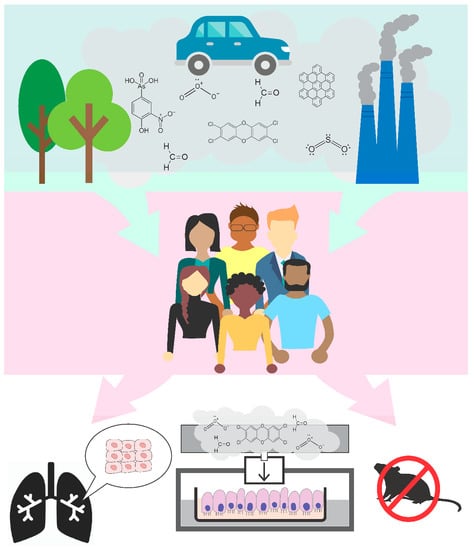
New Approach Methods to Evaluate Health Risks of Air Pollutants: Critical Design Considerations for In Vitro Exposure Testing
Abstract
1. Introduction
1.1. Introduction to Air Pollution Health Impacts
1.2. Methods to Evaluate Air Pollution Health Impacts
1.2.1. Epidemiological Studies in Air Pollution Research
1.2.2. Controlled Human Studies in Air Pollution Research
1.2.3. Animal Studies in Air Pollution Research
1.2.4. New Approach Methods in Air Pollution Research
1.3. Purpose of the Current Review
2. In Vitro Study Design Considerations
2.1. Cell Models
2.1.1. Monoculture Cell Lines
2.1.2. Cells from Human Donors
2.1.3. Lung-On-A-Chip Models
2.2. Cell Exposure Conditions
2.2.1. Exposures Using Submerged Conditions
2.2.2. Exposures Using Air–Liquid Interface Conditions
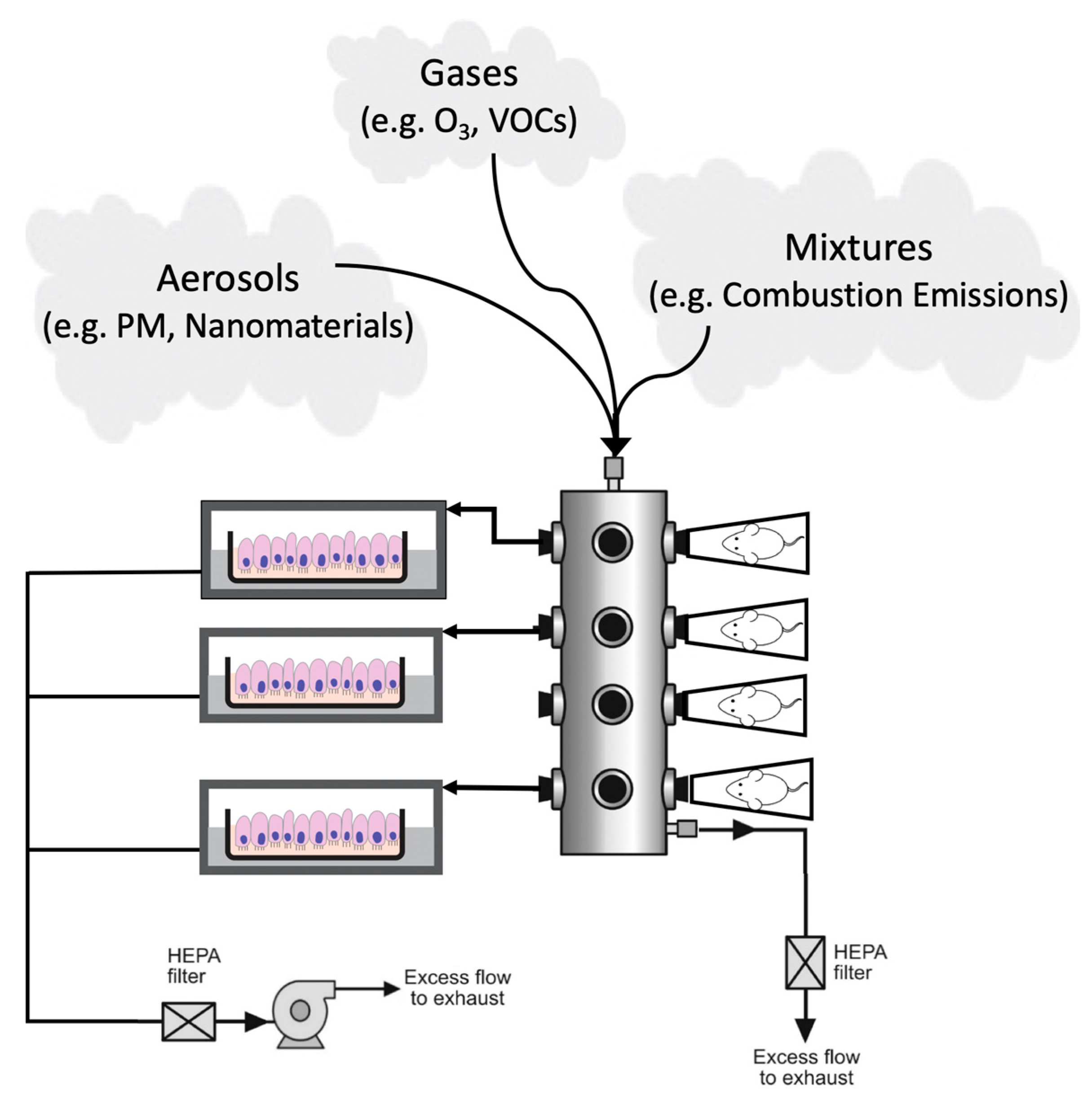
2.3. ALI Exposure Chambers
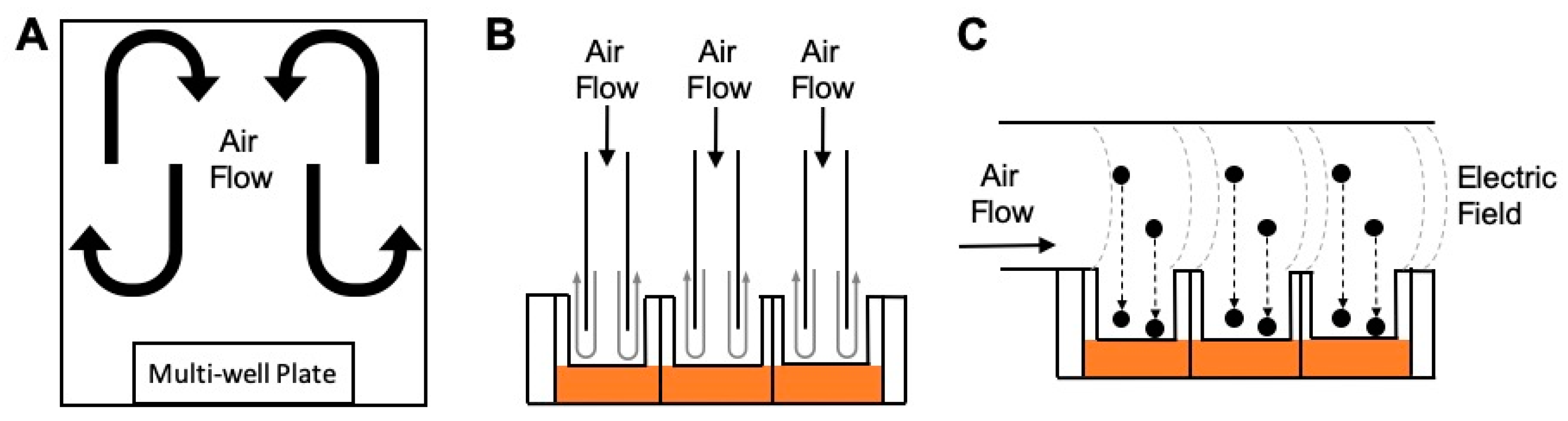
2.3.1. Undirected Flow
2.3.2. Perpendicular Flow
2.3.3. Horizontal Flow
2.3.4. Particle Deposition Forces
2.3.5. Exposure Parameters and Test Controls
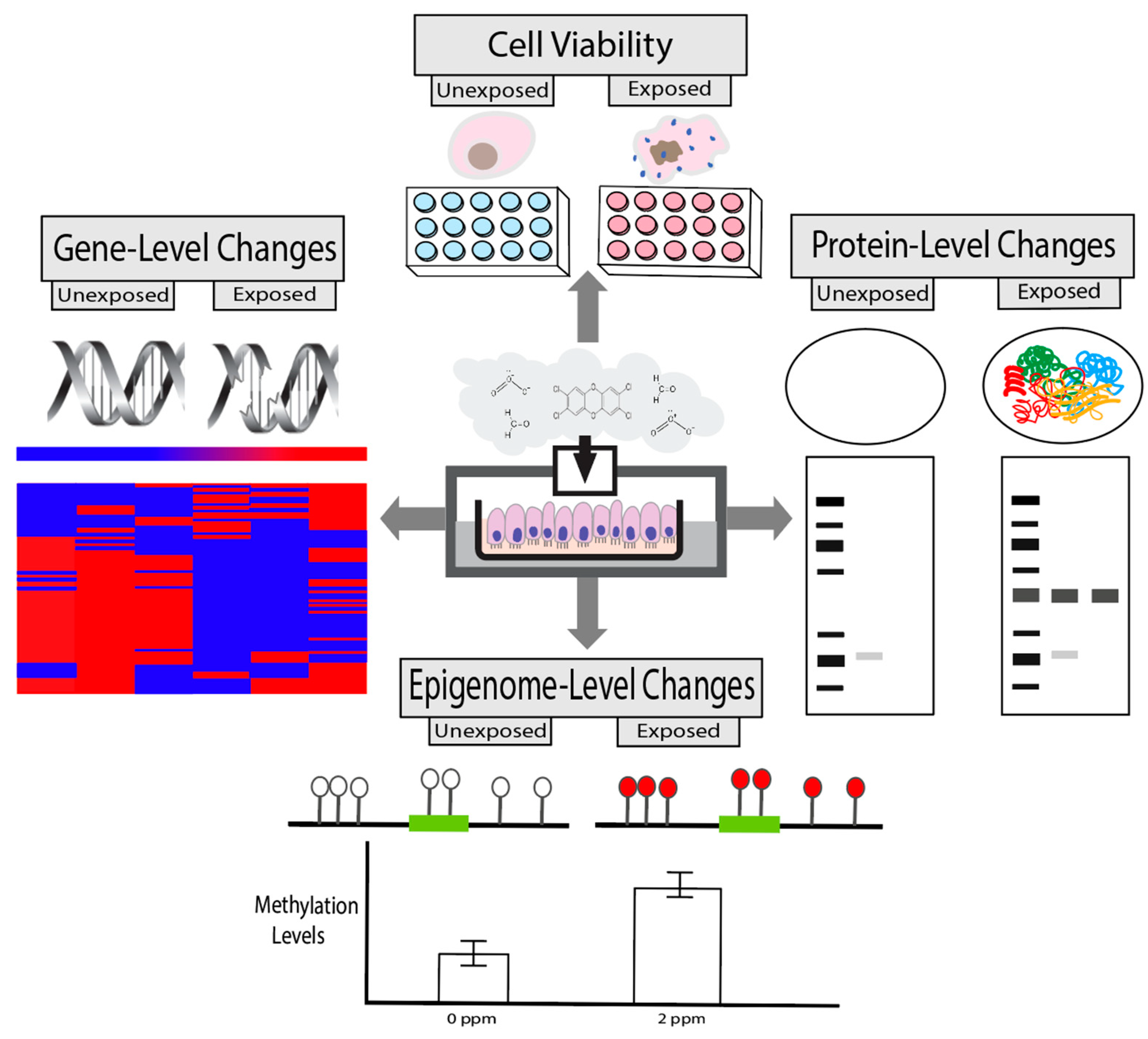
2.4. Toxicity Endpoints
2.4.1. Cell Viability
2.4.2. Gene-Level Changes
2.4.3. Protein-Level Changes
2.4.4. Epigenetic-Level Changes
3. Placing In Vitro Findings in the Context of Animal and Human Exposure Conditions
3.1. Experimental Methods to Compare In Vitro Findings to In Vivo Toxicology
3.2. Computational In Vitro-To-In Vivo Extrapolation Modeling
3.3. Current Limitations and Future Directions
4. Conclusions
Funding
Acknowledgments
Conflicts of Interest
References
- Mannucci, P.M.; Harari, S.; Martinelli, I.; Franchini, M. Effects on health of air pollution: A narrative review. Intern. Emerg. Med. 2015, 10, 657–662. [Google Scholar] [CrossRef] [PubMed]
- Koening, J.Q. Effect of Ozone on Respiratory Responses in Subjects with Asthma. Environ. Health Perspect. 1995, 103, 103–105. [Google Scholar] [CrossRef]
- Valavanidis, A.; Fiotakis, K.; Vlachogianni, T. Airborne particulate matter and human health: Toxicological assessment and importance of size and composition of particles for oxidative damage and carcinogenic mechanisms. J. Environ. Sci. Health C Environ. Carcinog. Ecotoxicol. Rev. 2008, 26, 339–362. [Google Scholar] [CrossRef] [PubMed]
- Doyle, M.; Sexton, K.G.; Jeffries, H.E.; Bridge, K.; Jaspers, I. Effects of 1-3-butadiene, isoprene, and the photochemical degradation products on human lung cells. Environ. Health Perspect. 2004, 112, 1488–1495. [Google Scholar] [CrossRef] [PubMed]
- Sexton, K.G.; Jeffries, H.E.; Jang, M.; Kamens, R.M.; Doyle, M.; Voicu, I.; Jaspers, I. Photochemical products in urban mixtures enhance inflammatory responses in lung cells. Inhal. Toxicol. 2004, 16 (Suppl. 1), 107–114. [Google Scholar] [CrossRef]
- Doyle, M.; Sexton, K.G.; Jeffries, H.; Jaspers, I. Atmospheric photochemical transformations enhance 1,3-butadiene-induced inflammatory responses in human epithelial cells: The role of ozone and other photochemical degradation products. Chem. Biol. Interact. 2007, 166, 163–169. [Google Scholar] [CrossRef]
- Rager, J.E.; Lichtveld, K.; Ebersviller, S.; Smeester, L.; Jaspers, I.; Sexton, K.G.; Fry, R.C. A toxicogenomic comparison of primary and photochemically altered air pollutant mixtures. Environ. Health Perspect. 2011, 119, 1583–1589. [Google Scholar] [CrossRef]
- Brauer, M.; Freedman, G.; Frostad, J.; van Donkelaar, A.; Martin, R.V.; Dentener, F.; van Dingenen, R.; Estep, K.; Amini, H.; Apte, J.S.; et al. Ambient Air Pollution Exposure Estimation for the Global Burden of Disease 2013. Environ. Sci. Technol. 2016, 50, 79–88. [Google Scholar] [CrossRef]
- Makri, A.; Stilianakis, N.I. Vulnerability to air pollution health effects. Int. J. Hyg. Environ. Health 2008, 211, 326–336. [Google Scholar] [CrossRef]
- Koken, P.J.; Piver, W.T.; Ye, F.; Elixhauser, A.; Olsen, L.M.; Portier, C.J. Temperature, air pollution, and hospitalization for cardiovascular diseases among elderly people in Denver. Environ. Health Perspect. 2003, 111, 1312–1317. [Google Scholar] [CrossRef]
- Cutter, S.L.; Boruff, B.J.; Shirley, W.L. Social Vulnerability to Environmental Hazards. Soc. Sci. Q 2003, 84, 242–261. [Google Scholar] [CrossRef]
- Anderson, H.R. Air Polution and Mortality: A History. Atmos 2009, 43, 142–152. [Google Scholar] [CrossRef]
- Bell, M.L.; Davis, D.L. Reassessment of the lethal London fog of 1952: Novel indicators of acute and chronic consequences of acute exposure to air pollution. Environ. Health Perspect. 2001, 109 (Suppl. 3), 389–394. [Google Scholar] [CrossRef] [PubMed]
- Wilkins, E.T. Air pollution and the London fog of December, 1952. J. R. Sanit. Inst. 1954, 74, 1–21. [Google Scholar] [CrossRef]
- Jacobs, E.T.; Burgess, J.L.; Abbott, M.B. The Donora Smog Revisited: 70 Years After the Event That Inspired the Clean Air Act. Am. J. Public Health 2018, 108, S85–S88. [Google Scholar] [CrossRef]
- Shima, M. Health Effects of Air Pollution: A Historical Review and Present Status. Nihon Eiseigaku Zasshi 2017, 72, 159–165. [Google Scholar] [CrossRef]
- Fuller, C.H.; Feeser, K.R.; Sarnat, J.A.; O’Neill, M.S. Air pollution, cardiovascular endpoints and susceptibility by stress and material resources: A systematic review of the evidence. Environ. Health 2017, 16, 58. [Google Scholar] [CrossRef]
- Brokamp, C.; Brandt, E.B.; Ryan, P.H. Assessing exposure to outdoor air pollution for epidemiological studies: Model-based and personal sampling strategies. J. Allergy Clin. Immunol. 2019, 143, 2002–2006. [Google Scholar] [CrossRef]
- Sujaritpong, S.; Dear, K.; Cope, M.; Walsh, S.; Kjellstrom, T. Quantifying the health impacts of air pollution under a changing climate-a review of approaches and methodology. Int. J. Biometeorol. 2014, 58, 149–160. [Google Scholar] [CrossRef]
- Ghio, A.J.; Sobus, J.R.; Pleil, J.D.; Madden, M.C. Controlled human exposures to diesel exhaust. Swiss Med. Wkly. 2012, 142, w13597. [Google Scholar] [CrossRef]
- Hackney, J.D.; Linn, W.S.; Buckley, R.D.; Pedersen, E.E.; Karuza, S.K.; Law, D.C.; Fischer, A. Experimental studies on human health effects of air pollutants: I. Design considerations. Arch. Environ. Health 1975, 30, 373–378. [Google Scholar] [CrossRef] [PubMed]
- Hackney, J.D.; Linn, W.S.; Mohler, J.G.; Pedersen, E.E.; Breisacher, P.; Russo, A. Experimental studies on human health effects of air pollutants. II. Four-hour exposure to ozone alone and in combination with other pollutant gases. Arch. Environ. Health 1975, 30, 379–384. [Google Scholar] [CrossRef] [PubMed]
- Dorato, M.A. Overview of Inhalation Toxicology. Environ. Health Perspect. 1990, 85, 163–170. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Phalen, R.F.; Mannix, R.C.; Drew, R.T. Inhalation exposure methodology. Environ. Health Perspect. 1984, 56, 23–34. [Google Scholar] [CrossRef] [PubMed]
- Dearman, R.J.; Caddick, H.; Stone, S.; Basketter, D.A.; Kimber, I. Characterization of antibody responses induced in rodents by exposure to food proteins: Influence of route of exposure. Toxicology 2001, 167, 217–231. [Google Scholar] [CrossRef]
- Jones, C.P.; Boyd, K.L.; Wallace, J.M. Evaluation of Mice Undergoing Serial Oral Gavage While Awake or Anesthetized. J. Am. Assoc. Lab. Anim. Sci. 2016, 55, 805–810. [Google Scholar]
- Liu, Z.; Ng, J.; Yuwono, A.; Lu, Y.; Tan, Y.K. Which is best method for instillation of topical therapy to the upper urinary tract? An in vivo porcine study to evaluate three delivery methods. Int. Braz. J. Urol. 2017, 43, 1084–1091. [Google Scholar] [CrossRef]
- Nielsen, T.B.; Yan, J.; Luna, B.; Spellberg, B. Murine Oropharyngeal Aspiration Model of Ventilator-associated and Hospital-acquired Bacterial Pneumonia. J. Vis. Exp. 2018. [Google Scholar] [CrossRef]
- Smulders, S.; Luyts, K.; Brabants, G.; Landuyt, K.V.; Kirschhock, C.; Smolders, E.; Golanski, L.; Vanoirbeek, J.; Hoet, P.H. Toxicity of nanoparticles embedded in paints compared with pristine nanoparticles in mice. Toxicol. Sci. 2014, 141, 132–140. [Google Scholar] [CrossRef]
- Morgan, D.L.; Flake, G.P.; Kirby, P.J.; Palmer, S.M. Respiratory toxicity of diacetyl in C57BL/6 mice. Toxicol. Sci. 2008, 103, 169–180. [Google Scholar] [CrossRef]
- Pratt, T.S.; Pincus, S.H.; Hale, M.L.; Moreira, A.L.; Roy, C.J.; Tchou-Wong, K.M. Oropharyngeal aspiration of ricin as a lung challenge model for evaluation of the therapeutic index of antibodies against ricin A-chain for post-exposure treatment. Exp. Lung Res. 2007, 33, 459–481. [Google Scholar] [CrossRef] [PubMed]
- Backus-Hazzard, G.S.; Howden, R.; Kleeberger, S.R. Genetic susceptibility to ozone-induced lung inflammation in animal models of asthma. Curr. Opin. Allergy Clin. Immunol. 2004, 4, 349–353. [Google Scholar] [CrossRef] [PubMed]
- Pinkerton, K.E.; Joad, J.P. Influence of air pollution on respiratory health during perinatal development. Clin. Exp. Pharmacol. Physiol. 2006, 33, 269–272. [Google Scholar] [CrossRef] [PubMed]
- Bolton, J.L.; Smith, S.H.; Huff, N.C.; Gilmour, M.I.; Foster, W.M.; Auten, R.L.; Bilbo, S.D. Prenatal air pollution exposure induces neuroinflammation and predisposes offspring to weight gain in adulthood in a sex-specific manner. FASEB J. 2012, 26, 4743–4754. [Google Scholar] [CrossRef] [PubMed]
- Swenberg, J.A.; Moeller, B.C.; Lu, K.; Rager, J.E.; Fry, R.C.; Starr, T.B. Formaldehyde carcinogenicity research: 30 years and counting for mode of action, epidemiology, and cancer risk assessment. Toxicol. Pathol. 2013, 41, 181–189. [Google Scholar] [CrossRef] [PubMed]
- Hanna, L.M.; Lou, S.R.; Su, S.; Jarabek, A.M. Mass transport analysis: Inhalation rfc methods framework for interspecies dosimetric adjustment. Inhal. Toxicol. 2001, 13, 437–463. [Google Scholar] [CrossRef] [PubMed]
- Brown, J.S.; Wilson, W.E.; Grant, L.D. Dosimetric comparisons of particle deposition and retention in rats and humans. Inhal. Toxicol. 2005, 17, 355–385. [Google Scholar] [CrossRef]
- Rothen-Rutishauser, B.; Blank, F.; Muhlfeld, C.; Gehr, P. In vitro models of the human epithelial airway barrier to study the toxic potential of particulate matter. Expert Opin. Drug Metab. Toxicol. 2008, 4, 1075–1089. [Google Scholar] [CrossRef] [PubMed]
- Ghasemi, M.; Dehpour, A.R. Ethical considerations in animal studies. J. Med. Ethics Hist. Med. 2009, 2, 12. [Google Scholar]
- EU. European Commission Ban on Animal Testing. Available online: https://ec.europa.eu/growth/sectors/cosmetics/animal-testing_en (accessed on 3 January 2020).
- EPA. Efforts to Reduce Animal Testing at EPA. Available online: https://www.epa.gov/research/efforts-reduce-animal-testing-epa (accessed on 3 January 2020).
- Kavlock, R.J.; Bahadori, T.; Barton-Maclaren, T.S.; Gwinn, M.R.; Rasenberg, M.; Thomas, R.S. Accelerating the Pace of Chemical Risk Assessment. Chem. Res. Toxicol. 2018, 31, 287–290. [Google Scholar] [CrossRef]
- ECHA. New Approach Methodologies in Regulatory Science. Available online: https://echa.europa.eu/documents/10162/22816069/scientific_ws_proceedings_en.pdf (accessed on 1 November 2019).
- Wambaugh, J.F.; Bare, J.C.; Carignan, C.C.; Dionisio, K.L.; Dodson, R.E.; Jolliet, O.; Liu, X.; Meyer, D.E.; Newton, S.R.; Phillips, K.A.; et al. New approach methodologies for exposure science. Curr. Opin. Toxicol. 2019, 15, 76–92. [Google Scholar] [CrossRef]
- Costa, D.L. Alternative test methods in inhalation toxicology: Challenges and opportunities. Exp. Toxicol. Pathol. 2008, 60, 105–109. [Google Scholar] [CrossRef] [PubMed]
- Arts, J.H.; Muijser, H.; Jonker, D.; van de Sandt, J.J.; Bos, P.M.; Feron, V.J. Inhalation toxicity studies: OECD guidelines in relation to REACH and scientific developments. Exp. Toxicol. Pathol. 2008, 60, 125–133. [Google Scholar] [CrossRef] [PubMed]
- Clippinger, A.J.; Allen, D.; Behrsing, H.; BeruBe, K.A.; Bolger, M.B.; Casey, W.; DeLorme, M.; Gaca, M.; Gehen, S.C.; Glover, K.; et al. Pathway-based predictive approaches for non-animal assessment of acute inhalation toxicity. Toxicol Vitro 2018, 52, 131–145. [Google Scholar] [CrossRef]
- Clippinger, A.J.; Allen, D.; Jarabek, A.M.; Corvaro, M.; Gaca, M.; Gehen, S.; Hotchkiss, J.A.; Patlewicz, G.; Melbourne, J.; Hinderliter, P.; et al. Alternative approaches for acute inhalation toxicity testing to address global regulatory and non-regulatory data requirements: An international workshop report. Toxicol. In Vitro 2018, 48, 53–70. [Google Scholar] [CrossRef]
- Hiemstra, P.S.; Grootaers, G.; van der Does, A.M.; Krul, C.A.M.; Kooter, I.M. Human lung epithelial cell cultures for analysis of inhaled toxicants: Lessons learned and future directions. Toxicol. In Vitro 2018, 47, 137–146. [Google Scholar] [CrossRef]
- Blank, F.; Rothen-Rutishauser, B.M.; Schurch, S.; Gehr, P. An optimized in vitro model of the respiratory tract wall to study particle cell interactions. J. Aerosol. Med. 2006, 19, 392–405. [Google Scholar] [CrossRef]
- Rager, J.E.; Smeester, L.; Jaspers, I.; Sexton, K.G.; Fry, R.C. Epigenetic changes induced by air toxics: Formaldehyde exposure alters miRNA expression profiles in human lung cells. Environ. Health Perspect. 2011, 119, 494–500. [Google Scholar] [CrossRef]
- Jaspers, I.; Flescher, E.; Chen, L.C. Ozone-induced IL-8 expression and transcription factor binding in respiratory epithelial cells. Am. J. Physiol. (Lung Cell Mol. Physiol.) 1997, 272, L504–L511. [Google Scholar] [CrossRef]
- Zavala, J.; O’Brien, B.; Lichtveld, K.; Sexton, K.G.; Rusyn, I.; Jaspers, I.; Vizuete, W. Assessment of biological responses of EpiAirway 3-D cell constructs versus A549 cells for determining toxicity of ambient air pollution. Inhal. Toxicol. 2016, 28, 251–259. [Google Scholar] [CrossRef]
- Judson, R.S.; Houck, K.A.; Kavlock, R.J.; Knudsen, T.B.; Martin, M.T.; Mortensen, H.M.; Reif, D.M.; Rotroff, D.M.; Shah, I.; Richard, A.M.; et al. In vitro screening of environmental chemicals for targeted testing prioritization: The ToxCast project. Environ. Health Perspect. 2010, 118, 485–492. [Google Scholar] [CrossRef] [PubMed]
- Ke, Y.; Reddel, R.R.; Gerwin, B.I.; Miyashita, M.; McMenamin, M.; Lechner, J.F.; Harris, C.C. Human bronchial epithelial cells with integrated SV40 virus T antigen genes retain the ability to undergo squamous differentiation. Differentiation 1988, 38, 60–66. [Google Scholar] [CrossRef]
- Li, Y.; Duan, J.; Yang, M.; Li, Y.; Jing, L.; Yu, Y.; Wang, J.; Sun, Z. Transcriptomic analyses of human bronchial epithelial cells BEAS-2B exposed to atmospheric fine particulate matter PM2.5. Toxicol. In Vitro 2017, 42, 171–181. [Google Scholar] [CrossRef] [PubMed]
- Øya, E.; Becher, R.; Ekeren, L.; Afanou, A.K.J.; Øvrevik, J.; Holme, J.A. Pro-Inflammatory Responses in Human Bronchial Epithelial Cells Induced by Spores and Hyphal Fragments of Common Damp Indoor Molds. Int. J. Environ. Res. Public Health 2019, 16, 1085. [Google Scholar] [CrossRef] [PubMed]
- Le Vee, M.; Bacle, A.; Jouan, E.; Lecureur, V.; Potin, S.; Fardel, O. Induction of multidrug resistance-associated protein 3 expression by diesel exhaust particle extract in human bronchial epithelial BEAS-2B cells. Toxicol. In Vitro 2019, 58, 60–68. [Google Scholar] [CrossRef] [PubMed]
- Schlesinger, R.B. Comparative deposition of inhaled aerosols in experimental animals and humans: A review. J. Toxicol. Environ. Health 1985, 15, 197–214. [Google Scholar] [CrossRef]
- Stanek, L.W.; Brown, J.S.; Stanek, J.; Gift, J.; Costa, D.L. Air pollution toxicology--a brief review of the role of the science in shaping the current understanding of air pollution health risks. Toxicol. Sci. 2011, 120 (Suppl. 1), S8–S27. [Google Scholar] [CrossRef]
- Upadhyay, S.; Palmberg, L. Air-Liquid Interface: Relevant In Vitro Models for Investigating Air Pollutant-Induced Pulmonary Toxicity. Toxicol. Sci. 2018, 164, 21–30. [Google Scholar] [CrossRef]
- Elisia, I.; Pae, H.B.; Lam, V.; Cederberg, R.; Hofs, E.; Krystal, G. Comparison of RAW264.7, human whole blood and PBMC assays to screen for immunomodulators. J. Immunol. Methods 2018, 452, 26–31. [Google Scholar] [CrossRef]
- Jalava, P.I.; Hirvonen, M.R.; Sillanpaa, M.; Pennanen, A.S.; Happo, M.S.; Hillamo, R.; Cassee, F.R.; Gerlofs-Nijland, M.; Borm, P.J.; Schins, R.P.; et al. Associations of urban air particulate composition with inflammatory and cytotoxic responses in RAW 246.7 cell line. Inhal. Toxicol. 2009, 21, 994–1006. [Google Scholar] [CrossRef]
- Jalava, P.I.; Aakko-Saksa, P.; Murtonen, T.; Happo, M.S.; Markkanen, A.; Yli-Pirila, P.; Hakulinen, P.; Hillamo, R.; Maki-Paakkanen, J.; Salonen, R.O.; et al. Toxicological properties of emission particles from heavy duty engines powered by conventional and bio-based diesel fuels and compressed natural gas. Part. Fibre Toxicol. 2012, 9, 37. [Google Scholar] [CrossRef] [PubMed]
- Müller, L.; Brighton, L.E.; Carson, J.L.; Fischer, W.A., 2nd; Jaspers, I. Culturing of human nasal epithelial cells at the air liquid interface. J. Vis. Exp. 2013. [Google Scholar] [CrossRef]
- Fulcher, M.L.; Randell, S.H. Human nasal and tracheo-bronchial respiratory epithelial cell culture. Methods Mol. Biol. 2013, 945, 109–121. [Google Scholar] [CrossRef] [PubMed]
- Chason, K.D.; Jaspers, I.; Parker, J.; Sellers, S.; Brighton, L.E.; Hunsucker, S.A.; Armistead, P.M.; Fischer, W.A., 2nd. Age-Associated Changes in the Respiratory Epithelial Response to Influenza Infection. J. Gerontol. A Biol. Sci. Med. Sci. 2018, 73, 1643–1650. [Google Scholar] [CrossRef] [PubMed]
- Speen, A.M.; Hoffman, J.R.; Kim, H.H.; Escobar, Y.N.; Nipp, G.E.; Rebuli, M.E.; Porter, N.A.; Jaspers, I. Small Molecule Antipsychotic Aripiprazole Potentiates Ozone-Induced Inflammation in Airway Epithelium. Chem. Res. Toxicol. 2019, 32, 1997–2005. [Google Scholar] [CrossRef] [PubMed]
- Mucilair. MucilAir™: A unique 3D Human Airway Epithelia reconstituted in vitro. Available online: http://www.epithelix.com/products/mucilair (accessed on 20 October 2019).
- MatTek. EpiAirway™. Available online: https://www.mattek.com/products/epiairway/ (accessed on 1 January 2020).
- Haycock, J.W. 3D cell culture: A review of current approaches and techniques. Methods Mol. Biol. 2011, 695, 1–15. [Google Scholar] [CrossRef]
- Chang, Y.; Siddens, L.K.; Heine, L.K.; Sampson, D.A.; Yu, Z.; Fischer, K.A.; Lohr, C.V.; Tilton, S.C. Comparative mechanisms of PAH toxicity by benzo[a]pyrene and dibenzo[def,p]chrysene in primary human bronchial epithelial cells cultured at air-liquid interface. Toxicol. Appl. Pharmacol. 2019, 379, 114644. [Google Scholar] [CrossRef]
- Rossner, P., Jr.; Cervena, T.; Vojtisek-Lom, M.; Vrbova, K.; Ambroz, A.; Novakova, Z.; Elzeinova, F.; Margaryan, H.; Beranek, V.; Pechout, M.; et al. The Biological Effects of Complete Gasoline Engine Emissions Exposure in a 3D Human Airway Model (MucilAir(TM)) and in Human Bronchial Epithelial Cells (BEAS-2B). Int. J. Mol. Sci. 2019, 20, 5710. [Google Scholar] [CrossRef]
- Anderson, S.E.; Khurshid, S.S.; Meade, B.J.; Lukomska, E.; Wells, J.R. Toxicological analysis of limonene reaction products using an in vitro exposure system. Toxicol. In Vitro 2013, 27, 721–730. [Google Scholar] [CrossRef]
- Balharry, D.; Sexton, K.; BeruBe, K.A. An in vitro approach to assess the toxicity of inhaled tobacco smoke components: Nicotine, cadmium, formaldehyde and urethane. Toxicology 2008, 244, 66–76. [Google Scholar] [CrossRef]
- Berube, K.; Prytherch, Z.; Job, C.; Hughes, T. Human primary bronchial lung cell constructs: The new respiratory models. Toxicology 2010, 278, 311–318. [Google Scholar] [CrossRef] [PubMed]
- Huh, D.; Leslie, D.C.; Matthews, B.D.; Fraser, J.P.; Jurek, S.; Hamilton, G.A.; Thorneloe, K.S.; McAlexander, M.A.; Ingber, D.E. A human disease model of drug toxicity-induced pulmonary edema in a lung-on-a-chip microdevice. Sci. Transl. Med. 2012, 4, 159ra147. [Google Scholar] [CrossRef] [PubMed]
- Wikswo, J.P.; Curtis, E.L.; Eagleton, Z.E.; Evans, B.C.; Kole, A.; Hofmeister, L.H.; Matloff, W.J. Scaling and systems biology for integrating multiple organs-on-a-chip. Lab. Hip 2013, 13, 3496–3511. [Google Scholar] [CrossRef] [PubMed]
- Benam, K.H.; Mazur, M.; Choe, Y.; Ferrante, T.C.; Novak, R.; Ingber, D.E. Human Lung Small Airway-on-a-Chip Protocol. Methods Mol. Biol. 2017, 1612, 345–365. [Google Scholar] [CrossRef] [PubMed]
- Benam, K.H.; Villenave, R.; Lucchesi, C.; Varone, A.; Hubeau, C.; Lee, H.H.; Alves, S.E.; Salmon, M.; Ferrante, T.C.; Weaver, J.C.; et al. Small airway-on-a-chip enables analysis of human lung inflammation and drug responses in vitro. Nat. Methods 2016, 13, 151–157. [Google Scholar] [CrossRef]
- Huh, D.D. A human breathing lung-on-a-chip. Ann. Am. Thoracic Soc. 2015, 12 (Suppl. 1), S42–S44. [Google Scholar] [CrossRef]
- Miller, P.G.; Chen, C.Y.; Wang, Y.I.; Gao, E.; Shuler, M.L. Multiorgan microfluidic platform with breathable lung chamber for inhalation or intravenous drug screening and development. Biotechnol. Bioeng. 2019. [Google Scholar] [CrossRef]
- Kimura, H.; Sakai, Y.; Fujii, T. Organ/body-on-a-chip based on microfluidic technology for drug discovery. Drug Metab. Pharmacokinet. 2018, 33, 43–48. [Google Scholar] [CrossRef]
- Wikswo, J.P. The relevance and potential roles of microphysiological systems in biology and medicine. Exp. Biol. Med. 2014, 239, 1061–1072. [Google Scholar] [CrossRef]
- Liu, Z.; Mackay, S.; Gordon, D.M.; Anderson, J.D.; Haithcock, D.W.; Garson, C.J.; Tearney, G.J.; Solomon, G.M.; Pant, K.; Prabhakarpandian, B.; et al. Co-cultured microfluidic model of the airway optimized for microscopy and micro-optical coherence tomography imaging. Biomed. Opt. express 2019, 10, 5414–5430. [Google Scholar] [CrossRef]
- Konar, D.; Devarasetty, M.; Yildiz, D.V.; Atala, A.; Murphy, S.V. Lung-On-A-Chip Technologies for Disease Modeling and Drug Development. Biomed. Eng. Comput. Biol. 2016, 7, 17–27. [Google Scholar] [CrossRef] [PubMed]
- Ghaemmaghami, A.M.; Hancock, M.J.; Harrington, H.; Kaji, H.; Khademhosseini, A. Biomimetic tissues on a chip for drug discovery. Drug Discov. Today 2012, 17, 173–181. [Google Scholar] [CrossRef] [PubMed]
- Dvorak, A.; Tilley, A.E.; Shaykhiev, R.; Wang, R.; Crystal, R.G. Do airway epithelium air-liquid cultures represent the in vivo airway epithelium transcriptome? Am. J. Respir. Cell Mol. Biol. 2011, 44, 465–473. [Google Scholar] [CrossRef] [PubMed]
- Paur, H.R.; Cassee, F.R.; Teeguarden, J.; Fissan, H.; Diabate, S.; Aufderheide, M.; Kreyling, W.G.; Hänninen, O.; Kasper, G.; Riediker, M.; et al. In-vitro cell exposure studies for the assessment of nanoparticle toxicity in the lung-a dialogue between aerosol science and biology. J. Aerosol. Sci. 2011, 42, 668–692. [Google Scholar] [CrossRef]
- de Bruijne, K.; Ebersviller, S.; Sexton, K.G.; Lake, S.; Leith, D.; Goodman, R.; Jetters, J.; Walters, G.W.; Doyle-Eisele, M.; Woodside, R.; et al. Design and testing of Electrostatic Aerosol in Vitro Exposure System (EAVES): An alternative exposure system for particles. Inhal. Toxicol. 2009, 21, 91–101. [Google Scholar] [CrossRef]
- Volckens, J.; Dailey, L.; Walters, G.; Devlin, R.B. Direct particle-to-cell deposition of coarse ambient particulate matter increases the production of inflammatory mediators from cultured human airway epithelial cells. Environ. Sci. Technol. 2009, 43, 4595–4599. [Google Scholar] [CrossRef]
- Lichtveld, K.M.; Ebersviller, S.M.; Sexton, K.G.; Vizuete, W.; Jaspers, I.; Jeffries, H.E. In vitro exposures in diesel exhaust atmospheres: Resuspension of PM from filters versus direct deposition of PM from air. Environ. Sci. Technol. 2012, 46, 9062–9070. [Google Scholar] [CrossRef]
- Secondo, L.E.; Liu, N.J.; Lewinski, N.A. Methodological considerations when conducting in vitro, air-liquid interface exposures to engineered nanoparticle aerosols. Crit. Rev. Toxicol. 2017, 47, 225–262. [Google Scholar] [CrossRef]
- Galezowska, G.; Chraniuk, M.; Wolska, L. In vitro assays as a tool for determination of VOCs toxic effect on respiratory system: A critical review. TrAC Trends Anal. Chem. 2016, 77, 14–22. [Google Scholar] [CrossRef]
- Zavala, J.; Ledbetter, A.D.; Morgan, D.S.; Dailey, L.A.; Puckett, E.; McCullough, S.D.; Higuchi, M. A new cell culture exposure system for studying the toxicity of volatile chemicals at the air-liquid interface. Inhal. Toxicol. 2018, 30, 169–177. [Google Scholar] [CrossRef]
- Clapp, P.W.; Lavrich, K.S.; van Heusden, C.A.; Lazarowski, E.R.; Carson, J.L.; Jaspers, I. Cinnamaldehyde in flavored e-cigarette liquids temporarily suppresses bronchial epithelial cell ciliary motility by dysregulation of mitochondrial function. Am. J. Physiol. Lung Cell Mol. Physiol. 2019, 316, L470–L486. [Google Scholar] [CrossRef] [PubMed]
- Carson, J.L.; Zhou, L.; Brighton, L.; Mills, K.H.; Zhou, H.; Jaspers, I.; Hazucha, M. Temporal structure/function variation in cultured differentiated human nasal epithelium associated with acute single exposure to tobacco smoke or E-cigarette vapor. Inhal. Toxicol. 2017, 29, 137–144. [Google Scholar] [CrossRef]
- Lenz, A.G.; Karg, E.; Lentner, B.; Dittrich, V.; Brandenberger, C.; Rothen-Rutishauser, B.; Schulz, H.; Ferron, G.A.; Schmid, O. A dose-controlled system for air-liquid interface cell exposure and application to zinc oxide nanoparticles. Part. Fibre Toxicol. 2009, 6, 32. [Google Scholar] [CrossRef] [PubMed]
- Zavala, J.; Greenan, R.; Krantz, Q.T.; DeMarini, D.M.; Higuchi, M.; Gilmour, M.I.; White, P.A. Regulating temperature and relative humidity in air-liquid interface in vitro systems eliminates cytotoxicity resulting from control air exposures. Toxicol. Res. (Camb.) 2017, 6, 448–459. [Google Scholar] [CrossRef] [PubMed]
- Jeannet, N.; Fierz, M.; Kalberer, M.; Burtscher, H.; Geiser, M. Nano aerosol chamber for in-vitro toxicity (NACIVT) studies. Nanotoxicology 2015, 9, 34–42. [Google Scholar] [CrossRef]
- Ritter, D.; Knebel, J. Investigations of the Biological Effects of Airborne and Inhalable Substances by Cell-Based In Vitro Methods: Fundamental Improvements to the ALI Concept. Adv. Toxicol. 2014, 185201, 11. [Google Scholar]
- Secondo, L.E.; Wygal, N.J.; Lewinski, N.A. A New Portable In Vitro Exposure Cassette for Aerosol Sampling. J. Vis. Exp. 2019. [Google Scholar] [CrossRef]
- Tilly, T.B.; Nelson, M.T.; Chakravarthy, K.B.; Shira, E.A.; Debrose, M.C.; Grabinski, C.M.; Salisbury, R.L.; Mattie, D.R.; Hussain, S.M. In Vitro Aerosol Exposure to Nanomaterials: From Laboratory to Environmental Field Toxicity Testing. Chem. Res. Toxicol. 2020. [Google Scholar] [CrossRef]
- Zavala, J.; Lichtveld, K.; Ebersviller, S.; Carson, J.L.; Walters, G.W.; Jaspers, I.; Jeffries, H.E.; Sexton, K.G.; Vizuete, W. The Gillings Sampler--an electrostatic air sampler as an alternative method for aerosol in vitro exposure studies. Chem. Biol. Interact. 2014, 220, 158–168. [Google Scholar] [CrossRef]
- MedTec. MedTec Biolab. Available online: http://www.medtecbiolab.com/ (accessed on 15 December 2019).
- Hinds, W.C. Aerosol. Technology: Property, Behavior, and Measurement of Airborne Particles, 2nd ed.; Wiley: Hoboken, NJ, USA, 1999; ISBN 978-0-471-19410-1. [Google Scholar]
- Brodbeck, C.; Ritter, D.; Knebel, J. Use of Computational Fluid Dynamics for optimization of cell-based in vitro methods in inhalation research. Available online: https://www.prit-systems.de/content/dam/scai/prit-systems/en/documents/Poster_PRIT_CFD_Eurotox_Sevilla_2016_DINA4.pdf (accessed on 15 December 2019).
- Aufderheide, M.; Scheffler, S.; Mohle, N.; Halter, B.; Hochrainer, D. Analytical in vitro approach for studying cyto- and genotoxic effects of particulate airborne material. Anal. Bioanal. Chem. 2011, 401, 3213–3220. [Google Scholar] [CrossRef]
- EPA. Particulate Matter (PM) Pollution. Available online: https://www.epa.gov/pm-pollution (accessed on 1 January 2020).
- Rager, J.E.; Fry, R.C. Systems biology and environmental exosures. In Network Biology; Zhang, W.J., Ed.; Nova Science Publishers: Hauppauge, NY, USA, 2013; pp. 81–132. ISBN 978-1-62618-942-3. [Google Scholar]
- Rager, J.E.; Carberry, C.; Fry, R.C. Use of Genome Editing Tools in Environmental Health Research. Curr. Opin. Toxicol. 2019, 18, 13–17. [Google Scholar] [CrossRef]
- Chappell, G.; Rager, J.E. Epigenetics in chemical-induced genotoxic carcinogenesis. Curr. Opin. Toxicol. 2017, 6, 10–17. [Google Scholar] [CrossRef]
- Fry, R.C.; Bangma, J.; Szilagyi, J.; Rager, J.E. Developing novel in vitro methods for the risk assessment of developmental and placental toxicants in the environment. Toxicol. Appl. Pharmacol. 2019, 378, 114635. [Google Scholar] [CrossRef] [PubMed]
- Judson, R.; Houck, K.; Martin, M.; Richard, A.M.; Knudsen, T.B.; Shah, I.; Little, S.; Wambaugh, J.; Setzer, R.W.; Kothiya, P.; et al. Analysis of the Effects of Cell Stress and Cytotoxicity on In Vitro Assay Activity Across a Diverse Chemical and Assay Space. Toxicol. Sci. 2016, 153, 409. [Google Scholar] [CrossRef]
- Cakmak, G.; Erturk Ari, P.; Emerce, E.; Ari, A.; Odabasi, M.; Schins, R.; Burgaz, S.; Gaga, E.O. Investigation of spatial and temporal variation of particulate matter in vitro genotoxicity and cytotoxicity in relation to the elemental composition. Mutat. Res. 2019, 842, 22–34. [Google Scholar] [CrossRef] [PubMed]
- Promega. CellTiter-Glo® Luminescent. Cell Viability Assay. Available online: file:///Users/anastasiafreedman/Downloads/CellTiterGlo%20Luminescent%20Cell%20Viability%20Assay%20TB288.pdf (accessed on 1 December 2019).
- Sigma-Aldrich. WST-1 Assay for Cell Proliferation and Viability. Available online: https://www.sigmaaldrich.com/technical-documents/protocols/biology/roche/cell-proliferation-reagent-wst-1.html (accessed on 1 December 2019).
- Scientific, T. Vybrant® MTT Cell Proliferation Assay Kit. Available online: https://www.thermofisher.com/us/en/home/references/protocols/cell-culture/mtt-assay-protocol/vybrant-mtt-cell-proliferation-assay-kit.html#prot3 (accessed on 15 October 2019).
- Sigma, M. Adenylate Kinase Activity Assay Kit (Colorimetric/Fluorometric). Available online: https://www.sigmaaldrich.com/catalog/product/sigma/mak235?lang=en®ion=US (accessed on 3 January 2020).
- Scientific, T. alamarBlue HS and alamarBlue Assays for Cell Viability. Available online: https://www.thermofisher.com/us/en/home/life-science/cell-analysis/fluorescence-microplate-assays/microplate-assays-cell-viability/alamarblue-assay-cell-viability.html (accessed on 1 December 2019).
- Scientific, T.; Calcein, A.M. cell-permeant dye. Available online: https://www.thermofisher.com/order/catalog/product/C1430#/C1430 (accessed on 3 January 2020).
- Shen, C.; Liu, J.; Zhu, F.; Lei, R.; Cheng, H.; Zhang, C.; Sui, X.; Ding, L.; Yang, M.; Chen, H.; et al. The effects of cooking oil fumes-derived PM2.5 on blood vessel formation through ROS-mediated NLRP3 inflammasome pathway in human umbilical vein endothelial cells. Ecotoxicol. Environ. Saf. 2019, 174, 690–698. [Google Scholar] [CrossRef] [PubMed]
- Konczol, M.; Goldenberg, E.; Ebeling, S.; Schafer, B.; Garcia-Kaufer, M.; Gminski, R.; Grobety, B.; Rothen-Rutishauser, B.; Merfort, I.; Giere, R.; et al. Cellular uptake and toxic effects of fine and ultrafine metal-sulfate particles in human A549 lung epithelial cells. Chem. Res. Toxicol. 2012, 25, 2687–2703. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, R.; Inoue, K.; Fujitani, Y.; Kiyono, M.; Hirano, S.; Takano, H. Effects of nanoparticle-rich diesel exhaust particles on IL-17 production in vitro. J. Immunotoxicol. 2012, 9, 72–76. [Google Scholar] [CrossRef]
- Velali, E.; Papachristou, E.; Pantazaki, A.; Besis, A.; Samara, C.; Labrianidis, C.; Lialiaris, T. In vitro cellular toxicity induced by extractable organic fractions of particles exhausted from urban combustion sources—Role of PAHs. Environ. Pollut. 2018, 243, 1166–1176. [Google Scholar] [CrossRef]
- Holder, A.L.; Goth-Goldstein, R.; Lucas, D.; Koshland, C.P. Particle-induced artifacts in the MTT and LDH viability assays. Chem. Res. Toxicol. 2012, 25, 1885–1892. [Google Scholar] [CrossRef]
- Neutral Red Cell Cytotoxicity Assay Kit 5/19. 2019. Available online: https://www.biovision.com/documentation/datasheets/K447.pdf?__cf_chl_jschl_tk__=3b83f40353302dc89508a336c06afb12a6331012-1584933474-0-AekYlDH6asMYzauezBrF6MlnHFeshd_iuNRuAPAdDOXma59Tv121unWzAMyOinuQvG_n2IcsOK2W7k_HIHzfA51xmRHjggLfmd9V_Au2nJnp--2BRNahB9jIy7wFdbE_Lm4UOWlULcXCikd-kAUbx0I2nk6G5q8SiPUymFdbIcJJdjH0hAS3ZTh4_UjaB_wjgdNbYlbpirODa59FdUnxwO7V2mHFAsH5sMustsNpTDVcjvrwLcuLMRBjNPskTpjVzbYfqvVfDmz0JvcncOXhbDBEompyjudXJrrotnYIyPBA7qaedbBnZqrnKEbXBUpEYQ (accessed on 13 February 2020).
- Combes, R.; Scott, K.; Dillon, D.; Meredith, C.; McAdam, K.; Proctor, C. The effect of a novel tobacco process on the in vitro cytotoxicity and genotoxicity of cigarette smoke particulate matter. Toxicol. In Vitro 2012, 26, 1022–1029. [Google Scholar] [CrossRef] [PubMed]
- Cervena, T.; Rossnerova, A.; Sikorova, J.; Beranek, V.; Vojtisek-Lom, M.; Ciganek, M.; Topinka, J.; Rossner, P., Jr. DNA Damage Potential of Engine Emissions Measured In Vitro by Micronucleus Test in Human Bronchial Epithelial Cells. Basic Clin. Pharmacol. Toxicol. 2017, 121 (Suppl. 3), 102–108. [Google Scholar] [CrossRef] [PubMed]
- Smith, M.T.; Guyton, K.Z.; Gibbons, C.F.; Fritz, J.M.; Portier, C.J.; Rusyn, I.; DeMarini, D.M.; Caldwell, J.C.; Kavlock, R.J.; Lambert, P.F.; et al. Key Characteristics of Carcinogens as a Basis for Organizing Data on Mechanisms of Carcinogenesis. Environ. Health Perspect. 2016, 124, 713–721. [Google Scholar] [CrossRef]
- Hamanaka, R.B.; Mutlu, G.M. Particulate Matter Air Pollution: Effects on the Cardiovascular System. Front. Endocrinol. 2018, 9, 680. [Google Scholar] [CrossRef]
- GSEA. View Gene Families for HALLMARK_INFLAMMATORY_RESPONSE. 2019. Available online: https://www.gsea-msigdb.org/gsea/msigdb/cards/HALLMARK_INFLAMMATORY_RESPONSE (accessed on 13 February 2020).
- Aghasafari, P.; George, U.; Pidaparti, R. A review of inflammatory mechanism in airway diseases. Inflamm. Res. 2019, 68, 59–74. [Google Scholar] [CrossRef]
- Rager, J.E.; Moeller, B.C.; Doyle-Eisele, M.; Kracko, D.; Swenberg, J.A.; Fry, R.C. Formaldehyde and epigenetic alterations: microRNA changes in the nasal epithelium of nonhuman primates. Environ. Health Perspect. 2013, 121, 339–344. [Google Scholar] [CrossRef][Green Version]
- Wang, T.; Wang, L.; Moreno-Vinasco, L.; Lang, G.D.; Siegler, J.H.; Mathew, B.; Usatyuk, P.V.; Samet, J.M.; Geyh, A.S.; Breysse, P.N.; et al. Particulate matter air pollution disrupts endothelial cell barrier via calpain-mediated tight junction protein degradation. Part. Fibre Toxicol. 2012, 9, 35. [Google Scholar] [CrossRef]
- Sadaria, M.R.; Smith, P.D.; Fullerton, D.A.; Justison, G.A.; Lee, J.H.; Puskas, F.; Grover, F.L.; Cleveland, J.C., Jr.; Reece, T.B.; Weyant, M.J. Cytokine expression profile in human lungs undergoing normothermic ex-vivo lung perfusion. Ann. Thorac. Surg. 2011, 92, 478–484. [Google Scholar] [CrossRef]
- Li, S.; Wang, J.; Wei, B.K.; Dong, G.; Wang, X. Protective Effect of Taurine on Paraquat-Induced Lung Epithelial Cell Injury. Adv. Exp. Med. Biol. 2019, 1155, 739–746. [Google Scholar] [CrossRef]
- Agraval, H.; Yadav, U.C.S. MMP-2 and MMP-9 mediate cigarette smoke extract-induced epithelial-mesenchymal transition in airway epithelial cells via EGFR/Akt/GSK3beta/beta-catenin pathway: Amelioration by fisetin. Chem. Biol. Interact. 2019, 314, 108846. [Google Scholar] [CrossRef] [PubMed]
- Yilmaz, Y.; Williams, G.; Walles, M.; Manevski, N.; Krahenbuhl, S.; Camenisch, G. Comparison of Rat and Human Pulmonary Metabolism Using Precision-cut Lung Slices (PCLS). Drug Metab. Lett. 2019, 13, 53–63. [Google Scholar] [CrossRef] [PubMed]
- Haubrich, B.A.; Swinney, D.C. Enzyme Activity Assays for Protein Kinases: Strategies to Identify Active Substrates. Curr. Drug Discov. Technol. 2016, 13, 2–15. [Google Scholar] [CrossRef] [PubMed]
- Kocbach, A.; Totlandsdal, A.I.; Lag, M.; Refsnes, M.; Schwarze, P.E. Differential binding of cytokines to environmentally relevant particles: A possible source for misinterpretation of in vitro results? Toxicol. Lett. 2008, 176, 131–137. [Google Scholar] [CrossRef]
- Kroll, A.; Pillukat, M.H.; Hahn, D.; Schnekenburger, J. Interference of engineered nanoparticles with in vitro toxicity assays. Arch. Toxicol. 2012, 86, 1123–1136. [Google Scholar] [CrossRef]
- Seagrave, J.; Knall, C.; McDonald, J.D.; Mauderly, J.L. Diesel particulate material binds and concentrates a proinflammatory cytokine that causes neutrophil migration. Inhal. Toxicol. 2004, 16 (Suppl. 1), 93–98. [Google Scholar] [CrossRef]
- Inbar-Feigenberg, M.; Choufani, S.; Butcher, D.T.; Roifman, M.; Weksberg, R. Basic concepts of epigenetics. Fertil. Steril. 2013, 99, 607–615. [Google Scholar] [CrossRef]
- Dolinoy, D.C.; Weidman, J.R.; Jirtle, R.L. Epigenetic gene regulation: Linking early developmental environment to adult disease. Reprod. Toxicol. 2007, 23, 297–307. [Google Scholar] [CrossRef]
- Jaspers, I.; Horvath, K.M.; Zhang, W.; Brighton, L.E.; Carson, J.L.; Noah, T.L. Reduced expression of IRF7 in nasal epithelial cells from smokers after infection with influenza. Am. J. Respir. Cell Mol. Biol. 2010, 43, 368–375. [Google Scholar] [CrossRef]
- Rager, J.E.; Bauer, R.N.; Muller, L.L.; Smeester, L.; Carson, J.L.; Brighton, L.E.; Fry, R.C.; Jaspers, I. DNA methylation in nasal epithelial cells from smokers: Identification of ULBP3-related effects. Am. J. Physiol. Lung Cell Mol. Physiol. 2013, 305, L432–L438. [Google Scholar] [CrossRef]
- Owens, E.O.; Patel, M.M.; Kirrane, E.; Long, T.C.; Brown, J.; Cote, I.; Ross, M.A.; Dutton, S.J. Framework for assessing causality of air pollution-related health effects for reviews of the National Ambient Air Quality Standards. Regul. Toxicol. Pharmacol. 2017, 88, 332–337. [Google Scholar] [CrossRef] [PubMed]
- Driscoll, K.E.; Costa, D.L.; Hatch, G.; Henderson, R.; Oberdorster, G.; Salem, H.; Schlesinger, R.B. Intratracheal instillation as an exposure technique for the evaluation of respiratory tract toxicity: Uses and limitations. Toxicol. Sci. 2000, 55, 24–35. [Google Scholar] [CrossRef] [PubMed]
- Vinall, J.; Simpson, H.; Baily, J.; Roper, C. Comparison of Histopathological and Cell Viability Results from In Vitro Rat and Human Airway™ Models for Translational Inhalation Toxicity Testing, Association of Inhalation Toxicologist Conference. Available online: https://www.criver.com/sites/default/files/resource-files/SP-AIT-18-comparison-of-histopathological-and-cell-viability-results-from-in-vitro-rat-and-human-Airway.pdf (accessed on 1 December 2019).
- OECD. Guidance document on acute inhalation toxicity testing. Series on testing and assessment. Number 39. Available online: https://ntp.niehs.nih.gov/iccvam/suppdocs/feddocs/oecd/oecd-gd39.pdf (accessed on 1 December 2019).
- Bell, S.M.; Chang, X.; Wambaugh, J.F.; Allen, D.G.; Bartels, M.; Brouwer, K.L.R.; Casey, W.M.; Choksi, N.; Ferguson, S.S.; Fraczkiewicz, G.; et al. In vitro to in vivo extrapolation for high throughput prioritization and decision making. Toxicol. In Vitro 2018, 47, 213–227. [Google Scholar] [CrossRef] [PubMed]
- Sipes, N.S.; Wambaugh, J.F.; Pearce, R.; Auerbach, S.S.; Wetmore, B.A.; Hsieh, J.H.; Shapiro, A.J.; Svoboda, D.; DeVito, M.J.; Ferguson, S.S. An Intuitive Approach for Predicting Potential Human Health Risk with the Tox21 10k Library. Environ. Sci. Technol 2017, 51, 10786–10796. [Google Scholar] [CrossRef]
- Coecke, S.; Pelkonen, O.; Leite, S.B.; Bernauer, U.; Bessems, J.G.; Bois, F.Y.; Gundert-Remy, U.; Loizou, G.; Testai, E.; Zaldivar, J.M. Toxicokinetics as a key to the integrated toxicity risk assessment based primarily on non-animal approaches. Toxicol. In Vitro 2013, 27, 1570–1577. [Google Scholar] [CrossRef] [PubMed]
- Algharably, E.A.H.; Kreutz, R.; Gundert-Remy, U. Importance of in vitro conditions for modeling the in vivo dose in humans by in vitro-in vivo extrapolation (IVIVE). Arch. Toxicol. 2019, 93, 615–621. [Google Scholar] [CrossRef] [PubMed]
- Blaauboer, B.J. Biokinetic modeling and in vitro-in vivo extrapolations. J. Toxicol. Environ. Health B Crit. Rev. 2010, 13, 242–252. [Google Scholar] [CrossRef]
- Yoon, M.; Campbell, J.L.; Andersen, M.E.; Clewell, H.J. Quantitative in vitro to in vivo extrapolation of cell-based toxicity assay results. Crit. Rev. Toxicol. 2012, 42, 633–652. [Google Scholar] [CrossRef]
- NRC. Methods for Assessing Health Risks of Reclaimed Water; National Acaedmies Press: Washington, DC, USA, 1998. [Google Scholar]
- Wetmore, B.A.; Wambaugh, J.F.; Ferguson, S.S.; Li, L.; Clewell, H.J., 3rd; Judson, R.S.; Freeman, K.; Bao, W.; Sochaski, M.A.; Chu, T.M.; et al. Relative impact of incorporating pharmacokinetics on predicting in vivo hazard and mode of action from high-throughput in vitro toxicity assays. Toxicol. Sci. 2013, 132, 327–346. [Google Scholar] [CrossRef]
- Wambaugh, J.F.; Setzer, R.W.; Pitruzzello, A.M.; Liu, J.; Reif, D.M.; Kleinstreuer, N.C.; Wang, N.C.; Sipes, N.; Martin, M.; Das, K.; et al. Dosimetric anchoring of in vivo and in vitro studies for perfluorooctanoate and perfluorooctanesulfonate. Toxicol. Sci. 2013, 136, 308–327. [Google Scholar] [CrossRef]
- Yoon, M.; Efremenko, A.; Blaauboer, B.J.; Clewell, H.J. Evaluation of simple in vitro to in vivo extrapolation approaches for environmental compounds. Toxicol. In Vitro 2014, 28, 164–170. [Google Scholar] [CrossRef] [PubMed]
- Wambaugh, J.F.; Hughes, M.F.; Ring, C.L.; MacMillan, D.K.; Ford, J.; Fennell, T.R.; Black, S.R.; Snyder, R.W.; Sipes, N.S.; Wetmore, B.A.; et al. Evaluating In Vitro-In Vivo Extrapolation of Toxicokinetics. Toxicol. Sci. 2018, 163, 152–169. [Google Scholar] [CrossRef] [PubMed]
- Wetmore, B.A. Quantitative in vitro-to-in vivo extrapolation in a high-throughput environment. Toxicology 2015, 332, 94–101. [Google Scholar] [CrossRef] [PubMed]
- Escher, B.I.; Glauch, L.; Konig, M.; Mayer, P.; Schlichting, R. Baseline Toxicity and Volatility Cutoff in Reporter Gene Assays Used for High-Throughput Screening. Chem. Res. Toxicol. 2019, 32, 1646–1655. [Google Scholar] [CrossRef]
- Armitage, J.M.; Wania, F.; Arnot, J.A. Application of mass balance models and the chemical activity concept to facilitate the use of in vitro toxicity data for risk assessment. Environ. Sci. Technol. 2014, 48, 9770–9779. [Google Scholar] [CrossRef] [PubMed]
- Schreiber, R.; Altenburger, R.; Paschke, A.; Kuster, E. How to deal with lipophilic and volatile organic substances in microtiter plate assays. Environ. Toxicol. Chem. 2008, 27, 1676–1682. [Google Scholar] [CrossRef] [PubMed]
- Quick, D.J.; Shuler, M.L. Use of in vitro data for construction of a physiologically based pharmacokinetic model for naphthalene in rats and mice to probe species differences. Biotechnol. Prog. 1999, 15, 540–555. [Google Scholar] [CrossRef]
- Jongeneelen, F.J.; Berge, W.F. A generic, cross-chemical predictive PBTK model with multiple entry routes running as application in MS Excel; design of the model and comparison of predictions with experimental results. Ann. Occup. Hyg. 2011, 55, 841–864. [Google Scholar] [CrossRef]
- Wetmore, B.A.; Wambaugh, J.F.; Ferguson, S.S.; Sochaski, M.A.; Rotroff, D.M.; Freeman, K.; Clewell, H.J., 3rd; Dix, D.J.; Andersen, M.E.; Houck, K.A.; et al. Integration of dosimetry, exposure, and high-throughput screening data in chemical toxicity assessment. Toxicol. Sci. 2012, 125, 157–174. [Google Scholar] [CrossRef]
- Wambaugh, J.F.; Wetmore, B.A.; Ring, C.L.; Nicolas, C.I.; Pearce, R.G.; Honda, G.S.; Dinallo, R.; Angus, D.; Gilbert, J.; Sierra, T.; et al. Assessing Toxicokinetic Uncertainty and Variability in Risk Prioritization. Toxicol. Sci. 2019, 172, 235–251. [Google Scholar] [CrossRef]
- Richard, A.M.; Judson, R.S.; Houck, K.A.; Grulke, C.M.; Volarath, P.; Thillainadarajah, I.; Yang, C.; Rathman, J.; Martin, M.T.; Wambaugh, J.F.; et al. ToxCast Chemical Landscape: Paving the Road to 21st Century Toxicology. Chem. Res. Toxicol. 2016, 29, 1225–1251. [Google Scholar] [CrossRef] [PubMed]
| Cell Model Category | Advantages | Disadvantages |
|---|---|---|
| Monoculture cell lines |
|
|
| Cells from human donors |
|
|
| Lung-on-a-chip |
|
|
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zavala, J.; Freedman, A.N.; Szilagyi, J.T.; Jaspers, I.; Wambaugh, J.F.; Higuchi, M.; Rager, J.E. New Approach Methods to Evaluate Health Risks of Air Pollutants: Critical Design Considerations for In Vitro Exposure Testing. Int. J. Environ. Res. Public Health 2020, 17, 2124. https://doi.org/10.3390/ijerph17062124
Zavala J, Freedman AN, Szilagyi JT, Jaspers I, Wambaugh JF, Higuchi M, Rager JE. New Approach Methods to Evaluate Health Risks of Air Pollutants: Critical Design Considerations for In Vitro Exposure Testing. International Journal of Environmental Research and Public Health. 2020; 17(6):2124. https://doi.org/10.3390/ijerph17062124
Chicago/Turabian StyleZavala, Jose, Anastasia N. Freedman, John T. Szilagyi, Ilona Jaspers, John F. Wambaugh, Mark Higuchi, and Julia E. Rager. 2020. "New Approach Methods to Evaluate Health Risks of Air Pollutants: Critical Design Considerations for In Vitro Exposure Testing" International Journal of Environmental Research and Public Health 17, no. 6: 2124. https://doi.org/10.3390/ijerph17062124
APA StyleZavala, J., Freedman, A. N., Szilagyi, J. T., Jaspers, I., Wambaugh, J. F., Higuchi, M., & Rager, J. E. (2020). New Approach Methods to Evaluate Health Risks of Air Pollutants: Critical Design Considerations for In Vitro Exposure Testing. International Journal of Environmental Research and Public Health, 17(6), 2124. https://doi.org/10.3390/ijerph17062124