Epigenetic Biomarkers for Environmental Exposures and Personalized Breast Cancer Prevention
Abstract
1. Introduction
1.1. Towards a Better Understanding of how Exposures Affect Personal Breast Cancer Risk
1.2. DNA Methylation as Molecular Markers of Environmental Exposures
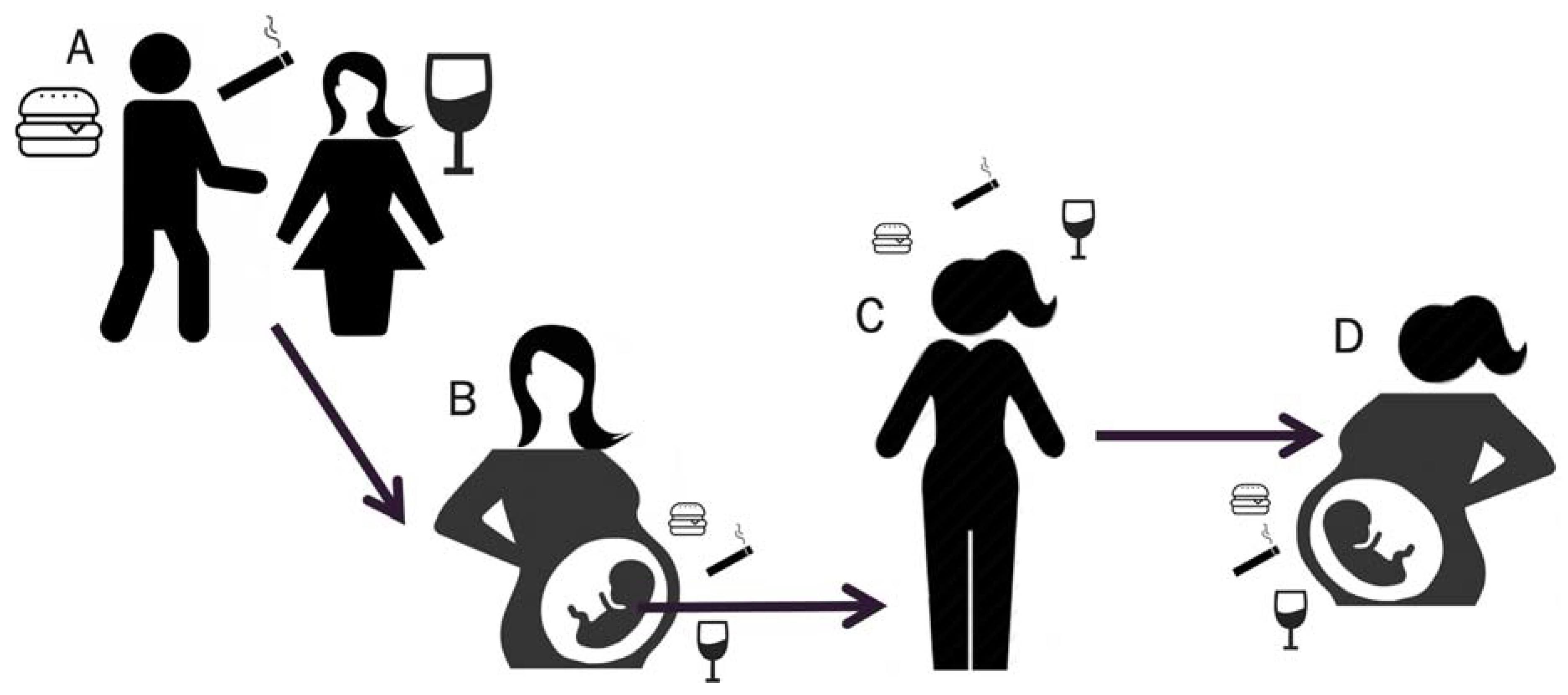
1.3. Towards a Better Understanding of how Exposures Affect Breast Cancer Risk in Offspring and Future Generations
1.4. Towards Personalized Prevention of Breast Cancer
2. Proposed Approaches and Expected Results
Funding
Acknowledgments
Conflicts of Interest
References
- Lichtenstein, P.; Holm, N.V.; Verkasalo, P.K.; Iliadou, A.; Kaprio, J.; Koskenvuo, M.; Pukkala, E.; Skytthe, A.; Hemminki, K. Environmental and heritable factors in the causation of cancer: Analyses of cohorts of twins from Sweden, Denmark, and Finland. N. Engl. J. Med. 2000, 343, 78–85. [Google Scholar] [CrossRef] [PubMed]
- Foulkes, W.D. Inherited susceptibility to common cancers. N. Engl. J. Med. 2008, 359, 2143. [Google Scholar] [CrossRef] [PubMed]
- Nelson, H.D.; Zakher, B.; Cantor, A.; Fu, R.; Griffin, J.; O’Meara, E.S.; Buist, D.S.M.; Kerlikowske, K.; van Ravesteyn, N.T.; Trentham-Dietz, A.; et al. Risk Factors for Breast Cancer for Women Age 40 to 49: A Systematic Review and Meta-analysis. Ann. Intern. Med. 2012, 156, 635. [Google Scholar] [CrossRef] [PubMed]
- Singletary, S.E. Rating the risk factors for breast cancer. Ann. Surg. 2003, 237, 474–482. [Google Scholar] [CrossRef] [PubMed]
- Clemons, M.; Loijens, L.; Goss, P. Breast cancer risk following irradiation for Hodgkin’s disease. Cancer Treat. Rev. 2000, 26, 291–302. [Google Scholar] [CrossRef] [PubMed]
- Hoover, R.N.; Hyer, M.; Pfeiffer, R.M.; Adam, E.; Bond, B.; Cheville, A.L.; Colton, T.; Hartge, P.; Hatch, E.E.; Herbst, A.L.; et al. Adverse Health Outcomes in Women Exposed In Utero to Diethylstilbestrol. N. Engl. J. Med. 2011, 365, 1304–1314. [Google Scholar] [CrossRef]
- Palmer, J.R.; Wise, L.A.; Hatch, E.E.; Troisi, R.; Titus-Ernstoff, L.; Strohsnitter, W.; Kaufman, R.; Herbst, A.L.; Noller, K.L.; Hyer, M.; et al. Prenatal diethylstilbestrol exposure and risk of breast cancer. Cancer Epidemiol. Biomarkers Prev. 2006, 15, 1509–1514. [Google Scholar] [CrossRef]
- Cohn, B.A.; La Merrill, M.; Krigbaum, N.Y.; Yeh, G.; Park, J.S.; Zimmermann, L.; Cirillo, P.M. DDT exposure in utero and breast cancer. J. Clin. Endocrinol. Metab. 2015, 100, 2865–2872. [Google Scholar] [CrossRef]
- Kehm, R.D.; Genkinger, J.M.; MacInnis, R.J.; John, E.M.; Phillips, K.-A.; Dite, G.S.; Milne, R.L.; Zeinomar, N.; Liao, Y.; Knight, J.A.; et al. Recreational physical activity is associated with reduced breast cancer risk in adult women at high risk for breast cancer: A cohort study of women selected for familial and genetic risk. Cancer Res. 2019, 80, 116–125. [Google Scholar] [CrossRef]
- Chlebowski, R.T.; Luo, J.; Anderson, G.L.; Barrington, W.; Reding, K.; Simon, M.S.; Manson, J.E.; Rohan, T.E.; Wactawski-Wende, J.; Lane, D.; et al. Weight loss and breast cancer incidence in postmenopausal women. Cancer 2019, 125, 205–212. [Google Scholar] [CrossRef]
- Terry, M.B.; Liao, Y.; Whittemore, A.S.; Leoce, N.; Buchsbaum, R.; Zeinomar, N.; Dite, G.S.; Chung, W.K.; Knight, J.A.; Southey, M.C.; et al. 10-year performance of four models of breast cancer risk: A validation study. Lancet Oncol. 2019, 20, 504–517. [Google Scholar] [CrossRef]
- Al-Ajmi, K.; Lophatananon, A.; Yuille, M.; Ollier, W.; Muir, K.R. Review of non-clinical risk models to aid prevention of breast cancer. Cancer Causes Control 2018, 29, 967–986. [Google Scholar] [CrossRef] [PubMed]
- Rosenstock, I.M.; Strecher, V.J.; Becker, M.H. Social learning theory and the Health Belief Model. Health Educ. Q. 1988, 15, 175–183. [Google Scholar] [CrossRef] [PubMed]
- Brody, J.G.; Rudel, R.A.; Michels, K.B.; Moysich, K.B.; Bernstein, L.; Attfield, K.R.; Gray, S. Environmental pollutants, diet, physical activity, body size, and breast cancer. Cancer 2007, 109, 2627–2634. [Google Scholar] [CrossRef] [PubMed]
- Rodgers, K.M.; Udesky, J.O.; Rudel, R.A.; Brody, J.G. Environmental chemicals and breast cancer: An updated review of epidemiological literature informed by biological mechanisms. Environ. Res. 2018, 160, 152–182. [Google Scholar] [CrossRef]
- SRNT Subcommittee on Biochemical Verification. Biochemical verification of tobacco use and cessation. Nicotine Tob. Res. 2002, 4, 149–159. [Google Scholar] [CrossRef]
- Dietz, P.M.; Homa, D.; England, L.J.; Burley, K.; Tong, V.T.; Dube, S.R.; Bernert, J.T. Estimates of nondisclosure of cigarette smoking among pregnant and nonpregnant women of reproductive age in the United States. Am. J. Epidemiol. 2011, 173, 355–359. [Google Scholar] [CrossRef]
- Shipton, D.; Tappin, D.M.; Vadiveloo, T.; Crossley, J.A.; Aitken, D.A.; Chalmers, J. Reliability of self reported smoking status by pregnant women for estimating smoking prevalence: A retrospective, cross sectional study. BMJ 2009, 339, 1241. [Google Scholar] [CrossRef]
- MacOn, M.B.; Fenton, S.E. Endocrine disruptors and the breast: Early life effects and later life disease. J. Mammary Gland Biol. Neoplasia 2013, 18, 43–61. [Google Scholar] [CrossRef]
- Gray, J.M.; Rasanayagam, S.; Engel, C.; Rizzo, J. State of the evidence 2017: An update on the connection between breast cancer and the environment. Environ. Health 2017, 16, 94. [Google Scholar] [CrossRef]
- Angerer, J.; Ewers, U.; Wilhelm, M. Human biomonitoring: State of the art. Int. J. Hyg. Environ. Health 2007, 210, 201–228. [Google Scholar] [CrossRef] [PubMed]
- Holland, N.T.; Smith, M.T.; Eskenazi, B.; Bastaki, M. Biological sample collection and processing for molecular epidemiological studies. Mutat. Res. Rev. Mutat. Res. 2003, 543, 217–234. [Google Scholar] [CrossRef]
- Rexrode, K.M.; Lee, I.M.; Cook, N.R.; Hennekens, C.H.; Buring, J.E. Baseline characteristics of participants in the women’s health study. J. Women’s Heal. Gender Based Med. 2000, 9, 19–27. [Google Scholar] [CrossRef] [PubMed]
- Riboli, E.; Hunt, K.J.; Slimani, N.; Ferrari, P.; Norat, T.; Fahey, M.; Charrondière, U.R.; Hémon, B.; Casagrande, C.; Vignat, J.; et al. European Prospective Investigation into Cancer and Nutrition (EPIC): Study populations and data collection. Public Health Nutr. 2002, 5, 1113–1124. [Google Scholar] [CrossRef] [PubMed]
- Bernstein, L.; Allen, M.; Anton-Culver, H.; Deapen, D.; Horn-Ross, P.L.; Peel, D.; Pinder, R.; Reynolds, P.; Sullivan-Halley, J.; West, D.; et al. High breast cancer incidence rates among California teachers: Results from the California Teachers Study (United States). Cancer Causes Control 2002, 13, 625–635. [Google Scholar] [CrossRef] [PubMed]
- Kwan, M.L.; Ambrosone, C.B.; Lee, M.M.; Barlow, J.; Krathwohl, S.E.; Ergas, I.J.; Ashley, C.H.; Bittner, J.R.; Darbinian, J.; Stronach, K.; et al. The Pathways Study: A prospective study of breast cancer survivorship within Kaiser Permanente Northern California. Cancer Causes Control 2008, 19, 1065–1076. [Google Scholar] [CrossRef]
- Ladd-Acosta, C.; Fallin, M.D. The role of epigenetics in genetic and environmental epidemiology. Epigenomics 2016, 8, 271–283. [Google Scholar] [CrossRef]
- Ladd-Acosta, C.; Fallin, M.D. DNA Methylation Signatures as Biomarkers of Prior Environmental Exposures. Curr. Epidemiol. Rep. 2019, 6, 1–13. [Google Scholar] [CrossRef]
- Vineis, P.; Chatziioannou, A.; Cunliffe, V.T.; Flanagan, J.M.; Hanson, M.; Kirsch-Volders, M.; Kyrtopoulos, S. Epigenetic memory in response to environmental stressors. FASEB J. 2017, 31, 2241–2251. [Google Scholar] [CrossRef]
- Bird, A. Perceptions of epigenetics. Nature 2007, 447, 396–398. [Google Scholar] [CrossRef]
- Szyf, M.; McGowan, P.; Meaney, M.J. The social environment and the epigenome. Environ. Mol. Mutagen. 2008, 49, 46–60. [Google Scholar] [CrossRef] [PubMed]
- Klutstein, M.; Nejman, D.; Greenfield, R.; Cedar, H. DNA Methylation in Cancer and Aging. Cancer Res. 2016, 76, 3446–3450. [Google Scholar] [CrossRef] [PubMed]
- Esteller, M.; Corn, P.G.; Baylin, S.B.; Herman, J.G. A gene hypermethylation profile of human cancer. Cancer Res. 2001, 61, 3225–3229. [Google Scholar] [PubMed]
- Esteller, M.; Manel, E. (REVIEW) Epigenetics in cancer. N. Engl. J. Med. 2008, 358, 1148–1159. [Google Scholar] [CrossRef]
- Gorber, S.C.; Schofield-Hurwitz, S.; Hardt, J.; Levasseur, G.; Tremblay, M. The accuracy of self-reported smoking: A systematic review of the relationship between self-reported and cotinine-assessed smoking status. Nicotine Tob. Res. 2009, 11, 12–24. [Google Scholar] [CrossRef]
- Gao, X.; Jia, M.; Zhang, Y.; Breitling, L.P.; Brenner, H. DNA methylation changes of whole blood cells in response to active smoking exposure in adults: A systematic review of DNA methylation studies. Clin. Epigenetics 2015, 7, 113. [Google Scholar] [CrossRef]
- Philibert, R.; Hollenbeck, N.; Andersen, E.; Osborn, T.; Gerrard, M.; Gibbons, F.X.; Wang, K. A quantitative epigenetic approach for the assessment of cigarette consumption. Front. Psychol. 2015, 6, 656. [Google Scholar] [CrossRef]
- Zhang, Y.; Schöttker, B.; Florath, I.; Stock, C.; Butterbach, K.; Holleczek, B.; Mons, U.; Brenner, H. Smoking-Associated DNA Methylation Biomarkers and Their Predictive Value for All-Cause and Cardiovascular Mortality. Environ. Health Perspect. 2016, 124, 67–74. [Google Scholar] [CrossRef]
- Zhang, Y.; Yang, R.; Burwinkel, B.; Breitling, L.P.; Brenner, H. F2RL3 Methylation as a Biomarker of Current and Lifetime Smoking Exposures. Environ. Health Perspect. 2014, 122, 131–137. [Google Scholar] [CrossRef]
- Philibert, R.; Dogan, M.; Beach, S.R.H.; Mills, J.A.; Long, J.D. AHRR methylation predicts smoking status and smoking intensity in both saliva and blood DNA. Am. J. Med. Genet. Part B Neuropsychiatr. Genet. 2019, 183, 51–60. [Google Scholar] [CrossRef]
- Horvath, S. DNA methylation age of human tissues and cell types. Genome Biol. 2013, 14, 3156. [Google Scholar] [CrossRef] [PubMed]
- Hannum, G.; Guinney, J.; Zhao, L.; Zhang, L.; Hughes, G.; Sadda, S.V.; Klotzle, B.; Bibikova, M.; Fan, J.B.; Gao, Y.; et al. Genome-wide Methylation Profiles Reveal Quantitative Views of Human Aging Rates. Mol. Cell 2013, 49, 359–367. [Google Scholar] [CrossRef] [PubMed]
- Geurts, Y.M.; Dugué, P.-A.; Joo, J.E.; Makalic, E.; Jung, C.-H.; Guan, W.; Nguyen, S.; Grove, M.L.; Wong, E.M.; Hodge, A.M.; et al. Novel associations between blood DNA methylation and body mass index in middle-aged and older adults. Int. J. Obes. 2018, 42, 887–896. [Google Scholar] [CrossRef] [PubMed]
- Dick, K.J.; Nelson, C.P.; Tsaprouni, L.; Sandling, J.K.; Aïssi, D.; Wahl, S.; Meduri, E.; Morange, P.-E.; Gagnon, F.; Grallert, H.; et al. DNA methylation and body-mass index: A genome-wide analysis. Lancet 2014, 383, 1990–1998. [Google Scholar] [CrossRef]
- Sayols-Baixeras, S.; Subirana, I.; Fernández-Sanlés, A.; Sentí, M.; Lluís-Ganella, C.; Marrugat, J.; Elosua, R. DNA methylation and obesity traits: An epigenome-wide association study. The REGICOR study. Epigenetics 2017, 12, 909–916. [Google Scholar] [CrossRef]
- Wahl, S.; Drong, A.; Lehne, B.; Loh, M.; Scott, W.R.; Kunze, S.; Tsai, P.C.; Ried, J.S.; Zhang, W.; Yang, Y.; et al. Epigenome-wide association study of body mass index, and the adverse outcomes of adiposity. Nature 2017, 541, 81–86. [Google Scholar] [CrossRef]
- Al Muftah, W.A.; Al-Shafai, M.; Zaghlool, S.B.; Visconti, A.; Tsai, P.C.; Kumar, P.; Spector, T.; Bell, J.; Falchi, M.; Suhre, K. Epigenetic associations of type 2 diabetes and BMI in an Arab population. Clin. Epigenetics 2016, 8, 13. [Google Scholar] [CrossRef]
- Demerath, E.W.; Guan, W.; Grove, M.L.; Aslibekyan, S.; Mendelson, M.; Zhou, Y.H.; Hedman, Å.K.; Sandling, J.K.; Li, L.A.; Irvin, M.R.; et al. Epigenome-wide association study (EWAS) of BMI, BMI change and waist circumference in African American adults identifies multiple replicated loci. Hum. Mol. Genet. 2015, 24, 4464–4479. [Google Scholar] [CrossRef]
- Wilson, L.E.; Harlid, S.; Xu, Z.; Sandler, D.P.; Taylor, J.A. An epigenome-wide study of body mass index and DNA methylation in blood using participants from the Sister Study cohort. Int. J. Obes. 2017, 41, 194–199. [Google Scholar] [CrossRef]
- Caspers, M.; Blocquiaux, S.; Charlier, R.; Knaeps, S.; Lefevre, J.; De Bock, K.; Thomis, M. Intensity-Specific Differential Leukocyte DNA Methylation in Physical (In) Activity: An Exploratory Approach. Twin Res. Hum. Genet. 2018, 21, 101–111. [Google Scholar] [CrossRef]
- VAN Roekel, E.H.; Dugué, P.-A.; Jung, C.-H.; Joo, J.E.; Makalic, E.; Wong, E.E.M.; English, D.R.; Southey, M.C.; Giles, G.G.; Lynch, B.M.; et al. Physical Activity, Television Viewing Time, and DNA Methylation in Peripheral Blood. Med. Sci. Sports Exerc. 2019, 51, 490–498. [Google Scholar] [CrossRef] [PubMed]
- Panni, T.; Mehta, A.J.; Schwartz, J.D.; Baccarelli, A.A.; Just, A.C.; Wolf, K.; Wahl, S.; Cyrys, J.; Kunze, S.; Strauch, K.; et al. Genome-Wide Analysis of DNA Methylation and Fine Particulate Matter Air Pollution in Three Study Populations: KORA F3, KORA F4, and the Normative Aging Study. Environ. Health Perspect. 2016, 124, 983–990. [Google Scholar] [CrossRef] [PubMed]
- Rider, C.F.; Carlsten, C. Air pollution and DNA methylation: Effects of exposure in humans. Clin. Epigenetics 2019, 11, 131. [Google Scholar] [CrossRef] [PubMed]
- Gondalia, R.; Baldassari, A.; Holliday, K.M.; Justice, A.E.; Méndez-Giráldez, R.; Stewart, J.D.; Liao, D.; Yanosky, J.D.; Brennan, K.J.M.; Engel, S.M.; et al. Methylome-wide association study provides evidence of particulate matter air pollution-associated DNA methylation. Environ. Int. 2019, 132, 104723. [Google Scholar] [CrossRef] [PubMed]
- Khulan, B.; Manning, J.R.; Dunbar, D.R.; Seckl, J.R.; Raikkonen, K.; Eriksson, J.G.; Drake, A.J. Epigenomic profiling of men exposed to early-life stress reveals DNA methylation differences in association with current mental state. Transl. Psychiatry 2014, 4, e448. [Google Scholar] [CrossRef]
- Papale, L.A.; Seltzer, L.J.; Madrid, A.; Pollak, S.D.; Alisch, R.S. Differentially Methylated Genes in Saliva are linked to Childhood Stress. Sci. Rep. 2018, 8, 10785. [Google Scholar] [CrossRef]
- Li, M.; D’Arcy, C.; Li, X.; Zhang, T.; Joober, R.; Meng, X. What do DNA methylation studies tell us about depression? A systematic review. Transl. Psychiatry 2019, 9, 1–14. [Google Scholar] [CrossRef]
- Clark, S.L.; Aberg, K.A.; Nerella, S.; Kumar, G.; Mcclay, J.L.; Chen, W.; Xie, L.Y.; Harada, A.; Shabalin, A.A.; Gao, G.; et al. Combined Whole Methylome and Genomewide Association Study Implicates CNTN4 in Alcohol Use. Alcohol. Clin. Exp. Res. 2015, 39, 1396–1405. [Google Scholar] [CrossRef]
- Philibert, R.; Dogan, M.; Noel, A.; Miller, S.; Krukow, B.; Papworth, E.; Cowley, J.; Knudsen, A.; Beach, S.R.H.; Black, D. Genome-wide and digital polymerase chain reaction epigenetic assessments of alcohol consumption. Am. J. Med. Genet. Part B Neuropsychiatr. Genet. 2018, 177, 479–488. [Google Scholar] [CrossRef]
- Liu, C.; Marioni, R.E.; Hedman, A.K.; Pfeiffer, L.; Tsai, P.C.; Reynolds, L.M.; Just, A.C.; Duan, Q.; Boer, C.G.; Tanaka, T.; et al. A DNA methylation biomarker of alcohol consumption. Mol. Psychiatry 2018, 23, 422–433. [Google Scholar] [CrossRef]
- Dugué, P.-A.; Wilson, R.; Lehne, B.; Jayasekara, H.; Wang, X.; Jung, C.-H.; Joo, J.E.; Makalic, E.; Schmidt, D.F.; Baglietto, L.; et al. Alcohol consumption is associated with widespread changes in blood DNA methylation: Analysis of cross-sectional and longitudinal data. Addict. Biol. 2019, e12855. [Google Scholar] [CrossRef] [PubMed]
- Philibert, R.; Miller, S.; Noel, A.; Dawes, K.; Papworth, E.; Black, D.W.; Beach, S.R.H.; Long, J.D.; Mills, J.A.; Dogan, M. A Four Marker Digital PCR Toolkit for Detecting Heavy Alcohol Consumption and the Effectiveness of Its Treatment. J. Insur. Med. 2019, 48, 90–102. [Google Scholar] [CrossRef] [PubMed]
- Liangpunsakul, S.; Lai, X.; Ross, R.A.; Yu, Z.; Modlik, E.; Westerhold, C.; Heathers, L.; Paul, R.; O’Connor, S.; Crabb, D.W.; et al. Novel serum biomarkers for detection of excessive alcohol use. Alcohol. Clin. Exp. Res. 2015, 39, 556–565. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Elgizouli, M.; Schöttker, B.; Holleczek, B.; Nieters, A.; Brenner, H. Smoking-associated DNA methylation markers predict lung cancer incidence. Clin. Epigenetics 2016, 8, 127. [Google Scholar] [CrossRef]
- Jordahl, K.M.; Phipps, A.I.; Randolph, T.W.; Tindle, H.A.; Liu, S.; Tinker, L.F.; Kelsey, K.T.; White, E.; Bhatti, P. Differential DNA methylation in blood as a mediator of the association between cigarette smoking and bladder cancer risk among postmenopausal women. Epigenetics 2019, 14, 1065–1073. [Google Scholar] [CrossRef]
- Breitling, L.P.; Salzmann, K.; Rothenbacher, D.; Burwinkel, B.; Brenner, H. Smoking, F2RL3 methylation, and prognosis in stable coronary heart disease. Eur. Heart J. 2012, 33, 2841–2848. [Google Scholar] [CrossRef]
- Zhang, Y.; Wilson, R.; Heiss, J.; Breitling, L.P.; Saum, K.U.; Schöttker, B.; Holleczek, B.; Waldenberger, M.; Peters, A.; Brenner, H. DNA methylation signatures in peripheral blood strongly predict all-cause mortality. Nat. Commun. 2017, 8, 1–11. [Google Scholar] [CrossRef]
- Li, Y.; Pan, X.; Roberts, M.L.; Liu, P.; Kotchen, T.A.; Cowley, A.W.; Mattson, D.L.; Liu, Y.; Liang, M.; Kidambi, S. Stability of global methylation profiles of whole blood and extracted DNA under different storage durations and conditions. Epigenomics 2018, 10, 797–811. [Google Scholar] [CrossRef]
- Groen, K.; Lea, R.A.; Maltby, V.E.; Scott, R.J.; Lechner-Scott, J. Letter to the editor: Blood processing and sample storage have negligible effects on methylation. Clin. Epigenetics 2018, 10, 22. [Google Scholar] [CrossRef]
- Wu, H.C.; Cohn, B.A.; Cirillo, P.M.; Santella, R.M.; Terry, M.B. DDT exposure during pregnancy and DNA methylation alterations in female offspring in the Child Health and Development Study. Reprod. Toxicol. 2019. [Google Scholar] [CrossRef]
- Walker, C.L.; Ho, S. Developmental reprogramming of cancer susceptibility. Nat. Rev. Cancer 2012, 12, 479–486. [Google Scholar] [CrossRef] [PubMed]
- Küpers, L.K.; Xu, X.; Jankipersadsing, S.A.; Vaez, A.; la Bastide-van Gemert, S.; Scholtens, S.; Nolte, I.M.; Richmond, R.C.; Relton, C.L.; Felix, J.F.; et al. DNA methylation mediates the effect of maternal smoking during pregnancy on birthweight of the offspring. Int. J. Epidemiol. 2015, 44, 1224–1237. [Google Scholar] [CrossRef] [PubMed]
- Witt, S.H.; Frank, J.; Gilles, M.; Lang, M.; Treutlein, J.; Streit, F.; Wolf, I.A.C.; Peus, V.; Scharnholz, B.; Send, T.S.; et al. Impact on birth weight of maternal smoking throughout pregnancy mediated by DNA methylation. BMC Genom. 2018, 19, 290. [Google Scholar] [CrossRef] [PubMed]
- Cardenas, A.; Lutz, S.M.; Everson, T.M.; Perron, P.; Bouchard, L.; Hivert, M.-F. Mediation by placental DNA Methylation of the Association of Prenatal Maternal Smoking and Birth Weight. Am. J. Epidemiol. 2019, 188, 1878–1886. [Google Scholar] [CrossRef]
- Ladd-Acosta, C.; Shu, C.; Lee, B.K.; Gidaya, N.; Singer, A.; Schieve, L.A.; Schendel, D.E.; Jones, N.; Daniels, J.L.; Windham, G.C.; et al. Presence of an epigenetic signature of prenatal cigarette smoke exposure in childhood. Environ. Res. 2016, 144, 139–148. [Google Scholar] [CrossRef]
- Richmond, R.C.; Simpkin, A.J.; Woodward, G.; Gaunt, T.R.; Lyttleton, O.; McArdle, W.L.; Ring, S.M.; Smith, A.D.A.C.; Timpson, N.J.; Tilling, K.; et al. Prenatal exposure to maternal smoking and offspring DNA methylation across the lifecourse: Findings from the Avon Longitudinal Study of Parents and Children (ALSPAC). Hum. Mol. Genet. 2015, 24, 2201–2217. [Google Scholar] [CrossRef]
- Ladd-Acosta, C.; Feinberg, J.I.; Brown, S.C.; Lurmann, F.W.; Croen, L.A.; Hertz-Picciotto, I.; Newschaffer, C.J.; Feinberg, A.P.; Fallin, M.D.; Volk, H.E. Epigenetic marks of prenatal air pollution exposure found in multiple tissues relevant for child health. Environ. Int. 2019, 126, 363–376. [Google Scholar] [CrossRef]
- Liu, X.; Ye, Y.; Chen, Y.; Li, X.; Feng, B.; Cao, G.; Xiao, J.; Zeng, W.; Li, X.; Sun, J.; et al. Effects of prenatal exposure to air particulate matter on the risk of preterm birth and roles of maternal and cord blood LINE-1 methylation: A birth cohort study in Guangzhou, China. Environ. Int. 2019, 133, 105177. [Google Scholar] [CrossRef]
- Stein, A.D.; Kahn, H.S.; Rundle, A.; Zybert, P.A.; Van Der Pal-De Bruin, K.; Lumey, L.H. Anthropometric measures in middle age after exposure to famine during gestation: Evidence from the Dutch famine. Am. J. Clin. Nutr. 2007, 85, 869–876. [Google Scholar] [CrossRef]
- Lumey, L.H.; Stein, A.D.; Kahn, H.S.; Romijn, J.A. Lipid profiles in middle-aged men and women after famine exposure during gestation: The Dutch Hunger Winter Families Study. Am. J. Clin. Nutr. 2009, 89, 1737–1743. [Google Scholar] [CrossRef]
- Tobi, E.W.; Slieker, R.C.; Luijk, R.; Dekkers, K.F.; Stein, A.D.; Xu, K.M.; Biobank-based Integrative Omics Studies Consortium; Slagboom, P.E.; van Zwet, E.W.; Lumey, L.H.; et al. DNA methylation as a mediator of the association between prenatal adversity and risk factors for metabolic disease in adulthood. Sci. Adv. 2018, 4, 4364. [Google Scholar] [CrossRef] [PubMed]
- Painter, R.C.; De Rooij, S.R.; Bossuyt, P.M.M.; Osmond, C.; Barker, D.J.P.; Bleker, O.P.; Roseboom, T.J. A possible link between prenatal exposure to famine and breast cancer: A preliminary study. Am. J. Hum. Biol. 2006, 18, 853–856. [Google Scholar] [CrossRef] [PubMed]
- He, D.; Fang, Y.; Gunter, M.J.; Xu, D.; Zhao, Y.; Zhou, J.; Fang, H.; Xu, W.H. Incidence of breast cancer in Chinese women exposed to the 1959–1961 great Chinese famine. BMC Cancer 2017, 17, 824. [Google Scholar] [CrossRef] [PubMed]
- Burton, G.J.; Fowden, A.L.; Thornburg, K.L. Placental origins of chronic disease. Physiol. Rev. 2016, 96, 1509–1565. [Google Scholar] [CrossRef]
- Misra, D.P.; Astone, N.; Lynch, C.D. Maternal smoking and birth weight: Interaction with parity and mother’s own in utero exposure to smoking. Epidemiology 2005, 16, 288–293. [Google Scholar] [CrossRef]
- Hyppönen, E.; Smith, G.D.; Power, C. Effects of grandmothers’ smoking in pregnancy on birth weight: Intergenerational cohort study. Br. Med. J. 2003, 327, 898–900. [Google Scholar] [CrossRef]
- Li, Y.F.; Langholz, B.; Salam, M.T.; Gilliland, F.D. Maternal and grandmaternal smoking patterns are associated with early childhood asthma. Chest 2005, 127, 1232–1241. [Google Scholar] [CrossRef]
- Spycher, B.D.; Lupatsch, J.E.; Huss, A.; Rischewski, J.; Schindera, C.; Spoerri, A.; Vermeulen, R.; Kuehni, C.E. Parental occupational exposure to benzene and the risk of childhood cancer: A census-based cohort study. Environ. Int. 2017, 108, 84–91. [Google Scholar] [CrossRef]
- Ali, R.; Yu, C.L.; Wu, M.T.; Ho, C.K.; Pan, B.J.; Smith, T.; Christiani, D.C. A case-control study of parental occupation, leukemia, and brain tumors in an industrial city in Taiwan. J. Occup. Environ. Med. 2004, 46, 985–992. [Google Scholar] [CrossRef]
- Monge, P.; Wesseling, C.; Guardado, J.; Lundberg, I.; Ahlbom, A.; Cantor, K.P.; Weiderpass, E.; Partanen, T. Parental occupational exposure to pesticides and the risk of childhood leukemia in Costa Rica. Scand. J. Work. Environ. Heal. 2007, 33, 293–303. [Google Scholar] [CrossRef]
- Infante-Rivard, C.; Sinnett, D. Preconceptional paternal exposure to pesticides and increased risk of childhood leukaemia [3]. Lancet 1999, 354, 1819. [Google Scholar] [CrossRef]
- Perera, F.; Herbstman, J. Prenatal environmental exposures, epigenetics, and disease. Reprod. Toxicol. 2011, 31, 363–373. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Liu, H.; Sun, Z. Lamarck rises from his grave: Parental environment-induced epigenetic inheritance in model organisms and humans. Biol. Rev. 2017, 92, 2084–2111. [Google Scholar] [CrossRef] [PubMed]
- Aarabi, M.; San Gabrie, M.C.; Chan, D.; Behan, N.A.; Caron, M.; Pastinen, T.; Bourque, G.; MacFarlane, A.J.; Zini, A.; Trasler, J. High-dose folic acid supplementation alters the human sperm methylome and is influenced by the MTHFR C677T polymorphism. Hum. Mol. Genet. 2015, 24, 6301–6313. [Google Scholar] [CrossRef] [PubMed]
- Ouko, L.A.; Shantikumar, K.; Knezovich, J.; Haycock, P.; Schnugh, D.J.; Ramsay, M. Effect of alcohol consumption on CpG methylation in the differentially methylated regions of H19 and IG-DMR in male gametes—Implications for fetal alcohol spectrum disorders. Alcohol. Clin. Exp. Res. 2009, 33, 1615–1627. [Google Scholar] [CrossRef]
- Donkin, I.; Versteyhe, S.; Ingerslev, L.R.; Qian, K.; Mechta, M.; Nordkap, L.; Mortensen, B.; Appel, E.V.R.; Jørgensen, N.; Kristiansen, V.B.; et al. Obesity and bariatric surgery drive epigenetic variation of spermatozoa in humans. Cell Metab. 2016, 23, 369–378. [Google Scholar] [CrossRef]
- Laqqan, M.; Tierling, S.; Alkhaled, Y.; Porto, C.L.; Solomayer, E.F.; Hammadeh, M.E. Aberrant DNA methylation patterns of human spermatozoa in current smoker males. Reprod. Toxicol. 2017, 71, 126–133. [Google Scholar] [CrossRef]
- Jenkins, T.G.; James, E.R.; Alonso, D.F.; Hoidal, J.R.; Murphy, P.J.; Hotaling, J.M.; Cairns, B.R.; Carrell, D.T.; Aston, K.I. Cigarette smoking significantly alters sperm DNA methylation patterns. Andrology 2017, 5, 1089–1099. [Google Scholar] [CrossRef]
- Miao, M.; Zhou, X.; Li, Y.; Zhang, O.; Zhou, Z.; Li, T.; Yuan, W.; Li, R.; Li, D.K. LINE-1 hypomethylation in spermatozoa is associated with Bisphenol A exposure. Andrology 2014, 2, 138–144. [Google Scholar] [CrossRef]
- Wu, H.; Estill, M.S.; Shershebnev, A.; Suvorov, A.; Krawetz, S.A.; Whitcomb, B.W.; Dinnie, H.; Rahil, T.; Sites, C.K.; Pilsner, J.R. Preconception urinary phthalate concentrations and sperm DNA methylation profiles among men undergoing IVF treatment: A cross-sectional study. Hum. Reprod. 2017, 32, 2159–2169. [Google Scholar] [CrossRef]
- Fontelles, C.C.; Guido, L.N.; Rosim, M.P.; de Oliveira Andrade, F.; Jin, L.; Inchauspe, J.; Pires, V.C.; de Castro, I.A.; Hilakivi-Clarke, L.; de Assis, S.; et al. Paternal programming of breast cancer risk in daughters in a rat model: Opposing effects of animal- and plant-based high-fat diets. Breast Cancer Res. 2016, 18, 71. [Google Scholar] [CrossRef] [PubMed]
- De Assis, S.; Warri, A.; Cruz, M.I.; Laja, O.; Tian, Y.; Zhang, B.; Wang, Y.; Huang, T.H.M.; Hilakivi-Clarke, L. High-fat or ethinyl-oestradiol intake during pregnancy increases mammary cancer risk in several generations of offspring. Nat. Commun. 2012, 3, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, N.M.; de Oliveira Andrade, F.; Jin, L.; Zhang, X.; Macon, M.; Cruz, M.I.; Benitez, C.; Wehrenberg, B.; Yin, C.; Wang, X.; et al. Maternal intake of high n-6 polyunsaturated fatty acid diet during pregnancy causes transgenerational increase in mammary cancer risk in mice. Breast Cancer Res. 2017, 19, 77. [Google Scholar] [CrossRef] [PubMed]
- Tehranifar, P.; Wu, H.C.; McDonald, J.A.; Jasmine, F.; Santella, R.M.; Gurvich, I.; Flom, J.D.; Terry, M.B. Maternal cigarette smoking during pregnancy and offspring DNA methylation in midlife. Epigenetics 2018, 13, 129–134. [Google Scholar] [CrossRef] [PubMed]
- Richmond, R.C.; Suderman, M.; Langdon, R.; Relton, C.L.; Davey Smith, G. DNA methylation as a marker for prenatal smoke exposure in adults. Int. J. Epidemiol. 2018, 47, 1120–1130. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, C.H.; Larsen, A.; Nielsen, A.L. DNA methylation alterations in response to prenatal exposure of maternal cigarette smoking: A persistent epigenetic impact on health from maternal lifestyle? Arch. Toxicol. 2016, 90, 231–245. [Google Scholar] [CrossRef]
- Barrès, R.; Yan, J.; Egan, B.; Treebak, J.T.; Rasmussen, M.; Fritz, T.; Caidahl, K.; Krook, A.; O’Gorman, D.J.; Zierath, J.R. Acute exercise remodels promoter methylation in human skeletal muscle. Cell Metab. 2012, 15, 405–411. [Google Scholar] [CrossRef]
- Alibegovic, A.C.; Sonne, M.P.; Højbjerre, L.; Bork-Jensen, J.; Jacobsen, S.; Nilsson, E.; Færch, K.; Hiscock, N.; Mortensen, B.; Friedrichsen, M.; et al. Insulin resistance induced by physical inactivity is associated with multiple transcriptional changes in skeletal muscle in young men. Am. J. Physiol. Endocrinol. Metab. 2010, 299, E752–E763. [Google Scholar] [CrossRef]
- Prunicki, M.; Stell, L.; Dinakarpandian, D.; de Planell-Saguer, M.; Lucas, R.W.; Hammond, S.K.; Balmes, J.R.; Zhou, X.; Paglino, T.; Sabatti, C.; et al. Exposure to NO2, CO, and PM2.5 is linked to regional DNA methylation differences in asthma. Clin. Epigenetics 2018, 10, 2. [Google Scholar] [CrossRef]
- Nadeau, K.; McDonald-Hyman, C.; Noth, E.M.; Pratt, B.; Hammond, S.K.; Balmes, J.; Tager, I. Ambient air pollution impairs regulatory T-cell function in asthma. J. Allergy Clin. Immunol. 2010, 126, 845–852. [Google Scholar] [CrossRef]
- Bind, M.-A.C.; Coull, B.A.; Peters, A.; Baccarelli, A.A.; Tarantini, L.; Cantone, L.; Vokonas, P.S.; Koutrakis, P.; Schwartz, J.D. Beyond the Mean: Quantile Regression to Explore the Association of Air Pollution with Gene-Specific Methylation in the Normative Aging Study. Environ. Health Perspect. 2015, 123, 759–765. [Google Scholar] [CrossRef] [PubMed]
- Bind, M.A.; Lepeule, J.; Zanobetti, A.; Gasparrini, A.; Baccarelli, A.; Coull, B.A.; Tarantini, L.; Vokonas, P.S.; Koutrakis, P.; Schwartz, J. Air pollution and gene-specific methylation in the Normative Aging Study:Association, effect modification, and mediation analysis. Epigenetics 2014, 9, 448–458. [Google Scholar] [CrossRef]
- Li, H.; Chen, R.; Cai, J.; Cui, X.; Huang, N.; Kan, H. Short-term exposure to fine particulate air pollution and genome-wide DNA methylation: A randomized, double-blind, crossover trial. Environ. Int. 2018, 120, 130–136. [Google Scholar] [CrossRef] [PubMed]
- Peng, C.; Bind, M.A.C.; Colicino, E.; Kloog, I.; Byun, H.M.; Cantone, L.; Trevisi, L.; Zhong, J.; Brennan, K.; Dereix, A.E.; et al. Particulate air pollution and fasting blood glucose in nondiabetic individuals: Associations and epigenetic mediation in the normative aging study, 2000–2011. Environ. Health Perspect. 2016, 124, 1715–1721. [Google Scholar] [CrossRef] [PubMed]
- Lepeule, J.; Bind, M.A.C.; Baccarelli, A.A.; Koutrakis, P.; Tarantini, L.; Litonjua, A.; Sparrow, D.; Vokonas, P.; Schwartz, J.D. Epigenetic influences on associations between air pollutants and lung function in elderly men: The normative aging study. Environ. Health Perspect. 2014, 122, 566–572. [Google Scholar] [CrossRef] [PubMed]
- Cantone, L.; Iodice, S.; Tarantini, L.; Albetti, B.; Restelli, I.; Vigna, L.; Bonzini, M.; Pesatori, A.C.; Bollati, V. Particulate matter exposure is associated with inflammatory gene methylation in obese subjects. Environ. Res. 2017, 152, 478–484. [Google Scholar] [CrossRef]
- Kim, T.Y.; Kim, S.J.; Chung, H.G.; Choi, J.H.; Kim, S.H.; Kang, J.I. Epigenetic alterations of the BDNF gene in combat-related post-traumatic stress disorder. Acta Psychiatr. Scand. 2017, 135, 170–179. [Google Scholar] [CrossRef]
- Li, S.; Chen, M.; Li, Y.; Tollefsbol, T.O. Prenatal epigenetics diets play protective roles against environmental pollution. Clin. Epigenetics 2019, 11, 82. [Google Scholar] [CrossRef]
- Santos, H.P.; Nephew, B.C.; Bhattacharya, A.; Tan, X.; Smith, L.; Alyamani, R.A.S.; Martin, E.M.; Perreira, K.; Fry, R.C.; Murgatroyd, C. Discrimination exposure and DNA methylation of stress-related genes in Latina mothers. Psychoneuroendocrinology 2018, 98, 131–138. [Google Scholar] [CrossRef]
- Kertes, D.A.; Kamin, H.S.; Hughes, D.A.; Rodney, N.C.; Bhatt, S.; Mulligan, C.J. Prenatal Maternal Stress Predicts Methylation of Genes Regulating the Hypothalamic-Pituitary-Adrenocortical System in Mothers and Newborns in the Democratic Republic of Congo. Child Dev. 2016, 87, 61–72. [Google Scholar] [CrossRef]
- Kertes, D.A.; Bhatt, S.S.; Kamin, H.S.; Hughes, D.A.; Rodney, N.C.; Mulligan, C.J. BNDF methylation in mothers and newborns is associated with maternal exposure to war trauma. Clin. Epigenetics 2017, 9, 68. [Google Scholar] [CrossRef] [PubMed]
- Peng, H.; Zhu, Y.; Strachan, E.; Fowler, E.; Bacus, T.; Roy-Byrne, P.; Goldberg, J.; Vaccarino, V.; Zhao, J. Childhood Trauma, DNA Methylation of Stress-Related Genes, and Depression: Findings from Two Monozygotic Twin Studies. Psychosom. Med. 2018, 80, 599–608. [Google Scholar] [CrossRef] [PubMed]
- Unternaehrer, E.; Meyer, A.H.; Burkhardt, S.C.A.; Dempster, E.; Staehli, S.; Theill, N.; Lieb, R.; Meinlschmidt, G. Childhood maternal care is associated with DNA methylation of the genes for brain-derived neurotrophic factor (BDNF) and oxytocin receptor (OXTR) in peripheral blood cells in adult men and women. Stress 2015, 18, 451–461. [Google Scholar] [CrossRef] [PubMed]
- Wrigglesworth, J.; Ancelin, M.L.; Ritchie, K.; Ryan, J. Association between DNA methylation of the KITLG gene and cortisol levels under stress: A replication study. Stress 2019, 22, 162–168. [Google Scholar] [CrossRef] [PubMed]
- Houtepen, L.C.; Vinkers, C.H.; Carrillo-Roa, T.; Hiemstra, M.; Van Lier, P.A.; Meeus, W.; Branje, S.; Heim, C.M.; Nemeroff, C.B.; Mill, J.; et al. Genome-wide DNA methylation levels and altered cortisol stress reactivity following childhood trauma in humans. Nat. Commun. 2016, 7, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Romens, S.E.; Mcdonald, J.; Svaren, J.; Pollak, S.D. Associations Between Early Life Stress and Gene Methylation in Children. Child Dev. 2015, 86, 303–309. [Google Scholar] [CrossRef]
- Van Der Knaap, L.J.; Riese, H.; Hudziak, J.J.; Verbiest, M.M.P.J.; Verhulst, F.C.; Oldehinkel, A.J.; Van Oort, F.V.A. Glucocorticoid receptor gene (NR3C1) methylation following stressful events between birth and adolescence. the TRAILS study. Transl. Psychiatry 2014, 4, e381. [Google Scholar] [CrossRef]
- Yehuda, R.; Daskalakis, N.P.; Lehrner, A.; Desarnaud, F.; Bader, H.N.; Makotkine, I.; Flory, J.D.; Bierer, L.M.; Meaney, M.J. Influences of maternal and paternal PTSD on epigenetic regulation of the glucocorticoid receptor gene in Holocaust survivor offspring. Am. J. Psychiatry 2014, 171, 872–880. [Google Scholar] [CrossRef]
- Yehuda, R.; Flory, J.D.; Bierer, L.M.; Henn-Haase, C.; Lehrner, A.; Desarnaud, F.; Makotkine, I.; Daskalakis, N.P.; Marmar, C.R.; Meaney, M.J. Lower Methylation of Glucocorticoid Receptor Gene Promoter 1F in Peripheral Blood of Veterans with Posttraumatic Stress Disorder. Biol. Psychiatry 2015, 77, 356–364. [Google Scholar] [CrossRef]
- Lee, R.S.; Mahon, P.B.; Zandi, P.P.; McCaul, M.E.; Yang, X.; Bali, U.; Wand, G.S. DNA methylation and sex-specific expression of FKBP5 as correlates of one-month bedtime cortisol levels in healthy individuals. Psychoneuroendocrinology 2018, 97, 164–173. [Google Scholar] [CrossRef]
- Kang, J.I.; Kim, T.Y.; Choi, J.H.; So, H.S.; Kim, S.J. Allele-specific DNA methylation level of FKBP5 is associated with post-traumatic stress disorder. Psychoneuroendocrinology 2019, 103, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Snijders, C.; Maihofer, A.X.; Ratanatharathorn, A.; Baker, D.G.; Boks, M.P.; Geuze, E.; Jain, S.; Kessler, R.C.; Pishva, E.; Risbrough, V.B.; et al. Longitudinal epigenome-wide association studies of three male military cohorts reveal multiple CpG sites associated with post-traumatic stress disorder. Clin. Epigenetics 2020, 12, 11. [Google Scholar] [CrossRef] [PubMed]
- Roy, B.; Shelton, R.C.; Dwivedi, Y. DNA methylation and expression of stress related genes in PBMC of MDD patients with and without serious suicidal ideation. J. Psychiatr. Res. 2017, 89, 115–124. [Google Scholar] [CrossRef] [PubMed]
- Won, E.; Choi, S.; Kang, J.; Kim, A.; Han, K.-M.; Chang, H.S.; Tae, W.S.; Son, K.R.; Joe, S.-H.; Lee, M.-S.; et al. Association between reduced white matter integrity in the corpus callosum and serotonin transporter gene DNA methylation in medication-naive patients with major depressive disorder. Transl. Psychiatry 2016, 6, e866. [Google Scholar] [CrossRef]
- Na, K.-S.; Won, E.; Kang, J.; Chang, H.S.; Yoon, H.-K.; Tae, W.S.; Kim, Y.-K.; Lee, M.-S.; Joe, S.-H.; Kim, H.; et al. Brain-derived neurotrophic factor promoter methylation and cortical thickness in recurrent major depressive disorder. Sci. Rep. 2016, 6, 21089. [Google Scholar] [CrossRef] [PubMed]
- Na, K.-S.; Chang, H.S.; Won, E.; Han, K.-M.; Choi, S.; Tae, W.S.; Yoon, H.-K.; Kim, Y.-K.; Joe, S.-H.; Jung, I.-K.; et al. Association between glucocorticoid receptor methylation and hippocampal subfields in major depressive disorder. PLoS ONE 2014, 9, e85425. [Google Scholar] [CrossRef] [PubMed]
- Chagnon, Y.C.; Potvin, O.; Hudon, C.; Préville, M. DNA methylation and single nucleotide variants in the brain-derived neurotrophic factor (BDNF) and oxytocin receptor (OXTR) genes are associated with anxiety/depression in older women. Front. Genet. 2015, 6, 230. [Google Scholar] [CrossRef]
- Kim, J.-M.; Stewart, R.; Kang, H.-J.; Bae, K.-Y.; Kim, S.-W.; Shin, I.-S.; Hong, Y.J.; Ahn, Y.; Jeong, M.H.; Yoon, J.-S. BDNF methylation and depressive disorder in acute coronary syndrome: The K-DEPACS and EsDEPACS studies. Psychoneuroendocrinology 2015, 62, 159–165. [Google Scholar] [CrossRef]
- Kim, J.-M.; Stewart, R.; Kang, H.-J.; Kim, S.-Y.; Kim, S.-W.; Shin, I.-S.; Park, M.-S.; Kim, H.-R.; Shin, M.-G.; Cho, K.-H.; et al. A longitudinal study of BDNF promoter methylation and genotype with poststroke depression. J. Affect. Disord. 2013, 149, 93–99. [Google Scholar] [CrossRef]
- Kim, J.-M.; Stewart, R.; Kang, H.-J.; Kim, S.-W.; Shin, I.-S.; Kim, H.-R.; Shin, M.-G.; Kim, J.-T.; Park, M.-S.; Cho, K.-H.; et al. A longitudinal study of SLC6A4 DNA promoter methylation and poststroke depression. J. Psychiatr. Res. 2013, 47, 1222–1227. [Google Scholar] [CrossRef]
- Januar, V.; Ancelin, M.L.; Ritchie, K.; Saffery, R.; Ryan, J. BDNF promoter methylation and genetic variation in late-life depression. Transl. Psychiatry 2015, 5, e619. [Google Scholar] [CrossRef] [PubMed]
- Carlberg, L.; Scheibelreiter, J.; Hassler, M.R.; Schloegelhofer, M.; Schmoeger, M.; Ludwig, B.; Kasper, S.; Aschauer, H.; Egger, G.; Schosser, A. Brain-derived neurotrophic factor (BDNF)—Epigenetic regulation in unipolar and bipolar affective disorder. J. Affect. Disord. 2014, 168, 399–406. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Goldberg, J.; Bremner, J.D.; Vaccarino, V. Association between promoter methylation of serotonin transporter gene and depressive symptoms: A monozygotic twin study. Psychosom. Med. 2013, 75, 523–529. [Google Scholar] [CrossRef] [PubMed]
- Kang, H.-J.; Kim, J.-M.; Stewart, R.; Kim, S.-Y.; Bae, K.-Y.; Kim, S.-W.; Shin, I.-S.; Shin, M.-G.; Yoon, J.-S. Association of SLC6A4 methylation with early adversity, characteristics and outcomes in depression. Prog. Neuropsychopharmacol. Biol. Psychiatry 2013, 44, 23–28. [Google Scholar] [CrossRef]
- Fuchikami, M.; Morinobu, S.; Segawa, M.; Okamoto, Y.; Yamawaki, S.; Ozaki, N.; Inoue, T.; Kusumi, I.; Koyama, T.; Tsuchiyama, K.; et al. DNA methylation profiles of the brain-derived neurotrophic factor (BDNF) gene as a potent diagnostic biomarker in major depression. PLoS ONE 2011, 6, e23881. [Google Scholar] [CrossRef]
- Nantharat, M.; Wanitchanon, T.; Amesbutr, M.; Tammachote, R.; Praphanphoj, V. Glucocorticoid receptor gene (NR3C1) promoter is hypermethylated in Thai females with major depressive disorder. Genet. Mol. Res. 2015, 14, 19071–19079. [Google Scholar] [CrossRef]
- Bustamante, A.C.; Aiello, A.E.; Galea, S.; Ratanatharathorn, A.; Noronha, C.; Wildman, D.E.; Uddin, M. Glucocorticoid receptor DNA methylation, childhood maltreatment and major depression. J. Affect. Disord. 2016, 206, 181–188. [Google Scholar] [CrossRef]
- Sikdar, S.; Joehanes, R.; Joubert, B.R.; Xu, C.J.; Vives-Usano, M.; Rezwan, F.I.; Felix, J.F.; Ward, J.M.; Guan, W.; Richmond, R.C.; et al. Comparison of smoking-related DNA methylation between newborns from prenatal exposure and adults from personal smoking. Epigenomics 2019, 11, 1487–1500. [Google Scholar] [CrossRef]
- Lee, K.W.K.; Richmond, R.; Hu, P.; French, L.; Shin, J.; Bourdon, C.; Reischl, E.; Waldenberger, M.; Zeilinger, S.; Gaunt, T.; et al. Prenatal exposure to maternal cigarette smoking and DNA methylation: Epigenome-wide association in a discovery sample of adolescents and replication in an independent cohort at birth through 17 years of age. Environ. Health Perspect. 2015, 123, 193–199. [Google Scholar] [CrossRef]
- Lussier, A.A.; Morin, A.M.; MacIsaac, J.L.; Salmon, J.; Weinberg, J.; Reynolds, J.N.; Pavlidis, P.; Chudley, A.E.; Kobor, M.S. DNA methylation as a predictor of fetal alcohol spectrum disorder. Clin. Epigenetics 2018, 10, 5. [Google Scholar] [CrossRef]
- Portales-Casamar, E.; Lussier, A.A.; Jones, M.J.; MacIsaac, J.L.; Edgar, R.D.; Mah, S.M.; Barhdadi, A.; Provost, S.; Lemieux-Perreault, L.-P.; Cynader, M.S.; et al. DNA methylation signature of human fetal alcohol spectrum disorder. Epigenetics Chromatin 2016, 9, 25. [Google Scholar] [CrossRef] [PubMed]
- Sharp, G.C.; Salas, L.A.; Monnereau, C.; Allard, C.; Yousefi, P.; Everson, T.M.; Bohlin, J.; Xu, Z.; Huang, R.C.; Reese, S.E.; et al. Maternal BMI at the start of pregnancy and offspring epigenome-wide DNA methylation: Findings from the pregnancy and childhood epigenetics (PACE) consortium. Hum. Mol. Genet. 2017, 26, 4067–4085. [Google Scholar] [CrossRef] [PubMed]
- Breton, C.V.; Gao, L.; Yao, J.; Siegmund, K.D.; Lurmann, F.; Gilliland, F. Particulate matter, the newborn methylome, and cardio-respiratory health outcomes in childhood. Environ. Epigenetics 2016, 2, dvw005. [Google Scholar] [CrossRef] [PubMed]
- Nagarajan, S.; Seddighzadeh, B.; Baccarelli, A.; Wise, L.A.; Williams, M.; Shields, A.E. Adverse maternal exposures, methylation of glucocorticoid-related genes and perinatal outcomes: A systematic review. Epigenomics 2016, 8, 925–944. [Google Scholar] [CrossRef]
- Palma-Gudiel, H.; Córdova-Palomera, A.; Eixarch, E.; Deuschle, M.; Fañanás, L. Maternal psychosocial stress during pregnancy alters the epigenetic signature of the glucocorticoid receptor gene promoter in their offspring: A meta-analysis. Epigenetics 2015, 10, 893–902. [Google Scholar] [CrossRef]
- Cardenas, A.; Faleschini, S.; Cortes Hidalgo, A.; Rifas-Shiman, S.L.; Baccarelli, A.A.; Demeo, D.L.; Litonjua, A.A.; Neumann, A.; Felix, J.F.; Jaddoe, V.W.V.; et al. Prenatal maternal antidepressants, anxiety, and depression and offspring DNA methylation: Epigenome-wide associations at birth and persistence into early childhood. Clin. Epigenetics 2019, 11, 56. [Google Scholar] [CrossRef]
- Radtke, K.M.; Ruf, M.; Gunter, H.M.; Dohrmann, K.; Schauer, M.; Meyer, A.; Elbert, T. Transgenerational impact of intimate partner violence on methylation in the promoter of the glucocorticoid receptor. Transl. Psychiatry 2011, 1, e21. [Google Scholar] [CrossRef]
- Perroud, N.; Rutembesa, E.; Paoloni-Giacobino, A.; Mutabaruka, J.; Mutesa, L.; Stenz, L.; Malafosse, A.; Karege, F. The Tutsi genocide and transgenerational transmission of maternal stress: Epigenetics and biology of the HPA axis. World J. Biol. Psychiatry 2014, 15, 334–345. [Google Scholar] [CrossRef]
- Tobi, E.W.; Goeman, J.J.; Monajemi, R.; Gu, H.; Putter, H.; Zhang, Y.; Slieker, R.C.; Stok, A.P.; Thijssen, P.E.; Müller, F.; et al. DNA methylation signatures link prenatal famine exposure to growth and metabolism. Nat. Commun. 2014, 5, 5592. [Google Scholar] [CrossRef]
- Wang, Z.; Song, J.; Li, Y.; Dong, B.; Zou, Z.; Ma, J. Early-Life Exposure to the Chinese Famine Is Associated with Higher Methylation Level in the INSR Gene in Later Adulthood. Sci. Rep. 2019, 9, 1–9. [Google Scholar] [CrossRef]
- Irwin, R.E.; Thursby, S.-J.; Ondičová, M.; Pentieva, K.; McNulty, H.; Richmond, R.C.; Caffrey, A.; Lees-Murdock, D.J.; McLaughlin, M.; Cassidy, T.; et al. A randomized controlled trial of folic acid intervention in pregnancy highlights a putative methylation-regulated control element at ZFP57. Clin. Epigenetics 2019, 11, 31. [Google Scholar] [CrossRef] [PubMed]
- Latendresse, S.J.; Musci, R.; Maher, B.S. Critical Issues in the Inclusion of Genetic and Epigenetic Information in Prevention and Intervention Trials. Prev. Sci. 2018, 19, 58–67. [Google Scholar] [CrossRef] [PubMed]
- Pavanello, S.; Campisi, M.; Tona, F.; Dal Lin, C.; Iliceto, S. Exploring Epigenetic Age in Response to Intensive Relaxing Training: A Pilot Study to Slow Down Biological Age. Int. J. Environ. Res. Public Health 2019, 16, 3074. [Google Scholar] [CrossRef]
- Chen, J.; Hutchison, K.E.; Bryan, A.D.; Filbey, F.M.; Calhoun, V.D.; Claus, E.D.; Lin, D.; Sui, J.; Du, Y.; Liu, J. Opposite Epigenetic Associations With Alcohol Use and Exercise Intervention. Front. Psychiatry 2018, 9, 594. [Google Scholar] [CrossRef] [PubMed]
- Rönn, T.; Volkov, P.; Davegårdh, C.; Dayeh, T.; Hall, E.; Olsson, A.H.; Nilsson, E.; Tornberg, Å.; Dekker Nitert, M.; Eriksson, K.F.; et al. A Six Months Exercise Intervention Influences the Genome-wide DNA Methylation Pattern in Human Adipose Tissue. PLoS Genet. 2013, 9, e1003572. [Google Scholar] [CrossRef]
- Bollepalli, S.; Kaye, S.; Heinonen, S.; Kaprio, J.; Rissanen, A.; Virtanen, K.A.; Pietiläinen, K.H.; Ollikainen, M. Subcutaneous adipose tissue gene expression and DNA methylation respond to both short- and long-term weight loss. Int. J. Obes. 2018, 42, 412–423. [Google Scholar] [CrossRef]
- Ozanne, E.M.; Wittenberg, E.; Garber, J.E.; Weeks, J.C. Breast cancer prevention: Patient decision making and risk communication in the high risk setting. Breast J. 2010, 16, 38–47. [Google Scholar] [CrossRef]
- Dolinoy, D.C.; Huang, D.; Jirtle, R.L. Maternal nutrient supplementation counteracts bisphenol A-induced DNA hypomethylation in early development. Proc. Natl. Acad. Sci. USA 2007, 104, 13056–13061. [Google Scholar] [CrossRef]
- Connolly, R.M.; Li, H.; Jankowitz, R.C.; Zhang, Z.; Rudek, M.A.; Jeter, S.C.; Slater, S.A.; Powers, P.; Wolff, A.C.; Fetting, J.H.; et al. Combination epigenetic therapy in advanced breast cancer with 5-azacitidine and entinostat: A phase II national cancer institute/stand up to cancer study. Clin. Cancer Res. 2017, 23, 2691–2701. [Google Scholar] [CrossRef]
- Patnaik, S. Anupriya Drugs targeting epigenetic modifications and plausible therapeutic strategies against colorectal cancer. Front. Pharmacol. 2019, 10, 588. [Google Scholar] [CrossRef]
- Rockhill, B.; Spiegelman, D.; Byrne, C.; Hunter, D.J.; Colditz, G.A. Validation of the Gail et al. model of breast cancer risk prediction and implications for chemoprevention. J. Natl. Cancer Inst. 2001, 93, 358–366. [Google Scholar] [CrossRef] [PubMed]
- Louro, J.; Posso, M.; Hilton Boon, M.; Román, M.; Domingo, L.; Castells, X.; Sala, M. A systematic review and quality assessment of individualised breast cancer risk prediction models. Br. J. Cancer 2019, 121, 76–85. [Google Scholar] [CrossRef] [PubMed]
- Moran, S.; Arribas, C.; Esteller, M. Validation of a DNA methylation microarray for 850,000 CpG sites of the human genome enriched in enhancer sequences. Epigenomics 2016, 8, 389–399. [Google Scholar] [CrossRef] [PubMed]
- Andra, S.S.; Austin, C.; Patel, D.; Dolios, G.; Awawda, M.; Arora, M. Trends in the application of high-resolution mass spectrometry for human biomonitoring: An analytical primer to studying the environmental chemical space of the human exposome. Environ. Int. 2017, 100, 32–61. [Google Scholar] [CrossRef] [PubMed]
- Jones, D.P. Sequencing the exposome: A call to action. Toxicol. Reports 2016, 3, 29–45. [Google Scholar] [CrossRef] [PubMed]
- National Research Council. Environmental Epidemiology: Volume 2: Use of the Gray Literature and Other Data in Environmental Epidemiology; National Academies Press (US): Washington, DC, USA, 1997.
- Pembrey, M.; Saffery, R.; Bygren, L.O. Human transgenerational responses to early-life experience: Potential impact on development, health and biomedical research. J. Med. Genet. 2014, 51, 563–572. [Google Scholar] [CrossRef]
| Exposure | Genes Containing Differentially Methylated CpG Sites or Regions | References |
|---|---|---|
| Self | ||
| Aging | Panel of 353 CpG sites (Horvath method); Panel of 71 CpG sites (Hannum method) | [41,42] |
| Cigarette smoking | AHRR, F2RL3, GPR15 | [36,37,38,39,40] |
| Alcohol | CNTN4, CDC42BPB | [58,59,61] |
| BMI | ABCG1, CPT1A, SREBF1, HIF1A | [43,44,46,47,48] |
| Physical activity | PPARGC1A | [107,108] |
| Air pollution | FOXP3, IFN-γ, ICAM-1, TLR-2 | [109,110,111,112,113,114,115,116] |
| Stress | BDNF, KITLG, NR3C1, FKBP5, MAD1L1, HEXCD | [117,118,119,120,121,122,123,124,125,126,127,128,129,130,131,132] |
| Depression | BDNF, SLC6A4, NR3C1 | [133,134,135,136,137,138,139,140,141,142,143,144,145,146,147] |
| In utero/Peri-conception (maternal exposures) | ||
| Cigarette smoking | AHRR, CYP1A1, MYO1G, GFI1 | [72,74,75,76,104,105,106,148,149] |
| Alcohol (in context of Fetal Alcohol Syndrome) | HLA-DPB1, FAM59B, CAPN10, DES, SLC6A3, SLC38A2, FAM24A, H19, TGFB1I1, PCDHB18, PCDHGA | [150,151] |
| BMI | CDHR3, ACTL10/NECAB3, POM121L1P, VIPR2, AGRN, GGTLC1 | [152] |
| Air pollution | COLEC11 | [153] |
| Stress | NR3C1 | [154,155] |
| Antidepressant use | ZNF575 | [156] |
| Interpartner violence | NR3C1 | [157,158] |
| Famine | INSR | [159,160] |
| Folic acid | ZFP57 | [161] |
| Pre-conception (paternal exposure) | ||
| Cigarette smoking | MAPK8IP3, TKR (in paternal sperm) | [97] |
© 2020 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Park, H.L. Epigenetic Biomarkers for Environmental Exposures and Personalized Breast Cancer Prevention. Int. J. Environ. Res. Public Health 2020, 17, 1181. https://doi.org/10.3390/ijerph17041181
Park HL. Epigenetic Biomarkers for Environmental Exposures and Personalized Breast Cancer Prevention. International Journal of Environmental Research and Public Health. 2020; 17(4):1181. https://doi.org/10.3390/ijerph17041181
Chicago/Turabian StylePark, Hannah Lui. 2020. "Epigenetic Biomarkers for Environmental Exposures and Personalized Breast Cancer Prevention" International Journal of Environmental Research and Public Health 17, no. 4: 1181. https://doi.org/10.3390/ijerph17041181
APA StylePark, H. L. (2020). Epigenetic Biomarkers for Environmental Exposures and Personalized Breast Cancer Prevention. International Journal of Environmental Research and Public Health, 17(4), 1181. https://doi.org/10.3390/ijerph17041181





