The Association between Influenza Vaccination and the Risk of SARS-CoV-2 Infection, Severe Illness, and Death: A Systematic Review of the Literature
Abstract
:1. Introduction
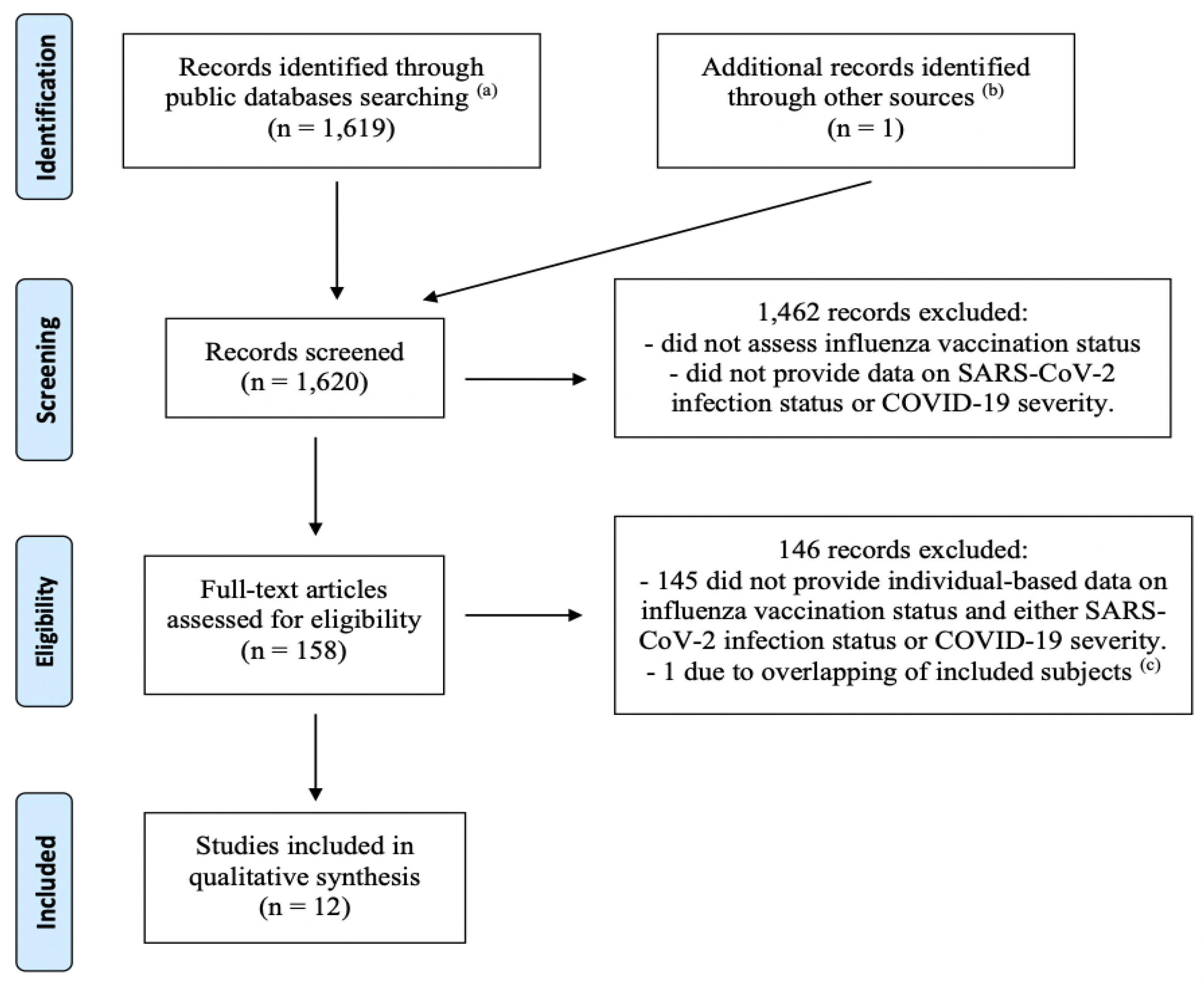
2. Materials and Methods
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Li, Q.; Tang, B.; Bragazzi, N.L.; Xiao, Y.; Wu, J. Modeling the impact of mass influenza vaccination and public health interventions on COVID-19 epidemics with limited detection capability. Math. Biosci. 2020, 325, 108378. [Google Scholar] [CrossRef] [PubMed]
- Zanettini, C.; Omar, M.; Dinalankara, W.; Imada, E.L.; Colantuoni, E.; Parmigiani, G.; Marchionni, L. Influenza Vaccination and COVID19 Mortality in the USA. medRxiv 2020. Available online: https://www.medrxiv.org/content/10.1101/2020.06.24.20129817v1 (accessed on 26 June 2020).
- Marín-Hernández, D.; Schwartz, R.E.; Nixon, D.F. Epidemiological evidence for association between higher influenza vaccine uptake in the elderly and lower COVID-19 deaths in Italy. J. Med. Virol. 2020. [Google Scholar] [CrossRef] [PubMed]
- Wells, G.; Shea, B.; O’Connel, D.; Peterson, J.; Welch, V.; Losos, M.; Tugwell, P. The Newcastle Ottawa Scale (NOS) for Assessing the Quality of Nonrandomized Studies in Meta-Analyses. 2013. Available online: http://www.ohri.ca/programs/clinical_epidemiology/oxford.asp (accessed on 19 October 2020).
- Santos, M.; Lucena, E.E.S.; Lima, K.C.; Brito, A.A.C.; Bonfada, D. Survival and predictors of deaths of patients hospitalized due to COVID-19 from a retrospective and multicenter cohort study in Brazil. medRxiv 2020. Available online: https://www.medrxiv.org/content/10.1101/2020.06.07.20125047v1 (accessed on 9 June 2020).
- Fink, G.; Orlova-Fink, N.; Schindler, T.; Grisi, S.; Ferrer, A.P.; Daubenberger, C.; Brentani, A. Inactivated trivalent influenza vaccine is associated with lower mortality among Covid-19 patients in Brazil. medRxiv 2020. Available online: https://www.medrxiv.org/content/10.1101/2020.06.29.20142505v1 (accessed on 1 July 2020).
- Aziz, N.A.; Corman, V.M.; Echterhoff, A.K.C.; Richter, A.; Schmandke, A.; Schmidt, M.L.; Schmidt, T.H.; De Vries, F.M.; Drosten, C.; Breteler, M.M.B. Seroprevalence and correlates of SARS-CoV-2 neutralizing antibodies: Results from a population-based study in Bonn, Germany. medRxiv 2020. Available online: https://www.medrxiv.org/content/10.1101/2020.08.24.20181206v1 (accessed on 29 August 2020).
- Caban-Martinez, A.J.; Schaefer-Solle, N.; Santiago, K.; Louzado-Feliciano, P.; Brotons, A.; Gonzalez, M.; Issenberg, S.B.; Kobetz, E. Epidemiology of SARS-CoV-2 antibodies among firefighters/paramedics of a US fire department: A cross-sectional study. Occup. Environ. Med. 2020. [Google Scholar] [CrossRef] [PubMed]
- Donato, M.F.; Invernizzi, F.; Lampertico, P.; Rossi, G. Health Status of Patients Who Underwent Liver Transplantation During the Coronavirus Outbreak at a Large Center in Milan, Italy. Clin. Gastroenterol. Hepatol. 2020, 18, 2131–2133. [Google Scholar] [CrossRef] [PubMed]
- Jehi, L.; Ji, X.; Milinovich, A.; Erzurum, S.; Rubin, B.P.; Gordon, S.; Young, J.B.; Kattan, M.W. Individualizing Risk Prediction for Positive Coronavirus Disease 2019 Testing: Results from 11,672 Patients. Chest 2020, 158, 1364–1375. [Google Scholar] [CrossRef] [PubMed]
- Noale, M.; Trevisan, C.; Maggi, S.; Antonelli Incalzi, R.; Pedone, C.; Di Bari, M.; Adorni, F.; Jesuthasan, N.; Sojic, A.; Galli, M.; et al. The Association between Influenza and Pneumococcal Vaccinations and SARS-Cov-2 Infection: Data from the EPICOVID19 Web-Based Survey. Vaccines 2020, 8, 471. [Google Scholar] [CrossRef] [PubMed]
- Pawlowski, C.; Puranik, A.; Bandi, H.; Venkatakrishnan, A.J.; Agarwal, V.; Kennedy, R.; O’Horo, J.C.; Gores, G.J.; Williams, A.W.; Halamka, J.; et al. Exploratory analysis of immunization records highlights decreased SARS-CoV-2 rates in individuals with recent non-COVID-19 vaccinations. medRxiv 2020. Available online: https://www.medrxiv.org/content/10.1101/2020.07.27.20161976v2 (accessed on 29 July 2020).
- Vila-Córcoles, Á.; Ochoa-Gondar, O.; Torrente-Fraga, C.; Vila-Rovira, Á.; Satué-Gracia, E.; Hospital-Guardiola, I.; de Diego-Cabanes, C.; Gómez-Bertomeu, F.; Basora-Gallisà, J. Evaluación de la incidencia y perfil de riesgo de Covid-19 según comorbilidad previa en adultos ≥50 años del área de Tarragona [Evaluation of incidence and risk profile for suffering Covid-19 infection by underlying conditions among middle-aged and older adults in Tarragona]. Rev. Esp. Salud Publica 2020, 94, e202006065. [Google Scholar] [PubMed]
- Jehi, L.; Ji, X.; Milinovich, A.; Erzurum, S.; Merlino, A.; Gordon, S.; Young, J.B.; Kattan, M.W. Development and validation of a model for individualized prediction of hospitalization risk in 4536 patients with COVID-19. PLoS ONE 2020, 15, e0237419. [Google Scholar] [CrossRef] [PubMed]
- Murillo-Zamora, E.; Trujillo, X.; Huerta, M.; Ríos-Silva, M.; Mendoza-Cano, O. Male gender and kidney illness associated with an increased risk of severe laboratory-confirmed coronavirus disease. BMC Infect. Dis. 2020, 20, 674. [Google Scholar] [CrossRef] [PubMed]
- Ortiz-Prado, E.; Simbana-Rivera, K.; Diaz, A.M.; Barreto, A.; Moyano, C.; Arcos, V.; Vasconez-Gonzalez, E.; Paz, C.; Simbana-Guaycha, F.; Molestina-Luzuriaga, M.; et al. Epidemiological, socio-demographic and clinical features of the early phase of the COVID-19 epidemic in Ecuador. medRxiv 2020. Available online: https://www.medrxiv.org/content/10.1101/2020.05.08.20095943v2 (accessed on 18 May 2020).
- Poblador-Plou, B.; Carmona-Pírez, J.; Ioakeim-Skoufa, I.; Poncel-Falcó, A.; Bliek-Bueno, K.; Cano-Del Pozo, M.; Gimeno-Feliú, L.A.; González-Rubio, F.; Aza-Pascual-Salcedo, M.; Bandrés-Liso, A.C.; et al. Baseline Chronic Comorbidity and Mortality in Laboratory-Confirmed COVID-19 Cases: Results from the PRECOVID Study in Spain. Int. J. Environ. Res. Public Health 2020, 17, 5171. [Google Scholar] [CrossRef] [PubMed]
- Gostin, L.O.; Salmon, D.A. The Dual Epidemics of COVID-19 and Influenza: Vaccine Acceptance, Coverage, and Mandates. JAMA 2020, 324, 335–336. [Google Scholar] [CrossRef] [PubMed]
- Wolff, G.G. Influenza vaccination and respiratory virus interference among Department of Defense personnel during the 2017–2018 influenza season. Vaccine 2020, 38, 350–354. [Google Scholar] [CrossRef] [PubMed]
- Ainslie, K.E.C.; Haber, M.; Orenstein, W.A. Challenges in estimating influenza vaccine effectiveness. Expert Rev. Vaccines 2019, 18, 615–628. [Google Scholar] [CrossRef] [PubMed]
| Author | Study Sample | Age (Years) | Laboratory Method | Main Result | Adjustment |
|---|---|---|---|---|---|
| Aziz et al. [7] | Subjects participating in a community-based cohort study in Bonn, Germany (n = 4755) | ≥30 (mean 55.2, SD 13.6) | ELISA and PRNT | No statistically significant association (no details available) | None |
| Caban-Martinez et al. [8] | Frontline firefighters and paramedics of a fire department in Florida, USA (n = 203) (a) | ≥21 | Point-of-care IgM-IgG LFIA | COVID-19 cases were significantly less likely to be vaccinated than controls (p = 0.027) | None |
| Donato et al. [9] | Liver transplant patients in Italy (n = 640) | ≥20 (median 63) | Not specified | No statistically significant association (p = 0.238) | None |
| Jehi et al. [10] | Subjects tested in several clinics in Ohio, USA (n = 11,672) (b) | Any age COVID-19 negative: median 46.9, IQR 31.6–62.9 COVID-19 positive: median 54.2, IQR 38.8–65.9 | RT-PCR | COVID-19 cases were significantly less likely to be vaccinated than controls (p < 0.001) | None |
| Subjects tested in several clinics in Florida, USA (n = 2295) (b) | Any age COVID-19 negative: median 56.0, IQR 41.9–67.5 COVID–19 positive: median 52.6, IQR 36.7–63.1 | COVID-19 cases were significantly less likely to be vaccinated than controls (p = 0.011) | |||
| Noale et al. [11] | Subjects years participating in a web-based survey in Italy (n = 6650) (c) | ≥18 (mean 48.0, SD 1 7.7) | Not specified | Vaccinated subjects <65 years old were significantly less likely to be infected (OR 0.85, 95% CI 0.74–0.98, p = 0.024). No statistically significant association among subjects ≥ 65 years (OR 0.87, 95% CI 0.59–1.28, p = 0.483) | Age, gender, education, comorbidities, other |
| Pawlowski et al. [12] | Subjects who received SARS-CoV-2 testing at Mayo Clinic, USA (n = 137,037) | Any age | PCR | Subjects vaccinated in the past year were significantly less likely to be infected (RR 0.85, 95% CI 0.75–0.96, p = 0.03). The association was stronger in the ≥ 65 years subgroup (RR 0.74, 95% CI 0.61–0.89, p < 0.01). | Propensity score matching (d) and multiple comparison |
| Vila-Córcoles et al. [13] | All subjects tested at primary healthcare center in Tarragona area, Spain (n = 79,071) | ≥50 | RT-PCR | Vaccinated subjects were significantly less likely to be infected (HR 0.63, 95% CI 0.43–0.92, p = 0.015) | Age, gender, vaccination history, comorbidities |
| Author | Study Sample | Age (Years) | Outcome | Main Result | Adjustment |
|---|---|---|---|---|---|
| Jehi et al. [14] | Laboratory-confirmed COVID-19 patients in Ohio and Florida, USA (n = 2852) (a) | Any age Not hospitalized: median 50.8, IQR 35.8–64.4 Hospitalized: median 64.4, IQR 54.8–76.6 | Severe illness | Severe cases (requiring hospitalization) were significantly less likely to be vaccinated than non-severe cases (p < 0.001) (b) | None |
| Laboratory-confirmed COVID-19 patients in Ohio and Florida, USA (n = 1684) (a) | Any age Not hospitalized: median 45.6, IQR 30.5–65.9 Hospitalized: median 64.9, IQR 52.5–76.8 | Severe illness | Severe cases (requiring hospitalization) were significantly less likely to be vaccinated than non-severe cases (p < 0.001) (b) | None | |
| Murillo-Zamora et al. [15] | Laboratory-confirmed COVID-19 patients in Mexico (n = 740) | ≥15 (mean 43.7, SD 14.9) | Severe illness | Severe cases (dyspnoea requiring hospitalization) were non-significantly less likely to be vaccinated than non-severe cases (p = 0.073) | None |
| Fink et al. [6] | All clinically confirmed COVID-19 patients in Brazil (n = 92,664, of which 84% laboratory-confirmed) (c) | Any age (median 59) | Severe illness | Vaccinated patients were significantly less likely to require intensive care (OR 0.92, 95% CI 0.86–0.99, p < 0.05) or respiratory support (OR 0.81, 95% CI 0.74–0.88, p < 0.01) | Age, SES, comorbidities, other |
| Death | Vaccinated patients were at significantly reduced risk of death (OR 0.82, 95% CI 0.75–0.89, p < 0.01) (d) | ||||
| Ortiz-Prado et al. [16] | All laboratory-confirmed COVID-19 patients in Ecuador (n = 9468) | Any age Men: median 42, IQR 32–56 Women: median 39, IQR 30–54 | Death | No statistically significant association (OR among vaccinated patients: 0.71, 95% CI 0.23–2.17) | Age, gender, comorbidities |
| Poblador-Plou et al. [17] | All laboratory-confirmed COVID-19 patients in Aragon, Spain, with follow-up ≥ 30 days (n = 4412) | Any age (mean 67.7, SD 20.7) | Death | No significant differences in the proportion of vaccination between deceased and alive patients after adjusting by age (p = 0.110 among men, p = 0.126 among women) | Age |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Del Riccio, M.; Lorini, C.; Bonaccorsi, G.; Paget, J.; Caini, S. The Association between Influenza Vaccination and the Risk of SARS-CoV-2 Infection, Severe Illness, and Death: A Systematic Review of the Literature. Int. J. Environ. Res. Public Health 2020, 17, 7870. https://doi.org/10.3390/ijerph17217870
Del Riccio M, Lorini C, Bonaccorsi G, Paget J, Caini S. The Association between Influenza Vaccination and the Risk of SARS-CoV-2 Infection, Severe Illness, and Death: A Systematic Review of the Literature. International Journal of Environmental Research and Public Health. 2020; 17(21):7870. https://doi.org/10.3390/ijerph17217870
Chicago/Turabian StyleDel Riccio, Marco, Chiara Lorini, Guglielmo Bonaccorsi, John Paget, and Saverio Caini. 2020. "The Association between Influenza Vaccination and the Risk of SARS-CoV-2 Infection, Severe Illness, and Death: A Systematic Review of the Literature" International Journal of Environmental Research and Public Health 17, no. 21: 7870. https://doi.org/10.3390/ijerph17217870
APA StyleDel Riccio, M., Lorini, C., Bonaccorsi, G., Paget, J., & Caini, S. (2020). The Association between Influenza Vaccination and the Risk of SARS-CoV-2 Infection, Severe Illness, and Death: A Systematic Review of the Literature. International Journal of Environmental Research and Public Health, 17(21), 7870. https://doi.org/10.3390/ijerph17217870









