A Parental Smoking Cessation Intervention in the Pediatric Emergency Setting: A Randomized Trial
Abstract
1. Introduction
2. Materials and Methods
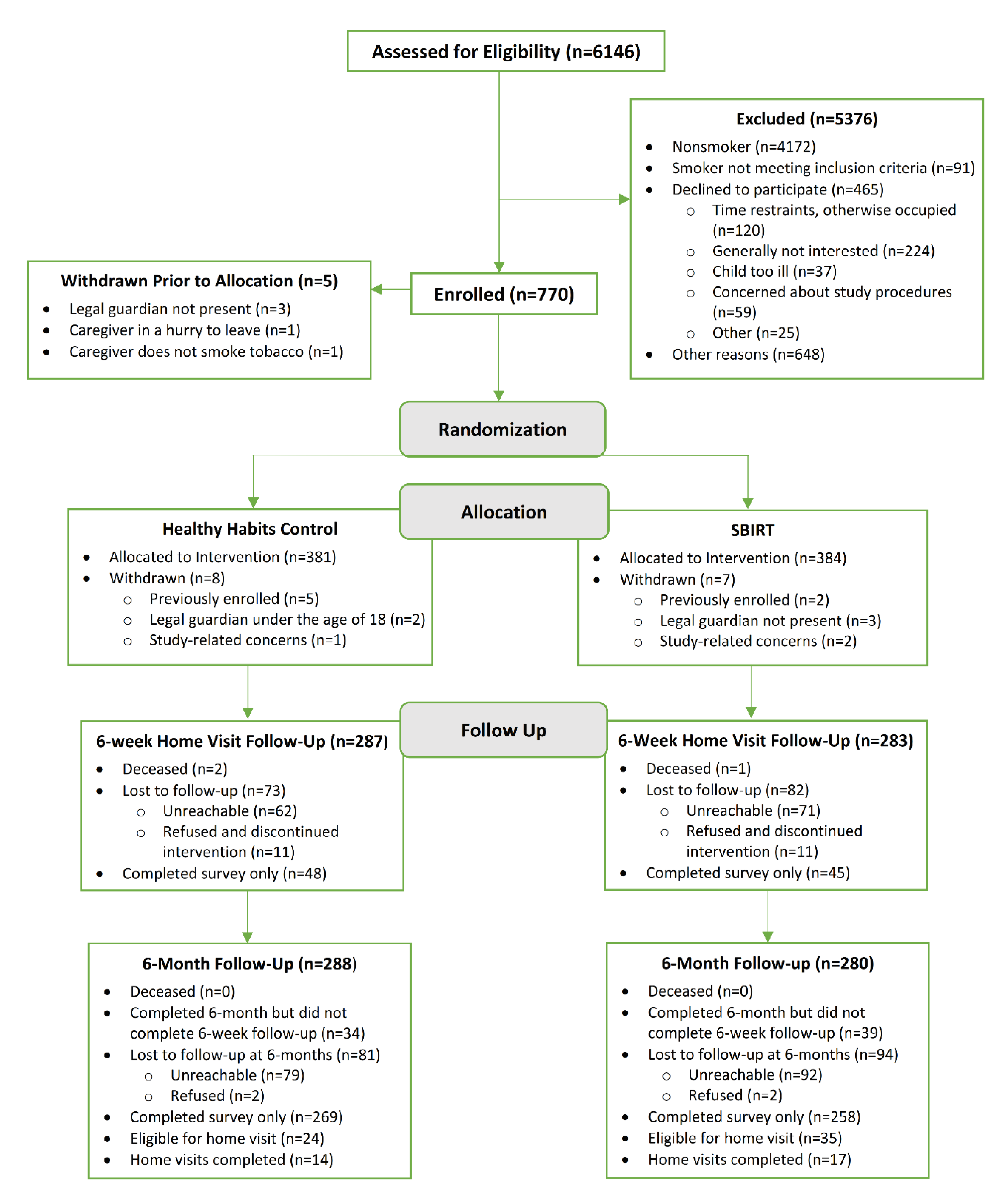
2.1. Design, Participant Recruitment and Screening
2.2. Randomization and Study Conditions
2.3. Parental Assessments
2.4. Child Measures, Saliva Collection and Analysis
2.5. Statistical Analysis
2.6. Sample Size and Power
3. Results
3.1. Participant Characteristics
3.2. Child T0 Cotinine Levels
3.3. Primary and Secondary Outcomes at T1 and T2
3.4. Cessation Resources Given to SBIRT Participants
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- McDermott, K.; Stocks, C.; Freeman, W. Statistical Brief #242. In Healthcare Cost and Utilization Project (HCUP); Agency for Healthcare Research and Quality: Rockville, MD, USA, 2018. [Google Scholar]
- Committee on Pediatric Emergency Medicine. Pediatric care recommendations for freestanding urgent care facilities. Pediatrics 2014, 133, 950–953. [Google Scholar] [CrossRef] [PubMed]
- Bornais, J.A.K.; Crawley, J.; El-Masri, M.M. One Stop: Examining the Reasons Patients Use the Emergency Department for Nonurgent Care and the Barriers They Face. J. Emerg. Nurs. 2020, 46, 163–170. [Google Scholar] [CrossRef] [PubMed]
- Farion, K.J.; Wright, M.; Zemek, R.; Neto, G.; Karwowska, A.; Tse, S.; Reid, S.; Jabbour, M.; Poirier, S.; Moreau, K.A.; et al. Understanding Low-Acuity Visits to the Pediatric Emergency Department. PLoS ONE 2015, 10, e0128927. [Google Scholar] [CrossRef]
- Long, C.M.; Mehrhoff, C.; Abdel-Latief, E.; Rech, M.; Laubham, M. Factors Influencing Pediatric Emergency Department Visits for Low-Acuity Conditions. Pediatr. Emerg. Care 2018. [Google Scholar] [CrossRef] [PubMed]
- Schlichting, L.E.; Rogers, M.L.; Gjelsvik, A.; Linakis, J.G.; Vivier, P.M. Pediatric Emergency Department Utilization and Reliance by Insurance Coverage in the United States. Acad. Emerg. Med. 2017, 24, 1483–1490. [Google Scholar] [CrossRef] [PubMed]
- Orlansky, A.; Smolij, C.; Moughan, B.; Aronoff, S.C. Factors that Affect Nonurgent Emergency Department Visits in a Publicly Insured Pediatric Population: An Observational Study. J. Healthc. Qual. 2016, 38, 195–201. [Google Scholar] [CrossRef]
- Burns, R.R.; Alpern, E.R.; Rodean, J.; Canares, T.; Lee, B.R.; Hall, M.; Montalbano, A. Factors Associated With Urgent Care Reliance and Outpatient Health Care Use Among Children Enrolled in Medicaid. JAMA Netw. Open 2020, 3, e204185. [Google Scholar] [CrossRef] [PubMed]
- Mahabee-Gittens, E.M.; Khoury, J.C.; Ho, M.; Stone, L.; Gordon, J.S. A smoking cessation intervention for low-income smokers in the ED. Am. J. Emerg. Med. 2015, 33, 1056–1061. [Google Scholar] [CrossRef] [PubMed]
- Mahabee-Gittens, E.M.; Stone, L.; Gordon, J.S. Pediatric emergency department is a promising venue for adult tobacco cessation interventions. Nicotine Tob. Res. 2013, 15, 1792–1793. [Google Scholar] [CrossRef][Green Version]
- Mahabee-Gittens, E.M.; Gordon, J.S.; Krugh, M.E.; Henry, B.; Leonard, A.C. A smoking cessation intervention plus proactive quitline referral in the pediatric emergency department: A pilot study. Nicotine Tob. Res. 2008, 10, 1745–1751. [Google Scholar] [CrossRef]
- Hall, N.; Hipple, B.; Friebely, J.; Ossip, D.J.; Winickoff, J.P. Addressing Family Smoking in Child Health Care Settings. J. Clin. Outcomes Manag. 2009, 16, 367–373. [Google Scholar] [PubMed]
- Mahabee-Gittens, E.M.; Gordon, J. Acceptability of tobacco cessation interventions in the pediatric emergency department. Pediatr. Emerg. Care 2008, 24, 214–216. [Google Scholar] [CrossRef]
- Mahabee-Gittens, E.M.; Merianos, A.L.; Stone, L.; Tabangin, M.E.; Khoury, J.C.; Gordon, J.S. Tobacco Use Behaviors and Perceptions of Parental Smokers in the Emergency Department Setting. Tob. Use Insights 2019, 12, 1179173x19841392. [Google Scholar] [CrossRef] [PubMed]
- Ralston, S.; Roohi, M. A randomized, controlled trial of smoking cessation counseling provided during child hospitalization for respiratory illness. Pediatr. Pulmonol. 2008, 43, 561–566. [Google Scholar] [CrossRef]
- Kells, M.; Rogers, J.; Oppenheimer, S.C.; Blaine, K.; McCabe, M.; McGrath, E.; Woodring, B.; Geller, A.C. The teachable moment captured: A framework for nurse-led smoking cessation interventions for parents of hospitalized children. Public Health Nurs. 2013, 30, 468–473. [Google Scholar] [CrossRef]
- Behbod, B.; Sharma, M.; Baxi, R.; Roseby, R.; Webster, P. Family and carer smoking control programmes for reducing children’s exposure to environmental tobacco smoke. Cochrane Database Syst. Rev. 2018, 1, CD001746. [Google Scholar] [CrossRef]
- Lemhoefer, C.; Rabe, G.L.; Wellmann, J.; Bernstein, S.L.; Cheung, K.W.; McCarthy, W.J.; Lauridsen, S.V.; Spies, C.; Neuner, B. Emergency Department-Initiated Tobacco Control: Update of a Systematic Review and Meta-Analysis of Randomized Controlled Trials. Prev. Chronic. Dis. 2017, 14, E89. [Google Scholar] [CrossRef] [PubMed]
- Rabe, G.L.; Wellmann, J.; Bagos, P.; Busch, M.A.; Hense, H.W.; Spies, C.; Weiss-Gerlach, E.; McCarthy, W.; Gareca Arizaga, M.J.; Neuner, B. Efficacy of emergency department-initiated tobacco control—Systematic review and meta-analysis of randomized controlled trials. Nicotine Tob. Res. 2013, 15, 643–655. [Google Scholar] [CrossRef]
- Bernstein, S.L.; Bijur, P.; Cooperman, N.; Jearld, S.; Arnsten, J.H.; Moadel, A.; Gallagher, E.J. A randomized trial of a multicomponent cessation strategy for emergency department smokers. Acad. Emerg. Med. 2011, 18, 575–583. [Google Scholar] [CrossRef]
- Bernstein, S.L.; D’Onofrio, G.; Rosner, J.; O’Malley, S.; Makuch, R.; Busch, S.; Pantalon, M.V.; Toll, B. Successful Tobacco Dependence Treatment in Low-Income Emergency Department Patients: A Randomized Trial. Ann. Emerg. Med. 2015, 66, 140–147. [Google Scholar] [CrossRef] [PubMed]
- Bernstein, S.L.; Rosner, J.; Toll, B. A Multicomponent Intervention Including Texting to Promote Tobacco Abstinence in Emergency Department Smokers: A Pilot Study. Acad. Emerg. Med. 2016, 23, 803–808. [Google Scholar] [CrossRef]
- Mahabee-Gittens, E.M.; Ammerman, R.T.; Khoury, J.; Stone, L.; Meyers, G.T.; Witry, J.K.; Merianos, A.L.; Mancuso, T.F.; Stackpole, K.; Bennett, B.L.; et al. Healthy families: Study protocol for a randomized controlled trial of a screening, brief intervention, and referral to treatment intervention for caregivers to reduce secondhand smoke exposure among pediatric emergency patients. BMC Public Health 2017, 17, 374. [Google Scholar] [CrossRef]
- Prochaska, J.O.; DiClemente, C.C.; Norcross, J.C. In search of how people change. Applications to addictive behaviors. Am. Psychol. 1992, 47, 1102–1114. [Google Scholar] [CrossRef]
- Fiore, M.; Jaen, C.; Baker, T.; Bailey, W.; Benowitz, N.L.; Curry, S.J.; Dorfman, S.F.; Froelicher, E.S.; Goldstein, M.G.; Healton, C.G.; et al. Treating Tobacco Use and Dependence: 2008 Update; US Department of Health and Human Services, Public Health Service: Rockville, MD, USA, 2008.
- Mahabee-Gittens, E.M.; Merianos, A.L.; Tabangin, M.E.; Stone, L.; Gordon, J.S.; Khoury, J.C. Provision of free nicotine replacement therapy to parental smokers in the pediatric emergency setting. Tob. Prev. Cessat. 2020, 6, 30. [Google Scholar] [CrossRef]
- Foltz, J.L.; Cook, S.R.; Szilagyi, P.G.; Auinger, P.; Stewart, P.A.; Bucher, S.; Dipl, D.; Baldwin, C.D. US Adolescent Nutrition, Exercise, and Screen Time Baseline Levels Prior to National Recommendations. Clin. Pediatr. 2011, 50, 424–433. [Google Scholar] [CrossRef]
- Rogers, V.W.; Hart, P.H.; Motyka, E.; Rines, E.N.; Vine, J.; Deatrick, D.A. Impact of Let’s Go! 5-2-1-0: A Community-Based, Multisetting Childhood Obesity Prevention Program. J. Pediatr. Psychol. 2013, 38, 1010–1020. [Google Scholar] [CrossRef]
- Harris, P.A.; Taylor, R.; Thielke, R.; Payne, J.; Gonzalez, N.; Conde, J.G. Research electronic data capture (REDCap)—A metadata-driven methodology and workflow process for providing translational research informatics support. J. Biomed. Inform. 2009, 42, 377–381. [Google Scholar] [CrossRef] [PubMed]
- Vinokur, A.D.; Price, R.H.; Caplan, R.D. Hard times and hurtful partners: How financial strain affects depression and relationship satisfaction of unemployed persons and their spouses. J. Personal. Soc. Psychol. 1996, 71, 166–179. [Google Scholar] [CrossRef]
- Pérez-Ríos, M.; I Santiago-Pérez, M.; Alonso, B.; Malvar, A.; Hervada, X.; De Leon, J. Fagerstrom test for nicotine dependence vs heavy smoking index in a general population survey. BMC Public Health 2009, 9, 493. [Google Scholar] [CrossRef]
- Chabrol, H.; Niezborala, M.; Chastan, E.; De Leon, J. Comparison of the Heavy Smoking Index and of the Fagerstrom Test for Nicotine Dependence in a sample of 749 cigarette smokers. Addict. Behav. 2005, 30, 1474–1477. [Google Scholar] [CrossRef]
- Biener, L.; Abrams, D.B. The Contemplation Ladder: Validation of a measure of readiness to consider smoking cessation. Health Psychol. 1991, 10, 360–365. [Google Scholar] [CrossRef] [PubMed]
- Benowitz, N.L.; Bernert, J.T.; Foulds, J.; Hecht, S.S.; Jacob, P.; Jarvis, M.J.; Joseph, A.; Oncken, C.; Piper, M.E. Biochemical Verification of Tobacco Use and Abstinence: 2019 Update. Nicotine Tob. Res. 2019, 22, 1086–1097. [Google Scholar] [CrossRef]
- Wagener, T.L.; Gregor, K.L.; Busch, A.M.; McQuaid, E.L.; Borrelli, B. Risk perception in smokers with children with asthma. J. Consult. Clin. Psychol. 2010, 78, 980–985. [Google Scholar] [CrossRef]
- Benowitz, N.L.; Hukkanen, J.; Jacob, P. Nicotine Chemistry, Metabolism, Kinetics and Biomarkers. Handb. Exp. Pharmacol. 2009, 29–60. [Google Scholar] [CrossRef]
- Murphy, S.E.; Wickham, K.M.; Lindgren, B.R.; Spector, L.G.; Joseph, A.M. Cotinine and trans 3′-hydroxycotinine in dried blood spots as biomarkers of tobacco exposure and nicotine metabolism. J. Expo. Sci. Environ. Epidemiol. 2013, 23, 513–518. [Google Scholar] [CrossRef]
- Mahabee-Gittens, E.M.; Mazzella, M.J.; Doucette, J.T.; Merianos, A.L.; Stone, L.; Wullenweber, C.A.; Busgang, S.A.; Matt, G.E. Comparison of Liquid Chromatography Mass Spectrometry and Enzyme-Linked Immunosorbent Assay Methods to Measure Salivary Cotinine Levels in Ill Children. Int. J. Environ. Res. Public Health 2020, 17, 1157. [Google Scholar] [CrossRef] [PubMed]
- Butz, A.; Tsoukleris, M.; Bollinger, M.E.; Jassal, M.; Bellin, M.H.; Kub, J.; Mudd, S.; Ogborn, C.J.; Lewis-Land, C.; Thompson, R.E. Association between second hand smoke (SHS) exposure and caregiver stress in children with poorly controlled asthma. J. Asthma 2019, 56, 915–926. [Google Scholar] [CrossRef]
- Schulz, K.F.; Altman, D.G.; Moher, D. Consort Group, CONSORT 2010 Statement: Updated guidelines for reporting parallel group randomised trials. BMC Med. 2010, 8, 18. [Google Scholar] [CrossRef] [PubMed]
- Gordon, J.S.; Andrews, J.A.; Albert, D.A.; Crews, K.M.; Payne, T.J.; Severson, H.H. Tobacco Cessation via Public Dental Clinics: Results of a Randomized Trial. Am. J. Public Health 2010, 100, 1307–1312. [Google Scholar] [CrossRef] [PubMed]
- Stead, L.F.; Perera, R.; Bullen, C.; Mant, D.; Hartmann-Boyce, J.; Cahill, K.; Lancaster, T. Nicotine replacement therapy for smoking cessation. Cochrane Database Syst. Rev. 2012, 11, CD000146. [Google Scholar] [CrossRef]
- Bernstein, S.L.; Toll, B.A. Ask about smoking, not quitting: A chronic disease approach to assessing and treating tobacco use. Addict. Sci. Clin. Pract. 2019, 14, 1–4. [Google Scholar] [CrossRef]
- Bernstein, S.; Dziura, J.; Weiss, J.; Harper Brooks, A.; Miller, T.; Vickerman, K.; Grau, L.; Pantalon, M.; Abroms, L.; Collins, L.; et al. Successful Treatment of Tobacco Dependence Treatment in the Emergency Department Using the Multiphase Optimization Strategy. In Annual Meeting of the Society for Research on Nicotine and Tobacco; SRNT: New Orleans, LA, USA, 2020. [Google Scholar]
- Bernstein, J.; Bernstein, E.; Heeren, T.C. Mechanisms of change in control group drinking in clinical trials of brief alcohol intervention: Implications for bias toward the null. Drug Alcohol Rev. 2010, 29, 498–507. [Google Scholar] [CrossRef]
- Bernstein, E.; Bernstein, J. Effectiveness of alcohol screening and brief motivational intervention in the emergency department setting. Ann. Emerg. Med. 2008, 51, 751–754. [Google Scholar] [CrossRef]
- D’Onofrio, G.; Pantalon, M.V.; DeGutis, L.C.; Fiellin, D.A.; Busch, S.H.; Chawarski, M.C.; Owens, P.H.; O’Connor, P.G. Brief Intervention for Hazardous and Harmful Drinkers in the Emergency Department. Ann. Emerg. Med. 2008, 51, 742–750. [Google Scholar] [CrossRef] [PubMed]
- Bernstein, S.L.; Bijur, P.; Cooperman, N.A.; Jearld, S.; Arnsten, J.H.; Moadel, A.; Gallagher, E.J. Efficacy of an emergency department-based multicomponent intervention for smokers with substance use disorders. J. Subst. Abus. Treat. 2013, 44, 139–142. [Google Scholar] [CrossRef][Green Version]
- Bock, B.C.; Becker, B.M.; Niaura, R.; Partridge, R.; Fava, J.L.; Trask, P. Smoking cessation among patients in an emergency chest pain observation unit: Outcomes of the Chest Pain Smoking Study (CPSS). Nicotine Tob. Res. 2008, 10, 1523–1531. [Google Scholar] [CrossRef]
- Nabi-Burza, E.; Drehmer, J.E.; Walters, B.H.; Rigotti, N.A.; Ossip, D.J.; Levy, D.E.; Klein, J.D.; Regan, S.; Gorzkowski, J.A.; Winickoff, J.P. Treating Parents for Tobacco Use in the Pediatric Setting. JAMA Pediatr. 2019, 173, 931–939. [Google Scholar] [CrossRef]
- Collins, B.N.; Lepore, S.J.; Winickoff, J.P.; Sosnowski, D.W. Parents’ self-efficacy for tobacco exposure protection and smoking abstinence mediate treatment effects on child cotinine at 12-month follow-up: Mediation results from the Kids Safe and Smokefree trial. Nicotine Tob. Res. 2019, 22, 1981–1988. [Google Scholar] [CrossRef]
- Mahabee-Gittens, E.M.; Merianos, A.L.; Dexheimer, J.W.; Meyers, G.T.; Stone, L.; Tabangin, M.; Khoury, J.C.; Gordon, J.S. Utilization of a Clinical Decision Support Tool to Reduce Child Tobacco Smoke Exposure in the Urgent Care Setting. Pediatr. Emerg. Care 2018. [Google Scholar] [CrossRef]
- Mahabee-Gittens, E.M.; Dexheimer, J.W.; Tabangin, M.; Khoury, J.C.; Merianos, A.L.; Stone, L.; Meyers, G.T.; Gordon, J.S. An Electronic Health Record−Based Strategy to Address Child Tobacco Smoke Exposure. Am. J. Prev. Med. 2018, 54, 64–71. [Google Scholar] [CrossRef] [PubMed]
- Jenssen, B.P.; Muthu, N.; Kelly, M.K.; Baca, H.; Shults, J.; Grundmeier, R.W.; Fiks, A.G. Parent eReferral to Tobacco Quitline: A Pragmatic Randomized Trial in Pediatric Primary Care. Am. J. Prev. Med. 2019, 57, 32–40. [Google Scholar] [CrossRef] [PubMed]
- Keller, P.A.; Lien, R.K.; Beebe, L.A.; Parker, J.; Klein, P.; Lachter, R.B.; Gillaspy, S. Replicating state Quitline innovations to increase reach: Findings from three states. BMC Public Health 2020, 20, 7–11. [Google Scholar] [CrossRef]
- Kruse, G.R.; Park, E.R.; Chang, Y.; Haberer, J.E.; Abroms, L.C.; Shahid, N.N.; Howard, S.; Haas, J.S.; Rigotti, N.A. Proactively Offered Text Messages and Mailed Nicotine Replacement Therapy for Smokers in Primary Care Practices: A Pilot Randomized Trial. Nicotine Tob. Res. 2020, 22, 1509–1514. [Google Scholar] [CrossRef]
- Chaiton, M.; Diemert, L.; Cohen, J.E.; Bondy, S.J.; Selby, P.; Philipneri, A.; Schwartz, R. Estimating the number of quit attempts it takes to quit smoking successfully in a longitudinal cohort of smokers. BMJ Open 2016, 6, e011045. [Google Scholar] [CrossRef]
- Bernstein, S.L.; Boudreaux, E.D.; Cydulka, R.K.; Rhodes, K.V.; Lettman, N.A.; Almeida, S.L.; McCullough, L.B.; Mizouni, S.; Kellermann, A.L. American College of Emergency Physicians Task Force on Smoking, C. Tobacco control interventions in the emergency department: A joint statement of emergency medicine organizations. Ann. Emerg. Med. 2006, 48, e417–e426. [Google Scholar] [CrossRef]
| Variable | Overall | SBIRT 1 | HHC 2 | p-Value |
|---|---|---|---|---|
| (N = 750) | (N = 377) | (N = 373) | SBIRT vs. HHC | |
| n (%) | n (%) | n (%) | ||
| Parent Age, Mean (SD) | 31.76 (7.65) | 31.97 (8.23) | 31.55 (7.03) | 0.46 * |
| Parent Sex-Female | 651 (86.80) | 330 (87.53) | 321 (86.06) | 0.55 |
| Parent Race/Ethnicity | 0.99 ** | |||
| White, non-Hispanic | 285 (39.39) | 149 (39.52) | 146 (39.25) | |
| Black, non-Hispanic | 395 (52.74) | 198 (52.52) | 197 (52.96) | |
| Other, non-Hispanic | 42 (5.61) | 22 (5.84) | 20 (5.38) | |
| Hispanic | 17 (2.27) | 8 (2.12) | 9 (2.42) | |
| Insurance | 0.75 | |||
| Public or None | 655 (87.33) | 328 (87.00) | 327 (87.67) | |
| Commercial | 80 (10.67) | 40 (10.61) | 40 (10.72) | |
| Other | 15 (2.00) | 9 (2.39) | 6 (1.61) | |
| Income | 0.06 | |||
| ≤$15,000 | 483 (64.57) | 231 (61.27) | 252 (67.92) | |
| >$15,000 | 265 (35.43) | 146 (38.73) | 119 (32.08) | |
| Financial Strain | 0.17 * | |||
| Mean (SD) | 2.41 (1.10) | 2.46 (1.10) | 2.35 (1.10) | |
| Median (IQR) | 2.33 (1.67, 3.00) | 2.33 (1.67, 3.33) | 2.00 (1.33, 3.00) | |
| Employed (yes) | 403 (54.24) | 208 (56.06) | 195 (52.42) | 0.32 |
| Highest Education Level | 0.43 | |||
| Less than HS/HS | 427 (56.93) | 220 (58.36) | 207 (55.50) | |
| ≥Some college | 323 (43.07) | 157 (41.64) | 166 (44.50) | |
| Child age, Mean (SD) | 4.91 (4.71) | 5.07 (4.88) | 4.74 (5.54) | 0.36 * |
| Number of Daily Cigarettes Smoked by the Parent | 0.39 | |||
| Mean (SD) | 10.23 (7.23) | 10.53 (7.47) | 9.93 (6.97) | |
| Median (IQR) | 10.00 (5, 15) | 10.00 (5, 15) | 10.00 (5, 15) | |
| Current electronic cigarette user | 39 (5.20) | 18 (4.83) | 21 (5.57) | 0.65 |
| Stage of Change | 0.94 | |||
| Pre-Contemplation | 111 (14.80) | 56 (14.85) | 55 (14.75) | |
| Contemplation | 402 (53.60) | 204 (54.11) | 198 (53.08) | |
| Preparation | 237 (31.60) | 117 (31.03) | 120 (32.17) | |
| Past Year Quit Attempts | 539 (71.87) | 262 (69.50) | 277 (74.26) | 0.15 |
| Number of Household SmokersMean (SD) | 2.29 (1.45) | 2.53 (1.48) | 2.44 (1.42) | 0.41 * |
| Home Smoking Ban | 212/485 (43.71) | 107/243 (44.03) | 105/242 (43.39) | 0.89 |
| Home and Car Ban | 93/485 (19.18) | 44/243 (18.11) | 49/242 (20.25) | 0.55 |
| Cotinine ng/mLMedian (IQR) | 6.10 (2.61, 12.05) | 6.74 (2.70, 12.58) | 5.60 (2.40, 11.65) | 0.40 * |
| Parent Believes Smoking Affects Child’s Health (PV 3 total), Mean (SD) | 11.95 (4.22) | 11.93 (4.16) | 11.97 (4.28) | 0.90 * |
| Parent Believes Quitting Would Benefit Child (PE 4 total), Mean (SD) | 13.35 (4.71) | 13.21 (4.70) | 13.49 (4.72) | 0.43 * |
| Variable | SBIRT3 | HHC4 | p-Value | SBIRT | HHC | p-Value |
|---|---|---|---|---|---|---|
| T1 | T1 | T2 | T2 | |||
| n (%) | n (%) | n (%) | n (%) | |||
| N | 283 | 288 | -- | 280 | 289 | -- |
| Abstinence, Biochemically Verified N/ Total N of Participants Who Self-Reported Quitting (%) | 9/10 (90) | 5/8 (62.5) | 0.27 + | 11/17 (64.7) | 10/14 (71.4) | 1.00 + |
| Abstinence, Self-report | 12 (4.24) | 12 (4.17) | 0.96 | 36 (12.9) | 24 (8.30) | 0.08 |
| Number of Daily CigarettesMedian (IQR) | 6 (3, 10) | 7 (4, 11.5) | 0.11 | 5 (2, 10) | 5 (2, 10) | 0.049 |
| Change in Number of Daily Cigarettes from Baseline Median (IQR) | −2 [−5, 0] | 0 [−4, 0] | 0.0008 | −4 [−9, −1] | −2 [−5, 0] | 0.0006 |
| Change in Number of Daily Cigarettes from T1 Median (IQR) | -- | -- | -- | 0 [−4, 1] | 0 [−2, 0] | 0.84 |
| Motivation to Quit | 0.03 | |||||
| Mean (SD) | 6.98 (2.47) | 6.46 (2.78) | 7.03 (2.58) | 6.77 (2.80) | 0.41 | |
| Median (IQR) | 8 (5, 9) | 6 (5, 9) | 8 (5, 9) | 7 (5, 9) | ||
| Quit Attempts Since Baseline | ||||||
| Mean (SD) | 2.69 (4.98) | 2.22 (2.53) | 0.99 | 3.82 (6.36) | 2.34 (3.24) | 0.003 |
| Median (IQR) | 2 (0,3) | 2 (0, 3) | 2 (1, 4) | 2 (0, 3) | ||
| Change in Number of Quit Attempts from Baseline | 0.11 | 0.02 | ||||
| Mean (SD) | 0.44 (5.54) | −0.25 (4.85) | 1.25 (6.5) | 0.02 (4.71) | ||
| Median (IQR) | 0 (−1,1) | 0 (−1, 1) | 0 (−2, 1) | 0 (−1, 1) | ||
| Number of Household Smokers | ||||||
| Mean (SD) | 1.49 (1.28) | 1.45 (0.76) | 0.9 | 1.22 (0.80) | 1.21 (0.64) | |
| Median (IQR) | 1 (1, 2) | 1 (1, 2) | 1 (1, 1) | 1 (1, 2) | 0.59 | |
| Home Smoking Ban | 82 (43.85) | 82 (44.09) | 0.96 | 111 (51.63) | 120 (52.63) | 0.83 |
| Home and Car Smoking Ban | 47 (25.13) | 38 (20.32) | 0.27 | 69 (32.09) | 84 (36.84) | 0.29 |
| Cotinine Change from T0 ng/mL * | 0.05 | 0.66 | ||||
| Median (IQR) | 1.81 (−2.94, 3.24) | 1.81 (−2.02, 5.52) | 2.44 (−1.61, 6.92) | 1.81 (−2.18, 5.52) |
| Variable | Yes, at T1 | No, at T1 | p-Value | Yes, at T2 | No, at T2 | p-Value |
|---|---|---|---|---|---|---|
| n (%) | n (%) | n (%) | n (%) | |||
| N | 24 | 547 | 60 | 509 | ||
| Financial Strain >2 at Baseline | 11 (45.83) | 201 (53.20) | 0.53 * | 27 (45.00) | 267 (52.46) | 0.27 |
| NRT 3 Given at Baseline † | 10/12 (83.33) | 138/271 (50.92) | 0.04 * | 20/36 (55.56) | 124/244 (50.82) | 0.60 |
| Number of Household Cigarette Smokers >1 | 9 (37.50) | 389 (71.12) | 0.001 * | 43 (71.67) | 353 (69.35) | 0.71 |
| Parent Believes Smoking Affects Child’s Health (PV 4 ≥12) at Baseline | 16 (66.67) | 288 (52.65) | 0.21 * | 31 (51.67) | 263 (51.67) | 1.00 |
| Parent Believes Quitting Would Benefit Child (PE 5 ≥14) at Baseline | 13 (54.17) | 270 (49.36) | 0.68 * | 32 (53.33) | 254 (49.90) | 0.62 |
| Parent Believes Smoking Affects Child’s Health (PV ≥ 12) at T1 | 16 (66.67) | 292/544 (53.68) | 0.30 * | 23/47 (48.94) | 247/446 (55.38) | 0.40 |
| Parent Believes Quitting Would Benefit Child (PE ≥ 14) at T1 | 15 (62.50) | 277/544 (50.92) | 0.30 * | 26/47 (55.32) | 229/446 (51.35) | 0.60 |
| Parent Believes Smoking Affects Child’s Health (PV ≥ 12) at T2 | 11/20 (55.00) | 253/476 (53.15) | 1.00 * | 34 (56.67) | 265 (52.06) | 0.50 |
| Parent Believes Quitting Would Benefit Child (PE ≥ 14) at T2 | 14/20 (70.00) | 261/476 (54.83) | 0.25 * | 44 (73.33) | 281 (55.21) | 0.007 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mahabee-Gittens, E.M.; Ammerman, R.T.; Khoury, J.C.; Tabangin, M.E.; Ding, L.; Merianos, A.L.; Stone, L.; Gordon, J.S. A Parental Smoking Cessation Intervention in the Pediatric Emergency Setting: A Randomized Trial. Int. J. Environ. Res. Public Health 2020, 17, 8151. https://doi.org/10.3390/ijerph17218151
Mahabee-Gittens EM, Ammerman RT, Khoury JC, Tabangin ME, Ding L, Merianos AL, Stone L, Gordon JS. A Parental Smoking Cessation Intervention in the Pediatric Emergency Setting: A Randomized Trial. International Journal of Environmental Research and Public Health. 2020; 17(21):8151. https://doi.org/10.3390/ijerph17218151
Chicago/Turabian StyleMahabee-Gittens, E. Melinda, Robert T. Ammerman, Jane C. Khoury, Meredith E. Tabangin, Lili Ding, Ashley L. Merianos, Lara Stone, and Judith S. Gordon. 2020. "A Parental Smoking Cessation Intervention in the Pediatric Emergency Setting: A Randomized Trial" International Journal of Environmental Research and Public Health 17, no. 21: 8151. https://doi.org/10.3390/ijerph17218151
APA StyleMahabee-Gittens, E. M., Ammerman, R. T., Khoury, J. C., Tabangin, M. E., Ding, L., Merianos, A. L., Stone, L., & Gordon, J. S. (2020). A Parental Smoking Cessation Intervention in the Pediatric Emergency Setting: A Randomized Trial. International Journal of Environmental Research and Public Health, 17(21), 8151. https://doi.org/10.3390/ijerph17218151






