Transition Metal Containing Particulate Matter Promotes Th1 and Th17 Inflammatory Response by Monocyte Activation in Organic and Inorganic Compounds Dependent Manner
Abstract
1. Introduction
2. Materials and Methods
2.1. TMCPM Preparation
2.2. Cell Isolation
2.3. Cell Culture and Immunostaining for Intracellular Proteins
2.4. Cytokine Detection by CBA
2.5. Endotoxin Determination
2.6. Statistics
3. Results
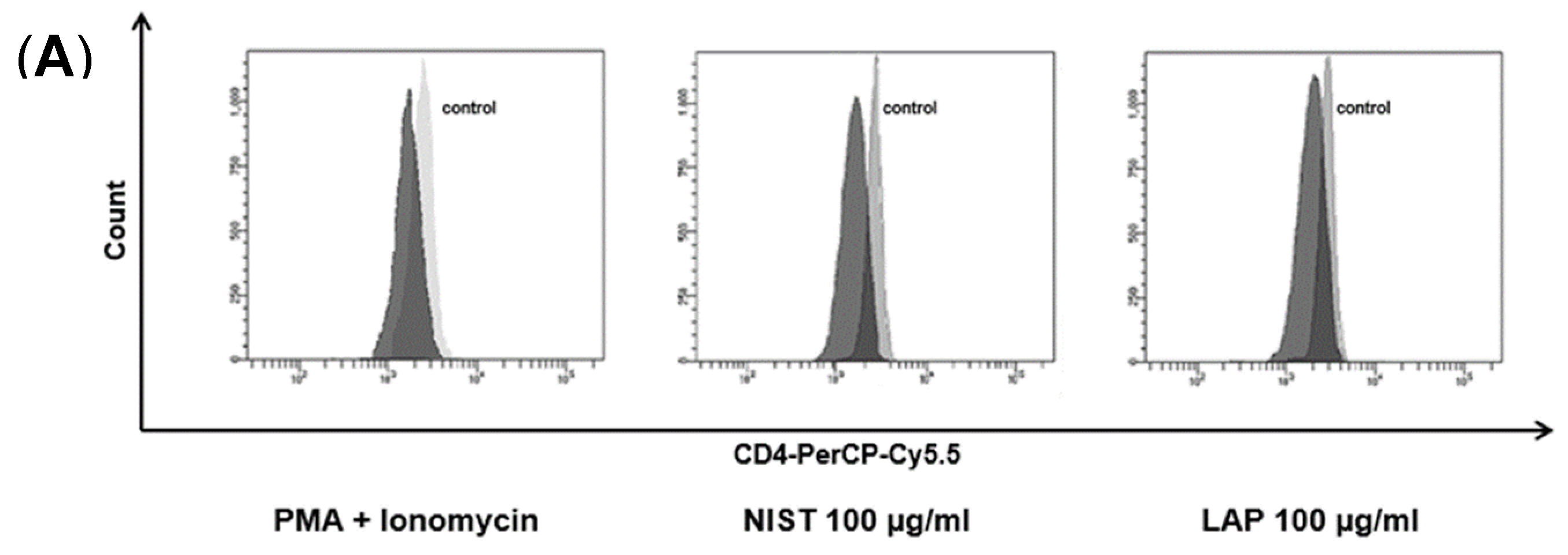
3.1. TMCPM Reduces the Expression Level of CD4 on the Surface of T Cells
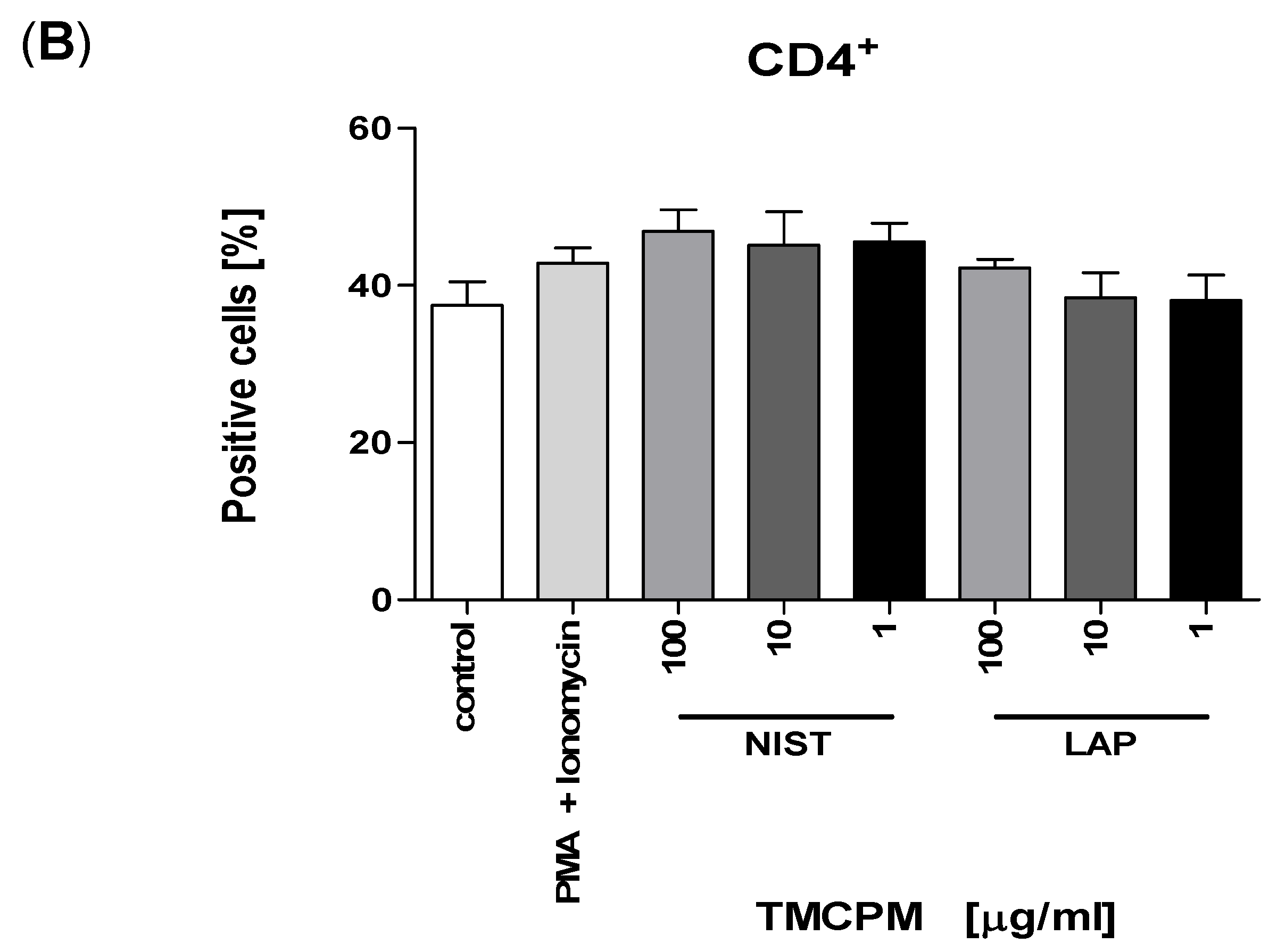
3.2. TMCPM Activates CD4+ T Lymphocytes Depending on the Organic Compounds Content
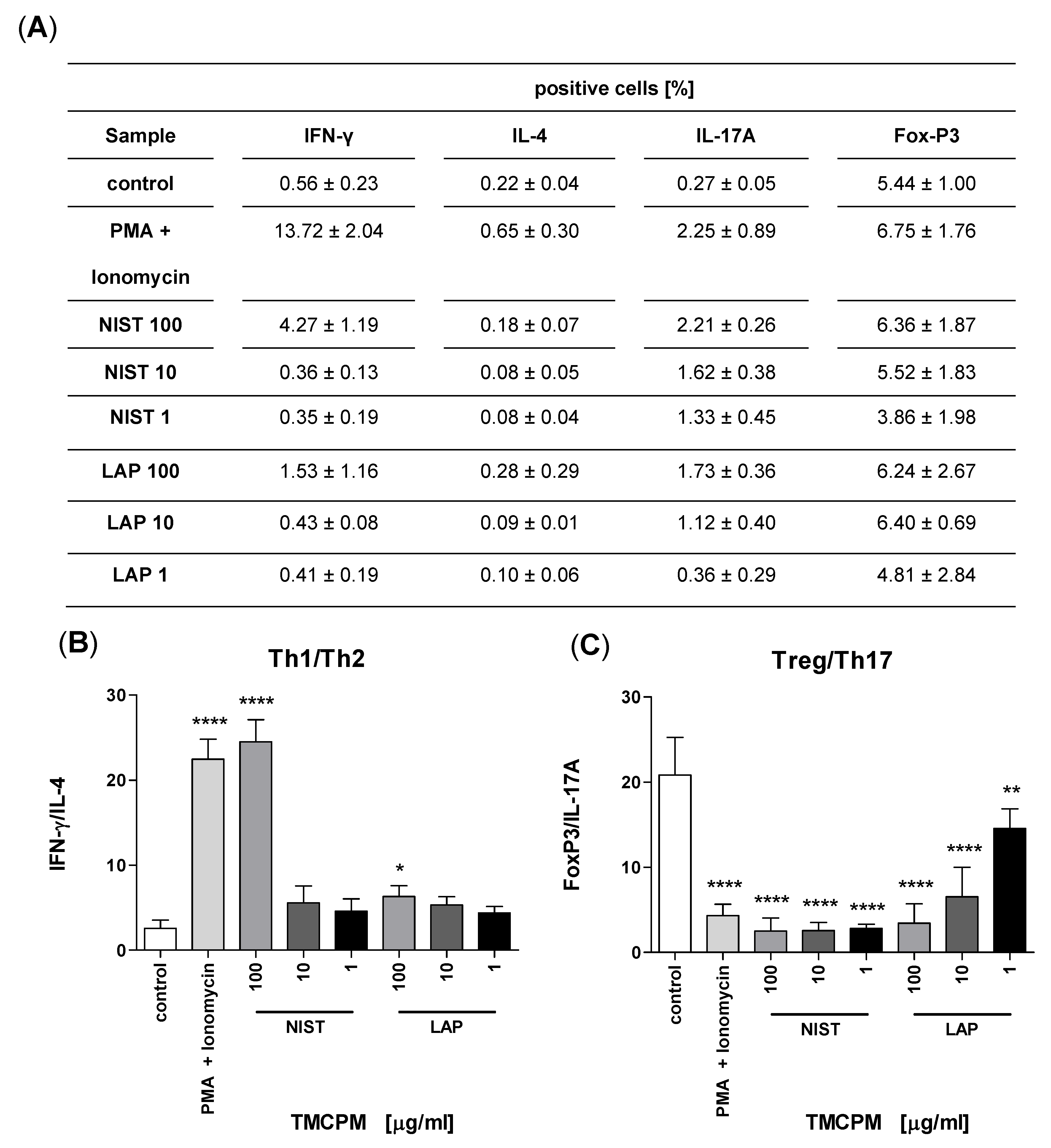
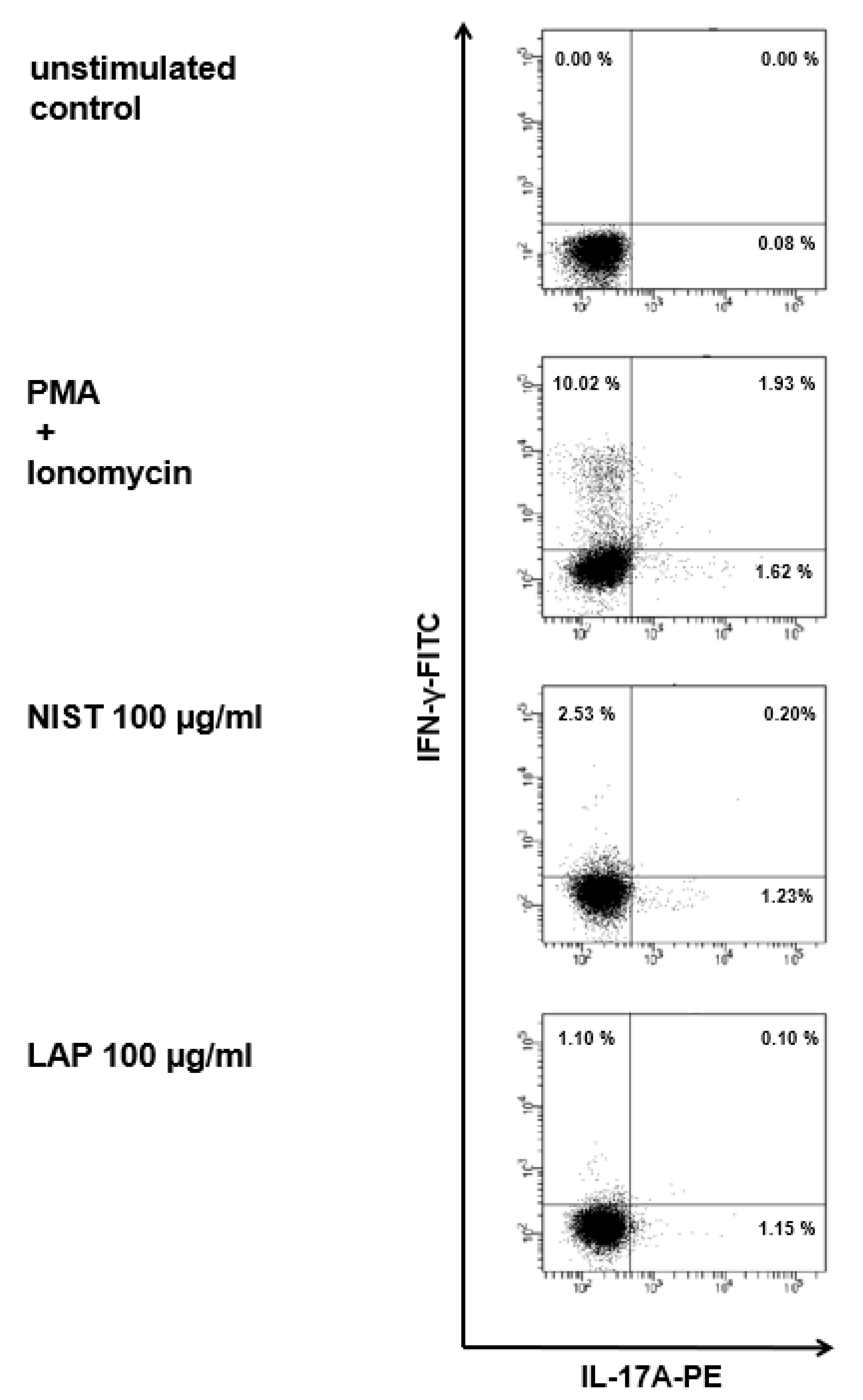
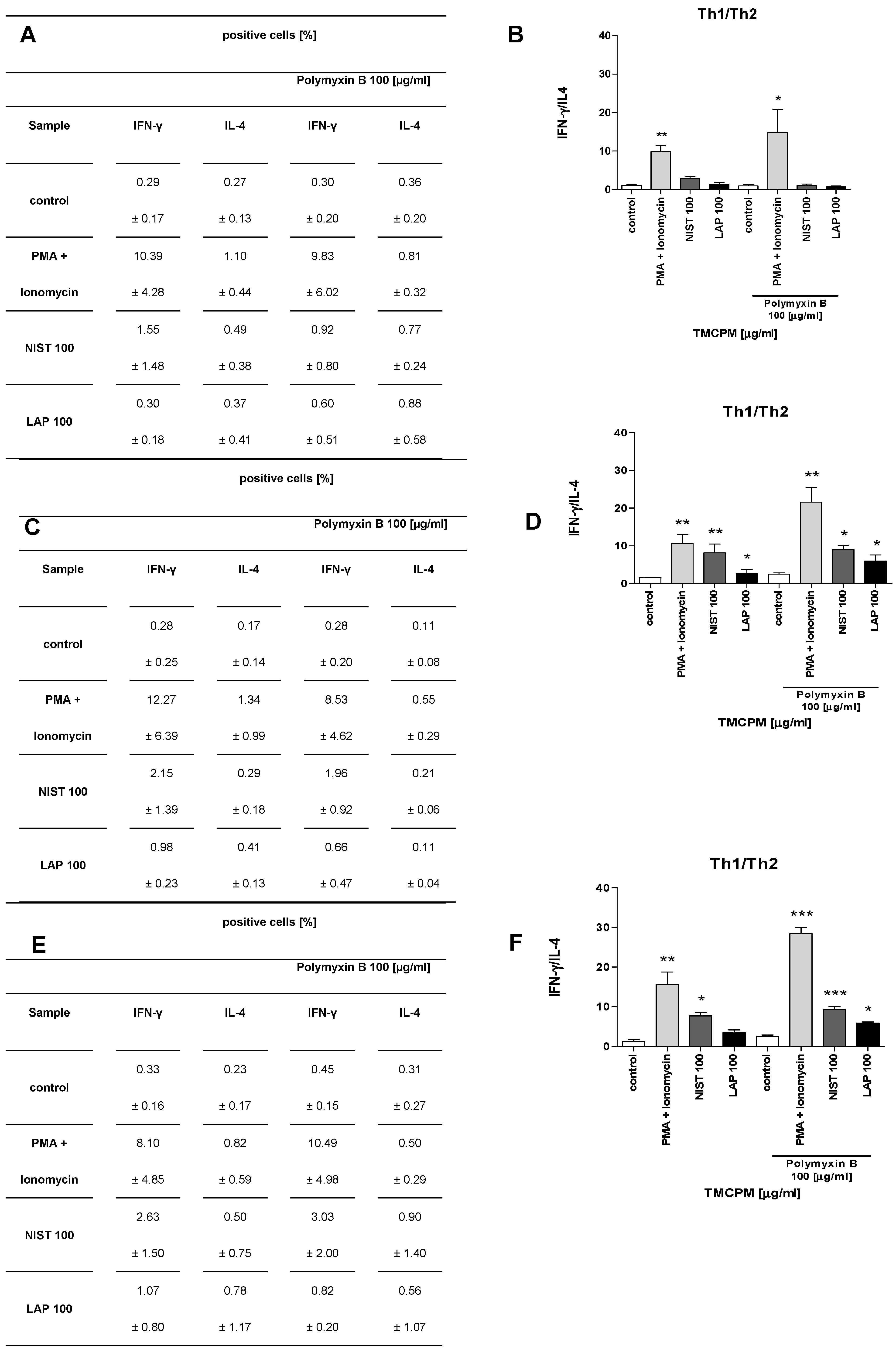
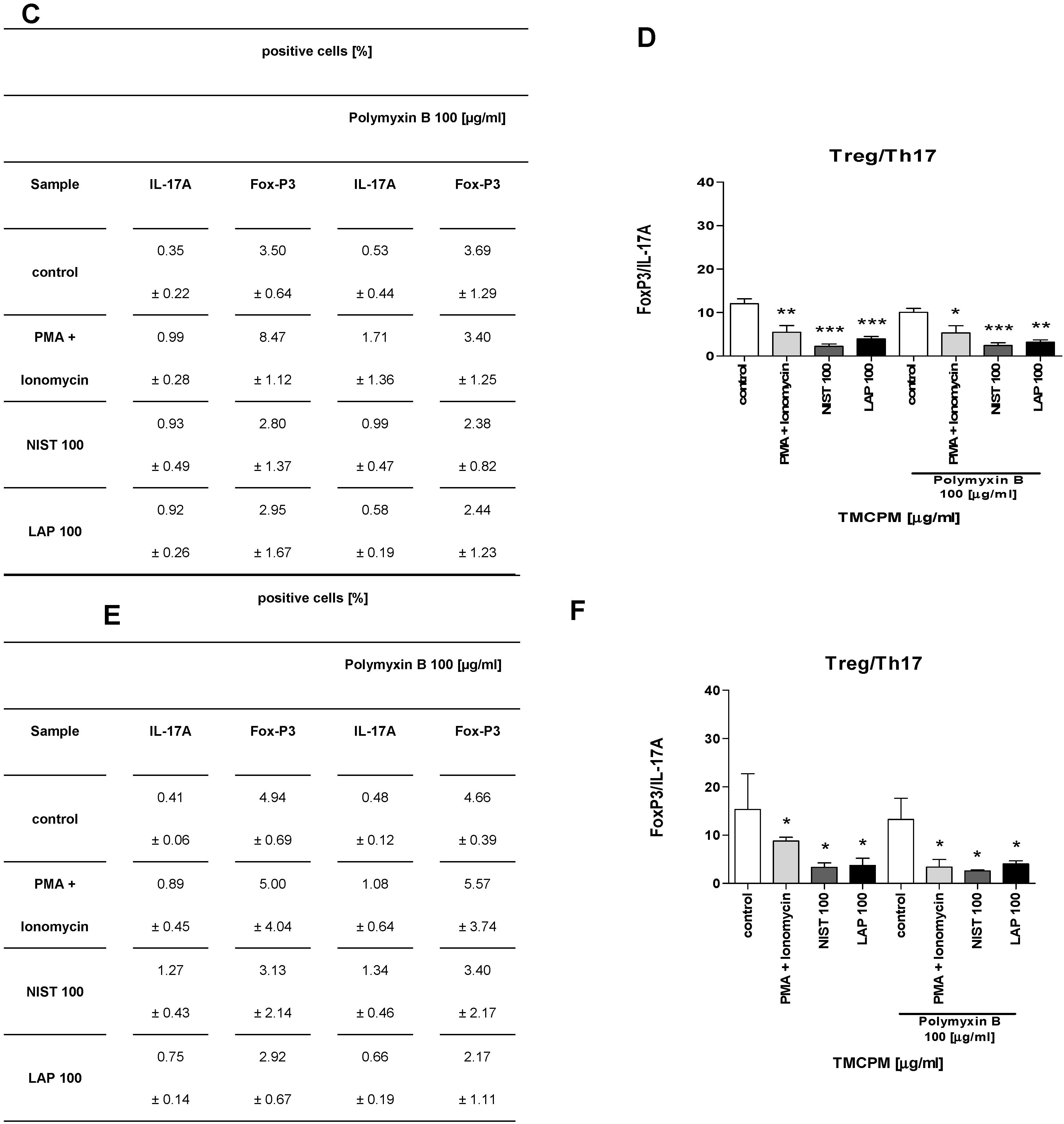
3.3. TMCPM Exposure Is Skewing the Balance of Th1/Th2 and Treg/Th17 Subsets
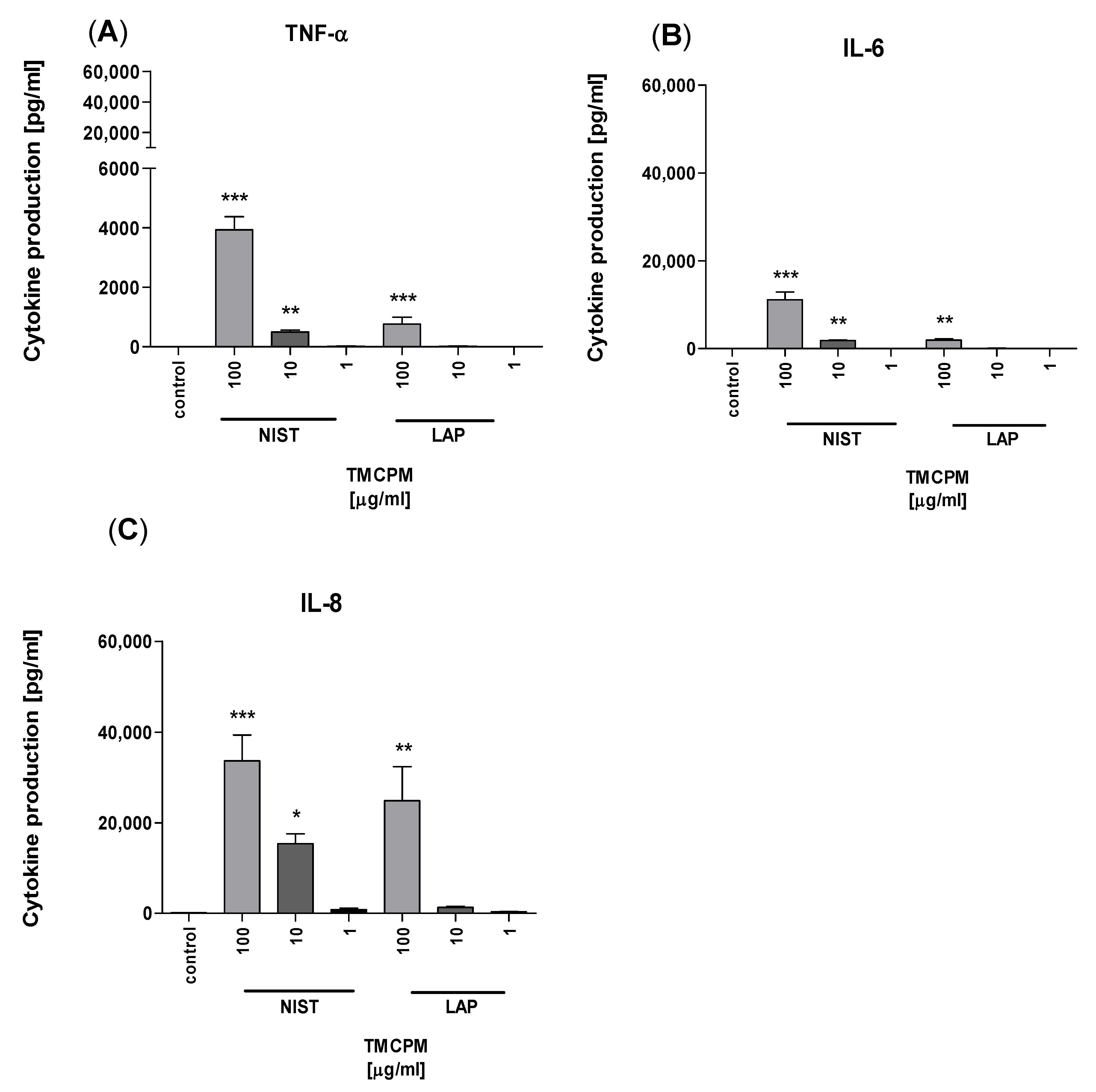
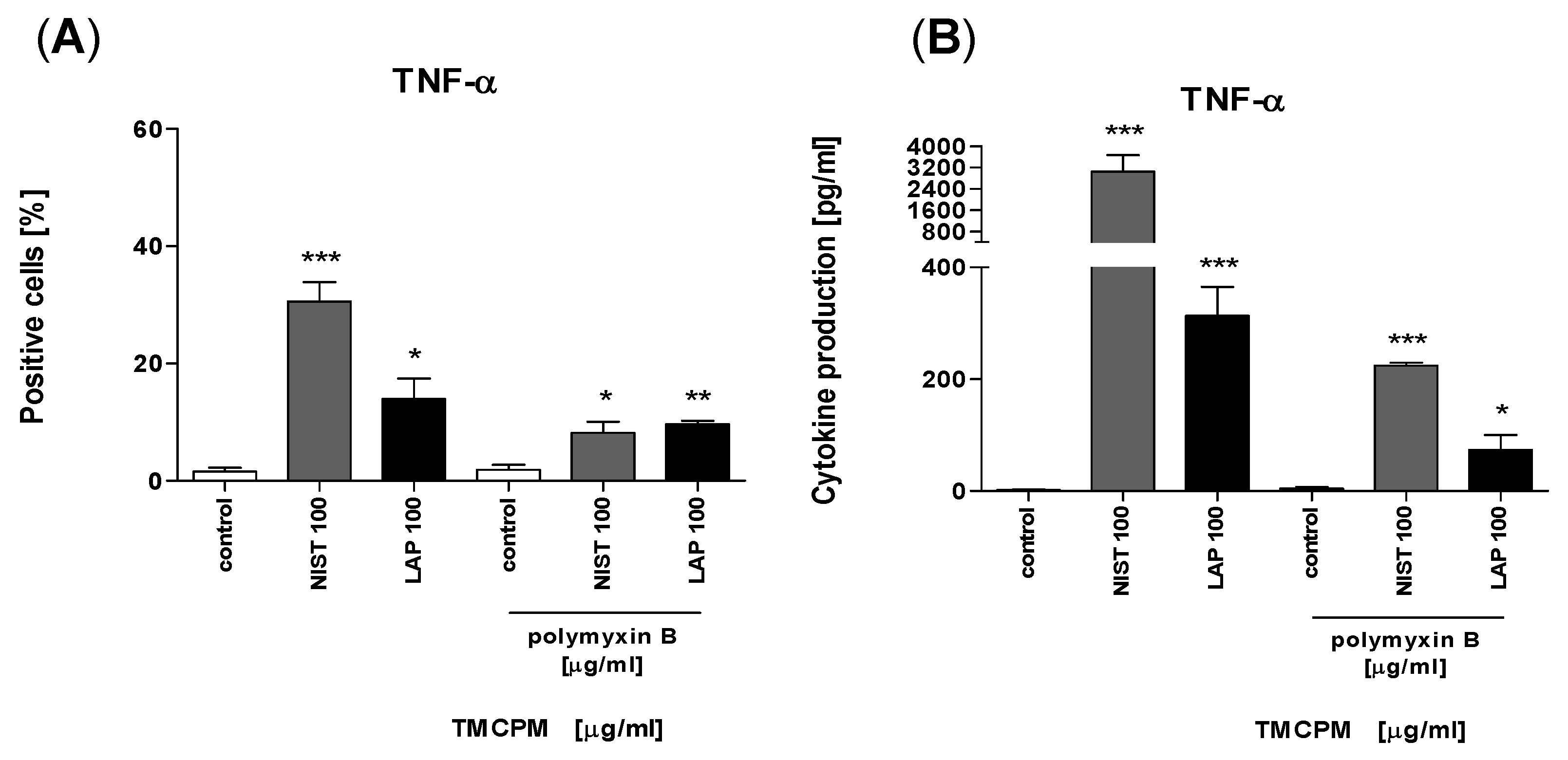
3.4. Monocytes Activation Is Required for TMCPM Induced Polarization of Th1 and Th17 Immune Response
3.5. Endotoxin Content Is Partially Responsible for Pro-Inflammatory Potency of TMCPM
3.6. Both Organic and Inorganic Components of TMCPM Might Be Responsible for Activation of Monocytes and Propagation of Inflammatory Response
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Liu, S.K.; Cai, S.; Chen, Y. The effect of pollutional haze on pulmonary function. J. Thorac. Dis. 2016, 8, E41–E56. [Google Scholar]
- Valavanidis, A.; Fiotakis, K.; Vlachogianni, T. Airborne particulate matter and human health: Toxicological assessment and importance of size and composition of particles for oxidative damage and carcinogenic mechanisms. J. Environ. Sci. Health C Environ. Carcinog. Ecotoxicol. Rev. Part C 2008, 26, 339–362. [Google Scholar] [CrossRef]
- Ritz, S.A. Air pollution as a potential contributor to the “epidemic” of autoimmune disease. Med. Hypotheses 2010, 74, 110–117. [Google Scholar] [CrossRef]
- Bateman, E.D.; Hurd, S.S.; Barnes, P.J.; Bousquet, J.; Drazen, J.M.; FitzGerald, J.M.; Gibson, P.; Ohta, K.; O’Byrne, P.; Pedersen, S.E.; et al. Global strategy for asthma management and prevention: GINA executive summary. Eur. Respir. J. 2008, 31, 143–178. [Google Scholar] [CrossRef]
- Gawda, A.; Majka, G.; Nowak, B.; Marcinkiewicz, J. Air pollution, oxidative stress, and exacerbation of autoimmune diseases. Cent. Eur. J. Immunol. 2017, 42, 305–312. [Google Scholar] [CrossRef]
- Ji, H.; Hershey, G.K.K. Genetic and epigenetic influence on the response to environmental particulate matter. J. Allergy Clin. Immunol. 2013, 129, 33–41. [Google Scholar] [CrossRef]
- Buzea, C.; Pacheco, I.I.; Robbie, K. Nanomaterials and nanoparticles: Sources and toxicity. Biointerphases 2007, 2, MR17–MR71. [Google Scholar] [CrossRef]
- Gan, W.Q.; FitzGerald, J.M.; Carlsten, C.; Sadatsafavi, M.; Brauer, M. Associations of ambient air pollution with chronic obstructive pulmonary disease hospitalization and mortality. Am. J. Respir. Crit. Care Med. 2013, 187, 721–727. [Google Scholar] [CrossRef]
- Brook, R.D.; Rajagopalan, S.; Pope, C.A.; Brook, J.R.; Bhatnagar, A.; Diez-Roux, A.V.; Holguin, F.; Hong, Y.; Luepker, R.V.; Mittleman, M.A.; et al. American Heart Association Council on Epidemiology and Prevention, Council on the Kidney in Cardiovascular Disease, and Council on Nutrition, Physical Activity and Metabolism. Particulate matter air pollution and cardiovascular disease: An update to the scientific statement from the American Heart Association. Circulation 2010, 121, 2331–2378. [Google Scholar]
- Ling, S.H.; van Eeden, S.F. Particulate matter air pollution exposure: Role in the development and exacerbation of chronic obstructive pulmonary disease. Int. J. Chron. Obstr. Pulm. Dis. 2009, 4, 233–243. [Google Scholar] [CrossRef]
- Pope, C.A.; Burnett, R.T.; Thun, M.J.; Calle, E.E.; Krewski, D.; Ito, K.; Thurstonet, G.D. Lung cancer, cardiopulmonary mortality, and long-term exposure to fine particulate air pollution. JAMA 2002, 287, 1132–1141. [Google Scholar] [CrossRef]
- Meng, Q.; Richmond-Bryant, J.; Lu, S.E.; Buckley, B.; Welsh, W.J.; Whitsel, E.A.; Hanna, A.; Yeatts, K.B.; Warren, J.; Herring, A.H.; et al. Cardiovascular outcomes and the physical and chemical properties of metal ions found in particulate matter air pollution: A QICAR study. Environ. Health Perspect. 2013, 121, 558–564. [Google Scholar] [CrossRef]
- Wright, R.O.; Baccarelli, A. Metals and neurotoxicology. J. Nutr. 2007, 137, 2809–2813. [Google Scholar] [CrossRef]
- Shi, T.; Schins, R.P.; Knaapen, A.M.; Kuhlbusch, T.; Pitz, M.; Heinrich, J.; Borm, P.J. Hydroxyl radical generation by electron paramagnetic resonance as a new method to monitor ambient particulate matter composition. J. Environ. Monit. 2003, 5, 550–556. [Google Scholar] [CrossRef]
- Sawyer, K.; Mundandhara, S.; Ghio, A.J.; Madden, M.C. The effects of ambient particulate matter on human alveolar macrophage oxidative and inflammatory responses. J. Toxicol. Environ. Health A 2009, 73, 41–57. [Google Scholar] [CrossRef]
- Nemmar, A.; Hoet, P.H.M.; Vanquickenborne, B.; Dinsdale, D.; Thomeer, M.; Hoylaerts, M.F.; Vanbilloen, H.; Mortelmans, L.; Nemery, B. Passage of inhaled particles into the blood circulation in humans. Circulation 2002, 105, 411–414. [Google Scholar] [CrossRef]
- Zembala, M.; Asherson, G.L. (Eds.) Human Monocytes, 1st ed.; Academic Press: London, UK, 1989. [Google Scholar]
- Lee, F.E.; Georas, S.N.; Beck, L.A. IL-17: Important for host defense, autoimmunity, and allergy? J. Investig. Dermatol. 2010, 130, 2540–2542. [Google Scholar]
- Hou, T.; Liao, J.; Zhang, C.; Sun, C.; Li, X.; Wang, G. Elevated expression of miR-146, miR-139 and miR-340 involved in regulating Th1/Th2 balance with acute exposure of fine particulate matter in mice. Int. Immunopharmacol. 2018, 54, 68–77. [Google Scholar] [CrossRef]
- Li, Y.; Dong, T.; Jiang, X.; Wang, C.; Zhang, Y.; Li, Y.; Zheng, G.; Li, X.; Bai, J.; Li, H. Chronic and low-level particulate matter exposure can sustainably mediate lung damage and alter CD4 T cells during acute lung injury. Mol. Immunol. 2019, 112, 51–58. [Google Scholar] [CrossRef]
- Samek, L.; Furman, L.; Mikrut, M.; Regiel-Futyra, A.; Macyk, W.; Stochel, G.; van Eldik, R. Chemical composition of submicron and fine particulate matter collected in Krakow, Poland. Consequences for the APARIC project. Chemosphere 2017, 187, 430–439. [Google Scholar] [CrossRef]
- Gawda, A.; Majka, G.; Nowak, B.; Śróttek, M.; Walczewska, M.; Marcinkiewicz, J. Air particulate matter SRM 1648a primes macrophages to hyperinflammatory response after LPS stimulation. Inflamm. Res. 2018, 67, 765–776. [Google Scholar] [CrossRef] [PubMed]
- Mikrut, M.; Regiel-Futyra, A.; Samek, L.; Macyk, W.; Stochel, G.; van Eldik, R. Generation of hydroxyl radicals and singlet oxygen by particulate matter and its inorganic components. Environ. Pollut. 2018, 238, 638–646. [Google Scholar] [CrossRef] [PubMed]
- Baran, J.; Guzik, K.; Hryniewicz, W.; Ernst, M.; Flad, H.D.; Pryjma, J. Apoptosis of monocytes and prolonged survival of granulocytes as a result of phagocytosis of bacteria. Infect. Immun. 1996, 64, 4242–4248. [Google Scholar] [CrossRef] [PubMed]
- Baran, J.; Kowalczyk, D.; Ożóg, M.; Zembala, M. Three-color flow cytometry detection of intracellular cytokines in peripheral blood mononuclear cells: Comparative analysis of phorbol myristate acetate-ionomycin and phytohemagglutinin stimulation. Clin. Diagn. Lab. Immunol. 2001, 8, 303–313. [Google Scholar] [CrossRef] [PubMed]
- Petersen, C.M.; Christensen, E.I.; Andresen, B.S.; Møller, B.K. Internalization, lysosomal degradation and new synthesis of surface membrane CD4 in phorbol ester-activated T-lymphocytes and U-937 cells. Exp. Cell Res. 1992, 201, 160–173. [Google Scholar] [CrossRef]
- Møller, B.K.; Andresen, B.S.; Christensen, E.I.; Petersen, C.M. Surface membrane CD4 turnover in phorbol ester stimulated T-lymphocytes. Evidence of degradation and increased synthesis. FEBS Lett. 1990, 276, 59–62. [Google Scholar] [CrossRef]
- Crane, I.J.; Forrester, J.V. Th1 and Th2 lymphocytes in autoimmune disease. Crit. Rev. Immunol. 2005, 25, 75–102. [Google Scholar] [CrossRef]
- Charlton, B.; Lafferty, K.J. The Th1/Th2 balance in autoimmunity. Curr. Opin. Immunol. 1995, 7, 793–798. [Google Scholar] [CrossRef]
- Druet, P.; Sheela, R.; Pelletier, L. Th1 and Th2 cells in autoimmunity. Clin. Exp. Immunol. 1995, 101, 9–12. [Google Scholar] [CrossRef]
- Pryjma, J.; Baran, J.; Ernst, M.; Woloszyn, M.; Flad, H.D. Altered antigen-presenting capacity of human monocytes after phagocytosis of bacteria. Infect. Immun. 1994, 62, 1961–1967. [Google Scholar] [CrossRef]
- Ma, Q.Y.; Huang, D.Y.; Zhang, H.J.; Wang, S.; Chen, X.F. Exposure to particulate matter 2.5 (PM2.5) induced macrophage-dependent inflammation, characterized by increased Th1/Th17 cytokine secretion and cytotoxicity. Int. Immunopharmacol. 2017, 50, 139–145. [Google Scholar] [CrossRef]
- Li, G.; Cao, Y.; Sun, Y.; Xu, R.; Zheng, Z.; Song, H. Ultrafine particles in the airway aggravated experimental lung injury through impairment in Treg function. Biochem. Biophys. Res. Commun. 2016, 478, 494–500. [Google Scholar] [CrossRef]
- Zhang, F.; Li, M.Y.; Lan, Y.T.; Wanga, C.B. Imbalance of Th17/Tregs in rats with smoke inhalation-induced acute lung injury. Sci. Rep. 2016, 6, 21348. [Google Scholar] [CrossRef]
- Duan, M.C.; Zhang, J.Q.; Liang, Y.; Liu, G.N.; Xiao, J.; Tang, H.J.; Liang, Y. Infiltration of IL-17-Producing T Cells and Treg Cells in a Mouse Model of Smoke-Induced Emphysema. Inflammation 2016, 39, 1334–1344. [Google Scholar] [CrossRef]
- Castañeda, A.R.; Pinkerton, K.E.; Bein, K.J.; Magaña-Méndez, A.; Yang, H.T.; Ashwood, P.; Vogel, C.F.A. Ambient particulate matter activates the aryl hydrocarbon receptor in dendritic cells and enhances Th17 polarization. Toxicol. Lett. 2018, 292, 85–96. [Google Scholar] [CrossRef]
- Becker, S.; Soukup, J.M.; Gilmour, M.I.; Devlin, R.B. Stimulation of human and rat alveolar macrophages by urban air particles: Effects on oxidant radical generation and cytokine production. Toxicol. Appl. Pharmacol. 1996, 141, 637–648. [Google Scholar] [CrossRef]
- Becker, S.; Soukup, J.M.; Sioutas, C.; Cassee, F.R. Response of human alveolar macrophages to ultrafine, fine, and coarse urban air pollution particles. Exp. Lung Res. 2003, 29, 29–44. [Google Scholar] [CrossRef]
- Dong, W.; Lewtas, J.; Luster, M. Role of endotoxin in tumor necrosis factor α expression from alveolar macrophages treated with urban air particles. Exp. Lung Res. 1996, 22, 577–592. [Google Scholar] [CrossRef]
- Long, C.M.; Suh, H.H.; Kobzik, L.; Catalano, P.J.; Ning, Y.Y.; Koutrakis, P. A pilot investigation of the relative toxicity of indoor and outdoor fine particles: In vitro effects of endotoxin and other particulate properties. Environ. Health Perspect. 2001, 109, 1019–1026. [Google Scholar] [CrossRef]
- Schins, R.P.; Lightbody, J.H.; Borm, P.J.; Shi, T.; Donaldson, K.; Stone, V. Inflammatory effects of coarse and fine particulate matter in relation to chemical and biological constituents. Toxicol. Appl. Pharmacol. 2004, 195, 1–11. [Google Scholar] [CrossRef]
- Monn, C.; Becker, S. Cytotoxicity and induction of proinflammatory cytokines from human monocytes exposed to fine (PM2.5) and coarse particles (PM10—2.5) in outdoor and indoor air. Toxicol. Appl. Pharmacol. 1999, 155, 245–252. [Google Scholar] [CrossRef]
- Soukup, J.M.; Becker, S. Human alveolar macrophage response to air pollution particles are associated with insoluble components of coarse material, including particulate endotoxin. Toxicol. Appl. Pharmacol. 2001, 171, 20–26. [Google Scholar] [CrossRef]
- Ghio, A.J.; Carraway, M.S.; Madden, M.C. Composition of air pollution particles and oxidative stress in cells, tissues, and living systems. J. Toxicol. Environ. Health B Crit. Rev. 2012, 15, 1–21. [Google Scholar] [CrossRef]




| Sample | LPS [EU/ml] | |||
|---|---|---|---|---|
| Polymyxin B 100 [µg/mL] | ||||
| E. coli | [ng/mL] | LPS 0.5 | 5.50 | 0.00 |
| TMCPM | [µg/mL] | NIST 100 | 1.75 | 0.10 |
| LAP 100 | 0.40 | 0.00 | ||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gałuszka, A.; Stec, M.; Węglarczyk, K.; Kluczewska, A.; Siedlar, M.; Baran, J. Transition Metal Containing Particulate Matter Promotes Th1 and Th17 Inflammatory Response by Monocyte Activation in Organic and Inorganic Compounds Dependent Manner. Int. J. Environ. Res. Public Health 2020, 17, 1227. https://doi.org/10.3390/ijerph17041227
Gałuszka A, Stec M, Węglarczyk K, Kluczewska A, Siedlar M, Baran J. Transition Metal Containing Particulate Matter Promotes Th1 and Th17 Inflammatory Response by Monocyte Activation in Organic and Inorganic Compounds Dependent Manner. International Journal of Environmental Research and Public Health. 2020; 17(4):1227. https://doi.org/10.3390/ijerph17041227
Chicago/Turabian StyleGałuszka, Adrianna, Małgorzata Stec, Kazimierz Węglarczyk, Anna Kluczewska, Maciej Siedlar, and Jarek Baran. 2020. "Transition Metal Containing Particulate Matter Promotes Th1 and Th17 Inflammatory Response by Monocyte Activation in Organic and Inorganic Compounds Dependent Manner" International Journal of Environmental Research and Public Health 17, no. 4: 1227. https://doi.org/10.3390/ijerph17041227
APA StyleGałuszka, A., Stec, M., Węglarczyk, K., Kluczewska, A., Siedlar, M., & Baran, J. (2020). Transition Metal Containing Particulate Matter Promotes Th1 and Th17 Inflammatory Response by Monocyte Activation in Organic and Inorganic Compounds Dependent Manner. International Journal of Environmental Research and Public Health, 17(4), 1227. https://doi.org/10.3390/ijerph17041227






