Enhancement of Toxic Efficacy of Alkylated Polycyclic Aromatic Hydrocarbons Transformed by Sphingobium quisquiliarum
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Culturing of Bacteria
2.3. Biodegradation Kinetics
2.4. Fractionation of Transformation Products
2.5. Identification of Metabolic Products of Alkylated PAHs
2.6. Luminescence Inhibition Assay
3. Results
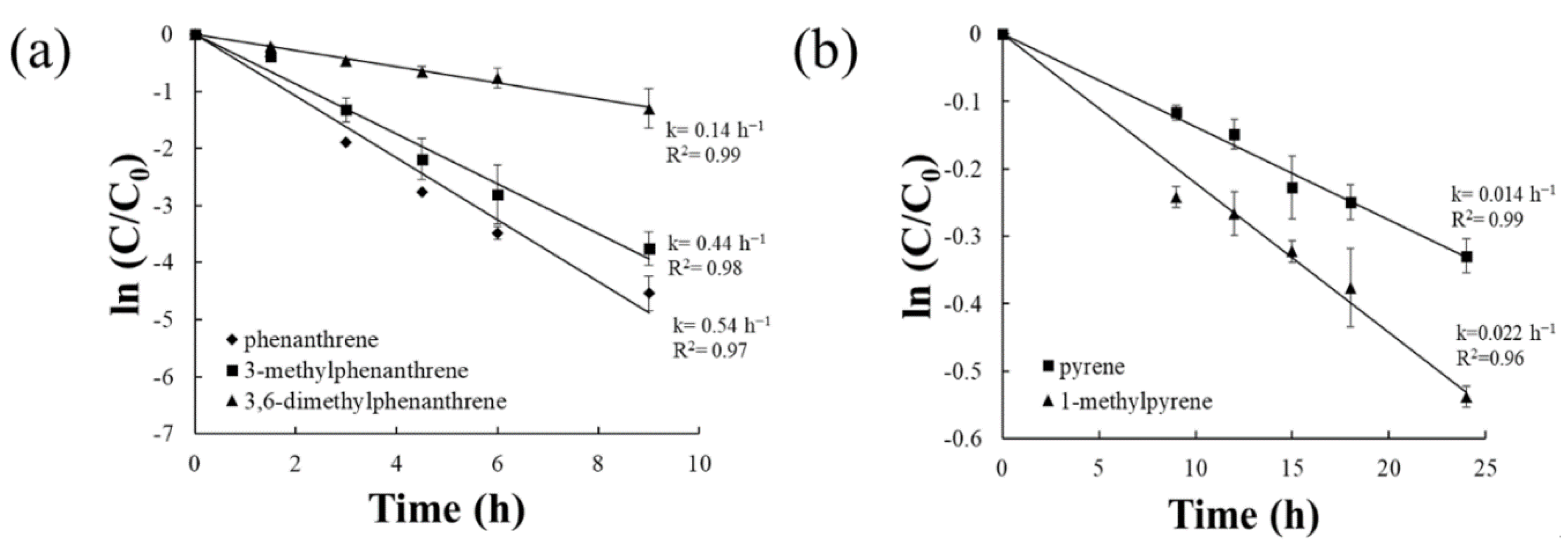
3.1. Biodegradation Kinetics of Alkylated PAHs
3.2. Fractionation of Transformation Products
3.3. Identification of Transformation Products
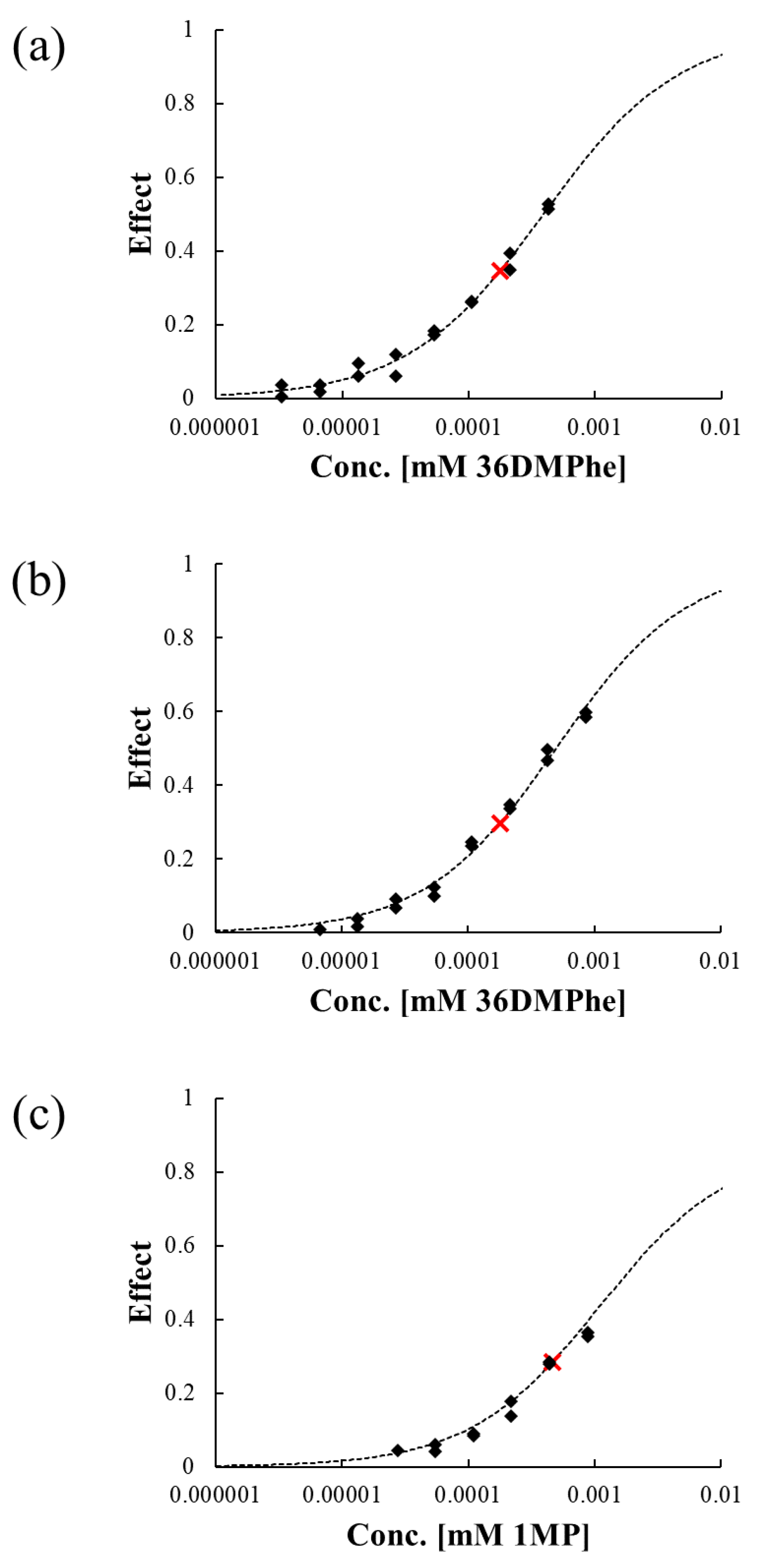
3.4. Evaluation of Luminescence Inhibition by Transformation Products
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Baklanov, A.; Hänninen, O.; Slørdal, L.H.; Kukkonen, J.; Bjergene, N.; Fay, B.; Finardi, S.; Hoe, S.C.; Jantunen, M.; Karppinen, A.; et al. Integrated systems for forecasting urban meteorology, air pollution and population exposure. Atmos. Chem. Phys. 2007, 7, 855–874. [Google Scholar] [CrossRef] [Green Version]
- Latimer, J.S.; Zheng, J. The Sources, Transport, and Fate of PAHs in the Marine Environment. In PAHs: An Ecotoxicological Perspective; John Wiley & Sons: New York, NY, USA, 2003. [Google Scholar] [CrossRef]
- Abdel-Shafy, H.I.; Mansour, M.S.M. A review on polycyclic aromatic hydrocarbons: Source, environmental impact, effect on human health and remediation. Egypt J. Pet. 2016, 25, 107–123. [Google Scholar] [CrossRef] [Green Version]
- Hawthorne, S.B.; Miller, D.J.; Kreitinger, J.P. Measurement of total polycyclic aromatic hydrocarbon concentrations in sediments and toxic units used for estimating risk to benthic invertebrates at manufactured gas plant sites. Environ. Toxicol. Chem. 2006, 25, 287–296. [Google Scholar] [CrossRef] [PubMed]
- Kang, H.-J.; Lee, S.-Y.; Roh, J.-Y.; Yim, U.H.; Shim, W.J.; Kwon, J.-H. Prediction of ecotoxicity of heavy crude oil: Contribution of measured components. Environ. Sci. Technol. 2014, 48, 2962–2970. [Google Scholar] [CrossRef] [PubMed]
- Ramseur, J.L. CRS Report for Congress Deepwater Horizon Oil Spill: The Fate of the Oil; Congressional Research Service: Washington, DC, USA, 2010. [Google Scholar]
- Camilli, R.; Reddy, C.M.; Yoerger, D.R.; Van Mooy, B.A.S.; Jakuba, M.V.; Kinsey, J.C.; McIntyre, C.P.; Sylva, S.P.; Maloney, J.V. Tracking hydrocarbon plume transport and biodegradation at deepwater horizon. Science 2010, 330, 201–204. [Google Scholar] [CrossRef] [PubMed]
- Hazen, T.C.; Dubinsky, E.A.; DeSantis, T.Z.; Andersen, G.L.; Piceno, Y.M.; Singh, N.; Jansson, J.K.; Probst, A.; Borglin, S.E.; Fortney, J.L.; et al. Deep-sea oil plume enriches indigenous oil-degrading bacteria. Science 2010, 330, 204–208. [Google Scholar] [CrossRef] [Green Version]
- Seo, J.-S.; Keum, Y.-S.; Li, Q.X. Bacterial degradation of aromatic compounds. Int. J. Environ. Res. Public Health 2010, 6, 278–309. [Google Scholar] [CrossRef]
- Cerniglia, C.E. Biodegradation of polycyclic aromatic hydrocarbons. Biodegradation 1992, 3, 351–368. [Google Scholar] [CrossRef]
- Waigi, M.G.; Kang, F.; Goikavi, C.; Ling, W.; Gao, Y. Phenanthrene biodegradation by sphingomonads and its application in the contaminated soils and sediments: A review. Int. Biodeterior. Biodegrad. 2015, 104, 333–349. [Google Scholar] [CrossRef]
- Peng, R.H.; Xiong, A.S.; Xue, Y.; Fu, X.Y.; Gao, F.; Zhao, W.; Tian, Y.S.; Yao, Q.H. Microbial biodegradation of polyaromatic hydrocarbons. FEMS Microbiol. Rev. 2008, 32, 927–955. [Google Scholar] [CrossRef] [Green Version]
- Haritash, A.K.; Kaushik, C.P. Biodegradation aspects of polycyclic aromatic hydrocarbons (PAHs): A review. J. Hazard Mater 2009, 169, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Rojo-nieto, E.; Perales-Vargas-Machuca, J.A. Microbial Degradation of PAHs: Organisms and Environmental Compartments. In Microbial Degradation of Xenobiotics; Singh, S.N., Ed.; Springer: Berlin/Heidelberg, Germany, 2012; pp. 263–290. [Google Scholar] [CrossRef]
- Ghosal, D.; Ghosh, S.; Dutta, T.K.; Ahn, Y. Current state of knowledge in microbial degradation of polycyclic aromatic hydrocarbons (PAHs): A review. Front. Microbiol. 2016, 7, 1369. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Basu, A.; Dixit, S.S.; Phale, P.S. Metabolism of benzyl alcohol via catechol ortho-pathway in methylnaphthalene-degrading Pseudomonas putida CSV86. Appl. Microbiol. Biotechnol. 2003, 62, 579–585. [Google Scholar] [CrossRef] [PubMed]
- Mahajan, M.C.; Phale, P.S.; Vaidyanathan, C.S. Evidence for the involvement of multiple pathways in the biodegradation of 1- and 2-methylnaphthalene by Pseudomonas putida CSV86. Arch. Microbiol. 1994, 161, 425–433. [Google Scholar] [CrossRef]
- Musat, F.; Galushko, A.; Jacob, J.; Widdel, F.; Kube, M.; Reinhardt, R.; Wilkes, H.; Schink, B.; Rabus, R. Anaerobic degradation of naphthalene and 2-methylnaphthalene by strains of marine sulfate-reducing bacteria. Environ. Microbiol. 2009, 11, 209–219. [Google Scholar] [CrossRef] [Green Version]
- Saftić, S.; Fedorak, P.M.; Andersson, J.T. Transformations of methyldibenzothiophenes by three Pseudomonas isolates. Environ. Sci. Technol. 1993, 27, 2577–2584. [Google Scholar] [CrossRef]
- Dutta, T.K.; Selifonov, S.A.; Gunsalus, I.C. Oxidation of methyl-substituted naphthalenes: Pathways in a versatile Sphingomonas paucimobilis strain. Appl. Environ. Microbiol. 1998, 64, 1884–1889. [Google Scholar] [CrossRef] [Green Version]
- Tøndervik, A.; Bruheim, P.; Berg, L.; Ellingsen, T.E.; Kotlar, H.K.; Valla, S.; Throne-Holst, M. Ralstonia sp. U2 naphthalene dioxygenase and Comamonas sp. JS765 nitrobenzene dioxygenase show differences in activity towards methylated naphthalenes. J. Biosci. Bioeng. 2012, 113, 173–178. [Google Scholar] [CrossRef]
- Zhong, J.; Luo, L.; Chen, B.; Sha, S.; Qing, Q.; Tam, N.F.Y.; Zhang, Y.; Luan, T. Degradation pathways of 1-methylphenanthrene in bacterial Sphingobium sp. MP9-4 isolated from petroleum-contaminated soil. Mar. Pollut. Bull 2017, 114, 926–933. [Google Scholar] [CrossRef]
- Wammer, K.H.; Peters, C.A. Polycyclic aromatic hydrocarbon biodegradation rates: A structure-based study. Environ. Sci. Technol. 2005, 39, 2571–2578. [Google Scholar] [CrossRef]
- Dimitriou-Christidis, P.; Autenrieth, R.L.; McDonald, T.J.; Desai, A.M. Measurement of biodegradability parameters for single unsubstituted and methylated polycyclic aromatic hydrocarbons in liquid bacterial suspensions. Biotechnol. Bioeng. 2007, 97, 922–932. [Google Scholar] [CrossRef] [PubMed]
- Desai, A.M.; Autenrieth, R.L.; Dimitriou-Christidis, P.; McDonald, T.J. Biodegradation kinetics of select polycyclic aromatic hydrocarbon (PAH) mixtures by Sphingomonas paucimobilis EPA505. Biodegradation 2008, 19, 223–233. [Google Scholar] [CrossRef] [PubMed]
- Knightes, C.D.; Peters, C.A. Aqueous phase biodegradation kinetics of 10 PAH compounds. Environ. Eng. Sci. 2003, 20, 207–218. [Google Scholar] [CrossRef]
- Siddiqi, M.A.; Yuan, Z.X.; Honey, S.A.; Kumar, S.; Sikka, H.C. Metabolism of PAHs and methyl-substituted PAHs by Sphingomonas paucimobilis strain EPA 505. Polycycl. Aromat. Compd. 2002, 22, 621–630. [Google Scholar] [CrossRef]
- Pampanin, D.M.; Sydnes, M.O. Chapter 5: Polycyclic Aromatic Hydrocarbons a Constituent of Petroleum: Presence and Influence in the Aquatic Environment. In Hydrocarbon; Kutcherov, V., Kolesnikov, A., Eds.; InTechOpen: London, UK, 2013; pp. 83–118. [Google Scholar] [CrossRef] [Green Version]
- Yim, U.H.; Ha, S.Y.; An, J.G.; Won, J.H.; Han, G.M.; Hong, S.H.; Kim, M.; Jung, J.H.; Shim, W.J. Fingerprint and weathering characteristics of stranded oils after the Hebei Spirit oil spill. J. Hazard Mater. 2011, 197, 60–69. [Google Scholar] [CrossRef]
- Neff, J.; Lee, K.; DeBlois, E.M. Produced Water: Overview of Composition, Fates, and Effects. In Produced Water: Environmental Risks and Advances in Mitigation Technologies; Lee, K., Neff, J., Eds.; Springer: New York, NY, USA, 2011; pp. 3–54. [Google Scholar] [CrossRef]
- Lundstedt, S.; White, P.A.; Lemieux, C.L.; Lynes, K.D.; Lambert, I.B.; Oberg, L.; Haglund, P.; Tysklind, M. Sources, fate, and toxic hazards of oxygenated polycyclic aromatic hydrocarbons (PAHs) at PAH-contaminated sites. Ambio 2007, 36, 475–485. [Google Scholar] [CrossRef]
- Arp, H.P.H.; Lundstedt, S.; Josefsson, S.; Cornelissen, G.; Enell, A.; Allard, A.S.; Kleja, D.B. Native oxy-PAHs, N-PACs, and PAHs in historically contaminated soils from Sweden, Belgium, and France: Their soil-porewater partitioning behavior, bioaccumulation in Enchytraeus crypticus, and bioavailability. Environ. Sci. Technol. 2014, 48, 11187–11195. [Google Scholar] [CrossRef]
- Frank, R.A.; Kavanagh, R.; Burnison, B.K.; Arsenault, G.; Headley, J.V.; Peru, K.M.; Van Der Kraak, G.; Solomon, K.R. Toxicity assessment of collected fractions from an extracted naphthenic acid mixture. Chemosphere 2008, 72, 1309–1314. [Google Scholar] [CrossRef]
- Jones, D.; Scarlett, A.G.; West, C.E.; Rowland, S.J. Toxicity of individual naphthenic acids to Vibrio fischeri. Environ. Sci. Technol. 2011, 45, 9776–9782. [Google Scholar] [CrossRef]
- Fallahtafti, S.; Rantanen, T.; Brown, R.S.; Snieckus, V.; Hodson, P.V. Toxicity of hydroxylated alkyl-phenanthrenes to the early life stages of Japanese medaka (Oryzias latipes). Aquat. Toxicol. 2012, 106–107, 56–64. [Google Scholar] [CrossRef]
- Diamante, G.; do Amaral e Silva Müller, G.; Menjivar-Cervantes, N.; Xu, E.G.; Volz, D.C.; Dias Bainy, A.C.; Schlenk, D. Developmental toxicity of hydroxylated chrysene metabolites in zebrafish embryos. Aquat. Toxicol. 2017, 189, 77–86. [Google Scholar] [CrossRef] [PubMed]
- Kawano, M.; Uno, S.; Koyama, J.; Kokushi, E.; McElroy, A. Effects of oxygenated polycyclic aromatic hydrocarbons on the early life stages of Japanese medaka. Environ. Sci. Pollut. Res. 2017, 24, 27670–27677. [Google Scholar] [CrossRef]
- Dogra, Y.; Scarlett, A.G.; Rowe, D.; Galloway, T.S.; Rowland, S.J. Predicted and measured acute toxicity and developmental abnormalities in zebrafish embryos produced by exposure to individual aromatic acids. Chemosphere 2018, 205, 98–107. [Google Scholar] [CrossRef] [PubMed]
- Wincent, E.; Jönsson, M.E.; Bottai, M.; Lundstedt, S.; Dreij, K. Aryl hydrocarbon receptor activation and developmental toxicity in zebrafish in response to soil extracts containing unsubstituted and oxygenated pahs. Environ. Sci. Technol. 2015, 49, 3869–3877. [Google Scholar] [CrossRef] [PubMed]
- Roh, J.-Y.; Lee, H.; Kwon, J.-H. Changes in the expression of cyp35a family genes in the soil nematode Caenorhabditis elegans under controlled exposure to chlorpyrifos using passive dosing. Environ. Sci. Technol. 2014, 48, 10475–10481. [Google Scholar] [CrossRef]
- Kwon, J.-H.; Wuethrich, T.; Mayer, P.; Escher, B.I. Development of a dynamic delivery method for in vitro bioassays. Chemosphere 2009, 76, 83–90. [Google Scholar] [CrossRef]
- Smith, K.E.C.; Rein, A.; Trapp, S.; Mayer, P.; Karlson, U.G. Dynamic passive dosing for studying the biotransformation of hydrophobic organic chemicals: Microbial degradation as an example. Environ. Sci. Technol. 2012, 46, 4852–4860. [Google Scholar] [CrossRef] [Green Version]
- Stanier, R.Y.; Palleroni, N.J.; Doudoroff, M. The aerobic Pseudomonads: A Taxonomic study. J. Gen. Microbiol. 1966, 43, 159–271. [Google Scholar] [CrossRef] [Green Version]
- Cohen-Bazire, G.; Sistrom, W.R.; Stanier, R.Y. Kinetic studies of pigment synthesis by non-sulfur purple bacteria. J. Cell Comp. Physiol. 1957, 49, 25–68. [Google Scholar] [CrossRef]
- US Environmental Protection Agency. EPIsuiteTM v.4.11. 2012. Available online: https://www.epa.gov/tsca-screening-tools/download-epi-suitetm-estimation-program-interface-v411 (accessed on 19 July 2017).
- Kwon, H.-C.; Kwon, J.-H. Measuring aqueous solubility in the presence of small cosolvent volume fractions by passive dosing. Environ. Sci. Technol. 2012, 46, 12550–12556. [Google Scholar] [CrossRef]
- Kang, H.-J.; Lee, S.-Y.; Kwon, J.-H. Physico-chemical properties and toxicity of alkylated polycyclic aromatic hydrocarbons. J. Hazard Mater. 2016, 312, 200–207. [Google Scholar] [CrossRef] [PubMed]
- van der Heijden, S.A.; Jonker, M.T.O. Evaluation of liposome-water partitioning for predicting bioaccumulation potential of hydrophobic organic chemicals. Environ. Sci. Technol. 2009, 43, 8854–8859. [Google Scholar] [CrossRef] [PubMed]
- Malmquist, L.M.V.; Selk, H.; Jørgensen, K.B.; Christensen, J.H. Polycyclic aromatic acids are primary metabolites of alkyl-PAHs—A case study with Nereis Diversicolor. Environ. Sci. Technol. 2015, 49, 5713–5721. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, I.; Jasmine, J.; Mukherji, S. Biodegradation of pyrene by a Pseudomonas aeruginosa strain RS1 isolated from refinery sludge. Bioresour. Technol. 2014, 166, 548–558. [Google Scholar] [CrossRef]
- Vila, J.; López, Z.; Sabaté, J.; Minguillón, C.; Solanas, A.M.; Grifoll, M. Identification of a novel metabolite in the degradation of pyrene by Mycobacterium sp. strain AP1: Actions of the isolate on two- and three-ring polycyclic aromatic hydrocarbons. Appl. Environ. Microbiol. 2001, 67, 5497–5505. [Google Scholar] [CrossRef] [Green Version]
- Zhong, Y.; Luan, T.; Zhou, H.; Lan, C.; Tam, N.F.Y. Metabolite production in degradation of pyrene alone or in a mixture with another polycyclic aromatic hydrocarbon by Mycobacterium sp. Environ. Toxicol. Chem. 2006, 25, 2853–2859. [Google Scholar] [CrossRef]
- Pagnout, C.; Rast, C.; Veber, A.-M.; Poupin, P.; Férard, J.-F. Ecotoxicological assessment of PAHs and their dead-end metabolites after degradation by Mycobacterium sp. strain SNP11. Ecotoxicol. Environ. Saf. 2006, 65, 151–158. [Google Scholar] [CrossRef]
- Šepič, E.; Bricelj, M.; Leskovšek, H. Toxicity of fluoranthene and its biodegradation metabolites to aquatic organisms. Chemosphere 2003, 52, 1125–1133. [Google Scholar] [CrossRef]
- Singh, S.N.; Kumari, B.; Upadhyay, S.K.; Mishra, S.; Kumar, D. Bacterial degradation of pyrene in minimal salt medium mediated by catechol dioxygenases: Enzyme purification and molecular size determination. Bioresour. Technol. 2013, 133, 293–300. [Google Scholar] [CrossRef]
- Kumar, S.; Upadhayay, S.K.; Kumari, B.; Tiwari, S.; Singh, S.N.; Singh, P.K. In vitro degradation of fluoranthene by bacteria isolated from petroleum sludge. Bioresour. Technol. 2011, 102, 3709–3715. [Google Scholar] [CrossRef]
- Lee, S.-Y.; Kang, H.-J.; Kwon, J.-H. Toxicity cutoff of aromatic hydrocarbons for luminescence inhibition of Vibrio fischeri. Ecotoxicol. Environ. Saf. 2013, 94, 16–22. [Google Scholar] [CrossRef] [PubMed]
- Verhaar, H.J.M.; van Leeuwen, C.J.; Hermens, J.L.M. Classifying environmental pollutants. 1. Structure-activity relationships for prediction of aquatic toxicity. Chemosphere 1992, 25, 471–491. [Google Scholar] [CrossRef]
| Chemical | Structure | MW (g mol−1) | S (mg L−1 at 25 °C) | Cini (mg L−1, mean ± SD) | log Kow 1 | log Klipw |
|---|---|---|---|---|---|---|
| phenanthrene | 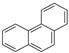 | 178 | 0.82 2 | 0.72 ± 0.03 | 4.46 | 5.05 4 |
| 3MPhe | 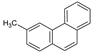 | 192.26 | 0.63 3 | 0.56 ± 0.02 | 4.89 | 5.24 3 |
| 36DMPhe | 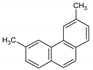 | 206.29 | 0.037 3 | 0.027 ± 0.001 | 5.44 | 5.58 3 |
| Pyrene | 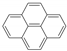 | 202 | 0.086 2 | 0.088 ± 0.003 | 5.18 | 5.74 4 |
| 1MP | 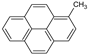 | 216.28 | 0.10 3 | 0.102 ± 0.005 | 5.48 | 5.76 3 |
| Chemical | Retention Time (min) | Mass Spectrum (m/z) |
|---|---|---|
| 3,6DMPhe | 16.79 | 206.1, 202.1, 191.1, 187.0 |
| 36DMPhe_F1 | 37.35 | 335.0, 293.0, 218.9, 204.0, 190.9, 183.8 |
| 36DMPhe_F2 | 28.32 | 336.1, 321.0, 279.1, 220.1, 205.0, 189.0 |
| 1MP | 22.02 | 216.1, 207.1, 199.9, 189.0, 163.0 |
| 1MP_F | 32.52 | 346.1, 289.1, 215.0, 200.0, 188.9, 133.9 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, S.-Y.; Kwon, J.-H. Enhancement of Toxic Efficacy of Alkylated Polycyclic Aromatic Hydrocarbons Transformed by Sphingobium quisquiliarum. Int. J. Environ. Res. Public Health 2020, 17, 6416. https://doi.org/10.3390/ijerph17176416
Lee S-Y, Kwon J-H. Enhancement of Toxic Efficacy of Alkylated Polycyclic Aromatic Hydrocarbons Transformed by Sphingobium quisquiliarum. International Journal of Environmental Research and Public Health. 2020; 17(17):6416. https://doi.org/10.3390/ijerph17176416
Chicago/Turabian StyleLee, So-Young, and Jung-Hwan Kwon. 2020. "Enhancement of Toxic Efficacy of Alkylated Polycyclic Aromatic Hydrocarbons Transformed by Sphingobium quisquiliarum" International Journal of Environmental Research and Public Health 17, no. 17: 6416. https://doi.org/10.3390/ijerph17176416
APA StyleLee, S.-Y., & Kwon, J.-H. (2020). Enhancement of Toxic Efficacy of Alkylated Polycyclic Aromatic Hydrocarbons Transformed by Sphingobium quisquiliarum. International Journal of Environmental Research and Public Health, 17(17), 6416. https://doi.org/10.3390/ijerph17176416





