Slackline Training in Children with Spastic Cerebral Palsy: A Randomized Clinical Trial
Abstract
1. Introduction
2. Materials & Methods
2.1. Trial Design
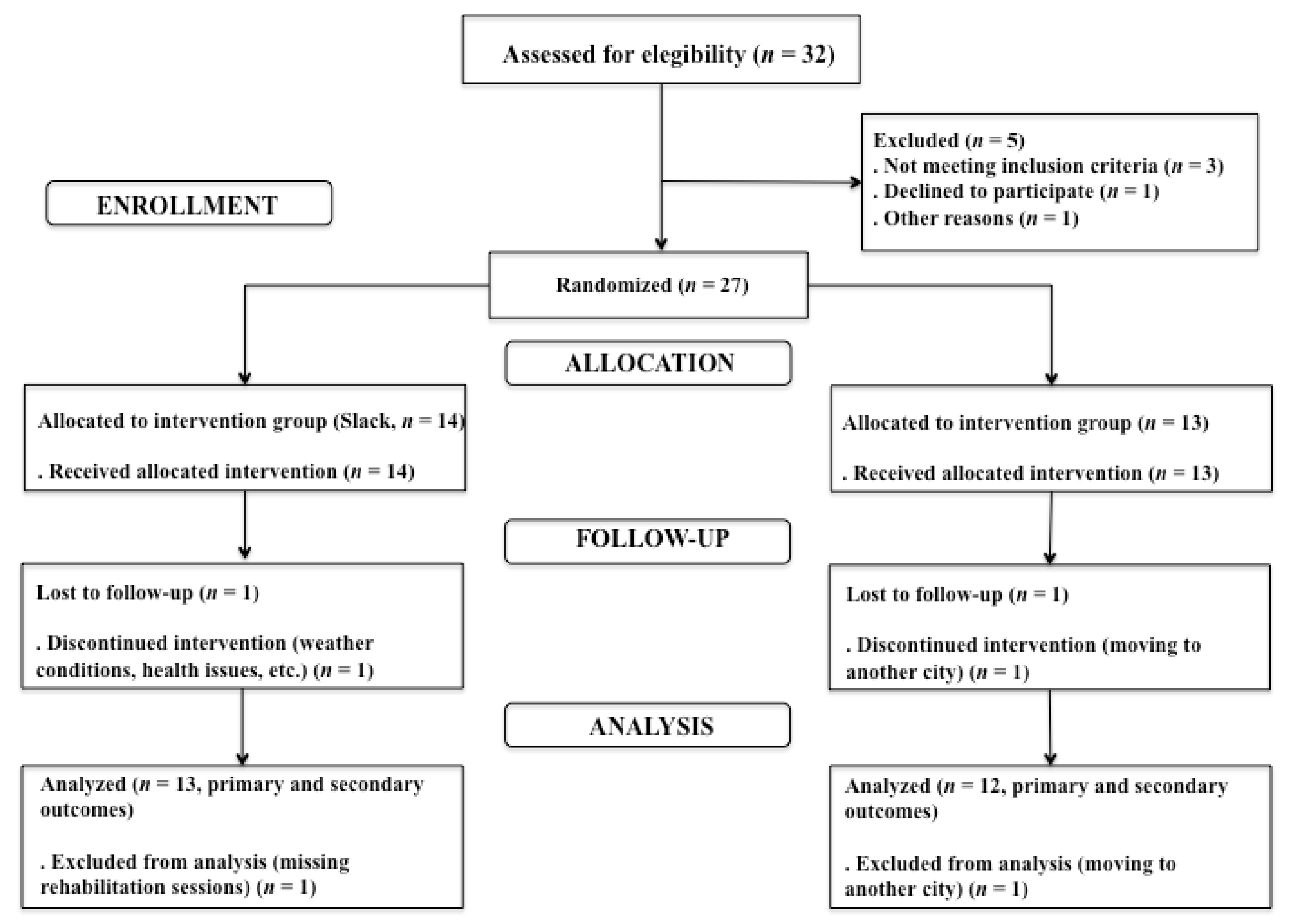
2.2. Participants
2.3. Intervention
2.4. Outcome Measures
2.4.1. Static Posturography
2.4.2. Myoelectrical Activity
2.4.3. Jump Performance
2.4.4. Perceived Exertion
2.5. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Data Availability
References
- Rosenbaum, P.; Paneth, N.; Leviton, A.; Goldstein, M.; Bax, M. A report: The definition and classification of cerebral palsy April 2006. Dev. Med. Child. Neurol. 2007, 49, 8–14. [Google Scholar]
- Bottos, M.; Gericke, C. Ambulatory capacity in cerebral palsy: Prognostic criteria and consequences for intervention. Dev. Med. Child. Neurol. 2003, 45, 786–790. [Google Scholar] [CrossRef]
- Eken, M.M.; Brændvik, S.M.; Bardal, E.M.; Houdijk, H.; Dallmeijer, A.J.; Roeleveld, K. Lower limb muscle fatigue during walking in children with cerebral palsy. Dev. Med. Child. Neurol. 2019, 61, 212–218. [Google Scholar] [CrossRef] [PubMed]
- Degelaen, M.; de Borre, L.; Kerckhofs, E.; de Meirleir, L.; Buyl, R.; Cheron, G.; Dan, B. Influence of botulinum toxin therapy on postural control and lower limb intersegmental coordination in children with spastic cerebral palsy. Toxins 2013, 5, 93–105. [Google Scholar] [CrossRef] [PubMed]
- Chaloupská, P.; Hrušová, D. Effect of Level of Coordination Abilities on Motor Learning Progress in Slacklining. In Proceedings of the 3rd International Conference on Health and Health Psychology. The European Proceedings of Social & Behavioural Sciences (EpSBS), Porto, Portugal, 5–7 July 2017. [Google Scholar]
- Pfusterschmied, J.; Stöggl, T.; Buchecker, M.; Lindinger, S.; Wagner, H.; Müller, E. Effects of 4-week slackline training on lower limb joint motion and muscle activation. J. Sci. Med. Sport 2013, 16, 562–566. [Google Scholar] [CrossRef] [PubMed]
- Santos, L.; Fernandez-Rio, J.; Fernández-García, B.; Jakobsen, M.D. The effects of supervised Slackline training on postural balance in judoists. Med. Sport 2014, 67, 539–553. [Google Scholar]
- Santos, L.; Fernández-Río, J.; Fernández-García, B.; Jakobsen, M.D.; González-Gómez, L.; Suman, O.E. Effects of slackline training on postural control, jump performance, and myoelectrical activity in female basketball players. J. Strength Cond. Res. 2016, 30, 653–664. [Google Scholar] [CrossRef]
- Jäger, T.; Kiefer, J.; Werner, I.; Federolf, P.A. Could Slackline Training Complement the FIFA 11+ Programme Regarding Training of Neuromuscular Control? Eur. J. Sport Sci. 2017, 17, 1021–1028. [Google Scholar] [CrossRef]
- Paoletti, P.; Mahadevan, L. Balancing on tightropes and slacklines. J. R. Soc. Interface 2012, 9, 2097–2108. [Google Scholar] [CrossRef][Green Version]
- Santos, L.; Fernandez-Rio, J.; Winge, K.; Barragán-Pérez, B.; Rodríguez-Pérez, V.; González-Díez, V.; Blanco-Traba, M.; Suman, O.E.; Gabel, C.P.; Rodríguez-Gómez, J. Effects of supervised slackline training on postural instability, freezing of gait, and falls efficacy in people with Parkinson’s disease. Disabil. Rehabil. 2017, 39, 1573–1580. [Google Scholar] [CrossRef]
- Palisano, R.; Rosenbaum, P.; Walter, S.; Russell, D.; Wood, E.; Galuppi, B. Development and reliability of a system to classify gross motor function in children with cerebral palsy. Dev. Med. Child. Neurol. 1997, 30, 214–223. [Google Scholar]
- Cohen, J. Statistical Power Analysis for the Behavioral Sciences; Lawrence Erlbaum Associates: Hillsdale, NJ, USA, 1988; pp. 8–14. [Google Scholar]
- Ruhe, A.; Fejer, R.; Walker, B. The test—Retest reliability of centre of pressure measures in bipedal static task conditions—A systematic review of the literature. Gait Posture 2010, 32, 436–445. [Google Scholar] [CrossRef] [PubMed]
- Sozzi, S.; Honeine, J.L.; Do, M.C.; Schieppatis, M. Leg muscle activity during tandem stance and the control of body balance in the frontal plane. Clin. Neurophysiol. 2013, 124, 1175–1186. [Google Scholar] [CrossRef] [PubMed]
- Hermens, H.J.; Frederiks, B. SENIAM: European Recommendations for Surface ElectromyoGraphy; Roessingh Research and Development: Enschede, The Netherlands, 1999. [Google Scholar]
- Bosco, C.; Luhtanen, P.; Komi, P. A Simple Method for Measurement of Mechanical Power in Jumping. Eur. J. Appl. Physiol. 1983, 50, 273–282. [Google Scholar] [CrossRef]
- Borg, G. Borg’s Perceived Exertion and Pain Scales; Human Kinetics: Champaign, IL, USA, 1998. [Google Scholar]
- Huber, P. Robust Statistics; Wiley: New York, NY, USA, 1981. [Google Scholar]
- Rosenthal, R. Meta-Analytic Procedures for Social Research; Sage: Newbury Park, CA, USA, 1991. [Google Scholar]
- Le Clair, K.; Riach, C. Postural stability measures: What to measure and for how long. Clin. Biomech. 1996, 11, 176–178. [Google Scholar] [CrossRef]
- Donath, L.; Roth, R.; Rueegge, A.; Groppa, M.; Zahner, L.; Faude, O. Effects of slackline training on balance, jump performance & muscle activity in young children. Int. J. Sports Med. 2013, 34, 1093–1098. [Google Scholar]
- Azzam, A.M.; Ebrahim, M.B.; El-aziz, A. Efficacy of Balance Training Program on Improvement of Knee Flexion Deformity in Spastic Diplegic Cerebral Palsied Children. Med. J. Cairo Univ. 2010, 78, 527–532. [Google Scholar]
- Abd El-Kafy, E.; El-Basatiny, H. Effect of postural balance training on gait parameters in children with cerebral palsy. Am. J. Phys. Med. Rehabil. 2014, 93, 938–947. [Google Scholar] [CrossRef]
- El-Shamy, S.M.; Abd El Kafy, E.M. Effect of balance training on postural balance control and risk of fall in children with diplegic cerebral palsy. Disabil. Rehabil. 2014, 36, 1176–1183. [Google Scholar] [CrossRef]
- Yun, C.-K.; Yoo, J.-N. The Effects of Visual Biofeedback Balance Training on Functional Ability in Children with Cerebral Palsy: A Pilot Study. J. Korean Soc. Phys. Med. 2016, 11, 133–139. [Google Scholar] [CrossRef][Green Version]
- Meyns, P.; Pans, L.; Plasmans, K.; Heyrman, L.; Desloovere, K.; Molenaers, G. The Effect of Additional Virtual Reality Training on Balance in Children with Cerebral Palsy after Lower Limb Surgery: A Feasibility Study. Games Health J. 2017, 6, 39–48. [Google Scholar] [CrossRef] [PubMed]
- Bonnechère, B.; Omelina, L.; Jansen, B.; Van Sint Jan, S. Balance improvement after physical therapy training using specially developed serious games for cerebral palsy children: Preliminary results. Disabil. Rehabil. 2017, 39, 403–406. [Google Scholar] [CrossRef] [PubMed]
- Paillard, T. Plasticity of the postural function to sport and/or motor experience. Neurosci. Biobehav. Rev. 2017, 72, 129–152. [Google Scholar] [CrossRef] [PubMed]
- Miller, M.; Thompson, S. Miller’s Review of Orthopaedics; Elsevier: Amsterdam, The Netherlands, 2016; pp. 468–475. [Google Scholar]
- Keller, M.; Pfusterschmied, J.; Buchecker, M.; Müller, E.; Taube, W. Improved postural control after slackline training is accompanied by reduced H-reflexes. Scand. J. Med. Sci. Sports 2012, 22, 471–477. [Google Scholar] [CrossRef] [PubMed]
- Donath, L.; Roth, R.; Zahner, L.; Faude, O. Slackline training and neuromuscular performance in seniors: A randomized controlled trial. Scand. J. Med. Sci. Sports 2016, 26, 275–283. [Google Scholar] [CrossRef] [PubMed]
- Benda, W.; McGibbon, N.; Grant, K. Improvements in Muscle Symmetry in Children with Cerebal Palsy after equine-assisted therapy (hippotherapy). J. Altern. Complementary Med. 2003, 9, 817–825. [Google Scholar] [CrossRef] [PubMed]
- Woollacott, M.; Shumway-Cook, A.; Hutchinson, S.; Ciol, M.; Price, R.; Kartin, D. Effect of balance training on muscle activity used in recovery of stability in children with cerebral palsy: A pilot study. Dev. Med. Child. Neurol. 2005, 47, 455–461. [Google Scholar] [CrossRef]
- Granacher, U.; Iten, N.; Roth, R.; Gollhofer, A. Slackline training for balance and strength promotion. Int. J. Sports Med. 2010, 31, 717–723. [Google Scholar] [CrossRef]
- Bobbert, M.F.; Van Soest, A.J. Effects of muscle strenghtening on vertical jump height: A simulation study. Med. Sci. Sports Exerc. 1994, 26, 1012–1020. [Google Scholar] [CrossRef]
- Russchen, H.; Slaman, J.; Stam, H.J.; van Markus-Doornbosch, F.; van den Berg-Emons, R.J.; Roebroeck, M.E. Focus on fatigue amongst young adults with spastic cerebral palsy. J. Neuroeng. Rehabil. 2014, 11, 161. [Google Scholar] [CrossRef]
| Weeks | Tasks Description |
|---|---|
| 1st | Task-1: Stand on the band using the right leg as support (with arms and the left leg free). The therapist supplies “major help”. Task-2: Repeat task 1 but using the left leg as support. Task-3: Tandem stance. Patients set the left leg as the rear one. The therapist supplies “major help”. Task-4: Repeat task 3 but using the right leg as the rear one. This set of 4 tasks should be carried out 4 times. |
| 2nd | Task-1: Stand on the band using the right leg as support (with arms and the left leg free). The therapist supplies “minor help”. Task-2: Repeat task 1 but using the left leg as support. Task-3: Tandem stance. Patients set the left leg as the rear one. The therapist supplies “minor help”. Task-4: Repeat task 3 but using the right leg as the rear one. This set of 4 tasks should be carried out 3 times. Task-5: Patients walk 4 forward steps. The therapist supplies “major help”. Task-6: Patients walk 6 forward steps. The therapist supplies “major help”. |
| 3rd | Task-1: Patients walk 4 forward steps. The therapist supplies “major help”. Task-2: Patients walk between 6 and 8 forward steps. The therapist supplies “major help”. Task-3: Patients walk 4 forward steps. The therapist supplies “minor help”. Task-4: Patients walk between 6 and 8 forward steps. The therapist supplies “minor help”. Task-5: Patients walk 4 forward steps. The therapist does not supply help. Task-6: Patients walk between 6 and 8 forward steps. The therapist does not supply help. This set of 6 tasks should be carried out twice. |
| 4th | Task-1: Patients walk 4 forward steps. The therapist supplies “minor help”. Task-2: Patients walk between 6 and 8 forward steps. The therapist supplies “minor help”. Task-3: Patients walk 4 forward steps. The therapist does not supply help. Task-4: Patients walk between 6 and 8 forward steps. The therapist does not supply help. Task-5: Patients walk 4 backward steps. The therapist supplies “major help”. Task-6: Patients walk between 4 and 6 backward steps. The therapist supplies “major help”. This set of 6 tasks should be carried out twice. |
| 5th | Task-1: Patients walk between 4 and 6 forward steps. The therapist does not supply help. Task-2: Patients walk 4 backward steps. The therapist supplies “major help”. Task-3: Patients walk between 4 and 6 backward steps. The therapist supplies “major help”. Task-4: Patients walk 4 backward steps. The therapist supplies “minor help”. Task-5: Patients walk between 4 and 6 backward steps. The therapist supplies “minor help”. Task-6: Patients walk 4 backward steps. The therapist does not supply help. This set of 6 tasks should be carried out twice. |
| 6th | Task-1: Patients walk 4 backward steps. The therapist supplies ‘minor help’. Task-2: Patients walk between 4 and 6 backward steps. The therapist supplies “minor help”. Task-3: Patients walk 4 backward steps. The therapist does not supply any help. Task-4: Patients walk between 4 and 6 backward steps. The therapist supplies “minor help”. Task-5: Patients make 2 90º turns towards right and left side while standing. The therapist supplies “major help”. Task-6: Patients make 2 90º turns towards right and left side while standing. The therapist supplies “minor help”. This set of 6 tasks should be carried out twice. |
| Characteristic | Intervention (n = 14) | Control (n = 13) | p-Value |
|---|---|---|---|
| Age (years) | 13 ± 2 | 12 ± 2 | 0.679 |
| Sex (male/female) | 7/7 | 8/5 | 0.688 |
| GMFCS (I/II) | 8/6 | 8/5 | 1.000 |
| SCP type (dip/hem) | 8/6 | 7/6 | 1.000 |
| BMI (kg.m−2) | 21.2 ± 4.2 | 19.5 ± 3.1 | 0.251 |
| Endpoints | Group | Baseline | Post-intervention | Group p-Value | Time p-Value | Interaction p-Value | Effect Size |
|---|---|---|---|---|---|---|---|
| Speed (mm/s) b | Intervention | 6.4 ± 3.6 | 4.1 ± 1.6 * | p = 0.041 * | p = 0.229 | p = 0.269 | r = 0.64 |
| Control | 7.5 ± 2.9 | 7.8 ± 2.9 | |||||
| Xspeed (mm/s) b | Intervention | 6.2 ± 3.07 | 2.1 ± 1.9 * | p < 0.000 * | p = 0.055 * | p = 0.006 * | r = 0.71 |
| Control | 7.04 ± 3.1 | 7.6 ± 3.2 | |||||
| Yspeed (mm/s) b | Intervention | 7.2 ± 3.5 | 4.1 ± 2.8 | p = 0.062 | p = 0.345 | p = 0.077 | -- |
| Control | 6.9 ± 4.1 | 8.2 ± 4.4 | |||||
| Soleus (%SEMGMVC) b | Intervention | 18.7 ± 9.5 | 19.4 ± 10.3 | p = 0.760 | p = 0.926 | p = 0.802 | --- |
| Control | 20.6 ± 13.4 | 19.7 ± 11.3 | |||||
| Tibialis anterior (%SEMGMVC) b | Intervention | 12.8 ± 6.5 | 13.1 ± 6.5 | p = 0.372 | p = 0.218 | p = 0.692 | --- |
| Control | 14.4 ± 7.5 | 16.6 ± 8.5 | |||||
| Peroneus longus (%SEMGMVC) b | Intervention | 14.5 ± 8.9 | 15.1 ± 8.4 | p = 0.511 | p = 0.656 | p = 0.863 | --- |
| Control | 16.9 ± 6.4 | 18.1 ± 6.8 | |||||
| CMJ (cm) a | Intervention | 12.4 ± 4.3 | 15.2 ± 6.2 | p = 0.435 | p = 0.405 | p = 0.214 | --- |
| Control | 11.5 ± 4.3 | 10.9 ± 3.8 | |||||
| Abalakov (cm) a | Intervention | 15.9 ± 6.2 | 22.7 ± 7.2 * | p = 0.230 | p = 0.055 * | p = 0.015 * | f = 1.99 |
| Control | 13.7 ± 5.7 | 12.7 ± 5 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
González, L.; Argüelles, J.; González, V.; Winge, K.; Iscar, M.; Olmedillas, H.; Blanco, M.; Valenzuela, P.L.; Lucia, A.; Federolf, P.A.; et al. Slackline Training in Children with Spastic Cerebral Palsy: A Randomized Clinical Trial. Int. J. Environ. Res. Public Health 2020, 17, 8649. https://doi.org/10.3390/ijerph17228649
González L, Argüelles J, González V, Winge K, Iscar M, Olmedillas H, Blanco M, Valenzuela PL, Lucia A, Federolf PA, et al. Slackline Training in Children with Spastic Cerebral Palsy: A Randomized Clinical Trial. International Journal of Environmental Research and Public Health. 2020; 17(22):8649. https://doi.org/10.3390/ijerph17228649
Chicago/Turabian StyleGonzález, Lucía, Juan Argüelles, Vicente González, Kristian Winge, Marta Iscar, Hugo Olmedillas, Miguel Blanco, Pedro L. Valenzuela, Alejandro Lucia, Peter A. Federolf, and et al. 2020. "Slackline Training in Children with Spastic Cerebral Palsy: A Randomized Clinical Trial" International Journal of Environmental Research and Public Health 17, no. 22: 8649. https://doi.org/10.3390/ijerph17228649
APA StyleGonzález, L., Argüelles, J., González, V., Winge, K., Iscar, M., Olmedillas, H., Blanco, M., Valenzuela, P. L., Lucia, A., Federolf, P. A., & Santos, L. (2020). Slackline Training in Children with Spastic Cerebral Palsy: A Randomized Clinical Trial. International Journal of Environmental Research and Public Health, 17(22), 8649. https://doi.org/10.3390/ijerph17228649









