Advancing Prevention of STIs by Developing Specific Serodiagnostic Targets: Trichomonas vginalis as a Model
Abstract
1. Introduction
2. Materials and Methods
2.1. Epitopes Unique to the T. vaginalis A, E and G Proteins
2.2. ACT-P2, a Truncated α-Actinin Protein Used for Screening of Patient Sera
2.3. Plasmid Encoding AEG::SOE2 and ACT-P2
2.4. Recombinant Proteins
2.5. Sera for Enzyme-Linked Immunosorbent Assay (ELISA) and Derivation of Positive/Negative (P/N) Scores
2.6. Mouse Anti-AEG::SOE2 Serum and Anti-T. vaginalis Serum and ELISA for Detecting Antibody to T. vaginalis
2.7. Reproducibility
3. Results
3.1. Epitopes of A, E, and G Unique to T. vaginalis and the AEG::SOE2 Protein Sequence
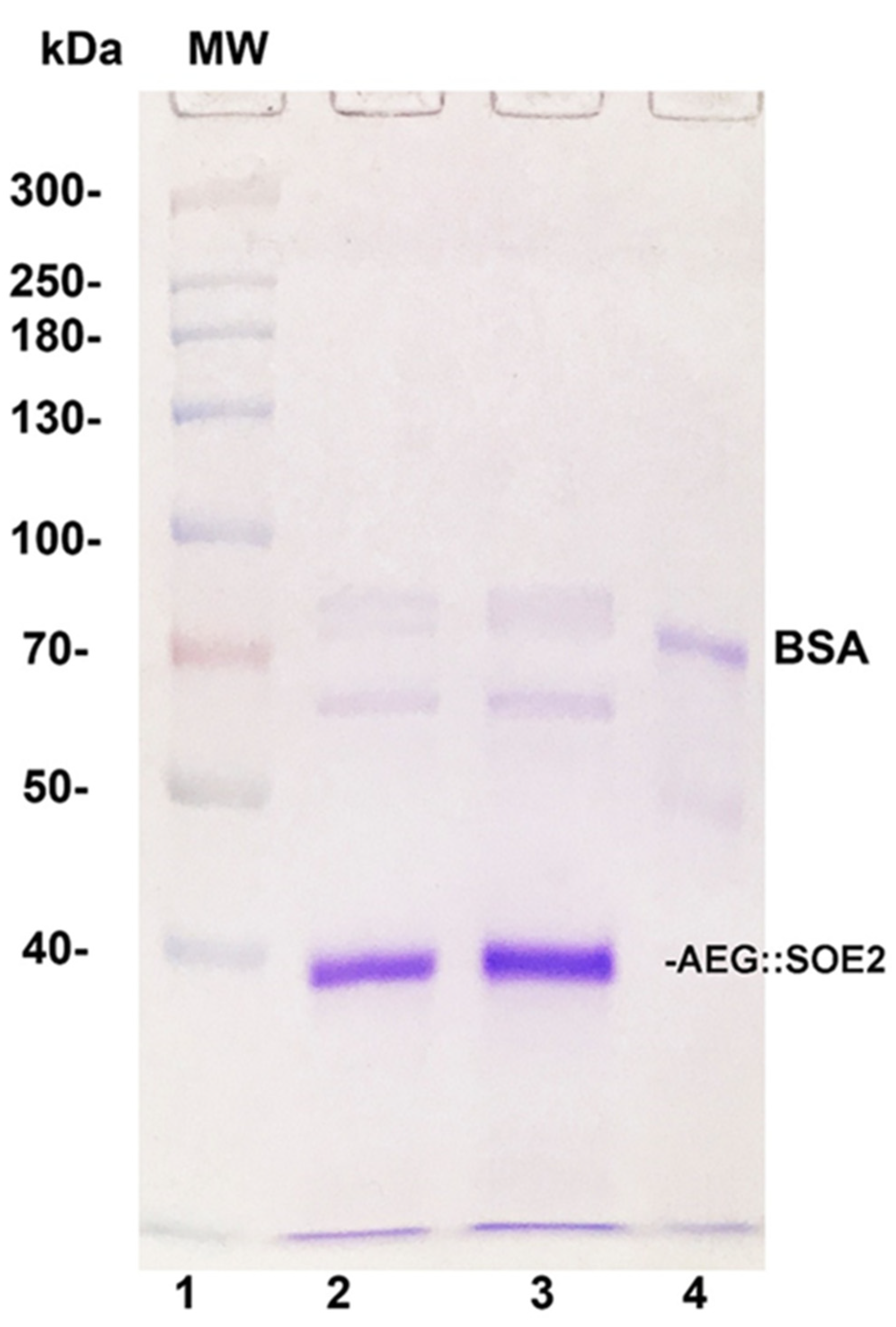
3.2. Purification of Recombinant AEG::SOE2
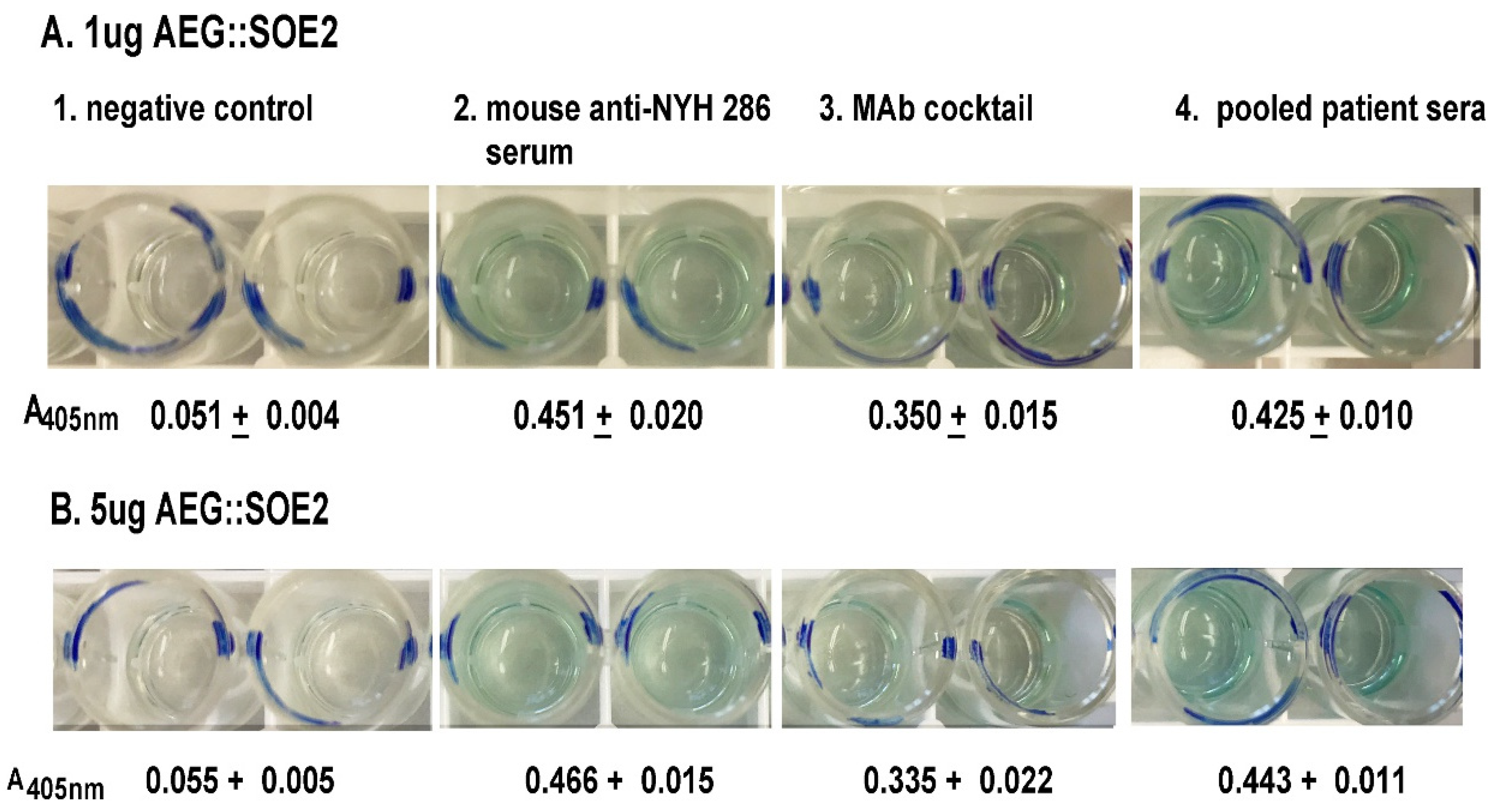
3.3. ELISA Using Different Amounts of AEG::SOE2-Coated Wells Probed with Different Antibodies
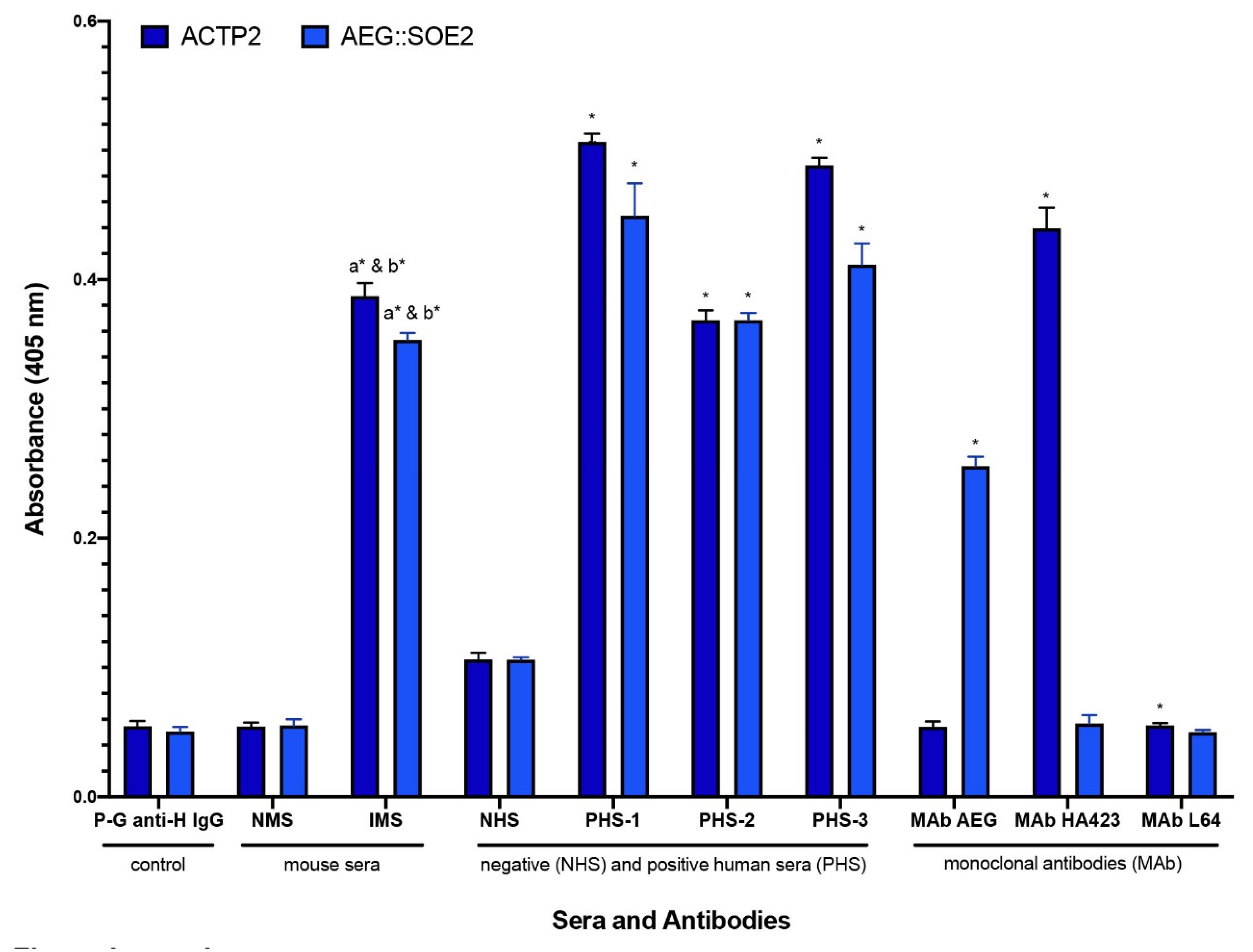
3.4. ELISA Comparing ACT-P2 and AEG::SOE2 with Different Antibodies
3.5. Different ELISA Experiments Testing Women and Men Sera of Different 0 to 4+ Scores
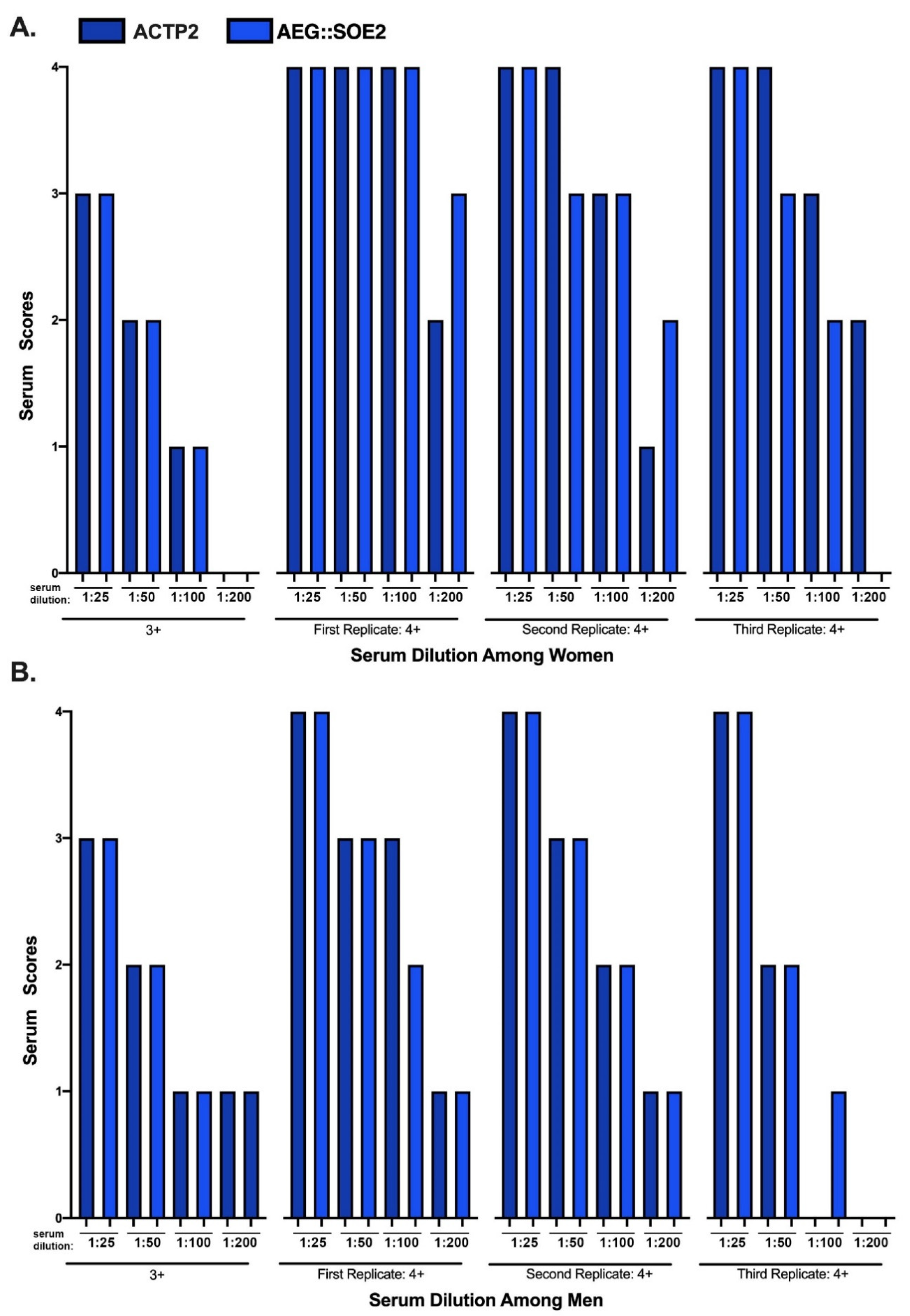
3.6. ELISA of Women and Men Sera of 3+ and 4+ Scores at Different Dilutions
3.7. ELISA Using ACT-P2 and AEG::SOE2 as Targets with 0 to 4+ Individual Sera
3.8. AEG::SOE2 is Immunogenic, and Anti-AEG::SOE2 Serum IgG Antibody Detects T. vaginalis Organisms Immobilized onto Microtiter Wells by ELISA
4. Discussion
5. Conclusions
6. Patent
Funding
Acknowledgments
Conflicts of Interest
References
- Hobbs, M.M.; Sena, A.C.; Swygard, H.; Schwebke, J.R. Trichomonas Vaginalis and Trichomoniasis. In Sexually Transmitted Diseases; Holmes, K.K., Sparling, P.F., Stamm, W.E., Piot, P., Wasserheit, J.N., Corey, L., Cohen, M.S., Watts, D.H., Eds.; McGraw-Hill MedicalHolmes: New York, NY, USA, 2008. [Google Scholar]
- Swygard, H.; Seña, A.C.; Hobbs, M.M.; Cohen, M.S. Trichomoniasis: Clinical manifestations, diagnosis and management. Sex. Transm. Infect. 2004, 80, 91–95. [Google Scholar] [CrossRef] [PubMed]
- Bachmann, L.H.; Hobbs, M.M.; Seña, A.C.; Sobel, J.D.; Schwebke, J.R.; Krieger, J.N.; McClelland, R.S.; Workowski, K.A. Trichomonas vaginalis genital infections: Progress and challenges. Clin. Infect. Dis. 2011, 53, S160–S172. [Google Scholar] [CrossRef] [PubMed]
- Krashin, J.W.; Koumans, E.H.; Bradshaw-Sydnor, A.C.; Braxton, J.R.; Secor, W.E.; Sawyer, M.K.; Markowitz, L.E. Trichomonas vaginalis prevalence, incidence, risk factors and antibiotic-resistance in an adolescent population. Sex. Transm. Dis. 2010, 37, 440–444. [Google Scholar] [CrossRef] [PubMed]
- Huppert, J.S. Trichomoniasis in teens: An update. Curr. Opin. Obstet. Gynecol. 2009, 21, 371–378. [Google Scholar] [CrossRef]
- Gardner, W.A., Jr.; Culberson, D.E.; Bennett, B.D. Trichomonas vaginalis in the prostate gland. Arch. Pathol. Lab. Med. 1986, 110, 430–432. [Google Scholar]
- Mitteregger, D.; Aberle, S.W.; Makristathis, A.; Walochnik, J.; Brozek, W.; Marberger, M.; Kramer, G. High detection rate of Trichomonas vaginalis in benign hyperplastic prostatic tissue. Med. Microbiol. Immunol. 2011, 201, 113–116. [Google Scholar] [CrossRef]
- Stark, J.R.; Judson, G.; Alderete, J.F.; Mundodi, V.; Kucknoor, A.S.; Giovannucci, E.L.; Platz, E.A.; Sutcliffe, S.; Fall, K.; Kurth, T.; et al. Prospective study of Trichomonas vaginalis infection and prostate cancer incidence and mortality: Physicians’ health study. J. Natl. Cancer Inst. 2009, 101, 1406–1411. [Google Scholar] [CrossRef]
- Sutcliffe, S.; Alderete, J.F.; Till, C.; Goodman, P.J.; Hsing, A.W.; Zenilman, J.M.; De Marzo, A.M.; Platz, E.A. Trichomonosis and subsequent risk of prostate cancer in the prostate cancer prevention trial. Int. J. Cancer 2009, 124, 2082–2087. [Google Scholar] [CrossRef]
- Sutcliffe, S.; Giovannucci, E.; Alderete, J.F.; Chang, T.-H.; Gaydos, C.A.; Zenilman, J.M.; De Marzo, A.M.; Willett, W.C.; Platz, E.A. Plasma antibodies against Trichomonas vaginalis and subsequent risk of prostate cancer. Cancer Epidemiol. Biomarkers Prev. 2006, 15, 939–945. [Google Scholar] [CrossRef]
- Sutcliffe, S.; Neace, C.; Magnuson, N.S.; Reeves, R.; Alderete, J.F. Trichomonosis, a common curable STI, and prostate carcinogenesis—A proposed molecular mechanism. PLOS Pathog. 2012, 8, e1002801. [Google Scholar] [CrossRef]
- Han, I.H.; Kim, J.H.; Kim, S.S.; Ahn, M.H.; Ryu, J.-S. Signalling pathways associated with IL-6 production and epithelial-mesenchymal transition induction in prostate epithelial cells stimulated with Trichomonas vaginalis. Parasite Immunol. 2016, 38, 678–687. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.-H.; Kim, S.-S.; Han, I.-H.; Sim, S.; Ahn, M.-H.; Ryu, J.-S. Proliferation of prostate stromal cell induced by benign prostatic hyperplasia epithelial cell stimulated with Trichomonas vaginalisvia crosstalk with mast cell. Prostate 2016, 76, 1431–1444. [Google Scholar] [CrossRef]
- Kim, J.-H.; Han, I.-H.; Kim, S.-S.; Park, S.-J.; Min, D.-Y.; Ahn, M.-H.; Ryu, J.-S. Interaction between Trichomonas vaginalis and the prostate epithelium. Korean J. Parasitol. 2017, 55, 213–218. [Google Scholar] [CrossRef] [PubMed]
- Twu, O.; Dessi’, D.; Vu, A.; Mercer, F.; Stevens, G.C.; De Miguel, N.; Rappelli, P.; Cocco, A.R.; Clubb, R.T.; Fiori, P.L.; et al. Trichomonas vaginalis homolog of macrophage migration inhibitory factor induces prostate cell growth, invasiveness, and inflammatory responses. Proc. Natl. Acad. Sci. USA 2014, 111, 8179–8184. [Google Scholar] [CrossRef] [PubMed]
- Pillay, A.; Lewis, J.; Ballard, R.C. Evaluation of Xenostrip-Tv, a rapid diagnostic test for Trichomonas vaginalis infection. J. Clin. Microbiol. 2004, 42, 3853–3856. [Google Scholar] [CrossRef]
- Lee, J.J.; Moon, H.S.; Lee, T.Y.; Hwang, H.S.; Ahn, M.H.; Ryu, J.S. PCR for diagnosis of male Trichomonas vaginalis infection with chronic prostatis and urethritis. Korean J. Parasitol. 2012, 50, 157–159. [Google Scholar] [CrossRef] [PubMed]
- Ginocchio, C.C.; Chapin, K.; Smith, J.S.; Aslanzadeh, J.; Snook, J.; Hill, C.S.; Gaydos, C.A. Prevalence of Trichomonas vaginalis and coinfection with chlamydia trachomatis and Neisseria gonorrhoeae in the United States as determined by the aptima Trichomonas vaginalis Nucleic acid amplification assay. J. Clin. Microbiol. 2012, 50, 2601–2608. [Google Scholar] [CrossRef]
- Yar, T.M.; Karakus, M.; Toz, S.; Karabulut, A.B.; Ozbel, Y.; Atambay, M. Diagnosis of trichomonosis in male patients on performing nested polymerase chain reaction. Turkiye Parzitol. Derg. 2017, 41, 130–134. [Google Scholar] [CrossRef]
- Toskin, I.; Murtagh, M.; Peeling, R.W.; Blondeel, K.; Cordero, J.P.; Kiarie, J. Advancing prevention of sexually transmitted infections through point-of-care testing: Target product profiles and landscape analysis. Sex. Transm. Infect. 2017, 93, S69–S80. [Google Scholar] [CrossRef]
- Alderete, J.F.; Neace, C.J. Identification, characterization, and synthesis of peptide epitopes and a recombinant six-epitope protein for Trichomonas vaginalis serodiagnosis. Immuno. Targets Ther. 2013, 2, 91–103. [Google Scholar] [CrossRef]
- Mundodi, V.; Kucknoor, A.S.; Alderete, J.F. α-Enolase is a surface-associated plasminogen-binding protein of Trichomonas vaginalis. Infect. Immun. 2008, 76, 523–531. [Google Scholar] [CrossRef] [PubMed]
- Lama, A.; Kucknoor, A.; Mundodi, V.; Alderete, J. Glyceraldehyde-3-phosphate dehydrogenase is a surface-associated, fibronectin-binding protein of Trichomonas vaginalis. Infect. Immun. 2009, 77, 2703–2711. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Neace, C.J.; Alderete, J.F. Epitopes of the highly immunogenic Trichomonas vaginalis—Actinin are serodiagnostic targets for both women and men. J. Clin. Microbiol. 2013, 51, 2483–2490. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Alderete, J. Epitopes within recombinant α-actinin protein is serodiagnostic target for Trichomonas vaginalis sexually transmitted infections. Heliyon 2017, 3, e00237. [Google Scholar] [CrossRef]
- Addis, M.F.; Rappelli, P.; De Andrade, A.M.P.; Rita, F.M.; Colombo, M.M.; Cappuccinelli, P.; Fiori, P.L. Identification of Trichomonas vaginalis? Actinin as the most common immunogen recognized by sera of women exposed to the parasite. J. Infect. Dis. 1999, 180, 1727–1730. [Google Scholar] [CrossRef]
- Addis, M.F.; Rappelli, P.; Delogu, G.; Carta, F.; Cappuccinelli, P.; Fiori, P.L. Cloning and molecular characterization of a cDNA clone coding for Trichomonas vaginalis alpha-actinin and intracellular localization of the protein. Infect. Immun. 1998, 66, 4924–4931. [Google Scholar] [CrossRef]
- Alderete, J.F.; Newton, E.; Dennis, C.; Neale, K.A. The vagina of women infected with Trichomonas vaginalis has numerous proteinases and antibody to trichomonad proteinases. Sex. Transm. Infect. 1991, 67, 469–474. [Google Scholar] [CrossRef][Green Version]
- Alderete, J.F.; Newton, E.; Dennis, C.; Neale, K.A. Antibody in sera of patients infected with Trichomonas vaginalis is to trichomonad proteinases. Sex. Transm. Infect. 1991, 67, 331–334. [Google Scholar] [CrossRef][Green Version]
- Alderete, J.F.; Newton, E.; Dennis, C.; Engbring, J.; Neale, K.A. Vaginal antibody of patients with trichomoniasis is to a prominent surface immunogen of Trichomonas vaginalis. Sex. Transm. Infect. 1991, 67, 220–225. [Google Scholar] [CrossRef][Green Version]
- Alderete, J.F. Antigen analysis of several pathogenic strains of Trichomonas vaginalis. Infect. Immun. 1983, 39, 1041–1047. [Google Scholar] [CrossRef]
- Cudmore, S.L.; Garber, G.E. Prevention or treatment: The benefits of Trichomonas vaginalis vaccine. J. Infect. Public Health. 2010, 3, 47–53. [Google Scholar] [CrossRef] [PubMed]
- Smith, J.; Garber, G.E. Current status and prospects for development of a vaccine against Trichomonas vaginalis infections. Vaccine 2014, 32, 1588–1594. [Google Scholar] [CrossRef] [PubMed]
- Sunblad, V.; Bussmann, L.; Chiauzzi, V.A.; Pancholi, V.; Charreau, E.H. Alpha-enolase: A novel autoantigen in patients with premature ovarian failure. Clin. Endocrinol. 2006, 65, 745–751. [Google Scholar] [CrossRef]
- Peterson, K.M.; Alderete, J.F. Host plasma proteins on the surface of pathogenic Trichomonas vaginalis. Infect. Immun. 1982, 37, 755–762. [Google Scholar] [CrossRef] [PubMed]
- Crouch, M.-L.; Alderete, J.F. Trichomonas vaginalis interactions with fibronectin and laminin. Microbiol. 1999, 145, 2835–2843. [Google Scholar] [CrossRef] [PubMed]
- Crouch, M.-L.; Benchimol, M.; Alderete, J. Binding of fibronectin by Trichomonas vaginalis is influenced by iron and calcium. Microb. Pathog. 2001, 31, 131–144. [Google Scholar] [CrossRef]
- Ibáñez-Escribano, A.; Nogal-Ruiz, J.J.; Serrano, J.P.; Barrio, G.; Escario, J.A.; Alderete, J. Sequestration of host-CD59 as potential immune evasion strategy of Trichomonas vaginalis. Acta. Trop. 2015, 149, 1–7. [Google Scholar] [CrossRef]
- Kim, S.R.; Kim, J.H.; Park, S.J.; Lee, H.Y.; Kim, Y.S.; Kim, Y.M.; Hong, Y.C.; Ryu, J.S. Comparison between mixed lysate antigen and α-actinin antigen in ELISA for serodiagnosis of trichomonosis. Parasitol. Int’l. 2015, 64, 405–407. [Google Scholar] [CrossRef]
- Dodet, B. Current barriers, challenges and opportunities for the development of effective STI vaccines: Point of view of vaccine producers, biotech companies and funding agencies. Vaccine 2014, 32, 1624–1629. [Google Scholar] [CrossRef]
- Xie, Y.-T.; Gao, J.-M.; Wu, Y.-P.; Tang, P.; Hide, G.; Lai, D.-H.; Lun, Z.-R. Recombinant α-actinin subunit antigens of Trichomonas vaginalis as potential vaccine candidates in protecting against trichomoniasis. Parasite. Vector. 2017, 10, 83. [Google Scholar] [CrossRef]

| Proteins | |||
|---|---|---|---|
| No. | A | E | G |
| 1 | HTYTRPEEVODFVSK | AEHDAIVKECIAEEAA | RACRKLYPKDIQVVA |
| 2 | LGEARTKLTEMYMRK | VKYLGRVTLAARSSA | NVYLLKYDTAHRAFP |
| 3 | VTLAARSSAPSGAST | QEFTVGEGADKWVVK | |
| 4 | TDGTVLKKNIGGNAC | WVVKSIGGRLGPSQL | |
| 5 | DKVPKKFKLPSPFFN | VVLESTGIFRTKAEK | |
| 6 | KLGGLLVKKYGLSAK | KDAEGKIKKDDGYDGH | |
| 7 | ILVKKYGLSAKNLDEF | TLNNAFGIRNGFMTTV | |
| 8 | ASSEFYDEEKKLYEV | ||
| 9 | DYENWTKLNARLGQRV |
| Expt | Serum Reaction Scores Against ACTP2: | |||||||||||||
| 0‡‡ | 0 | 1+ | 1+ | 2+ | 2+ | 3+ | 3+ | 4+ | 4+ | 4+ | 4+ | 4+ | 4+ | |
| W | M | W | M | W | M | W | M | W | M | W | M | W | M | |
| expt 1 | 0 | 0 | 1 | 1 | 2 | 2 | 3 | 3 | 4 | 4 | 4 | 4 | 4 | 4 |
| expt 2 | 0 | 0 | 1 | 1 | 2 | 2 | 3 | 3 | 4 | 4 | 4 | 4 | 4 | 4 |
| expt 3 | 0 | 0 | 1 | 1 | 2 | 2 | 3 | 3 | 4 | 4 | 4 | 4 | 4 | 4 |
| expt 4 | 0 | 0 | 1 | 1 | 2 | 2 | 3 | 3 | 4 | 4 | 4 | 4 | 4 | 4 |
| Expt | Serum Reaction Scores Against AEG::SOE2: | |||||||||||||
| 0 | 0 | 1+ | 1+ | 2+ | 2+ | 3+ | 3+ | 4+ | 4+ | 4+ | 4+ | 4+ | 4+ | |
| W | M | W | M | W | M | W | M | W | M | W | M | W | M | |
| expt 1 | 0 | 0 | 1 | 1 | 2 | 2 | 3 | 3 | 4 | 4 | 4 | 4 | 4 | 4 |
| expt 2 | 0 | 0 | 1 | 1 | 2 | 2 | 3 | 3 | 4 | 4 | 4 | 4 | 4 | 4 |
| expt 3 | 0 | 0 | 1 | 1 | 2 | 2 | 3 | 3 | 4 | 4 | 4 | 4 | 4 | 4 |
| expt 4 | 0 | 0 | 1 | 1 | 2 | 2 | 3 | 3 | 4 | 4 | 4 | 4 | 4 | 4 |
| No.‡ | Scores # | Scores | No. | Scores | Scores | No. | Scores | Scores | No. | Scores | Scores |
|---|---|---|---|---|---|---|---|---|---|---|---|
| AEG::SOE2 | ACTP2 | AEG::SOE2 | ACTP2 | AEG::SOE2 | ACTP2 | AEG::SOE2 | ACTP2 | ||||
| 1 | 0 | 0 | 22 | 4+ | 3++ | 43 | 4+ | 4+ | 64 | 3+ | 2 |
| 2 | 4+ | 4+ | 23 | 3+ | 3+ | 44 | 3+ | 3+ | 65 | 4+ | 3++ |
| 3 | 3+ | 2++ | 24 | 4+ | 4+ | 45 | 3+ | 3+ | 66 | 2+ | 2+ |
| 4 | 1+ | 2+ | 25 | 4+ | 4+ | 46 | 4+ | 3+ | 67 | 3+ | 3++ |
| 5 | 2+ | 3+ | 26 | 3++ | 3+ | 47 | 2+ | 2+ | 68 | 2+ | 2+ |
| 6 | 2+ | 2+ | 27 | 4+ | 3++ | 48 | 3++ | 3+ | 69 | 2+ | 2+ |
| 7 | 3+ | 4+ | 28 | 4+ | 3++ | 49 | 3+ | 3+ | 70 | 3+ | 3++ |
| 8 | 2+ | 2+ | 29 | 2+ | 2+ | 50 | 2+ | 3+ | 71 | 1+ | 1+ |
| 9 | 2+ | 2+ | 30 | 1+ | 1+ | 51 | 3+ | 2+ | 72 | 0 | 0 |
| 10 | 4+ | 4+ | 31 | 1+ | 1+ | 52 | 4+ | 3++ | 73 | 1+ | 1+ |
| 11 | 4+ | 3++ | 32 | 1+ | 2+ | 53 | 3+ | 2+ | 74 | 0 | 0 |
| 12 | 2+ | 3+ | 33 | 4+ | 3++ | 54 | 3+ | 1+ | 75 | 2+ | 3+ |
| 13 | 1+ | 34 | 4+ | 3+ | 55 | 3++ | 3+ | 76 | 0 | 1+ | |
| 14 | 4+ | 4+ | 35 | 3++ | 4+ | 56 | 2+ | 2+ | 77 | 4+ | 4+ |
| 15 | 0 | 0 | 36 | 3+ | 2+ | 57 | 3+ | 3++ | 78 | 3++ | 3++ |
| 16 | 2+ | 2+ | 37 | 3+ | 3+ | 58 | 4+ | 4+ | 79 | 3+ | 3+ |
| 17 | 4+ | 4+ | 38 | 3+ | 2+ | 59 | 3+ | 3+ | 80 | 3+ | 3+ |
| 18 | 3+ | 3+ | 39 | 3++ | 3+ | 60 | 4+ | 4+ | 81 | 4+ | 4+ |
| 19 | 4+ | 3++ | 40 | 1+ | 1+ | 61 | 4+ | 3++ | 82 | 3+ | 3+ |
| 20 | 3++ | 4+ | 41 | 2+ | 1+ | 62 | 3+ | 3+ | 83 | 4+ | 4+ |
| 21 | 4+ | 4+ | 42 | 3+ | 3+ | 63 | 3+ | 3+ | 84 | 4+ | 4+ |
| Experiment No. (Mean ± SD) | |||
|---|---|---|---|
| Antibodies * | 1 | 2 | 3 |
| goat anti-mouse IgG alone | 0.075 ± 0.001 ‡ | 0.123 ± 0.005 | 0.130 ± 0.015 |
| NMS (1:10) | 0.160 ± 0.001 | 0.157 ± 0.002 | 0.140 ± 0.010 |
| anti-T. vaginalis serum (1:10) | ND ‡‡ | 0.364 ± 0.005 | 0.455 ± 0.005 |
| anti-T. vaginalis serum (1:100) | 0.345 ± 0.005 | ND | 0.432 ± 0.001 |
| anti-AEG::SOE2 serum (1:10) | 0.225 ± 0.002 | ND | ND |
| anti-AEG::SOE2 serum (1:10) | 0.207 ±0.002 | 0.305 ± 0.005 | 0.190 ± 0.005 |
| MAb cocktail (1:1:1:1, v/v) | 0.275 ± 0.005 | 0.218 ± 0.005 | 0.228 ± 0.005 |
| MAb ALD30A | 0.335 ± 0.010 | 0.220 ± 0.010 | 0.253 ± 0.008 |
| MAb L64 | 0.055, 0.055 | 0.051, 0.052 | 0.051, 0.050 |
© 2020 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alderete, J.F. Advancing Prevention of STIs by Developing Specific Serodiagnostic Targets: Trichomonas vginalis as a Model. Int. J. Environ. Res. Public Health 2020, 17, 5783. https://doi.org/10.3390/ijerph17165783
Alderete JF. Advancing Prevention of STIs by Developing Specific Serodiagnostic Targets: Trichomonas vginalis as a Model. International Journal of Environmental Research and Public Health. 2020; 17(16):5783. https://doi.org/10.3390/ijerph17165783
Chicago/Turabian StyleAlderete, John F. 2020. "Advancing Prevention of STIs by Developing Specific Serodiagnostic Targets: Trichomonas vginalis as a Model" International Journal of Environmental Research and Public Health 17, no. 16: 5783. https://doi.org/10.3390/ijerph17165783
APA StyleAlderete, J. F. (2020). Advancing Prevention of STIs by Developing Specific Serodiagnostic Targets: Trichomonas vginalis as a Model. International Journal of Environmental Research and Public Health, 17(16), 5783. https://doi.org/10.3390/ijerph17165783





