Wastewater-Based Epidemiology (WBE) and Viral Detection in Polluted Surface Water: A Valuable Tool for COVID-19 Surveillance—A Brief Review
Abstract
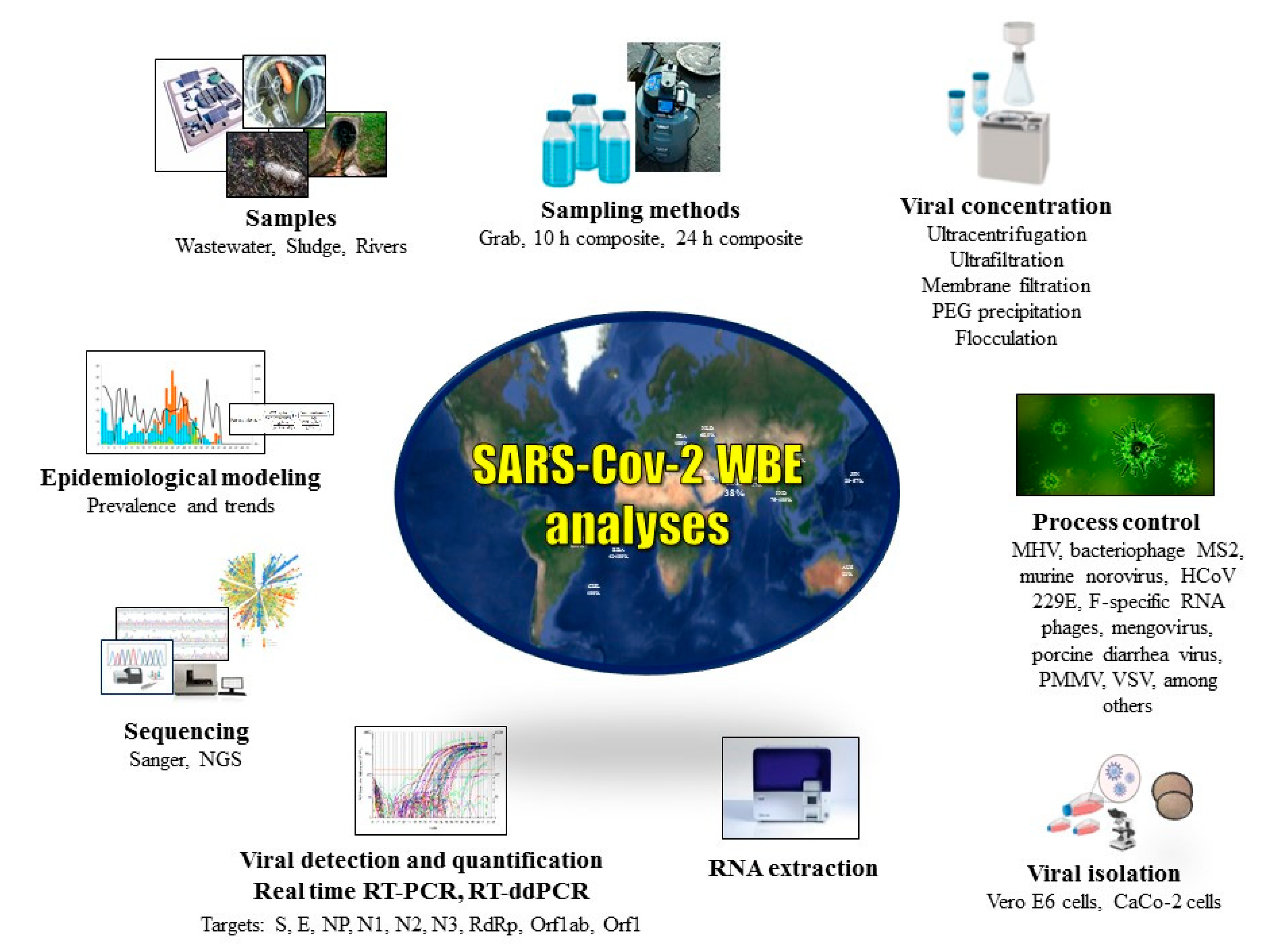
1. Introduction
2. Global Experience in SARS-CoV-2 WBE Surveillance
2.1. WBE in the Americas
2.2. WBE in Europe
2.3. WBE in Asia
2.4. WBE in Oceania and Middle-East
3. SARS-CoV-2 RNA Detection in Surface Water
4. WBE as a Surveillance Tool: Advantages and Challenges
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Disclaimer
References
- Wu, F.; Zhao, S.; Yu, B.; Chen, Y.-M.; Wang, W.; Song, Z.-G.; Hu, Y.; Tao, Z.-W.; Tian, J.-H.; Pei, Y.-Y.; et al. A new coronavirus associated with human respiratory disease in China. Nature 2020, 579, 265–269. [Google Scholar] [CrossRef] [PubMed]
- Zhou, P.; Yang, X.-L.; Wang, X.-G.; Hu, B.; Zhang, L.; Zhang, W.; Si, H.-R.; Zhu, Y.; Li, B.; Huang, C.-L.; et al. A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature 2020, 579, 270–273. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. WHO Director-General’s Opening Remarks at the Media Briefing on COVID-19. Available online: https://www.who.int/dg/speeches/detail/who-director-general-s-opening-remarks-at-the-media-briefing-on-covid-19---11-march-2020 (accessed on 3 July 2020).
- World Health Organization. Coronavirus Disease 2019 (COVID-19)—Situation Report 190. Available online: https://www.who.int/docs/default-source/coronaviruse/situation-reports/20200728-covid-19-sitrep-190.pdf?sfvrsn=fec17314_2 (accessed on 29 July 2020).
- Di Gennaro, F.; Pizzol, D.; Marotta, C.; Antunes, M.; Racalbuto, V.; Veronese, N.; Smith, L. Coronavirus Diseases (COVID-19) Current Status and Future Perspectives: A Narrative Review. Int. J. Env. Res. Public Health 2020, 17, 2690. [Google Scholar] [CrossRef] [PubMed]
- Ge, H.; Wang, X.; Yuan, X.; Xiao, G.; Wang, C.; Deng, T.; Yuan, Q.; Xiao, X. The epidemiology and clinical information about COVID-19. Eur. J. Clin. Microbiol. Infect. Dis. 2020, 39, 1–9. [Google Scholar] [CrossRef]
- Agarwal, A.; Chen, A.; Ravindran, N.; To, C.; Thuluvath, P.J. Gastrointestinal and Liver Manifestations of COVID-19. J. Clin. Exp. Hepatol. 2020, 10, 263–265. [Google Scholar] [CrossRef] [PubMed]
- Cholankeril, G.; Podboy, A.; Aivaliotis, V.I.; Tarlow, B.; Pham, E.A.; Spencer, S.; Kim, D.; Hsing, A.; Ahmed, A. High Prevalence of Concurrent Gastrointestinal Manifestations in Patients with SARS-CoV-2: Early Experience from California. Gastroenterology 2020, 159, 775–777. [Google Scholar] [CrossRef]
- Tian, Y.; Rong, L.; Nian, W.; He, Y. Review article: Gastrointestinal features in COVID-19 and the possibility of faecal transmission. Aliment. Pharm. 2020, 51, 843–851. [Google Scholar] [CrossRef]
- Yan, R.; Zhang, Y.; Li, Y.; Xia, L.; Guo, Y.; Zhou, Q. Structural basis for the recognition of SARS-CoV-2 by full-length human ACE2. Science 2020, 367, 1444–1448. [Google Scholar] [CrossRef]
- D’Amico, F.; Baumgart, D.C.; Danese, S.; Peyrin-Biroulet, L. Diarrhea During COVID-19 Infection: Pathogenesis, Epidemiology, Prevention, and Management. Clin. Gastroenterol. Hepatol. 2020, 18, 1663–1672. [Google Scholar] [CrossRef]
- Lamers, M.M.; Beumer, J.; van der Vaart, J.; Knoops, K.; Puschhof, J.; Breugem, T.I.; Ravelli, R.B.G.; Paul van Schayck, J.; Mykytyn, A.Z.; Duimel, H.Q.; et al. SARS-CoV-2 productively infects human gut enterocytes. Science 2020, 369, 50–54. [Google Scholar] [CrossRef]
- Xiao, F.; Tang, M.; Zheng, X.; Liu, Y.; Li, X.; Shan, H. Evidence for Gastrointestinal Infection of SARS-CoV-2. Gastroenterology 2020, 158, 1831–1833.e3. [Google Scholar] [CrossRef]
- Lin, L.; Jiang, X.; Zhang, Z.; Huang, S.; Zhang, Z.; Fang, Z.; Gu, Z.; Gao, L.; Shi, H.; Mai, L.; et al. Gastrointestinal symptoms of 95 cases with SARS-CoV-2 infection. Gut 2020, 69, 997–1001. [Google Scholar] [CrossRef] [PubMed]
- Park, S.-K.; Lee, C.-W.; Park, D.-I.; Woo, H.-Y.; Cheong, H.S.; Shin, H.C.; Ahn, K.; Kwon, M.-J.; Joo, E.-J. Detection of SARS-CoV-2 in Fecal Samples from Patients with Asymptomatic and Mild COVID-19 in Korea. Clin. Gastroenterol. Hepatol. 2020. [Google Scholar] [CrossRef] [PubMed]
- Xiao, F.; Sun, J.; Xu, Y.; Li, F.; Huang, X.; Li, H.; Zhao, J.; Huang, J.; Zhao, J. Infectious SARS-CoV-2 in Feces of Patient with Severe COVID-19. Emerg. Infect. Dis. 2020, 26, 1920–1922. [Google Scholar] [CrossRef] [PubMed]
- Xing, Y.-H.; Ni, W.; Wu, Q.; Li, W.-J.; Li, G.-J.; Wang, W.-D.; Tong, J.-N.; Song, X.-F.; Wing-Kin Wong, G.; Xing, Q.-S. Prolonged viral shedding in feces of pediatric patients with coronavirus disease 2019. J. Microbiol. Immunol. Infect. 2020, 53, 473–480. [Google Scholar] [CrossRef]
- Wang, W.; Xu, Y.; Gao, R.; Lu, R.; Han, K.; Wu, G.; Tan, W. Detection of SARS-CoV-2 in Different Types of Clinical Specimens. JAMA 2020, 323, 1843–1844. [Google Scholar] [CrossRef]
- Chen, C.; Gao, G.; Xu, Y.; Pu, L.; Wang, Q.; Wang, L.; Wang, W.; Song, Y.; Chen, M.; Wang, L.; et al. SARS-CoV-2-Positive Sputum and Feces After Conversion of Pharyngeal Samples in Patients With COVID-19. Ann. Intern. Med. 2020, 172, 832–834. [Google Scholar] [CrossRef] [PubMed]
- Lescure, F.-X.; Bouadma, L.; Nguyen, D.; Parisey, M.; Wicky, P.-H.; Behillil, S.; Gaymard, A.; Bouscambert-Duchamp, M.; Donati, F.; Le Hingrat, Q.; et al. Clinical and virological data of the first cases of COVID-19 in Europe: A case series. Lancet Infect. Dis. 2020, 20, 697–706. [Google Scholar] [CrossRef]
- Wölfel, R.; Corman, V.M.; Guggemos, W.; Seilmaier, M.; Zange, S.; Müller, M.A.; Niemeyer, D.; Jones, T.C.; Vollmar, P.; Rothe, C.; et al. Virological assessment of hospitalized patients with COVID-2019. Nature 2020, 581, 465–469. [Google Scholar] [CrossRef]
- Zheng, S.; Fan, J.; Yu, F.; Feng, B.; Lou, B.; Zou, Q.; Xie, G.; Lin, S.; Wang, R.; Yang, X.; et al. Viral load dynamics and disease severity in patients infected with SARS-CoV-2 in Zhejiang province, China, January-March 2020: Retrospective cohort study. BMJ 2020, 369, m1443. [Google Scholar] [CrossRef]
- Wu, Y.; Guo, C.; Tang, L.; Hong, Z.; Zhou, J.; Dong, X.; Yin, H.; Xiao, Q.; Tang, Y.; Qu, X.; et al. Prolonged presence of SARS-CoV-2 viral RNA in faecal samples. Lancet Gastroenterol. Hepatol. 2020, 5, 434–435. [Google Scholar] [CrossRef]
- Xu, Y.; Li, X.; Zhu, B.; Liang, H.; Fang, C.; Gong, Y.; Guo, Q.; Sun, X.; Zhao, D.; Shen, J.; et al. Characteristics of pediatric SARS-CoV-2 infection and potential evidence for persistent fecal viral shedding. Nat. Med. 2020, 26, 502–505. [Google Scholar] [CrossRef]
- Ianiro, G.; Mullish, B.H.; Kelly, C.R.; Sokol, H.; Kassam, Z.; Ng, S.; Fischer, M.; Allegretti, J.R.; Masucci, L.; Zhang, F.; et al. Screening of faecal microbiota transplant donors during the COVID-19 outbreak: Suggestions for urgent updates from an international expert panel. Lancet Gastroenterol. Hepatol. 2020, 5, 430–432. [Google Scholar] [CrossRef]
- Zhang, Y.; Chen, C.; Zhu, S.; Shu, C.; Wang, D.; Song, J.; Song, Y.; Zhen, W.; Feng, Z.; Wu, G.; et al. Isolation of 2019-nCoV from a Stool Specimen of a Laboratory-Confirmed Case of the Coronavirus Disease 2019 (COVID-19). China Cdc Wkly. 2020, 2, 123–124. [Google Scholar] [CrossRef]
- Heller, L.; Mota, C.R.; Greco, D.B. COVID-19 faecal-oral transmission: Are we asking the right questions? Sci. Total Environ. 2020, 729, 138919. [Google Scholar] [CrossRef]
- Kim, J.-M.; Kim, H.M.; Lee, E.J.; Jo, H.J.; Yoon, Y.; Lee, N.-J.; Son, J.; Lee, Y.-J.; Kim, M.S.; Lee, Y.-P.; et al. Detection and Isolation of SARS-CoV-2 in Serum, Urine, and Stool Specimens of COVID-19 Patients from the Republic of Korea. Osong Public Health Res. Perspect. 2020, 11, 112–117. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. United Nations World Water Development Report 2017. Available online: https://www.unwater.org/publications/world-water-development-report-2017 (accessed on 26 July 2020).
- La Rosa, G.; Libera, S.D.; Iaconelli, M.; Ciccaglione, A.R.; Bruni, R.; Taffon, S.; Equestre, M.; Alfonsi, V.; Rizzo, C.; Tosti, M.E.; et al. Surveillance of hepatitis A virus in urban sewages and comparison with cases notified in the course of an outbreak, Italy 2013. BMC Infect. Dis. 2014, 14, 419. [Google Scholar] [CrossRef]
- Prevost, B.; Lucas, F.S.; Goncalves, A.; Richard, F.; Moulin, L.; Wurtzer, S. Large scale survey of enteric viruses in river and waste water underlines the health status of the local population. Environ. Int. 2015, 79, 42–50. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Guidelines for Environmental Surveillance of Poliovirus Circulation. Available online: https://apps.who.int/iris/handle/10665/67854 (accessed on 5 May 2020).
- World Health Organization. Polio Surveillance: Tracking Progress towards Eradication Worldwide. 2013–2014. Available online: https://www.who.int/wer/2015/wer9021/en/ (accessed on 3 July 2020).
- Medema, G.; Been, F.; Heijnen, L.; Petterson, S. Implementation of environmental surveillance for SARS-CoV-2 virus to support public health decisions: Opportunities and challenges. Curr. Opin. Environ. Sci. Health 2020, S2468584420300635. [Google Scholar] [CrossRef]
- World Health Organization. Status of Environmental Surveillance for SARS-CoV-2 Virus: Scientific Brief, 5 August 2020. Available online: https://apps.who.int/iris/handle/10665/333670 (accessed on 10 October 2020).
- Secretaria Estadual de Saúde do Rio Grande do Sul. Centro Estadual de Vigilância em Saúde do Estado do Rio Grande do Sul Monitoramento ambiental de SARS-CoV-2. Boletim de acompanhamento no 2 2020. Available online: https://cevs-admin.rs.gov.br/upload/arquivos/202009/21115806-boletim-informativo-n-2-final.pdf (accessed on 20 September 2020).
- Secretaria Municipal de Saúde de Niterói, Acompanhamento dos Casos de Coronavirus (COVID19) Niterói—Painel de Análise Evolução Covid-19. Available online: https://geoniteroi.maps.arcgis.com/apps/MapSeries/index.html?appid=1d5a2d1006094558a58be3db42c54759 (accessed on 14 September 2020).
- Agência Nacional de Águas; Instituto Nacional de Ciência e Tecnologia em Estações Sustentáveis de Tratamento de Esgoto; Companhia de Saneamento de Minas Gerais (Copasa); Instituto Mineiro de Gestão das Águas (Igam); Secretaria de Estado de Saúde de Minas Gerais Detecção e quantificação do novo coronavírus em amostras de esgoto nas cidades de Belo Horizonte e Contagem—Monitoramento COVID Esgotos. Boletim de Acompanhamento No. 12 2020. Available online: https://www.ana.gov.br/panorama-das-aguas/qualidade-da-agua/boletins-monitoramento-covid-esgotos/boletim-de-acompanhamento-no-122020.pdf (accessed on 23 September 2020).
- Tlhagale, M.; Bhagwan, J.; Jonas, K. The Water Research Comission Launch a Programme to Monitor the Spread of COVID-19 in Communities Using a Water and Sanitation Based Approach. Available online: http://www.waterjpi.eu/images/newsletter/wrc-covid-19-surveillance-programme-article-final-2-june-2020.pdf (accessed on 12 October 2020).
- Prado, A.; Fumian, T.; Mannarino, C.F.; Maranhão, A.G.; Siqueira, M.M.; Miagostovich, M.P. Preliminary results of SARS-CoV-2 detection in sewerage system in Niterói municipality, Rio de Janeiro, Brazil. Memórias do Instituto Oswaldo Cruz 2020, 115, e200196. [Google Scholar] [CrossRef]
- Fongaro, G.; Hermes Stoco, P.; Sobral Marques Souza, D.; Grisard, E.C.; Magri, M.E.; Rogovski, P.; Schorner, M.A.; Hartmann Barazzetti, F.; Christoff, A.P.; de Oliveira, L.F.V.; et al. SARS-CoV-2 in human sewage in Santa Catalina, Brazil, November 2019. medRxiv 2020. [Google Scholar] [CrossRef]
- Conselho Nacional de Secretários de Saúde Painel CONASS—COVID-19. Available online: https://www.conass.org.br/painelconasscovid19/ (accessed on 16 September 2020).
- BRASIL. Ministério da Saúde Boletim Epidemiologico N.30—Doença Pelo Coronavirus. Available online: https://www.saude.gov.br/images/pdf/2020/September/09/Boletim-epidemiologico-COVID-30.pdf (accessed on 16 September 2020).
- Wu, F.; Xiao, N.; Zhang, J.; Gu, X.; Lee, W.L.; Kauffman, K.; Hanage, W.; Matus, M.; Ghaeli, N.; Endo, N.; et al. SARS-CoV-2 titers in wastewater are higher than expected from clinically confirmed cases. mSystems 2020, 5. [Google Scholar] [CrossRef]
- Wu, F.; Xiao, A.; Zhang, J.; Moniz, K.; Endo, N.; Armas, F.; Bonneau, R.; Brown, M.A.; Bushman, M.; Chai, P.R.; et al. SARS-CoV-2 titers in wastewater foreshadow dynamics and clinical presentation of new COVID-19 cases. medRxiv 2020. [Google Scholar] [CrossRef]
- Green, H.; Wilder, M.; Middleton, F.A.; Collins, M.; Fenty, A.; Gentile, K.; Kmush, B.; Zeng, T.; Larsen, D.A. Quantification of SARS-CoV-2 and cross-assembly phage (crAssphage) from wastewater to monitor coronavirus transmission within communities. medRxiv 2020. [Google Scholar] [CrossRef]
- Peccia, J.; Zulli, A.; Brackney, D.E.; Grubaugh, N.D.; Kaplan, E.H.; Casanovas-Massana, A.; Ko, A.I.; Malik, A.A.; Wang, D.; Wang, M.; et al. SARS-CoV-2 RNA concentrations in primary municipal sewage sludge as a leading indicator of COVID-19 outbreak dynamics. medRxiv 2020. [Google Scholar] [CrossRef]
- Nemudryi, A.; Nemudraia, A.; Wiegand, T.; Surya, K.; Buyukyoruk, M.; Cicha, C.; Vanderwood, K.K.; Wilkinson, R.; Wiedenheft, B. Temporal Detection and Phylogenetic Assessment of SARS-CoV-2 in Municipal Wastewater. Cell Rep. Med. 2020, 1, 100098. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez, R.; Curtis, K.; Bivins, A.; Bibby, K.; Weir, M.H.; Yetka, K.; Thompson, H.; Keeling, D.; Mitchell, J.; Gonzalez, D. COVID-19 surveillance in Southeastern Virginia using wastewater-based epidemiology. Water Res. 2020, 186, 116296. [Google Scholar] [CrossRef] [PubMed]
- Sherchan, S.P.; Shahin, S.; Ward, L.M.; Tandukar, S.; Aw, T.G.; Schmitz, B.; Ahmed, W.; Kitajima, M. First detection of SARS-CoV-2 RNA in wastewater in North America: A study in Louisiana, USA. Sci. Total Environ. 2020, 743, 140621. [Google Scholar] [CrossRef]
- Ampuero, M.; Valenzuela, S.; Valiente-Echeverria, F.; Soto-Rifo, R.; Barriga, G.P.; Chnaiderman, J.; Rojas, C.; Guajardo-Leiva, S.; Diez, B.; Gaggero, A. SARS-CoV-2 Detection in Sewage in Santiago, Chile—Preliminary results. medRxiv 2020. [Google Scholar] [CrossRef]
- D’Aoust, P.M.; Mercier, E.; Montpetit, D.; Jia, J.-J.; Alexandrov, I.; Neault, N.; Baig, A.T.; Mayne, J.; Zhang, X.; Alain, T.; et al. Quantitative analysis of SARS-CoV-2 RNA from wastewater solids in communities with low COVID-19 incidence and prevalence. medRxiv 2020. [Google Scholar] [CrossRef]
- Medema, G.; Heijnen, L.; Elsinga, G.; Italiaander, R.; Brouwer, A. Presence of SARS-Coronavirus-2 in sewage. medRxiv 2020. [Google Scholar] [CrossRef]
- Medema, G.; Heijnen, L.; Elsinga, G.; Italiaander, R.; Brouwer, A. Presence of SARS-Coronavirus-2 RNA in Sewage and Correlation with Reported COVID-19 Prevalence in the Early Stage of the Epidemic in The Netherlands. Environ. Sci. Technol. Lett. 2020, 7, 511–516. [Google Scholar] [CrossRef]
- Randazzo, W.; Cuevas-Ferrando, E.; Sanjuán, R.; Domingo-Calap, P.; Sánchez, G. Metropolitan wastewater analysis for COVID-19 epidemiological surveillance. Int. J. Hyg. Environ. Health 2020, 230, 113621. [Google Scholar] [CrossRef] [PubMed]
- Randazzo, W.; Truchado, P.; Cuevas-Ferrando, E.; Simón, P.; Allende, A.; Sánchez, G. SARS-CoV-2 RNA in wastewater anticipated COVID-19 occurrence in a low prevalence area. Water Res. 2020, 181, 115942. [Google Scholar] [CrossRef] [PubMed]
- Chavarria-Miró, G.; Anfruns-Estrada, E.; Guix, S.; Paraira, M.; Galofré, B.; Sáanchez, G.; Pintó, R.; Bosch, A. Sentinel surveillance of SARS-CoV-2 in wastewater anticipates the occurrence of COVID-19 cases. medRxiv 2020. [Google Scholar] [CrossRef]
- Balboa, S.; Mauricio-Iglesias, M.; Rodríguez, S.; Martínez-Lamas, L.; Vasallo, F.J.; Regueiro, B.; Lema, J.M. The fate of SARS-CoV-2 in wastewater treatment plants points out the sludge line as a suitable spot for incidence monitoring. medRxiv 2020. [Google Scholar] [CrossRef]
- Wurtzer, S.; Marechal, V.; Mouchel, J.-M.; Moulin, L. Time course quantitative detection of SARS-CoV-2 in Parisian wastewaters correlates with COVID-19 confirmed cases. medRxiv 2020. [Google Scholar] [CrossRef]
- Wurtzer, S.; Marechal, V.; Mouchel, J.-M.; Maday, Y.; Teyssou, R.; Richard, E.; Almayrac, J.L.; Moulin, L. Evaluation of lockdown impact on SARS-CoV-2 dynamics through viral genome quantification in Paris wastewaters. medRxiv 2020. [Google Scholar] [CrossRef]
- Trottier, J.; Darques, R.; Ait Mouheb, N.; Partiot, E.; Bakhache, W.; Deffieu, M.S.; Gaudin, R. Post-lockdown detection of SARS-CoV-2 RNA in the wastewater of Montpellier, France. One Health 2020, 10, 100157. [Google Scholar] [CrossRef]
- La Rosa, G.; Iaconelli, M.; Mancini, P.; Bonanno Ferraro, G.; Veneri, C.; Bonadonna, L.; Lucentini, L.; Suffredini, E. First detection of SARS-CoV-2 in untreated wastewaters in Italy. Sci. Total Environ. 2020, 736, 139652. [Google Scholar] [CrossRef]
- La Rosa, G.; Mancini, P.; Bonanno Ferraro, G.; Veneri, C.; Iaconelli, M.; Bonadonna, L.; Lucentini, L.; Suffredini, E. SARS-CoV-2 has been circulating in northern Italy since December 2019: Evidence from environmental monitoring. Sci. Total Environ. 2020, 750, 141711. [Google Scholar] [CrossRef] [PubMed]
- Zehender, G.; Lai, A.; Bergna, A.; Meroni, L.; Riva, A.; Balotta, C.; Tarkowski, M.; Gabrieli, A.; Bernacchia, D.; Rusconi, S.; et al. Genomic characterization and phylogenetic analysis of SARS-COV-2 in Italy. J. Med. Virol. 2020, 92, 1637–1640. [Google Scholar] [CrossRef] [PubMed]
- Westhaus, S.; Weber, F.-A.; Schiwy, S.; Linnemann, V.; Brinkmann, M.; Widera, M.; Greve, C.; Janke, A.; Hollert, H.; Wintgens, T.; et al. Detection of SARS-CoV-2 in raw and treated wastewater in Germany—Suitability for COVID-19 surveillance and potential transmission risks. Sci. Total Environ. 2020, 751, 141750. [Google Scholar] [CrossRef]
- Alpaslan Kocamemi, B.; Kurt, H.; Sait, A.; Sarac, F.; Saatci, A.M.; Pakdemirli, B. SARS-CoV-2 Detection in Istanbul Wastewater Treatment Plant Sludges. medRxiv 2020. [Google Scholar] [CrossRef]
- Hata, A.; Honda, R.; Hara-Yamamura, H.; Meuchi, Y. Detection of SARS-CoV-2 in wastewater in Japan by multiple molecular assays-implication for wastewater-based epidemiology (WBE). medRxiv 2020. [Google Scholar] [CrossRef]
- Haramoto, E.; Malla, B.; Thakali, O.; Kitajima, M. First environmental surveillance for the presence of SARS-CoV-2 RNA in wastewater and river water in Japan. Sci. Total Environ. 2020, 737, 140405. [Google Scholar] [CrossRef]
- Zhang, D.; Yang, Y.; Huang, X.; Jiang, J.; Li, M.; Zhang, X.; Ling, H.; Li, J.; Liu, Y.; Li, G.; et al. SARS-CoV-2 spillover into hospital outdoor environments. medRxiv 2020. [Google Scholar] [CrossRef]
- Wang, J.; Feng, H.; Zhang, S.; Ni, Z.; Ni, L.; Chen, Y.; Zhuo, L.; Zhong, Z.; Qu, T. SARS-CoV-2 RNA detection of hospital isolation wards hygiene monitoring during the Coronavirus Disease 2019 outbreak in a Chinese hospital. Int. J. Infect. Dis. 2020, 94, 103–106. [Google Scholar] [CrossRef]
- Zhang, D.; Ling, H.; Huang, X.; Li, J.; Li, W.; Yi, C.; Zhang, T.; Jiang, Y.; He, Y.; Deng, S.; et al. Potential spreading risks and disinfection challenges of medical wastewater by the presence of Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) viral RNA in septic tanks of fangcang hospital. medRxiv 2020. [Google Scholar] [CrossRef]
- Arora, S.; Nag, A.; Sethi, J.; Rajvanshi, J.; Saxena, S.; Shrivastava, S.K.; Gupta, A.B. Sewage surveillance for the presence of SARS-CoV-2 genome as a useful wastewater based epidemiology (WBE) tracking tool in India. medRxiv 2020. [Google Scholar] [CrossRef]
- Kumar, M.; Patel, A.K.; Shah, A.V.; Raval, J.; Rajpara, N.; Joshi, M.; Joshi, C.G. First proof of the capability of wastewater surveillance for COVID-19 in India through detection of genetic material of SARS-CoV-2. Sci. Total Environ. 2020, 746, 141326. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, W.; Angel, N.; Edson, J.; Bibby, K.; Bivins, A.; O’Brien, J.W.; Choi, P.M.; Kitajima, M.; Simpson, S.L.; Li, J.; et al. First confirmed detection of SARS-CoV-2 in untreated wastewater in Australia: A proof of concept for the wastewater surveillance of COVID-19 in the community. Sci. Total Environ. 2020, 728, 138764. [Google Scholar] [CrossRef] [PubMed]
- Bar Or, I.; Yaniv, K.; Shagan, M.; Ozer, E.; Erster, O.; Mendelson, E.; Mannasse, B.; Shirazi, R.; Kramarsky-Winter, E.; Nir, O.; et al. Regressing SARS-CoV-2 sewage measurements onto COVID-19 burden in the population: A proof-of-concept for quantitative environmental surveillance. medRxiv 2020. [Google Scholar] [CrossRef] [PubMed]
- Elsamadony, M.; Fujii, M.; Miura, T.; Watanabe, T. Possible transmission of viruses from contaminated human feces and sewage: Implications for SARS-CoV-2. Sci. Total Environ. 2020, 755, 142575. [Google Scholar] [CrossRef] [PubMed]
- Sunkari, E.D.; Korboe, H.M.; Abu, M.; Kizildeniz, T. Sources and routes of SARS-CoV-2 transmission in water systems in Africa: Are there any sustainable remedies? Sci. Total Environ. 2020, 753, 142298. [Google Scholar] [CrossRef]
- Adelodun, B.; Ajibade, F.O.; Ibrahim, R.G.; Bakare, H.O.; Choi, K.-S. Snowballing transmission of COVID-19 (SARS-CoV-2) through wastewater: Any sustainable preventive measures to curtail the scourge in low-income countries? Sci. Total Environ. 2020, 742, 140680. [Google Scholar] [CrossRef] [PubMed]
- Parikh, P.; Karim, Y.B.; Paulose, J.; Factor-Litvak, P.; Nix, E.; Aisyah, D.N.; Chaturvedi, H.; Manikam, L.; Lakhanpaul, M. COVID-19 and informal settlements—Implications for water, sanitation and health in India and Indonesia. Ucl Open Environ. 2020, 1, 1–5. [Google Scholar]
- IBGE. Síntese de Indicadores Sociais: Uma Análise das Condições de Vida da População Brasileira; IBGE: Rio de Janeiro, Brazil, 2018.
- Guerrero-Latorre, L.; Ballesteros, I.; Villacrés-Granda, I.; Granda, M.G.; Freire-Paspuel, B.; Ríos-Touma, B. SARS-CoV-2 in river water: Implications in low sanitation countries. Sci. Total Environ. 2020, 743, 140832. [Google Scholar] [CrossRef]
- Rimoldi, S.G.; Stefani, F.; Gigantiello, A.; Polesello, S.; Comandatore, F.; Mileto, D.; Maresca, M.; Longobardi, C.; Mancon, A.; Romeri, F.; et al. Presence and infectivity of SARS-CoV-2 virus in wastewaters and rivers. Sci. Total Environ. 2020, 744, 140911. [Google Scholar] [CrossRef]
- Street, R.; Malema, S.; Mahlangeni, N.; Mathee, A. Wastewater surveillance for Covid-19: An African perspective. Sci. Total Environ. 2020, 743, 140719. [Google Scholar] [CrossRef]
- Gwenzi, W. Leaving no stone unturned in light of the COVID-19 faecal-oral hypothesis? A water, sanitation and hygiene (WASH) perspective targeting low-income countries. Sci. Total Environ. 2020, 753, 141751. [Google Scholar] [CrossRef] [PubMed]
- Odih, E.E.; Afolayan, A.O.; Akintayo, I.; Okeke, I.N. Could Water and Sanitation Shortfalls Exacerbate SARS-CoV-2 Transmission Risks? Am. J. Trop. Med. Hyg. 2020, 103, 554–557. [Google Scholar] [CrossRef] [PubMed]
- Carducci, A.; Federigi, I.; Liu, D.; Thompson, J.R.; Verani, M. Making Waves: Coronavirus detection, presence and persistence in the water environment: State of the art and knowledge needs for public health. Water Res. 2020, 179, 115907. [Google Scholar] [CrossRef] [PubMed]
- Paleologos, E.K.; O’Kelly, B.C.; Tang, C.-S.; Cornell, K.; Rodríguez-Chueca, J.; Abuel-Naga, H.; Koda, E.; Farid, A.; Vaverková, M.D.; Kostarelos, K.; et al. Post Covid-19 water and waste water management to protect public health and geoenvironment. Environ. Geotech. 2020, 1–15. [Google Scholar] [CrossRef]
- Pandey, D.; Verma, S.; Verma, P.; Mahanty, B.; Dutta, K.; Daverey, A.; Arunachalam, K. SARS-CoV-2 in wastewater: Challenges for developing countries. Int. J. Hyg. Environ. Health 2020, 231, 113634. [Google Scholar] [CrossRef] [PubMed]
- Carraturo, F.; Del Giudice, C.; Morelli, M.; Cerullo, V.; Libralato, G.; Galdiero, E.; Guida, M. Persistence of SARS-CoV-2 in the environment and COVID-19 transmission risk from environmental matrices and surfaces. Environ. Pollut. 2020, 265, 115010. [Google Scholar] [CrossRef]
- Foladori, P.; Cutrupi, F.; Segata, N.; Manara, S.; Pinto, F.; Malpei, F.; Bruni, L.; La Rosa, G. SARS-CoV-2 from faeces to wastewater treatment: What do we know? A review. Sci. Total Environ. 2020, 743, 140444. [Google Scholar] [CrossRef]
- Sharif, S.; Ikram, A.; Khurshid, A.; Salman, M.; Mehmood, N.; Arshad, Y.; Ahmad, J.; Angez, M.; Alam, M.M.; Rehman, L.; et al. Detection of SARS-Coronavirus-2 in wastewater, using the existing environmental surveillance network: An epidemiological gateway to an early warning for COVID-19 in communities. medRxiv 2020. [Google Scholar] [CrossRef]
- Ahmed, F.; Islam, M.A.; Kumar, M.; Hossain, M.; Bhattacharya, P.; Islam, M.T.; Hossen, F.; Hossain, M.S.; Islam, M.S.; Uddin, M.M.; et al. First detection of SARS-CoV-2 genetic material in the vicinity of COVID-19 isolation centre through wastewater surveillance in Bangladesh. medRxiv 2020. [Google Scholar] [CrossRef]
- Lodder, W.; de Roda Husman, A.M. SARS-CoV-2 in wastewater: Potential health risk, but also data source. Lancet Gastroenterol. Hepatol. 2020, 5, 533–534. [Google Scholar] [CrossRef]
- Mlejnkova, H.; Sovova, K.; Vasickova, P.; Ocenaskova, V.; Jasikova, L.; Juranova, E. Preliminary Study of Sars-Cov-2 Occurrence in Wastewater in the Czech Republic. IJERPH 2020, 17, 5508. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. WHO Global Epidemiological Surveillance Standards for Influenza. Available online: https://www.who.int/influenza/resources/documents/influenza_surveillance_manual/en/ (accessed on 17 September 2020).
- Sims, N.; Kasprzyk-Hordern, B. Future perspectives of wastewater-based epidemiology: Monitoring infectious disease spread and resistance to the community level. Environ. Int. 2020, 139, 105689. [Google Scholar] [CrossRef] [PubMed]
- Mao, K.; Zhang, K.; Du, W.; Ali, W.; Feng, X.; Zhang, H. The potential of wastewater-based epidemiology as surveillance and early warning of infectious disease outbreaks. Curr. Opin. Environ. Sci. Health 2020, 17, 1–7. [Google Scholar] [CrossRef]
- Daughton, C.G. Wastewater surveillance for population-wide Covid-19: The present and future. Sci. Total Environ. 2020, 736, 139631. [Google Scholar] [CrossRef]
- Orive, G.; Lertxundi, U.; Barcelo, D. Early SARS-CoV-2 outbreak detection by sewage-based epidemiology. Sci. Total Environ. 2020, 732, 139298. [Google Scholar] [CrossRef]
- Daughton, C. The international imperative to rapidly and inexpensively monitor community-wide Covid-19 infection status and trends. Sci. Total Environ. 2020, 726, 138149. [Google Scholar] [CrossRef]
- Kitajima, M.; Ahmed, W.; Bibby, K.; Carducci, A.; Gerba, C.P.; Hamilton, K.A.; Haramoto, E.; Rose, J.B. SARS-CoV-2 in wastewater: State of the knowledge and research needs. Sci. Total Environ. 2020, 739, 139076. [Google Scholar] [CrossRef] [PubMed]
- Polo, D.; Quintela-Baluja, M.; Corbishley, A.; Jones, D.L.; Singer, A.C.; Graham, D.W.; Romalde, J.L. Making waves: Wastewater-based epidemiology for COVID-19 – approaches and challenges for surveillance and prediction. Water Res. 2020, 186, 116404. [Google Scholar] [CrossRef] [PubMed]
- Xagoraraki, I.; O’Brien, E. Wastewater-Based Epidemiology for Early Detection of Viral Outbreaks. In Women in Water Quality: Investigations by Prominent Female Engineers; O’Bannon, D.J., Ed.; Springer, International Publishing: Cham, Switzerland, 2020; pp. 75–97. [Google Scholar]
- Olds, H.T.; Corsi, S.R.; Dila, D.K.; Halmo, K.M.; Bootsma, M.J.; McLellan, S.L. High levels of sewage contamination released from urban areas after storm events: A quantitative survey with sewage specific bacterial indicators. Plos Med. 2018, 15. [Google Scholar] [CrossRef]
- Pereira, B.M.T.; Morales, W.; Cardoso, R.G.; Fiorelli, R.; Fraga, G.P.; Briggs, S.M. Lessons learned from a landslide catastrophe in Rio de Janeiro, Brazil. Am. J. Disaster Med. 2013, 8, 253–258. [Google Scholar] [CrossRef]
- Vikas. The Southwest Monsoon During COVID-19 Pandemic: A Potential Concern. Asia Pac. J. Public Health 2020, 32, 374–375. [Google Scholar] [CrossRef] [PubMed]
- Cashdollar, J.L.; Wymer, L. Methods for primary concentration of viruses from water samples: A review and meta-analysis of recent studies. J. Appl. Microbiol. 2013, 115, 1–11. [Google Scholar] [CrossRef]
- Haramoto, E.; Kitajima, M.; Hata, A.; Torrey, J.R.; Masago, Y.; Sano, D.; Katayama, H. A review on recent progress in the detection methods and prevalence of human enteric viruses in water. Water Res. 2018, 135, 168–186. [Google Scholar] [CrossRef] [PubMed]
- Bofill-Mas, S.; Rusiñol, M. Recent trends on methods for the concentration of viruses from water samples. Curr. Opin. Environ. Sci. Health 2020, 16, 7–13. [Google Scholar] [CrossRef]
- Bivins, A.; North, D.; Ahmad, A.; Ahmed, W.; Alm, E.; Been, F.; Bhattacharya, P.; Bijlsma, L.; Boehm, A.B.; Brown, J.; et al. Wastewater-Based Epidemiology: Global Collaborative to Maximize Contributions in the Fight Against COVID-19. Environ. Sci. Technol. 2020, 54, 7754–7757. [Google Scholar] [CrossRef]
- O’Reilly, K.M.; Allen, D.J.; Fine, P.; Asghar, H. The challenges of informative wastewater sampling for SARS-CoV-2 must be met: Lessons from polio eradication. Lancet Microbe 2020, 1, e189–e190. [Google Scholar] [CrossRef]
- Michael-Kordatou, I.; Karaolia, P.; Fatta-Kassinos, D. Sewage analysis as a tool for the COVID-19 pandemic response and management: The urgent need for optimised protocols for SARS-CoV-2 detection and quantification. J. Environ. Chem. Eng. 2020, 8, 104306. [Google Scholar] [CrossRef]
- Rusiñol, M.; Martínez-Puchol, S.; Forés, E.; Itarte, M.; Girones, R.; Bofill-Mas, S. Concentration methods for the quantification of coronavirus and other potentially pandemic enveloped virus from wastewater. Curr. Opin. Environ. Sci. Health 2020, 17, 21–28. [Google Scholar] [CrossRef]
- Ahmed, W.; Harwood, V.J.; Gyawali, P.; Sidhu, J.P.S.; Toze, S. Comparison of Concentration Methods for Quantitative Detection of Sewage-Associated Viral Markers in Environmental Waters. Appl. Environ. Microbiol. 2015, 81, 2042–2049. [Google Scholar] [CrossRef]
- World Health Organization. Leveraging GISRS for COVID-19 Surveillance. Available online: http://www.who.int/influenza/gisrs_laboratory/covid19/en/ (accessed on 16 September 2020).
- World Health Organization. Water, Sanitation, Hygiene, and Waste management for SARS-CoV-2, the Virus that Causes COVID-19. Available online: https://www.who.int/publications-detail-redirect/WHO-2019-nCoV-IPC-WASH-2020.4 (accessed on 19 August 2020).
- Han, J.; He, S. Urban flooding events pose risks of virus spread during the novel coronavirus (COVID-19) pandemic. Sci. Total Environ. 2020, 755, 142491. [Google Scholar] [CrossRef]
- Gardy, J.; Loman, N.J.; Rambaut, A. Real-time digital pathogen surveillance—The time is now. Genome Biol. 2015, 16, 155. [Google Scholar] [CrossRef] [PubMed]
| Country | Localities | Period | Type of Water Sample | Main Outcomes and Remarks | References |
|---|---|---|---|---|---|
| Brazil | Rio de Janeiro, Minas Gerais, Rio Grande do Sul, Santa Catarina | Oct. 2019–Aug, 2020 |
|
| [36,38,40,41] |
| Chile | Santiago | Mar–Jun, 2020 |
|
| [51] |
| Ecuador | Quito | Jun, 2020 |
|
| [81] |
| Canada | Ottawa and Gatineau | Apr–Jun, 2020 |
|
| [52] |
| USA | Connecticut, Massachusetts, Montana, New York, Virginia, Louisiana | Jan–Jul, 2020 |
|
| [44,45,47,48,49,50] |
| China | Wuhan | Feb–Apr, 2020 |
|
| [69,70,71] |
| India | Jaipur | May–Jun, 2020 |
|
| [72,73] |
| Japan | Ishikawa, Toyama and Yamanashi | Mar–May, 2020 |
|
| [67,68] |
| Pakistan | Islamabad | Mar–Apr, 2020 |
|
| [91] |
| Bangladesh | Noakhali | Jul–Aug, 2020 |
|
| [92] |
| Australia | Queensland | Mar–Apr, 2020 |
|
| [74] |
| France | Paris, Montpellier | Mar–Jul, 2020 |
|
| [59,60,61] |
| Italy | Milan, Monza, Rome, Turin/Piedmont and Bologna/Emilia Romagna | Feb–Apr, 2020 |
|
| [62,63,82] |
| Netherlands | Amsterdam, Hague, Utrecht, Apeldoorn, Tilburg and Schiphol airport | Feb–Mar, 2020 |
|
| [54,93] |
| Spain | Murcia, Totana, Lorca, Cartagena, Cieza, Molina de Segura, Valencia, Barcelona, Santiago de Compostela | Feb–May, 2020 |
|
| [55,56,57,58] |
| Germany | North Westphalia | 8 April, 2020 |
|
| [65] |
| Czech Republic | Different localities, not reported | Apr–Jun, 2020 |
|
| [94] |
| Turkey | Istanbul | 7 May, 2020 |
|
| [66] |
| Israel | Tel Aviv | NR |
|
| [75] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aguiar-Oliveira, M.d.L.; Campos, A.; R. Matos, A.; Rigotto, C.; Sotero-Martins, A.; Teixeira, P.F.P.; Siqueira, M.M. Wastewater-Based Epidemiology (WBE) and Viral Detection in Polluted Surface Water: A Valuable Tool for COVID-19 Surveillance—A Brief Review. Int. J. Environ. Res. Public Health 2020, 17, 9251. https://doi.org/10.3390/ijerph17249251
Aguiar-Oliveira MdL, Campos A, R. Matos A, Rigotto C, Sotero-Martins A, Teixeira PFP, Siqueira MM. Wastewater-Based Epidemiology (WBE) and Viral Detection in Polluted Surface Water: A Valuable Tool for COVID-19 Surveillance—A Brief Review. International Journal of Environmental Research and Public Health. 2020; 17(24):9251. https://doi.org/10.3390/ijerph17249251
Chicago/Turabian StyleAguiar-Oliveira, Maria de Lourdes, Aline Campos, Aline R. Matos, Caroline Rigotto, Adriana Sotero-Martins, Paulo F. P. Teixeira, and Marilda M. Siqueira. 2020. "Wastewater-Based Epidemiology (WBE) and Viral Detection in Polluted Surface Water: A Valuable Tool for COVID-19 Surveillance—A Brief Review" International Journal of Environmental Research and Public Health 17, no. 24: 9251. https://doi.org/10.3390/ijerph17249251
APA StyleAguiar-Oliveira, M. d. L., Campos, A., R. Matos, A., Rigotto, C., Sotero-Martins, A., Teixeira, P. F. P., & Siqueira, M. M. (2020). Wastewater-Based Epidemiology (WBE) and Viral Detection in Polluted Surface Water: A Valuable Tool for COVID-19 Surveillance—A Brief Review. International Journal of Environmental Research and Public Health, 17(24), 9251. https://doi.org/10.3390/ijerph17249251





