–60. [Google Scholar] [CrossRef]
- Fraley, R.C.; Roisman, G.I.; Haltigan, J.D. The Legacy of Early Experiences in Development: Formalizing Alternative Models of How Early Experiences Are Carried Forward over Time. Dev. Psychol. 2013, 49, 109–126. [Google Scholar] [CrossRef]
- Lyons-Ruth, K.; Spielman, E. Disorganized Infant Attachment Strategies and Helpless-Fearful Profiles of Parenting: Integrating Attachment Research with Clinical Intervention. Infant Ment. Health J. 2004, 25, 318–335. [Google Scholar] [CrossRef] [PubMed]
- Feldman, R. Infant-Mother and Infant-Father Synchrony: The Coregulation of Positive Arousal. Infant Ment. Health J. 2003, 24, 1–23. [Google Scholar] [CrossRef]
- Grossmann, K.; Grossmann, K.E.; Fremmer-Bombik, E.; Kindler, H.; Scheuerer-Englisch, H.; Zimmermann, P. The Uniqueness of the Child-Father Attachment Relationship: Fathers’ Sensitive and Challenging Play as a Pivotal Variable in a 16-Year Longitudinal Study. Soc. Dev. 2002, 11, 301–337. [Google Scholar] [CrossRef]
- Grossmann, K.; Grossmann, K.E.; Kindler, H.; Zimmermann, P. A Wider View of Attachment and Exploration: The Influence of Mothers and Fathers on the Development of Psychological Security from Infancy to Young Adulthood, In Handbook of Attachment: Theory, Research, and Clinical Applications; Cassidy, J., Shaver, P.R., Eds.; Guilford Press: New York, NY, USA, 2008; pp. 857–879. [Google Scholar]
- Paquette, D. Theorizing the Father-Child Relationship: Mechanisms and Developmental Outcomes. Hum. Dev. 2004, 47, 193–219. [Google Scholar] [CrossRef]
- Feldman, R. The Development of Regulatory Functions From Birth to 5 Years: Insights From Premature Infants: Regulatory Functions. Child Dev. 2009, 80, 544–561. [Google Scholar] [CrossRef] [PubMed]
- Rutherford, H.J.V.; Wallace, N.S.; Laurent, H.K.; Mayes, L.C. Emotion Regulation in Parenthood. Dev. Rev. 2015, 36, 1–14. [Google Scholar] [CrossRef]
- Barrett, L.F.; Gross, J.; Christensen, T.C.; Benvenuto, M. Knowing What You’re Feeling and Knowing What to Do about It: Mapping the Relation between Emotion Differentiation and Emotion Regulation. Cogn. Emot. 2001, 15, 713–724. [Google Scholar] [CrossRef]
- Grynberg, D.; Chang, B.; Corneille, O.; Maurage, P.; Vermeulen, N.; Berthoz, S.; Luminet, O. Alexithymia and the Processing of Emotional Facial Expressions (EFEs): Systematic Review, Unanswered Questions and Further Perspectives. PLoS ONE 2012, 7, e42429. [Google Scholar] [CrossRef]
- Moriguchi, Y.; Maeda, M.; Igarashi, T.; Ishikawa, T.; Shoji, M.; Kubo, C.; Komaki, G. Age and Gender Effect on Alexithymia in Large, Japanese Community and Clinical Samples: A Cross-Validation Study of the Toronto Alexithymia Scale (TAS-20). Biopsychosoc. Med. 2007, 1, 7. [Google Scholar] [CrossRef]
- Luminet, O.; Bagby, R.M.; Taylor, G.J. An Evaluation of the Absolute and Relative Stability of Alexithymia in Patients with Major Depression. Psychother. Psychosom. 2001, 70, 254–260. [Google Scholar] [CrossRef]
- Taylor, G.J. Recent Developments in Alexithymia Theory and Research. Can. J. Psychiatry 2000, 45, 134–142. [Google Scholar] [CrossRef] [PubMed]
- Franz, M.; Popp, K.; Schaefer, R.; Sitte, W.; Schneider, C.; Hardt, J.; Decker, O.; Braehler, E. Alexithymia in the German General Population. Soc. Psychiatry Psychiatr. Epidemiol. 2008, 43, 54–62. [Google Scholar] [CrossRef] [PubMed]
- Schimmenti, A.; Jonason, P.K.; Passanisi, A.; La Marca, L.; Di Dio, N.; Gervasi, A.M. Exploring the Dark Side of Personality: Emotional Awareness, Empathy, and the Dark Triad Traits in an Italian Sample. Curr. Psychol. 2019, 38, 100–109. [Google Scholar] [CrossRef]
- Rieffe, C.; Oosterveld, P.; Miers, A.C.; Meerum Terwogt, M.; Ly, V. Emotion Awareness and Internalising Symptoms in Children and Adolescents: The Emotion Awareness Questionnaire Revised. Personal. Individ. Differ. 2008, 45, 756–761. [Google Scholar] [CrossRef]
- Bagby, R.M.; Taylor, G.J. Affect Dyregulation and Alexithymia. In Disorders of Affect Regulation: Alexithymia in Medical and Psychiatric Illness; Taylor, G.J., Bagby, R.M., Parker, J.D.A., Eds.; Cambridge University Press: Cambridge, UK, 1997; pp. 26–45. [Google Scholar]
- Aust, S.; Alkan Härtwig, E.; Koelsch, S.; Heekeren, H.R.; Heuser, I.; Bajbouj, M. How Emotional Abilities Modulate the Influence of Early Life Stress on Hippocampal Functioning. Soc. Cogn. Affect. Neurosci. 2014, 9, 1038–1045. [Google Scholar] [CrossRef]
- Donges, U.-S.; Suslow, T. Alexithymia and Automatic Processing of Emotional Stimuli: A Systematic Review. Rev. Neurosci. 2017, 28, 247–264. [Google Scholar] [CrossRef]
- Yürümez, E.; Akça, Ö.F.; Uğur, Ç.; Uslu, R.I.; Kılıç, B.G. Mothers’ Alexithymia, Depression and Anxiety Levels and Their Association with the Quality of Mother-Infant Relationship: A Preliminary Study. Int. J. Psychiatry Clin. Pract. 2014, 18, 190–196. [Google Scholar] [CrossRef]
- Fukunishi, I.; Kawamura, N.; Ishikawa, T.; Ago, Y.; Sei, H.; Morita, Y.; Rahe, R.H. Mothers’ Low Care in the Development of Alexithymia: A Preliminary Study in Japanese College Students. Psychol. Rep. 1997, 80, 143–146. [Google Scholar] [CrossRef]
- Kooiman, C.G.; van Rees Vellinga, S.; Spinhoven, P.; Draijer, N.; Trijsburg, R.W.; Rooijmans, H.G.M. Childhood Adversities as Risk Factors for Alexithymia and Other Aspects of Affect Dysregulation in Adulthood. Psychother. Psychosom. 2004, 73, 107–116. [Google Scholar] [CrossRef]
- Roque, L.; Veríssimo, M. Emotional Context, Maternal Behavior and Emotion Regulation. Infant Behav. Dev. 2011, 34, 617–626. [Google Scholar] [CrossRef]
- Levant, R.F.; Kopecky, G. Masculinity Reconstructed: Changing the Rules of Manhood- at Work, in Relationships and in Family Life; Dutton: New York, NY, USA, 1995. [Google Scholar]
- Montebarocci, O.; Codispoti, M.; Baldaro, B.; Rossi, N. Adult Attachment Style and Alexithymia. Personal. Individ. Differ. 2004, 36, 499–507. [Google Scholar] [CrossRef]
- Schimmenti, A.; Caretti, V. Attachment, Trauma, and Alexithymia. In Alexithymia: Advances in Research, Theory, and Clinical Practice; Luminet, O., Bagby, R.M., Taylor, G.J., Eds.; Cambridge University Press: New York, NY, USA, 2018; pp. 127–141. [Google Scholar]
- Kaitz, M.; Good, A.; Rokem, A.M.; Eidelman, A.I. Mothers’ Recognition of Their Newborns by Olfactory Cues. Dev. Psychobiol. 1987, 20, 587–591. [Google Scholar] [CrossRef] [PubMed]
- Kaitz, M.; Rokem, A.M.; Eidelman, A.I. Infants’ Face-Recognition by Primiparous and Multiparous Women. Percept. Mot. Skills 1988, 67, 495–502. [Google Scholar] [CrossRef] [PubMed]
- Kinsley, C.H.; Lambert, K.G. The Maternal Brain. Sci. Am. 2006, 294, 72–79. [Google Scholar] [CrossRef]
- Purhonen, M.; Kilpeläinen-Lees, R.; Pääkkönen, A.; Yppärilä, H.; Lehtonen, J.; Karhu, J. Effects of Maternity on Auditory Event-Related Potentials to Human Sound. Neuroreport 2001, 12, 2975–2979. [Google Scholar] [CrossRef]
- Mayes, L.C.; Swain, J.E.; Leckman, J.F. Parental Attachment Systems: Neural Circuits, Genes, and Experiential Contributions to Parental Engagement. Clin. Neurosci. Res. 2005, 4, 301–313. [Google Scholar] [CrossRef]
- Panksepp, J. Affective Neuroscience: The Foundations of Human and Animal Emotions; Oxford Univ. Press: Oxford, UK, 2005. [Google Scholar]
- Insel, T.R.; Young, L.J. The Neurobiology of Attachment. Nat. Rev. Neurosci. 2001, 2, 129–136. [Google Scholar] [CrossRef]
- Gordon, I.; Zagoory-Sharon, O.; Leckman, J.F.; Feldman, R. Oxytocin and the Development of Parenting in Humans. Biol. Psychiatry 2010, 68, 377–382. [Google Scholar] [CrossRef]
- Storey, A. Hormonal Correlates of Paternal Responsiveness in New and Expectant Fathers. Evol. Hum. Behav. 2000, 21, 79–95. [Google Scholar] [CrossRef]
- Paul, S.; Austin, J.; Elliott, R.; Ellison-Wright, I.; Wan, M.W.; Drake, R.; Downey, D.; Elmadih, A.; Mukherjee, I.; Heaney, L.; et al. Neural Pathways of Maternal Responding: Systematic Review and Meta-Analysis. Arch. Womens Ment. Health 2019, 22, 179–187. [Google Scholar] [CrossRef]
- Swain, J.E.; Kim, P.; Spicer, J.; Ho, S.S.; Dayton, C.J.; Elmadih, A.; Abel, K.M. Approaching the Biology of Human Parental Attachment: Brain Imaging, Oxytocin and Coordinated Assessments of Mothers and Fathers. Brain Res. 2014, 1580, 78–101. [Google Scholar] [CrossRef] [PubMed]
- Bernhardt, B.C.; Singer, T. The Neural Basis of Empathy. Annu. Rev. Neurosci. 2012, 35, 1–23. [Google Scholar] [CrossRef] [PubMed]
- Bethlehem, R.A.I.; van Honk, J.; Auyeung, B.; Baron-Cohen, S. Oxytocin, Brain Physiology, and Functional Connectivity: A Review of Intranasal Oxytocin FMRI Studies. Psychoneuroendocrinology 2013, 38, 962–974. [Google Scholar] [CrossRef] [PubMed]
- Feldman, R. The Adaptive Human Parental Brain: Implications for Children’s Social Development. Trends Neurosci. 2015, 38, 387–399. [Google Scholar] [CrossRef]
- Meyer-Lindenberg, A.; Domes, G.; Kirsch, P.; Heinrichs, M. Oxytocin and Vasopressin in the Human Brain: Social Neuropeptides for Translational Medicine. Nat. Rev. Neurosci. 2011, 12, 524–538. [Google Scholar] [CrossRef]
- Navratilova, E.; Porreca, F. Reward and Motivation in Pain and Pain Relief. Nat. Neurosci. 2014, 17, 1304–1312. [Google Scholar] [CrossRef]
- Swain, J.E. The Human Parental Brain: In Vivo Neuroimaging. Prog. Neuropsychopharmacol. Biol. Psychiatry 2011, 35, 1242–1254. [Google Scholar] [CrossRef]
- Kim, P.; Leckman, J.F.; Mayes, L.C.; Feldman, R.; Wang, X.; Swain, J.E. The Plasticity of Human Maternal Brain: Longitudinal Changes in Brain Anatomy during the Early Postpartum Period. Behav. Neurosci. 2010, 124, 695–700. [Google Scholar] [CrossRef]
- Gallese, V. Mirror Neurons, Embodied Simulation, and the Neural Basis of Social Identification. Psychoanal. Dialogues 2009, 19, 519–536. [Google Scholar] [CrossRef]
- Lenzi, D.; Trentini, C.; Pantano, P.; Macaluso, E.; Iacoboni, M.; Lenzi, G.L.; Ammaniti, M. Neural Basis of Maternal Communication and Emotional Expression Processing during Infant Preverbal Stage. Cereb. Cortex 2009, 19, 1124–1133. [Google Scholar] [CrossRef]
- Lenzi, D.; Trentini, C.; Pantano, P.; Macaluso, E.; Lenzi, G.L.; Ammaniti, M. Attachment Models Affect Brain Responses in Areas Related to Emotions and Empathy in Nulliparous Women. Hum. Brain Mapp. 2013, 34, 1399–1414. [Google Scholar] [CrossRef] [PubMed]
- Rizzolatti, G.; Craighero, L. The Mirror-Neuron System. Annu. Rev. Neurosci. 2004, 27, 169–192. [Google Scholar] [CrossRef] [PubMed]
- LeDoux, J. The emotional brain, fear, and the amygdala. Cell. Mol. Neurobiol. 2003, 23, 727–738. [Google Scholar] [CrossRef]
- Carr, L.; Iacoboni, M.; Dubeau, M.-C.; Mazziotta, J.C.; Lenzi, G.L. Neural Mechanisms of Empathy in Humans: A Relay from Neural Systems for Imitation to Limbic Areas. Proc. Natl. Acad. Sci. USA 2003, 100, 5497–5502. [Google Scholar] [CrossRef] [PubMed]
- Picard, N.; Strick, P.L. Motor Areas of the Medial Wall: A Review of Their Location and Functional Activation. Cereb. Cortex 1996, 6, 342–353. [Google Scholar] [CrossRef]
- Frith, U.; Frith, C.D. Development and Neurophysiology of Mentalizing. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2003, 358, 459–473. [Google Scholar] [CrossRef]
- Frith, C.D.; Frith, U. Mechanisms of Social Cognition. Annu. Rev. Psychol. 2012, 63, 287–313. [Google Scholar] [CrossRef]
- Kanat, M.; Heinrichs, M.; Domes, G. Oxytocin and the Social Brain: Neural Mechanisms and Perspectives in Human Research. Brain Res. 2014, 1580, 160–171. [Google Scholar] [CrossRef]
- Yang, D.Y.-J.; Rosenblau, G.; Keifer, C.; Pelphrey, K.A. An Integrative Neural Model of Social Perception, Action Observation, and Theory of Mind. Neurosci. Biobehav. Rev. 2015, 51, 263–275. [Google Scholar] [CrossRef]
- Nitschke, J.B.; Nelson, E.E.; Rusch, B.D.; Fox, A.S.; Oakes, T.R.; Davidson, R.J. Orbitofrontal Cortex Tracks Positive Mood in Mothers Viewing Pictures of Their Newborn Infants. NeuroImage 2004, 21, 583–592. [Google Scholar] [CrossRef]
- Menon, V. Large-Scale Brain Networks and Psychopathology: A Unifying Triple Network Model. Trends Cogn. Sci. 2011, 15, 483–506. [Google Scholar] [CrossRef] [PubMed]
- Schore, A.N. The Right Brain Implicit Self Lies at the Core of Psychoanalysis. Psychoanal. Dialogues 2011, 21, 75–100. [Google Scholar] [CrossRef]
- Bush, G.; Vogt, B.A.; Holmes, J.; Dale, A.M.; Greve, D.; Jenike, M.A.; Rosen, B.R. Dorsal Anterior Cingulate Cortex: A Role in Reward-Based Decision Making. Proc. Natl. Acad. Sci. USA 2002, 99, 523–528. [Google Scholar] [CrossRef] [PubMed]
- Rushworth, M.F.S.; Behrens, T.E.J.; Rudebeck, P.H.; Walton, M.E. Contrasting Roles for Cingulate and Orbitofrontal Cortex in Decisions and Social Behaviour. Trends Cogn. Sci. 2007, 11, 168–176. [Google Scholar] [CrossRef]
- Pollatos, O.; Gramann, K.; Schandry, R. Neural Systems Connecting Interoceptive Awareness and Feelings. Hum. Brain Mapp. 2007, 28, 9–18. [Google Scholar] [CrossRef]
- Spielberger, C.D. Manual for the State-Trait Anxiety Inventory STAI (Form Y) (“self-Evaluation Questionnaire”); Consulting Psychologists Press: Palo Alto, CA, USA, 1983. [Google Scholar]
- Pedrabissi, L.; Santinello, M. Inventario per l’ansia Di Stato e Di Tratto (Forma Y); Organizzazioni Speciali: Florence, Italy, 1989. [Google Scholar]
- Cox, J.L.; Holden, J.M.; Sagovsky, R. Detection of Postnatal Depression: Development of the 10-Item Edinburgh Postnatal Depression Scale. Br. J. Psychiatry 1987, 150, 782–786. [Google Scholar] [CrossRef]
- Benvenuti, P. The Edinburgh Postnatal Depression Scale: Validation for an Italian Sample. J. Affect. Disord. 1999, 53, 137–141. [Google Scholar] [CrossRef]
- Edmondson, O.J.H.; Psychogiou, L.; Vlachos, H.; Netsi, E.; Ramchandani, P.G. Depression in Fathers in the Postnatal Period: Assessment of the Edinburgh Postnatal Depression Scale as a Screening Measure. J. Affect. Disord. 2010, 125, 365–368. [Google Scholar] [CrossRef]
- Massoudi, P.; Hwang, C.P.; Wickberg, B. How Well Does the Edinburgh Postnatal Depression Scale Identify Depression and Anxiety in Fathers? A Validation Study in a Population Based Swedish Sample. J. Affect. Disord. 2013, 149, 67–74. [Google Scholar] [CrossRef]
- Taylor, G.J.; Bagby, M.; Parker, J.D.A. The Revised Toronto Alexithymia Scale: Some Reliability, Validity, and Normative Data. Psychother. Psychosom. 1992, 57, 34–41. [Google Scholar] [CrossRef]
- Bressi, C.; Taylor, G.; Parker, J.; Bressi, S.; Brambilla, V.; Aguglia, E.; Allegranti, I.; Bongiorno, A.; Giberti, F.; Bucca, M.; et al. Cross Validation of the Factor Structure of the 20-Item Toronto Alexithymia Scale: An Italian Multicenter Study. J. Psychosom. Res. 1996, 41, 551–559. [Google Scholar] [CrossRef]
- Lenzi, D.; Trentini, C.; Macaluso, E.; Graziano, S.; Speranza, A.M.; Pantano, P.; Ammaniti, M. Mothers with Depressive Symptoms Display Differential Brain Activations When Empathizing with Infant Faces. Psychiatry Res. Neuroimaging 2016, 249, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Oster, H.; Hegley, D.; Nagel, L. Adult Judgments and Fine-Grained Analysis of Infant Facial Expressions: Testing the Validity of a Priori Coding Formulas. Dev. Psychol. 1992, 28, 1115–1131. [Google Scholar] [CrossRef]
- Izard, C. A System for Identifying Affect Expressions by Holistic Judgements (AFFEX); Instructional Resources Center: Newark, DE, USA, 1983. [Google Scholar]
- Sullivan, M.W.; Lewis, M. Emotional Expressions of Young Infants and Children: A Practitioner’s Primer. Infants Young Child. 2003, 16, 120–142. [Google Scholar] [CrossRef][Green Version]
- Litvak, V.; Mattout, J.; Kiebel, S.; Phillips, C.; Henson, R.; Kilner, J.; Barnes, G.; Oostenveld, R.; Daunizeau, J.; Flandin, G.; et al. EEG and MEG Data Analysis in SPM8. Comput. Intell. Neurosci. 2011, 2011, 1–32. [Google Scholar] [CrossRef]
- Sharma, N.; Jones, P.S.; Carpenter, T.A.; Baron, J.-C. Mapping the Involvement of BA 4a and 4p during Motor Imagery. NeuroImage 2008, 41, 92–99. [Google Scholar] [CrossRef]
- Olson, I.R.; McCoy, D.; Klobusicky, E.; Ross, L.A. Social Cognition and the Anterior Temporal Lobes: A Review and Theoretical Framework. Soc. Cogn. Affect. Neurosci. 2013, 8, 123–133. [Google Scholar] [CrossRef]
- Hein, G.; Singer, T. I Feel How You Feel but Not Always: The Empathic Brain and Its Modulation. Curr. Opin. Neurobiol. 2008, 18, 153–158. [Google Scholar] [CrossRef]
- Steele, J.D.; Lawrie, S.M. Segregation of Cognitive and Emotional Function in the Prefrontal Cortex: A Stereotactic Meta-Analysis. NeuroImage 2004, 21, 868–875. [Google Scholar] [CrossRef]
- Ramnani, N.; Owen, A.M. Anterior Prefrontal Cortex: Insights into Function from Anatomy and Neuroimaging. Nat. Rev. Neurosci. 2004, 5, 184–194. [Google Scholar] [CrossRef]
- Gusnard, D.A.; Akbudak, E.; Shulman, G.L.; Raichle, M.E. Medial Prefrontal Cortex and Self-Referential Mental Activity: Relation to a Default Mode of Brain Function. Proc. Natl. Acad. Sci. USA 2001, 98, 4259–4264. [Google Scholar] [CrossRef] [PubMed]
- Collette, F.; Salmon, E.; Van der Linden, M.; Chicherio, C.; Belleville, S.; Degueldre, C.; Delfiore, G.; Franck, G. Regional Brain Activity during Tasks Devoted to the Central Executive of Working Memory. Cogn. Brain Res. 1999, 7, 411–417. [Google Scholar] [CrossRef]
- The American Association for Research into Nervous and Mental Diseases; Posner, M.I.; Rothbart, M.K. Attention, Self–Regulation and Consciousness. Philos. Trans. R. Soc. Lond. B Biol. Sci. 1998, 353, 1915–1927. [Google Scholar] [CrossRef] [PubMed]
- Brunet, E.; Sarfati, Y.; Hardy-Baylé, M.-C.; Decety, J. A PET Investigation of the Attribution of Intentions with a Nonverbal Task. NeuroImage 2000, 11, 157–166. [Google Scholar] [CrossRef]
- Ida Gobbini, M.; Leibenluft, E.; Santiago, N.; Haxby, J.V. Social and Emotional Attachment in the Neural Representation of Faces. NeuroImage 2004, 22, 1628–1635. [Google Scholar] [CrossRef]
- Rossion, B.; Schiltz, C.; Crommelinck, M. The Functionally Defined Right Occipital and Fusiform “Face Areas” Discriminate Novel from Visually Familiar Faces. NeuroImage 2003, 19, 877–883. [Google Scholar] [CrossRef]
- Schupp, H.T.; Öhman, A.; Junghöfer, M.; Weike, A.I.; Stockburger, J.; Hamm, A.O. The Facilitated Processing of Threatening Faces: An ERP Analysis. Emotion 2004, 4, 189–200. [Google Scholar] [CrossRef]
- Knauff, M.; Mulack, T.; Kassubek, J.; Salih, H.R.; Greenlee, M.W. Spatial Imagery in Deductive Reasoning: A Functional MRI Study. Cogn. Brain Res. 2002, 13, 203–212. [Google Scholar] [CrossRef]
- Smith, A.T.; Cotillon-Williams, N.M.; Williams, A.L. Attentional Modulation in the Human Visual Cortex: The Time-Course of the BOLD Response and Its Implications. NeuroImage 2006, 29, 328–334. [Google Scholar] [CrossRef]
- Waberski, T.D.; Gobbelé, R.; Lamberty, K.; Buchner, H.; Marshall, J.C.; Fink, G.R. Timing of Visuo-Spatial Information Processing: Electrical Source Imaging Related to Line Bisection Judgements. Neuropsychologia 2008, 46, 1201–1210. [Google Scholar] [CrossRef]
- Ciaramelli, E.; Grady, C.L.; Moscovitch, M. Top-down and Bottom-up Attention to Memory: A Hypothesis (AtoM) on the Role of the Posterior Parietal Cortex in Memory Retrieval. Neuropsychologia 2008, 46, 1828–1851. [Google Scholar] [CrossRef] [PubMed]
- Newland, L.A.; Coyl, D.D.; Freeman, H. Predicting Preschoolers’ Attachment Security from Fathers’ Involvement, Internal Working Models, and Use of Social Support. Early Child Dev. Care 2008, 178, 785–801. [Google Scholar] [CrossRef]
- Kazura, K. Fathers’ Qualitative and Quantitative Involvement: An Investigation of Attachment, Play, and Social Interactions. J. Mens Stud. 2000, 9, 41–57. [Google Scholar] [CrossRef]
- Assaf, M.; Hyatt, C.J.; Wong, C.G.; Johnson, M.R.; Schultz, R.T.; Hendler, T.; Pearlson, G.D. Mentalizing and Motivation Neural Function during Social Interactions in Autism Spectrum Disorders. NeuroImage Clin. 2013, 3, 321–331. [Google Scholar] [CrossRef] [PubMed]
- Morin, A.; Hamper, B. Self-Reflection and the Inner Voice: Activation of the Left Inferior Frontal Gyrus During Perceptual and Conceptual Self-Referential Thinking. Open Neuroimaging J. 2012, 6, 78–89. [Google Scholar] [CrossRef] [PubMed]
- Platek, S. How Much Paternal Resemblance Is Enough? Sex Differences in Hypothetical Investment Decisions but Not in the Detection of Resemblance. Evol. Hum. Behav. 2003, 24, 81–87. [Google Scholar] [CrossRef]
- Platek, S.M.; Raines, D.M.; Gallup, G.G.; Mohamed, F.B.; Thomson, J.W.; Myers, T.E.; Panyavin, I.S.; Levin, S.L.; Davis, J.A.; Fonteyn, L.C.M.; et al. Reactions to Children’s Faces: Males Are More Affected by Resemblance than Females Are, and so Are Their Brains. Evol. Hum. Behav. 2004, 25, 394–405. [Google Scholar] [CrossRef]
- Platek, S.M.; Keenan, J.P.; Mohamed, F.B.; Taylor, G.J.; Bagby, R.M.; Parker, J.D.A. Disorders of Affect Regulation: Alexithymia in Medical and Psychiatric Illness. NeuroImage 2005, 25, 1336–1344. [Google Scholar] [CrossRef]
- Winnicott, D.W. Primary Maternal Preoccupation. In Collected Papers: Through Pediatrics to Psycho-Analysis; Basic Books: New York, NY, USA, 1956; pp. 78–101. [Google Scholar]
- Leckman, J.F.; Feldman, R.; Swain, J.E.; Eicher, V.; Thompson, N.; Mayes, L.C. Primary Parental Preoccupation: Circuits, Genes, and the Crucial Role of the Environment. J. Neural Transm. 2004, 111, 753–771. [Google Scholar] [CrossRef]
- Tambelli, R.; Trentini, C.; Dentale, D. Predictive and Incremental Validity of Parental Representations during Pregnancy on Child Attachment. Front. Psychol. 2020. under review. [Google Scholar]
- Ammaniti, M.; Trentini, C.; Menozzi, F.; Tambelli, R. Transition to Parenthood: Studies of Intersubjectivity in Mothers and Fathers. In Early Parenting and the Prevention of Disorders; Emde, R.N., Leuzinger-Bohleber, M., Eds.; Publisher Karnac: London, UK, 2014; pp. 131–166. [Google Scholar]
- Fava Vizziello, G.; Antonioli, M.E.; Cocci, V.; Invernizzi, R. From Pregnancy to Motherhood: The Structure of Representative and Narrative Change. Infant Ment. Health J. 1993, 12, 4–16. [Google Scholar] [CrossRef]
- Viaux-Savelon, S.; Dommergues, M.; Rosenblum, O.; Bodeau, N.; Aidane, E.; Philippon, O.; Mazet, P.; Vibert-Guigue, C.; Vauthier-Brouzes, D.; Feldman, R.; et al. Prenatal Ultrasound Screening: False Positive Soft Markers May Alter Maternal Representations and Mother-Infant Interaction. PLoS ONE 2012, 7, e30935. [Google Scholar] [CrossRef] [PubMed]
- Schoppe-Sullivan, S.J.; Brown, G.L.; Cannon, E.A.; Mangelsdorf, S.C.; Sokolowski, M.S. Maternal Gatekeeping, Coparenting Quality, and Fathering Behavior in Families with Infants. J. Fam. Psychol. 2008, 22, 389–398. [Google Scholar] [CrossRef] [PubMed]
- Lebovici, S.; Stoléru, S. Le Nourrisson, La Mère et Le Psychanalyste. Les Interactions Précoces; Le Centurion: Paris, France, 1983. [Google Scholar]
- Stern, D.N. The Motherhood Constellation: A Unified View of Parent-Infant Psychotherapy; Basic Books: New York, NY, USA, 1995. [Google Scholar]
- Larsen, J.K.; van Strien, T.; Eisinga, R.; Engels, R.C.M.E. Gender Differences in the Association between Alexithymia and Emotional Eating in Obese Individuals. J. Psychosom. Res. 2006, 60, 237–243. [Google Scholar] [CrossRef]
- Parker, J.D.A.; Taylor, G.J.; Bagby, R.M. The 20-Item Toronto Alexithymia Scale: III. Reliability and Factorial Validity in a Community Population. J. Psychosom. Res. 2003, 55, 269–275. [Google Scholar] [CrossRef]
- Levant, R.F.; Halter, M.J.; Hayden, E.W.; Williams, C.M. The Efficacy of Alexithymia Reduction Treatment: A Pilot Study. J. Mens Stud. 2009, 17, 75–84. [Google Scholar] [CrossRef]
- Olafsen, K.S.; Rønning, J.A.; Handegård, B.H.; Ulvund, S.E.; Dahl, L.B.; Kaaresen, P.I. Regulatory Competence and Social Communication in Term and Preterm Infants at 12 Months Corrected Age. Results from a Randomized Controlled Trial. Infant Behav. Dev. 2012, 35, 140–149. [Google Scholar] [CrossRef]
- Fonagy, P.; Gergely, G.; Jurist, E.; Target, M. Affect Regulation, Mentalization, and the Development of the Self; Other Press Professional: New York, NY, USA, 2001. [Google Scholar]
| CONTRAST | MOTHERS | FATHERS | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| X | Y | z | T | Cluster | Region | BA | x | Y | Z | T | Cluster | Region | BA | |
| D > N | 16 | −24 | 68 | 10.28 | 610 | r preSMA | 6 | - | - | - | - | - | - | - |
| −4 | −34 | 70 | 9.16 | 688 | l preSMA | 6 | - | - | - | - | - | - | - | |
| −14 | −26 | 68 | 9.60 | 688 | l PrG | 4 | - | - | - | - | - | - | - | |
| 6 | −40 | 70 | 14.74 | 610 | r PrG | 4 | - | - | - | - | - | - | - | |
| - | - | - | - | - | - | - | 40 | 38 | 0 | 13.49 | 811 | r IFG | 47 | |
| - | - | - | - | - | - | - | 48 | 30 | 12 | 13.20 | 811 | r MFG | 46 | |
| - | - | - | - | - | - | - | 0 | 6 | 20 | 8.96 | 568 | l ACC | 33 | |
| 10 | −72 | 54 | 6.24 | 610 | r SPL | 7 | 34 | −66 | 46 | 8.31 | 104 | r SPL | 7 | |
| - | - | - | - | - | - | - | 24 | −90 | 2 | 12.69 | 352 | r IOG | 19 | |
| - | - | - | - | - | - | - | −30 | −84 | 8 | 7.10 | 173 | l IOG | 19 | |
| - | - | - | - | - | - | - | −18 | −94 | 4 | 8.07 | 173 | l Striate | 17 | |
| - | - | - | - | - | - | - | 38 | −48 | −20 | 16.02 | 346 | r FuG | 37 | |
| - | - | - | - | - | - | - | −36 | −52 | −20 | 16.15 | 323 | l FuG | 37 | |
| 58 | −38 | −26 | 8.11 | 155 | r ITG | 20 | 48 | −12 | −38 | 7.32 | 290 | r ITG | 20 | |
| 64 | −16 | −18 | 9.07 | 155 | r MTG | 21 | - | - | - | - | - | - | - | |
| −50 | 4 | −34 | 9.67 | 106 | l MTG | 21 | - | - | - | - | - | - | - | |
| 14 | −34 | −4 | 13.03 | 294 | r MTL | 27 | ||||||||
| −46 | 12 | −32 | 7.97 | 106 | l TP | 38 | - | - | - | - | - | - | - | |
| H > N | - | - | - | - | - | - | - | −30 | −68 | 50 | 10.41 | 138 | l SPL | 7 |
| - | - | - | - | - | - | - | −54 | −62 | 8 | 11.00 | 145 | l AG | 39 | |
| - | - | - | - | - | - | - | 48 | −76 | −4 | 8.61 | 234 | r IOG | 19 | |
| - | - | - | - | - | - | - | 40 | −66 | 0 | 8.89 | 234 | r FuG | 37 | |
| - | - | - | - | - | - | - | 46 | −18 | −28 | 11.94 | 456 | r ITG | 20 | |
| - | - | - | - | - | - | - | −44 | −14 | −32 | 11.28 | 251 | l ITG | 20 | |
| - | - | - | - | - | - | - | 52 | −62 | 14 | 11.42 | 136 | r STG | 22 | |
| A > N | - | - | - | - | - | - | - | 40 | 40 | −4 | 13.65 | 890 | r IFG | 47 |
| - | - | - | - | - | - | - | 38 | 34 | 8 | 13.89 | 890 | r MFG | 46 | |
| - | - | - | - | - | - | - | 38 | 46 | 10 | 8.35 | 890 | r FPC | 10 | |
| - | - | - | - | - | - | - | −16 | 54 | 26 | 8.40 | 237 | l FPC | 10 | |
| 40 | 16 | 42 | 12.83 | 165 | r DLPFC | 9 | 16 | 52 | 24 | 8.36 | 208 | r DLPFC | 9 | |
| - | - | - | - | - | - | - | −6 | 52 | 30 | 7.31 | 237 | l DLPFC | 9 | |
| - | - | - | - | - | - | - | −46 | −26 | 58 | 15.06 | 149 | l PoG | 3 | |
| - | - | - | - | - | - | - | 38 | −68 | 46 | 6.91 | 121 | r SPL | 7 | |
| - | - | - | - | - | - | - | 32 | −84 | 6 | 13.02 | 335 | r IOG | 19 | |
| - | - | - | - | - | - | - | −34 | −76 | 12 | 14.05 | 371 | l IOG | 19 | |
| - | - | - | - | - | - | - | 18 | −100 | 8 | 7.57 | 335 | r mOG | 18 | |
| - | - | - | - | - | - | - | 48 | −12 | −38 | 7.84 | 365 | r ITG | 20 | |
| 52 | 8 | −32 | 7.60 | 198 | r MTG | 21 | - | - | - | - | - | - | - | |
| Clinical Scales | Contrast | X | Y | z | T | Cluster | Region | BA | |
|---|---|---|---|---|---|---|---|---|---|
| TAS-20 | F2 | D > N | 10 | −72 | 54 | 6.24 | 239 | r SPL | 7 |
| −12 | −70 | 54 | 7.23 | 370 | l SPL | 7 | |||
| Clinical Scales | Contrast | X | Y | Z | T | Cluster | Region | BA | |
|---|---|---|---|---|---|---|---|---|---|
| TAS−20 | F1 | D > N | 48 | −12 | −36 | 7.20 | 133 | r ITG | 20 |
| H > N | 42 | −72 | −8 | 8.35 | 133 | r IOG | 19 | ||
| 40 | −66 | 0 | 8.89 | 142 | r FuG | 37 | |||
| F3 | D > N | 48 | −12 | −38 | 7.32 | 269 | r ITG | 20 | |
| H > N | 42 | −72 | −8 | 8.35 | 111 | r IOG | 19 | ||
| 46 | −16 | −34 | 10.55 | 153 | r ITG | 20 | |||
| A > N | 48 | −12 | −38 | 7.84 | 306 | r ITG | 20 | ||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Trentini, C.; Pagani, M.; Lauriola, M.; Tambelli, R. Neural Responses to Infant Emotions and Emotional Self-Awareness in Mothers and Fathers during Pregnancy. Int. J. Environ. Res. Public Health 2020, 17, 3314. https://doi.org/10.3390/ijerph17093314
Trentini C, Pagani M, Lauriola M, Tambelli R. Neural Responses to Infant Emotions and Emotional Self-Awareness in Mothers and Fathers during Pregnancy. International Journal of Environmental Research and Public Health. 2020; 17(9):3314. https://doi.org/10.3390/ijerph17093314
Chicago/Turabian StyleTrentini, Cristina, Marco Pagani, Marco Lauriola, and Renata Tambelli. 2020. "Neural Responses to Infant Emotions and Emotional Self-Awareness in Mothers and Fathers during Pregnancy" International Journal of Environmental Research and Public Health 17, no. 9: 3314. https://doi.org/10.3390/ijerph17093314
APA StyleTrentini, C., Pagani, M., Lauriola, M., & Tambelli, R. (2020). Neural Responses to Infant Emotions and Emotional Self-Awareness in Mothers and Fathers during Pregnancy. International Journal of Environmental Research and Public Health, 17(9), 3314. https://doi.org/10.3390/ijerph17093314





