Poor Oral Hygiene and High Levels of Inflammatory Cytokines in Saliva Predict the Risk of Overweight and Obesity
Abstract
:1. Introduction
2. Materials and Methods
3. Results
3.1. Basic Statistics
3.2. Logistic Regression Models
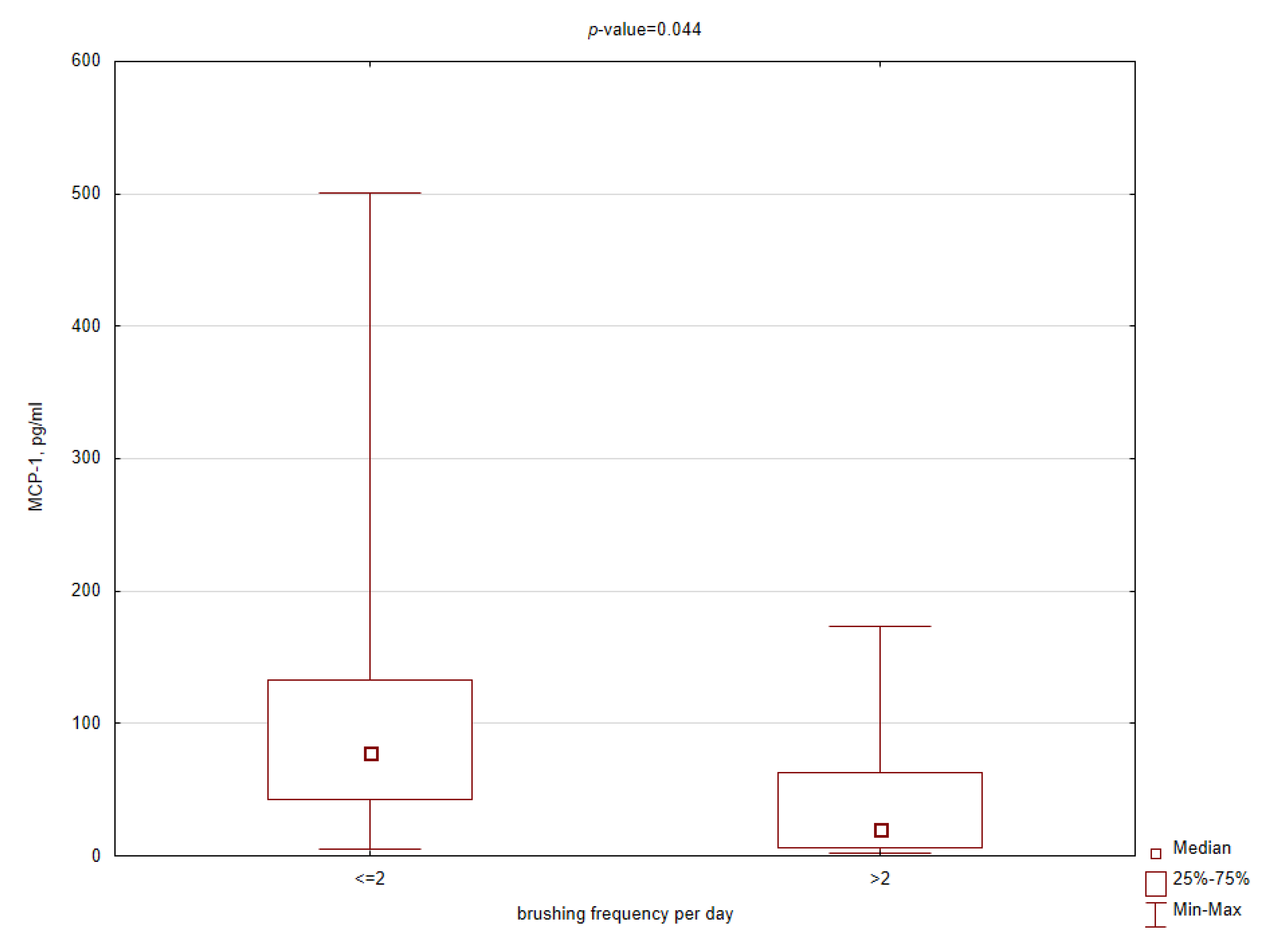
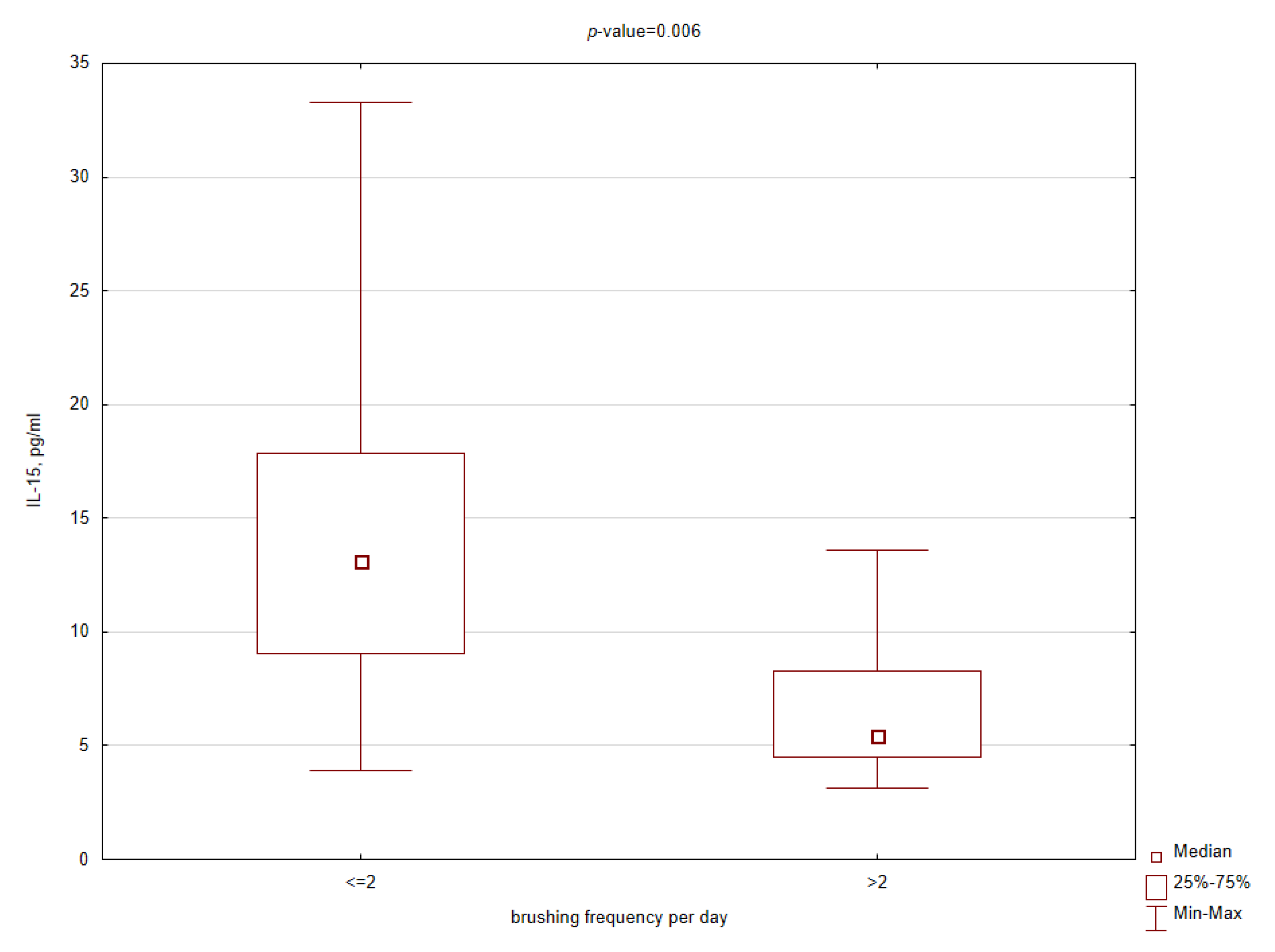
3.3. Comparative Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- World Health Organization. Obesity: Preventing and Managing the Global Epidemic; Report of a WHO Consultation, World Health Organization Technical Report Series 894; World Health Organization: Geneva, Switzerland, 2000; pp. 1–253. [Google Scholar]
- Duffles, L.F.; Hermont, A.P.; Abreu, L.G.; Pordeus, I.A.; Silva, T.A. Association between obesity and adipokines levels in saliva and gingival crevicular fluid: A systematic review and meta-analysis. J. Evid. Based Med. 2019, 12, 313–324. [Google Scholar] [CrossRef]
- Goodson, J.M.; Kantarci, A.; Hartman, M.-L.; Denis, G.V.; Stephens, D.; Hasturk, H.; Yaskell, T.; Vargas, J.; Wang, X.; Cugini, M.; et al. Metabolic Disease Risk in Children by Salivary Biomarker Analysis. PLoS ONE 2014, 9, e98799. [Google Scholar] [CrossRef]
- Mamali, I.; Roupas, N.D.; Armeni, A.K.; Theodoropoulou, A.; Markou, K.B.; Georgopoulos, N.A. Measurement of salivary resistin, visfatin and adiponectin levels. Peptides 2012, 33, 120–124. [Google Scholar] [CrossRef] [PubMed]
- Nigro, E.; Piombino, P.; Scudiero, O.; Monaco, M.L.; Schettino, P.; Chambery, A.; Daniele, A. Evaluation of salivary adiponectin profile in obese patients. Peptides 2015, 63, 150–155. [Google Scholar] [CrossRef] [PubMed]
- Thanakun, S.; Watanabe, H.; Thaweboon, S.; Izumi, Y. Comparison of salivary and plasma adiponectin and leptin in patients with metabolic syndrome. Diabetol. Metab. Syndr. 2014, 6, 19. [Google Scholar] [CrossRef] [Green Version]
- Kim, J.S.; Kim, S.Y.; Byon, M.J.; Lee, J.H.; Jeong, S.H.; Kim, J.B. Association between Periodontitis and Metabolic Syndrome in a Korean Nationally Representative Sample of Adults Aged 35–79 Years. Int. J. Environ. Res. Public Health 2019, 16, 2930. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- De Moura-Grec, P.G.; Marsicano, J.A.; de Carvalho, C.A.P.; Sales-Peres, S.H.D.C. Obesity and periodontitis: Systematic review and meta-analysis. Cien. Saude Colet. 2014, 19, 1763–1772. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Suvan, J.E.; Finer, N.; D’Aiuto, F. Periodontal complications with obesity. Periodontol. 2000 2018, 78, 98–128. [Google Scholar] [CrossRef]
- Goodson, J.M.; Groppo, D.; Halem, S.; Carpino, E. Is Obesity an Oral Bacterial Disease? J. Dent. Res. 2009, 88, 519–523. [Google Scholar] [CrossRef]
- Andreeva, V.A.; Egnell, M.; Galan, P.; Feron, G.; Hercberg, S.; Julia, C. Association of the Dietary Index Underpinning the Nutri-Score Label with Oral Health: Preliminary Evidence from a Large, Population-Based Sample. Nutrients 2019, 11, 1998. [Google Scholar] [CrossRef] [Green Version]
- Garcia, R.I.; Kleinman, D.; Holt, K.; Battrell, A.; Casamassimo, P.; Grover, J.; Tinanoff, N. Healthy Futures: Engaging the oral health community in childhood obesity prevention—Conference summary and recommendations. J. Public Health Dent. 2017, 77, S136–S140. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kantovitz, K.R.; Pascon, F.M.; Rontani, R.M.P.; Gavião, M.B.D. Obesity and dental caries—A systematic review. Oral Health Prev. Dent. 2006, 4, 137–144. [Google Scholar] [PubMed]
- Mervish, N.A.; Hu, J.; Hagan, L.A.; Arora, M.; Frau, C.; Choi, J.; Attaie, A.; Ahmed, M.; Teitelbaum, S.L.; Wolff, M.S. Associations of the Oral Microbiota with Obesity and Menarche in Inner City Girls. J. Child. Obes. 2019, 4, 2. [Google Scholar] [CrossRef] [PubMed]
- Lehmann, A.P.; Nijakowski, K.; Swora-Cwynar, E.; Łuczak, J.; Czepulis, N.; Surdacka, A. Characteristics of salivary inflammation profile in obesity. Pol. Arch. Intern. Med. 2020, 130, 297–303. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Amar, S.; Zhou, Q.; Shaik-Dasthagirisaheb, Y.; Leeman, S.E. Diet-induced obesity in mice causes changes in immune responses and bone loss manifested by bacterial challenge. Proc. Natl. Acad. Sci. USA 2007, 104, 20466–20471. [Google Scholar] [CrossRef] [Green Version]
- Modéer, T.; Blomberg, C.C.; Wondimu, B.; Julihn, A.; Marcus, C. Association Between Obesity, Flow Rate of Whole Saliva, and Dental Caries in Adolescents. Obesity 2010, 18, 2367–2373. [Google Scholar] [CrossRef]
- Roa, I.; Del Sol, M. Obesity, salivary glands and oral pathology. Colomb. Med. 2018, 49, 280–287. [Google Scholar] [CrossRef] [Green Version]
- Rangé, H.; Léger, T.; Huchon, C.; Ciangura, C.; Diallo, D.; Poitou, C.; Meilhac, O.; Bouchard, P.; Chaussain, C. Salivary proteome modifications associated with periodontitis in obese patients. J. Clin. Periodontol. 2012, 39, 799–806. [Google Scholar] [CrossRef]
- Akram, Z.; Abduljabbar, T.; Abu Hassan, M.I.; Javed, F.; Vohra, F. Cytokine Profile in Chronic Periodontitis Patients with and without Obesity: A Systematic Review and Meta-Analysis. Dis. Markers 2016, 2016, 1–12. [Google Scholar] [CrossRef] [Green Version]
- Hujoel, P.; Kressin, N.R.; Cunha-Cruz, J. Spurious associations in oral epidemiological research: The case of dental flossing and obesity. J. Clin. Periodontol. 2006, 33, 520–523. [Google Scholar] [CrossRef]
- Konopka, T.; Matuszewska, A.; Chrzęszczyk, D.; Zawada, D. Body Mass Index and selected periodontal clinical parameters. Dent. Med. Probl. 2011, 48, 189–197. [Google Scholar]
- Park, J.B.; Nam, G.E.; Han, K.; Ko, Y.; Park, Y.G. Obesity in relation to oral health behaviors: An analysis of the Korea National Health and Nutrition Examination Survey 2008–2010. Exp. Ther. Med. 2016, 12, 3093–3100. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Franchini, R.; Petri, A.; Migliario, M.; Rimondini, L. Poor oral hygiene and gingivitis are associated with obesity and overweight status in paediatric subjects. J. Clin. Periodontol. 2011, 38, 1021–1028. [Google Scholar] [CrossRef]
- Ferraz, E.G.; Silva, L.R.; Sarmento, V.A.; Campos, E.D.J.; De Oliveira, T.F.L.; Magalhães, J.C.; Paraguassú, G.M.; Sorte, N.C.B. Association between childhood obesity and oral hygiene status. Nutr. Hosp. 2014, 30, 253–259. [Google Scholar] [PubMed]
- Lehmann-Kalata, A.; Miechowicz, I.; Korybalska, K.; Swora-Cwynar, E.; Czepulis, N.; Łuczak, J.; Orzechowska, Z.; Grzymisławski, M.; Surdacka, A.; Witowski, J. Salivary fingerprint of simple obesity. Cytokine 2018, 110, 174–180. [Google Scholar] [CrossRef]
- Porcelli, I.C.D.S.; Corsi, N.M.; Fracasso, M.D.L.C.; Pascotto, R.C.; Cardelli, A.A.M.; Poli-Frederico, R.C.; Nasser, D.; Maciel, S.M. Oral Health Promotion in Patients with Morbid Obesity After Gastroplasty: A Randomized Clinical Trial. Arq. Bras. Cir. Dig. 2019, 32, e1437. [Google Scholar] [CrossRef] [PubMed]



| Category | BMI, kg/m2 | n |
|---|---|---|
| Underweight | <18.5 | 6 |
| Normal | [18.5–25) | 48 |
| Overweight | [25–30) | 11 |
| Obese class I | [30–35) | 13 |
| Obese class II | [35–40) | 9 |
| Obese class III | >40 | 7 |
| Parameter | All | BMI < 25 | BMI ≥ 25 | p-Value |
|---|---|---|---|---|
| DMF-T | 9.5 | 8.5 | 11.0 | 0.002 * |
| DMF-S | 13.0 | 11.0 | 19.5 | <0.001 * |
| Approximal plaque index, % | 60.0 | 40.0 | 80.0 | <0.001 * |
| Plaque index | 0.4 | 0.3 | 0.7 | 0.003 * |
| Gingival index | 0.3 | 0.1 | 0.5 | 0.013 * |
| Sulcus bleeding index, % | 7.7 | 1.9 | 15.4 | <0.001 * |
| Periodontal probing depth, mm | 0.9 | 0.8 | 1.1 | 0.029 * |
| Brushing frequency/day | 2.0 | 2.0 | 2.0 | 0.004 * |
| Brushing time, min | 2.0 | 3.0 | 2.0 | 0.010 * |
| Cleaning index | 6.0 | 6.0 | 4.0 | <0.001 * |
| Cytokine, pg/mL | All | BMI < 25 | BMI ≥ 25 | p-Value |
|---|---|---|---|---|
| TNFα-R1 | 154.6 | 98.4 | 230.6 | <0.001 * |
| TNFα-R2 | 53.0 | 42.6 | 71.1 | 0.026 * |
| PTX-3 | 314.2 | 246.5 | 386.8 | 0.005 * |
| IL-15 | 9.9 | 9.0 | 12.0 | 0.012 * |
| MCP-1 | 39.3 | 20.0 | 62.6 | 0.007 * |
| sICAM-1 | 418.1 | 325.8 | 596.9 | 0.002 * |
| sCD40L | 4.6 | 4.7 | 3.1 | 0.003 * |
| β | SE | WaldStat. | p-Value | Odds Ratio | Confidence OR −95% | Confidence OR 95% | |
|---|---|---|---|---|---|---|---|
| intercept | −3.537 | 1.054 | 11.269 | <0.001 | 0.029 | 0.004 | 0.229 |
| cleaning index > 4 | −1.913 | 0.601 | 10.128 | 0.001 | 0.148 | 0.045 | 0.480 |
| API > 40% | 2.087 | 0.678 | 9.471 | 0.002 | 8.061 | 2.134 | 30.457 |
| TNF-R1, pg/mL | 0.008 | 0.002 | 11.402 | <0.001 | 1.008 | 1.003 | 1.012 |
| IL-15, pg/mL | 0.136 | 0.054 | 6.350 | 0.012 | 1.145 | 1.031 | 1.273 |
| β | SE | Wald Stat. | p-Value | Odds Ratio | Confidence OR −95% | Confidence OR 95% | |
|---|---|---|---|---|---|---|---|
| intercept | −4.165 | 1.048 | 15.788 | <0.001 | 0.016 | 0.002 | 0.121 |
| cleaning index > 4 | −0.858 | 0.609 | 1.983 | 0.159 | 0.424 | 0.128 | 1.400 |
| API > 40% | 2.012 | 0.627 | 10.296 | 0.001 | 7.476 | 2.188 | 25.550 |
| TNF-R1, pg/ml | 0.007 | 0.002 | 11.350 | <0.001 | 1.007 | 1.003 | 1.011 |
| IL-15, pg/ml | 0.145 | 0.051 | 8.244 | 0.004 | 1.157 | 1.047 | 1.277 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nijakowski, K.; Lehmann, A.; Rutkowski, R.; Korybalska, K.; Witowski, J.; Surdacka, A. Poor Oral Hygiene and High Levels of Inflammatory Cytokines in Saliva Predict the Risk of Overweight and Obesity. Int. J. Environ. Res. Public Health 2020, 17, 6310. https://doi.org/10.3390/ijerph17176310
Nijakowski K, Lehmann A, Rutkowski R, Korybalska K, Witowski J, Surdacka A. Poor Oral Hygiene and High Levels of Inflammatory Cytokines in Saliva Predict the Risk of Overweight and Obesity. International Journal of Environmental Research and Public Health. 2020; 17(17):6310. https://doi.org/10.3390/ijerph17176310
Chicago/Turabian StyleNijakowski, Kacper, Anna Lehmann, Rafał Rutkowski, Katarzyna Korybalska, Janusz Witowski, and Anna Surdacka. 2020. "Poor Oral Hygiene and High Levels of Inflammatory Cytokines in Saliva Predict the Risk of Overweight and Obesity" International Journal of Environmental Research and Public Health 17, no. 17: 6310. https://doi.org/10.3390/ijerph17176310
APA StyleNijakowski, K., Lehmann, A., Rutkowski, R., Korybalska, K., Witowski, J., & Surdacka, A. (2020). Poor Oral Hygiene and High Levels of Inflammatory Cytokines in Saliva Predict the Risk of Overweight and Obesity. International Journal of Environmental Research and Public Health, 17(17), 6310. https://doi.org/10.3390/ijerph17176310







