Identification of Tools for the Functional and Subjective Assessment of Patients in an Aquatic Environment: A Systematic Review
Abstract
1. Introduction
2. Material and Methods
2.1. Data Sources and Searches
2.2. Study Selection
2.3. Data Extraction and Quality Assessment
2.4. Data Synthesis and Analysis
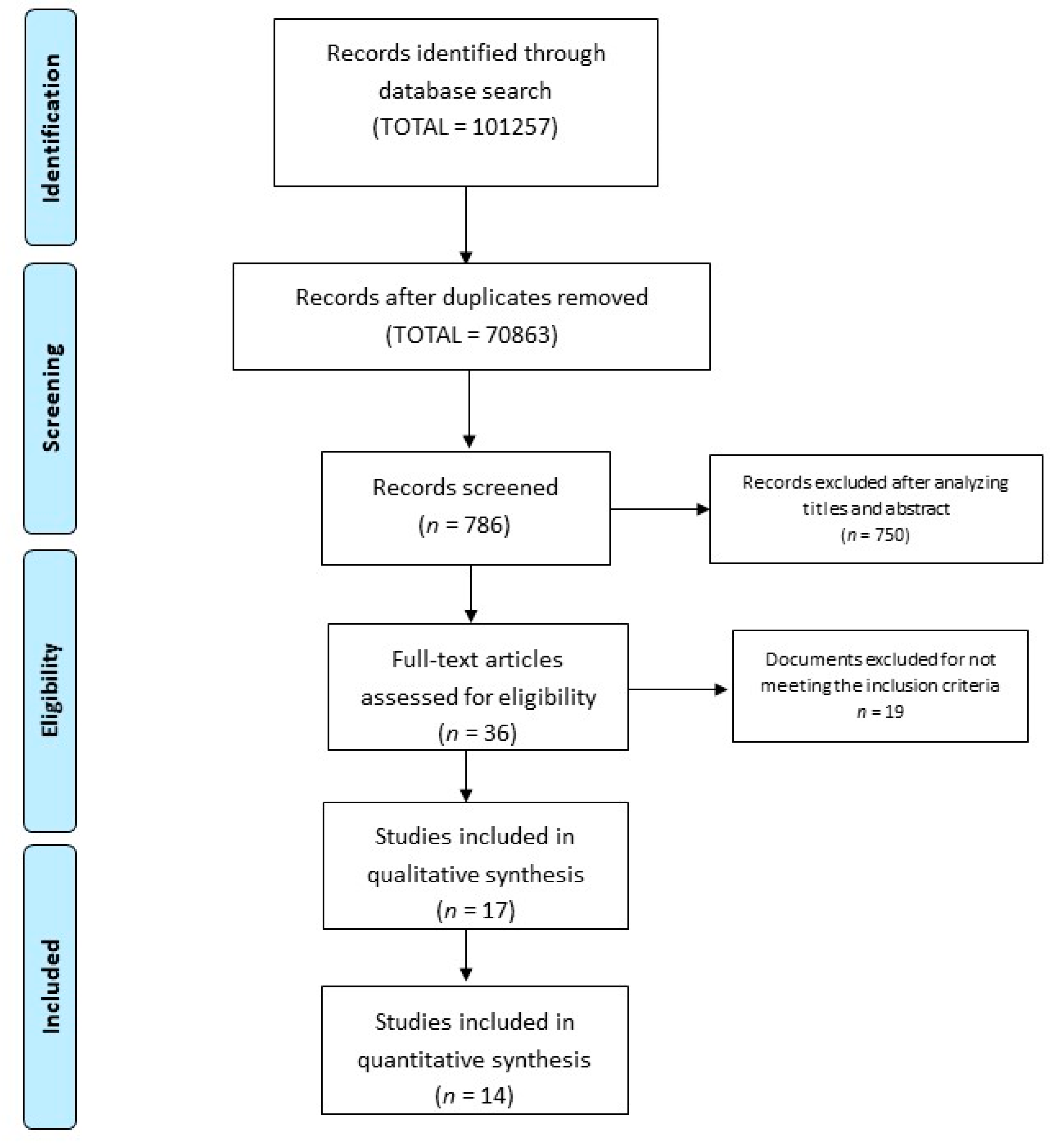
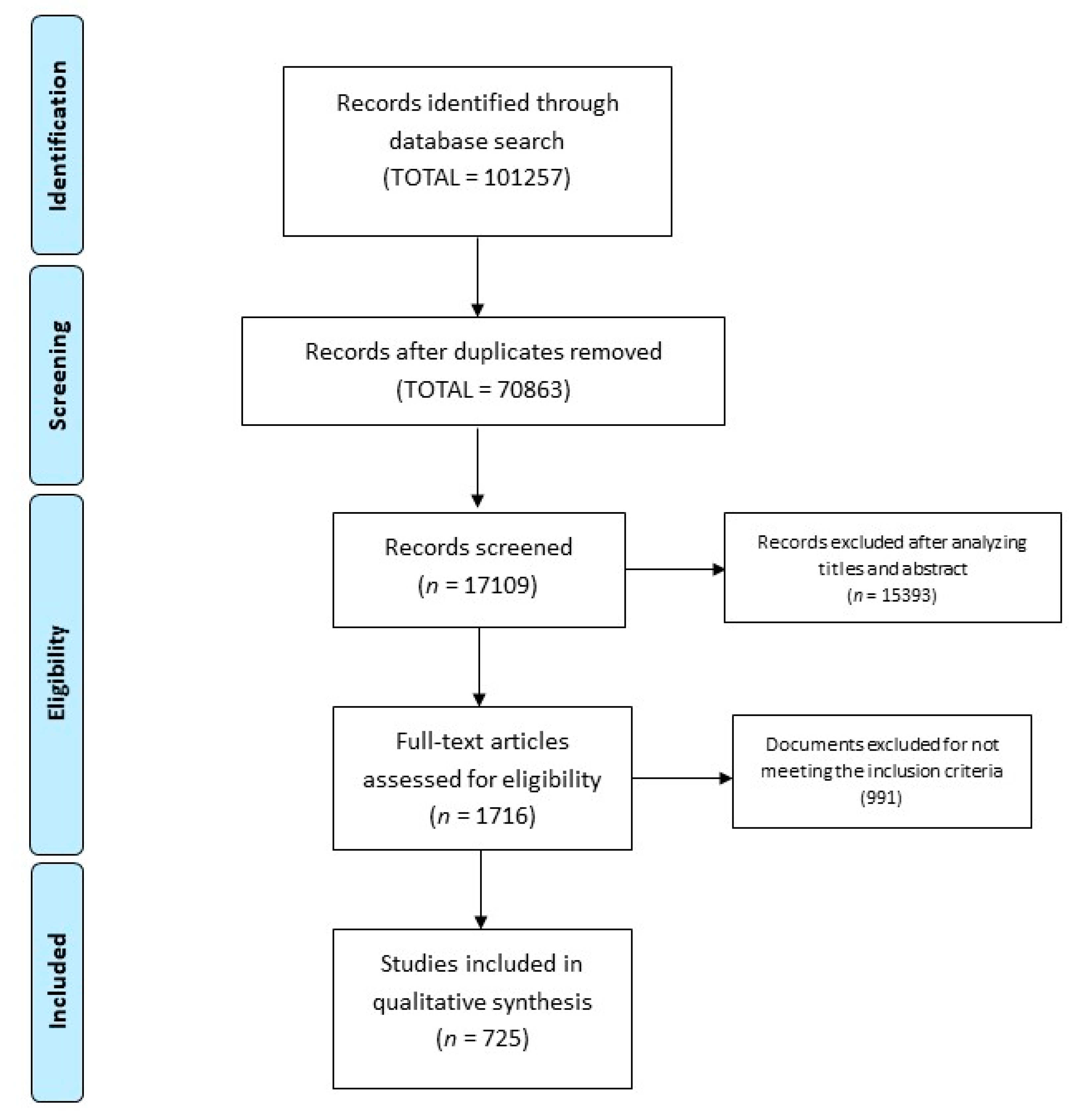
3. Results
4. Discussion
4.1. Walking
4.2. Deep Water Running
4.3. Shoulder Scaption
4.4. Sit to Stand
4.5. Time Get Up and Go
4.6. Jump
4.7. Questionnaire Assessment
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Mooventhan, A.; Nivethitha, L. Scientific evidence-based effects of hydrotherapy on various systems of the body. N. Am. J. Med. Sci. 2014, 6, 199–209. [Google Scholar] [CrossRef] [PubMed]
- Becker, B.E. Aquatic therapy: Scientific foundations and clinical rehabilitation applications. PM&R 2009, 1, 859–872. [Google Scholar]
- Simas, V.; Hing, W.; Pope, R.; Climstein, M. Effects of water-based exercise on bone health of middle-aged and older adults: A systematic review and meta-analysis. Open Access J. Sports Med. 2017, 8, 39–60. [Google Scholar] [CrossRef]
- Higgins, T.R.; Greene, D.A.; Baker, M.K. Effects of Cold Water Immersion and Contrast Water Therapy for Recovery from Team Sport: A Systematic Review and Meta-analysis. J. Strength Cond. Res. 2017, 31, 1443–1460. [Google Scholar] [CrossRef]
- Marinho-Buzelli, A.R.; Bonnyman, A.M.; Verrier, M.C. The effects of aquatic therapy on mobility of individuals with neurological diseases: A systematic review. Clin. Rehabil. 2015, 29, 741–751. [Google Scholar] [CrossRef] [PubMed]
- Basar, S.; Citaker, S.; Kanatli, U.; Ozturk, B.Y.; Kilickap, S.; Kafa, N.K. Assessment of function in patients with rotator cuff tears: Functional test versus self-reported questionnaire. Int. J. Shoulder Surg. 2014, 8, 107–113. [Google Scholar] [CrossRef]
- Tucher, G.; de Souza Castro, F.A.; da Silva, A.J.R.M.; Garrido, N.D. The Functional Test for Agility Performance is a Reliable Quick Decision-Making Test for Skilled Water Polo Players. J. Hum. Kinet. 2015, 46, 157–165. [Google Scholar] [CrossRef]
- Liang, Z.; Fu, C.; Zhang, Q.; Xiong, F.; Peng, L.; Chen, L.; He, C.; Wei, Q. Effects of water therapy on disease activity, functional capacity, spinal mobility and severity of pain in patients with ankylosing spondylitis: A systematic review and meta-analysis. Disabil. Rehabil. 2019, 1–8. [Google Scholar] [CrossRef]
- Karagülle, M.; Karagülle, M.Z. Effectiveness of balneotherapy and spa therapy for the treatment of chronic low back pain: A review on latest evidence. Clin. Rheumatol. 2015, 34, 207–214. [Google Scholar] [CrossRef]
- Santos, D.G.D.; Pegoraro, A.S.N.; Abrantes, C.V.; Jakaitis, F.; Gusman, S.; Bifulco, S.C. Evaluation of functional mobility of patients with stroke sequela after treatment in hydrotherapy pool using the Timed Up and Go Test. Einstein Sao Paulo Braz. 2011, 9, 302–306. [Google Scholar] [CrossRef]
- Araújo, J.P.; Neto, G.R.; Loenneke, J.P.; Bemben, M.G.; Laurentino, G.C.; Batista, G.; Silva, J.C.; Freitas, E.D.; Sousa, M.S. The effects of water-based exercise in combination with blood flow restriction on strength and functional capacity in post-menopausal women. Age Dordr. Neth. 2015, 37, 110. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Tripp, F.; Krakow, K. Effects of an aquatic therapy approach (Halliwick-Therapy) on functional mobility in subacute stroke patients: A randomized controlled trial. Clin. Rehabil. 2014, 28, 432–439. [Google Scholar] [CrossRef] [PubMed]
- Liberati, A.; Altman, D.G.; Tetzlaff, J.; Mulrow, C.; Gøtzsche, P.C.; Ioannidis, J.P.A.; Clarke, M.; Devereaux, P.J.; Kleijnen, J.; Moher, D. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: Explanation and elaboration. PLoS Med. 2009, 6, e1000100. [Google Scholar] [CrossRef]
- Bossuyt, P.M.; Reitsma, J.B.; Bruns, D.E.; Gatsonis, C.A.; Glasziou, P.P.; Irwig, L.; Lijmer, J.; Moher, D.; Rennie, D.; de Vet, H.; et al. STARD 2015: An updated list of essential items for reporting diagnostic accuracy studies. BMJ 2015, 351, h5527. [Google Scholar] [CrossRef] [PubMed]
- Cuesta-Vargas, A.I.; Heywood, S. Aerobic fitness testing in chronic nonspecific low back pain: A comparison of deep-water running with cycle ergometry. Am. J. Phys. Med. Rehabil. 2011, 90, 1030–1035. [Google Scholar] [CrossRef] [PubMed]
- Gayda, M.; Juneau, M.; Guiraud, T.; Lambert, J.; Nigam, A. Optimization and reliability of a deep water running test in healthy adults older than 45 years. Am. J. Phys. Med. Rehabil. 2010, 89, 722–730. [Google Scholar] [CrossRef] [PubMed]
- Chevutschi, A.; Lensel, G.; Vaast, D.; Thevenon, A. An electromyographic study of human gait both in water and on dry ground. J. Physiol. Anthropol. 2007, 26, 467–473. [Google Scholar] [CrossRef]
- Masumoto, K.; Hamada, A.; Tomonaga, H.; Kodama, K.; Hotta, N. Physiological responses, rating of perceived exertion, and stride characteristics during walking on dry land and walking in water, both with and without a water current. J. Sport Rehabil. 2012, 21, 175–181. [Google Scholar] [CrossRef]
- Masumoto, K.; Shono, T.; Hotta, N.; Fujishima, K. Muscle activation, cardiorespiratory response, and rating of perceived exertion in older subjects while walking in water and on dry land. J. Electromyogr. Kinesiol. 2008, 18, 581–590. [Google Scholar] [CrossRef]
- Masumoto, K.; Applequist, B.C.; Mercer, J.A. Muscle activity during different styles of deep water running and comparison to treadmill running at matched stride frequency. Gait Posture 2013, 37, 558–563. [Google Scholar] [CrossRef]
- Alberton, C.L.; Cadore, E.L.; Pinto, S.S.; Tartaruga, M.P.; da Silva, E.M.; Kruel, L.F.M. Cardiorespiratory, neuromuscular and kinematic responses to stationary running performed in water and on dry land. Eur. J. Appl. Physiol. 2011, 111, 1157–1166. [Google Scholar] [CrossRef] [PubMed]
- Mikos, V.; Yen, S.-C.; Tay, A.; Heng, C.-H.; Chung, C.L.H.; Liew, S.H.X.; Tan, D.M.L.; Au, W.L. Regression analysis of gait parameters and mobility measures in a healthy cohort for subject-specific normative values. PLoS ONE 2018, 13, e0199215. [Google Scholar] [CrossRef] [PubMed]
- Gimunová, M.; Zvonař, M.; Mikeska, O. The effect of aging and gender on plantar pressure distribution during the gait in elderly. Acta Bioeng. Biomech. 2018, 20, 139–144. [Google Scholar] [PubMed]
- Artaud, F.; Singh-Manoux, A.; Dugravot, A.; Tzourio, C.; Elbaz, A. Decline in Fast Gait Speed as a Predictor of Disability in Older Adults. J. Am. Geriatr. Soc. 2015, 63, 1129–1136. [Google Scholar] [CrossRef]
- Nagle, E.F.; Sanders, M.E.; Franklin, B.A. Aquatic High Intensity Interval Training for Cardiometabolic Health: Benefits and Training Design. Am. J. Lifestyle Med. 2016, 11, 64–76. [Google Scholar] [CrossRef] [PubMed]
- Masumoto, K.; Soucy, M.T.; Bailey, J.P.; Mercer, J.A. Muscle activity during backward and forward running with body weight support. Hum. Mov. Sci. 2017, 55, 276–286. [Google Scholar] [CrossRef] [PubMed]
- Castillo-Lozano, R.; Cuesta-Vargas, A.; Gabel, C.P. Analysis of arm elevation muscle activity through different movement planes and speeds during in-water and dry-land exercise. J. Shoulder Elbow Surg. 2014, 23, 159–165. [Google Scholar] [CrossRef] [PubMed]
- Cuesta-Vargas, A.I.; Cano-Herrera, C.L.; Heywood, S. Analysis of the neuromuscular activity during rising from a chair in water and on dry land. J. Electromyogr. Kinesiol. 2013, 23, 1446–1450. [Google Scholar] [CrossRef]
- Cuesta-Vargas, A.I.; Cano-Herrera, C.; Formosa, D.; Burkett, B. Electromyographic responses during time get up and go test in water (wTUG). SpringerPlus 2013, 2, 217. [Google Scholar] [CrossRef]
- Colado, J.C.; Garcia-Masso, X.; González, L.-M.; Triplett, N.T.; Mayo, C.; Merce, J. Two-leg squat jumps in water: An effective alternative to dry land jumps. Int. J. Sports Med. 2010, 31, 118–122. [Google Scholar] [CrossRef]
- Louder, T.; Dolny, D.; Bressel, E. Biomechanical Comparison of Countermovement Jumps Performed on Land and in Water: Age Effects. J. Sport Rehabil. 2018, 27, 249–256. [Google Scholar] [CrossRef]
- Sattler, M.C.; Jaunig, J.; Tösch, C.; Watson, E.D.; Mokkink, L.B.; Dietz, P.; van Poppel, M.N.M. Current Evidence of Measurement Properties of Physical Activity Questionnaires for Older Adults: An Updated Systematic Review. Sports Med. 2020, 50, 1271–1315. [Google Scholar] [CrossRef] [PubMed]
- Souza, A.C.D.; Alexandre, N.M.C.; Guirardello, E.D.B. Psychometric properties in instruments evaluation of reliability and validity. Epidemiol. Serv. Saúde 2017, 26, 649–659. [Google Scholar] [CrossRef]
- Stevenson, T.J. Detecting change in patients with stroke using the Berg Balance Scale. Aust. J. Physiother. 2001, 47, 29–38. [Google Scholar] [CrossRef]
- Huang, T.-T.; Wang, W.-S. Comparison of three established measures of fear of falling in community-dwelling older adults: Psychometric testing. Int. J. Nurs. Stud. 2009, 46, 1313–1319. [Google Scholar] [CrossRef] [PubMed]
- Lemay, J.-F.; Nadeau, S. Standing balance assessment in ASIA D paraplegic and tetraplegic participants: Concurrent validity of the Berg Balance Scale. Spinal Cord 2010, 48, 245–250. [Google Scholar] [CrossRef] [PubMed]
- Bernspång, B.; Fisher, A.G. Differences between persons with right or left cerebral vascular accident on the Assessment of Motor and Process Skills. Arch. Phys. Med. Rehabil. 1995, 76, 1144–1151. [Google Scholar] [CrossRef]
- Nemmers, T.M.; Miller, J.W. Factors influencing balance in healthy community-dwelling women age 60 and older. J. Geriatr. Phys. Ther. 2008, 31, 93–100. [Google Scholar] [CrossRef]
- Richardson, J.; Iezzi, A.; Khan, M.A.; Maxwell, A. Validity and reliability of the Assessment of Quality of Life (AQoL)-8D multi-attribute utility instrument. Patient 2014, 7, 85–96. [Google Scholar] [CrossRef]
- Harrison, J.E.; Preston, S.; Blunt, S.B. Measuring symptom change in patients with Parkinson’s disease. Age Ageing 2000, 29, 41–45. [Google Scholar] [CrossRef]
- Tripathy, S.; Myatra, S.N. Are the instruments for quality of life assessment comparable between cultures? No. Intensive Care Med. 2020. [Google Scholar] [CrossRef] [PubMed]
- Lajoie, Y.; Gallagher, S.P. Predicting falls within the elderly community: Comparison of postural sway, reaction time, the Berg balance scale and the Activities-specific Balance Confidence (ABC) scale for comparing fallers and non-fallers. Arch. Gerontol. Geriatr. 2004, 38, 11–26. [Google Scholar] [CrossRef]
- Berg, K.O.; Wood-Dauphinee, S.L.; Williams, J.I.; Maki, B. Measuring balance in the elderly: Validation of an instrument. Can. J. Public Health 1992, 83 (Suppl. 2), S7–S11. [Google Scholar] [PubMed]
- Delbaere, K.; Close, J.C.T.; Mikolaizak, A.S.; Sachdev, P.S.; Brodaty, H.; Lord, S.R. The Falls Efficacy Scale International (FES-I). A comprehensive longitudinal validation study. Age Ageing 2010, 39, 210–216. [Google Scholar] [CrossRef]
- Botner, E.M.; Miller, W.C.; Eng, J.J. Measurement properties of the Activities-specific Balance Confidence Scale among individuals with stroke. Disabil. Rehabil. 2005, 27, 156–163. [Google Scholar] [CrossRef]
- Lohnes, C.A.; Earhart, G.M. External validation of abbreviated versions of the activities-specific balance confidence scale in Parkinson’s disease. Mov. Disord. 2010, 25, 485–489. [Google Scholar] [CrossRef]
- Peretz, C.; Herman, T.; Hausdorff, J.M.; Giladi, N. Assessing fear of falling: Can a short version of the Activities-specific Balance Confidence scale be useful? Mov. Disord. 2006, 21, 2101–2105. [Google Scholar] [CrossRef]
- Wrisley, D.M.; Kumar, N.A. Functional gait assessment: Concurrent, discriminative, and predictive validity in community-dwelling older adults. Phys. Ther. 2010, 90, 761–773. [Google Scholar] [CrossRef]
- Landers, M.R.; Durand, C.; Powell, D.S.; Dibble, L.E.; Young, D.L. Development of a scale to assess avoidance behavior due to a fear of falling: The Fear of Falling Avoidance Behavior Questionnaire. Phys. Ther. 2011, 91, 1253–1265. [Google Scholar] [CrossRef]
- Hatch, J.; Gill-Body, K.M.; Portney, L.G. Determinants of balance confidence in community-dwelling elderly people. Phys. Ther. 2003, 83, 1072–1079. [Google Scholar] [CrossRef]
- Dal Bello-Haas, V.; Klassen, L.; Sheppard, M.S.; Metcalfe, A. Psychometric Properties of Activity, Self-Efficacy, and Quality-of-Life Measures in Individuals with Parkinson Disease. Physiother. Can. 2011, 63, 47–57. [Google Scholar] [CrossRef] [PubMed]
- Steffen, T.; Seney, M. Test-retest reliability and minimal detectable change on balance and ambulation tests, the 36-item short-form health survey, and the unified Parkinson disease rating scale in people with parkinsonism. Phys. Ther. 2008, 88, 733–746. [Google Scholar] [CrossRef] [PubMed]
- Meenan, R.F.; Mason, J.H.; Anderson, J.J.; Guccione, A.A.; Kazis, L.E. AIMS2. The content and properties of a revised and expanded Arthritis Impact Measurement Scales Health Status Questionnaire. Arthritis Rheum. 1992, 35, 1–10. [Google Scholar] [CrossRef]
- Guillemin, F.; Coste, J.; Pouchot, J.; Ghézail, M.; Bregeon, C.; Sany, J. The AIMS2-SF: A short form of the Arthritis Impact Measurement Scales 2. French Quality of Life in Rheumatology Group. Arthritis Rheum. 1997, 40, 1267–1274. [Google Scholar] [PubMed]
- Atamaz, F.; Hepguler, S.; Oncu, J. Translation and validation of the Turkish version of the arthritis impact measurement scales 2 in patients with knee osteoarthritis. J. Rheumatol. 2005, 32, 1331–1336. [Google Scholar] [PubMed]
- Spies-Dorgelo, M.N.; Terwee, C.B.; Stalman, W.A.B.; van der Windt, D.A.W.M. Reproducibility and responsiveness of the Symptom Severity Scale and the hand and finger function subscale of the Dutch arthritis impact measurement scales (Dutch-AIMS2-HFF) in primary care patients with wrist or hand problems. Health Qual. Life Outcomes 2006, 4, 87. [Google Scholar] [CrossRef] [PubMed]
- Haslam, J.; Pépin, G.; Bourbonnais, R.; Grignon, S. Processes of task performance as measured by the Assessment of Motor and Process Skills (AMPS): A predictor of work-related outcomes for adults with schizophrenia? Work 2010, 37, 53–64. [Google Scholar] [CrossRef] [PubMed]
- Pan, A.W.; Fisher, A.G. The Assessment of Motor and Process Skills of persons with psychiatric disorders. Am. J. Occup. Ther. 1994, 48, 775–780. [Google Scholar] [CrossRef] [PubMed]
- Fisher, A.G.; Jones, K.B. Assessment of Motor and Process Skills: Volume 1: Development, Standardization, and Administration Manual; Three Star Press: Fort Collins, CO, USA, 2010. [Google Scholar]
- Lomi, C.; Nordholm, L.A. Validation of a Swedish version of the Arthritis Self-efficacy Scale. Scand. J. Rheumatol. 1992, 21, 231–237. [Google Scholar] [CrossRef]
- Merritt, B.K. Validity of using the Assessment of Motor and Process Skills to determine the need for assistance. Am. J. Occup. Ther. 2011, 65, 643–650. [Google Scholar] [CrossRef]
- Peny-Dahlstrand, M.; Gosman-Hedström, G.; Krumlinde-Sundholm, L. Are there cross-cultural differences of ADL ability in children measured with the Assessment of Motor and Process Skills (AMPS)? Scand. J. Occup. Ther. 2012, 19, 26–32. [Google Scholar] [CrossRef] [PubMed]
- Hawthorne, G.; Richardson, J.; Osborne, R. The Assessment of Quality of Life (AQoL) instrument: A psychometric measure of health-related quality of life. Qual. Life Res. 1999, 8, 209–224. [Google Scholar] [CrossRef] [PubMed]
- Lorig, K.; Chastain, R.L.; Ung, E.; Shoor, S.; Holman, H.R. Development and evaluation of a scale to measure perceived self-efficacy in people with arthritis. Arthritis Rheum. 1989, 32, 37–44. [Google Scholar] [CrossRef] [PubMed]
- Barlow, J.H.; Williams, B.; Wright, C.C. The reliability and validity of the arthritis self-efficacy scale in a UK context. Psychol. Health Med. 1997, 2, 3–17. [Google Scholar] [CrossRef]
- Flansbjer, U.-B.; Blom, J.; Brogårdh, C. The reproducibility of Berg Balance Scale and the Single-leg Stance in chronic stroke and the relationship between the two tests. Pm&R 2012, 4, 165–170. [Google Scholar]
- Newstead, A.H.; Hinman, M.R.; Tomberlin, J.A. Reliability of the Berg Balance Scale and balance master limits of stability tests for individuals with brain injury. J. Neurol. Phys. Ther. JNPT 2005, 29, 18–23. [Google Scholar] [CrossRef]
- Chou, C.-Y.; Chien, C.-W.; Hsueh, I.-P.; Sheu, C.-F.; Wang, C.-H.; Hsieh, C.-L. Developing a short form of the Berg Balance Scale for people with stroke. Phys. Ther. 2006, 86, 195–204. [Google Scholar] [CrossRef]
- Bogle Thorbahn, L.D.; Newton, R.A. Use of the Berg Balance Test to predict falls in elderly persons. Phys. Ther. 1996, 76, 576–583; discussion 584–585. [Google Scholar] [CrossRef]
- Shumway-Cook, A.; Baldwin, M.; Polissar, N.L.; Gruber, W. Predicting the probability for falls in community-dwelling older adults. Phys. Ther. 1997, 77, 812–819. [Google Scholar] [CrossRef]
- Liston, R.A.; Brouwer, B.J. Reliability and validity of measures obtained from stroke patients using the Balance Master. Arch. Phys. Med. Rehabil. 1996, 77, 425–430. [Google Scholar] [CrossRef]
- Edmunds, S.; Garratt, A.; Haines, L.; Blair, M. Child Health Assessment at School Entry (CHASE) project: Evaluation in 10 London primary schools. Child. Care Health Dev. 2005, 31, 143–154. [Google Scholar] [CrossRef] [PubMed]
- Launois, R.; Reboul-Marty, J.; Henry, B. Construction and validation of a quality of life questionnaire in chronic lower limb venous insufficiency (CIVIQ). Qual. Life Res. 1996, 5, 539–554. [Google Scholar] [CrossRef] [PubMed]
- Erevnidou, K.; Launois, R.; Katsamouris, A.; Lionis, C. Translation and validation of a quality of life questionnaire for chronic lower limb venous insufficiency into greek. Int. Angiol. 2004, 23, 394–399. [Google Scholar]
- Shapiro, J.P.; Welker, C.J.; Jacobson, B.J. The Youth Client Satisfaction Questionnaire: Development, construct validation, and factor structure. J. Clin. Child. Psychol. 1997, 26, 87–98. [Google Scholar] [CrossRef]
- Matsubara, C.; Green, J.; Astorga, L.T.; Daya, E.L.; Jervoso, H.C.; Gonzaga, E.M.; Jimba, M. Reliability tests and validation tests of the client satisfaction questionnaire (CSQ-8) as an index of satisfaction with childbirth-related care among Filipino women. BMC Pregnancy Childbirth 2013, 13, 235. [Google Scholar] [CrossRef]
- Siemons, L.; ten Klooster, P.M.; Vonkeman, H.E.; van de Laar, M.A.F.J.; Glas, C.A.W. Further optimization of the reliability of the 28-joint disease activity score in patients with early rheumatoid arthritis. PLoS ONE 2014, 9, e100544. [Google Scholar] [CrossRef] [PubMed]
- Leeb, B.F.; Andel, I.; Sautner, J.; Fassl, C.; Nothnagl, T.; Rintelen, B. The Disease Activity Score in 28 joints in rheumatoid arthritis and psoriatic arthritis patients. Arthritis Rheum. 2007, 57, 256–260. [Google Scholar] [CrossRef] [PubMed]
- Leeb, B.F.; Bird, H.A. A disease activity score for polymyalgia rheumatica. Ann. Rheum. Dis. 2004, 63, 1279–1283. [Google Scholar] [CrossRef] [PubMed]
- Salaffi, F.; Ciapetti, A.; Carotti, M.; Gasparini, S.; Citera, G.; Gutierrez, M. Construct validity and responsiveness of the simplified version of Ankylosing Spondylitis Disease Activity Score (SASDAS) for the evaluation of disease activity in axial spondyloarthritis. Health Qual. Life Outcomes 2014, 12, 129. [Google Scholar] [CrossRef]
- Van Agt, H.M.; Essink-Bot, M.L.; Krabbe, P.F.; Bonsel, G.J. Test-retest reliability of health state valuations collected with the EuroQol questionnaire. Soc. Sci. Med. 1994, 39, 1537–1544. [Google Scholar] [CrossRef]
- Brazier, J.; Jones, N.; Kind, P. Testing the validity of the Euroqol and comparing it with the SF-36 health survey questionnaire. Qual. Life Res. 1993, 2, 169–180. [Google Scholar] [CrossRef]
- Golicki, D.; Niewada, M.; Karlińska, A.; Buczek, J.; Kobayashi, A.; Janssen, M.F.; Pickard, A.S. Comparing responsiveness of the EQ-5D-5L, EQ-5D-3L and EQ VAS in stroke patients. Qual. Life Res. 2015, 24, 1555–1563. [Google Scholar] [CrossRef] [PubMed]
- Hauer, K.; Yardley, L.; Beyer, N.; Kempen, G.; Dias, N.; Campbell, M.; Becker, C.; Tood, C. Validation of the Falls Efficacy Scale and Falls Efficacy Scale International in geriatric patients with and without cognitive impairment: Results of self-report and interview-based questionnaires. Gerontology 2010, 56, 190–199. [Google Scholar] [CrossRef]
- Dewan, N.; MacDermid, J.C. Fall Efficacy Scale-International (FES-I). J. Physiother. 2014, 60, 60. [Google Scholar] [CrossRef] [PubMed]
- Van de Vliet, P.; Rintala, P.; Fröjd, K.; Verellen, J.; van Houtte, S.; Daly, D.J.; Vanlandewijck, Y.C. Physical fitness profile of elite athletes with intellectual disability. Scand. J. Med. Sci. Sports. 2006, 16, 417–425. [Google Scholar] [CrossRef]
- Visschedijk, J.H.M.; Terwee, C.B.; Caljouw, M.A.A.; van Eijk, M.S.; van Balen, R.; Achterberg, W.P. Reliability and validity of the Falls Efficacy Scale-International after hip fracture in patients aged ≥ 65 years. Disabil. Rehabil. 2015, 37, 2225–2232. [Google Scholar] [CrossRef]
- Burckhardt, C.S.; Clark, S.R.; Bennett, R.M. The fibromyalgia impact questionnaire: Development and validation. J. Rheumatol. 1991, 18, 728–733. [Google Scholar] [PubMed]
- Sarzi-Puttini, P.; Atzeni, F.; Fiorini, T.; Panni, B.; Randisi, G.; Turiel, M.; Carraba, M. Validation of an Italian version of the Fibromyalgia Impact Questionnaire (FIQ-I). Clin. Exp. Rheumatol. 2003, 21, 459–464. [Google Scholar]
- Osada, K.; Oka, H.; Isomura, T.; Nakamura, I.; Tominaga, K.; Takahashi, S.; Kojima, A.; Nishioka, K. Development of the Japanese version of the Fibromyalgia Impact Questionnaire (JFIQ): Psychometric assessments of reliability and validity. Int. J. Rheum. Dis. 2011, 14, 74–80. [Google Scholar] [CrossRef]
- Rivera, J.; González, T. The Fibromyalgia Impact Questionnaire: A validated Spanish version to assess the health status in women with fibromyalgia. Clin. Exp. Rheumatol. 2004, 22, 554–560. [Google Scholar]
- Spitzer, R.L.; Kroenke, K.; Williams, J.B.W.; Löwe, B. A brief measure for assessing generalized anxiety disorder: The GAD-7. Arch. Intern. Med. 2006, 166, 1092–1097. [Google Scholar] [CrossRef]
- Maska, L.; Anderson, J.; Michaud, K. Measures of functional status and quality of life in rheumatoid arthritis: Health Assessment Questionnaire Disability Index (HAQ), Modified Health Assessment Questionnaire (MHAQ), Multidimensional Health Assessment Questionnaire (MDHAQ), Health Assessment Questionnaire II (HAQ-II), Improved Health Assessment Questionnaire (Improved HAQ), and Rheumatoid Arthritis Quality of Life (RAQoL). Arthritis Care Res. 2011, 63 (Suppl. 11), S4–S13. [Google Scholar]
- Wolfe, F. A reappraisal of HAQ disability in rheumatoid arthritis. Arthritis Rheum. 2000, 43, 2751–2761. [Google Scholar] [CrossRef]
- Cole, J.C.; Motivala, S.J.; Khanna, D.; Lee, J.Y.; Paulus, H.E.; Irwin, M.R. Validation of single-factor structure and scoring protocol for the Health Assessment Questionnaire-Disability Index. Arthritis Rheum. 2005, 53, 536–542. [Google Scholar] [CrossRef] [PubMed]
- Salavati, M.; Akhbari, B.; Mohammadi, F.; Mazaheri, M.; Khorrami, M. Knee injury and Osteoarthritis Outcome Score (KOOS); reliability and validity in competitive athletes after anterior cruciate ligament reconstruction. Osteoarthr. Cartil. 2011, 19, 406–410. [Google Scholar] [CrossRef]
- Van de Graaf, V.A.; Wolterbeek, N.; Scholtes, V.A.B.; Mutsaerts, E.L.A.R.; Poolman, R.W. Reliability and Validity of the IKDC, KOOS, and WOMAC for Patients with Meniscal Injuries. Am. J. Sports Med. 2014, 42, 1408–1416. [Google Scholar] [CrossRef]
- Abdolahi, A.; Scoglio, N.; Killoran, A.; Dorsey, E.R.; Biglan, K.M. Potential reliability and validity of a modified version of the Unified Parkinson’s Disease Rating Scale that could be administered remotely. Parkinsonism Relat. Disord. 2013, 19, 218–221. [Google Scholar] [CrossRef] [PubMed]
- Vanbellingen, T.; Nyffeler, T.; Nef, T.; Kwakkel, G.; Bohlhalter, S.; van Wegen, E.E.H. Reliability and validity of a new dexterity questionnaire (DextQ-24) in Parkinson’s disease. Parkinsonism Relat. Disord. 2016, 33, 78–83. [Google Scholar] [CrossRef]
- Martinez-Martin, P.; Chaudhuri, K.R.; Rojo-Abuin, J.M.; Rodriguez-Blazquez, C.; Alvarez-Sanchez, M.; Arakaki, T.; Bergareche-Yarza, A.; Chade, A.; Garretto, N.; Gershanik, O.; et al. Assessing the non-motor symptoms of Parkinson’s disease: MDS-UPDRS and NMS Scale. Eur. J. Neurol. 2015, 22, 37–43. [Google Scholar] [CrossRef]
- Monticone, M.; Ferrante, S.; Ambrosini, E.; Rocca, B.; Secci, C.; Foti, C. Development of the Tampa Scale of Kinesiophobia for Parkinson’s disease: Confirmatory factor analysis, reliability, validity and sensitivity to change. Int. J. Rehabil. Res. 2015, 38, 113–120. [Google Scholar] [CrossRef]
- Horváth, K.; Aschermann, Z.; Ács, P.; Deli, G.; Janszky, J.; Komoly, S.; Balazs, E.; Takacs, K.; Karadi, K.; Kovacs, N. Minimal clinically important difference on the Motor Examination part of MDS-UPDRS. Parkinsonism Relat. Disord. 2015, 21, 1421–1426. [Google Scholar] [CrossRef] [PubMed]
- Washburn, R.A.; McAuley, E.; Katula, J.; Mihalko, S.L.; Boileau, R.A. The physical activity scale for the elderly (PASE): Evidence for validity. J. Clin. Epidemiol. 1999, 52, 643–651. [Google Scholar] [CrossRef]
- Svege, I.; Kolle, E.; Risberg, M.A. Reliability and validity of the Physical Activity Scale for the Elderly (PASE) in patients with hip osteoarthritis. BMC Musculoskelet. Disord. 2012, 13, 26. [Google Scholar] [CrossRef] [PubMed]
- Harada, N.D.; Chiu, V.; King, A.C.; Stewart, A.L. An evaluation of three self-report physical activity instruments for older adults. Med. Sci. Sports Exerc. 2001, 33, 962–970. [Google Scholar] [CrossRef] [PubMed]
- Ziropada, L.; Stefanova, E.; Potrebić, A.; Kostić, V.S. Quality of life in Serbian patients with Parkinson’s disease. Qual. Life Res. 2009, 18, 833–839. [Google Scholar] [CrossRef]
- Jenkinson, C.; Fitzpatrick, R.; Norquist, J.; Findley, L.; Hughes, K. Cross-cultural evaluation of the Parkinson’s Disease Questionnaire: Tests of data quality, score reliability, response rate, and scaling assumptions in the United States, Canada, Japan, Italy, and Spain. J. Clin. Epidemiol. 2003, 56, 843–847. [Google Scholar] [CrossRef]
- Schrag, A.; Selai, C.; Jahanshahi, M.; Quinn, N.P. The EQ-5D—A generic quality of life measure-is a useful instrument to measure quality of life in patients with Parkinson’s disease. J. Neurol. Neurosurg. Psychiatry 2000, 69, 67–73. [Google Scholar] [CrossRef] [PubMed]
- Peto, V.; Jenkinson, C.; Fitzpatrick, R. Determining minimally important differences for the PDQ-39 Parkinson’s disease questionnaire. Age Ageing 2001, 30, 299–302. [Google Scholar] [CrossRef]
- Speksnijder, C.M.; Koppenaal, T.; Knottnerus, J.A.; Spigt, M.; Staal, J.B.; Terwee, C.B. Measurement Properties of the Quebec Back Pain Disability Scale in Patients with Nonspecific Low Back Pain: Systematic Review. Phys. Ther. 2016, 96, 1816–1831. [Google Scholar] [CrossRef]
- Cheak-Zamora, N.C.; Wyrwich, K.W.; McBride, T.D. Reliability and validity of the SF-12v2 in the medical expenditure panel survey. Qual. Life Res. 2009, 18, 727–735. [Google Scholar] [CrossRef]
- Díaz-Arribas, M.J.; Fernández-Serrano, M.; Royuela, A.; Kovacs, F.M.; Gallego-Izquierdo, T.; Ramos-Sánchez, M.; Llorca-Palomera, R.; Pardo-Hervás, P.; Martín-Pariente, O.S. Minimal Clinically Important Difference in Quality of Life for Patients With Low Back Pain. Spine 2017, 42, 1908–1916. [Google Scholar] [CrossRef] [PubMed]
- Zhou, K.; Zhuang, G.; Zhang, H.; Liang, P.; Yin, J.; Kou, L.; Hao, M.; You, L. Psychometrics of the Short Form 36 Health Survey version 2 (SF-36v2) and the Quality of Life Scale for Drug Addicts (QOL-DAv2.0) in Chinese mainland patients with methadone maintenance treatment. PLoS ONE 2013, 8, e79828. [Google Scholar] [CrossRef]
- Brown, C.A.; Cheng, E.M.; Hays, R.D.; Vassar, S.D.; Vickrey, B.G. SF-36 includes less Parkinson Disease (PD)-targeted content but is more responsive to change than two PD-targeted health-related quality of life measures. Qual. Life Res. 2009, 18, 1219–1237. [Google Scholar] [CrossRef] [PubMed]
- Leonardi, M.; Raggi, A.; Pagani, M.; Carella, F.; Soliveri, P.; Albanese, A.; Romito, L. Relationships between disability, quality of life and prevalence of nonmotor symptoms in Parkinson’s disease. Parkinsonism Relat. Disord. 2012, 18, 35–39. [Google Scholar] [CrossRef] [PubMed]
- Dorman, P.J.; Dennis, M.; Sandercock, P. How do scores on the EuroQol relate to scores on the SF-36 after stroke? Stroke 1999, 30, 2146–2151. [Google Scholar] [CrossRef]
- Wilkinson, P.R.; Wolfe, C.D.; Warburton, F.G.; Rudd, A.G.; Howard, R.S.; Ross-Russell, R.W.; Beech, R. Longer term quality of life and outcome in stroke patients: Is the Barthel index alone an adequate measure of outcome? Qual. Health Care QHC 1997, 6, 125–130. [Google Scholar] [CrossRef]
- Emanuelson, I.; Andersson Holmkvist, E.; Björklund, R.; Stålhammar, D. Quality of life and post-concussion symptoms in adults after mild traumatic brain injury: A population-based study in western Sweden. Acta Neurol. Scand. 2003, 108, 332–338. [Google Scholar] [CrossRef]
- Elfving, B.; Andersson, T.; Grooten, W.J.A. Low levels of physical activity in back pain patients are associated with high levels of fear-avoidance beliefs and pain catastrophizing. Physiother. Res. Int. 2007, 12, 14–24. [Google Scholar] [PubMed]
- Marston, W.A.; Vasquez, M.A.; Lurie, F.; Wakefield, T.W.; Rabe, E.; Shortell, C.K.; Lohr, J.; Passman, M.A.; McLafferty, R. American Venous Forum Outcomes Working Group. Multicenter assessment of the repeatability and reproducibility of the revised Venous Clinical Severity Score (rVCSS). J. Vasc. Surg. Venous Lymphat. Disord. 2013, 1, 219–224. [Google Scholar] [CrossRef] [PubMed]
- Stratford, P.W.; Kennedy, D.M.; Woodhouse, L.J.; Spadoni, G.F. Measurement properties of the WOMAC LK 3.1 pain scale. Osteoarthr. Cartil. 2007, 15, 266–272. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Basaran, S.; Guzel, R.; Seydaoglu, G.; Guler-Uysal, F. Validity, reliability, and comparison of the WOMAC osteoarthritis index and Lequesne algofunctional index in Turkish patients with hip or knee osteoarthritis. Clin. Rheumatol. 2010, 29, 749–756. [Google Scholar] [CrossRef] [PubMed]
- Brooks, L.O.; Rolfe, M.I.; Cheras, P.A.; Myers, S.P. The comprehensive osteoarthritis test: A simple index for measurement of treatment effects in clinical trials. J. Rheumatol. 2004, 31, 1180–1186. [Google Scholar] [PubMed]
- Pua, Y.-H.; Cowan, S.M.; Wrigley, T.V.; Bennell, K.L. Discriminant validity of the Western Ontario and McMaster Universities Osteoarthritis index physical functioning subscale in community samples with hip osteoarthritis. Arch. Phys. Med. Rehabil. 2009, 90, 1772–1777. [Google Scholar] [CrossRef] [PubMed]
- Greco, N.J.; Anderson, A.F.; Mann, B.J.; Cole, B.J.; Farr, J.; Nissen, C.W.; Irrgang, J.J. Responsiveness of the International Knee Documentation Committee Subjective Knee Form in comparison to the Western Ontario and McMaster Universities Osteoarthritis Index, modified Cincinnati Knee Rating System, and Short Form 36 in patients with focal articular cartilage defects. Am. J. Sports Med. 2010, 38, 891–902. [Google Scholar]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cuesta-Vargas, A.; Martin-Martin, J.; Gonzalez-Sanchez, M.; Merchan-Baeza, J.A.; Perez-Cruzado, D. Identification of Tools for the Functional and Subjective Assessment of Patients in an Aquatic Environment: A Systematic Review. Int. J. Environ. Res. Public Health 2020, 17, 5690. https://doi.org/10.3390/ijerph17165690
Cuesta-Vargas A, Martin-Martin J, Gonzalez-Sanchez M, Merchan-Baeza JA, Perez-Cruzado D. Identification of Tools for the Functional and Subjective Assessment of Patients in an Aquatic Environment: A Systematic Review. International Journal of Environmental Research and Public Health. 2020; 17(16):5690. https://doi.org/10.3390/ijerph17165690
Chicago/Turabian StyleCuesta-Vargas, Antonio, Jaime Martin-Martin, Manuel Gonzalez-Sanchez, Jose Antonio Merchan-Baeza, and David Perez-Cruzado. 2020. "Identification of Tools for the Functional and Subjective Assessment of Patients in an Aquatic Environment: A Systematic Review" International Journal of Environmental Research and Public Health 17, no. 16: 5690. https://doi.org/10.3390/ijerph17165690
APA StyleCuesta-Vargas, A., Martin-Martin, J., Gonzalez-Sanchez, M., Merchan-Baeza, J. A., & Perez-Cruzado, D. (2020). Identification of Tools for the Functional and Subjective Assessment of Patients in an Aquatic Environment: A Systematic Review. International Journal of Environmental Research and Public Health, 17(16), 5690. https://doi.org/10.3390/ijerph17165690





