Interactions of Cortisol and Prolactin with Other Selected Menstrual Cycle Hormones Affecting the Chances of Conception in Infertile Women
Abstract
1. Introduction
2. Materials and Methods
- Assessment of the course of the menstrual cycle:
- ○
- Determination of hormone levels
- On the third day of the cycle-assessment of FSH, LH, AMH, TSH, PRL, COR;
- During ovulation, as determined using the ovulation test strips-assessment of E2, P, LH, COR;
- 7 days after ovulation-assessment of E2, P, COR;
- Determination of pregnancy: 2 weeks after ovulation-assessment of HCG.
- ○
- Ultrasound examination
- During ovulation: assessment of the amount and size of dominant follicles (larger or equal to 17 mm), assessment of endometrial thickness;
- Seven days after ovulation: assessment of follicular rupture and assessment of endometrial thickness.
- Collection of sociodemographic data.
3. Results
3.1. Characteristics of the Groups
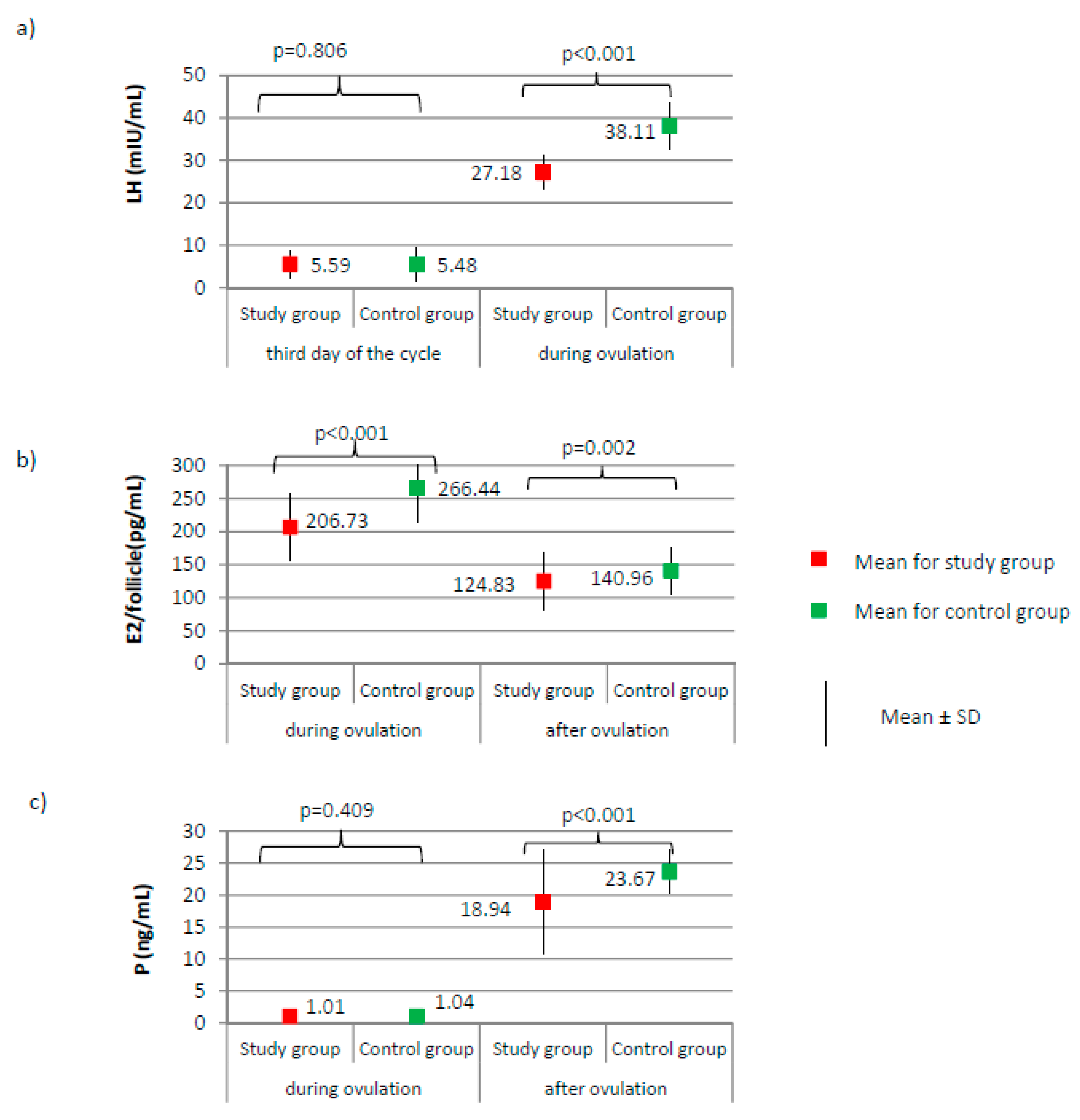
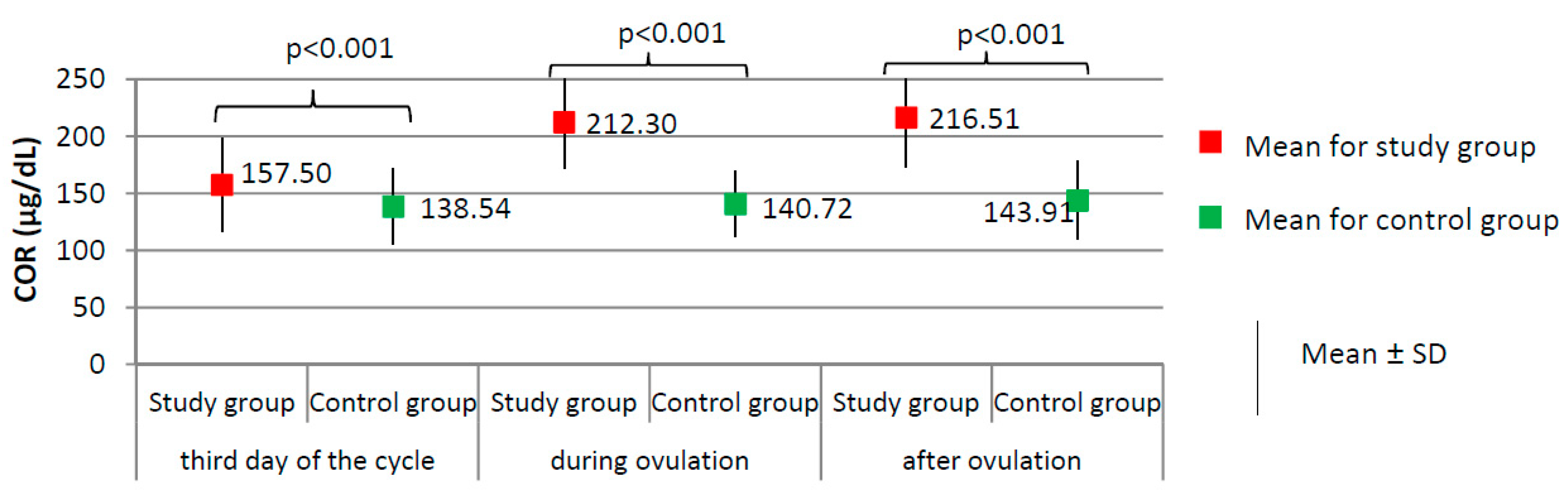
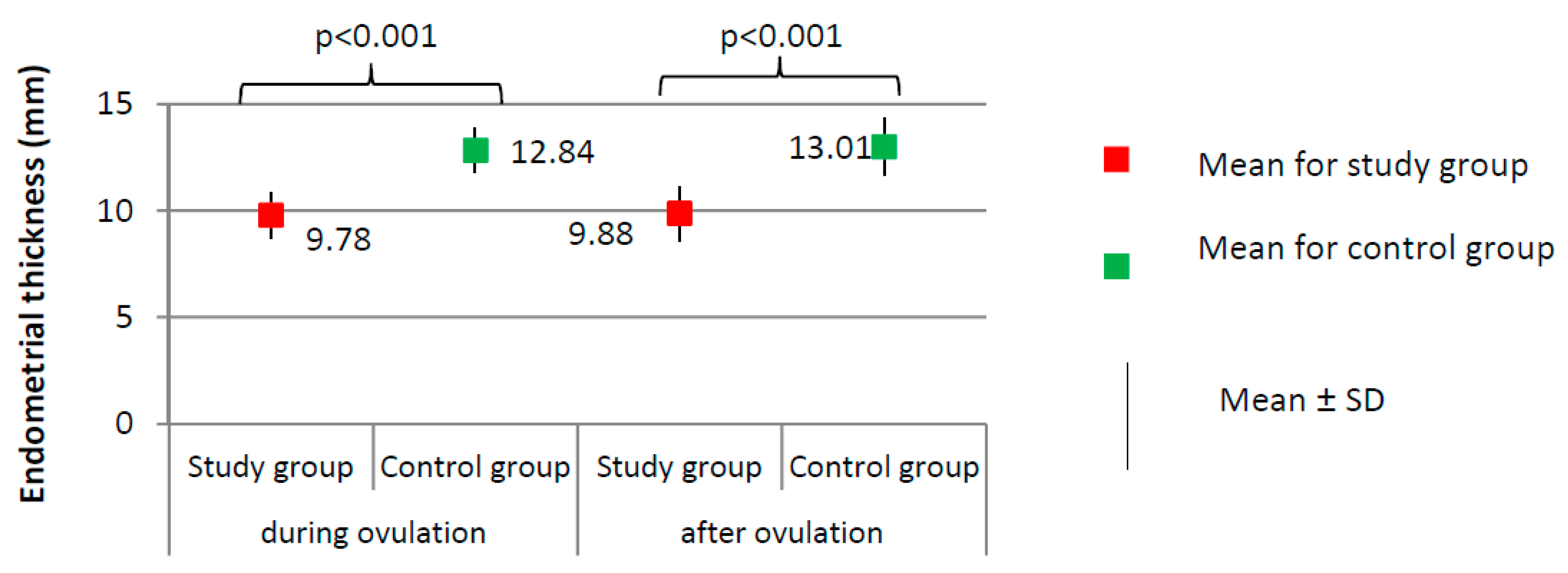
3.2. Comparison of the Menstrual Cycle Course between Study Group and Control Group
3.3. Correlations between Prolactin and Cortisol, and Menstrual Cycle Course in the Study Group and in the Control Group
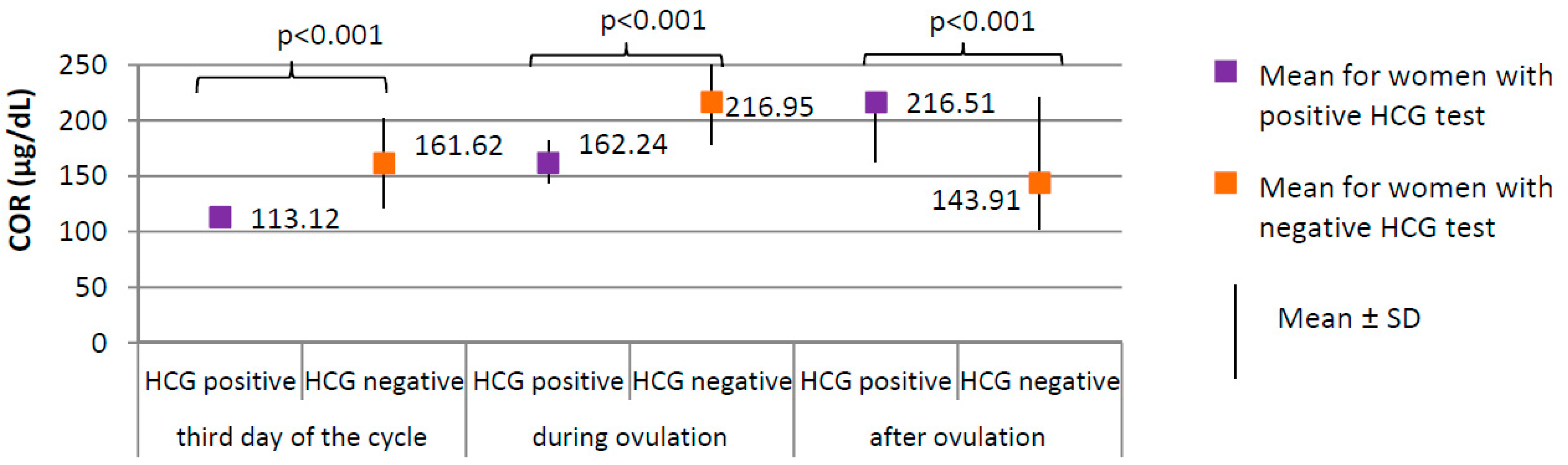
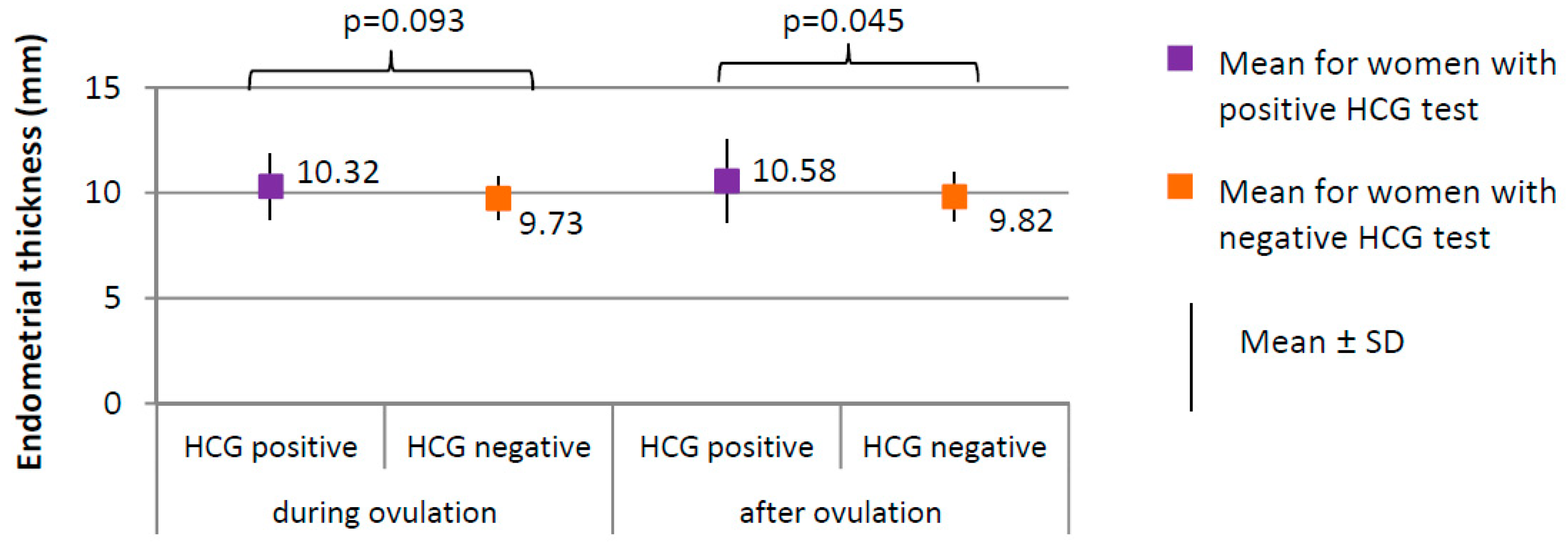
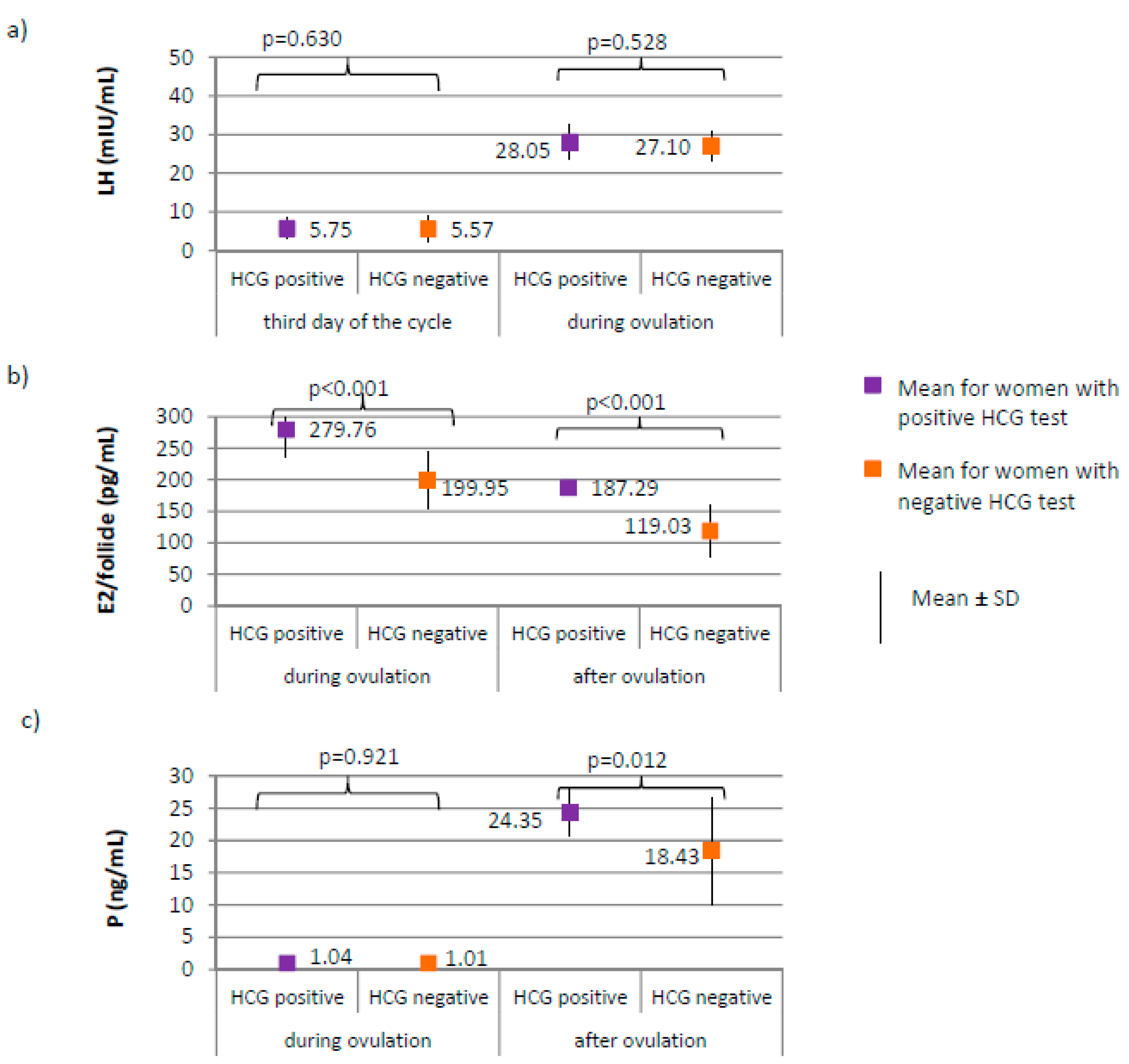
3.4. The Menstrual Cycle Course in the Study Group—Comparison between Women with Positive HCG Test and Women with Negative HCG Test
4. Discussions
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- World Health Organization. Meeting to Develop a Global Consensus on Preconception Care to Reduce Maternal and Childhood Mortality and Morbidity:World Health Organization Headquarters, Geneva, 6–7 February 2012: Meeting Report; World Health Organization: Geneva, Switzerland, 2013. [Google Scholar]
- Łukaszuk, K.; Kozioł, K.; Jakiel, G.; Jakimiuk, A.; Jędrzejczak, P.; Kuczyński, W.; Kurzawa, R.; Pawelczyk, L.; Radwan, M.; Spaczyński, R.; et al. Diagnostyka i Leczenie Niepłodności—Rekomendacje Polskiego Towarzystwa Medycyny Rozrodu i Embriologii (PTMRiE) Oraz Polskiego Towarzystwa Ginekologów i Położników (PTGP). Ginekol. Perinatol. Prakt. 2018, 3, 112–140. [Google Scholar]
- Sominsky, L.; Hodgson, D.M.; McLaughlin, E.A.; Smith, R.; Wall, H.M.; Spencer, S.J. Linking Stress and Infertility: A Novel Role for Ghrelin. Endocr. Rev. 2017, 38, 432–467. [Google Scholar] [CrossRef] [PubMed]
- Patel, S.S.; Bamigboye, V. Hyperprolactinaemia. J. Obstet. Gynaecol. 2007, 27, 455–459. [Google Scholar] [CrossRef] [PubMed]
- Stouffer, R.L.; Bishop, C.V.; Bogan, R.L.; Xu, F.; Hennebold, J.D. Endocrine and Local Control of the Primate Corpus Luteum. Reprod. Biol. 2013, 13, 259–271. [Google Scholar] [CrossRef] [PubMed]
- Filicori, M.; Butler, J.P.; Crowley, W.F. Neuroendocrine Regulation of the Corpus Luteum in the Human. Evidence for Pulsatile Progesterone Secretion. J. Clin. Investig. 1984, 73, 1639–1647. [Google Scholar] [CrossRef] [PubMed]
- Gaggiotti-Marre, S.; Martinez, F.; Coll, L.; Garcia, S.; Álvarez, M.; Parriego, M.; Barri, P.N.; Polyzos, N.; Coroleu, B. Low Serum Progesterone the Day Prior to Frozen Embryo Transfer of Euploid Embryos Is Associated with Significant Reduction in Live Birth Rates. Gynecol. Endocrinol. 2019, 35, 439–442. [Google Scholar] [CrossRef]
- Griesinger, G.; Trevisan, S.; Cometti, B. Endometrial Thickness on the Day of Embryo Transfer Is a Poor Predictor of IVF Treatment Outcome. Hum. Reprod. Open 2018, 1–8. [Google Scholar] [CrossRef]
- Wdowiak, A.; Bojar, I. Ovarian Stimulation with Human and Recombinant Gonadotropin—Comparison of in Vitro Fertilization Efficiency with Use of Time-Lapse Monitoring. Reprod. Health 2015, 12, 113. [Google Scholar] [CrossRef]
- Wdowiak, A.; Wdowiak, E.; Stec, M.; Bojar, I. Post-Laparoscopy Predictive Factors of Achieving Pregnancy in Patients Treated for Infertility. Videosurg. Other Miniinvasive Tech. 2016, 11, 253–258. [Google Scholar] [CrossRef]
- Younis, J.S. The Role of Progesterone/Estradiol Ratio in Exploring the Mechanism of Late Follicular Progesterone Elevation in Low Ovarian Reserve Women. Med. Hypotheses 2019, 125, 126–128. [Google Scholar] [CrossRef]
- Moretti, C.; Toscano, V. Dynamic Evaluation of Ovarian Reserve and Abnormal Androgen Excess in Women. J. Endocrinol. Investig. 2003, 26, 114–123. [Google Scholar]
- Maruo, T.; Hayashi, M.; Matsuo, H.; Yamamoto, T.; Okada, H.; Mochizuki, M. The Role of Thyroid Hormone as a Biological Amplifier of the Actions of Follicle-Stimulating Hormone in the Functional Differentiation of Cultured Porcine Granulosa Cells. Endocrinology 1987, 121, 123–1241. [Google Scholar] [CrossRef] [PubMed]
- Fujimoto, N.; Jinno, N.; Kitamura, S. Activation of Estrogen Response Element Dependent Transcription by Thyroid Hormone with Increase in Estrogen Receptor Levels in a Rat Pituitary Cell Line, GH3. J. Endocrinol. 2004, 181, 77–84. [Google Scholar] [CrossRef]
- Zegers-Hochschild, F.; Adamson, G.D.; de Mouzon, J.; Ishihara, O.; Mansour, R.; Nygren, K.; Sullivan, E.; Vanderpoel, S. International Committee for Monitoring Assisted Reproductive Technology (ICMART) and the World Health Organization (WHO) Revised Glossary of ART Terminology, 2009. Hum. Repord. 2009, 92, 1520–1524. [Google Scholar] [CrossRef]
- Cooper, T.G.; Noonan, E.; Von Eckardstein, S.; Auger, J.; Baker, H.W.G.; Behre, H.M.; Haugen, T.B.; Kruger, T.; Wang, C.; Mbizvo, M.T.; et al. World Health Organization Reference Values for Human Semen Characteristics. Hum. Reprod. Update 2010, 16, 231–245. [Google Scholar] [CrossRef]
- National Institute for Clinical Excellence. Fertility: Assessment & Treatment for People with Fertility Problems. NICE Clinical Guideline 11, February 2004; RCOG and National Collaborating Centre for Women’s and Children’s Health: London, UK, 2013. [Google Scholar]
- Babor, T.; Higgins-Biddle, J.C.; Saunders, J.B.; Monteiro, M.G. The Alcohol Use Disorders Identification Test: Guidelines for Use in Primary Care; World Health Organization: Geneva, Switzerland, 2001; WHO/MSD/MSB/01.6a. [Google Scholar]
- Csemiczky, G.; Landgren, B.M.; Collins, A. The Influence of Stress and State Anxiety on the Outcome of IVF-Treatment: Psychological and Endocrinological Assessment of Swedish Women Entering IVF-Treatment. Acta Obstet. Gynecol. Scand. 2000, 79, 113–118. [Google Scholar] [CrossRef]
- Cesta, C.E.; Johansson, A.L.V.; Hreinsson, J.; Rodriguez-Wallberg, K.A.; Olofsson, J.I.; Holte, J.; Wramsby, H.; Wramsby, M.; Cnattingius, S.; Skalkidou, A.; et al. A Prospective Investigation of Perceived Stress, Infertility-Related Stress, and Cortisol Levels in Women Undergoing in Vitro Fertilization: Influence on Embryo Quality and Clinical Pregnancy Rate. Acta Obstet. Gynecol. Scand. 2018, 97, 258–268. [Google Scholar] [CrossRef] [PubMed]
- Nouri, K.; Litschauer, B.; Huber, J.C.; Buerkle, B.; Tiringer, D.; Tempfer, C.B. Saliva Cortisol Levels and Subjective Stress Are Not Associated with Number of Oocytes after Controlled Ovarian Hyperstimulation in Patients Undergoing in Vitro Fertilization. Fertil. Steril. 2011, 96, 69–72. [Google Scholar] [CrossRef]
- An, Y.; Sun, Z.; Li, L.; Zhang, Y.; Ji, H. Relationship between Psychological Stress and Reproductive Outcome in Women Undergoing in Vitro Fertilization Treatment: Psychological and Neurohormonal Assessment. J. Assist. Reprod. Genet. 2013, 30, 35–41. [Google Scholar] [CrossRef]
- Nepomnaschy, P.A.; Welch, K.B.; McConnell, D.S.; Low, B.S.; Strassmann, B.I.; England, B.G. Cortisol Levels and Very Early Pregnancy Loss in Humans. Proc. Natl. Acad. Sci. USA 2006, 103, 3938–3942. [Google Scholar] [CrossRef]
- Zorn, J.V.; Schür, R.R.; Boks, M.P.; Kahn, R.S.; Joëls, M.; Vinkers, C.H. Cortisol Stress Reactivity across Psychiatric Disorders: A Systematic Review and Meta-Analysis. Psychoneuroendocrinology 2017, 77, 25–36. [Google Scholar] [CrossRef]
- Durlinger, A.L.L.; Gruijters, M.J.G.; Kramer, P.; Karels, B.; Ingraham, H.A.; Nachtigal, M.W.; Uilenbroek, J.T.J.; Anton Grootegoed, J.; Themmen, A.P.N. Anti-Müllerian Hormone Inhibits Initiation of Primordial Follicle Growth in the Mouse Ovary. Endocrinology 2002, 143, 1076–1084. [Google Scholar] [CrossRef] [PubMed]
- Josso, N.; Di Clemente, N.; Gouédard, L. Anti-Müllerian Hormone and Its Receptors. Mol. Cell. Endocrinol. 2001, 179, 25–32. [Google Scholar] [CrossRef]
- Lehmann, P.; Vélez, M.P.; Saumet, J.; Lapensée, L.; Jamal, W.; Bissonnette, F.; Phillips, S.; Kadoch, I.J. Anti-Müllerian Hormone (AMH): A Reliable Biomarker of Oocyte Quality in IVF. J. Assist. Reprod. Genet. 2014, 31, 493–498. [Google Scholar] [CrossRef] [PubMed]
- Hussain, M.; Cahill, D.; Akande, V.; Gordon, U. Discrepancies between Antimullerian Hormone and Follicle Stimulating Hormone in Assisted Reproduction. Obstet. Gynecol. Int. 2013, 2013, 1–6. [Google Scholar] [CrossRef]
- Reichman, D.E.; Goldschlag, D.; Rosenwaks, Z. Value of Antimüllerian Hormone as a Prognostic Indicator of in Vitro Fertilization Outcome. Fertil. Steril. 2014, 101, 1012–1018.e1. [Google Scholar] [CrossRef] [PubMed]
- Seckin, B.; Tokmak, A.; Yumusak, O.H. The Role of Anti-Müllerian Hormone in Prediction of Pregnancy in Young and Older Women with Unexplained Infertility Undergoing Intrauterine Insemination. J. Chin. Med. Assoc. 2019, 82, 300–304. [Google Scholar] [CrossRef]
- Casadei, L.; Manicuti, C.; Puca, F.; Madrigale, A.; Emidi, E.; Piccione, E. Can Anti-Müllerian Hormone Be Predictive of Spontaneous Onset of Pregnancy in Women with Unexplained Infertility. J. Obstet. Gynaecol. 2013, 33, 857–861. [Google Scholar] [CrossRef]
- Pacheco, A.; Cruz, M.; Iglesias, C.; García-Velasco, J.A. Very Low Anti-Müllerian Hormone Concentrations Are Not an Independent Predictor of Embryo Quality and Pregnancy Rate. Reprod. Biomed. Online 2018, 37, 113–119. [Google Scholar] [CrossRef]
- Ramachandran, A.; Jamdade, K.; Kumar, P.; Kumar Adiga, S.; Bhat, R.G.; Ferrao, S.R. Is There a Need for Luteinizing Hormone (LH) Estimation in Patients Undergoing Ovarian Stimulation with Gonadotropin-Releasing Hormone Antagonists and Recombinant Follicle-Stimulating Hormone? J. Clin. Diagn. Res. 2014, 8, 90–92. [Google Scholar] [CrossRef]
- Manno, M.; Cervi, M.; Zadro, D.; Fuggetta, G.; Adamo, V.; Tomei, F. Different ART Outcomes at Increasing Peak Estradiol Levels with Long and Antagonist Protocols: Retrospective Insights from Ten Years Experience. J. Assist. Reprod. Genet. 2011, 28, 693–698. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Muñoz, M.; Cruz, M.; Humaidan, P.; Garrido, N.; Pérez-Cano, I.; Meseguer, M. Dose of Recombinant FSH and Oestradiol Concentration on Day of HCG Affect Embryo Development Kinetics. Reprod. Biomed. Online 2012, 25, 382–389. [Google Scholar] [CrossRef] [PubMed]
- Murto, T.; Bjuresten, K.; Landgren, B.M.; Stavreus-Evers, A. Predictive Value of Hormonal Parameters for Live Birth in Women with Unexplained Infertility and Male Infertility. Reprod. Biol. Endocrinol. 2013, 11, 1–61. [Google Scholar] [CrossRef]
- Diedrich, K.; Fauser, B.C.J.M.; Devroey, P.; Griesinger, G. The Role of the Endometrium and Embryo in Human Implantation. Hum. Reprod. Update 2007, 13, 365–377. [Google Scholar] [CrossRef]
- Simón, C.; Martín, J.C.; Pellicer, A. Paracrine Regulators of Implantation. Best Pract. Res. Clin. Obstet. Gynaecol. 2000, 14, 815–826. [Google Scholar] [CrossRef]
- Michael, A.E.; Papageorghiou, A.T. Potential Significance of Physiological and Pharmacological Glucocorticoids in Early Pregnancy. Hum. Reprod. Update 2008, 14, 497–517. [Google Scholar] [CrossRef] [PubMed]
- Dinelli, L.; Courbière, B.; Achard, V.; Jouve, E.; Deveze, C.; Gnisci, A.; Grillo, J.M.; Paulmyer-Lacroix, O. Prognosis Factors of Pregnancy after Intrauterine Insemination with the Husband’s Sperm: Conclusions of an Analysis of 2019 Cycles. Fertil. Steril. 2014, 101, 994–1000. [Google Scholar] [CrossRef] [PubMed]
- Gao, G.; Cui, X.; Li, S.; Ding, P.; Zhang, S.; Zhang, Y. Endometrial Thickness and IVF Cycle Outcomes: A Meta-Analysis. Reprod. Biomed. Online 2020, 40, 124–133. [Google Scholar] [CrossRef]
- Prasad, S.; Tiwari, M.; Pandey, A.N.; Shrivastav, T.G.; Chaube, S.K. Impact of Stress on Oocyte Quality and Reproductive Outcome. J. Biomed. Sci. 2016, 23, 26. [Google Scholar] [CrossRef]
| Demographic Data | Study Group (N = 205) | Control Group (N = 100) | χ2 or t, p |
|---|---|---|---|
| Age—years M ± SD (min–max) | 26.7 ± 1.9 (23–30) | 26.8 ± 1.8 (23–30) | t = 2.266 p = 0.790 |
| Place of residence n (%) | |||
| Urban | 90 (45%) | 45 (45%) | χ2 = 0.000 |
| Rural | 115 (55%) | 55 (55%) | p = 1.000 |
| Education n (%) | |||
| Higher | 49 (24%) | 21 (21%) | χ2 = 2.790 |
| Secondary school | 78 (38%) | 31 (31%) | p = 0.248 |
| Primary school | 78 (38%) | 48 (48%) | |
| Type of occupation n (%) | |||
| Physical | 84 (41%) | 47 (47%) | χ2 = 1.029 |
| Intellectual | 82 (40%) | 35 (35%) | p = 0.598 |
| Mixed | 31 (19%) | 18 (18%) | |
| Number of hours of work weekly n (%) | |||
| 34–39 | 135 (66.5%) | 68 (68%) | χ2 = 1.951 |
| 40–44 | 33 (16%) | 11 (11%) | p = 0.583 |
| 45–49 | 17 (8%) | 8 (8%) | |
| 50–55 | 20 (9.5%) | 13 (13%) | |
| Family income n (%) | |||
| High | 46 (22.5%) | 30 (30%) | χ2 = 3.434 |
| Mediocre | 111 (54%) | 43 (43%) | p = 0.180 |
| Low | 48 (23.5%) | 27 (27%) | |
| Alcohol consumption n (%) | |||
| No | 191 (94%) | 64 (64%) | χ2 = 44.643 |
| Yes | 14 (6%) | 36 (36%) | p < 0.001 |
| Age of the spouse—years (exclusively study group) M ± SD (min–max) | 33.0 ± 5.7 (23–49) | - | - |
| Hormones | Menstrual Cycle Phase | PRL (ng/mL) | COR (µg/dL) | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Third Day of the Cycle | Third Day of the Cycle | Ovulation | After Ovulation | ||||||
| r | p | r | p | r | p | r | p | ||
| FSH (mIU/mL) | Third day of the cycle | 0.096 | 0.176 | 0.000 | 0.997 | 0.052 | 0.468 | 0.015 | 0.832 |
| LH (mIU/mL) | Third day of the cycle | 0.016 | 0.821 | −0.011 | 0.879 | 0.015 | 0.831 | 0.006 | 0.937 |
| Ovulation | −0.187 | 0.008 | −0.224 | 0.001 | −0.165 | 0.020 | −0.226 | 0.001 | |
| E2/follicle (pg/mL) | Ovulation | −0.569 | <0.001 | −0.857 | <0.001 | −0.820 | <0.001 | −0.882 | <0.001 |
| After ovulation | −0.670 | <0.001 | −0.890 | <0.001 | −0.884 | <0.001 | −0.899 | <0.001 | |
| P (ng/mL) | Ovulation | −0.001 | 0.996 | −0.033 | 0.641 | −0.008 | 0.910 | −0.035 | 0.620 |
| After ovulation | −0.705 | <0.001 | −0.840 | <0.001 | −0.765 | <0.001 | −0.823 | <0.001 | |
| Endometrial thickness (mm) | Ovulation | −0.259 | <0.001 | −0.246 | <0.001 | −0.333 | <0.001 | −0.321 | <0.001 |
| After ovulation | −0.268 | <0.001 | −0.269 | <0.001 | −0.357 | <0.001 | −0.349 | <0.001 | |
| Follicle size (cm) | Ovulation | −0.150 | 0.034 | −0.130 | 0.066 | −0.108 | 0.128 | −0.152 | 0.032 |
| Hormones | Menstrual Cycle Phase | PRL (ng/mL) | COR (µg/dL) | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Third Day of the Cycle | Third Day of the Cycle | Ovulation | After Ovulation | ||||||
| r | p | r | p | r | p | r | p | ||
| FSH (mIU/mL) | Third day of the cycle | 0.175 | 0.082 | −0.071 | 0.481 | −0.065 | 0.524 | −0.053 | 0.600 |
| LH (mIU/mL) | Third day of the cycle | −0.008 | 0.941 | 0.040 | 0.691 | 0.059 | 0.557 | 0.019 | 0.852 |
| Ovulation | −0.017 | 0.871 | −0.369 | <0.001 | −0.031 | 0.763 | −0.046 | 0.653 | |
| E2/follicle (pg/mL) | Ovulation | −0.117 | 0.245 | −0.125 | 0.216 | 0.019 | 0.851 | −0.041 | 0.683 |
| After ovulation | 0.227 | 0.023 | −0.520 | <0.001 | −0.118 | 0.241 | −0.141 | 0.161 | |
| P (ng/mL) | Ovulation | −0.018 | 0.860 | 0.027 | 0.792 | −0.034 | 0.737 | −0.046 | 0.652 |
| After ovulation | −0.024 | 0.814 | −0.109 | 0.282 | 0.004 | 0.969 | −0.003 | 0.974 | |
| Endometrial thickness (mm) | Ovulation | 0.159 | 0.115 | 0.164 | 0.104 | −0.045 | 0.659 | −0.015 | 0.882 |
| After ovulation | 0.177 | 0.078 | 0.093 | 0.358 | −0.066 | 0.512 | −0.033 | 0.746 | |
| Follicle size (cm) | Ovulation | −0.221 | 0.027 | 0.193 | 0.054 | −0.033 | 0.744 | −0.048 | 0.636 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wdowiak, A.; Raczkiewicz, D.; Janczyk, P.; Bojar, I.; Makara-Studzińska, M.; Wdowiak-Filip, A. Interactions of Cortisol and Prolactin with Other Selected Menstrual Cycle Hormones Affecting the Chances of Conception in Infertile Women. Int. J. Environ. Res. Public Health 2020, 17, 7537. https://doi.org/10.3390/ijerph17207537
Wdowiak A, Raczkiewicz D, Janczyk P, Bojar I, Makara-Studzińska M, Wdowiak-Filip A. Interactions of Cortisol and Prolactin with Other Selected Menstrual Cycle Hormones Affecting the Chances of Conception in Infertile Women. International Journal of Environmental Research and Public Health. 2020; 17(20):7537. https://doi.org/10.3390/ijerph17207537
Chicago/Turabian StyleWdowiak, Artur, Dorota Raczkiewicz, Paula Janczyk, Iwona Bojar, Marta Makara-Studzińska, and Anita Wdowiak-Filip. 2020. "Interactions of Cortisol and Prolactin with Other Selected Menstrual Cycle Hormones Affecting the Chances of Conception in Infertile Women" International Journal of Environmental Research and Public Health 17, no. 20: 7537. https://doi.org/10.3390/ijerph17207537
APA StyleWdowiak, A., Raczkiewicz, D., Janczyk, P., Bojar, I., Makara-Studzińska, M., & Wdowiak-Filip, A. (2020). Interactions of Cortisol and Prolactin with Other Selected Menstrual Cycle Hormones Affecting the Chances of Conception in Infertile Women. International Journal of Environmental Research and Public Health, 17(20), 7537. https://doi.org/10.3390/ijerph17207537







