Intimate Partner Violence: A Risk Factor for Gestational Diabetes
Abstract
:1. Introduction
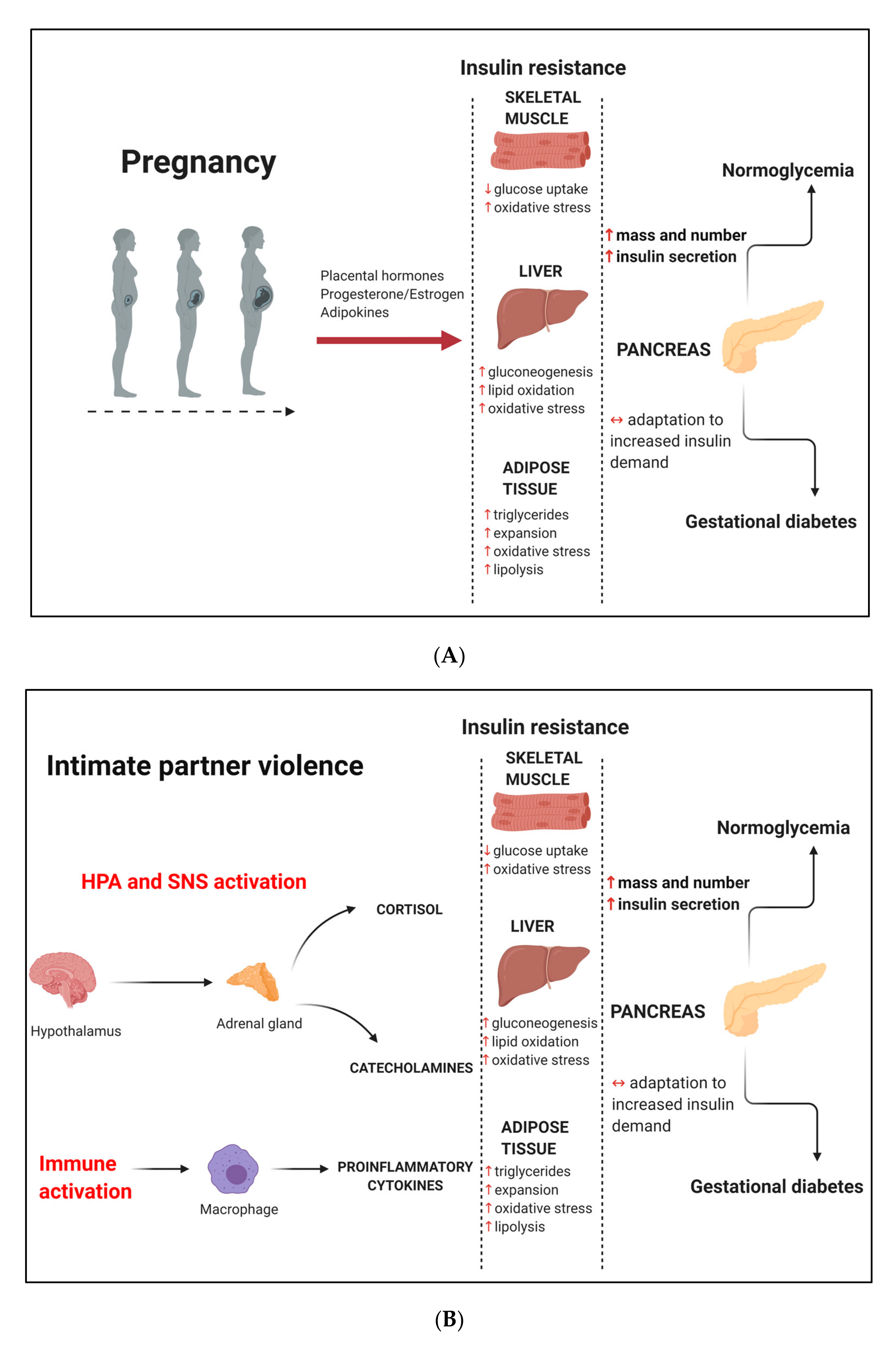
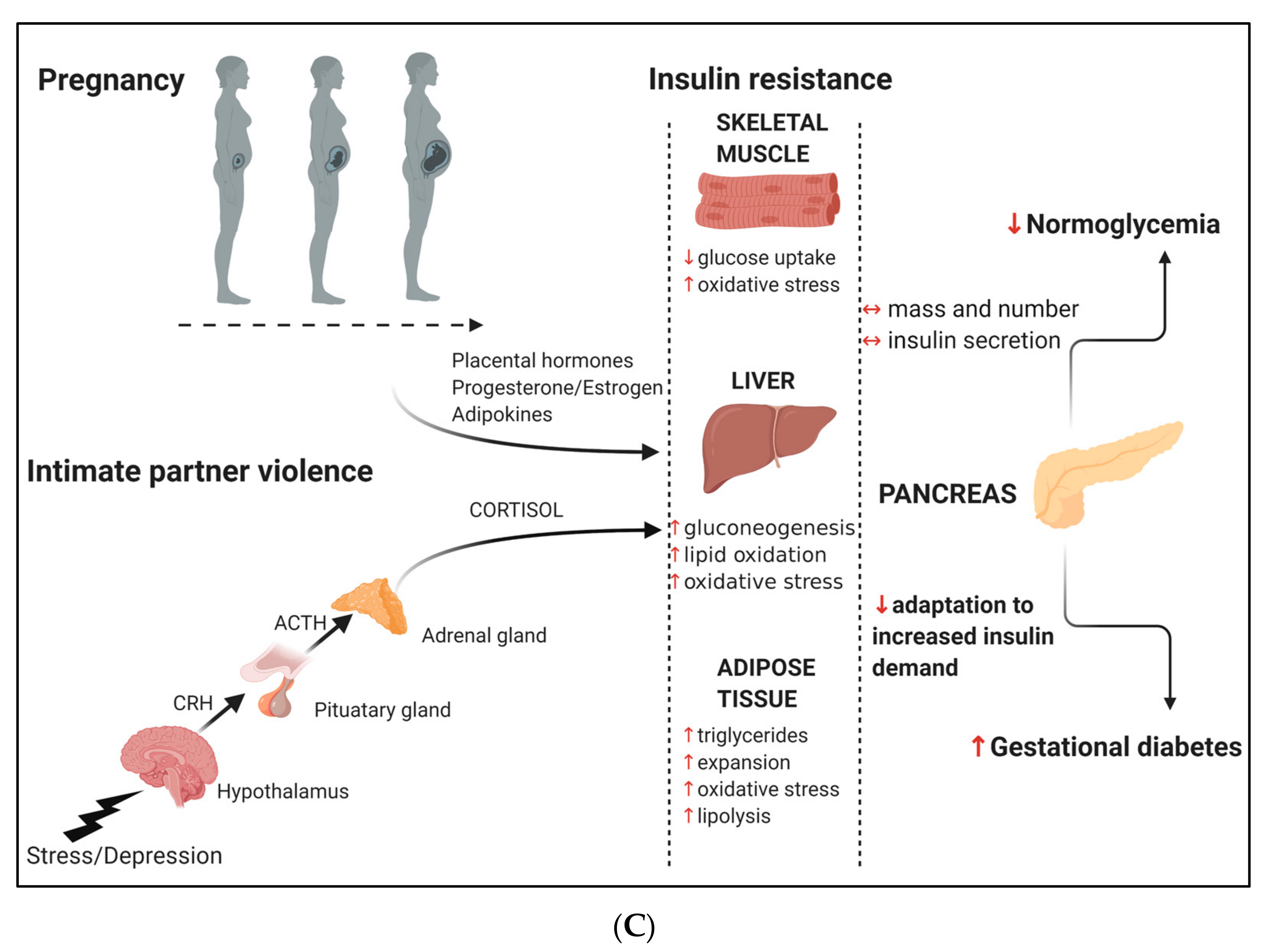
2. Pregnancy: An Insulin-Resistant State
3. Gestational Diabetes Mellitus
4. Intimate Partner Violence
5. Association between IPV and GDM
6. Screening for IPV as a Risk Factor for GDM
7. Limitations
8. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Hod, M.; Kapur, A.; Sacks, D.A.; Hadar, E.; Agarwal, M.; Di Renzo, G.C.; Cabero Roura, L.; McIntyre, H.D.; Morris, J.L.; Divakar, H. The International Federation of Gynecology and Obstetrics (FIGO) Initiative on gestational diabetes mellitus: A pragmatic guide for diagnosis, management, and care. Int. J. Gynaecol. Obstet. 2015, 131 (Suppl. 3), S173–S211. [Google Scholar] [CrossRef] [Green Version]
- International Diabetes Federation. IDF Diabetes Atlas, 7th ed.; International Diabetes Federation: Brussels, Belgium, 2017. [Google Scholar]
- Bellamy, L.; Casas, J.-P.; Hingorani, A.D.; Williams, D. Type 2 diabetes mellitus after gestational diabetes: A systematic review and meta-analysis. Lancet 2009, 373, 1773–1779. [Google Scholar] [CrossRef]
- Mitanchez, D.; Yzydorczyk, C.; Siddeek, B.; Boubred, F.; Benahmed, M.; Simeoni, U. The offspring of the diabetic mother--short- and long-term implications. Best Pract. Res. Clin. Obstet. Gynaecol. 2015, 29, 256–269. [Google Scholar] [CrossRef]
- Ratner, R.E. Prevention of Type 2 Diabetes in Women With Previous Gestational Diabetes. Diabetes Care 2007, 30, S242. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dias, S.; Pheiffer, C.; Rheeder, P.; Adam, S. Screening and diagnosis of gestational diabetes mellitus in South Africa: What we know so far. S. Afr. Med. J. 2019, 109, 457–462. [Google Scholar] [CrossRef]
- Devries, K.M.; Kishor, S.; Johnson, H.; Stöckl, H.; Bacchus, L.J.; Garcia-Moreno, C.; Watts, C. Intimate partner violence during pregnancy: Analysis of prevalence data from 19 countries. Reprod. Health Matters 2010, 18, 158–170. [Google Scholar] [CrossRef]
- Ribeiro, M.R.C.; da Silva, A.A.M.; e Alves, M.T.S.S. de B.; Batista, R.F.L.; de Rocha, L.M.L.N.; Schraiber, L.B.; Medeiros, N.L.; Costa, D.C.S.; Bettiol, H.; Barbieri, M.A. Psychological violence against pregnant women in a prenatal care cohort: Rates and associated factors in São Luís, Brazil. BMC Pregnancy Childbirth 2014, 14, 66. [Google Scholar] [CrossRef] [Green Version]
- Zar, H.J.; Pellowski, J.A.; Cohen, S.; Barnett, W.; Vanker, A.; Koen, N.; Stein, D.J. Maternal health and birth outcomes in a South African birth cohort study. PLoS ONE 2019, 14, e0222399. [Google Scholar] [CrossRef]
- Martin-de-las-Heras, S.; Velasco, C.; de Dios Luna-del-Castillo, J.; Khan, K.S. Maternal outcomes associated to psychological and physical intimate partner violence during pregnancy: A cohort study and multivariate analysis. PLoS ONE 2019, 14, e0218255. [Google Scholar] [CrossRef] [Green Version]
- Boeckel, M.G.; Viola, T.W.; Daruy-Filho, L.; Martinez, M.; Grassi-Oliveira, R. Intimate partner violence is associated with increased maternal hair cortisol in mother–child dyads. Compr. Psychiatry 2017, 72, 18–24. [Google Scholar] [CrossRef]
- Hammen, C. Stress and depression. Annu. Rev. Clin. Psychol. 2005, 1, 293–319. [Google Scholar] [CrossRef] [Green Version]
- Ghandour, R.M.; Campbell, J.C.; Lloyd, J. Screening and counseling for Intimate Partner Violence: A vision for the future. J. Womens Health (Larchmt.) 2015, 24, 57–61. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sonagra, A.D.; Biradar, S.M.; Dattatreya, K.; Jayaprakash Murthy , D.S. Normal pregnancy—A state of insulin resistance. J. Clin. Diagn. Res. 2014, 8, CC01–CC03. [Google Scholar] [CrossRef]
- Moyce, B.L.; Dolinsky, V.W. Maternal β-Cell Adaptations in Pregnancy and Placental Signalling: Implications for Gestational Diabetes. Int. J. Mol. Sci. 2018, 19, 3467. [Google Scholar] [CrossRef] [Green Version]
- Soma-Pillay, P.; Nelson-Piercy, C.; Tolppanen, H.; Mebazaa, A. Physiological changes in pregnancy. Cardiovasc. J. Afr. 2016, 27, 89–94. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sivan, E.; Homko, C.J.; Chen, X.; Reece, E.A.; Boden, G. Effect of insulin on fat metabolism during and after normal pregnancy. Diabetes 1999, 48, 834–838. [Google Scholar] [CrossRef]
- Wilcox, G. Insulin and insulin resistance. Clin. Biochem. Rev. 2005, 26, 19–39. [Google Scholar]
- Tripathy, D.; Chavez, A.O. Defects in insulin secretion and action in the pathogenesis of type 2 diabetes mellitus. Curr. Diab. Rep. 2010, 10, 184–191. [Google Scholar] [CrossRef]
- Zavalza-Gómez, A.B.; Anaya-Prado, R.; Rincón-Sánchez, A.R.; Mora-Martínez, J.M. Adipokines and insulin resistance during pregnancy. Diabetes Res. Clin. Pract. 2008, 80, 8–15. [Google Scholar] [CrossRef] [PubMed]
- Catalano, P.M.; Huston, L.; Amini, S.B.; Kalhan, S.C. Longitudinal changes in glucose metabolism during pregnancy in obese women with normal glucose tolerance and gestational diabetes mellitus. Am. J. Obstet. Gynecol. 1999, 180, 903–916. [Google Scholar] [CrossRef]
- Kahn, S.E.; Hull, R.L.; Utzschneider, K.M. Mechanisms linking obesity to insulin resistance and type 2 diabetes. Nature 2006, 444, 840–846. [Google Scholar] [CrossRef] [PubMed]
- Hardy, O.T.; Czech, M.P.; Corvera, S. What causes the insulin resistance underlying obesity? Curr. Opin. Endocrinol. Diabetes Obes. 2012, 19, 81–87. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- McIntyre, H.D.; Chang, A.M.; Callaway, L.K.; Cowley, D.M.; Dyer, A.R.; Radaelli, T.; Farrell, K.A.; Huston-Presley, L.; Amini, S.B.; Kirwan, J.P.; et al. Hormonal and Metabolic Factors Associated With Variations in Insulin Sensitivity in Human Pregnancy. Diabetes Care 2010, 33, 356. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Barbour, L.A.; McCurdy, C.E.; Hernandez, T.L.; Kirwan, J.P.; Catalano, P.M.; Friedman, J.E. Cellular mechanisms for insulin resistance in normal pregnancy and gestational diabetes. Diabetes Care 2007, 30 (Suppl. 2), S112–S119. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nien, J.K.; Mazaki-Tovi, S.; Romero, R.; Kusanovic, J.P.; Erez, O.; Gotsch, F.; Pineles, B.L.; Friel, L.A.; Espinoza, J.; Goncalves, L.; et al. Resistin: A hormone which induces insulin resistance is increased in normal pregnancy. J. Perinat. Med. 2007, 35, 513–521. [Google Scholar] [CrossRef] [Green Version]
- Kühl, C. Insulin secretion and insulin resistance in pregnancy and GDM. Implications for diagnosis and management. Diabetes 1991, 40 (Suppl. 2), 18–24. [Google Scholar] [CrossRef]
- Ryan, E.A.; O’Sullivan, M.J.; Skyler, J.S. Insulin action during pregnancy. Studies with the euglycemic clamp technique. Diabetes 1985, 34, 380–389. [Google Scholar] [CrossRef]
- Dias, S.; Adam, S.; Rheeder, P.; Pheiffer, C. Prevalence of and risk factors for gestational diabetes mellitus in South Africa. S. Afr. Med. J. 2019, 109, 463–467. [Google Scholar] [CrossRef]
- Kolu, P.; Raitanen, J.; Rissanen, P.; Luoto, R. Health care costs associated with gestational diabetes mellitus among high-risk women—Results from a randomised trial. BMC Pregnancy Childbirth 2012, 12, 71. [Google Scholar] [CrossRef] [Green Version]
- Chen, Y.; Quick, W.W.; Yang, W.; Zhang, Y.; Baldwin, A.; Moran, J.; Moore, V.; Sahai, N.; Dall, T.M. Cost of gestational diabetes mellitus in the United States in 2007. Popul. Health Manag. 2009, 12, 165–174. [Google Scholar] [CrossRef] [Green Version]
- Gentner, M.B.; Leppert, M.L.O. Environmental influences on health and development: Nutrition, substance exposure, and adverse childhood experiences. Dev. Med. Child Neurol. 2019, 61, 1008–1014. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- International Association of Diabetes and Pregnancy Study Groups Consensus Panel. International Association of Diabetes and Pregnancy Study Groups Recommendations on the Diagnosis and Classification of Hyperglycemia in Pregnancy. Diabetes Care 2010, 33, 676–682. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- HAPO Study Cooperative Research Group; Metzger, B.E.; Lowe, L.P.; Dyer, A.R.; Trimble, E.R.; Chaovarindr, U.; Coustan, D.R.; Hadden, D.R.; McCance, D.R.; Hod, M.; et al. Hyperglycemia and adverse pregnancy outcomes. N. Engl. J. Med. 2008, 358, 1991–2002. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Matta-Coelho, C.; Monteiro, A.M.; Fernandes, V.; Pereira, M.L.; Souto, S.B.; Portuguese Diabetes and Pregnancy Study Group. Universal vs. risk-factor-based screening for gestational diabetes—An analysis from a 5-Year Portuguese Cohort. Endocrine 2019, 63, 507–512. [Google Scholar] [CrossRef] [Green Version]
- Adam, S.; Rheeder, P. Screening for gestational diabetes mellitus in a South African population: Prevalence, comparison of diagnostic criteria and the role of risk factors. S. Afr. Med. J. 2017, 107, 523–527. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dias, S.; Pheiffer, C.; Abrahams, Y.; Rheeder, P.; Adam, S. Molecular Biomarkers for Gestational Diabetes Mellitus. Int. J. Mol. Sci. 2018, 19, 2926. [Google Scholar] [CrossRef] [Green Version]
- Schoenaker, D.A.J.M.; Callaway, L.K.; Mishra, G.D. The Role of Childhood Adversity in the Development of Gestational Diabetes. Am. J. Prev. Med. 2019, 57, 302–310. [Google Scholar] [CrossRef]
- Roberts, A.L.; Lyall, K.; Rich-Edwards, J.W.; Ascherio, A.; Weisskopf, M.G. Association of maternal exposure to childhood abuse with elevated risk for autism in offspring. JAMA Psychiatry 2013, 70, 508–515. [Google Scholar] [CrossRef] [Green Version]
- Mason, S.M.; Tobias, D.K.; Clark, C.J.; Zhang, C.; Hu, F.B.; Rich-Edwards, J.W. Abuse in Childhood or Adolescence and Gestational Diabetes: A Retrospective Cohort Study. Am. J. Prev. Med. 2016, 50, 436–444. [Google Scholar] [CrossRef] [Green Version]
- Devries, K.M.; Mak, J.Y.T.; García-Moreno, C.; Petzold, M.; Child, J.C.; Falder, G.; Lim, S.; Bacchus, L.J.; Engell, R.E.; Rosenfeld, L.; et al. Global health. The global prevalence of intimate partner violence against women. Science 2013, 340, 1527–1528. [Google Scholar] [CrossRef]
- Moran, P.S.; Wuytack, F.; Turner, M.; Normand, C.; Brown, S.; Begley, C.; Daly, D. Economic burden of maternal morbidity—A systematic review of cost-of-illness studies. PLoS ONE 2020, 15, e0227377. [Google Scholar] [CrossRef] [PubMed]
- Jewkes, R. Intimate partner violence: Causes and prevention. Lancet 2002, 359, 1423–1429. [Google Scholar] [CrossRef]
- Bohn, D.K.; Tebben, J.G.; Campbell, J.C. Influences of income, education, age, and ethnicity on physical abuse before and during pregnancy. J. Obstet. Gynecol. Neonatal. Nurs. 2004, 33, 561–571. [Google Scholar] [CrossRef] [PubMed]
- Gottlieb, A.S. Intimate partner violence: A clinical review of screening and intervention. Womens Health 2008, 4, 529–539. [Google Scholar] [CrossRef] [Green Version]
- McFarlane, J.; Campbell, J.C.; Sharps, P.; Watson, K. Abuse during pregnancy and femicide: Urgent implications for women’s health. Obstet. Gynecol. 2002, 100, 27–36. [Google Scholar] [CrossRef]
- García-Moreno, C.; Jansen, H.A.F.M.; Ellsberg, M.; Heise, L.; Watts, C. WHO Multi-country Study on Women’s Health and Domestic Violence against Women; World Health Organization: Geneva, Switzerland, 2005. [Google Scholar]
- Alhusen, J.L.; Ray, E.; Sharps, P.; Bullock, L. Intimate partner violence during pregnancy: Maternal and neonatal outcomes. J. Womens Health (Larchmt.) 2015, 24, 100–106. [Google Scholar] [CrossRef] [Green Version]
- James, L.; Brody, D.; Hamilton, Z. Risk factors for domestic violence during pregnancy: A meta-analytic review. Violence Vict. 2013, 28, 359–380. [Google Scholar] [CrossRef]
- Pastor-Moreno, G.; Ruiz-Pérez, I.; Henares-Montiel, J.; Escribà-Agüir, V.; Higueras-Callejón, C.; Ricci-Cabello, I. Intimate partner violence and perinatal health: A systematic review. BJOG 2020, 127, 537–547. [Google Scholar] [CrossRef]
- Mason, S.M.; Wright, R.J.; Hibert, E.N.; Spiegelman, D.; Forman, J.P.; Rich-Edwards, J.W. Intimate partner violence and incidence of hypertension in women. Ann. Epidemiol. 2012, 22, 562–567. [Google Scholar] [CrossRef] [Green Version]
- Mason, S.M.; Wright, R.J.; Hibert, E.N.; Spiegelman, D.; Jun, H.-J.; Hu, F.B.; Rich-Edwards, J.W. Intimate partner violence and incidence of type 2 diabetes in women. Diabetes Care 2013, 36, 1159–1165. [Google Scholar] [CrossRef] [Green Version]
- Chandan, J.S.; Thomas, T.; Bradbury-Jones, C.; Taylor, J.; Bandyopadhyay, S.; Nirantharakumar, K. Risk of Cardiometabolic Disease and All-Cause Mortality in Female Survivors of Domestic Abuse. J. Am. Heart Assoc. 2020, 9, e014580. [Google Scholar] [CrossRef] [PubMed]
- Pantell, M.S.; Prather, A.A.; Downing, J.M.; Gordon, N.P.; Adler, N.E. Association of Social and Behavioral Risk Factors With Earlier Onset of Adult Hypertension and Diabetes. JAMA Netw. Open 2019, 2, e193933. [Google Scholar] [CrossRef] [PubMed]
- Stene, L.E.; Jacobsen, G.W.; Dyb, G.; Tverdal, A.; Schei, B. Intimate partner violence and cardiovascular risk in women: A population-based cohort study. J. Womens Health (Larchmt.) 2013, 22, 250–258. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rosmond, R. Stress induced disturbances of the HPA axis: A pathway to Type 2 diabetes? Med. Sci. Monit. 2003, 9, RA35–RA39. [Google Scholar] [PubMed]
- Joseph, J.J.; Golden, S.H. Cortisol dysregulation: The bidirectional link between stress, depression, and type 2 diabetes mellitus. Ann. N. Y. Acad. Sci. 2017, 1391, 20–34. [Google Scholar] [CrossRef]
- Champaneri, S.; Wand, G.S.; Malhotra, S.S.; Casagrande, S.S.; Golden, S.H. Biological basis of depression in adults with diabetes. Curr. Diab. Rep. 2010, 10, 396–405. [Google Scholar] [CrossRef]
- Black, P.H. The inflammatory consequences of psychologic stress: Relationship to insulin resistance, obesity, atherosclerosis and diabetes mellitus, type II. Med. Hypotheses 2006, 67, 879–891. [Google Scholar] [CrossRef]
- Shoelson, S.E.; Lee, J.; Goldfine, A.B. Inflammation and insulin resistance. J. Clin. Investig. 2006, 116, 1793–1801. [Google Scholar] [CrossRef]
- McNelis, J.C.; Olefsky, J.M. Macrophages, immunity, and metabolic disease. Immunity 2014, 41, 36–48. [Google Scholar] [CrossRef] [Green Version]
- Ramamoorthy, S.; Cidlowski, J.A. Corticosteroids: Mechanisms of Action in Health and Disease. Rheum. Dis. Clin. N. Am. 2016, 42, 15–31. [Google Scholar] [CrossRef] [Green Version]
- Seedat, S.; Stein, M.B.; Kennedy, C.M.; Hauger, R.L. Plasma cortisol and neuropeptide Y in female victims of intimate partner violence. Psychoneuroendocrinology 2003, 28, 796–808. [Google Scholar] [CrossRef]
- Kiely, M.; El-Mohandes, A.A.E.; Gantz, M.G.; Chowdhury, D.; Thornberry, J.S.; El-Khorazaty, M.N. Understanding the association of biomedical, psychosocial and behavioral risks with adverse pregnancy outcomes among African-Americans in Washington, DC. Matern. Child Health J. 2011, 15 (Suppl. 1), S85–S95. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Leone, J.M.; Lane, S.D.; Koumans, E.H.; DeMott, K.; Wojtowycz, M.A.; Jensen, J.; Aubry, R.H. Effects of intimate partner violence on pregnancy trauma and placental abruption. J. Womens Health (Larchmt.) 2010, 19, 1501–1509. [Google Scholar] [CrossRef] [PubMed]
- Mogos, M.F.; Araya, W.N.; Masho, S.W.; Salemi, J.L.; Shieh, C.; Salihu, H.M. The Feto-Maternal Health Cost of Intimate Partner Violence Among Delivery-Related Discharges in the United States, 2002–2009. J. Interpers. Violence 2016, 31, 444–464. [Google Scholar] [CrossRef] [PubMed]
- Connelly, C.D.; Hazen, A.L.; Baker-Ericzén, M.J.; Landsverk, J.; Horwitz, S.M. Is screening for depression in the perinatal period enough? The co-occurrence of depression, substance abuse, and intimate partner violence in culturally diverse pregnant women. J. Womens Health (Larchmt.) 2013, 22, 844–852. [Google Scholar] [CrossRef] [Green Version]
- Silveira, M.L.; Whitcomb, B.W.; Pekow, P.; Braun, B.; Markenson, G.; Dole, N.; Manson, J.E.; Solomon, C.G.; Carbone, E.T.; Chasan-Taber, L. Perceived psychosocial stress and glucose intolerance among pregnant Hispanic women. Diabetes Metab. 2014, 40, 466–475. [Google Scholar] [CrossRef] [Green Version]
- Arafa, A.; Dong, J.-Y. Depression and risk of gestational diabetes: A meta-analysis of cohort studies. Diabetes Res. Clin. Pract. 2019, 156, 107826. [Google Scholar] [CrossRef]
- Dahlen, H.G.; Barnett, B.; Kohlhoff, J.; Drum, M.E.; Munoz, A.M.; Thornton, C. Obstetric and psychosocial risk factors for Australian-born and non-Australian born women and associated pregnancy and birth outcomes: A population based cohort study. BMC Pregnancy Childbirth 2015, 15, 292. [Google Scholar] [CrossRef]
- Dahlen, H.G.; Munoz, A.M.; Schmied, V.; Thornton, C. The relationship between intimate partner violence reported at the first antenatal booking visit and obstetric and perinatal outcomes in an ethnically diverse group of Australian pregnant women: A population-based study over 10 years. BMJ Open 2018, 8, e019566. [Google Scholar] [CrossRef]
- Wilson, B.L.; Dyer, J.M.; Latendresse, G.; Wong, B.; Baksh, L. Exploring the Psychosocial Predictors of Gestational Diabetes and Birth Weight. J. Obstet. Gynecol. Neonatal. Nurs. 2015, 44, 760–771. [Google Scholar] [CrossRef]
- Rabin, R.F.; Jennings, J.M.; Campbell, J.C.; Bair-Merritt, M.H. Intimate partner violence screening tools: A systematic review. Am. J. Prev. Med. 2009, 36, 439–445.e4. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Paladine, H.L.; Blenning, C.E.; Strangas, Y. Postpartum Care: An Approach to the Fourth Trimester. Am. Fam. Physician 2019, 100, 485–491. [Google Scholar]
- Ware, W.R. Psychological stress, insulin resistance, inflammation and the assessment of heart disease risk. Time for a paradigm shift? Med. Hypotheses 2008, 71, 45–52. [Google Scholar] [CrossRef] [PubMed]
- Salmon, D.; Baird, K.M.; White, P. Women’s views and experiences of antenatal enquiry for domestic abuse during pregnancy. Health Expect. 2015, 18, 867–878. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Campbell, J.C. Helping women understand their risk in situations of intimate partner violence. J. Interpers. Violence 2004, 19, 1464–1477. [Google Scholar] [CrossRef] [PubMed]
- Renker, P.R.; Tonkin, P. Women’s views of prenatal violence screening: Acceptability and confidentiality issues. Obstet. Gynecol. 2006, 107, 348–354. [Google Scholar] [CrossRef]
- Groves, A.K.; Moodley, D.; McNaughton-Reyes, L.; Martin, S.L.; Foshee, V.; Maman, S. Prevalence, rates and correlates of intimate partner violence among South African women during pregnancy and the postpartum period. Matern. Child Health J. 2015, 19, 487–495. [Google Scholar] [CrossRef]
- Moats, C.C.; Edwards, F.D.; Files, J.A. More than meets the eye: The importance of screening for intimate partner violence. J. Womens Health (Larchmt.) 2014, 23, 275–277. [Google Scholar] [CrossRef]
- Walton-Moss, B.J.; Manganello, J.; Frye, V.; Campbell, J.C. Risk factors for intimate partner violence and associated injury among urban women. J. Community Health 2005, 30, 377–389. [Google Scholar] [CrossRef]
- Taillieu, T.L.; Brownridge, D.A. Violence against pregnant women: Prevalence, patterns, risk factors, theories, and directions for future research. Aggress. Violent Behav. 2010, 15, 14–35. [Google Scholar] [CrossRef]
- Liang, Y.-Z.; Dong, J.; Zhang, J.; Wang, S.; He, Y.; Yan, Y.-X. Identification of Neuroendocrine Stress Response-Related Circulating MicroRNAs as Biomarkers for Type 2 Diabetes Mellitus and Insulin Resistance. Front. Endocrinol. 2018, 9, 132. [Google Scholar] [CrossRef] [PubMed]
- Alkhatib, A.; Tsang, C.; Tiss, A.; Bahorun, T.; Arefanian, H.; Barake, R.; Khadir, A.; Tuomilehto, J. Functional Foods and Lifestyle Approaches for Diabetes Prevention and Management. Nutrients 2017, 9, 1310. [Google Scholar] [CrossRef] [Green Version]
- Gilbert, L.; Gross, J.; Lanzi, S.; Quansah, D.Y.; Puder, J.; Horsch, A. How diet, physical activity and psychosocial well-being interact in women with gestational diabetes mellitus: An integrative review. BMC Pregnancy Childbirth 2019, 19, 60. [Google Scholar] [CrossRef]
- Nguyen-Ngo, C.; Willcox, J.C.; Lappas, M. Anti-Diabetic, Anti-Inflammatory, and Anti-Oxidant Effects of Naringenin in an In Vitro Human Model and an In Vivo Murine Model of Gestational Diabetes Mellitus. Mol. Nutr. Food Res. 2019, 63, e1900224. [Google Scholar] [CrossRef] [PubMed]
- Lu, X.; Wu, F.; Jiang, M.; Sun, X.; Tian, G. Curcumin ameliorates gestational diabetes in mice partly through activating AMPK. Pharm. Biol. 2019, 57, 250–254. [Google Scholar] [CrossRef] [Green Version]
- Tuomilehto, J.; Schwarz, P.E.H. Preventing Diabetes: Early Versus Late Preventive Interventions. Diabetes Care 2016, 39 (Suppl. 2), S115–S120. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| Author | Study Details | Country | Sample Size (n) | Outcome | Estimate (95% CI) |
|---|---|---|---|---|---|
| [51] | A prospective population-based cohort study conducted between 2001 and 2007 to investigate the association between IPV and hypertension. Risk was measured using hazard ratios. | USA | 51,434 | Hypertension Physical IPV Sexual IPV Psychological IPV | 1.1 (1.0;1.1) 1.0 (0.9;1.1) 1.2 (1.0;1.5) |
| [52] | A prospective population-based cohort study conducted between 2001 and 2007 to investigate the association between IPV and type 2 diabetes risk. Risk was measured using hazard ratios. | USA | 64,732 | Type 2 diabetes Physical IPV Psychological IPV | 1.1 (0.9–1.3) 1.6 (1.1–2.4) |
| [53] | A retrospective cohort study of primary care patients conducted between 1995–2017 to investigate the association between IPV and cardiometabolic risk. Risk was measured using incidence rate ratios. | UK | 18,547 | Cardiovascular disease Hypertension Type 2 diabetes mellitus All-cause mortality | 1.3 (1.1;1.6) 1.0 (0.9;1.1) 1.5 (1.3;1.8) 1.4 (1.2;1.7) |
| [54] | A prospective population-based cohort study conducted between 2005–2016 to investigate the association between IPV cardiometabolic risk. Risk was measured using hazard ratios. | USA | 18,133 | Type 2 diabetes Hypertension | 1.7 (1.1;2.5) 1.0 (0.6;1.6) |
| [55] | A prospective population-based cohort study conducted between 2000–2002 to investigate the association between IPV and cardiometabolic risk. Risk was measured using incidence rate ratios. | Norway | 5593 | Increased metabolic risk Abdominal obesity, low high-density lipoprotein cholesterol, and elevated triglycerides. Antihypertensive medication | 1.4 (1.1;1.7) |
| Author | Study Description | Country | Sample Size (n) | Outcome | Estimate(95% CI) |
|---|---|---|---|---|---|
| Sociodemographic, Behavioral and Psychosocial Risk Factors | |||||
| [10] | A population-based study to investigate the association between IPV and adverse pregnancy outcomes. | Spain | 779 | No association between IPV and GDM was observed. | NE |
| [30] | A systematic review investigating the economic burden of common health problems associated with pregnancy and childbirth. Of the 38 studies included in the review 16 focused on GDM and 1 on IPV | USA, Ireland, Finland, Sweden, Italy, Brazil, South Korea China (GDM) USA (IPV) | GDM 3,027,237 IPV 32,658,259 | Costs per case GDM IPV | €263-€13,680 €1,410 |
| [64] | Population-based prospective study of high-risk, urban, African American mothers to investigate the association between biomedical, psychosocial and behavioral risks and adverse pregnancy outcomes. | USA | 918 | Predictor of poor pregnancy outcome IPV GDM | 4th 17th |
| [65] | Population-based, retrospective cross-sectional study conducted to examine the association between IPV and pregnancy outcomes. Risk was measured using odds ratios. | USA | 2,873 | IPV Pregnancy trauma Placental abruption (odds ratio) GDM | 14.4 (8.1;25.9) 4.0 (1.5;10.5) NE |
| [70] | A population-based cross-sectional study to investigate the association between psychosocial risk factors and pregnancy and birth outcomes. Risk was measured using risk ratios. | Australia | 3,092 | Psychosocial issues IPV risk ratio GDM risk ratio | 7.3 (4.2;12.7) 1.9 (1.1;3.0) |
| [71] | A retrospective population-based study to investigate the association between IPV and pregnancy outcomes. | Australia | 33, 542 | GDM prevalence higher in women who reported IPV although statistically significant. | 9.4% vs. 8.6% |
| [72] | Retrospective cross-sectional study conducted to investigate the association between sociodemographic and behavioral factors and risk of developing GDM. | USA | 4,682 | No association between abuse and GDM was observed. | NE |
| [74] | A review describing the importance of postpartum care. | USA | NA | Recommendations towards postpartum care: GDM screening Biopsychosocial assessment (e.g., depression, IPV) | NA |
| Abuse during childhood or adolescence | |||||
| [38] | A population-based cohort study conducted between 1996-2015 to investigate the association between childhood adverse events and risk of developing GDM. Risk was measured using risk ratios. | Australia | 6,317 | GDM in women with preconception depressive symptoms. Three adverse childhood events Four adverse child events | 1.7 (1.0;3.0) 1.8 (1.0;3.0) |
| [39] | A population-based longitudinal cohort study of the Nurses’ Health Study II to determine whether childhood abuse is associated with risk of autism, and other pregnancy outcomes including GDM. | USA | 52,949 | GDM prevalence higher in women exposed to the highest level of abuse compared to women who were not exposed to abuse. | 5.3% vs. 2.7% |
| [40] | A retrospective cohort study conducted in the Nurses’ Health Study II to investigate whether childhood or adolescent abuse victimization is associated with a risk of developing GDM. Risk was measured using risk ratios. | USA | 45,550 | GDM Severe physical abuse Forced sexual activity | 1.4 (1.2;1.7) 1.3 (1.1;1.5) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pheiffer, C.; Dias, S.; Adam, S. Intimate Partner Violence: A Risk Factor for Gestational Diabetes. Int. J. Environ. Res. Public Health 2020, 17, 7843. https://doi.org/10.3390/ijerph17217843
Pheiffer C, Dias S, Adam S. Intimate Partner Violence: A Risk Factor for Gestational Diabetes. International Journal of Environmental Research and Public Health. 2020; 17(21):7843. https://doi.org/10.3390/ijerph17217843
Chicago/Turabian StylePheiffer, Carmen, Stephanie Dias, and Sumaiya Adam. 2020. "Intimate Partner Violence: A Risk Factor for Gestational Diabetes" International Journal of Environmental Research and Public Health 17, no. 21: 7843. https://doi.org/10.3390/ijerph17217843
APA StylePheiffer, C., Dias, S., & Adam, S. (2020). Intimate Partner Violence: A Risk Factor for Gestational Diabetes. International Journal of Environmental Research and Public Health, 17(21), 7843. https://doi.org/10.3390/ijerph17217843






