New Intravenous Calcimimetic Agents: New Options, New Problems. An Example on How Clinical, Economical and Ethical Considerations Affect Choice of Treatment
Abstract
1. Introduction
2. Materials and Methods
2.1. Setting and Patients
2.2. Selection Criteria and Treatment Evaluation
2.3. Statistical Analysis
2.4. Ethical Discussion
2.5. Ethical Issues
3. Results
3.1. Overall Data
3.2. Overall Indications for Treatment
3.3. Ethical Issues: Insufficient Efficacy with or without Low Tolerance to Oral Calcimimetic Agents
3.4. Ethical Issues: Non Compliance
3.5. Ethical Issues: Other Complex Indications
3.6. Clinical Results: Why Treatment Is Being Continued
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| CCI | Charlson Comorbidity Index |
| CKD | MBD Chronic Kidney Disease–Mineral and Bone Disorder |
| HD | Haemodilaysis |
| HDF | Haemodiafiltration |
| IQR | Inter-quartile range |
| MIS | Malnutrition Inflammation Score |
| PTH | Parathyroid hormone |
| RRT | Renal replacement therapy |
| SD | Standard deviation |
| SGA | Subjective Global Assessment |
References
- Yamamoto, S.; Kazama, J.; Maruyama, H.; Nishi, S.; Narita, I.; Gejyo, F. Patients undergoing dialysis therapy for 30 years or more survive with serious osteoarticular disorders. Clin. Nephrol. 2008, 70, 496–502. [Google Scholar] [CrossRef]
- Otsubo, S.; Kimata, N.; Okutsu, I.; Oshikawa, K.; Ueda, S.; Sugimoto, H.; Mitobe, M.; Uchida, K.; Otsubo, K.; Nitta, K.; et al. Characteristics of dialysis-related amyloidosis in patients on haemodialysis therapy for more than 30 years. Nephrol. Dial. Transplant. 2009, 24, 1593–1598. [Google Scholar] [CrossRef] [PubMed]
- Otsubo, S.; Otsubo, K.; Sugimoto, H.; Ueda, S.; Otsubo, Y.; Otsubo, O.; Yajima, A.; Yagi, S.; Kataoka, H.; Iwasaki, T.; et al. Characteristics of Patients on Hemodialysis Therapy for More Than 30 Years. Ther. Apher. Dial. 2007, 11, 274–279. [Google Scholar] [CrossRef] [PubMed]
- Harris, S.A.; Brown, E. Patients surviving more than 10 years on haemodialysis. The natural history of the complications of treatment. Nephrol. Dial. Transplant. 1998, 13, 1226–1233. [Google Scholar] [CrossRef] [PubMed]
- Piccoli, G.B.; Mezza, E.; Anania, P.; Iadarola, A.M.; Vischi, M.; Torazza, M.C.; Fop, F.; Guarena, C.; Martina, G.; Messina, M.; et al. Patients on renal replacement therapy for 20 or more years: A clinical profile. Nephrol. Dial. Transplant. 2002, 17, 1440–1449. [Google Scholar] [CrossRef]
- Heaf, J.; Nielsen, A.H.; Hansen, H.P. Long-term haemodialysis survival. Clin. Kidney J. 2012, 5, 168–169. [Google Scholar] [CrossRef]
- Cozzolino, M.; Urena-Torres, P.; Vervloet, M.G.; Brandenburg, V.; Bover, J.; Goldsmith, D.; Larsson, T.E.; Massy, Z.A.; Mazzaferro, S.; on behalf of the CKD-MBD Working Group of ERA-EDTA. Is chronic kidney disease-mineral bone disorder (CKD-MBD) really a syndrome? Nephrol. Dial. Transplant. 2014, 29, 1815–1820. [Google Scholar] [CrossRef]
- Reiss, A.B.; Miyawaki, N.; Moon, J.; Kasselman, L.J.; Voloshyna, I.; D’Avino, R.; De Leon, J. CKD, arterial calcification, atherosclerosis and bone health: Inter-relationships and controversies. Atherosclerosis 2018, 278, 49–59. [Google Scholar] [CrossRef]
- Lunyera, J.; Scialla, J.J. Update on Chronic Kidney Disease Mineral and Bone Disorder in Cardiovascular Disease. Semin. Nephrol. 2018, 38, 542–558. [Google Scholar] [CrossRef]
- Rao, D.S.; Shih, M.-S.; Mohini, R. Effect of Serum Parathyroid Hormone and Bone Marrow Fibrosis on the Response to Erythropoietin in Uremia. N. Engl. J. Med. 1993, 328, 171–175. [Google Scholar] [CrossRef]
- Tanaka, M.; Komaba, H.; Fukagawa, M. Emerging Association between Parathyroid Hormone and Anemia in Hemodialysis Patients. Ther. Apher. Dial. 2018, 22, 242–245. [Google Scholar] [CrossRef] [PubMed]
- Altay, H.; Altın, C.; Coner, A.; Muderrisoglu, H.; Giray, S. Parathyroid Hormone and Ischemic Cerebrovascular Event. Endocr. Metab. Immune Disord. Drug Targets 2019, 19, 1134–1140. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Zhang, D.-Z. Circulating parathyroid hormone and risk of hypertension: A meta-analysis. Clin. Chim. Acta 2018, 482, 40–45. [Google Scholar] [CrossRef] [PubMed]
- Sofronie, A.C.; Kooij, I.; Bursot, C.; Santagati, G.; Coindre, J.-P.; Piccoli, G.B. Full normalization of severe hypertension after parathryoidectomy—A case report and systematic review. BMC Nephrol. 2018, 19, 112. [Google Scholar] [CrossRef] [PubMed]
- Park, J.H.; Kim, S.B.; Shin, H.S.; Jung, G.H.; Jung, Y.S.; Rim, H. Spontaneous and serial rupture of both Achilles tendons associated with secondary hyperparathyroidism in a patient receiving long-term hemodialysis. Int. Urol. Nephrol. 2013, 45, 587–590. [Google Scholar] [CrossRef] [PubMed]
- Chou, F.-F.; Chen, J.-B.; Hsieh, K.-C.; Liou, C.-W. Cognitive changes after parathyroidectomy in patients with secondary hyperparathyroidism. Surgery 2008, 143, 526–532. [Google Scholar] [CrossRef]
- Lourida, I.; Thompson-Coon, J.; Dickens, C.M.; Soni, M.; Kuźma, E.; Kos, K.; Llewellyn, D.J. Parathyroid Hormone, Cognitive Function and Dementia: A Systematic Review. PLoS ONE 2015, 10, e0127574. [Google Scholar] [CrossRef]
- Cermik, T.F.; Kaya, M.; Ugur-Altun, B.; Bedel, D.; Berkarda, S.; Yiğitbaşı, Ö.N.; Yigitbasi, O.N. Regional cerebral blood flow abnormalities in patients with primary hyperparathyroidism. Neuroradiology 2007, 49, 379–385. [Google Scholar] [CrossRef]
- Papageorgiou, S.G.; Christou, Y.; Kontaxis, T.; Bonakis, A.; Anagnostouli, M.; Potagas, C.; Kalfakis, N. Dementia as presenting symptom of primary hyperparathyroidism: Favourable outcome after surgery. Clin. Neurol. Neurosurg. 2008, 110, 1038–1040. [Google Scholar] [CrossRef]
- Goldstein, D.A.; Chui, L.A.; Massry, S.G. Effect of Parathyroid Hormone and Uremia on Peripheral Nerve Calcium and Motor Nerve Conduction Velocity. J. Clin. Investig. 1978, 62, 88–93. [Google Scholar] [CrossRef]
- Massry, S.G. Neurotoxicity of parathyroid hormone in uremia. Kidney Int. Suppl. 1985, 17, S5–S11. [Google Scholar] [PubMed]
- Komaba, H.; Fukagawa, M. Secondary Hyperparathyroidism and Protein-Energy Wasting in End-Stage Renal Disease. Ther. Apher. Dial. 2018, 22, 246–250. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, S.; Karaboyas, A.; Komaba, H.; Taniguchi, M.; Nomura, T.; Bieber, B.A.; De Sequera, P.; Christensson, A.; Pisoni, R.L.; Robinson, B.M.; et al. Mineral and bone disorder management in hemodialysis patients: Comparing PTH control practices in Japan with Europe and North America: The Dialysis Outcomes and Practice Patterns Study (DOPPS). BMC Nephrol. 2018, 19, 253. [Google Scholar] [CrossRef] [PubMed]
- Tentori, F.; Blayney, M.J.; Albert, J.M.; Gillespie, B.W.; Kerr, P.G.; Bommer, J.; Young, E.W.; Akizawa, T.; Akiba, T.; Pisoni, R.L.; et al. Mortality Risk for Dialysis Patients With Different Levels of Serum Calcium, Phosphorus, and PTH: The Dialysis Outcomes and Practice Patterns Study (DOPPS). Am. J. Kidney Dis. 2008, 52, 519–530. [Google Scholar] [CrossRef] [PubMed]
- Kimata, N.; Albert, J.M.; Akiba, T.; Yamazaki, S.; Kawaguchi, Y.; Fukuhara, S.; Akizawa, T.; Saito, A.; Asano, Y.; Kurokawa, K.; et al. Association of mineral metabolism factors with all-cause and cardiovascular mortality in hemodialysis patients: The Japan dialysis outcomes and practice patterns study. Hemodial. Int. 2007, 11, 340–348. [Google Scholar] [CrossRef] [PubMed]
- Avram, M.M.; Mittman, N.; Myint, M.M.; Fein, P. Importance of low serum intact parathyroid hormone as a predictor of mortality in hemodialysis and peritoneal dialysis patients: 14 years of prospective observation. Am. J. Kidney Dis. 2001, 38, 1351–1357. [Google Scholar] [CrossRef]
- Tentori, F.; Wang, M.; Bieber, B.A.; Karaboyas, A.; Li, Y.; Jacobson, S.H.; Andreucci, V.E.; Fukagawa, M.; Frimat, L.; Mendelssohn, D.C.; et al. Recent changes in therapeutic approaches and association with outcomes among patients with secondary hyperparathyroidism on chronic hemodialysis: The DOPPS study. Clin. J. Am. Soc. Nephrol. 2015, 7, 98–109. [Google Scholar] [CrossRef]
- Saab, G.; Navaneethan, S.D.; Slatopolsky, E. Targeting Parathyroid Hormone Levels in Dialysis Patients. Semin. Dial. 2014, 27, 562–565. [Google Scholar] [CrossRef]
- Ketteler, M.; Block, G.A.; Evenepoel, P.; Fukagawa, M.; Herzog, C.A.; McCann, L.; Moe, S.M.; Shroff, R.; Tonelli, M.A.; Toussaint, N.D.; et al. Diagnosis, Evaluation, Prevention, and Treatment of Chronic Kidney Disease–Mineral and Bone Disorder: Synopsis of the Kidney Disease: Improving Global Outcomes 2017 Clinical Practice Guideline Update. Ann. Intern. Med. 2018, 168, 422. [Google Scholar] [CrossRef]
- Isakova, T.; Nickolas, T.L.; Denburg, M.; Yarlagadda, S.; Weiner, D.E.; Gutiérrez, O.M.; Bansal, V.; Rosas, S.E.; Nigwekar, S.; Yee, J.; et al. KDOQI US Commentary on the 2017 KDIGO Clinical Practice Guideline Update for the Diagnosis, Evaluation, Prevention, and Treatment of Chronic Kidney Disease–Mineral and Bone Disorder (CKD-MBD). Am. J. Kidney Dis. 2017, 70, 737–751. [Google Scholar] [CrossRef]
- Kidney Disease: Improving Global Outcomes (KDIGO) CKD-MBD Update Work Group. KDIGO 2017 Clinical Practice Guideline Update for the Diagnosis, Evaluation, Prevention, and Treatment of Chronic Kidney Disease–Mineral and Bone Disorder (CKD-MBD). Kidney Int. Suppl. 2017, 7, 1–59. [Google Scholar] [CrossRef] [PubMed]
- Etelcalcetide for Treating Secondary Hyperparathyroidism: Technology Appraisal Guidance [TA448]; National Institute for Health and Care Excellence: London, UK, 2017.
- Lin, E.; Watnick, S. Calcimimetics and Bundled Reimbursement. Am. J. Kidney Dis. 2019, 73, 385–390. [Google Scholar] [CrossRef] [PubMed]
- Piccoli, G.B.; Cabiddu, G.; Breuer, C.; Jadeau, C.; Testa, A.; Brunori, G. Dialysis Reimbursement: What Impact Do Different Models Have on Clinical Choices? J. Clin. Med. 2019, 8, 276. [Google Scholar] [CrossRef] [PubMed]
- Fuller, D.S.; Xing, S.; Belozeroff, V.; Yehoshua, A.; Morgenstern, H.; Robinson, B.M.; Rubin, R.J.; Bhatt, N.; Pisoni, R.L. Variability in Cinacalcet Prescription across US Hemodialysis Facilities. Clin. J. Am. Soc. Nephrol. 2019, 14, 241–249. [Google Scholar] [CrossRef]
- Spoendlin, J.; Schneeweiss, S.; Tsacogianis, T.; Paik, J.M.; Fischer, M.A.; Kim, S.C.; Desai, R.J. Association of Medicare’s Bundled Payment Reform With Changes in Use of Vitamin D Among Patients Receiving Maintenance Hemodialysis: An Interrupted Time-Series Analysis. Am. J. Kidney Dis. 2018, 72, 178–187. [Google Scholar] [CrossRef] [PubMed]
- Turenne, M.; Baker, R.; Pearson, J.; Cogan, C.; Mukhopadhyay, P.; Cope, E. Payment Reform and Health Disparities: Changes in Dialysis Modality under the New Medicare Dialysis Payment System. Health Serv. Res. 2018, 53, 1430–1457. [Google Scholar] [CrossRef]
- Karaboyas, A.; Zee, J.; Morgenstern, H.; Nolen, J.G.; Hakim, R.; Kalantar-Zadeh, K.; Zager, P.; Pisoni, R.L.; Port, F.K.; Robinson, B.M. Understanding the Recent Increase in Ferritin Levels in United States Dialysis Patients: Potential Impact of Changes in Intravenous Iron and Erythropoiesis-Stimulating Agent Dosing. Clin. J. Am. Soc. Nephrol. 2015, 10, 1814–1821. [Google Scholar] [CrossRef]
- Swaminathan, S.; Mor, V.; Mehrotra, R.; Trivedi, A.N. Effect of Medicare dialysis payment reform on use of erythropoiesis stimulating agents. Health Serv. Res. 2015, 50, 790–808. [Google Scholar] [CrossRef]
- Jovanovich, A.; Kendrick, J. Personalized Management of Bone and Mineral Disorders and Precision Medicine in End-Stage Kidney Disease. Semin. Nephrol. 2018, 38, 397–409. [Google Scholar] [CrossRef]
- Hall, Y.N.; Himmelfarb, J. The CKD Classification System in the Precision Medicine Era. Clin. J. Am. Soc. Nephrol. 2017, 12, 346–348. [Google Scholar] [CrossRef]
- Société Francophone de Néphrologie Dialyse et Transplantation (SFNDT). Report on Chronic Dialysis in France in 2016; Société Francophone de Néphrologie Dialyse et Transplantation: Saint-Sorlin, France, May 2016. [Google Scholar]
- Piccoli, G.B.; Cabiddu, G.; Moio, M.R.; Fois, A.; Cao, R.; Molfino, I.; Kaniassi, A.; Lippi, F.; Froger, L.; Pani, A.; et al. Efficiency and nutritional parameters in an elderly high risk population on hemodialysis and hemodiafiltration in Italy and France: Different treatments with similar names? BMC Nephrol. 2018, 19, 171. [Google Scholar] [CrossRef] [PubMed]
- Piccoli, G.B.; Nielsen, L.; Gendrot, L.; Fois, A.; Cataldo, E.; Cabiddu, G. Prescribing Hemodialysis or Hemodiafiltration: When One Size Does Not Fit All the Proposal of a Personalized Approach Based on Comorbidity and Nutritional Status. J. Clin. Med. 2018, 7, 331. [Google Scholar] [CrossRef]
- Cupisti, A.; Brunori, G.; Di Iorio, B.R.; D’Alessandro, C.; Pasticci, F.; Cosola, C.; Bellizzi, V.; Bolasco, P.; Capitanini, A.; Fantuzzi, A.L.; et al. Nutritional treatment of advanced CKD: Twenty consensus statements. J. Nephrol. 2018, 31, 457–473. [Google Scholar] [CrossRef]
- Gillon, R. Medical ethics: Four principles plus attention to scope. BMJ 1994, 309, 184. [Google Scholar] [CrossRef] [PubMed]
- McCarthy, J. Principlism or narrative ethics: Must we choose between them? Med. Humanit. 2003, 29, 65–71. [Google Scholar] [CrossRef] [PubMed]
- Butler, C.R.; Mehrotra, R.; Tonelli, M.R.; Lam, D.Y. The Evolving Ethics of Dialysis in the United States: A Principlist Bioethics Approach. Clin. J. Am. Soc. Nephrol. 2016, 11, 704–709. [Google Scholar] [CrossRef]
- Paris, J.J. Rationing: A “decent minimum” or a “consumer driven” health care system? Am. J. Bioeth. 2011, 11, 16–18. [Google Scholar] [CrossRef]
- Anonymous. Who owns medical technology? Lancet 1995, 345, 1125–1126. [Google Scholar] [CrossRef]
- Block, G.A.; Bushinsky, D.A.; Cheng, S.; Cunningham, J.; Dehmel, B.; Drueke, T.B.; Ketteler, M.; Kewalramani, R.; Martin, K.J.; Moe, S.M.; et al. Effect of Etelcalcetide vs. Cinacalcet on Serum Parathyroid Hormone in Patients Receiving Hemodialysis With Secondary Hyperparathyroidism: A Randomized Clinical Trial. JAMA 2017, 317, 156–164. [Google Scholar] [CrossRef]
- Block, G.A.; Bushinsky, D.A.; Cunningham, J.; Drueke, T.B.; Ketteler, M.; Kewalramani, R.; Martin, K.J.; Mix, T.C.; Moe, S.M.; Patel, U.D.; et al. Effect of Etelcalcetide vs. Placebo on Serum Parathyroid Hormone in Patients Receiving Hemodialysis With Secondary Hyperparathyroidism: Two Randomized Clinical Trials. JAMA 2017, 317, 146–155. [Google Scholar] [CrossRef]
- Block, G.A.; Chertow, G.M. Dosing of Etelcalcetide vs Cinacalcet for Secondary Hyperparathyroidism-Reply. JAMA 2017, 317, 2132–2133. [Google Scholar] [CrossRef] [PubMed]
- Hai, M.T.T.; Guettier, J.-M.; Rosebraugh, C.J. Dosing of Etelcalcetide and Cinacalcet for Secondary Hyperparathyroidism. JAMA 2017, 317, 2132. [Google Scholar] [CrossRef]
- Fukagawa, M.; Yokoyama, K.; Shigematsu, T.; Akiba, T.; Fujii, A.; Kuramoto, T.; Odani, M.; Akizawa, T.; ONO-5163 Study Group. A phase 3, multicentre, randomized, double-blind, placebo-controlled, parallel-group study to evaluate the efficacy and safety of etelcalcetide (ONO-5163/AMG 416), a novel intravenous calcimimetic, for secondary hyperparathyroidism in Japanese haemodialysis patients. Nephrol. Dial. Transplant. 2017, 32, 1723–1730. [Google Scholar]
- Xipell, M.; Montagud-Marrahi, E.; Rubio, M.V.; Ojeda, R.; Arias-Guillén, M.; Fontseré, N.; Rodas, L.; Vera, M.; Broseta, J.J.; Torregrosa, V.; et al. Improved Control of Secondary Hyperparathyroidism in Hemodialysis Patients Switching from Oral Cinacalcet to Intravenous Etelcalcetide, Especially in Nonadherent Patients. Blood Purif. 2019, 48, 106–114. [Google Scholar] [CrossRef]
- Shigematsu, T.; Fukagawa, M.; Yokoyama, K.; Akiba, T.; Fujii, A.; Odani, M.; Akizawa, T. Effects of the Intravenous Calcimimetic Etelcalcetide on Bone Turnover and Serum Fibroblast Growth Factor 23: Post Hoc Analysis of an Open-label Study. Clin. Ther. 2018, 40, 2099–2111. [Google Scholar] [CrossRef] [PubMed]
- Harada, K.; Fujioka, A.; Konno, M.; Inoue, A.; Yamada, H.; Hirota, Y. Pharmacology of Parsabiv® (etelcalcetide, ONO-5163/AMG 416), a novel allosteric modulator of the calcium-sensing receptor, for secondary hyperparathyroidism in hemodialysis patients. Eur. J. Pharmacol. 2019, 842, 139–145. [Google Scholar] [CrossRef] [PubMed]
- Mima, A.; Tansho, K.; Nagahara, D.; Watase, K. Treatment of secondary hyperparathyroidism in patients on hemodialysis using a novel synthetic peptide calcimimetic, etelcalcetide: A short-term clinical study. J. Int. Med. Res. 2018, 46, 4578–4585. [Google Scholar] [CrossRef]
- Yoshimura, K.; Funakoshi, Y.; Terawaki, H. Dramatic Regression of Parathyroid Gland Swelling After Conversion of Calcimimetic Medication From Cinacalcet to Etelcalcetide. Ther. Apher. Dial. 2018, 22, 553–554. [Google Scholar] [CrossRef]
- Yokoyama, K.; Fukagawa, M.; Shigematsu, T.; Akiba, T.; Fujii, A.; Yamauchi, A.; Odani, M.; Akizawa, T.; ONO-5163 Study Group. A Single- and Multiple-Dose, Multicenter Study of Etelcalcetide in Japanese Hemodialysis Patients With Secondary Hyperparathyroidism. Kidney Int. Rep. 2017, 2, 634–644. [Google Scholar] [CrossRef]
- Shigematsu, T.; Fukagawa, M.; Yokoyama, K.; Akiba, T.; Fujii, A.; Odani, M.; Akizawa, T. ONO-5163 Study Group. Long-term effects of etelcalcetide as intravenous calcimimetic therapy in hemodialysis patients with secondary hyperparathyroidism. Clin. Exp. Nephrol. 2018, 22, 426–436. [Google Scholar] [CrossRef]
- Harada, K.; Inoue, A.; Yamauchi, A.; Fujii, A. The pharmacological profile and the clinical efficacy of the world’s 1st intravenous calcimimetics; etelcalcetide hydrochloride (Parsabiv®). Folia Pharmacol. Jpn. 2017, 150, 98–113. [Google Scholar] [CrossRef] [PubMed]
- Yokoyama, K.; Fukagawa, M.; Shigematsu, T.; Akiba, T.; Fujii, A.; Odani, M.; Akizawa, T. A 12-week dose-escalating study of etelcalcetide (ONO-5163/AMG 416), a novel intravenous calcimimetic, for secondary hyperparathyroidism in Japanese hemodialysis patients. Clin. Nephrol. 2017, 88, 68–78. [Google Scholar] [CrossRef] [PubMed]
- Patel, J.; Bridgeman, M.B. Etelcalcetide (Parsabiv) for Secondary Hyperparathyroidism in Adults with Chronic Kidney Disease on Hemodialysis. Pharm. Ther. 2018, 43, 396–399. [Google Scholar]
- Eidman, K.E.; Wetmore, J.B. Treatment of secondary hyperparathyroidism: How do cinacalcet and etelcalcetide differ? Semin. Dial. 2018, 31, 440–444. [Google Scholar] [CrossRef]
- Friedl, C.; Zitt, E. Role of etelcalcetide in the management of secondary hyperparathyroidism in hemodialysis patients: A review on current data and place in therapy. Drug Des. Dev. Ther. 2018, 12, 1589–1598. [Google Scholar] [CrossRef]
- Ye, J.; Deng, G.; Gao, F. Theoretical overview of clinical and pharmacological aspects of the use of etelcalcetide in diabetic patients undergoing hemodialysis. Drug Des. Dev. Ther. 2018, 12, 901–909. [Google Scholar] [CrossRef]
- Nemeth, E.F.; Van Wagenen, B.C.; Balandrin, M.F. Discovery and Development of Calcimimetic and Calcilytic Compounds. Prog. Med. Chem. 2018, 57, 1–86. [Google Scholar]
- Ochoa, P.V.; Heras, M.S.; Balcázar, A.Z.; Escudero, P.S.; Palomares, J.R.R.; Fuente, G.D.A.D.L. A light in the control of secondary hyperparathyroidism. Etelcalcetide IV in haemodialysis. Nefrología (Engl. Ed.) 2018, 38, 677–678. [Google Scholar] [CrossRef]
- Wu, B.; Melhem, M.; Subramanian, R.; Chen, P.; Sloey, B.J.; Fouqueray, B.; Hock, M.B.; Skiles, G.L.; Chow, A.T.; Lee, E. Clinical Pharmacokinetics and Pharmacodynamics of Etelcalcetide, a Novel Calcimimetic for Treatment of Secondary Hyperparathyroidism in Patients With Chronic Kidney Disease on Hemodialysis. J. Clin. Pharmacol. 2018, 58, 717–726. [Google Scholar] [CrossRef]
- Eidman, K.E.; Wetmore, J.B. Managing hyperparathyroidism in hemodialysis: Role of etelcalcetide. Int. J. Nephrol. Renov. Dis. 2018, 11, 69–80. [Google Scholar] [CrossRef]
- Pereira, L.; Meng, C.; Marques, D.; Frazão, J.M. Old and new calcimimetics for treatment of secondary hyperparathyroidism: Impact on biochemical and relevant clinical outcomes. Clin. Kidney J. 2018, 11, 80–88. [Google Scholar] [CrossRef] [PubMed]
- Baker, D.E. Formulary Drug Review: Etelcalcetide. Hosp. Pharm. 2017, 52, 669–674. [Google Scholar] [CrossRef]
- Ureña Torres, P.A.; Bover, J.; Cohen-Solal, M. Etelcalcetide: Injectable calcimimetic for the treatment of secondary hyperparathyroidism in hemodialysis-dependent patients. Drugs Today (Barc.) 2017, 53, 489–500. [Google Scholar] [CrossRef] [PubMed]
- Yu, L.; Tomlinson, J.E.; Alexander, S.T.; Hensley, K.; Han, C.-Y.; Dwyer, D.; Stolina, M.; Dean, C.; Goodman, W.G.; Richards, W.G.; et al. Etelcalcetide, A Novel Calcimimetic, Prevents Vascular Calcification in A Rat Model of Renal Insufficiency with Secondary Hyperparathyroidism. Calcif. Tissue Int. 2017, 101, 641–653. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Yu, L.; Asuncion, F.; Grisanti, M.; Alexander, S.; Hensley, K.; Han, C.-Y.; Niu, Q.-T.; Dwyer, D.; Villasenor, K.; et al. Etelcalcetide (AMG 416), a peptide agonist of the calcium-sensing receptor, preserved cortical bone structure and bone strength in subtotal nephrectomized rats with established secondary hyperparathyroidism. Bone 2017, 105, 163–172. [Google Scholar] [CrossRef]
- Cozzolino, M.; Galassi, A.; Conte, F.; Mangano, M.; Di Lullo, L.; Bellasi, A. Treatment of secondary hyperparathyroidism: The clinical utility of etelcalcetide. Ther. Clin. Risk Manag. 2017, 13, 679–689. [Google Scholar] [CrossRef]
- Chen, P.; Narayanan, A.; Wu, B.; Gisleskog, P.O.; Gibbs, J.P.; Chow, A.T.; Melhem, M. Population Pharmacokinetic and Pharmacodynamic Modeling of Etelcalcetide in Patients with Chronic Kidney Disease and Secondary Hyperparathyroidism Receiving Hemodialysis. Clin. Pharm. 2018, 57, 71–85. [Google Scholar] [CrossRef]
- Hamano, N.; Komaba, H.; Fukagawa, M. Etelcalcetide for the treatment of secondary hyperparathyroidism. Expert Opin. Pharmacother. 2017, 18, 529–534. [Google Scholar] [CrossRef]
- Blair, H.A. Etelcalcetide: First Global Approval. Drugs 2016, 76, 1787–1792. [Google Scholar] [CrossRef]
- Chen, P.; Gisleskog, P.O.; Perez-Ruixo, J.J.; Xiao, J.; Wilkins, J.; Narayanan, A.; Gibbs, J.P.; Melhem, M. Population Pharmacokinetics and Pharmacodynamics of the Calcimimetic Etelcalcetide in Chronic Kidney Disease and Secondary Hyperparathyroidism Receiving Hemodialysis. CPT Pharmacomet. Syst. Pharmacol. 2016, 5, 484–494. [Google Scholar] [CrossRef]
- Subramanian, R.; Zhu, X.; Hock, M.B.; Sloey, B.J.; Wu, B.; Wilson, S.F.; Egbuna, O.; Slatter, J.G.; Xiao, J.; Skiles, G.L. Pharmacokinetics, Biotransformation, and Excretion of [14C]Etelcalcetide (AMG 416) Following a Single Microtracer Intravenous Dose in Patients with Chronic Kidney Disease on Hemodialysis. Clin. Pharm. 2017, 56, 179–192. [Google Scholar] [CrossRef] [PubMed]
- Edson, K.Z.; Wu, B.M.; Iyer, A.; Goodman, W.; Skiles, G.L.; Subramanian, R. Determination of Etelcalcetide Biotransformation and Hemodialysis Kinetics to Guide the Timing of Its Dosing. Kidney Int. Rep. 2016, 1, 24–33. [Google Scholar] [CrossRef]
- Bellasi, A.; Cozzolino, M.; Malberti, F.; Cancarini, G.; Esposito, C.; Guastoni, C.M.; Ondei, P.; Pontoriero, G.; Teatini, U.; Vezzoli, G.; et al. New scenarios in secondary hyperparathyroidism: etelcalcetide. Position paper of working group on CKD-MBD of the Italian Society of Nephrology. J. Nephrol. 2019. [Google Scholar] [CrossRef] [PubMed]
- Rose, M.; Shepherd, J.; Harris, P.; Pickett, K.; Lord, J. Etelcalcetide for Treating Secondary Hyperparathyroidism: An Evidence Review Group Evaluation of a NICE Single Technology Appraisal. PharmacoEconomics 2018, 36, 1299–1308. [Google Scholar] [CrossRef] [PubMed]
- Stollenwerk, B.; Iannazzo, S.; Akehurst, R.; Adena, M.; Briggs, A.; Dehmel, B.; Parfrey, P.; Belozeroff, V. A Decision-Analytic Model to Assess the Cost-Effectiveness of Etelcalcetide vs. Cinacalcet. PharmacoEconomics 2018, 36, 603–612. [Google Scholar] [CrossRef]
- Stollenwerk, B.; Iannazzo, S.; Cooper, K.; Belozeroff, V. Exploring the potential value of improved care for secondary hyperparathyroidism with a novel calcimimetic therapy. J. Med. Econ. 2017, 20, 1–6. [Google Scholar] [CrossRef]
- Morrow, T. Parsabiv Could Be Pricey Addition in Care for CKD, ESRD Patients. Manag. Care 2017, 26, 33–34. [Google Scholar] [PubMed]
- Grubbs, V.; Moss, A.H.; Cohen, L.M.; Fischer, M.J.; Germain, M.J.; Jassal, S.V.; Perl, J.; Weiner, D.E.; Mehrotra, R. Dialysis Advisory Group of the American Society of Nephrology. A palliative approach to dialysis care: A patient-centered transition to the end of life. Clin. J. Am. Soc. Nephrol. 2014, 9, 2203–2209. [Google Scholar] [CrossRef]
- Ubel, P.A.; Arnold, R.M.; Caplan, A.L. Rationing failure. The ethical lessons of the retransplantation of scarce vital organs. JAMA 1993, 270, 2469–2474. [Google Scholar] [CrossRef]
- Dobbels, F.; Hames, A.; Aujoulat, I.; Heaton, N.; Samyn, M. Should we retransplant a patient who is non-adherent? A literature review and critical reflection. Pediatr. Transplant. 2012, 16, 4–11. [Google Scholar] [CrossRef]
- Dunn, T.B.; Browne, B.J.; Gillingham, K.J.; Kandaswamy, R.; Humar, A.; Payne, W.D.; Sutherland, D.E.R.; Matas, A.J. Selective retransplant after graft loss to nonadherence: Success with a second chance. Arab. Archaeol. Epigr. 2009, 9, 1337–1346. [Google Scholar] [CrossRef] [PubMed]
- Huesch, M.D. One and done? Equality of opportunity and repeated access to scarce, indivisible medical resources. BMC Med. Ethic. 2012, 13, 11. [Google Scholar] [CrossRef] [PubMed]
- Hofmann, B. Stuck in the Middle: The Many Moral Challenges with Bariatric Surgery. Am. J. Bioeth. 2010, 10, 3–11. [Google Scholar] [CrossRef] [PubMed]
- Conly, S. The right to preventive health care. Theor. Med. Bioeth. 2016, 37, 307–321. [Google Scholar] [CrossRef]
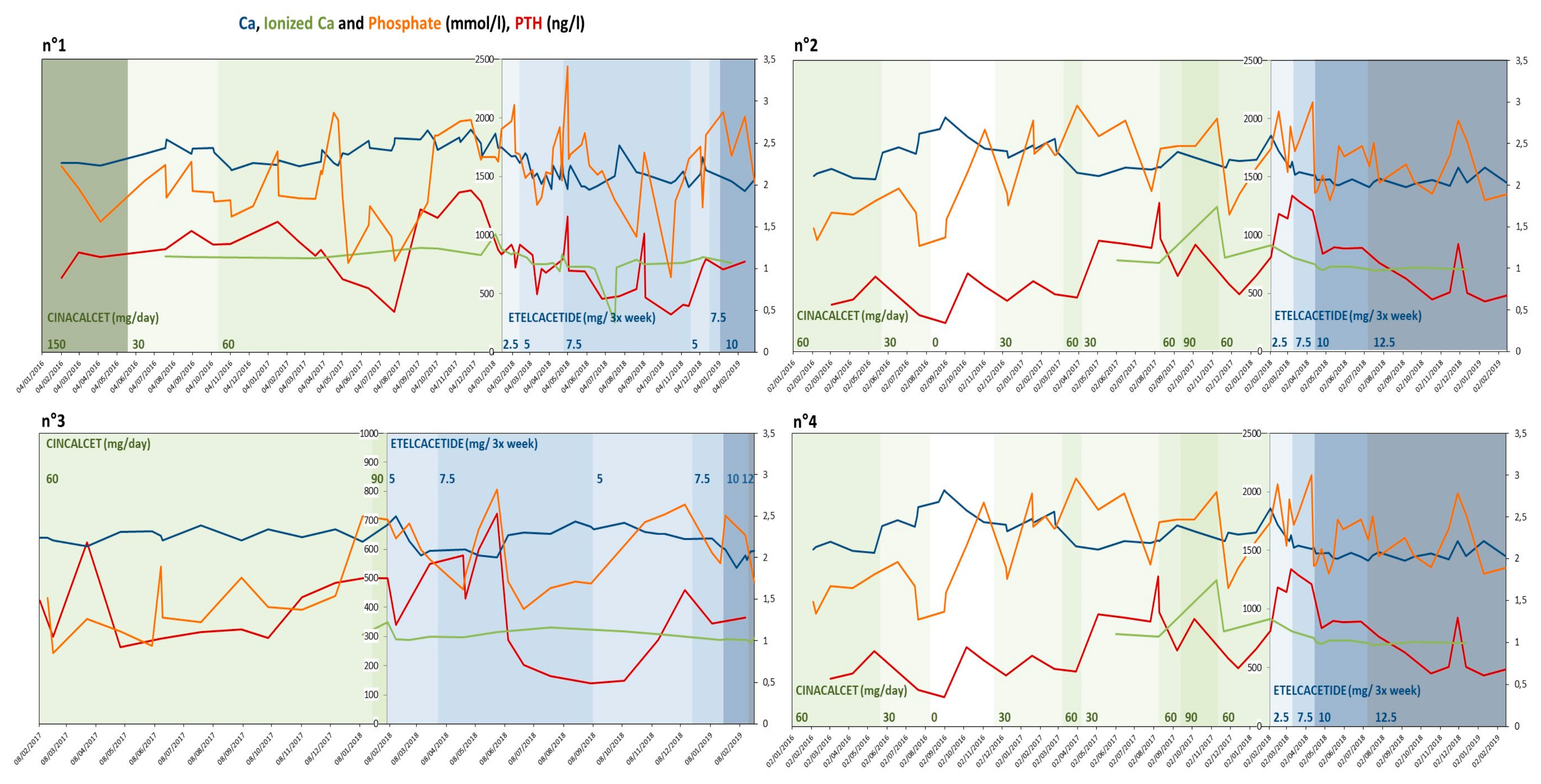
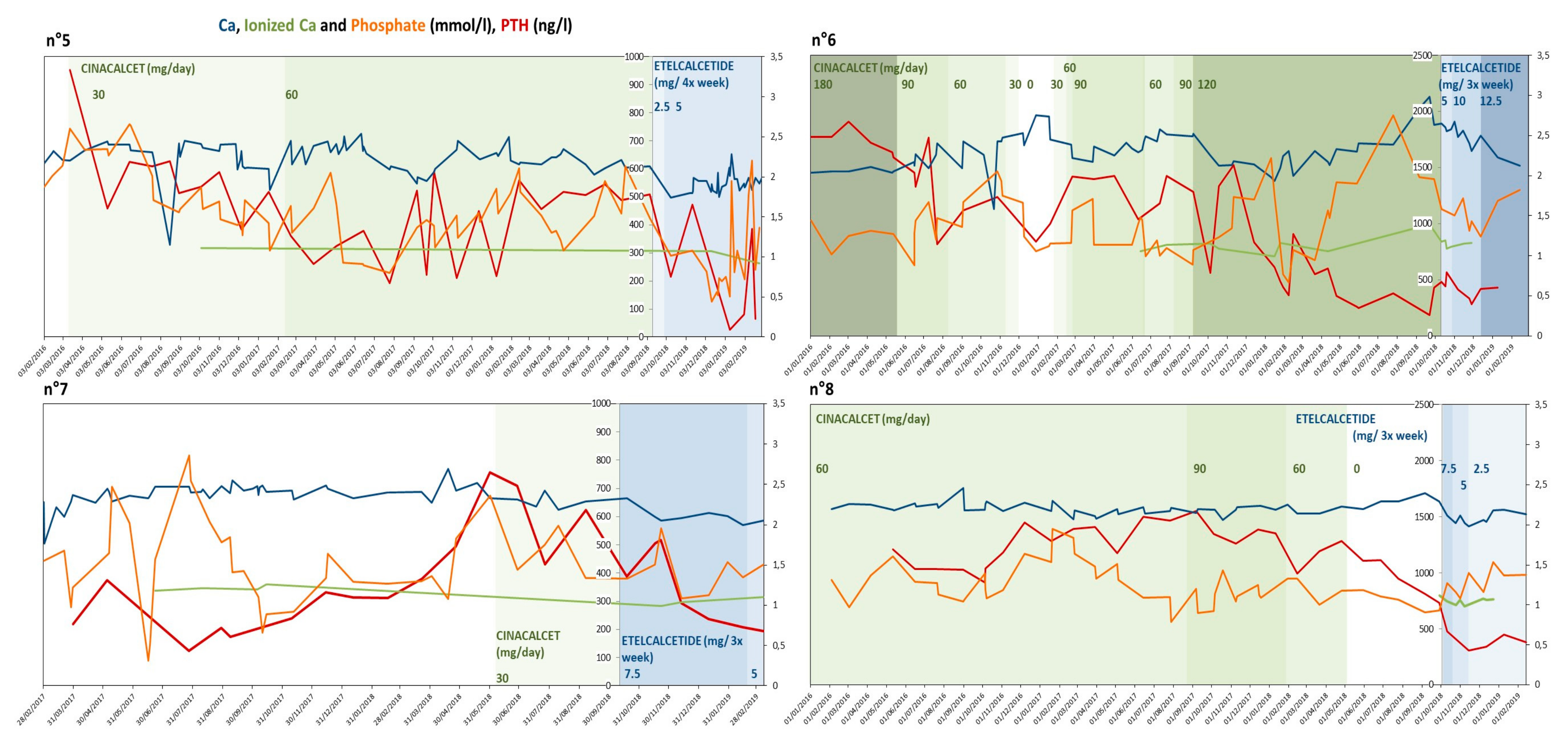
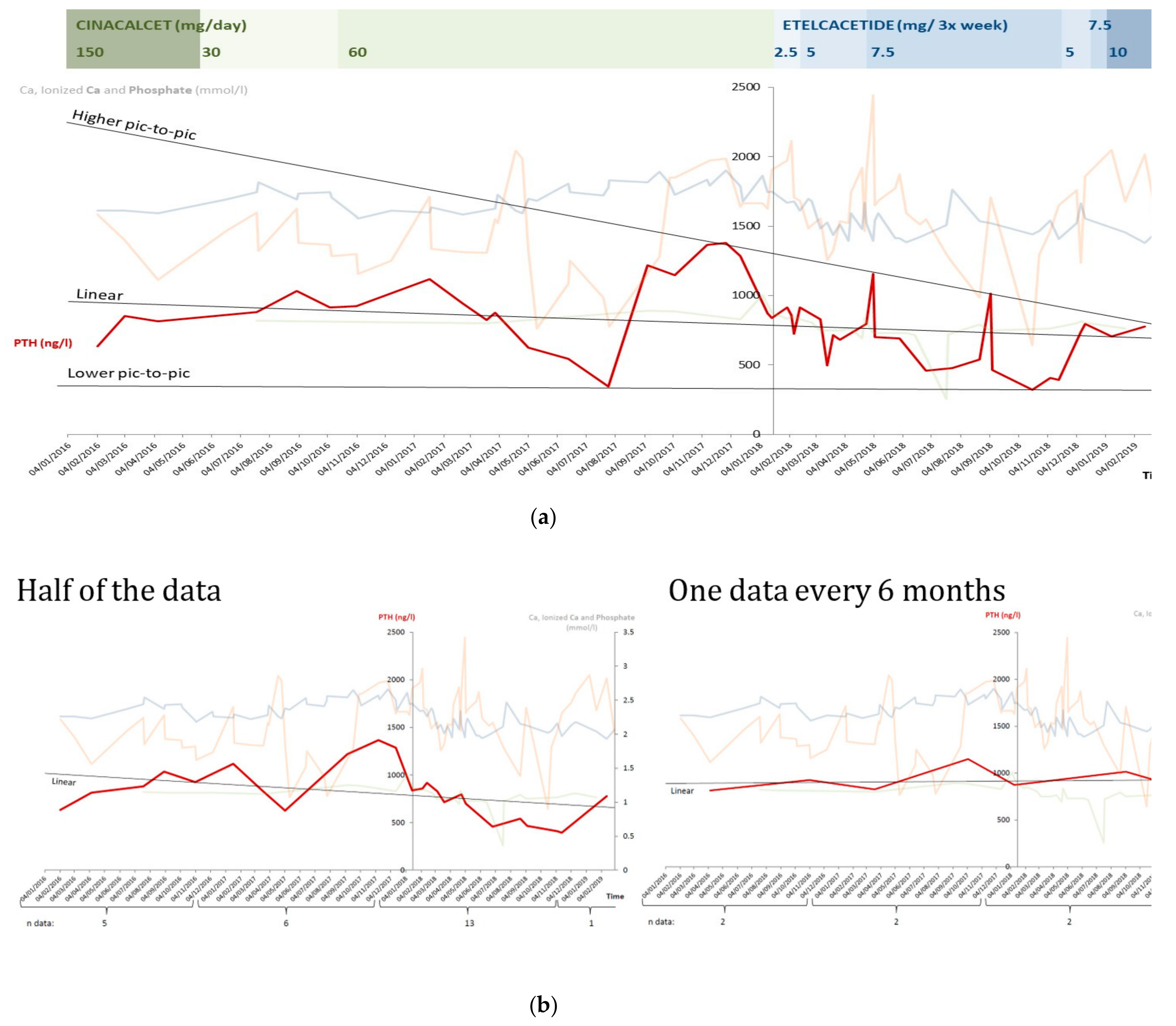
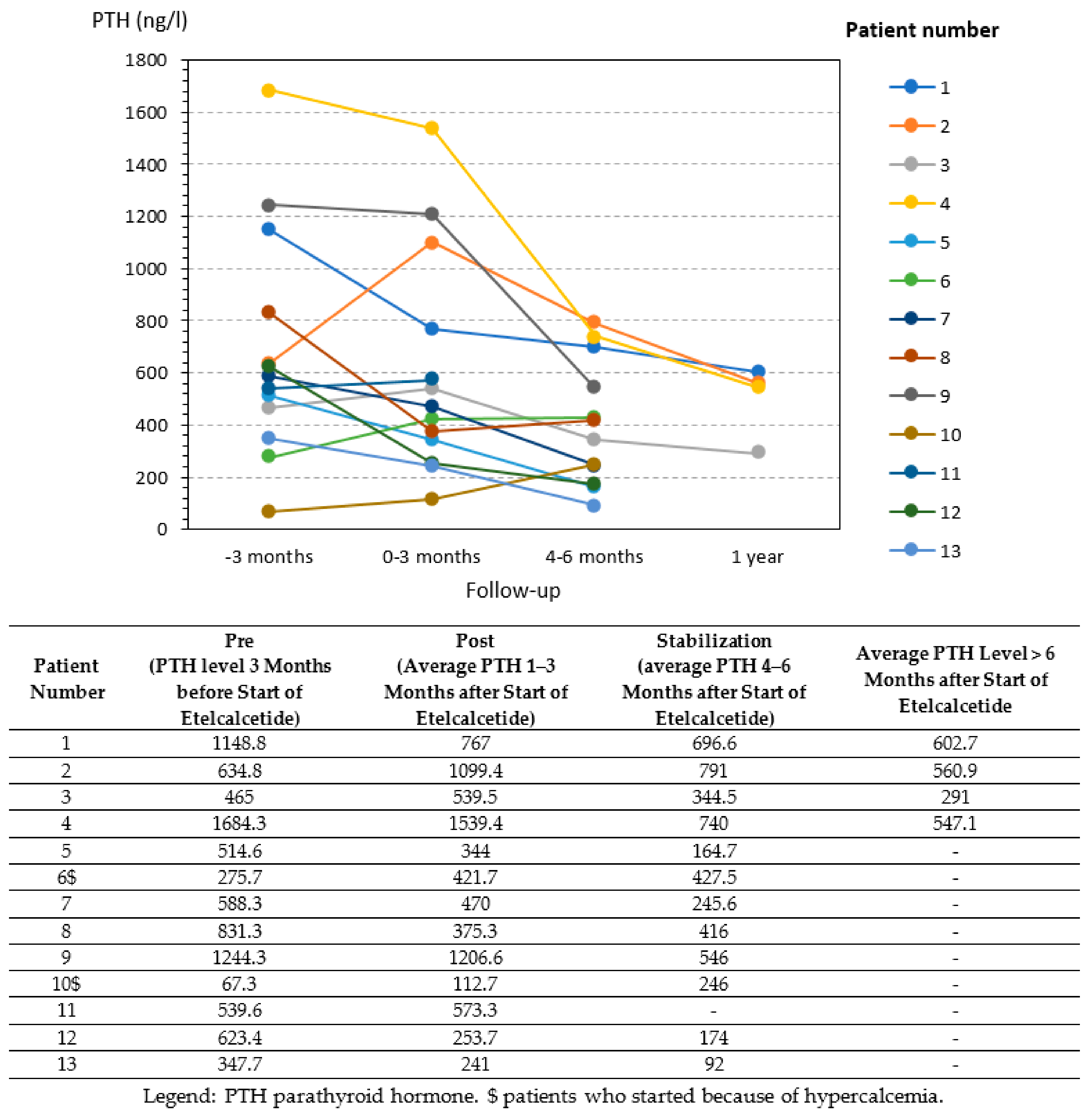
| Patient | Age | Sex | Start | CCI | Dialysis Vintage (years) | Main Indication for Etelcalcetide | Dialysis Schedule | Kt/V | Beta2M | Metabolic Bone Disease |
|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 78 | F | 01/2018 | 6 | 8 | Low tolerance, insufficient PTH control | HDF-Pre 4 h × 3 | 1.5 | 26.4 | Osteopenia |
| 2 | 82 | F | 02/2018 | 8 | 9 | Pathologic fractures, insufficient PTH control | HD 4 h × 3 | 1.9 | 27.4 | Pathologic fractures |
| 3 | 57 | M | 02/2018 | 3 | 15 | Insufficient PTH control, long RRT vintage | HDF post 4 h × 3 | 1.79 | 33.5 | B2M amyloidosis |
| 4 | 83 | F | 02/2018 | 10 | 12 | Insufficient PTH control | HDF post 3 h 30 × 3 | 1.59 | 23.5 | Osteopenia |
| 5 | 79 | F | 09/2018 | 10 | 7 | Insufficient PTH control, severe vascular disease | HDF Pre 3 h 30 × 4 | 1.50 | 18.6 | Osteopenia |
| 6 | 52 | M | 10/2018 | 6 | 11 | Need for high calcimimetic dose | HD 3 h 30 × 3 | 1.24 | 33.1 | B2M amyloidosis, Osteopenia, deformities |
| 7 | 59 | F | 10/2018 | 9 | 2 | Absorption problems, low tolerance, low compliance | HDF post 4 h × 3 | 1.30 | 36.6 | - |
| 8 | 35 | M | 10/2018 | 4 | 6 | Low compliance | HDF post 4 h × 3 | 1.33 | 29.6 | - |
| 9 | 37 | F | 11/2018 | 11 | 7 | Irregular compliance | HDF pre 4 h × 3 | 1.71 | 61.3 | - |
| 10 | 54 | F | 11/2018 to ** 02/2019 | 4 | 1 | Hypercalcemia, low tolerance | HD 4 h × 3 | 1.51 | 26.2 | - |
| 11 | 70 | F | 11/2018to * 23/2018 | 9 | 15 | Mild hypercalcemia, amyloidosis | HD 4 h × 3 | 1.6 | 14.5 | B2M amyloidosis, osteopenia |
| 12 | 43 | M | 11/2018 | 6 | 1 | Low compliance, diffuse calcifications | HD 4 h × 3 | 1.08 | 16.1 | - |
| 13 | 63 | M | 11/2018 | 7 | 20 | Insufficient PTH control, long RRT vintage | HDF post 4 h × 3 | 1.41 | 24.1 | B2M amyloidosis |
| 14 | 83 | F | 02/2019 | 9 | 10 | Low compliance, severe vascular disease | HDF pre 4 h × 3 | 1.32 | 31.1 | Osteopenia |
| 15 | 67 | F | 02/2019 | 9 | 40 | Insufficient PTH control, long RRT vintage | HDF Mid | 1.52 | 21.6 | B2M amyloidosis, osteopenia |
| Median | 63 | 10 F | 8 | 9 | 1.51 | 26.8 | ||||
| Min | 35 | 5 M | 3 | 1 | 1.08 | 14.5 | ||||
| Max | 83 | 11 | 40 | 1.90 | 61.3 | |||||
| Patient | Age | Main Indication for Etelcalcetide | PROS | CONS |
|---|---|---|---|---|
| 1 | 78 | Low tolerance, insufficient PTH control | Interest in ameliorating metabolic control in an elderly woman with relatively long vintage, high fracture risk and diffuse vascular calcifications. | Reversing bone damage is probably difficult, short life expectancy. No increase in alkaline phosphatase, possible low-turnover bone disease. |
| 2 | 82 | Pathologic fractures, insufficient PTH control | Interest in reducing risk of fractures in an elderly woman with relatively long vintage, previous fractures and diffuse vascular calcifications. | Reversing bone damage is probably difficult, short life expectancy. No increase in alkaline phosphatase, possible low-turnover bone disease. |
| 3 | 57 | Insufficient PTH control, Long RRT vintage | Interest in reducing all the long- term effects of dialysis in a young patient with diffuse vascular calcifications and low transplant chances (hyper immunized). | Reversing bone damage is probably difficult. No clear relationship with Beta2 amyloidosis. |
| 4 | 83 | Insufficient PTH control | Interest in ameliorating metabolic control in an elderly woman with relatively long vintage, high fracture risk and diffuse vascular calcifications. | Reversing bone damage is probably difficult, very short life expectancy. No increase in alkaline phosphatase, possible low-turnover bone disease. |
| 5 | 79 | Severe vascular calcifications, insufficient PTH control | Interest in ameliorating metabolic control in an elderly woman with relatively long vintage, severe and diffuse vascular calcifications. | Reversing bone damage is difficult, very short life expectancy. No increase in alkaline phosphatase, probably a low-turnover bone disease. No demonstration of efficacy in retarding vascular disease, if Ca-P balance is acceptable. |
| 6 | 52 | Need for high calcimimetic dose, bone fractures and deformities. Severe hypercalcemia | Interest in reducing risk of fractures in a young patient with a very long RRT vintage, previous fractures, deformities and diffuse vascular calcifications. | Reversing bone damage is probably difficult. No increase in alkaline phosphatase, probably a low-turnover bone disease. No demonstration of efficacy in retarding vascular disease, if Ca-P balance is acceptable. |
| 7 | 59 | Absorption problems, low tolerance, low compliance | Interest in ameliorating metabolic control in a relatively young woman at very high risk for vascular events (aphasic and with motor deficit after a cerebral accident). Reducing pill burden may improve compliance for other drugs. | No demonstration of efficacy in retarding vascular disease, if Ca-P balance is acceptable. Low compliance may offset the advantages of the drug, in particular in the presence of hyperphosphatemia. |
| 8 | 35 | Low compliance | Interest in ameliorating metabolic control in a young man with low compliance. Reducing pill burden may improve compliance for other drugs. | Low compliance may offset the advantages of the drug, in particular in the presence of hyperphosphatemia. |
| 9 | 37 | Irregular compliance | Interest in ameliorating metabolic control in a young woman with very high comorbidity. Reducing pill burden may improve compliance for other drugs. | No demonstration of efficacy in retarding vascular disease, if Ca-P balance is acceptable. Low compliance may offset treatment advantages. |
| 10 | 54 | Hypercalcemia, low tolerance | Interest in ameliorating metabolic control in a relatively young woman with hypercalcemia and unadapted PTH. | The cause of hypercalcemia is probably a granulomatous disease, less prone to being improved by treatment. |
| 11 | 70 | Hypercalcemia, amyloidosis | Interest in ameliorating metabolic control in woman with hypercalcemia and unadapted PTH and rheumatologic disease; high comorbidity, long dialysis vintage. | Reversing bone damage is difficult, short life expectancy. No increase in alkaline phosphatase, probably a low-turnover bone disease. No clear efficacy of PTH normalization on long-term dialysis-related comorbidity. |
| 12 | 43 | Low compliance, diffuse calcifications | Interest in ameliorating metabolic control in a young diabetic man with severe vascular calcifications. Low compliance. Reducing pill burden may improve compliance for other drugs. | No demonstration of efficacy in retarding vascular disease, if Ca-P balance is acceptable. Low compliance may offset the advantages of the drug, in the presence of hyperphosphatemia. |
| 13 | 63 | Insufficient PTH control, long RRT vintage | Interest in reducing all the long-term effects of dialysis in a young patient with diffuse vascular calcifications and low transplant chances (hyper immunized). | Reversing bone damage is difficult. No clear efficacy of PTH normalization on long-term dialysis-related comorbidity, in particular B2 microglobulin deposition. |
| 14 | 83 | Low compliance, diffuse calcifications | Interest in ameliorating metabolic control in a young diabetic man with severe vascular calcifications. Low compliance. Reducing burden pill may improve compliance for other drugs. | No demonstration of efficacy in retarding vascular disease. Low compliance may offset the advantages of the drug, in particular in the presence of hyperphosphatemia. |
| 15 | 67 | Insufficient PTH control, long RRT vintage | Interest in reducing all the long-term effects of dialysis in a relatively young patient, without transplant chances (neoplasia) and with dialysis related amyloidosis. | Reversing bone damage is probably difficult. No clear efficacy of PTH normalization on long-term dialysis related comorbidity, in particular B2 microglobulin deposition. |
| Patient | Age | CCI | Dialysis vintage RRT | Beneficience | Non-Maleficience | Justice | Autonomy |
|---|---|---|---|---|---|---|---|
| 1 | 78 | 6 | 8 | Better PTH control; possible, but unsure reduction of vascular and fracture risk | No contra-indication | Expensive treatment without clear benefits on the main targets (bone and vascular) | Patient agreement, appreciation of the lower oral drug load. |
| 2 | 82 | 8 | 9 | Better PTH control; possible, but unsure reduction mainly of fracture risk | No contra-indication | Expensive treatment without clear benefits on the main targets (bone and vascular) in a patient with short life expectancy | Patient agreement |
| 3 | 57 | 3 | 15 | Better PTH control; possible, but unsure reduction mainly of vascular risk | No contra-indication | Expensive treatment without clear benefits on the main targets (mainly vascular) | Patient agreement, appreciation of the lower oral drug load, appreciation of metabolic improvement |
| 4 | 83 | 10 | 12 | Better PTH control; possible, but unsure reduction mainly of fracture risk | No contra-indication | Expensive treatment without clear benefits on the main targets (bone and vascular) in a patient with short life expectancy | Patient agreement |
| 5 | 79 | 10 | 7 | Better PTH control; possible, but unsure reduction mainly of vascular risk | No contra-indication | Expensive treatment without clear benefits on the main targets (mainly vascular) in a patient with short life expectancy | Patient agreement |
| 6 | 52 | 6 | 11 | Better PTH control; possible, but unsure reduction of vascular risk and of progression of metabolic bone disease | No contra-indication | Expensive treatment without clear benefits on the main targets (bone and vascular) in a patient with advanced end-organ damage | Patient agreement, appreciation of metabolic improvement |
| 7 | 59 | 9 | 2 | Better PTH control; possible reduction of vascular risk. Lack of adherence may impair reaching synergic targets (phosphate) | No contra-indication | Expensive treatment without clear benefits on the main targets (mainly vascular) in a patient with short life expectancy and in which the lack of adherence may impair reaching synergic targets | Patient agreement |
| 8 | 35 | 4 | 6 | Better PTH control. Possible improvement of adherence reducing the pill burden. Lack of adherence may impair reaching synergic targets | No contra-indication | Expensive treatment in a patient in which the lack of adherence may impair reaching the synergic targets (phosphate) | Patient agreement |
| 9 | 37 | 14 | 7 | Better PTH control. Possible improvement of adherence reducing the pill burden. Lack of adherence may impair reaching synergic targets | No contra-indication, provided mild hypocalcemia is closely monitored | Expensive treatment without clear benefits in a patient with short life expectancy and in which the lack of adherence may impair reaching the other targets combined with PTH (calcium) | Patient agreement. Appreciation of the lower oral drug load, appreciation of metabolic improvement |
| 10 | 54 | 4 | 1 | Hypercalcemia, low tolerance. Unclear cause of hypercalcemia, unclear whether treatment will produce improvement | No contra-indication | Expensive treatment without clear benefits in the absence of clear diagnosis. | Patient agreement |
| 11 | 70 | 9 | 15 | Hypercalcemia, in a context of probable moderate hyper PTH not responsive to oral calcimimetics, not well tolerated at high doses. | No contra-indication | Expensive treatment without clear benefits in the absence of clear diagnosis. | Patient agreement |
| 12 | 43 | 6 | 1 | Better PTH control; possible reduction of vascular risk. Possible improvement of adherence reducing the pill burden. Lack of adherence may impair reaching synergic targets | No contra-indication | Expensive treatment without clear benefits on the main targets (mainly vascular) in a patient in which the lack of adherence may impair reaching the other targets combined with PTH (phosphate) | Patient agreement. Appreciation of the lower oral drug load, appreciation of metabolic improvement |
| 13 | 63 | 7 | 20 | Better PTH control; possible, but unsure reduction of vascular risk | No contra-indication | Expensive treatment without clear benefits on the main targets (mainly vascular) | Patient agreement, appreciation of the lower oral drug load, appreciation of metabolic improvement |
| 14 | 9 | 10 | Better PTH control; possible reduction of vascular risk. Possible improvement of adherence reducing the pill burden. Lack of adherence may impair reaching synergic targets | No contra-indication | Expensive treatment without clear benefits on the main targets (mainly vascular) in a patient in which the lack of adherence may impair reaching the other targets combined with PTH (phosphate) | Patient agreement. Appreciation of the lower oral drug load, appreciation of metabolic improvement | |
| 15 | 9 | 40 | Better PTH control; possible, but unsure reduction of vascular risk | No contra-indication | Expensive treatment without clear benefits on the main targets (mainly vascular) | Patient agreement, appreciation of the lower oral drug load, appreciation of metabolic improvement |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Piccoli, G.B.; Trabace, T.; Chatrenet, A.; Carranza de La Torre, C.A.; Gendrot, L.; Nielsen, L.; Fois, A.; Santagati, G.; Saulnier, P.; Panocchia, N. New Intravenous Calcimimetic Agents: New Options, New Problems. An Example on How Clinical, Economical and Ethical Considerations Affect Choice of Treatment. Int. J. Environ. Res. Public Health 2020, 17, 1238. https://doi.org/10.3390/ijerph17041238
Piccoli GB, Trabace T, Chatrenet A, Carranza de La Torre CA, Gendrot L, Nielsen L, Fois A, Santagati G, Saulnier P, Panocchia N. New Intravenous Calcimimetic Agents: New Options, New Problems. An Example on How Clinical, Economical and Ethical Considerations Affect Choice of Treatment. International Journal of Environmental Research and Public Health. 2020; 17(4):1238. https://doi.org/10.3390/ijerph17041238
Chicago/Turabian StylePiccoli, Giorgina Barbara, Tiziana Trabace, Antoine Chatrenet, Carlos Alberto Carranza de La Torre, Lurlinys Gendrot, Louise Nielsen, Antioco Fois, Giulia Santagati, Patrick Saulnier, and Nicola Panocchia. 2020. "New Intravenous Calcimimetic Agents: New Options, New Problems. An Example on How Clinical, Economical and Ethical Considerations Affect Choice of Treatment" International Journal of Environmental Research and Public Health 17, no. 4: 1238. https://doi.org/10.3390/ijerph17041238
APA StylePiccoli, G. B., Trabace, T., Chatrenet, A., Carranza de La Torre, C. A., Gendrot, L., Nielsen, L., Fois, A., Santagati, G., Saulnier, P., & Panocchia, N. (2020). New Intravenous Calcimimetic Agents: New Options, New Problems. An Example on How Clinical, Economical and Ethical Considerations Affect Choice of Treatment. International Journal of Environmental Research and Public Health, 17(4), 1238. https://doi.org/10.3390/ijerph17041238






