Abstract
The Coronavirus disease 2019 (COVID-19) pandemic and its associated governmental recommendations and restrictions have influenced many aspects of human life, including exercise and mental health. This study aims to explore the influence of COVID-19 on exercise behavior and its impact on mood states, as well as predict changes in exercise behavior during a similar future pandemic in Taiwan. A cross-sectional online survey was conducted between 7 April and 13 May 2020 (n = 1114). Data on exercise behavior pre and during the pandemic and mood states were collected. A cumulative link model was used to predict changes in exercise frequency during a similar future pandemic by exercise frequency during the pandemic. A linear model was used to predict the influence of exercise frequency before and during the pandemic on mood states during the pandemic. A total of 71.2%, 67.3%, and 58.3% of respondents maintained their exercise intensity, frequency, and duration, respectively, during the pandemic. Frequent exercisers are more likely to maintain their exercise frequency during a similar pandemic (p < 0.001). Higher exercise frequencies during the pandemic were associated with better mood states (p < 0.05). Moreover, the effects of prepandemic exercise frequency on mood states are moderated by changes in exercise frequency during the pandemic (p < 0.05). Additionally, maintenance of exercise frequency during a pandemic specifically for frequent exercisers are recommended to preserve mood states. These results may provide evidence for health policies on exercise promotion and mental health before and during a future pandemic.
1. Introduction
Coronavirus disease 2019 (COVID-19), generated by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) [1], has spread rapidly and globally, with 12,552,765 confirmed cases and 561,617 deaths reported worldwide as of 12 July 2020 [2] after the first case reported in Wuhan, China in December 2019 [3]. This crisis, yet, has affected each country differently. Taiwan was anticipated to have the second-highest number of cases because of proximity and frequent travels to and from China [4]. However, learning from severe acute respiratory syndrome (SARS), Taiwan took speedy responses, proactive deployments, as well as novel strategies to identify and contain the COVID-19 [4,5,6,7], resulting in only 443 cases and seven deaths as of 7 June 2020 which is the official date Taiwan loosened COVID-19 measures. It is worth mentioning that Taiwan did not enforce any lockdown measures similar to other governments around the world.
The COVID-19 pandemic has led to numerous behavior changes such as social distancing, staying at home, avoiding crowds, or avoiding the usual venues of exercise such as gyms and fitness clubs [2,8]. Therefore, this pandemic has created a special situation for researchers to examine changes in exercise behavior. In Taiwan specifically, in order to suppress the transmission, recommendations included strict social distancing, self-isolation, quarantine, mandatory wearing of medical face masks in public, measuring body temperature at all entrances, and restricting the number of people in the entertainment venues [9], which may have created a novel and unique barrier to maintaining exercise behavior among Taiwanese.
In addition to this novel situation created by the pandemic, the physiological and psychological benefits of exercise are other important reasons to investigate exercise behavior changes at this time. Regular exercise of moderate intensity is positively linked to immune function [10] and reduction in respiratory symptoms [11], the dominant clinical manifestation of COVID-19 [12,13]. The beneficial effects on exercise may also lead to a lower COVID-19 mortality rate due to its effect on COVID-19 related cardiovascular complications (e.g., cardiovascular disease and myocardial injury) and its comorbidities (e.g., hypertension, type-2 diabetes, and obesity) [14], which are recognized as critical risk factors of COVID-19 [13,15]. Additionally, physical inactivity significantly elevates the risk of COVID-19 even after adjusting for age, sex, and other lifestyle factors (e.g., smoking and alcohol intake) [16]. Exercise is also linked to better mood states and better subjective well-being [17]. A large-scale nationwide study conducted in the United States has shown a negative association between exercise levels and self-reported mental health burdens such as stress, depression, and problems with emotions [18]. Additionally, exercise termination has been shown to have adverse consequences for mental health [19]. These studies provide indirect evidence for the potential role of exercise on mental health during this pandemic. This is specifically important as the COVID-19 pandemic is linked to many psychological adverse effects [20,21,22,23]. For example, the general population might become anxious and fearful due to the high transmission rate of COVID-19 [24]; healthcare providers might experience stress, anxiety, or insomnia due to the high susceptibility to COVID-19 [25]; those who have a positive diagnosis, might encounter discrimination or emotional isolation [26], and the survivors might develop anxiety, depression, and post-traumatic stress disorder [27].
It is evident that exploring exercise behavior changes and its potential consequences on mental health during the COVID-19 pandemic is essential. Additionally, how the COVID-19 pandemic and future similar pandemics affect exercise behavior and mental health in Taiwan remain unknown. This study aims to determine whether and how COVID-19 has affected exercise behavior and mental health status in Taiwan. Additionally, a model was established to predict the changes in exercise behavior during similar future pandemics, and potential moderators (e.g., age, education, and gender) were examined.
2. Methods
2.1. Study Design
The study used a cross-sectional design to investigate changes in exercise behavior and its relation to changes in mood states during the COVID-19 pandemic in Taiwan. This study is part of a larger project that was initiated in Potsdam Germany by the “International Research Group on COVID and exercise (International Research Group (IRG) on COVID and exercise)” and was translated into 18 languages. Overall, 16,353 respondents from 99 countries participated in this study (i.e., completed the questionnaire) [28]. This study is specifically the report of the results from Taiwan. Data were collected with an online survey using the Unipark™ web-based survey software. The study was conducted from April 7 to 13 May 2020, and the questionnaires were translated into traditional Chinese for use in Taiwan. General Data Protection Regulations (EU) and compliance with the APA Ethical Guidelines for Research were fulfilled and were also acknowledged by the participants. Ethical approval was waived for this study as the survey was completely anonymous.
2.2. Participants
Participants were recruited through social media, including Facebook, Line, Instagram, Twitter, as well as personal referrals. We did not use any screening questionnaire for underlying psychological conditions to exclude respondents from participating in the survey. No statistical method was used to predetermine the sample size and as many participants as possible were sampled during the time period of global lockdown. A post-hoc power analysis revealed that our sample (n = 1104) corresponds to the recommendation for sufficient power in linear models [29]. All participants completed an informed consent before participating in the survey. The participants did not receive any incentive for their participation. Additionally, they were able to skip any questions they did not want to answer or stop answering all questions at any point in time. We collected information on the presence of COVID-19 symptoms or a positive diagnosis to exclude these individuals from the analysis.
2.3. Variables
2.3.1. Demographics
Information on age, gender, educational level, income level, and living environment was collected. Income was measured with the question “compared with the average income in your country, which one is your household income?” For statistical analysis, the answers “I don’t have an income at the moment”, “very low income”, and “low income” were combined as “low income”, “high income”, and “very high income” were combined as “high income”, and “medium income” stayed the same.
In order to demonstrate the unique influence of COVID-19 on human behaviors in Taiwan, the presence of governmental pandemic control strategies (i.e., restrictions and recommendations) and the status of recreational facilities (e.g., gyms, clubs, and outdoor facilities) internationally were compared. Additionally, participants’ compliance with those regulations were presented.
2.3.2. Exercise Behavior
Exercise frequency during the COVID-19 pandemic was measured with one question on how often people exercised during the pandemic. We defined exercise for participants as any activity they chose to do as their exercise (e.g., workouts at home, running outside, etc...). Participants were also informed that any physical activity as part of their occupation must not be included unless they are a professional fitness coach or have a similar profession. For statistical analysis, the answers “never”, “once in a while” and “1 day per week” were combined as “1 day or less”, the answers “2 days per week” and “3 days per week” were combined as “2–3 days per week”, and “4–5 days per week”, and “6 days per week”, and “every day” answers were combined as “4 days per week or more”. Exercise frequency before the COVID-19 pandemic was measured and processed in the same format. Participants were also asked about their exercise intensity both during and prepandemic, and they could respond by choosing “low”, “moderate”, “high”, or “very high” intensity. The options “high” and “very high” intensities were combined as “high intensity” for the analysis. Participants were also asked about their exercise session length during the COVID-19 pandemic compared to prepandemic and could choose between “shorter”, “longer”, or “they were of about the same duration”.
2.3.3. Mood
Mood was measured with 16 items from the Profile of Mood States (POMS; [30]). The POMS is a heavily used psychometric questionnaire that measures general well-being in the clinical field both with the general population and people with chronic disease, as well as in sport and exercise psychology research [31]. In this study, participants were asked to report how they felt in the last few days during the pandemic.
For this study, we used the 16-item POMS from a German short screening version, which was psychometrically tested using data from a large national and representative sample [32]. The German items were then matched with the English originals as thoroughly as possible, and then translated from English into traditional Chinese by the research group in Taiwan. These 16 items allow subscores for “depression/anxiety”, “vigor”, “fatigue”, and “irritability”; however, we only used the total score in our analysis. The higher values on POMS indicate more positive mood states. In our study, the 16-item POMS total score achieved an internal consistency (reliability) of Cronbach’s α = 0.88. Mean total scores were calculated if at least 10 items of the scale were answered by the participants.
2.4. Statistical Analysis
Cumulative link models (CLM) were employed to analyze which variables were significant predictors of the exercise frequency during a similar pandemic condition. Three levels of exercise frequency per week during the pandemic (i.e., “1 day or less”, “2–3 days”, “4 or more days”) were predicted by three levels of exercise frequency per week prepandemic (i.e., “1 day or less”, “2–3 days”, “4 or more days”) with covariates such as age, gender, and income.
A linear model with a priori contrasts was used to analyze the influence of exercise behavior on mood states. This model was run with mood as the numerical response variable and “prepandemic exercise frequency” and “exercise frequency during the pandemic” as categorical predictors (with three levels of “1 day or less”, “2–3 days”, “4 or more days”). We specified a priori contrasts to compare mood scores of different levels of exercise frequency pre and during the COVID-19 pandemic, with exercising “1 day or less” as the intercept. The significance level was set to α = 0.05.
All analyses were performed with R software [33] and the ordinal package [34] for cumulative link models.
3. Results
3.1. Demographics
A total of 1174 participants filled out the questionnaire in Taiwan. We excluded participants who reported either presence of COVID-19 symptoms or diagnosis at the time of this study (n = 60). Therefore, a total of 1114 participants (mean (M)age = 35.90, standard deviation (SD)age = 15.16, 53.9% female) were included in the analysis. Descriptive statistics of the participants, including gender distributions, age, educational level, living environment, and income, are summarized in Table 1.

Table 1.
Participants’ demographic data (n = 1114).
Participants were also asked about the presence of social restrictions and recommendations and their level of compliance. Although participants reported almost no formal restrictions (92.4%), 81.8% reported that there were recommendations on social distancing, and the majority (86.1%) stated that they did their best to follow these recommendations. This is in contrast to the international data of the IRG on COVID and exercise project, indicating that the majority of participants (79.7%) reported the presence of strict formal restrictions by their governments.
Participants were also asked about the status of the recreational facilities (e.g., gyms, clubs, outdoor facilities, and parks) where they live. A total of 34.2% reported that the gyms and clubs were closed and only 9.5% reported that the outdoor facilities and parks were closed. This is in contrast with the international data indicating 91.1% reported closures of gyms and clubs, and 76.3% reported closures of outdoor facilities and parks. Table 2 provides more detail on this information in Taiwan and a comparison with the international data.

Table 2.
Participants’ report on governmental restrictions and recommendations, and the status of the recreational facilities in Taiwan vs. international data.
3.2. Exercise Behavior
3.2.1. Exercise Behavior Change during the COVID-19 Pandemic
The results show that 67.3% of participants reported the same exercise frequency, 19.7% reported a decrease in exercise frequency, and 12.9% reported an increase in exercise frequency during the COVID-19 pandemic compared to prepandemic.
Of those who exercised during the COVID-19 pandemic, 71.2% reported being physically active at similar, 16.7% at lighter, and 4.8% at higher exercise intensities. Additionally, 58.3% reported the same exercise duration, 23.3% reported shorter, and 11.1% reported longer exercise duration. This information is presented in Figure 1 in more detail.
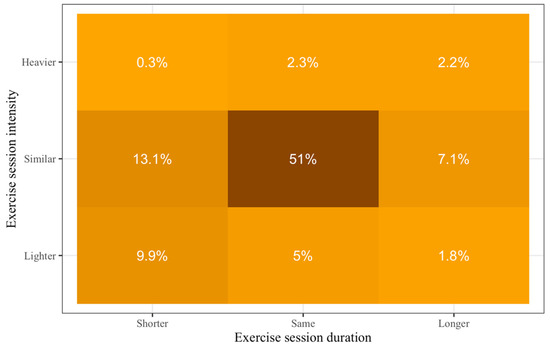
Figure 1.
Changes in exercise levels during the Coronavirus disease 2019 (COVID-19) pandemic compared to prepandemic. Darker colors show higher percentages.
3.2.2. Exercise Behavior Change Predictions
A CLM was employed to predict changes in exercise frequency during a pandemic from prepandemic exercise frequency for use in future conditions similar to the current pandemic. The results show that those who exercise 2–3 days per week before a similar pandemic have a significantly higher probability of maintaining their exercise frequency or do more during such pandemics compared to people who exercise one day or less before a similar pandemic (beta coefficient (b)pre2-1 = 1.95, p < 0.001). Those who exercise four days or more per week before a similar pandemic have a significantly higher probability of maintaining their exercise frequency or do more during such pandemics compared to those who exercise 2–3 days per week before a similar pandemic (bpre3-2 = 3.01, p < 0.001). Prepandemic exercise behavior could explain 56.4% of the variance in exercise behavior during a pandemic (R2Nagelkerke).
Calculating the category probabilities from the models’ prediction and location coefficients, we can see that the majority of the Taiwanese will maintain their prepandemic exercise frequency during similar pandemics (Figure 2). Specifically, the probabilities of maintaining exercise frequency during a similar pandemic for those who exercise one day or less per week, 2–3 days per week, and four days or more per week, are 73.3%, 56.8%, and 78.3%, respectively.
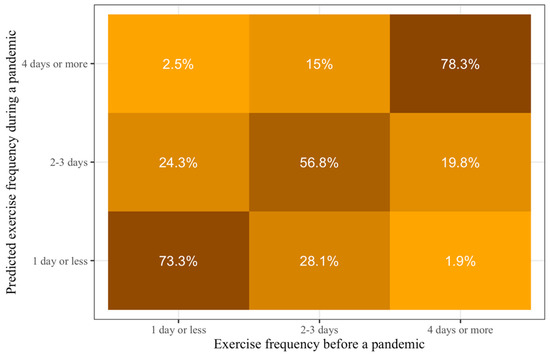
Figure 2.
Predictions of probabilities of exercise frequency during conditions similar to COVID-19 pandemic based on prepandemic exercise frequency. Lighter colors show smaller probabilities and darker colors show larger probabilities. The darkest colors are all on the diagonal from the bottom left to the top right, which means that people who exercise at a specific frequency before such pandemics would be most likely to exercise at the same frequency during it.
We also included gender, age, education, and income as covariates in separate models to predict exercise behavior during similar future pandemics. The results show that there was a main effect of age (bage = 0.01, p < 0.01) for exercise frequency during a pandemic. This means that older individuals are more likely to have higher exercise frequency during similar pandemics compared to younger individuals.
There were no significant main effects of gender or education. However, levels of these covariates showed significant interaction effects, meaning that the relationship between exercise behavior before a pandemic and exercise behavior during a pandemic is different for specific predictor levels. Specifically, females who exercise one day or less before a pandemic are more likely to stay inactive compared to others (bpre2-1*Female = 0.63, p = 0.03). Additionally, those who have “completed vocational school or college” and exercise one day or less before a pandemic are more likely to increase their exercise frequency during similar pandemics (bpre2-1*Education4 = –0.88, p < 0.01) compared to other respondents.
Finally, income was a significant predictor of exercise frequency during similar pandemics with those reporting a high level of income (compared to the average income level) being more likely to have higher exercise frequency compared to those with low levels of income (bIncomeHigh = 0.43, p = 0.03). However, when taking prepandemic exercise frequency into account, income is no longer a significant predictor of exercise frequency during a pandemic (bIncomeHigh = 0.23, p = 0.37).
Importantly, prepandemic exercise frequency remained a significant predictor of exercise frequency in all models when controlling for possible covariates. Complete statistical results can be seen in Table 3.

Table 3.
Change in exercise behavior. Statistical results for the cumulative link models (CLM) predicting exercise frequency during a pandemic with prepandemic exercise frequency and different covariates in separate models.
3.3. Exercise and Mood
In this analysis, the mood state was predicted by both prepandemic exercise frequency and exercise frequency during the COVID-19 pandemic. The results show that there was a significant main effect of exercise frequency during the pandemic on mood states. Those who exercised four days or more had significantly higher mood states compared to those who exercised for 2–3 days (bduring3-2 = 0.14, p = 0.04), and those exercised for 2–3 days had significantly higher mood states compared to those who exercised one day or less per week during the pandemic (bduring2-1 = 0.29, p < 0.001). There was also a significant main effect of prepandemic exercise frequency on mood states. Specifically, those who exercised four days or more per week prepandemic had a significantly lower mood state during the pandemic, compared to those who exercised for 2–3 days per week prepandemic (bpre3-2 = −0.16, p = 0.03). However, there was a significant interaction effect on exercise frequency levels during the pandemic × prepandemic exercise frequency levels on mood (bpre*during = −0.48–0.42, p = 0.01–0.03). Meaning, the effects of prepandemic exercise frequency on mood were dependent on exercise frequency during the pandemic (Figure 3). Table 4 summarizes the complete statistical results.
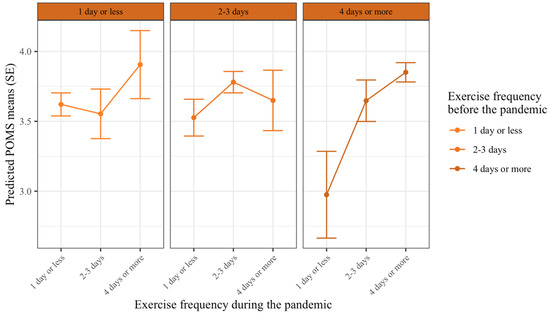
Figure 3.
The effect of exercise frequency during the COVID-19 pandemic on mood states depending on prepandemic exercise frequency. Lines indicate values for mood during the pandemic, (higher values are better mood states). Each column indicates exercise frequency before the pandemic, and exercise frequency levels within each column are exercise frequency levels during the pandemic. Error bars indicate 95% confidence intervals. There was a significant difference in mood for those who exercised 4 days or more before the pandemic (right columns) and decreased their exercise frequency during the pandemic. For those, who exercised for 2–3 days before the pandemic (middle column), only those who exercised 1 day or less reported significantly lower mood than those who maintained their exercise frequency.

Table 4.
Exercise frequency and change in mood states. Statistical results for the linear model analyzing mood states (POMS) with exercise frequency pre and during the pandemic.
Along with post-hoc contrasts (see Table 5), Figure 3 shows that those who exercised four days or more before the COVID-19 pandemic and decreased their exercise frequency during the pandemic experienced a decline in their mood states. Specifically, individuals who decreased their exercise frequency to 2–3 days per week, had significantly lower mood states than those who maintained their exercise frequency (bpre3: during2-3 = −0.20, p = 0.01), and if they decreased their exercise frequency to one day or less per week, they experienced even a more significant decline in mood states (bpre3: during1-3 = −0.88, p < 0.001).

Table 5.
Post-hoc contrasts comparing mood states with the pairwise test for different exercise frequency groups pre and during the pandemic.
Those who exercised for 2–3 days per week prepandemic and were able to maintain their exercise frequency, had higher mood states compared to those who decreased their exercise frequency to one day or less during the COVID-19 pandemic (bpre2:during1-2 = −0.25, p < 0.01). Exercise frequency during the COVID-19 pandemic had no significant effect on mood states for those who exercised one day or less prepandemic (p > 0.05). Overall, exercise behavior could explain 5.2% of the variability in mood states.
4. Discussion
The study presents the data from a larger study, “IRG on COVID and exercise”, to examine changes in exercise behavior and its relation to mood in Taiwan during the COVID-19 pandemic.
Our results showed that the majority of respondents were able to maintain their exercise behavior during this pandemic. Prediction analysis further revealed that Taiwanese are likely to maintain their prepandemic exercise frequency during future similar pandemics. Additionally, those who exercise more frequently before a similar pandemic have higher probabilities of maintaining their exercise frequency during such pandemics. Notably, individuals who “completed vocational school or college”, and are rarely active (i.e., exercise one day or less) before a pandemic are more likely to increase their exercise frequency during a future pandemic, while females who are rarely active (i.e., exercise one day or less) before a pandemic tend to maintain their exercise frequency during a future pandemic. Additionally, it seems that older individuals and those with higher levels of income are more likely to engage in higher exercise frequencies.
The relationship between exercise frequency and mood during the COVID-19 pandemic was dependent on the change in exercise behavior (before vs. during). In general, higher frequencies of exercise during the COVID-19 pandemic resulted in better mood states. Additionally, exercising “2–3 days” or “four days or more” before a pandemic was associated with worse mood states if these individuals reduced their exercise frequency during the pandemic.
4.1. Exercise Behavior before and during a Pandemic
Generally, a decline in exercise levels is expected during the COVID-19 pandemic and our study indicated that nearly 20% of individuals decreased their exercise frequency. However, our results are inconsistent with previous studies showing a dramatic decrease in physical activity during the COVID-19 pandemic in other parts of the world [8,23,35]; that is, the majority of Taiwanese maintained their exercise frequency, duration, or intensity during the COVID-19 pandemic. These differences may be partially the result of a relatively safe living environment and Taiwanese self-discipline in epidemic prevention. Given the geographical proximity to and the number of visits from mainland China, Taiwan Central Epidemic Control Center (CECC) of Taiwan Centers for Disease Control has been on constant alert about the epidemic in China. After the initial suspicious unknown acute respiratory syndrome case reported in December 2019 in China [3], Taiwan CECC has quickly mobilized and established comprehensive and proactive deployments to counteract and reduce the transmission of COVID-19. These included air and sea border control (e.g., assessing passengers for fever and pneumonia symptoms and restrictions at entries of international and cross-strait ports), case identification and containment (e.g., rapid screening tests for COVID-19, digital contact tracing, and quarantining suspicious cases), increase production rates and control of the domestic market price of medical face masks, and measuring body temperature at all entrances [4,5,6,7,36]. Furthermore, a high percentage of Taiwanese (86.1%) reported that they were willing to comply with Taiwan CECC derived public propaganda and school education for COVID-19 (e.g., maintaining social distance, regular disinfected living areas, and wearing medical face masks in public places) during the early days of the outbreak, which further decreased the spread of COVID-19. Therefore, Taiwan remained free from lockdown restrictions and kept the majority of health and fitness gyms and outdoor recreation facilities (e.g., parks, and playgrounds) open, and was able to maintain social activities per usual which reflect the successful containment of COVID-19.
The prediction analysis demonstrates that exercise frequency during the pandemic is dependent on prepandemic exercise frequency. Specifically, those who are more frequent exercisers prepandemic are more likely to stay active during a similar future pandemic. This finding reflects the importance of “prevention is better than cure” for exercise behavior during a future pandemic (e.g., second wave of the COVID-19 pandemic). Notably, some demographic variables might moderate exercise behavior. Specifically, older individuals are more likely to have higher exercise frequency during similar pandemics. The result is similar to other studies showing that older adults in Taiwan and mainland China exercise more regularly compared to younger individuals [35,37]. Additionally, our data show that inactive individuals who have “completed vocational school or college” are more likely to increase their activity levels compared to the rest of our sample. This is not surprising as generally, individuals with higher educational levels might have better financial resources [38] and more access to health information [39,40]. Finally, we observed that females who are rarely active are more likely to maintain their low levels of exercise frequency. A lower prevalence of physical activity among females compared to males has been previously reported [41,42]. It is plausible that females, in general, perceive more barriers to exercise, such as lack of time due to multitasking (e.g., requiring to take care of others while working) and therefore, they are less likely to change their exercise behavior during such pandemics [43,44]. Collectively, given that age, education, and gender can influence exercise behavior, specific strategies are required to consider and implement for these populations.
Interestingly, approximately 13% of participants reported an increase in exercise frequency and the prediction analysis showed that 26.8% of the individuals who exercise one day or less per week, and 15% of individuals who exercise 2–3 days per week, might increase their exercise frequencies during a future similar pandemic. This shows that inactive or rarely active individuals might increase their exercise frequency or adopt exercise behavior during a pandemic. Our finding is similar to data that showed increased health-seeking behavior (e.g., spending more time exercising) after the SARS epidemic in Hong Kong [45]. Additionally, the perceived severity and susceptibility to disease is associated with both increased likelihoods of conducting health-seeking behavior according to the Health Belief Model [46], and moving to the higher stages of the Transtheoretical Model of behavior change (e.g., stages of action and maintenance) [47]. That is, COVID-19 or a similar pandemic would increase awareness of health-seeking behavior such as exercise, and further facilitate the motivation to initiate and maintain the behavior. Therefore, public health policy-makers should not only consider strategies to encourage exercise before a pandemic, but also take this pandemic as an opportunity to promote exercise for future similar pandemics.
4.2. Exercise and Mood
We observed that exercise frequency both pre and during the COVID-19 pandemic impacts mood states. Generally, higher exercise frequency during COVID-19 was associated with better mood states, while the effect of prepandemic exercise frequency on mood states was moderated by the change in exercise frequency (before vs. during). Specifically, those who frequently exercised before the pandemic (i.e., exercised for 2–3 days or four days or more) experienced a significant decline in their mood states when they decreased their exercise frequency during the pandemic.
These results are in line with other research on the positive psychological benefits of exercise. For example, a meta-analytic study indicated an association between exercise and improved mood states, with an overall effect size of 0.24 and a mean change effect size of 0.38 for the treatment versus the control group and pretest-posttest studies, respectively [48]. Frequent exercise or physical activity might lead to adaptation of biological systems, including changes in neural hormones and endorphins [49], increasing density and efficiency of mineralocorticoid receptors and diminishing the cortisol synthesis [50], as well as improving cardiorespiratory fitness and strength [51]. Furthermore, exercise also elevates self-efficacy, self-esteem, feelings of mastery [51,52,53], and cognitive function [54,55,56]. These positive physiological and psychological changes might be the mechanisms for improvements in mood states associated with both exercise before and during the pandemic.
Notably, higher total POMS scores reflect either higher positive mood states (e.g., vigor) or less negative mood states (e.g., depression/anxiety, fatigue, or irritability). Research has shown that the ratings of the arousing emotional pictures were significantly decreased after an exercise session [57], suggesting exercise reduces anxiety and increases resilience toward emotional stressors. Additionally, alleviation of negative mood states [58], a decrease in trait anxiety [59,60], and a decrease in the emergence of depression [61] have also been observed with exercise. Based on the cross-stressor adaptation hypothesis [62], exercise might result in the adaptation of the sympathetic nervous system and the hypothalamus–pituitary–adrenal axis [49,63,64], which in turn lead to anxiolytic effects. This suggests that frequent exercisers might benefit from anxiolytic effects of exercise, especially during the COVID-19 pandemic [65,66,67].
It should be noted that prepandemic frequent exercisers (e.g., frequency of four days or more) experienced worse mood states if they decreased their exercise frequency during the pandemic. Decrease in exercise levels, known as detraining [68], can have adverse effects both on exercise-induced physiological adaptation [69,70], as well as psychological adaptations [71]. Other studies have shown that the cessation of regular exercise is linked to negative mood states [72,73], as well as increases in somatic depressive symptoms (e.g., fatigue, Berlin, Kop, and Deuster [72]). It is possible that a decrease in cardiorespiratory fitness [72], self-efficacy [51,74], or changes in obligatory exercise beliefs [75] contributed to declines in mood states when frequent exercisers decreased their exercise frequency during the COVID-19 pandemic. In sum, maintenance of exercise behavior patterns, particularly in the active population, is essential to preserve mood states during the COVID-19 pandemic or any similar future pandemics.
4.3. Limitation and Future Directions
Some drawbacks of the study should be acknowledged. The translated questionnaires used in this study were not validated due to time limitations. In order to conduct this study during the brief window of governmental recommendations and restrictions related to COVID-19, the questionnaires were translated from English into 18 different languages including traditional Chinese for use in Taiwan, resulting in the lack of time to proceed with the validation of the questionnaires as well as cultural adaptations. An additional limitation is the cross-sectional design of the study and the use of self-report data. However, the “IRG on COVID and exercise study” is planning to conduct a second wave after the cessation of the COVID-19 pandemic to investigate the trends of exercise behavior and mood states of the respondents over time. Finally, collecting data through the internet might potentially limit the participation of certain groups (e.g., individuals who do not have internet access). However, the internet penetration rate in Taiwan reached 89.6% in 2019, and the internet access rate for individuals aged 12 and above is as high as 88.8% [76], suggesting the high accessibility and usage of the internet by Taiwanese.
Our findings indicate that exercise behavior before a pandemic outbreak is likely to affect exercise behavior and mood states during a pandemic. Specifically, the results show that age, gender, and educational level played a role in exercise behavior change during the pandemic. Therefore, exercise professionals and policy-makers should work together to provide comprehensive and practical strategies specifically for the populations who are at risk of decreasing their exercise frequency before a similar pandemic in the future. For example, exercise training sessions can shift from in-person to online settings [77]. This shift might require exercise practitioners acquiring several new skills such as online communication strategies, online exercise program design, and online evaluation of the clients’ progress. Additionally, sending motivational messages through the internet, such as EdTech [78] and social media (e.g., Facebook, Wojcicki et al. [79]), could be incorporated to promote exercise participation.
5. Conclusions
Our study is the first study conducted to investigate the changes in exercise behavior and mood states during the COVID-19 pandemic in Taiwan. The majority of Taiwanese were able to maintain their exercise behavior with respect to frequency, intensity, and duration during the COVID-19 pandemic, and predictive data shows that they are also likely to maintain their exercise frequency during a future similar pandemic. Additionally, the mood state was affected by exercise frequency both during and before the pandemic, and it was negatively impacted if active individuals decreased their exercise frequency during the pandemic. This study thus provides evidence of the importance of promoting exercise both before and during a pandemic.
Author Contributions
Conceptualization, Y.-K.C., C.-L.H., S.T., S.N., and C.-H.C.; Methodology, S.T., and S.N.; Formal Analysis, S.T., and S.N.; Visualization, C.-L.H., S.T., and S.N.; Writing—Original Draft Preparation, All authors; Writing—Review & Editing, All authors. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by a grant from the Ministry of Science and Technology in Taiwan (107-2628-H-003-003-MY3), and the National Taiwan Normal University from the Higher Education Sprout Project by the Ministry of Education in Taiwan.
Acknowledgments
We thank Ralf Brand to initiate The International Research Group (IRG) on COVID and exercise.
Conflicts of Interest
The authors report no conflicts of interest.
Data Availability Statement
Data are available upon request.
References
- Del Rio, C.; Malani, P.N. COVID-19—New insights on a rapidly changing epidemic. JAMA 2020, 323, 1339–1340. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Coronavirus Disease (COVID-19) Pandemic. Available online: https://www.who.int/emergencies/diseases/novel-coronavirus-2019 (accessed on 14 July 2020).
- Zhu, N.; Zhang, D.; Wang, W.; Li, X.; Yang, B.; Song, J.; Zhao, X.; Huang, B.; Shi, W.; Lu, R.; et al. A novel coronavirus from patients with pneumonia in China, 2019. N Engl. J. Med. 2020, 382, 727–733. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.J.; Ng, C.Y.; Brook, R.H. Response to COVID-19 in Taiwan: Big data analytics, new technology, and proactive testing. JAMA 2020, 323, 1341–1342. [Google Scholar] [CrossRef] [PubMed]
- Steinbrook, R. Contact tracing, testing, and control of COVID-19-kearning from Taiwan. JAMA Intern. Med. 2020. [Google Scholar] [CrossRef] [PubMed]
- Cheng, H.Y.; Li, S.Y.; Yang, C.H. Initial rapid and proactive response for the COVID-19 outbreak—Taiwan’s experience. J. Formos. Med. Assoc. 2020, 119, 771–773. [Google Scholar] [CrossRef]
- Lee, T.L. Legal preparedness as part of COVID-19 response: The first 100 days in Taiwan. BMJ Glob. Health 2020, 5, e002608. [Google Scholar] [CrossRef]
- Xiang, M.; Zhang, Z.; Kuwahara, K. Impact of COVID-19 pandemic on children and adolescents’ lifestyle behavior larger than expected. Prog. Cardiovasc. Dis. 2020. [Google Scholar] [CrossRef]
- Chiu, W.T.; Laporte, R.P.; Wu, J. Determinants of Taiwan’s early containment of COVID-19 incidence. Am. J. Public Health 2020, 110, 943–944. [Google Scholar] [CrossRef]
- Simpson, R.J.; Campbell, J.P.; Gleeson, M.; Krüger, K.; Nieman, D.C.; Pyne, D.B.; Turner, J.E.; Walsh, N.P. Can exercise affect immune function to increase susceptibility to infection? Exerc. Immunol. Rev. 2020, 26, 8–22. [Google Scholar]
- Nieman, D.C.; Wentz, L.M. The compelling link between physical activity and the body’s defense system. J. Sport Health Sci. 2019, 8, 201–217. [Google Scholar] [CrossRef]
- Xu, Z.; Shi, L.; Wang, Y.; Zhang, J.; Huang, L.; Zhang, C.; Liu, S.; Zhao, P.; Liu, H.; Zhu, L. Pathological findings of COVID-19 associated with acute respiratory distress syndrome. Lancet Respir. Med. 2020, 8, 420–422. [Google Scholar] [CrossRef]
- Nieman, D.C. Coronavirus disease-2019: A tocsin to our aging, unfit, corpulent, and immunodeficient society. J. Sport Health Sci. 2020, 9, 293–301. [Google Scholar] [CrossRef] [PubMed]
- Piercy, K.L.; Troiano, R.P.; Ballard, R.M.; Carlson, S.A.; Fulton, J.E.; Galuska, D.A.; George, S.M.; Olson, R.D. The Physical Activity Guidelines for Americans. JAMA 2018, 320, 2020–2028. [Google Scholar] [CrossRef] [PubMed]
- Guo, T.; Fan, Y.; Chen, M.; Wu, X.; Zhang, L.; He, T.; Wang, H.; Wan, J.; Wang, X.; Lu, Z. Cardiovascular implications of fatal outcomes of patients with coronavirus disease 2019 (COVID-19). JAMA Cardiol. 2020. [Google Scholar] [CrossRef]
- Hamer, M.; Kivimaki, M.; Gale, C.R.; Batty, G.D. Lifestyle risk factors, inflammatory mechanisms, and COVID-19 hospitalization: A community-based cohort study of 387,109 adults in UK. Brain Behav. Immun. 2020, 87, 184–187. [Google Scholar] [CrossRef]
- Seino, S.; Kitamura, A.; Tomine, Y.; Tanaka, I.; Nishi, M.; Taniguchi, Y.U.; Yokoyama, Y.; Amano, H.; Fujiwara, Y.; Shinkai, S. Exercise arrangement is associated with physical and mental health in older adults. Med. Sci. Sports Exerc. 2019, 51, 1146–1153. [Google Scholar] [CrossRef]
- Chekroud, S.R.; Gueorguieva, R.; Zheutlin, A.B.; Paulus, M.; Krumholz, H.M.; Krystal, J.H.; Chekroud, A.M. Association between physical exercise and mental health in 1·2 million individuals in the USA between 2011 and 2015: A cross-sectional study. Lancet Psychiatry 2018, 5, 739–746. [Google Scholar] [CrossRef]
- Weinstein, A.A.; Koehmstedt, C.; Kop, W.J. Mental health consequences of exercise withdrawal: A systematic review. Gen. Hosp. Psychiatry 2017, 49, 11–18. [Google Scholar] [CrossRef] [PubMed]
- The Lancet Psychiatry. Mental health and COVID-19: Change the conversation. Lancet Psychiatry 2020, 7, 463. [Google Scholar] [CrossRef]
- Wang, C.; Pan, R.; Wan, X.; Tan, Y.; Xu, L.; McIntyre, R.S.; Choo, F.N.; Tran, B.; Ho, R.; Sharma, V.K.; et al. A longitudinal study on the mental health of general population during the COVID-19 epidemic in China. Brain Behav. Immun. 2020, 87, 40–48. [Google Scholar] [CrossRef] [PubMed]
- Tang, F.; Liang, J.; Zhang, H.; Kelifa, M.; He, Q.; Wang, P. COVID-19 related depression and anxiety among quarantined respondents. Psychol. Health 2020, 1–15. [Google Scholar] [CrossRef]
- Qi, M.; Li, P.; Moyle, W.; Weeks, B.; Jones, C. Physical activity, health-related quality of life, and stress among the Chinese adult population during the COVID-19 pandemic. Int. J. Environ. Res. Public Health 2020, 17, 6494. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Pan, R.; Wan, X.; Tan, Y.; Xu, L.; Ho, C.S.; Ho, R.C. Immediate psychological responses and associated factors during the initial stage of the 2019 Coronavirus Disease (COVID-19) epidemic among the general population in China. Int. J. Environ. Res. Public Health 2020, 17, 1729. [Google Scholar] [CrossRef] [PubMed]
- Spoorthy, M.S.; Pratapa, S.K.; Mahant, S. Mental health problems faced by healthcare workers due to the COVID-19 pandemic-A review. Asian J. Psychiatr. 2020, 51, 102119. [Google Scholar] [CrossRef] [PubMed]
- Zhai, Y.; Du, X. Mental health care for international Chinese students affected by the COVID-19 outbreak. Lancet Psychiatry 2020, 7, e22. [Google Scholar] [CrossRef]
- Pfefferbaum, B.; North, C.S. Mental health and the Covid-19 pandemic. N. Engl. J. Med. 2020. [Google Scholar] [CrossRef]
- Brand, R.; Timme, S.; Nosrat, S. When pandemic hits: Exercise frequency and subjective well-being during COVID-19 pandemic. Front. Psychol. 2020, 11. [Google Scholar] [CrossRef]
- Faul, F.; Erdfelder, E.; Buchner, A.; Lang, A. Statistical power analyses using G*Power 3.1: Tests for correlation and regression analyses. Behav Res Methods 2009, 41, 1149–1160. [Google Scholar] [CrossRef]
- McNair, D.M.; Lorr, M.; Droppleman, L.F. Manual for the Profile of Mood States; Educational and Industrial Testing Services: San Diego, CA, USA, 1971. [Google Scholar]
- Leunes, A.; Burger, J. Profile of mood states research in sport and exercise psychology: Past, present, and future. J. Appl. Sport Psychol. 2000, 12, 5–15. [Google Scholar] [CrossRef]
- Petrowski, K.; Schmalbach, B.; Albani, C.; Beutel, M.E.; Brähler, E.; Zenger, M. Revised short screening version of the Profile of Mood States (POMS) from the German general population. 2020. under review. [Google Scholar]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2019. [Google Scholar]
- Christensen, R.H.B. Cumulative link models for ordinal regression with the R package ordinal. J. Stat. Softw. 2018. [Google Scholar]
- Qin, F.; Song, Y.; Nassis, G.P.; Zhao, L.; Cui, S.; Lai, L.; Wu, Z.; Xu, M.; Qu, C.; Dong, Y.; et al. Prevalence of insufficient physical activity, sedentary screen time and emotional well-being during the early days of the 2019 novel coronavirus (COVID-19) outbreak in China: A national cross-sectional study. SSRN Electron. J. 2020. [Google Scholar] [CrossRef]
- Ministry of Health and Welfare Address. Measures of COVID-19; Ministry of Health and Welfare: Taipei, Taiwan, 2020.
- Chiang, C.; Chiou, S.; Liao, Y.; Liou, Y. The perceived neighborhood environment is associated with health-enhancing physical activity among adults: A cross-sectional survey of 13 townships in Taiwan. BMC Public Health 2019, 19, 524. [Google Scholar] [CrossRef]
- Schneeweis, N.; Skirbekk, V.; Winter-Ebmer, R. Does education improve cognitive performance four decades after school completion? Demography 2014, 51, 619–643. [Google Scholar] [CrossRef] [PubMed]
- Muller, A. Education, income inequality, and mortality: A multiple regression analysis. BMJ 2002, 324, 23–25. [Google Scholar] [CrossRef]
- Brunello, G.; Fort, M.; Schneeweis, N.; Winter-Ebmer, R. The causal effect of education on health: What is the role of health behaviors? Health Econ. 2016, 25, 314–336. [Google Scholar] [CrossRef] [PubMed]
- Bauman, A.; Bull, F.; Chey, T.; Craig, C.L.; Ainsworth, B.E.; Sallis, J.F.; Bowles, H.R.; Hagstromer, M.; Sjostrom, M.; Pratt, M.; et al. The international prevalence study on physical activity: Results from 20 countries. Int. J. Behav. Nutr. Phys. Act. 2009, 6, 21. [Google Scholar] [CrossRef]
- Guthold, R.; Stevens, G.A.; Riley, L.M.; Bull, F.C. Worldwide trends in insufficient physical activity from 2001 to 2016: A pooled analysis of 358 population-based surveys with 1.9 million participants. Lancet Glob. Health 2018, 6, e1077–e1086. [Google Scholar] [CrossRef]
- Bauman, A.E.; Reis, R.S.; Sallis, J.F.; Wells, J.C.; Loos, R.J.F.; Martin, B.W. Correlates of physical activity: Why are some people physically active and others not? Lancet 2012, 380, 258–271. [Google Scholar] [CrossRef]
- Salmon, J.; Owen, N.; Crawford, D.; Bauman, A.; Sallis, J. Physical activity and sedentary behavior: A population-based study of barriers, enjoyment, and preference. Health Psychol. 2003, 22, 178–188. [Google Scholar] [CrossRef]
- Lau, J.T.F.; Yang, X.; Tsui, H.Y.; Kim, J.H. Impacts of SARS on health-seeking behaviors in general population in Hong Kong. Prev. Med. 2005, 41, 454–462. [Google Scholar] [CrossRef] [PubMed]
- Champion, V.L.; Skinner, C.S. The health belief model. In Health Behavior and Health Education: Theory, Research, and Practice, 4th ed.; Jossey-Bass: San Francisco, CA, USA, 2008; pp. 45–65. [Google Scholar]
- Juniper, K.C.; Oman, R.F.; Hamm, R.M.; Kerby, D.S. The relationships among constructs in the health belief model and the transtheoretical model among African-American college women for physical activity. Am. J. Health Promot. 2004, 18, 354–357. [Google Scholar] [CrossRef] [PubMed]
- Arent, S.M.; Landers, D.M.; Etnier, J.L. The effects of exercise on mood in older adults: A meta-analytic review. J. Aging Phys. Act. 2000, 8, 407–430. [Google Scholar] [CrossRef]
- Heijnen, S.; Hommel, B.; Kibele, A.; Colzato, L.S. Neuromodulation of aerobic exercise—A review. Front. Psychol. 2016, 6, 1890. [Google Scholar] [CrossRef] [PubMed]
- Forcier, K.; Stroud, L.R.; Papandonatos, G.D.; Hitsman, B.; Reiches, M.; Krishnamoorthy, J.; Niaura, R. Links between physical fitness and cardiovascular reactivity and recovery to psychological stressors: A meta-analysis. Health Psychol. 2006, 25, 723–739. [Google Scholar] [CrossRef] [PubMed]
- Netz, Y.; Wu, M.J.; Becker, B.J.; Tenenbaum, G. Physical activity and psychological well-being in advanced age: A meta-analysis of intervention studies. Psychol. Aging 2005, 20, 272–284. [Google Scholar] [CrossRef] [PubMed]
- Bandura, A. Self-efficacy: Toward a unifying theory of behavioral change. Psychol. Rev. 1977, 84, 191–215. [Google Scholar] [CrossRef]
- Motl, R.W.; Konopack, J.F.; McAuley, E.; Elavsky, S.; Jerome, G.J.; Marquez, D.X. Depressive symptoms among older adults: Long-term reduction after a physical activity intervention. J. Behav. Med. 2005, 28, 385–394. [Google Scholar] [CrossRef]
- Bento-Torres, J.; Bento-Torres, N.V.O.; Stillman, C.M.; Grove, G.A.; Huang, H.; Uyar, F.; Watt, J.C.; Wollam, M.E.; Erickson, K.I. Associations between cardiorespiratory fitness, physical activity, intraindividual variability in behavior, and cingulate cortex in younger adults. J. Sport Health Sci. 2019, 8, 315–324. [Google Scholar] [CrossRef]
- Etnier, J.L.; Chang, Y.K. Exercise, cognitive function, and the brain: Advancing our understanding of complex relationships. J. Sport Health Sci. 2019, 8, 299–300. [Google Scholar] [CrossRef]
- Audiffren, M.; André, N. The exercise–cognition relationship: A virtuous circle. J. Sport Health Sci. 2019, 8, 339–347. [Google Scholar] [CrossRef] [PubMed]
- Smith, J.C. Effects of emotional exposure on state anxiety after acute exercise. Med. Sci. Sports Exerc. 2013, 45, 372–378. [Google Scholar] [CrossRef] [PubMed]
- Reed, J.; Buck, S. The effect of regular aerobic exercise on positive-activated affect: A meta-analysis. Psychol. Sport Exerc. 2009, 10, 581–594. [Google Scholar] [CrossRef]
- Hallgren, M.Å.; Moss, N.D.; Gastin, P. Regular exercise participation mediates the affective response to acute bouts of vigorous exercise. J. Sports Sci. Med. 2010, 9, 629–637. [Google Scholar] [PubMed]
- Hopkins, M.E.; Davis, F.C.; Vantieghem, M.R.; Whalen, P.J.; Bucci, D.J. Differential effects of acute and regular physical exercise on cognition and affect. Neuroscience 2012, 215, 59–68. [Google Scholar] [CrossRef] [PubMed]
- Schuch, F.B.; Vancampfort, D.; Firth, J.; Rosenbaum, S.; Ward, P.B.; Silva, E.S.; Hallgren, M.; Ponce De Leon, A.; Dunn, A.L.; Deslandes, A.C.; et al. Physical activity and incident depression: A meta-analysis of prospective cohort studies. Am. J. Psychiatry 2018, 175, 631–648. [Google Scholar] [CrossRef]
- Sothmann, M.S. The cross-stressor adaptation hypothesis and exercise training. In Psychobiology of Physical Activity; Human Kinetics: Champaign, IL, USA, 2006; pp. 149–160. [Google Scholar]
- Jankord, R.; Herman, J.P. Limbic regulation of hypothalamo-pituitary-adrenocortical function during acute and chronic stress. Ann. N. Y. Acad. Sci. 2008, 1148, 64–73. [Google Scholar] [CrossRef]
- Herman, J.P.; Ostrander, M.M.; Mueller, N.K.; Figueiredo, H. Limbic system mechanisms of stress regulation: Hypothalamo-pituitary-adrenocortical axis. Prog. Neuropsychopharmacol. Biol. Psychiatry 2005, 29, 1201–1213. [Google Scholar] [CrossRef]
- Fardin, M.A. COVID-19 and anxiety: A review of psychological impacts of infectious disease outbreaks. Arch. Clin. Infect. Dis. 2020, 15, e102779. [Google Scholar] [CrossRef]
- Elbay, R.Y.; Kurtulmuş, A.; Arpacıoğlu, S.; Karadere, E. Depression, anxiety, stress levels of physicians and associated factors in Covid-19 pandemics. Psychiatry Res. 2020, 290, 113130. [Google Scholar] [CrossRef]
- Hossain, M.D.; Tasnim, S.; Sultana, A.; Faizah, F.; Mazumder, H.; Zou, L.; McKyer, E.L.; Ahmed, H.; Ma, P. Epidemiology of mental health problems in COVID-19: A review. F1000Research 2020, 9, 636. [Google Scholar] [CrossRef]
- Mujika, I.; Padilla, S. Detraining: Loss of training-induced physiological and performance adaptations. Part II: Long term insufficient training stimulus. Sports Med. 2000, 30, 145–154. [Google Scholar] [CrossRef] [PubMed]
- Elliott, K.J.; Sale, C.; Cable, N.T. Effects of resistance training and detraining on muscle strength and blood lipid profiles in postmenopausal women. Br. J. Sports Med. 2002, 36, 340–344. [Google Scholar] [CrossRef] [PubMed]
- Van Roie, E.; Walker, S.; Van Driessche, S.; Baggen, R.; Coudyzer, W.; Bautmans, I.; Delecluse, C. Training load does not affect detraining’s effect on muscle volume, muscle strength and functional capacity among older adults. Exp. Gerontol. 2017, 98, 30–37. [Google Scholar] [CrossRef] [PubMed]
- Esain, I.; Gil, S.M.; Bidaurrazaga-Letona, I.; Rodriguez-Larrad, A. Effects of 3 months of detraining on functional fitness and quality of life in older adults who regularly exercise. Aging Clin. Exp. Res. 2019, 31, 503–510. [Google Scholar] [CrossRef] [PubMed]
- Berlin, A.A.; Kop, W.J.; Deuster, P.A. Depressive mood symptoms and fatigue after exercise withdrawal: The potential role of decreased fitness. Psychosom. Med. 2006, 68, 224–230. [Google Scholar] [CrossRef]
- Kop, W.J.; Weinstein, A.A.; Deuster, P.A.; Whittaker, K.S.; Tracy, R.P. Inflammatory markers and negative mood symptoms following exercise withdrawal. Brain Behav. Immun. 2008, 22, 1190–1196. [Google Scholar] [CrossRef]
- McAuley, E.; Courneya, K.S. Self-efficacy relationships with affective and exertion responses to exercise. J. Appl. Soc. Psychol. 1992, 22, 312–326. [Google Scholar] [CrossRef]
- LePage, M.L.; Price, M.; O’Neil, P.; Crowther, J.H. The effect of exercise absence on affect and body dissatisfaction as moderated by obligatory exercise beliefs and eating disordered beliefs and behaviors. Psychol. Sport Exerc. 2012, 13, 500–508. [Google Scholar] [CrossRef]
- Taiwan Network Information Center. 2019 Taiwan, 2019 ed.; Taiwan Network Information Center: Taipei, Taiwan, 2020. [Google Scholar]
- Wilke, J.; Mohr, L.; Tenforde, A.S.; Edouard, P.; Fossati, C.; González-Gross, M.; Ramirez, C.S.; Laiño, F.; Tan, B.; Pillay, J.D.; et al. Restrictercise! Preferences regarding digital home training programs during confinements associated with the COVID-19 pandemic. Int. J. Environ. Res. Public Health 2020, 17, 6515. [Google Scholar] [CrossRef]
- Hurling, R.; Fairley, B.W.; Dias, M.B. Internet-based exercise intervention systems: Are more interactive designs better? Psychol. Health 2006, 21, 757–772. [Google Scholar] [CrossRef]
- Wojcicki, T.R.; Grigsby-Toussaint, D.; Hillman, C.H.; Huhman, M.; McAuley, E. Promoting physical activity in low-active adolescents via Facebook: A pilot randomized controlled trial to test feasibility. JMIR Res. Protoc. 2014, 3, e56. [Google Scholar] [CrossRef] [PubMed]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).