The Impact of Device Settings, Use Patterns, and Flavorings on Carbonyl Emissions from Electronic Cigarettes
Abstract
1. Introduction
2. Materials and Methods
2.1. E-Vapor Generation Conditions and Materials
2.2. Carbonyl Collection and Analysis
2.3. Statistical Analysis
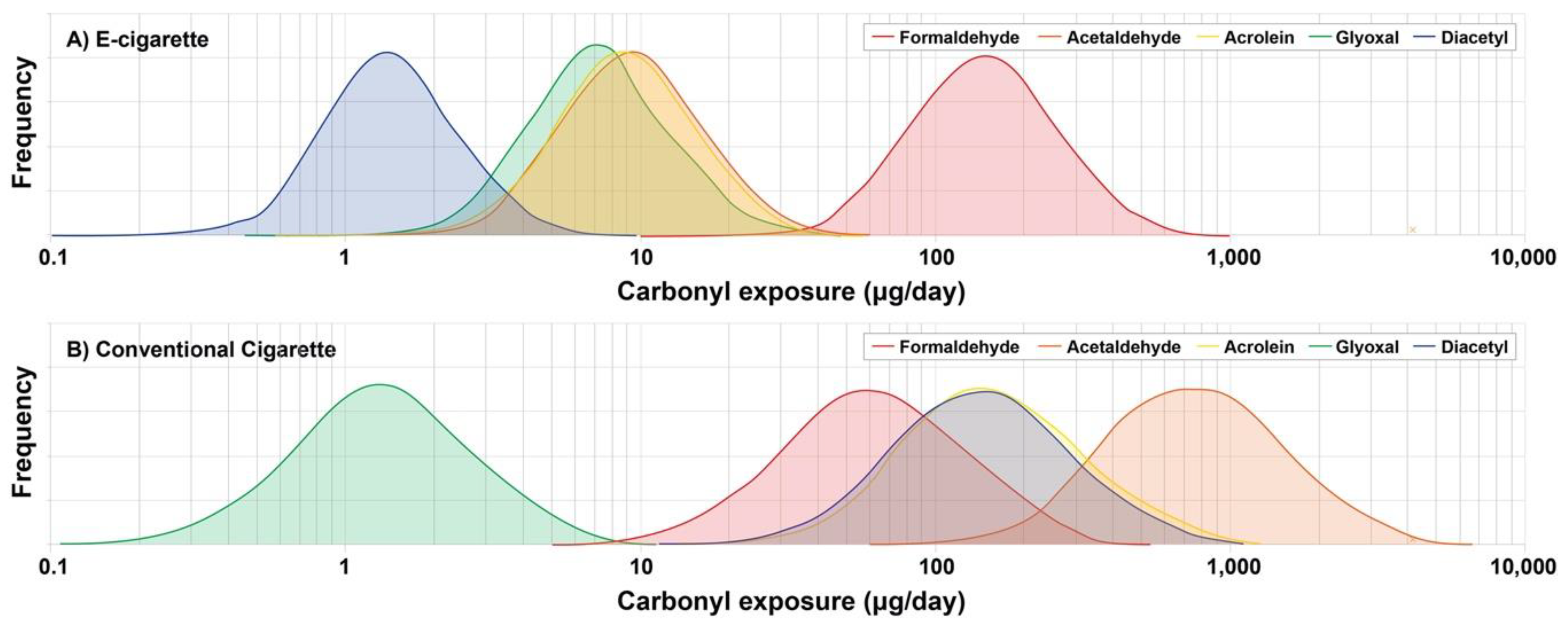
3. Results
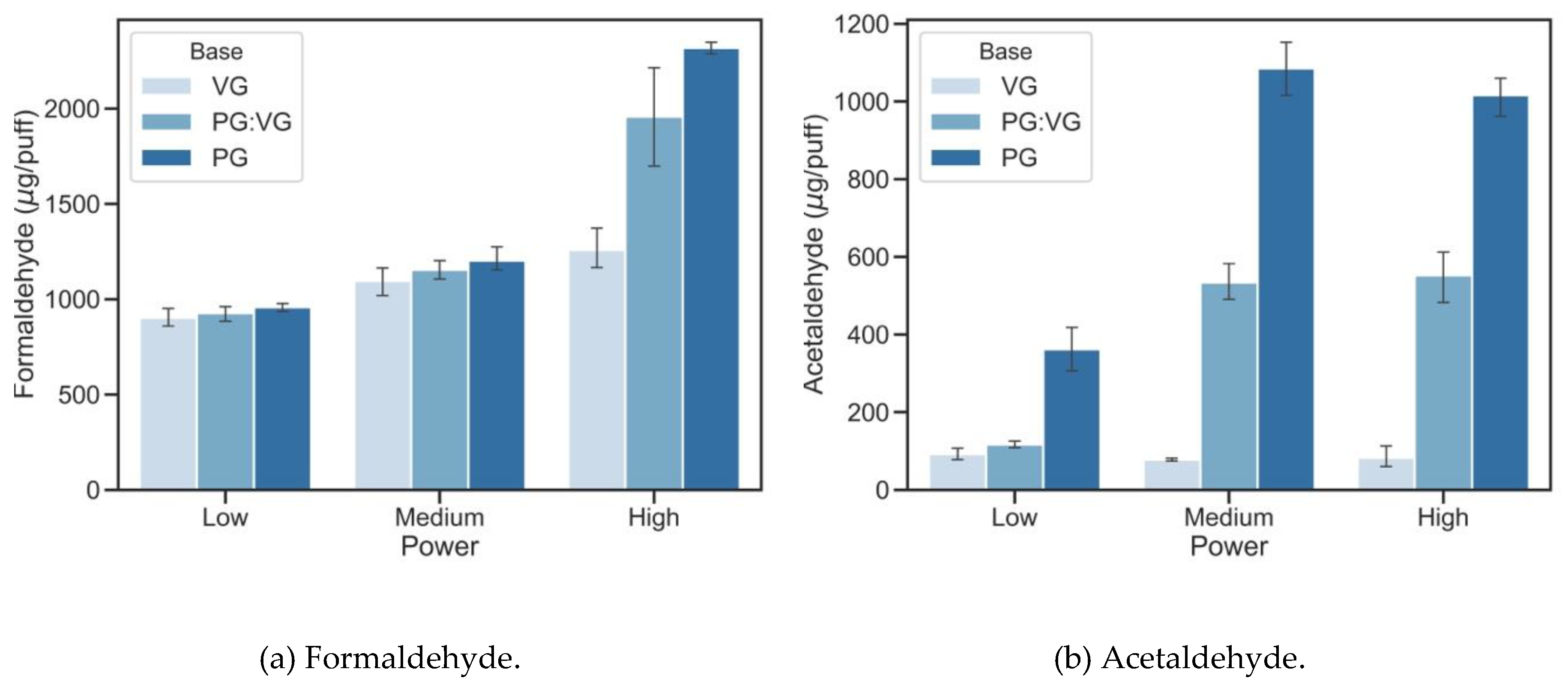
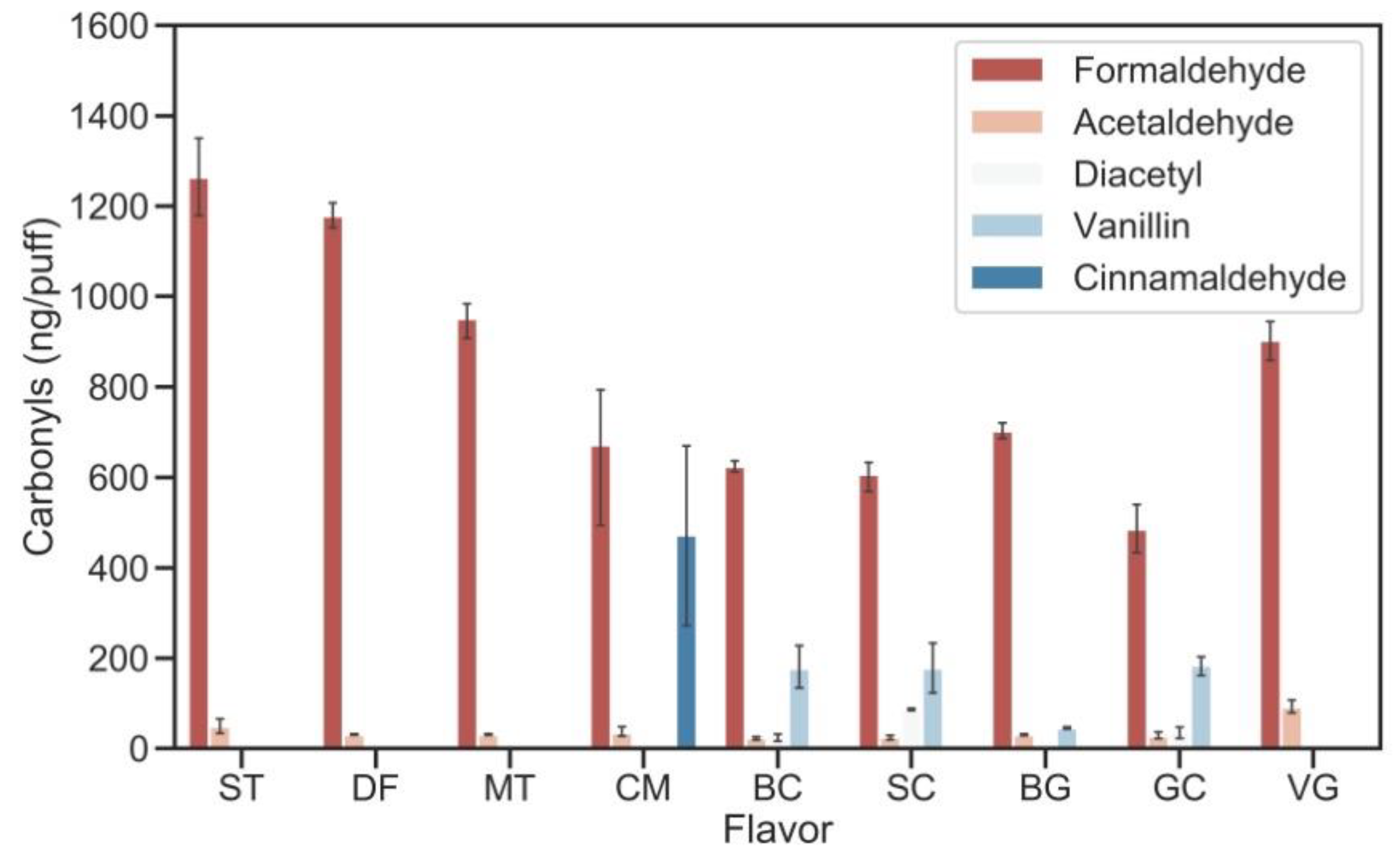
3.1. The Impact of E-Liquid Base Materials and Power Outputs
3.2. The Impact of E-Liquid Base Materials and Power Outputs
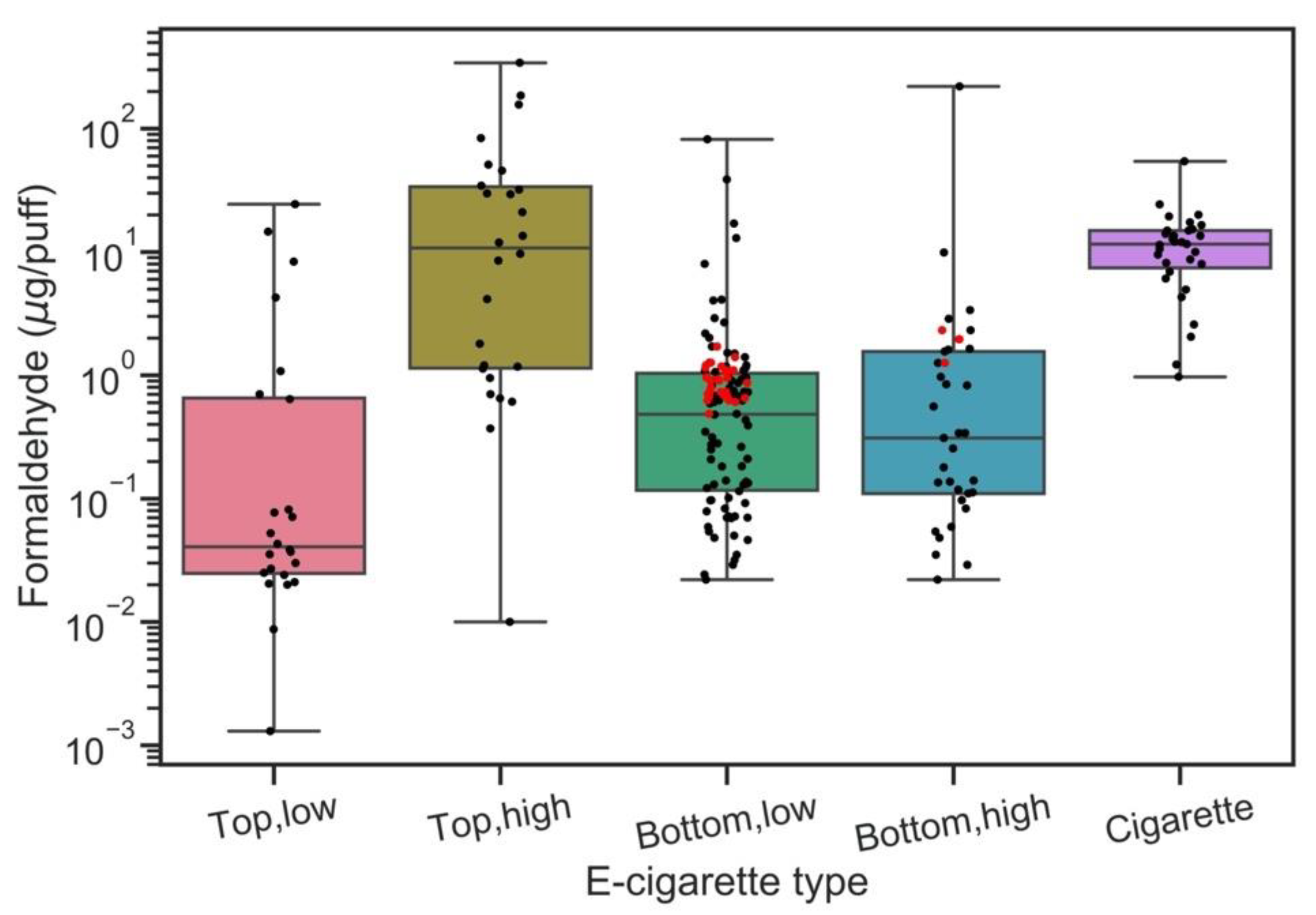
3.3. The Impact of Vaping Topography
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Flora, J.W.; Meruva, N.; Huang, C.B.; Wilkinson, C.T.; Ballentine, R.; Smith, D.C.; Werley, M.S.; McKinney, W.J. Characterization of potential impurities and degradation products in electronic cigarette formulations and aerosols. Regul. Toxicol. Pharmacol. 2016, 74, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Goniewicz, M.L.; Knysak, J.; Gawron, M.; Kosmider, L.; Sobczak, A.; Kurek, J.; Prokopowicz, A.; Jablonska-Czapla, M.; Rosik-Dulewska, C.; Havel, C. Levels of selected carcinogens and toxicants in vapour from electronic cigarettes. Tob. Control. 2014, 23, 133–139. [Google Scholar] [CrossRef] [PubMed]
- Schripp, T.; Markewitz, D.; Uhde, E.; Salthammer, T. Does e-cigarette consumption cause passive vaping? Indoor Air 2013, 23, 25–31. [Google Scholar] [CrossRef] [PubMed]
- Uchiyama, S.; Ohta, K.; Inaba, Y.; Kunugita, N. Determination of carbonyl compounds generated from the E-cigarette using coupled silica cartridges impregnated with hydroquinone and 2, 4-dinitrophenylhydrazine, followed by high-performance liquid chromatography. Anal. Sci. 2013, 29, 1219–1222. [Google Scholar] [CrossRef] [PubMed]
- US EPA. Integrated Risk Information System (IRIS). Available online: https://www.epa.gov/iris (accessed on 20 June 2020).
- Uter, W.; Schwanitz, H.; Lessmann, H.; Schnuch, A. Glyoxal is an important allergen for (medical care) cleaning staff. Int. J. Hyg. Environ. Health 2001, 204, 251–253. [Google Scholar] [CrossRef]
- Bahl, V.; Lin, S.; Xu, N.; Davis, B.; Wang, Y.-H.; Talbot, P. Comparison of electronic cigarette refill fluid cytotoxicity using embryonic and adult models. Reprod. Toxicol. 2012, 34, 529–537. [Google Scholar] [CrossRef]
- Behar, R.; Davis, B.; Wang, Y.; Bahl, V.; Lin, S.; Talbot, P. Identification of toxicants in cinnamon-flavored electronic cigarette refill fluids. Toxicol. Vitr. 2014, 28, 198–208. [Google Scholar] [CrossRef]
- Allen, J.G.; Flanigan, S.S.; LeBlanc, M.; Vallarino, J.; MacNaughton, P.; Stewart, J.H.; Christiani, D.C. Flavoring Chemicals in E-Cigarettes: Diacetyl, 2, 3-Pentanedione, and Acetoin in a Sample of 51 Products, Including Fruit-, Candy-, and Cocktail-Flavored E-Cigarettes. Environ. Health Perspect. 2016, 124, 733–739. [Google Scholar] [CrossRef]
- Rigler, M.W.; Longo, W.E. Emission of diacetyl (2, 3 butanedione) from natural butter, microwave popcorn butter flavor powder, paste, and liquid products. Int. J. Occup. Environ. Health 2010, 16, 291–302. [Google Scholar] [CrossRef]
- Geiss, O.; Bianchi, I.; Barahona, F.; Barrero-Moreno, J. Characterisation of mainstream and passive vapours emitted by selected electronic cigarettes. Int. J. Hyg. Environ. Health 2015, 218, 169–180. [Google Scholar] [CrossRef]
- Geiss, O.; Bianchi, I.; Barrero-Moreno, J. Correlation of volatile carbonyl yields emitted by e-cigarettes with the temperature of the heating coil and the perceived sensorial quality of the generated vapours. Int. J. Hyg. Environ. Health 2016, 219, 268–277. [Google Scholar] [CrossRef] [PubMed]
- Gillman, I.; Kistler, K.; Stewart, E.; Paolantonio, A. Effect of variable power levels on the yield of total aerosol mass and formation of aldehydes in e-cigarette aerosols. Regul. Toxicol. Pharmacol. 2016, 75, 58–65. [Google Scholar] [CrossRef] [PubMed]
- Kosmider, L.; Sobczak, A.; Fik, M.; Knysak, J.; Zaciera, M.; Kurek, J.; Goniewicz, M.L. Carbonyl compounds in electronic cigarette vapors—Effects of nicotine solvent and battery output voltage. Nicotine Tob. Res. 2014, 16, 1319–1326. [Google Scholar] [CrossRef] [PubMed]
- Uchiyama, S.; Senoo, Y.; Hayashida, H.; Inaba, Y.; Nakagome, H.; Kunugita, N. Determination of Chemical compounds generated from Second-generation E-cigarettes using a Sorbent Cartridge followed by a Two-step Elution Method. Anal. Sci. 2016, 32, 549–555. [Google Scholar] [CrossRef]
- Wang, P.; Chen, W.; Liao, J.; Matsuo, T.; Ito, K.; Fowles, J.; Shusterman, D.; Mendell, M.; Kumagai, K. A Device-Independent Evaluation of Carbonyl Emissions from Heated Electronic Cigarette Solvents. PLoS ONE 2017, 12, e0169811. [Google Scholar] [CrossRef]
- Ogunwale, M.A.; Li, M.; Ramakrishnam Raju, M.V.; Chen, Y.; Nantz, M.H.; Conklin, D.J.; Fu, X.-A. Aldehyde Detection in Electronic Cigarette Aerosols. ACS Omega 2017, 2, 1207–1214. [Google Scholar] [CrossRef]
- Baker, R.R.; Massey, E.D.; Smith, G. An overview of the effects of tobacco ingredients on smoke chemistry and toxicity. Food Chem. Toxicol. 2004, 42, 53–83. [Google Scholar] [CrossRef]
- Khlystov, A.; Samburova, V. Flavoring compounds dominate toxic aldehyde production during e-cigarette vaping. Environ. Sci. Technol. 2016, 50, 13080–13085. [Google Scholar] [CrossRef] [PubMed]
- Qu, Y.; Kim, K.-H.; Szulejko, J.E. The effect of flavor content in e-liquids on e-cigarette emissions of carbonyl compounds. Environ. Res. 2018, 166, 324–333. [Google Scholar] [CrossRef]
- Jensen, R.P.; Strongin, R.M.; Peyton, D.H. Solvent chemistry in the electronic cigarette reaction vessel. Sci. Rep. 2017, 7, 42549. [Google Scholar] [CrossRef]
- EL-Hellani, A.; Salman, R.; El-Hage, R.; Talih, S.; Malek, N.; Baalbaki, R.; Karaoghlanian, N.; Nakkash, R.; Shihadeh, A.; Saliba, N.A. Nicotine and carbonyl emissions from popular electronic cigarette products: Correlation to liquid composition and design characteristics. Nicotine Tob. Res. 2018, 20, 215–223. [Google Scholar] [CrossRef] [PubMed]
- Zhao, T.; Shu, S.; Guo, Q.; Zhu, Y. Effects of design parameters and puff topography on heating coil temperature and mainstream aerosols in electronic cigarettes. Atmos. Environ. 2016, 134, 61–69. [Google Scholar] [CrossRef]
- Flora, J.W.; Wilkinson, C.T.; Wilkinson, J.W.; Lipowicz, P.J.; Skapars, J.A.; Anderson, A.; Miller, J.H. Method for the Determination of Carbonyl Compounds in E-Cigarette Aerosols. J. Chromatogr. Sci. 2017, 55, 142–148. [Google Scholar] [CrossRef] [PubMed]
- Beauval, N.; Antherieu, S.; Soyez, M.; Gengler, N.; Grova, N.; Howsam, M.; Hardy, E.M.; Fischer, M.; Appenzeller, B.M.; Goossens, J.-F. Chemical evaluation of electronic cigarettes: Multicomponent analysis of liquid refills and their corresponding aerosols. J. Anal. Toxicol. 2017, 41, 670–678. [Google Scholar] [CrossRef] [PubMed]
- CORESTA. Method No. 81—Routine Analytical Machine for E-Cigarette Aerosol Generation and Collection—Definitions and Standard Conditions (CRM Developed into ISO 20768:2018); Cooperation Centre for Scientific Research Relative to Tobacco: Paris, France, 2015. [Google Scholar]
- Son, Y.; Weisel, C.; Wackowski, O.; Mainelis, G.; Schwander, S.; Delnevo, C.; Kipen, H.; Meng, Q. Evaluation of e-vapor nicotine and nicotyrine concentrations emitted under various e-liquid compositions, device settings and vaping topographies. Chem. Res. Toxicol. 2018, 31, 861–868. [Google Scholar] [CrossRef]
- Son, Y.; Mishin, V.; Laskin, J.D.; Mainelis, G.; Wackowski, O.A.; Delnevo, C.; Schwander, S.; Khlystov, A.; Samburova, V.; Meng, Q. Hydroxyl Radicals in E-Cigarette Vapor and E-Vapor Oxidative Potentials under Different Vaping Patterns. Chem. Res. Toxicol. 2019, 32, 1087–1095. [Google Scholar] [CrossRef]
- Son, Y.; Mainelis, G.; Delnevo, C.; Wackowski, O.A.; Schwander, S.; Meng, Q. Investigating E-Cigarette Particle Emissions and Human Airway Depositions under Various E-Cigarette-Use Conditions. Chem. Res. Toxicol. 2019, 33, 343–352. [Google Scholar] [CrossRef]
- Behar, R.Z.; Hua, M.; Talbot, P. Puffing Topography and Nicotine Intake of Electronic Cigarette Users. PLoS ONE 2015, 10, e0117222. [Google Scholar] [CrossRef]
- Dautzenberg, B.; Bricard, D. Real-Time Characterization of E-Cigarettes Use: The 1 Million Puffs Study. J. Addict. Res. Ther. 2015, 6, 1–5. [Google Scholar] [CrossRef]
- Meng, Q.; Schwander, S.; Son, Y.; Rivas, C.; Delnevo, C.; Graber, J.; Giovenco, D.; Bruen, U.; Mathew, R.; Robson, M. Has the mist been peered through? Revisiting the building blocks of human health risk assessment for electronic cigarette use. Hum. Ecol. Risk Assess. Int. J. 2016, 22, 558–579. [Google Scholar] [CrossRef]
- Norton, K.J.; June, K.M.; O’Connor, R.J. Initial puffing behaviors and subjective responses differ between an electronic nicotine delivery system and traditional cigarettes. Tob. Induc. Dis. 2014, 12, 17. [Google Scholar] [CrossRef]
- Robinson, R.; Hensel, E.; Morabito, P.; Roundtree, K. Electronic cigarette topography in the natural environment. PLoS ONE 2015, 10, e0129296. [Google Scholar] [CrossRef]
- Spindle, T.R.; Breland, A.B.; Karaoghlanian, N.V.; Shihadeh, A.L.; Eissenberg, T. Preliminary results of an examination of electronic cigarette user puff topography: The effect of a mouthpiece-based topography measurement device on plasma nicotine and subjective effects. Nicotine Tob. Res. 2014, 17, 142–149. [Google Scholar] [CrossRef]
- Hua, M.; Yip, H.; Talbot, P. Mining data on usage of electronic nicotine delivery systems (ENDS) from YouTube videos. Tob. Control. 2013, 22, 103–106. [Google Scholar] [CrossRef]
- Farsalinos, K.E.; Kistler, K.A.; Gillman, G.; Voudris, V. Evaluation of electronic cigarette liquids and aerosol for the presence of selected inhalation toxins. Nicotine Tob. Res. 2014, 17, 168–174. [Google Scholar] [CrossRef] [PubMed]
- Etter, J.F.; Zäther, E.; Svensson, S. Analysis of refill liquids for electronic cigarettes. Addiction 2013, 108, 1671–1679. [Google Scholar] [CrossRef] [PubMed]
- ELiquidRecipes. e-Liquid Recipes. Available online: http://e-liquid-recipes.com/ (accessed on 20 June 2020).
- US EPA. Compendium Method TO-11A: Determination of Formaldehyde in Ambient Air Using Adsorbent Cartridge Followed by High. Performance Liquid Chromatography (HPLC) [Active Sampling Methodology], 2nd ed.; (EPA/625/R-96/010b); Center for Environmental Research Information Office of Research and Development, U.S. Environmental Protection Agency: Cincinnati, OH, USA, 1999.
- NIOSH. Criteria for a Recommended Standard: Occupational Exposure to Diacetyl and 2,3-pentanedione DHHS (NIOSH) Publication No. 2016-111; Department of Health and Human Services, Centers for Disease Control and Prevention, National Institute for Occupational Safety and Health: Cincinnati, OH, USA, 2016.
- Lin, Y.-C. Catalytic valorization of glycerol to hydrogen and syngas. Int. J. Hydrog. Energy 2013, 38, 2678–2700. [Google Scholar] [CrossRef]
- Díaz, E.; Sad, M.E.; Iglesia, E. Homogeneous oxidation reactions of propanediols at low temperatures. ChemSusChem 2010, 3, 1063–1070. [Google Scholar] [CrossRef]
- Miner, C.S.; Dalton, N.N. Glycerol; Reinhold Pub. Corp.: New York, NY, USA, 1953. [Google Scholar]
- Son, Y.; Bhattarai, C.; Samburova, V.; Khlystov, A. Carbonyls and Carbon Monoxide Emissions from Electronic Cigarettes Affected by Device Type and Use Patterns. Int. J. Environ. Res. Public Health 2020, 17, 2767. [Google Scholar] [CrossRef]
- Uchiyama, S.; Noguchi, M.; Sato, A.; Ishitsuka, M.; Inaba, Y.; Kunugita, N. Determination of Thermal Decomposition Products Generated from E-cigarettes. Chem. Res. Toxicol. 2020, 33, 576–583. [Google Scholar] [CrossRef]
- Farsalinos, K.E.; Kistler, K.A.; Pennington, A.; Spyrou, A.; Kouretas, D.; Gillman, G. Aldehyde levels in e-cigarette aerosol: Findings from a replication study and from use of a new-generation device. Food Chem. Toxicol. 2018, 111, 64–70. [Google Scholar] [CrossRef]
- Farsalinos, K.E.; Voudris, V.; Poulas, K. E-cigarettes generate high levels of aldehydes only in ‘dry puff’conditions. Addiction 2015, 110, 1352–1356. [Google Scholar] [CrossRef] [PubMed]
- Sleiman, M.; Logue, J.M.; Montesinos, V.N.; Russell, M.L.; Litter, M.I.; Gundel, L.A.; Destaillats, H. Emissions from electronic cigarettes: Key parameters affecting the release of harmful chemicals. Environ. Sci. Technol. 2016, 50, 9644–9651. [Google Scholar] [CrossRef]
- Fujioka, K.; Shibamoto, T. Determination of toxic carbonyl compounds in cigarette smoke. Environ. Toxicol. 2006, 21, 47–54. [Google Scholar] [CrossRef] [PubMed]
- Moir, D.; Rickert, W.S.; Levasseur, G.; Larose, Y.; Maertens, R.; White, P.; Desjardins, S. A comparison of mainstream and sidestream marijuana and tobacco cigarette smoke produced under two machine smoking conditions. Chem. Res. Toxicol. 2007, 21, 494–502. [Google Scholar] [CrossRef] [PubMed]
- IARC. IARC Monographs on the Evaluation of Carcinogenic Risks to Humans Volume 83. In Tobacco Smoke and Involuntary Smoking; World Health Organization, International Agency for Research on Cancer: Lyon, France, 2004. [Google Scholar]
- Tierney, P.A.; Karpinski, C.D.; Brown, J.E.; Luo, W.; Pankow, J.F. Flavour chemicals in electronic cigarette fluids. Tob. Control. 2015, 25, e10–e15. [Google Scholar] [CrossRef] [PubMed]
- McAdam, K.; Vas, C.; Waters, G.; Margham, J.; Costigan, S.; Liu, C.; Proctor, C. Diacetyl in e-cigarettes: Sources, stability and aerosol transfer. In Proceedings of the SRNT 23rd Annual Meeting, Firenze Fiera Congress & Exhibition Center, Florence, Italy, 8–11 March 2017. [Google Scholar]
- Morgan, D.L.; Flake, G.P.; Kirby, P.J.; Palmer, S.M. Respiratory toxicity of diacetyl in C57BL/6 mice. Toxicol. Sci. 2008, 103, 169–180. [Google Scholar] [CrossRef]
- Lee, W.H.; Ong, S.-G.; Zhou, Y.; Tian, L.; Bae, H.R.; Baker, N.; Whitlatch, A.; Mohammadi, L.; Guo, H.; Nadeau, K.C. Modeling Cardiovascular Risks of E-Cigarettes with Human-Induced Pluripotent Stem Cell–Derived Endothelial Cells. J. Am. Coll. Cardiol. 2019, 73, 2722–2737. [Google Scholar] [CrossRef]
- National Academies of Sciences, Engineering, and Medicine. Public Health Consequences of E-Cigarettes; National Academies Press: Washington, DC, USA, 2018. [Google Scholar]
- Jamal, A.; Phillips, E.; Gentzke, A.S.; Homa, D.M.; Babb, S.D.; King, B.A.; Neff, L.J. Current cigarette smoking among adults—United States, 2016. Morb. Mortal. Wkly. Rep. 2018, 67, 53. [Google Scholar] [CrossRef]
- Shangari, N.; O’Brien, P.J. The cytotoxic mechanism of glyoxal involves oxidative stress. Biochem. Pharmacol. 2004, 68, 1433–1442. [Google Scholar] [CrossRef]
- US EPA. Toxicological Review of Acrylamide (CAS No. 7906-1) in Support of Summary Information on the Integrated Risk Information System (IRIS); U.S. Environmental Protection Agency: Washington, DC, USA, 2010.
- Yasuhara, A.; Tanaka, Y.; Hengel, M.; Shibamoto, T. Gas chromatographic investigation of acrylamide formation in browning model systems. J. Agric. Food Chem. 2003, 51, 3999–4003. [Google Scholar] [CrossRef] [PubMed]
- Schulte-Ladbeck, R.; Lindahl, R.; Levin, J.-O.; Karst, U. Characterization of chemical interferences in the determination of unsaturated aldehydes using aromatic hydrazine reagents and liquid chromatography. J. Environ. Monit. 2001, 3, 306–310. [Google Scholar] [CrossRef] [PubMed]
- FDA. Enforcement Priorities for Electronic Nicotine Delivery System (ENDS) and Other Deemed Products on the Market. Without Premarket Authorization (FDA-2019-D-0661); Center for Tobacco Products, Food and Drug Administration: Silver Spring, MD, USA, 2020.
| Experiments | Factors | Settings | Other Settings |
|---|---|---|---|
| E-liquid flavor | Flavor | 8 flavors 2 | 6.4 watt, 90 mL puff volume, 3.8 s puff duration, VG |
| Flavoring level (%) | 10% (v/v) 3 | ||
| E-liquid base | Base material | VG, PG:VG = 1:1 (v/v), PG | 6.4 watt, 90 mL puff volume, 3.8 s puff duration |
| Nicotine (mg/mL) | 12 | ||
| Device setting | Device power (watt) | 6.4, 14.7, 31.3 | 90 mL puff volume, 3.8 s puff duration, 12 mg/mL nicotine in VG |
| Vaping topography | Puff volume (mL) | 35, 90, 170 | 6.4 W, 12 mg/mL nicotine in VG |
| Puff duration (s) | 2, 3.8 |
| Carbonyl | Puff Volume and Duration † | |||||
|---|---|---|---|---|---|---|
| 35 mL | 90 mL | 170 mL | ||||
| 2 s | 3.8 s | 2 s | 3.8 s | 2 s | 3.8 s | |
| n = 3 | n = 3 | n = 3 | n = 5 | n = 3 | n = 3 | |
| Glyoxal | ND †† | ND †† | ND †† | ND †† | ND †† | ND †† |
| Formaldehyde | 683.0 ± 32.3 | 730.0 ± 53.8 | 790.0 ± 32.3 | 903.0 ± 56.2 | 747.0 ± 47.2 | 867.0 ± 32.7 |
| Acetaldehyde | 41.0 ± 9.35 | 39.9 ± 4.35 | <LOQ †††† | 91.7 ± 18.1 | <LOQ †††† | 47.1 ± 1.14 |
| Acetone | <LOQ †††† | <LOD ††† | <LOQ †††† | <LOD ††† | <LOQ †††† | <LOD ††† |
| Acrolein | 17.6 ± 1.32 | 32.0 ± 1.81 | 32.7 ± 1.27 | <LOQ | 28.9 ± 0.52 | 38.1 ± 1.52 |
| Propionaldehyde | ND †† | ND †† | ND †† | ND †† | ND †† | ND †† |
| Crotonaldehyde | 56.4 ± 3.82 | 34.9 ± 28.9 | 40.8 ± 0.56 | 29.8 ± 6.02 | 42.4 ± 0.67 | ND †† |
| n-Butylaldehyde | ND †† | ND †† | ND †† | ND †† | ND †† | 37.9 ± 1.80 |
| Benzaldehyde | 45.8 ± 1.41 | 31.4 ± 4.14 | 32.5 ± 0.46 | 23.1 ± 12.4 | 30.3 ± 2.45 | 29.5 ± 2.47 |
| Isovaleraldehyde | ND †† | 23.3 ± 5.56 | 33.4 ± 0.57 | ND †† | ND †† | 34.0 ± 1.55 |
| n-Valeraldehyde | 32.9 ± 12.8 | ND †† | 30.7 ± 2.18 | 81.1 ± 19.4 | 29.4 ± 0.92 | ND †† |
| o-Tolualdehyde | 134.1 ± 4.2 | 95.3 ± 17.9 | 196.0 ± 4.38 | ND †† | 186.0 ± 1.11 | 114. ± 4.19 |
| p-Tolualdehyde | 17.1 ± 0.26 | 21.6 ± 3.22 | 17.1 ± 0.18 | 18.1 ± 1.63 | 17.8 ± 0.15 | 20.4 ± 1.89 |
| n-Hexaldehyde | ND †† | 128.0 ± 8.06 | ND †† | 248.0 ± 65.1 | ND †† | 413.0 ± 4.65 |
| Dimethylbenzaldehyde | 33.1 ± 4.33 | ND †† | 28.4 ± 1.02 | ND †† | ND †† | ND †† |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Son, Y.; Weisel, C.; Wackowski, O.; Schwander, S.; Delnevo, C.; Meng, Q. The Impact of Device Settings, Use Patterns, and Flavorings on Carbonyl Emissions from Electronic Cigarettes. Int. J. Environ. Res. Public Health 2020, 17, 5650. https://doi.org/10.3390/ijerph17165650
Son Y, Weisel C, Wackowski O, Schwander S, Delnevo C, Meng Q. The Impact of Device Settings, Use Patterns, and Flavorings on Carbonyl Emissions from Electronic Cigarettes. International Journal of Environmental Research and Public Health. 2020; 17(16):5650. https://doi.org/10.3390/ijerph17165650
Chicago/Turabian StyleSon, Yeongkwon, Clifford Weisel, Olivia Wackowski, Stephan Schwander, Cristine Delnevo, and Qingyu Meng. 2020. "The Impact of Device Settings, Use Patterns, and Flavorings on Carbonyl Emissions from Electronic Cigarettes" International Journal of Environmental Research and Public Health 17, no. 16: 5650. https://doi.org/10.3390/ijerph17165650
APA StyleSon, Y., Weisel, C., Wackowski, O., Schwander, S., Delnevo, C., & Meng, Q. (2020). The Impact of Device Settings, Use Patterns, and Flavorings on Carbonyl Emissions from Electronic Cigarettes. International Journal of Environmental Research and Public Health, 17(16), 5650. https://doi.org/10.3390/ijerph17165650